INVITED COMMENTARY ON HOT ARTICLES
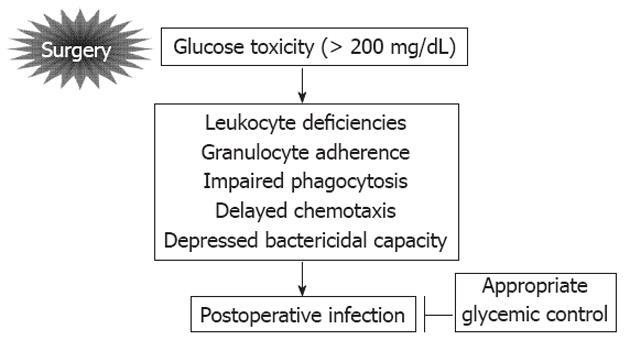
It is well known that perioperative hyperglycemia is the main cause of infectious complications after surgery[1]. Figure 1 shows the relationship between hyperglycemia and postoperative infection (POI). Glucose toxicity is caused by surgical stress induced hyperglycemia such as a level of more than 200 mg/dL. Glucose toxicity leads to the leukocyte deficiencies, granulocyte adherence, impaired phagocytosis, delayed chemotaxis, and depressed bactericidal capacity. These abnormalities are the principal causes of POI and they can be improved by appropriate glycemic control[1]. However, optimal blood glucose range to prevent postoperative infectious complications remains unclear in various surgical settings[1]. To improve the methods for perioperative glycemic control, we would like to recommend an interesting paper published recently on this topic[2]. At the same time, we will introduce a novel perioperative tight glycemic control (TGC) using an artificial pancreas (AP) in patients undergoing pancreatic resection aimed to reduce postoperative infectious complications including surgical site infection (SSI).
Figure 1 Relationship between hyperglycemia and postoperative infection.
We read with great interest the article published in Annals of Surgery entitled “Early postoperative hyperglycemia is associated with postoperative complications after pancreatoduodenectomy” by Eshuis et al[2]. Among 330 consecutive patients undergoing pancreatoduodenectomy (PD), the average glucose levels were controlled at 135 mg/dL (preoperative), 133 mg/dL (intraoperative) and 142 mg/dL (early postoperative). Pre- and intraoperative glucose levels were not associated with postoperative complications. However, early postoperative glucose levels more than 140 mg/dL was significantly associated with complications after PD[2]. Recent reports indicate that postoperative hyperglycemia increases the risk of postoperative infectious complications and prolongs hospital stay[3-5]. Since we recommend that perioperative TGC is an effective method to prevent postoperative complications including SSI after distal, proximal, and total pancreatic resection[6,7], we support strongly the conclusion drawn in this article[2]. Undoubtedly, this is a significant paper in our understanding of the efficacy of strict perioperative glucose control for patients undergoing PD. However, if early postoperative glucose control in patients undergoing PD was administrated by conventional method such as sliding scale approach as described in this article[2], it seems to be difficult to maintain strict glycemic control with less variability of blood glucose concentration recommended by the authors, including the targeting blood glucose zone of less than 140 mg/dL because pancreatogenic diabetes after pancreatic resection is likely to occur, either hypoglycemia or hyperglycemia, so called brittle diabetes[8,9]. Therefore, we would like to share our opinions regarding more effective and safe TGC against pancreatogenic diabetes after pancreatic resection including PD.
In 2005, we reported that perioperative glycemic control using a closed-loop AP for total pancreatectomized dogs could maintain a stable blood glucose near the normoglycemia[10]. Based on this experimental study, since 2006, we have introduced clinically perioperative glycemic control using an AP[8,11]. As described previously[8,11,12], the Nikkiso Company (Tokyo, Japan) developed a bed-side type AP with closed-loop glycemic control system as STG-22 in conventional device[8] and STG-55 in current device[12] (Figure 2). Detailed mechanisms and characteristics of STG-22 and/or STG-55 were reported previously[8,11,12]. Briefly, peripheral venous blood for glucose monitoring was sampled continuously at less than 2 mL/h. STG-55 (Figure 2)[12] is capable of measuring continuously the blood glucose with its glucose sensor, and automatically infuses insulin and/or glucose to adjust the blood glucose level in accordance with a target blood glucose value, which is the so called closed-loop system[13].
Figure 2 STG-55, a bedside-type artificial endocrine pancreas with closed-loop system.
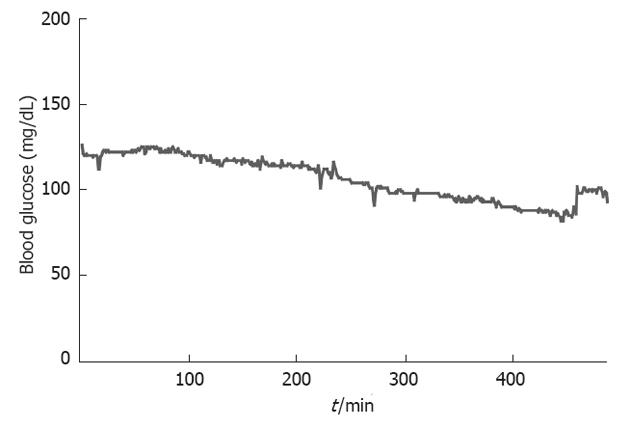
As a result in the clinical surgical settings, our previous report[7] suggested that perioperative TGC using an AP (targeted blood glucose zone of 80-110 mg/dL) in patients undergoing pancreatectomy decreased significantly SSI as compared with that of conventional glycemic control by sliding scale method (targeted blood glucose zone of 150-200 mg/dL). In the sliding scale group, postoperative blood glucose levels rose initially before reaching a plateau of approximately 200 mg/dL between 4 and 6 h after pancreatectomy. The levels remained high for 18 h postoperatively. In the AP group, blood glucose levels reduced steadily, reaching the target zone (80-110 mg/dL) by 6 h after surgery. The total insulin dose administered per patient during the first postoperative 18 h was significantly higher in the AP group (mean ± SD, 107 ± 109 IU) than the sliding scale group (8 ± 6 IU; P < 0.01). Neither group showed hypoglycemia[7]. In addition, this novel glycemic control provided for high achievement of targeting blood glucose levels with stable blood glucose concentration[7,8,14]. Moreover, surprisingly, we have never observed occurrence of hypoglycemia less than 40 mg/dL in more than 400 patients undergoing general surgery[12]. Up to dates, we have performed perioperative TGC with target blood glucose levels of 80-110 mg/dL using an AP in more than 100 pancreatectomized patients including more than 50 PD and 10 total pancreatectomies. Of note, every pancreatectomized patient had stable perioperative blood glucose near the normoglycemia not only without hyperglycemia and/or hypoglycemia but also with less variability of blood glucose concentration, even in a total pancreatectomized patient (Figure 3) who often presented with the most serious pancreatogenic diabetes[9]. Based on these findings from our experimental and clinical studies, we suggest that the AP helps us accomplish an effective and safe perioperative TGC in patients undergoing pancreatic resection.
Figure 3 Continuous perioperative blood glucose levels in a case after total pancreatectomy.
Interestingly, this article[2] suggests that an early postoperative level of at least more than 140 mg/dL is not recommended for improvement of morbidity after PD. Unfortunately, however, the ideal blood glucose range to reduce mortality and morbidity after PD remains unclear. Therefore, we promote a prospective randomized clinical trial to compared targeted blood glucose range of 80-110 mg/dL group and that of 140-180 mg/dL group in patients undergoing distal, proximal, and total pancreatic resection. Primary end point of this study is incidence of SSI and secondary end points are other postoperative complications and mortality (data not shown). We believe that this novel perioperative glucose control using AP is an easy and a reliable method to maintain targeted blood glucose zone, such as 80-110 mg/dL, 110-140 mg/dL and 140-180 mg/dL, which can be determined freely even in patients undergoing pancreatic resection. In the future perspectives, it is essential to find the optimal blood glucose range to improve morbidity and mortality in patients undergoing pancreatic resection, and then AP will be a useful device to maintain the range.
In conclusion, we suggest that the TGC using an artificial endocrine pancreas with a closed-loop system may play an important role in the effective control of infection after pancreatic resection. This novel perioperative glycemic control will enable us to improve surgical outcome by reducing the postoperative infectious complications due to surgical stress induced hyperglycemia.