Published online Apr 21, 2013. doi: 10.3748/wjg.v19.i15.2313
Revised: January 8, 2013
Accepted: January 18, 2013
Published online: April 21, 2013
Processing time: 135 Days and 19.5 Hours
Celiac disease, an immune-mediated enteropathy induced in genetically susceptible individuals by the ingestion of gluten, is the most frequent disorder associated with splenic hypofunction or atrophy. Defective splenic function affects more than one-third of adult patients with celiac disease, and it may predispose to a higher risk of infections by encapsulated bacteria and thromboembolic and autoimmune complications, particularly when celiac patients have concomitant pre-malignant and malignant complications (refractory celiac disease, ulcerative jejunoileitis and enteropathy-associated T-cell lymphoma). However, the clinical management of patients with celiac disease does not take into account the evaluation of splenic function, and in patients with high degree of hyposplenism or splenic atrophy the prophylactic immunization with specific vaccines against the polysaccharide antigens of encapsulated bacteria is not currently recommended. We critically re-evaluate clinical and diagnostic aspects of spleen dysfunction in celiac disease, and highlight new perspectives in the prophylactic management of infections in this condition.
- Citation: Di Sabatino A, Brunetti L, Carnevale Maffè G, Giuffrida P, Corazza GR. Is it worth investigating splenic function in patients with celiac disease? World J Gastroenterol 2013; 19(15): 2313-2318
- URL: https://www.wjgnet.com/1007-9327/full/v19/i15/2313.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i15.2313
Functional hyposplenism has been regarded as an acquired disorder, potentially associated with several diseases, sometimes accompanied by a reduction in spleen size, and burdened by the same complications occurring in surgical asplenia[1,2]. The spleen, apart from acting as a phagocytic filter, thus removing ageing and damaged cells, is crucial in regulating immune homeostasis by linking innate and adaptive immunity, and in protecting against infections by encapsulated bacteria[3-5].
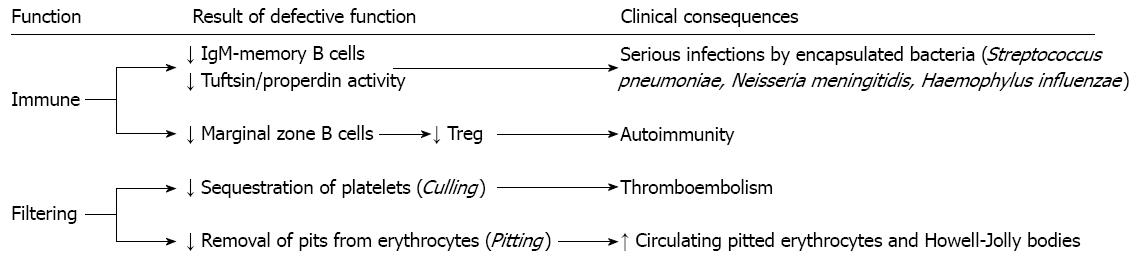
Removal of encapsulated bacteria in the course of initial infection requires natural antibodies produced by immunoglobulin M (IgM)-memory B cells, a unique B cell population resident in the marginal zone of the spleen, which, unlike switched-memory, are responsible for a T-independent response against bacteria[6-9]. The key role of the spleen in mounting an immune response against encapsulated bacteria is supported by the dramatic reduction of the IgM-memory B cell pool following removal of the spleen[10,11]. An impairment of the immune function of the spleen results in (1) reduced number of IgM-memory B cells and defective activity of opsonizing molecules, i.e., properdin and tuftsin, thus predisposing to infections caused by encapsulated bacteria (mainly Streptococcus pneumoniae, Neisseria meningitidis and Haemophylus influenzae); and (2) decreased number of marginal zone B cells which predisposes to the emergence of autoreactive T-cell clones as a consequence of T-regulatory cells depletion, with subsequent development of autoimmunity[8,9,12]. On the other hand, an impairment of the filtering function results in (1) reduced platelet sequestration, which predisposes to thromboembolism; and (2) defective removal of pits from erythrocytes with consequent increase of circulating Howell-Jolly bodies and pitted red cells (Figure 1)[1-3].
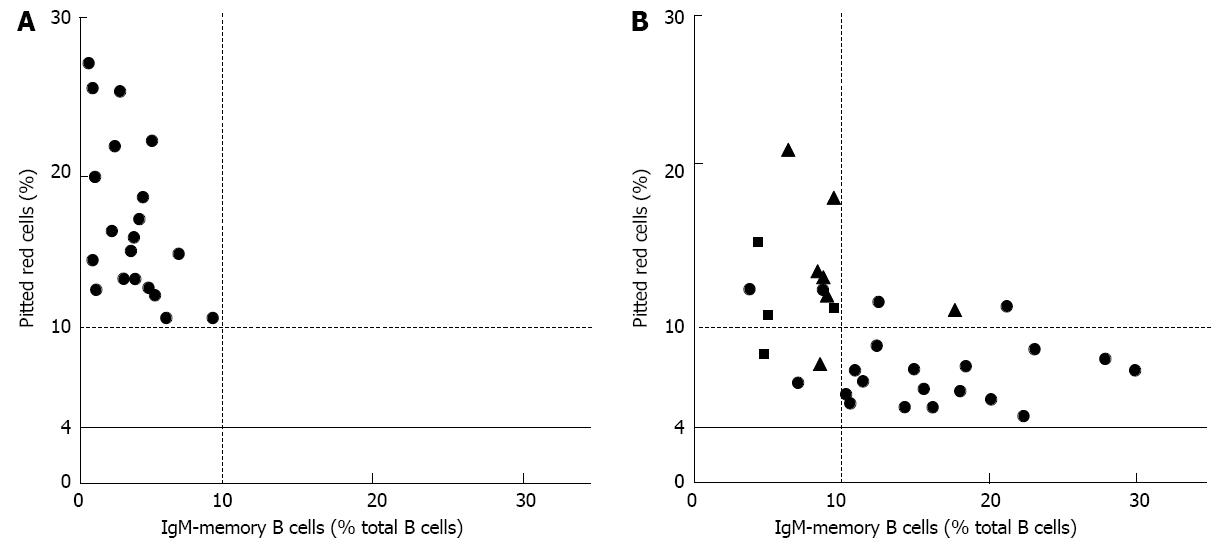
Detection of pitted red cells by phase-interference microscopy[13] is considered an accurate method to assess splenic function, quite easy to perform, less expensive and invasive than radioisotopic methods, and more accurate than Howell-Jolly bodies detection[14], particularly in the quantitation of mild degrees of hyposplenism[3]. The inverse correlation observed in asplenic and hyposplenic patients between pitted red cells and IgM-memory B cells suggests the possible use in clinical practice of flow cytometric B cell analysis as a quantitative alternative test[15]. The little we know about the natural history of hyposplenism leads us to believe that it evolves from a reversible mild impairment of splenic function - as occurs in responder Crohn’s disease patients after anti-tumor necrosis factor treatment with infliximab[16] - to an irreversible impairment of splenic function, up to severe splenic atrophy.
Among all the various diseases associated with hyposplenism, celiac disease is the most frequent[2,17]. Hyposplenism, assessed by pitted red cell counting, affects more than one-third of celiac patients[18]. Defective filtering function, measured by pitted red cell counting, is paralleled by a defect in the frequency of circulating IgM memory B cells and serum tuftsin activity, and both these parameters significantly correlate with the degree of splenic function in untreated celiac disease[18,19]. Hyposplenism does not complicate celiac disease in infancy[20]; in adults its incidence correlates with the duration of pre-exposure to gluten as shown by the correlation with age at diagnosis[18], and a gluten-free diet is effective in restoring splenic function[21]. When the data are split according to clinical severity, the prevalence of hyposplenism increases from 19% to 80% in celiacs with premalignant or malignant complications[19]. Both splenic atrophy and mesenteric lymph node cavitation are recognised as poor prognostic factors in celiac disease[19,22,23].
Major infections have been reported in a number of hyposplenic celiac patients in the last 25 years, variably associated with spleen atrophy and mesenteric lymph node cavitation (Table 1)[23-29]. However, it was only in 2008 that two ad hoc studies highlighted the importance of this predisposition by showing a significantly higher relative risk of pneumococcal sepsis in adult celiacs, which is still significant when hospitalised patients are considered as reference individuals[30,31]. The absolute risk of sepsis turned out to be even higher than that of hip fracture and lymphoma in the celiac cohort[32]. These findings fit with the demonstration of an increased mortality due to infections (in particular septicemia) and respiratory diseases (mainly pneumonia) in the Swedish celiac cohort[33].
| Ref. | No. of cases | Type of infection | Supplementary findings |
| Corazza et al[23] | 1 | Pneumoccoccal pneumonia | Splenic atrophy |
| Matuchansky et al[24] | 2 | Pneumococcal pneumonia, infectious pericarditis | Splenic atrophy, MLNC |
| O’Donoghue[25] | 1 | Pneumococcal septicemia | Splenic atrophy |
| Logan et al[26] | 2 | Pneumococcal meningitis, septicemia by Salmonella | Splenic atrophy |
| Stevens et al[27] | 3 | Lung abscesses by Staphyloccus and Klebsiella | Splenic atrophy |
| Howat et al[28] | 2 | Fatal chest infection, septicemia | Splenic atrophy, MLNC |
| Harmon et al[29] | 1 | Septicemia by Klebsiella | Splenic atrophy |
Although anti-pneumococcal vaccination has been shown to reduce the prevalence of major infections in asplenic patients[34-38], it is dramatically underused as shown by these data collected in England and Wales by examining 3584 patients with celiac disease or sickle cell anemia[39]. Vaccines currently used in patients at risk of pneumococcal infections are the 23-valent pneumococcal polysaccharide vaccine[40], whose protective action is based on the production of opsonising anti-capsular antibodies by means of a T-independent mechanism (it is actually recommended in asplenic/hyposplenic adults and children older than 5 years), and the 13-valent protein-conjugate pneumococcal vaccine (PCV-13, Prevnar)[41], in which the CRM197 diphtheria protein changes the nature of the response from T-independent to T-dependent, making this vaccine particularly suitable in infants, especially below the age of 2 years, when the splenic IgM-memory B cell pool is still immature (Table 2)[42-45]. Similarly, adult hyposplenic patients, in whom IgM-memory B cell are depleted, would benefit from PCV-13, as its T-dependent mechanism is supposed to bypass the immunological impairment due to the lack of IgM-memory B cells. Nevertheless, Prevnar is recommended by current guidelines only in infancy (Table 2)[46].
| Vaccine | Brand name | Structure | Mechanism | Serotype | Indication |
| PPV23 | Pneumovax® | Polysaccharide | T-cell independent | 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F, 33F | Asplenic or hyposplenic adults Asplenic or hyposplenic children > 5 yr |
| PCV13 | Prevnar® | Protein-conjugate (CRM197 protein) | T-cell dependent | 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F, 23F | Asplenic or hyposplenic children < 5 yr |
Celiac disease is frequently associated with a number of autoimmune disorders, including Hashimoto’s thyroiditis, insulin-dependent diabetes mellitus, Sjögren’s syndrome, Addison disease, systemic lupus erythematosus, rheumatoid arthritis[47,48]. The evidence that autoantibodies may develop within months of splenectomy[49], together with the demonstration that celiac patients with blood film features of hyposplenism have a higher prevalence of autoantibodies[50], have led to the hypothesis that defective splenic function might predispose the development of autoimmunity in celiac disease[51,52].
The nature of the link between hyposplenism and autoimmune manifestations of celiac disease is not known, and it is not clear whether autoimmune disorders precede and cause splenic hypofunction or atrophy, or vice versa, or whether additional factors influence both conditions. The finding that hyposplenism in nonceliac patients with autoimmune disorders did not differ significantly from that of healthy controls supports the hypothesis that the higher risk for splenic hypofuncion in celiac patients with autoimmune disorders might be related to celiac disease, rather than to autoimmunity per se[19]. Of note, both hyposplenism and autoimmune disorders increase their prevalence with the length of pre-exposure to gluten in celiac disease[18,53]. When we looked at the prevalence of celiac disease-associated hyposplenism, we found that it increases from 19% in uncomplicated patients to 59% in those with associated autoimmune diseases. Moreover, patients with celiac disease-associated autoimmune disorders have a significantly lower percentage of IgM memory B cells in comparison to uncomplicated celiac patients[19]. This finding is quite interesting when considering that memory B cells resident in the marginal zone of the spleen play a role in the tolerance of autoantigens through the B cell receptor[54]. A similar role is exerted by marginal zone dendritic cells which internalise apoptotic leucocytes thus preventing autoantigens exposed on the surface of apoptotic bodies from causing autoantibody formation[55]. Moreover, both marginal zone B cells and dendritic cells may favour the expansion of regulatory T cells which maintain tolerance through the up-regulation of anti-inflammatory cytokines, such as transforming growth factor-β and interleukin-10[56]. The perturbation of these regulatory mechanisms have been shown to predispose to the development of autoimmunity in splenectomised or hyposplenic subjects[19,49,57].
Impaired spleen sequestration of circulating platelets and increased blood viscosity are supposed to be implicated in the development of thromboembolic events in splenectomised patients or in other hyposplenism-associated disorders[58]. In the latter, the risk of thrombosis is difficult to assess as many of these disorders are associated with increased incidence of thrombosis per se. The hyperviscosity secondary to defective splenic function may be the result in part of the persistence of aged and damaged red cells in the circulation as well as intracellular inclusions, such as Howell-Jolly bodies, siderotic granules, and Heinz bodies, all of which promote decreased erythrocyte deformability[59]. An increased risk of thromboembolism has been recently demonstrated in celiac disease, where it correlates with the duration of pre-exposure to gluten[60]. However, in that study no data is available concerning the weight of the thromboembolic risk in the hyposplenic celiac patients, nor hyposplenism is mentioned among the possible factors predisposing to thromboembolism.
There is a number of critical issues that remain to be elucidated to define the optimal management of hyposplenic celiac patients and to clarify the pathogenic mechanisms underlying spleen hypofunction[61,62]. We propose that splenic function is determined in patients with pre-malignant and malignant complications, concomitant autoimmune disorders, old age at diagnosis, previous history of major infections/sepsis or thromboembolism, mesenteric lymph node cavitation and/or spleen atrophy (Table 3). As a diagnostic tool, pitted red cell counting remains an accurate, quantitative and inexpensive method, albeit observer-dependent[63]. Flow cytometric analysis of memory B cells could be an alternative quantitative test, although studies assessing its sensitivity and specificity are lacking[64]. We believe that protein-conjugate vaccines[65-68] should be recommended in patients with major hyposplenism, defined -on the basis of data derived from asplenic patients- by a pitted red cells value higher than 10% and/or an IgM memory B cell frequency lower than 10%. Of note, most of the patients identified by these parameters are refractory or have concomitant autoimmune disorders (Figure 2). Understanding the pathogenic mechanisms underlying spleen dysfunction in celiac disease requires a greater knowledge of the connections between gut and spleen. The demonstration that spleen function is crucial for the presence of IgA-producing plasma cells in the gut of both asplenic mice and patients[69], and that oral tolerance to gluten is predominantly mounted in the spleen[70] represent preliminary attempts in this direction.
| Patients with complications (RCD, UJI, EATL, collagenous sprue) |
| Patients with concomitant autoimmune disorders |
| Patients with old age at diagnosis |
| Patients with previous history of major infections/sepsis and/or thromboembolism |
| Patients with mesenteric lymph node cavitation and/or splenic atroph |
P- Reviewer Ciaccio EJ S- Editor Jiang L L- Editor A E- Editor Li JY
| 1. | William BM, Corazza GR. Hyposplenism: a comprehensive review. Part I: basic concepts and causes. Hematology. 2007;12:1-13. [PubMed] |
| 2. | William BM, Thawani N, Sae-Tia S, Corazza GR. Hyposplenism: a comprehensive review. Part II: clinical manifestations, diagnosis, and management. Hematology. 2007;12:89-98. [PubMed] |
| 3. | Di Sabatino A, Carsetti R, Corazza GR. Post-splenectomy and hyposplenic states. Lancet. 2011;378:86-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 437] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 4. | Mebius RE, Kraal G. Structure and function of the spleen. Nat Rev Immunol. 2005;5:606-616. [PubMed] |
| 5. | Sills RH. Splenic function: physiology and splenic hypofunction. Crit Rev Oncol Hematol. 1987;7:1-36. [PubMed] |
| 6. | Klein U, Rajewsky K, Küppers R. Human immunoglobulin (Ig)M+IgD+ peripheral blood B cells expressing the CD27 cell surface antigen carry somatically mutated variable region genes: CD27 as a general marker for somatically mutated (memory) B cells. J Exp Med. 1998;188:1679-1689. [PubMed] |
| 7. | Shi Y, Agematsu K, Ochs HD, Sugane K. Functional analysis of human memory B-cell subpopulations: IgD+CD27+ B cells are crucial in secondary immune response by producing high affinity IgM. Clin Immunol. 2003;108:128-137. [PubMed] |
| 8. | Kruetzmann S, Rosado MM, Weber H, Germing U, Tournilhac O, Peter HH, Berner R, Peters A, Boehm T, Plebani A. Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen. J Exp Med. 2003;197:939-945. [PubMed] |
| 9. | Carsetti R, Rosado MM, Wardmann H. Peripheral development of B cells in mouse and man. Immunol Rev. 2004;197:179-191. [PubMed] |
| 10. | Carsetti R, Pantosti A, Quinti I. Impairment of the antipolysaccharide response in splenectomized patients is due to the lack of immunoglobulin M memory B cells. J Infect Dis. 2006;193:1189-1190. [PubMed] |
| 11. | Wasserstrom H, Bussel J, Lim LC, Cunningham-Rundles C. Memory B cells and pneumococcal antibody after splenectomy. J Immunol. 2008;181:3684-3689. [PubMed] |
| 12. | Weller S, Braun MC, Tan BK, Rosenwald A, Cordier C, Conley ME, Plebani A, Kumararatne DS, Bonnet D, Tournilhac O. Human blood IgM “memory” B cells are circulating splenic marginal zone B cells harboring a prediversified immunoglobulin repertoire. Blood. 2004;104:3647-3654. [PubMed] |
| 13. | Corazza GR, Bullen AW, Hall R, Robinson PJ, Losowsky MS. Simple method of assessing splenic function in coeliac disease. Clin Sci (Lond). 1981;60:109-113. [PubMed] |
| 14. | Corazza GR, Ginaldi L, Zoli G, Frisoni M, Lalli G, Gasbarrini G, Quaglino D. Howell-Jolly body counting as a measure of splenic function. A reassessment. Clin Lab Haematol. 1990;12:269-275. [PubMed] |
| 15. | Di Sabatino A, Rosado MM, Ciccocioppo R, Cazzola P, Morera R, Corazza GR, Carsetti R. Depletion of immunoglobulin M memory B cells is associated with splenic hypofunction in inflammatory bowel disease. Am J Gastroenterol. 2005;100:1788-1795. [PubMed] |
| 16. | Di Sabatino A, Rosado MM, Cazzola P, Biancheri P, Tinozzi FP, Laera MR, Cantoro L, Vanoli A, Carsetti R, Corazza GR. Splenic function and IgM-memory B cells in Crohn’s disease patients treated with infliximab. Inflamm Bowel Dis. 2008;14:591-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Ferguson A, Hutton MM, Maxwell JD, Murray D. Adult coeliac disease in hyposplenic patients. Lancet. 1970;1:163-164. [PubMed] |
| 18. | Corazza GR, Zoli G, Di Sabatino A, Ciccocioppo R, Gasbarrini G. A reassessment of splenic hypofunction in celiac disease. Am J Gastroenterol. 1999;94:391-397. [PubMed] |
| 19. | Di Sabatino A, Rosado MM, Cazzola P, Riboni R, Biagi F, Carsetti R, Corazza GR. Splenic hypofunction and the spectrum of autoimmune and malignant complications in celiac disease. Clin Gastroenterol Hepatol. 2006;4:179-186. [PubMed] |
| 20. | Corazza GR, Lazzari R, Frisoni M, Collina A, Gasbarrini G. Splenic function in childhood coeliac disease. Gut. 1982;23:415-416. [PubMed] |
| 21. | Corazza GR, Frisoni M, Vaira D, Gasbarrini G. Effect of gluten-free diet on splenic hypofunction of adult coeliac disease. Gut. 1983;24:228-230. [PubMed] |
| 22. | Di Sabatino A, Rosado MM, Miele L, Capolunghi F, Cazzola P, Biancheri P, Carsetti R, Gasbarrini G, Corazza GR. Impairment of splenic IgM-memory but not switched-memory B cells in a patient with celiac disease and splenic atrophy. J Allergy Clin Immunol. 2007;120:1461-1463. [PubMed] |
| 23. | Corazza GR, Frisoni M, Vaira D, Gasbarrini G. Fatal pneumonia in a patient with coeliac disease and splenic atrophy. Ital J Gastroenterol. 1984;16:300-301. |
| 24. | Matuchansky C, Colin R, Hemet J, Touchard G, Babin P, Eugene C, Bergue A, Zeitoun P, Barboteau MA. Cavitation of mesenteric lymph nodes, splenic atrophy, and a flat small intestinal mucosa. Report of six cases. Gastroenterology. 1984;87:606-614. [PubMed] |
| 25. | O’Donoghue DJ. Fatal pneumococcal septicaemia in coeliac disease. Postgrad Med J. 1986;62:229-230. [PubMed] |
| 26. | Logan RF, Rifkind EA, Turner ID, Ferguson A. Mortality in celiac disease. Gastroenterology. 1989;97:265-271. [PubMed] |
| 27. | Stevens FM, Connolly CE, Murray JP, McCarthy CF. Lung cavities in patients with coeliac disease. Digestion. 1990;46:72-80. [PubMed] |
| 28. | Howat AJ, McPhie JL, Smith DA, Aqel NM, Taylor AK, Cairns SA, Thomas WE, Underwood JC. Cavitation of mesenteric lymph nodes: a rare complication of coeliac disease, associated with a poor outcome. Histopathology. 1995;27:349-354. [PubMed] |
| 29. | Harmon GS, Lee JS. Splenic atrophy in celiac disease. Clin Gastroenterol Hepatol. 2010;8:A22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 30. | Ludvigsson JF, Olén O, Bell M, Ekbom A, Montgomery SM. Coeliac disease and risk of sepsis. Gut. 2008;57:1074-1080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 94] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 31. | Thomas HJ, Wotton CJ, Yeates D, Ahmad T, Jewell DP, Goldacre MJ. Pneumococcal infection in patients with coeliac disease. Eur J Gastroenterol Hepatol. 2008;20:624-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 32. | Walters JR, Bamford KB, Ghosh S. Coeliac disease and the risk of infections. Gut. 2008;57:1034-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 33. | Peters U, Askling J, Gridley G, Ekbom A, Linet M. Causes of death in patients with celiac disease in a population-based Swedish cohort. Arch Intern Med. 2003;163:1566-1572. [PubMed] |
| 34. | Melles DC, de Marie S. Prevention of infections in hyposplenic and asplenic patients: an update. Neth J Med. 2004;62:45-52. [PubMed] |
| 35. | Bisharat N, Omari H, Lavi I, Raz R. Risk of infection and death among post-splenectomy patients. J Infect. 2001;43:182-186. [PubMed] |
| 36. | Hansen K, Singer DB. Asplenic-hyposplenic overwhelming sepsis: postsplenectomy sepsis revisited. Pediatr Dev Pathol. 2001;4:105-121. [PubMed] |
| 37. | Brigden ML, Pattullo AL. Prevention and management of overwhelming postsplenectomy infection--an update. Crit Care Med. 1999;27:836-842. [PubMed] |
| 38. | Brigden ML, Pattullo A, Brown G. Pneumococcal vaccine administration associated with splenectomy: the need for improved education, documentation, and the use of a practical checklist. Am J Hematol. 2000;65:25-29. [PubMed] |
| 39. | Pebody RG, Hippisley-Cox J, Harcourt S, Pringle M, Painter M, Smith G. Uptake of pneumococcal polysaccharide vaccine in at-risk populations in England and Wales 1999-2005. Epidemiol Infect. 2008;136:360-369. [PubMed] |
| 40. | French N. Use of pneumococcal polysaccharide vaccines: no simple answers. J Infect. 2003;46:78-86. [PubMed] |
| 41. | Durando P, Alicino C, De Florentiis D, Martini M, Icardi G. Improving the protection against Streptococcus pneumoniae with the new generation 13-valent pneumococcal conjugate vaccine. J Prev Med Hyg. 2012;53:68-77. [PubMed] |
| 42. | Breukels MA, Zandvoort A, van Den Dobbelsteen GP, van Den Muijsenberg A, Lodewijk ME, Beurret M, Klok PA, Timens W, Rijkers GT. Pneumococcal conjugate vaccines overcome splenic dependency of antibody response to pneumococcal polysaccharides. Infect Immun. 2001;69:7583-7587. [PubMed] |
| 43. | Mikoluc B, Kayhty H, Bernatowska E, Motkowski R. Immune response to the 7-valent pneumococcal conjugate vaccine in 30 asplenic children. Eur J Clin Microbiol Infect Dis. 2008;27:923-928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 44. | Musher DM, Ceasar H, Kojic EM, Musher BL, Gathe JC, Romero-Steiner S, White AC. Administration of protein-conjugate pneumococcal vaccine to patients who have invasive disease after splenectomy despite their having received 23-valent pneumococcal polysaccharide vaccine. J Infect Dis. 2005;191:1063-1067. [PubMed] |
| 45. | Vernacchio L, Neufeld EJ, MacDonald K, Kurth S, Murakami S, Hohne C, King M, Molrine D. Combined schedule of 7-valent pneumococcal conjugate vaccine followed by 23-valent pneumococcal vaccine in children and young adults with sickle cell disease. J Pediatr. 1998;133:275-278. [PubMed] |
| 46. | Shatz DV. Vaccination considerations in the asplenic patient. Expert Rev Vaccines. 2005;4:27-34. [PubMed] |
| 47. | Di Sabatino A, Corazza GR. Coeliac disease. Lancet. 2009;373:1480-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 460] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 48. | Lewis NR, Holmes GK. Risk of morbidity in contemporary celiac disease. Expert Rev Gastroenterol Hepatol. 2010;4:767-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 49. | Robertson DA, Simpson FG, Gowland G, Losowsky MS. Splenectomy causes auto-antibody formation. J Clin Lab Immunol. 1983;11:63-65. [PubMed] |
| 50. | Bullen AW, Hall R, Gowland G, Rajah S, Losowsky MS. Hyposplenism, adult coeliac disease, and autoimmunity. Gut. 1981;22:28-33. [PubMed] |
| 51. | Corazza GR, Gasbarrini G. Defective splenic function and its relation to bowel disease. Clin Gastroenterol. 1983;12:651-669. [PubMed] |
| 53. | Ventura A, Magazzù G, Greco L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Gastroenterology. 1999;117:297-303. [PubMed] |
| 54. | Rathmell JC, Townsend SE, Xu JC, Flavell RA, Goodnow CC. Expansion or elimination of B cells in vivo: dual roles for CD40- and Fas (CD95)-ligands modulated by the B cell antigen receptor. Cell. 1996;87:319-329. [PubMed] |
| 55. | Steinman RM, Pack M, Inaba K. Dendritic cells in the T-cell areas of lymphoid organs. Immunol Rev. 1997;156:25-37. [PubMed] |
| 56. | Morelli AE, Larregina AT, Shufesky WJ, Zahorchak AF, Logar AJ, Papworth GD, Wang Z, Watkins SC, Falo LD, Thomson AW. Internalization of circulating apoptotic cells by splenic marginal zone dendritic cells: dependence on complement receptors and effect on cytokine production. Blood. 2003;101:611-620. [PubMed] |
| 57. | Balsalobre B, Hernández-Godoy J, Planelles D. Autoantibodies in splenectomized patients as a consequence of abdominal trauma. J Investig Allergol Clin Immunol. 1992;2:91-95. [PubMed] |
| 58. | Cappellini MD, Grespi E, Cassinerio E, Bignamini D, Fiorelli G. Coagulation and splenectomy: an overview. Ann N Y Acad Sci. 2005;1054:317-324. [PubMed] |
| 59. | Crary SE, Buchanan GR. Vascular complications after splenectomy for hematologic disorders. Blood. 2009;114:2861-2868. [PubMed] |
| 60. | Ludvigsson JF, Welander A, Lassila R, Ekbom A, Montgomery SM. Risk of thromboembolism in 14,000 individuals with coeliac disease. Br J Haematol. 2007;139:121-127. [PubMed] |
| 61. | Davies JM, Lewis MP, Wimperis J, Rafi I, Ladhani S, Bolton-Maggs PH. Review of guidelines for the prevention and treatment of infection in patients with an absent or dysfunctional spleen: prepared on behalf of the British Committee for Standards in Haematology by a working party of the Haemato-Oncology task force. Br J Haematol. 2011;155:308-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 175] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 62. | Castagnola E, Fioredda F. Prevention of life-threatening infections due to encapsulated bacteria in children with hyposplenia or asplenia: a brief review of current recommendations for practical purposes. Eur J Haematol. 2003;71:319-326. [PubMed] |
| 63. | de Porto AP, Lammers AJ, Bennink RJ, ten Berge IJ, Speelman P, Hoekstra JB. Assessment of splenic function. Eur J Clin Microbiol Infect Dis. 2010;29:1465-1473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 123] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 64. | Cameron PU, Jones P, Gorniak M, Dunster K, Paul E, Lewin S, Woolley I, Spelman D. Splenectomy associated changes in IgM memory B cells in an adult spleen registry cohort. PLoS One. 2011;6:e23164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 65. | Croxtall JD, Dhillon S. Meningococcal quadrivalent (serogroups A, C, W135 and Y) tetanus toxoid conjugate vaccine (Nimenrix™). Drugs. 2012;72:2407-2430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 66. | Palmu AA, Jokinen J, Borys D, Nieminen H, Ruokokoski E, Siira L, Puumalainen T, Lommel P, Hezareh M, Moreira M. Effectiveness of the ten-valent pneumococcal Haemophilus influenzae protein D conjugate vaccine (PHiD-CV10) against invasive pneumococcal disease: a cluster randomised trial. Lancet. 2013;381:214-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 178] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 67. | Gruber WC, Scott DA, Emini EA. Development and clinical evaluation of Prevnar 13, a 13-valent pneumocococcal CRM197 conjugate vaccine. Ann N Y Acad Sci. 2012;1263:15-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 68. | Frenck RW, Gurtman A, Rubino J, Smith W, van Cleeff M, Jayawardene D, Giardina PC, Emini EA, Gruber WC, Scott DA. Randomized, controlled trial of a 13-valent pneumococcal conjugate vaccine administered concomitantly with an influenza vaccine in healthy adults. Clin Vaccine Immunol. 2012;19:1296-1303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 69. | Di Sabatino A, Rosado MM, Carsetti R, Cascioli S, Giorda E, Scarsella M, Petrini S, Tinozzi FP, Brunetti L, Milito C. Depletion of circulating memory B cells in splenectomised and common variable immune deficiency patients is associated with decreased IgA plasma cells in the gut mucosa. Intern Emerg Med. 2011;6:S142. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 70. | Du Pré MF, Kozijn AE, van Berkel LA, ter Borg MN, Lindenbergh-Kortleve D, Jensen LT, Kooy-Winkelaar Y, Koning F, Boon L, Nieuwenhuis EE. Tolerance to ingested deamidated gliadin in mice is maintained by splenic, type 1 regulatory T cells. Gastroenterology. 2011;141:610-620, 620.e1-2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |