Published online Feb 21, 2011. doi: 10.3748/wjg.v17.i7.867
Revised: December 8, 2010
Accepted: December 15, 2010
Published online: February 21, 2011
Several advances in genetics, diagnosis and palliation of pancreatic cancer (PC) have occurred in the last decades. A multidisciplinary approach to this disease is therefore recommended. PC is relatively common as it is the fourth leading cause of cancer related mortality. Most patients present with obstructive jaundice, epigastric or back pain, weight loss and anorexia. Despite improvements in diagnostic modalities, the majority of cases are still detected in advanced stages. The only curative treatment for PC remains surgical resection. No more than 20% of patients are candidates for surgery at the time of diagnosis and survival remains quite poor as adjuvant therapies are not very effective. A small percentage of patients with borderline non-resectable PC might benefit from neo-adjuvant chemoradiation therapy enabling them to undergo resection; however, randomized controlled studies are needed to prove the benefits of this strategy. Patients with unresectable PC benefit from palliative interventions such as biliary decompression and celiac plexus block. Further clinical trials to evaluate new chemo and radiation protocols as well as identification of genetic markers for PC are needed to improve the overall survival of patients affected by PC, as the current overall 5-year survival rate of patients affected by PC is still less than 5%. The aim of this article is to review the most recent high quality literature on this topic.
- Citation: Sharma C, Eltawil KM, Renfrew PD, Walsh MJ, Molinari M. Advances in diagnosis, treatment and palliation of pancreatic carcinoma: 1990-2010. World J Gastroenterol 2011; 17(7): 867-897
- URL: https://www.wjgnet.com/1007-9327/full/v17/i7/867.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i7.867
The vast majority (90%) of pancreatic cancers (PC) are malignant tumors originating from pancreatic ductal cells[1]. Anatomically, 78% of PCs are located in the head, and the remaining 22% are equally distributed in the body and in the tail[2]. The most common clinical presentations are progressive weight loss and anorexia, mid abdominal pain and jaundice[3-5]. Over the past two decades many advances in the diagnosis, therapy and palliation of PC have taken place although the overall survival of affected patients has not improved significantly. The aim of this article is to review the most recent high quality literature on this topic.
The literature search was targeted at studies that reported at least one of the following aspects of PC: epidemiology, diagnosis, therapy (e.g. surgery, radiotherapy, chemotherapy) and palliation. Randomized controlled trials (RCT) and prospective observational studies were given preference. Each of the topics was searched in MEDLINE, Ovid MEDLINE In-process, Cochrane Database of Systematic Reviews, Database of Systematic Reviews, Database of Abstracts of Review of Effects, EMBASE, PubMed, National Library of Medicine Gateway by established systematic review methods (Jadad Scale for RCT, as well as Downs and Black checklist for observational studies)[6-8]. Articles from the authors’ libraries and reference lists were further reviewed. We limited our search to English-language articles published from January 1990 to September 2010. We then developed a comprehensive and current database to catalog the medical literature on PC. To identify all potential papers, we searched the medical subject headings reported in Table 1. Three authors (Sharma C, Eltawil KM and Molinari M) independently performed the selection of the articles based on the content of titles and abstracts. When in doubt, each article was reviewed entirely. The decision to include articles in this review was reached by consensus. For conciseness, a full list of search strategies, search results, and quality assessment for each included study are available on request from the corresponding author.
| Primary MeSH terms | Secondary MeSH terms (epidemiology, diagnosis) | Secondary MeSH terms (treatment, palliation) |
| Pancreatic neoplasm(s) | Epidemiology | Pancreaticoduodenectomy |
| Adenocarcinoma(s) | Classification | Resection |
| Carcinoma(s) | Diagnosis | Therapeutic(s) |
| Pancreatic diseases | Differential diagnosis | Treatment outcome(s) |
| Pancreas | Risk factor(s) | Surgery |
| Carcinoma, pancreatic ductal | Diagnostic imaging | Surgical procedures |
| Pancreatic duct(s) | Magnetic resonance imaging | Clinical trial(s) |
| Humans | Endosonography | Controlled clinical trial(s) |
| Adult | Ultrasonography | Randomized controlled trial(s) |
| Emission computed tomography | Clinical trial (phase I) | |
| Radionuclide imaging | Clinical trial (phase II) | |
| Positron emission tomography | Clinical trial (phase III) | |
| Tomography | Clinical trial (phase IV) | |
| X-ray computed | Drug therapy | |
| Biopsy (fine needle) | Chemotherapy | |
| Biopsy (needle) | Neoadjuvant therapy | |
| Cytology | Adjuvant | |
| Cytodiagnosis | Antineoplastic combined chemotherapy protocols | |
| Tumor markers (biological) antigen(s) | Antineoplastic agent(s) | |
| Carcinoembryonic antigen | Antimetabolites, antineoplastic | |
| Ca 19-9 antigen | Combined modality therapeutic antineoplastic | |
| Ca 125 antigen | Combined chemotherapy protocols neoadjuvant | |
| Antigens, tumor-associated, carbohydrate | Therapy | |
| Endoscopic retrograde cholangiopancreatography | Radiotherapy | |
| Computed assisted image processing | Drainage | |
| Sensitivity and specificity | Cholestasis | |
| Endoscopy | Obstructive jaundice | |
| Celiac plexus | ||
| Autonomic nerve block | ||
| Nerve block | ||
| Ethanol | ||
| Injections, intralesional | ||
| Cisplatin | ||
| Deoxycytidine | ||
| Epidermal growth factor | ||
| Fluorouracil | ||
| Endostatin | ||
| Biological products | ||
| Neoplasm proteins | ||
| Immunotherapy | ||
| Antibodies, monoclonal |
PC is the fourth leading cause of cancer related mortality in the United States with an estimated 42 500 new cases and 35 000 deaths from the disease each year[9]. In industrialized countries, the incidence of PC (11 per 100 000 individuals) ranks second after colorectal cancer among all gastrointestinal malignancies[10]. While the mortality rate for males has decreased by 0.4% from 1990 to 2005, the mortality rate for females has increased by 4.4%[9]. More than 80% of PCs are diagnosed in patients older than 60 and almost 50% have distant metastases at the time of presentation[10-12]. Men are more frequently affected than women [relative risk (RR) = 1.3] and individuals of African American descent in comparison to Caucasians (RR = 1.5)[10]. Analysis of overall survival shows that the prognosis of PC is still quite poor despite the fact that 1-year survival has increased from 15.2% (period between 1977-1981) to 21.6% (period between 1997-2001) and 5-year survival has increased from 3% (period between 1977-1986) to 5% (period between 1996-2004)[10].
The risk of PC in smokers ranks second to lung cancer[13] and it is proportionate to the frequency [≥ 30 cigarettes per day: odds ratio (OR) = 1.75], duration (≥ 50 years: OR = 2.13) and cumulative smoking dose (≥ 40 pack/years: OR = 1.78)[14]. A meta-analysis of 82 studies from 4 continents has shown that cigarette smokers were diagnosed at significantly younger age and had a 75% increased risk of developing PC in comparison to the regular population[15] and the risk persisted for 5 to 15 years after cessation[16]. In a case-control study of 808 PC patients matched against 808 healthy controls, female smokers were at increased risk in comparison to males as they suffered from a synergistic interaction between cigarette smoking, diabetes mellitus (OR = 9.3) and family history of PC (OR = 12.8)[17].
Nearly 80% of PC patients have either frank diabetes or impaired glucose tolerance[18]. Diabetes is usually diagnosed either concomitantly or during the two years preceding the diagnosis[19]. Several studies have assessed the role of diabetes in PC with conflicting results. A meta-analysis of 11 cohort studies found that the relative risk for diabetics was 2.1 [95% confidence interval (95% CI): 1.6-2.8][20]. These findings were supported by another cohort study of 100 000 Danish diabetic patients which found a standardized incidence ratio of 2.1 (95% CI: 1.9-2.4) in a 4-year follow-up[21]. A large prospective cohort study of 20 475 men and 15 183 women in the United States, has shown that the relative risk of PC mortality adjusted for age, race, cigarette smoking, and body mass index (BMI) was proportionate to the severity of abnormal glucose metabolism: RR was 1.65 for post load plasma glucose levels between 6.7 and 8.8 mmol/L; 1.60 for levels between 8.9 and 11.0 mmol/L, and 2.15 for levels equal or more than 11.1 mmol/L[22]. Diabetes can be an early manifestation of PC as about 1% of new onset of diabetes in patients older than 50 is linked to PC[23], but there is no evidence that screening for recent onset diabetes would reduce the mortality[12] or lead to early diagnosis[24].
The link between abnormal glucose and PC exists only for type II diabetes. A meta analysis of 36 studies has shown that the OR of PC for patients with type II diabetes for more than 5 years was 2.1[25], while there are no reports on the association between PC and type I diabetes[26].
Family history of diabetes does not appear to be a risk for PC. Compared to subjects with no family history, diabetics with a positive family history have an OR of 0.8 while non-diabetics with a positive family history have an OR of 1.0[27].
A recent prospective study found that women with gestational diabetes have a relative risk of PC of 7.1 (95% CI: 2.8-18.0)[28]. Gapstur and colleagues have proposed a mechanism to explain these findings[22] by the fact that at high levels, insulin binds to the insulin-like growth factor I (IGF1) receptor[24] and downregulates IGF binding protein 1[25] causing an increase in cell growth in PC cell lines[29,30].
The role of alcohol is controversial and several studies have shown inconsistent findings. This might be attributed to multiple associations with confounding variables mainly smoking, socio-economic status[31] and pancreatitis[30]. A recent pooled analysis of 14 cohort studies with a sample of 862 664 individuals has shown a slight positive association between PC and alcohol intake only for consumption above 30 g/d (RR = 1.22; 95% CI: 1.03-1.45)[32]. Contrasting findings were reported by a European epidemiological study with a smaller sample size (n = 555) that did not show any association between PC and alcohol consumption[33].
Compared with light drinkers, men consuming large amounts of hard liquor suffered from a 62% increased risk of PC (95% CI: 1.24-2.10)[16,34], but this was not observed for women or for beer and wine drinkers[34].
Although moderate alcohol consumption is not a risk factor, African Americans were found to have a significantly higher OR when adjusted for their drinking habits, suggesting that racial differences might play a role in the development of PC[35].
Several studies have shown a positive association between PC and history of pancreatitis, although the magnitude is still controversial[36,37]. An international epidemiological study reported that both genders with chronic pancreatitis had an increased risk independently of the cause of pancreatitis[37]. A large case-control study showed that chronic pancreatitis lasting more than 7 years was associated with a higher risk of PC (RR = 2.04; 95% CI: 1.53-2.72)[38]. A large Italian study from 1983 to 1992 found similar results, as the risk increased after 5 or more years of chronic pancreatitis (RR in the first 4 years = 2.1, RR after 5 years = 6.9)[34]. These findings have been challenged by an international study, as the risk was significantly increased only in the early years after diagnosis. This would suggest that pancreatitis might represent a manifestation of PC that becomes apparent only several years later, rather than a risk factor. The risk of PC in chronic pancreatitis has been shown to be especially true for patients affected by hereditary pancreatitis, who were found to have 53 times the risk in comparison to normal individuals[39]. This was confirmed by another study that estimated a 40% cumulative risk of PC in patients with hereditary pancreatitis by the age of 70. For patients with paternal inheritance, the cumulative risk of PC was even higher with risk up to 75%[40]. Cytokines, reactive oxygen molecules and pro-inflammatory compounds seem to be responsible, as inflammation is a risk factor for many other solid tumors[38].
Genetic predisposing factors have been a topic of intense research in the last decades. Case reports of families with multiple affected members suggest that PC might have a hereditary background[41]. Yet, a large population study on twins identified hereditary factors for prostatic, breast and colorectal cancers, but not for PC[42]. A Canadian study on patients with suspected hereditary cancer syndromes found that the standardized incidence rate of PC was 4.5 (CI 0.54-16.) when cancer affected one 1st degree relative, and increased to 6.4 (CI 1.8-16.4) and 32 (CI 10.4-74.7) when two and three 1st degree relatives were affected, respectively[43]. This translates to an estimated incidence of PC of 41, 58 and 288 per 100 000 individuals, respectively, compared to 9 per 100 000 for the general population[44].
Brentnall et al[45] and Meckler et al[46] described examples of autosomal dominant PC in individuals presenting at early age (median age 43 years) and with high genetic penetrance (more than 80%). A mutation causing a proline (hydrophobic) to serine (hydrophilic) amino acid change (P239S) within a highly conserved region of the gene encoding paladin (PALLD) was found in all affected family members and was absent in non-affected individuals of the same family (family X). Another study has shown that the P239S mutation was only specific for family X and was not a common finding in other individuals with suspected familial PC[47]. Currently, genetic predisposition is thought to be responsible for 7% to 10% of all PC[48]. Genetic factors including germline mutations in p16/CDKN2A[49], BRCA2[50-52] and STK 11[53] genes increase the risk of PC. The combination of all these known genetic factors accounts for less than 20% of the familial aggregation of PC, suggesting the role of other additional genes.
A systematic review and meta analysis of studies that quantified familial risk of PC has shown that individuals with positive family history have an almost two-fold increased risk (RR = 1.80, CI 1.48-2.12)[54]. Therefore, families with two or more cases may benefit from a comprehensive risk assessment involving collection of detailed family history information and data regarding other risk factors[55]. A case-control study of PC in two Canadian provinces (Ontario and Quebec) assessed a total of 174 PC cases and 136 healthy controls that were compared for their family histories of cancer. Information regarding the ages and sites of cancer was obtained in 966 first degree relatives of the PC patients and for 903 first degree relatives of the control group. PC was the only malignancy in excess in relatives of patients with PC, compared to the control group (RR = 5, P = 0.01). The lifetime risk of PC was 4.7% for the first degree relatives and the risk was 7.2% for relatives of patients diagnosed before the age of 60[56].
Besides the isolated aggregation of PC in some families, several other hereditary disorders predispose to PC in known familial cancer conditions[57]. These include hereditary pancreatitis, Puetz-Jeghers syndrome, familial atypical multiple mole melanoma, familial breast and ovarian cancer, Li-Fraumeni syndrome, Fanconi anaemia, Ataxia-telangiectasia, familial adenomatous polyposis, cystic fibrosis and possible hereditary non-polyposis colon cancer or Lynch syndrome[11,55,58-60].
As the prognosis of PC is generally poor, there has been a strong interest in detecting genes or other markers that could help identify high risk patients at an early stage. Although a precise genetic marker for this scope is not currently available, geneticists and epidemiologists have been profiling traits of high risk families enrolled in registries established in North America and Europe[61]. Even if there is no standardized definition for familial PC, most authors apply the term to families with at least two first degree relatives affected by PC in the absence of other predisposing familial conditions[61]. The creation of familial PC registries has been used not only for identification of genetic mutations, but also for the screening of high risk individuals. In selected centers in North America and Europe, screening programs for high risk individuals have been implemented with the use of endoscopic ultrasound (EUS) and computed tomography (CT) scanning or magnetic resonance imaging (MRI). Such early diagnosis of PC within a comprehensive screening program is hoped to ultimately result in improved survival[62]. The discovery of the genetic bases of inherited PC continues to be an active area of research, and in 2001 a multi-center linkage was formed to conduct studies aimed at the localization and identification of PC susceptibility genes (PACGENE)[63]. The complex nature of pedigree data makes it difficult to accurately assess risk based upon the simple counting of the number of affected family members, as it does not adjust for family size, age of onset of PC, and the exact relationship between affected family members. Therefore, computer programs have been developed to integrate these complex risk factors and pedigree data. In April 2007, the 1st risk prediction tool for PC, PanaPro was released[64]. This model provides accurate risk assessment for kindreds with familial PC as the receiver operating characteristic (ROC) curve was 0.75 which is considered good for predictive models.
A number of studies have explored the relationship between BMI, lifestyle, diet and the risk of PC, but uncertainty regarding the strength of this relationship still exists. A recent case-control study of 841 patients and 754 healthy controls showed that individuals with a BMI of 25-29.9 had an OR of 1.67 (95% CI: 1.20-2.34) in comparison to obese patients (BMI of ≥ 30) who had an OR of 2.58 (95% CI: 1.70-3.90) independently of their diabetes status[65]. The duration of being overweight was significantly longer among patients with PC than controls. Being obese or overweight, particularly in early adulthood, resulted in earlier onset of PC (age at presentation of PC was 61 years for overweight patients and 59 years for obese) when compared to the median age of diagnosis (64 years) in the general population[66]. A number of studies reported that central weight gain measured by waist circumference and/or waist-to-hip ratio had a statistically significant increased risk compared to those with peripheral weight gain (RR = 1.45, 95% CI: 1.02-2.07)[67,68]. The known risk factors for PC are summarized in Table 2.
| Age (more than 60 yr) |
| Smoking |
| Diabetes |
| Type II |
| Gestational diabetes |
| Impaired glucose tolerance |
| Alcohol |
| Pancreatitis |
| Acute |
| Chronic |
| Genetic predisposition |
| Family history |
| Hereditary disorders |
| Hereditary pancreatitis |
| Puetz-Jeghers syndrome |
| FAMMM |
| Familial breast and ovarian cancer |
| Li-Fraumeni syndrome |
| Fanconi anaemia |
| Ataxia-telangiectasia |
| Familial adenomatous polyposis |
| Cystic fibrosis |
| HNPCC |
| Lynch syndrome |
| Obesity |
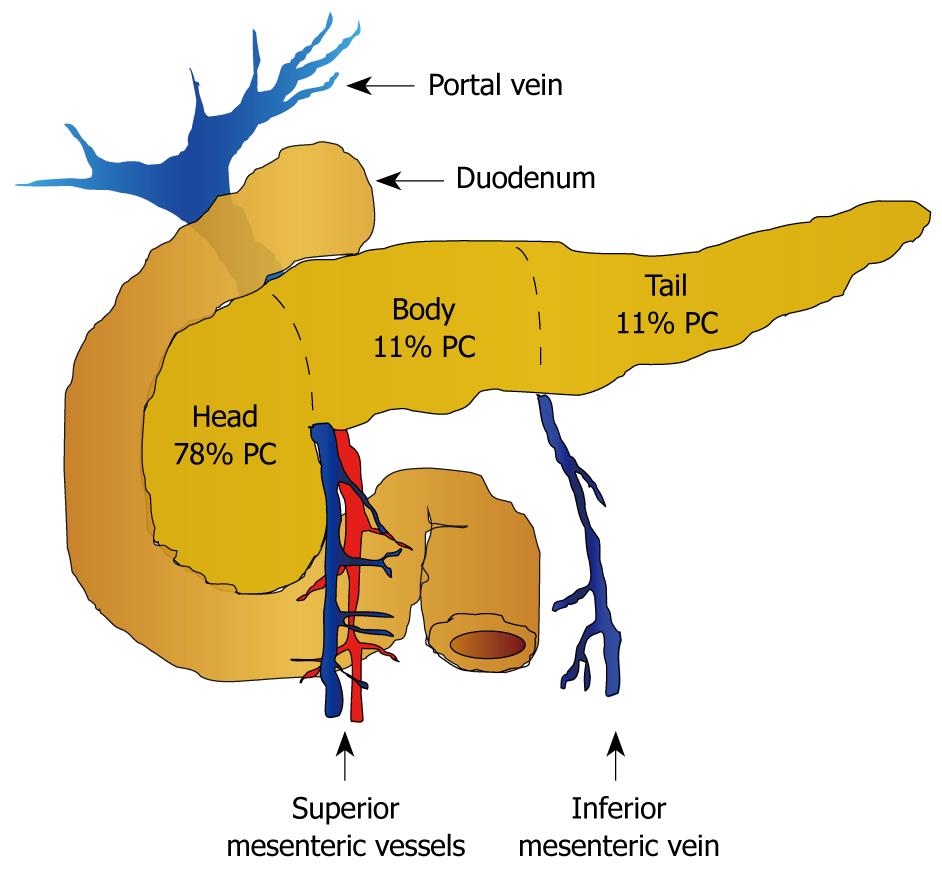
According to the location, PC can be divided in three groups: tumors of the head, body and tail. PCs of the head are at the right side of the superior mesenteric vessels, and tumors of the neck and body are located between the superior mesenteric vessels and the inferior mesenteric vein. PCs of the tail are located to the left of the inferior mesenteric vein.
A large epidemiological study[2] of 100,313 patients in the United States has shown that 78% of PC presents in the head, 11% in the body and 11% in the tail (Figure 1).
Recent advances in surgical pathology techniques integrated with molecular biology have allowed advances in the modern classification of PC. A summary of the clinico-pathological features of the different categories of PC is shown in Table 3.
| Classification | Frequency (%) | Author | yr | Survival (5-yr survival after surgical resection) |
| DIA (incidence per 100 000 patients at risk = 8.37)[69] | 85-90[1] | Conlon et al[70] | 1996 | 10% |
| Winter et al[71] | 2006 | 18% | ||
| Poultsides et al[72] | 2010 | 19% | ||
| SPPN (incidence per 100 000 patients at risk = NA)[69] | 0.1-3[73] | Papavramidis et al[74] | 2005 | 95% |
| IPMN (incidence per 100 000 patients at risk = 0.03)[69] | Shin et al[76] | 2010 | Benign: 95% | |
| Malignant: 64% | ||||
| IPMN with simultaneous DIA: (incidence per 100 000 patients at risk = NA)[69] | 5[75] | Poultsides et al[72] | 2010 | 42% |
| Fan et al[77] | 2010 | 57% | ||
| Sohn et al[78] | 2004 | 43% | ||
| Pancreatoblastoma (incidence per 100 000 patients at risk = NA)[69] | 0.50[79] | Dhebri et al[80] | 2004 | 50% |
| Saif et al[79] | 2007 | 80% | ||
| Undifferentiated (incidence per 100 000 patients at risk = 0.03)[69] | 2-7[81] | Paal et al[82] | 2001 | 3% (3-yr survival) |
| Connolly et al[83] | 1987 | 5 mo (average survival) | ||
| Medullary carcinoma (incidence per 100 000 patients at risk = NA)[69] | NA | Wilentz et al[84] | 2000 | 11% |
| 14 mo (average survival) | ||||
| Mucinous cystadenocarcinoma (incidence per 100 000 patients at risk = 0.43)[69] | 1 | Ridder et al[85] | 1996 | 56% |
| Adenosquamous carcinoma (incidence per 100 000 patients at risk = 0.05)[69] | 4 | Madura et al[86] | 1999 | 5-7 mo (median survival) |
| Mulkeen et al[87] | 2006 | |||
| Acinar cell carcinoma (incidence per 100 000 patients at risk = 0.02)[69] | 2 | Holen et al[88] | 2002 | 38 mo after surgical resection (median survival) |
| 14 mo for unresectable disease (median survival) |
Ductal infiltrating adenocarcinoma (DIA) represents the most common type (85%-90%) of PC originating from ductal epithelial cells. Most DIAs appear as whitish masses, hard at palpation and poorly defined from surrounding tissues, predominantly solid although cystic degenerations can be seen in larger tumors[89]. The microscopic appearance of DIA ranges from well-differentiated neoplasms difficult to distinguish from reactive gland, to poorly differentiated. The majority of DIAs are moderately to poorly differentiated and develop a dense desmoplastic stroma[89]. Mutations in the KRAS2 or p16/CDKN2A genes are observed in 90% of patients, TP53 gene abnormalities in more than 75% and more than 55% of cases have changes in MADH4/DPC4 genes. Tumors showing loss of DPC4 expression have a worse outcome than those with intact DPC4[90], and immunolabeling for DPC2 protein can help to classify metastatic carcinomas of unknown primary etiology[91].
Solid pseudopapillary neoplasms (SPPN) represent a small proportion of PCs (3%) and present as solid, or solid and cystic masses. They are malignant epithelial neoplasms made of poorly cohesive cells that form pseudo-papillae around the blood vessels[91]. The majority of SPNN are grossly well demarcated, but typically do not have a well formed capsule. The majority are solid, yellowish and soft[92]. Larger tumors usually develop cystic degeneration filled with blood and necrotic debris. Cases that are almost completely cystic without a solid component have also been reported[91].
Molecular analyses have shown that SPNN are different from ductal adenocarcinomas as they do not harbor mutations in the Kras 2, p16/CDKN2A, TP53, MADH4/DPC4 genes[93]. In contrast, 90% of SPNN have a mutation on chromosome 3p (CTNNB1) responsible for the metabolism of β-catenin protein causing its accumulation in the cytoplasm and nucleus of neoplastic cells[94]. As a result alteration in β-catenin protein expression disrupts E-cadherin which is a key regulator of cell junctions causing poor adhesion of neoplastic cells[95]. Although there is some histological overlap between SPNN and other tumors of the pancreas, immunolabeling for β-catenin protein may help establish the diagnosis.
Intraductal papillary mucinous neoplasms (IPMNs) represent 5% of all PCs and are papillary epithelial mucin-producing neoplasms arising in the main pancreatic duct or in one of its branches. IPMNs are relatively common with increasing age of the population[91] and the mean age at presentation is 65 years[96]. IPMN is a potential pre-malignant condition and the risks of developing invasive adenocarcinoma increase with tumor size and when originating in the main pancreatic duct.
Adenocarcinoma is present in up to one-third of patients with IPMN and current guidelines recommend surgical resection when IPMNs are greater than 3 cm, in the presence of main pancreatic duct dilatation and when mural nodules are detected[97].
Neoplastic cells of IPMN are columnar with gene profiles similar to infiltrating ductal carcinoma. About 25% of patients show loss of heterozygosity of the STK11/LKB1 gene[98,99]. Other frequent gene mutations are TP53, KRAS2, and P16/CDKN2A[100].
Pancreatic intraepithelial neoplasia (PanIN) represents a neoplastic proliferation of mucin producing epithelial cells confined to the smaller pancreatic ducts and is considered a precursor to invasive ductal carcinoma[101].
PanINs are usually characterized by lesions too small to be symptomatic or to be detected by current imaging technologies[89]. Microscopically, PanINs are classified into three grades (PanIN-1, PanIN-2 and PanIN-3) based on the progressive degree of architecture abnormality and cellular atypia[102]. PanIN-1 shows minimum cellular atypia, PanIN-2 moderate changes and PanIN-3 is equivalent to PC-in-situ. The discovery of specific molecular changes present in both PanIN and PC has helped to establish that these small lesions are the precursors to DIA[103]. Early abnormalities of IPMNs are telomerase shortening and activating point mutations in the KRAS2 gene while intermediate mutation is the activation of the p16/CDKN2A gene and late events are alterations in the TP53, MADH4/DPC4, and BRCA2 genes[102]. The understanding that many DIAs arise from PanIN lesions has prompted screening efforts on the detection of these small and potentially curable lesions[104].
Pancreatoblastoma is a rare malignant tumor (0.5% of PC) usually presenting in the pediatric age group. Generally, it appears as a soft and well demarcated mass with epithelial or acinar differentiation, but often it has cells with endocrine and mesenchymal characteristics[79]. Most pancreato-blastomas affect children with a mean age of 5 years and are frequently associated with elevated levels of serum alpha fetoprotein. The median survival of patients with pancreato-blastomas is 48 mo and the 5-year survival rate after successful resection is 50% (95% CI: 37%-62%)[80,105].
The majority of pancreato-blastomas have loss of heterozygosity of chromosome 11p from the maternal side[106]. These molecular findings unite pancreatoblastoma with other primitive neoplasms such as hepatoblastoma and nephroblastoma[107]. Genetic alterations in the adenomatous polyposis coli (APC)/β-catenin pathway have also been detected in most pancreato-blastomas including mutations in β-catenin (CTNNB1) and APC genes[107].
Undifferentiated PC (UPC) lacks differentiation direction[91] and presents with symptoms similar to patients with DIA, but has a worse prognosis as it has a more aggressive behavior and tends to metastasize and infiltrate surrounding organs in early stages[82]. The average time from diagnosis to death is about 5 mo and only 3% of patients are alive at 5 years after undergoing surgical resection. UPCs can form large locally aggressive masses and may present with severe hemorrhage and necrosis. The majority of UPCs have KRAS2 gene mutation suggesting that they arise from pre-existing ductal adenocarcinomas that transform into poorly differentiated tumors during their progression[108].
Medullary carcinoma (MC) is a variant of PC characterized by poor differentiation and syncytial growth that has been described and recognized only in recent years[84]. Patients with MC have a better prognosis and are more likely to have a family history of any kind of cancer[109]. MC does not differ significantly from other classes of PC in its clinical presentation, age and gender. These tumors tend to form well demarcated soft masses and microscopically they are usually poorly differentiated with pushing rather than infiltrating features[110]. Focal necrosis and intratumoral lymphocytic infiltration can be prominent similar to MC of the colon and other tumors with microsatellite instability[89]. MCs have been shown to have loss of expression of one of the DNA mismatch repair proteins (M1h1 and Msh2) and mutation in the BRAF gene, which is a downstream effector of the k-ras pathway[111]. Patients with MC and their families may benefit from genetic counseling and more frequent screening for early detection of other common cancers. The prognosis of MC is better than adenocarcinoma, although it is not responsive to adjuvant chemotherapy based on fluorouracil (5-FU), similar to colon cancer with microsatellite instability[112].
Mucinous cystadenocarcinoma: Malignant cystic neoplasms are rare entities that account for only 1% of all pancreatic tumors[113]. Both serous and mucinous cystic neoplasms are tumors of the exocrine pancreas with different biological behaviors. Serous cystadenomas are considered benign tumors with almost no malignant potential often managed expectantly unless symptomatic. However, the preoperative differentiation between a benign serous cystadenoma and malignant serous cystadenocarcinoma remains difficult[114]. Histologically, cystadenocarcinomas appear identical to serous cystadenomas and are distinguished only by the presence of lymphovascular invasion or metastases[115]. Mucinous cystadenocarcinomas resemble DIAs although some cell populations can present with undifferentiated features and other histological characteristics such as osteoclast-like giant cells, adenosquamous carcinoma, choriocarcinoma, or high-grade sarcoma[116-119]. Mucinous cystic neoplasms of the pancreas are slowly growing and only about 20% show invasive features[120,121].
The prognosis of cystadenocarcinoma is favorable compared to DIA with 5-year survival rates of 56% after radical resection[85]. There is limited evidence on the role of chemotherapy for cystadenocarcinomas of the pancreas as they appear to be unresponsive to current chemotherapy agents and radiation therapy[122,123].
Adenosquamous carcinoma: Adenosquamous carcinoma has previously been referred as adenoachantoma, mixed squamous and adenocarcinoma, and mucoepidermoid carcinoma. Histologically, they are characterized by mixed populations of adenomatous cells and cells with varying amount of keratinized squamous features. Usually this tumor affects patients in their seventh decade of life, with symptoms and pancreatic distribution similar to DIAs. Although it is reported that adenosquamous carcinomas represents 4% of all PCs (range 3%-11%), the literature on the natural history and survival is limited to case series only[86]. The prognosis seems to be worse than DIAs, with a mean survival of 5-7 mo even after surgical resection[86,87]. Lymphovascular and perineural invasion appear to be common and early features of adenosquamous carcinomas and the role of adjuvant chemo and radiation therapy is still not clear[124].
Acinar cell carcinoma: Acinar cell carcinomas (ACCs) represent less than 2% of all pancreatic malignancies[87,88]. ACCs are predominantly constituted by neoplastic cells with immunohistochemical staining characteristic for exocrine enzymes such as trypsin, chymotripsin or lipase, and they present in older patients than DIAs and the prognosis is slightly better, although the literature is somewhat limited[125,126]. Symptoms at presentation are aspecific and include abdominal pain and weight loss that are similar to all other PCs[125]. Very rarely, patients with ACC can develop subcutaneous fat necrosis secondary to exceedingly high concentrations of serum lipase and contrary to DIAs, bile duct obstruction causing jaundice is not as common[125]. Median survival for ACC confined to the pancreas treated by surgical resection is 38 mo, whereas it is 14 mo for individuals with unresectable disease[88]. For the majority of patients, surgical management is not curative as distant recurrent disease is more frequent than in DIA, suggesting the presence of early micrometastases even when the tumors are in the early stages[88]. Because ACCs are rare, there is a lack of studies on the role of chemotherapy, although radiation therapy seems to provide good responses in patients with regional unresectable disease[88].
Early symptoms of PC are notoriously difficult to measure as educational and economic factors influence their perception and reporting[127,128]. Cholestatic symptoms are more common in early PC of the head, while abdominal and back pain are more common in patients with distal PC and in patients with tumors infiltrating peripancreatic nerve tissue[129]. The appearance of these symptoms usually indicates advanced disease (Table 4)[129,130].
| Symptom | Percentage |
| Abdominal pain | 78-82 |
| Anorexia | 64 |
| Early satiety | 62 |
| Jaundice | 56-80 |
| Sleep disorders | 54 |
| Weight loss | 66-84 |
| Diabetes | 97 |
| Back pain | 48 |
| Nausea and weight loss | 50-86 |
Early symptoms are usually vague such as anorexia, moderate weight loss, and early satiety[131]. Diabetes might be a sign of PC particularly when presenting during or beyond the sixth decade of life in the absence of risk factors and family history[20]. Diabetes is detected in 60%[132] to 81%[133] of PC patients within two years of their diagnosis. Early detection is possible if symptoms raise clinicians’ suspicion, as 25% of patients report upper abdominal discomfort up to 6 mo prior to their diagnosis[134,135].
In two European studies[128,130], weight loss was present in 66%-84% of patients, jaundice (bilirubin level > 3 mg/dL) in 56%-61%, recent onset of diabetes in 97% and distended palpable gall bladder in 12%-94%, energy loss in 86%, abdominal pain in 78%, back pain in 48%, nausea in 50%, clay-coloured stools in 54%, dark urine in 58%, jaundice in 56% and pruritis in 32% of patients.
Several serum tumor markers are associated with PC, however, to date, no single marker has been found to be optimal for screening.
Carbohydrate antigen 19-9: Carbohydrate antigens have been used as markers for several cancers[136,137]. The production of these antigens seems to be caused by the upregulation of glycosyl transferase genes[138]. Among these carbohydrate antigen epitopes, Sialyl Lewisa (sLea) detected by the 1116NS19-9 monoclonal antibody is commonly called carbohydrate antigen 19-9 (CA19-9)[139]. The serum levels of CA19-9 at the time of diagnosis and during follow-up of PC provide useful diagnostic and prognostic information[140,141]. Its sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) are 70%-90%, 43%-91%, 72% and 81%, respectively[142-145]. A worse survival was observed in patients with pre-operative CA19-9 levels above 370 U/mL (median survival 4.4 mo vs 9.5 mo if CA19-9 < 370 U/mL, P value < 0.01)[146]. In another study, serum levels of CA19-9 > 200 U/mL were associated with a survival rate of 8 mo compared to 22 mo for patients with lower tumor antigen levels (P < 0.001)[147]. In a prospective study of patients undergoing curative resection for PC, post-operative CA19-9 < 37 U/mL was associated with a longer median and disease-free survival compared to the control group[148-150]. One of the limitations of CA19-9 is that high serum bilirubin can falsely increase its level and therefore the risk of false positive results in patients with jaundice. This is not observed for other markers such as carcinoembryonic antigen (CEA) and carbohydrate antigen 242 (CA 242)[141].
CEA: CEA is part of a subgroup of glycoproteins functioning as intracellular adhesion molecules. CEA was first detected in pancreatic secretions, and several studies have shown high levels of CEA in the pancreatic juice of patients with PC[151-153]. A Japanese study found significantly higher CEA levels in the pancreatic juice of PC patients compared to those with benign pancreatic diseases. When the CEA cut off level in pancreatic juice was 50 ng/mL, the PPV, NPV, and the accuracy for diagnosis of carcinoma were 77%, 95% and 85%, respectively. CEA levels in pancreatic juice were higher in smaller tumors in comparison to advanced PC due to the incomplete obstruction of the pancreatic duct[154]. A recent study examining single vs combined efficacy of tumor markers showed that CEA (> 5 ng/mL) alone had a sensitivity of 45% and a specificity of 75% in comparison to CA19-9 which had a sensitivity of 80% but lower specificity (43%) (P = 0.005)[141,155]. The combination of CEA (> 5 ng/mL) and CA 19-9 (> 37 U/mL) decreased the sensitivity to 37%, but increased the specificity to 84%. Similarly, the combination of CEA (> 5 ng/mL) and CA242 (> 20 U/mL) decreased the sensitivity to 34% and increased the specificity to 92%. Yet, CEA and CA242 are currently not used as single tumor markers for PC, and the simultaneous use of CEA and CA19-9 provides the same information as CA19-9 alone[156-158].
CA 242: CA 242, a sialylated carbohydrate was first defined by Lindholm et al in 1985 and has been used for diagnostic and prognostic purposes[159,160]. For PC, its diagnostic sensitivity and specificity are 60% (P = 0.073) and 76% (P = 0.197), respectively, comparable to CEA. It also seems to be valuable in differentiating PC from benign pancreatic tumors as well as other hepatobiliary cancers and to predict outcomes as survival rates in CA 242 positive patients are lower than those with negative serum levels (P = 0.002)[141].
In a study comparing CA 242 and CA19-9[161], CA 242 appeared to be an independent prognostic factor for patients with resectable disease as serum levels of CA 242 < 25 U/mL were associated with a significantly better survival (P < 0.05). For patients with unresectable disease, poorer outcomes were observed when CA 242 levels were > 100 U/mL.
Similar results have been confirmed by Ni et al, who found that CA 242 is an independent prognostic factor in PC yielding more information than CA 19-9[142,161]. In this study the use of combined tumor markers resulted in lower sensitivity, but higher specificity (Table 5). Despite these findings, CA 242 is not used in clinical practice as commonly as Ca 19-9 due to the limited number of laboratories equipped to run this test.
| Serum tumor marker | Author | Yr | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) |
| CA19-9 | Boeck et al[141] | 2006 | 70-90 | 43-91 | 72 | 81 | 67 |
| Ni et al[142] | 2005 | ||||||
| Steinberg et al[143] | 1990 | ||||||
| Safi et al[144] | 1997 | ||||||
| Mu et al[162] | 2003 | ||||||
| CEA in pancreatic juice | Ozkan et al[155] | 2003 | NA | NA | 77 | 95 | 85 |
| Futakawa et al[154] | 2000 | ||||||
| Ni et al[142] | 2005 | ||||||
| CEA in serum | Boeck et al[141] | 2006 | 45 | 75 | NA | NA | NA |
| CA19-9 + CEA | Ni et al[142] | 2005 | 37 | 84 | 91 | 90 | 89 |
| Ozkan et al[155] | 2003 | ||||||
| Ma et al[163] | 2009 | ||||||
| CA 242 | Nilsson et al[160] | 1992 | 60 | 76 | 63 | 61 | 71 |
| Röthlin et al[164] | 1993 | ||||||
| Carpelan-Holmström et al[165] | 2002 | ||||||
| Pålsson et al[166] | 1993 | ||||||
| CEA + CA 242 | Ni et al[142] | 2005 | 34 | 92 | 67 | 90 | 87 |
| Ozkan et al[155] | 2003 | ||||||
| Hall et al[167] | 1994 | ||||||
| CA19-9 + CA 242 | Ni et al[142] | 2005 | 59 | 77 | 65.3 | 87.8 | 65.1 |
| Röthlin et al[164] | 1993 | ||||||
| Jiang et al[158] | 2004 | ||||||
| CA19-9 + CA 242 + CEA | Ni et al[142] | 2005 | 29 | 96 | NA | NA | NA |
Recent studies have identified other serum molecules such as CA494[168], CEACAM1[169], PTHrP[170], TuM2-PK[171], CAM 17.1[172] and serum beta HCG[173] as potential markers for PC. Although preliminary results appear promising with sensitivity and specificity comparable and sometimes superior to CA19-9 and CEA, their clinical use has to be confirmed in larger studies and their role is currently confined to a limited number of medical centers and for research purposes.
Although PC may be detected with one particular diagnostic test, proper staging often requires the use of several imaging modalities[174].
Abdominal ultrasound: Trans-abdominal ultrasound (US) is currently used as a screening test for patients with suspected PC[175]. Its sensitivity ranges between 48%[176] and 89%[177], specificity between 40%[178] and 91%[179] and accuracy between 46%[176] and 64%[180]. PCs measuring less than 1 cm are detected by US in only 50% of cases, while the sensitivity increases to 95.8% for tumors larger than 3 cm[177]. Other factors affecting the sensitivity of US are the operator’s experience[181] and the technical characteristics of the machine. Newer US machines such as tissue harmonic imaging decrease artefacts and improve tissue contrast and therefore diagnostic accuracy[182]. US has a relatively low performance profile for the staging of PC as its sensitivity for lymph node involvement only ranges between 8%[159] and 57%[177].
Color Doppler US has been used to assess the possible involvement of the portal vein and superior mesenteric vessels with a sensitivity ranging between 50%[183] and 94%[184], specificity between 80% and 100%[183] and accuracy between 81% and 95%[175].
The recent introduction of intravenous contrast has been shown to improve evaluation of the vascularity of pancreatic lesions allowing differentiation between PC and other conditions with 90% sensitivity, 100% specificity and 93% accuracy[185]. Currently, US is considered a useful imaging modality for the initial screening of PC based on its ability to document unresectability (PPV = 94%)[176]. However, the PPV for resectabiltiy is only 55%[186], therefore, other imaging techniques are usually employed for better staging.
EUS: EUS provides high resolution images of the pancreas without interference by bowel gas[187]. Despite the advancement of CT scans, EUS appears to have a higher sensitivity in detecting small PCs (98%) in comparison to CT (86%)[188]. EUS has higher sensitivity compared to CT for local tumor staging (67% vs 41%), similar sensitivity for lymph node involvement (44% vs 47%) and potential tumor resectability (68% vs 64%)[185]. EUS has a NPV of 100% for PC of the head[186,189] and an accuracy of 90% for the assessment of portal and splenic vein invasion[178,190]. On the other hand, EUS does not appear to be accurate enough in assessing the invasion of SMA and superior mesenteric vein (SMV) with a NPV of 82% and sensitivity of only 50%[191,192].
In order to improve EUS performance in PC staging, recent studies have assessed the benefits of using parenteral contrast agents. This technique has shown 92% sensitivity, 100% specificity, 100% PPV, 86% NPV and 95% accuracy[193]. Although EUS is becoming a leading modality for staging and diagnosis of PC, drawbacks of this technique are the fact that it is invasive, highly operator dependent, costly and associated with a small risk of pancreatitis (0.85%)[194], bleeding and duodenal perforation.
CT: On contrast CT, PC appears as an ill-defined, hypo-attenuating focal mass with dilatation of the upstream pancreatic and or biliary duct[174]. Optimum visualization of the pancreas requires imaging acquisition obtained during both arterial and portal phases[195]. Sensitivity and specificity of thin section triple phase helical CT is 77% and 100%, respectively, for lesions less than 2 cm[196]. In a multicentric trial, the diagnostic accuracy of CT for resectability was 73% with a PPV for non resectability of 90%[197].
With the advent of multi detector CT scanners (MDCT), the pancreas can be imaged at a very high spatial and temporal resolution[198,199]. The dual phase pancreatic protocol MDCT using 1 to 3 mm slice collimation is one of the most sensitive techniques for metastatic disease to the liver and peritoneum[186,200,201]. Recent studies have shown that MDCT has a NPV of 87% for tumor resectability compared to a NPV of 79% for conventional helical CT[202] and with an accuracy between 85% and 95%[203,204].
Images from MDCT can be used to visualize the biliary tree and normal vascular variants such as replaced hepatic arteries before surgical planning. Gangi et al[198] reported that pancreatic ductal dilatation in asymptomatic patients could be identified between 0 to 50 mo before PC diagnosis was confirmed. The sensitivity, specificity and accuracy of CT in the presence of hypo-attenuated pancreatic lesions, pancreatic ductal dilatation with cut-off, distal pancreatic atrophy, pancreatic contour abnormalities and common bile duct dilatation are reported in Table 6[205].
| CT finding | Sensitivity (%) | Specificity (%) | Accuracy (%) |
| Hypoattenuation | 75 | 84 | 81 |
| Ductal dilatation | 50 | 78 | 70 |
| Ductal interruption | 45 | 82 | 70 |
| Distal pancreatic atrophy | 45 | 96 | 81 |
| Pancreatic contour anomalies | 15 | 92 | 70 |
| CBD dilatation | 5 | 92 | 67 |
Despite these improvements, interpretation of the CT scan is quite challenging in the setting of pancreatitis forming mass effects[206] and in the presence of loco-regional lymph node involvement and small hepatic metastasis[207].
Magnetic resonance imaging-magnetic resonance cholangiopancreatography: In most institutions, MRI is performed when other imaging modalities provide insufficient data for the clinical staging of the tumor, or when treatment planning can not be based on the images obtained by other techniques. Several studies have shown that MRI is superior to CT for the detection and staging of PC (100% vs 94%, respectively)[208-211]. However, recent evidence has challenged this belief. The use of MRI-magnetic resonance cholangiopancreatography (MRCP) to better characterize PC is supported by a prospective analysis that compared these two modalities in patients with periampullary cancers[212]. MRI-MRCP was superior to CT in differentiating malignant from benign lesions (ROC = 0.96 vs 0.81, P < 0.05) and MRI-MRCP had better sensitivity (92% vs 76%), specificity (85% vs 69%), accuracy (90% vs 75%), PPV (95% vs 88%) and NPV (79% vs 50%) compared to CT. Another study confirmed the previous results with MRI-MRCP showing 97% sensitivity, 81% specificity and 89% accuracy[213].
On the other hand, other studies comparing gadolinium-enhanced MRI with MDCT have shown that MRI and CT had equivalent sensitivity and specificity (83%-85% vs 83% and 63% vs 63%-75%, respectively). Both techniques had good to excellent agreement between radiologists, although MRI had a superior agreement for the evaluation of distant metastases (inter-observer agreement between MRI and CT scan; 0.78 vs 0.59 P = 0.1)[214]. On the other hand, with the improvement in CT scan technology, recent studies have shown that MRI might have lower sensitivity in comparison to MDCT (82%-94% vs 100%)[215]. This was confirmed by a recent meta-analysis comparing the accuracy of several imaging modalities which showed that helical CT had superior sensitivity compared to MRI (91% vs 84%) and transabdominal US (91% vs 76%)[216]. Sensitivity for resectability of the tumor was equal for both MRI and helical CT (82% vs 81%, respectively)[216].
Positron emission tomography:18F-2fluoro-2-deoxy-D-glucose (FDG) accumulated by tumor cells provides positron emission tomography (PET) with the advantage of combining metabolic activity and imaging characteristics. Newly developed PET scanners can detect small PCs up to 7 mm in diameter and diagnose metastatic disease in about 40% of cases[217,218]. A Japanese study found that the overall sensitivity of PET-CT was superior to contrast CT (92% vs 88%) and that PET was better at detecting bone metastases (100% vs 12%). However, CT scanning was superior for the evaluation of vascular invasion (100% vs 22%), involvement of para aortic regional lymph nodes (78% vs 57%), identification of peritoneal dissemination (57% vs 42%) and hepatic metastases (73% vs 52%)[219]. Another Japanese study confirmed that PET had a sensitivity of 87%, a specificity of 67% and accuracy of 85%, and that tumors with metastatic disease had significantly higher standardized uptake values [SUV = tissue concentration (millicuries/g)/injection dose (millicuries)/body weight (g)] than those without metastases[220]. PET had superior sensitivity (100% vs 65%), specificity (77% vs 61%), NPV (100% vs 31%), PPV (94% vs 87%) and accuracy (95% vs 65%) in an American study comparing PET-CT with a SUV cut off of 2.0 vs contrast CT[221]. A recent study enrolling 59 PC patients showed similar results, with 91% PPV and 64% NPV for PET-CT. One of the most interesting results was that the clinical management of patients undergoing PET was changed in 16% of cases deemed resectable after routine staging (P = 0.031) preventing unnecessary surgery because of distant metastases[222].
Diffuse uptake of FDG is frequent in pancreatitis in comparison to PC (53% vs 3%, P < 0.001), and therefore PET is extremely useful in distinguishing these two conditions in controversial cases[218,223]. Animal studies have shown that 11C-acetate-PET appears to be superior to FDG PET for the detection of early PC and might be useful in differentiating inflammatory processes from malignancies as 11C-acetate-PET is less affected by the presence of inflammation in human tissues[224].
Another very important characteristic of PET-CT is its ability to provide useful information on tumor viability, and this technique also allows monitoring of tumor response to treatment[217] and the metabolic features of PET help predict the prognosis as a SUV less than 3 appears to be a positive predictive factor[222,225-229].
Similar results were found by Zimny et al[230] who showed that better survival trends were noted in patients with PC and a SUV less than 6.0 in comparison to those with a higher SUV. Sensitivity and specificity of imaging modalities are summarized in Table 7.
| Diagnostic modality | Author | Yr | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) |
| US | Giovannini et al[176] | 1994 | 48-95 | 40-91 | 92 | 100 | 46-64 |
| Böttger et al[177] | 1998 | ||||||
| Rösch et al[178] | 1991 | ||||||
| Niederau et al[179] | 1992 | ||||||
| Palazzo et al[180] | 1993 | ||||||
| Tanaka et al[231] | 1996 | ||||||
| Doppler US | Candiani et al[232] | 1998 | 50-94 | 80-100 | 79 | 88 | 81-95 |
| Casadei et al[184] | 1998 | ||||||
| Calculli et al[233] | 2002 | ||||||
| EUS | Akahoshi et al[234] | 1998 | 98 | 97 | 94 | 100 | 90 |
| Legmann et al[235] | 1998 | ||||||
| Contrast enhanced US | Dietrich et al[185] | 2008 | 90 | 100 | 100 | 86 | 93 |
| CT | Bronstein et al[196] | 2004 | 77 | 100 | NA | NA | 73 |
| Megibow et al[197] | 1995 | ||||||
| MDCT | Park et al[214] | 2009 | 83-91 | 63-75 | 80 | 87 | 85-95 |
| Vargas et al[202] | 2004 | ||||||
| Diehl et al[203] | 1998 | ||||||
| Schima et al[208] | 2002 | ||||||
| MRI-MRCP | Andersson et al[212] | 2005 | 83-92 | 63-85 | 95 | 79 | 89 |
| PET | Maemura et al[217] | 2006 | 87-100 | 67-77 | 94 | 100 | 85-95 |
| Delbeke et al[221] | 1999 |
In the 7th edition of the American Joint Committee on Cancer the different categories of PC are classified according to only one TNM staging system, even if neuroendocrine tumors have a different biology and a better prognosis than ductal carcinomas. Yet, the TNM system provides a reasonable discrimination and prognostic validity for these patients[236].
The TNM system classifies PC into 3 clinically important categories: (1) patients with Tis-T2 PC have localized cancer within the pancreas; (2) patients with T3 cancer have locally invasive disease; and (3) patients with T4 tumors have unresectable PC[237] (Table 8).
| AJCC 6th edition TNM staging system for pancreatic cancer | ||
| TX | Primary tumor cannot be assessed | |
| T0 | No evidence of primary tumor | |
| Tis | Carcinoma in situ | |
| T1 | Tumor limited to the pancreas, 2 cm or less in greatest diameter | |
| T2 | Tumor limited to the pancreas, greater than 2 cm at greatest diameter | |
| T3 | Tumor extends beyond pancreas but no involvement of celiac axis or superior mestenteric artery | |
| T4 | Tumor involves the celiac axis or the superior mestenteric artery (unresectable) | |
| NX | Regional nodes cannot be assessed | |
| N0 | No regional lymph node metastasis | |
| N1 | Regional lymph node metastasis | |
| MX | Distant metastasis cannot be assessed | |
| M0 | No distant metastasis | |
| M1 | Distant metastasis | |
| Stage grouping | ||
| Stage 0 | Tis N0 M0 | Localized within pancreas |
| Stage IA | T1 N0 M0 | Localized within pancreas |
| Stage IB | T2 N0 M0 | Localized within pancreas |
| Stage IIA | T3 N0 M0 | Locally invasive, resectable |
| Stage IIB | T1, 2, or 3 N1 M0 | Locally invasive, resectable |
| Stage III | T4 Any N M0 | Locally advanced, unresectable |
| Stage IV | Any T Any N M1 | Distant metastases |
Prognostic features of PC include perineural and lymphovascular invasion, elevated serum CA19-9 levels and incomplete tumor resection. Therefore, gross and microscopic assessment of the resection margins is of major importance even if it is not included in the TNM staging system. Patients undergoing resections with grossly or microscopically positive margins have no survival benefits compared to individuals undergoing palliative chemo- radiation therapy alone.
Surgery is the only chance of cure and the presence of negative resection margins of the primary tumor represent the strongest prognostic factor. Preoperative staging modalities include the combination of several imaging techniques such as CT scan, MRI, EUS, staging laparoscopy and laparoscopic ultrasound which aim to identify patients with resectable disease. There is consensus that patients with distant metastases (liver, lungs, peritoneum) or local invasion of the surrounding organs (stomach, colon, small bowel) are usually not surgical candidates.
The criteria for unresectability of PC include tumor encroachment (defined as tumor surrounding the vessel more than 180 degrees) of arteries such as the celiac artery, hepatic artery, superior mesenteric artery (SMA) or massive venous invasion with thrombosis. Portal or superior mesenteric venous invasion without thrombosis or obliteration of vessels can still be classified as resectable PC[204,238]. A recent study comparing the roles of EUS, CT, MRI and angiography in the assessment of PC staging and resectability has shown that CT scanning was the most accurate in assessing the stage of the tumor (73%), loco-regional invasion (74%), vascular involvement (83%), distant metastases (88%), final TNM stage (46%) and overall tumor resectablity (83%)[239]. EUS appeared to be superior in detecting smaller tumors not visualized by CT. A decision analysis demonstrated that the best strategy to assess tumor resectability was based on CT as an initial test and the use of EUS to confirm the results of resectability by CT[221].
Diagnostic laparoscopy for PC was first introduced as a staging procedure in the late 1980s by Cuschieri et al[240] and Warshaw[241,242]. Staging laparoscopy is considered a simple, minimally invasive technique to identify radiographically occult distant metastatic disease and to prevent non-therapeutic laparotomies. Laparoscopic examination allows direct visualization of intra-abdominal contents and has been reported to identify hepatic and peritoneal metastases not shown by other modalities[243] as reported in some studies where 20%-48% of patients considered resectable by CT were found to be unresectable during surgery[244-246].
Diagnostic laparoscopy involves a general exploration of the abdominal surfaces including palpation of the liver with two instruments when necessary. The hilum of the liver is visualized, the foramen of Winslow is examined and periportal lymph nodes are biopsied when enlarged. The transverse colon and omentum are reflected cephalad and the base of the transverse mesocolon is examined with particular attention to the mesocolic vessels. The gastrocolic ligament/omentum is incised and the lesser sac is examined[247].
Laparoscopic ultrasonography (LUS) has been introduced as an additional procedure to increase the detection of intrahepatic metastases, identify enlarged and suspicious lymph nodes and to evaluate local growth in the vascular structures[248]. Some studies have demonstrated that LUS has improved the accuracy of predicting resectability up to 98%[249-251].
Despite these results, the routine use of staging laparoscopy and LUS in patients with radiographically resectable PC remains controversial as imaging modalities have significantly improved, thus reducing the risk of discovering non-resectable disease at the time of surgery. In addition, staging laparoscopy adds costs and it can be time consuming. Sustainers of staging laparoscopy are supported by a study by Kwon et al[250], which revealed that staging laparoscopy was able to detect unsuspected metastases and changed the surgical approach in 37% of patients even when using CT, MRI, ERCP and angiography for preoperative staging. Another study by Conlon et al[247], supported the use of staging laparoscopy as only 67 out of 115 patients (58%) with PC had resectable disease after completion of the laparoscopic examination. On the other hand, a more recent study from the same group at the Memorial Sloan-Kettering Cancer Center has shown that the yield of staging laparoscopy was only 8.4% when good imaging modalities were obtained at the referral center[252].
Based on the fact that minimally invasive approaches for the diagnosis of PC as well as radiological imaging techniques will continue to advance, the selective use of staging laparoscopy and LUS would play a role in cases where detection of unresectable disease is more likely. Factors which suggest a higher yield with diagnostic laparoscopy include a large primary tumor (diameter larger than 4 cm), a tumor in the body or tail of the pancreas, equivocal findings after imaging tests, severe weight loss, abdominal or back pain, hypoalbuminemia and significantly elevated tumor markers[240].
Patients with suspected or confirmed diagnosis of PC should be assessed by a multidisciplinary team and stratified as resectable (stage I or II), borderline resectable (stage IIa or IIb), locally advanced unresectable (stage III) or metastatic disease (stage IV). Treatment should be planned according to local expertise and established guidelines, as resectable and borderline patients should be referred to surgeons, unresectable and metastatic patients should be referred to medical and radiation oncologists and palliative care teams. A multidisciplinary approach to PC is necessary to improve the overall outcome of these patients, especially for borderline resectable or unresectable disease as neo-adjuvant chemo-radiation therapy may play a role in downstaging and the conversion to potentially curable disease[253,254].
Surgical treatment is the only potential cure for PC[255]. Although pancreatic surgery is considered challenging and technically demanding, improvements in surgical techniques and advances in perioperative supportive care have reduced the mortality rates to less than 5% in high-volume centers[256-258]. According to the United States Surveillance and Epidemiology End Results registries, the 5-year relative survival for the period between 1999 and 2006 was 22.5% for localized and 1.9% for metastasizing PC (Table 9)[259].
| Stage at diagnosis | Stage distribution (%) | 5-yr relative survival (%) |
| Localized (confirmed to primary site) | 8 | 22.5 |
| Regional (spread to regional LNs) | 26 | 8.8 |
| Distant (cancer had metastasized) | 53 | 1.9 |
| Unknown (unstaged) | 14 | 5 |
Because only 20% of patients with PC are candidates for radical resection at the time of diagnosis[260], accurate staging is important in identifying surgical candidates and sparing the risk and cost of surgery for patients who are affected by advanced disease[261]. Unresectable PC is commonly defined when there is tumor invasion of the SMA, inferior vena cava, aorta or celiac arteries; encasement or occlusion of the SMV-portal venous system or by distant metastasis (e.g. hepatic, extra-abdominal, peritoneum, omentum, lymph nodes outside the resection zone)[262]. An Italian study has recently demonstrated that the duration of symptoms (mainly jaundice and celiac pain) of more than 40 d, CA 19-9 levels above 200 U/mL and G3-G4 histological grade of the tumor are poor prognostic parameters, even if the disease is resectable by pre-operative staging[263].
Preoperative biliary decompression vs immediate surgical resection: Obstructive jaundice is a common presentation for tumors located in the periampullary area or in the head of the pancreas. To reduce perioperative complications and mortality in patients with obstructive jaundice undergoing pancreaticoduodenectomy (PD), preoperative biliary drainage appears to have a positive impact supported by the findings of several observational studies[264-266]. On the other hand, several other non randomized studies failed to show any advantage of preoperative biliary decompression in these patients, as they developed a higher incidence of bacteriobilia and fungal colonization causing more wound infections, postoperative sepsis and longer hospital stay[267-270]. Two meta-analyses of randomized controlled trials and a systematic review of descriptive series have shown that the outcome of patients undergoing biliary decompression prior to PD was inferior to early surgery as they had higher rates of infectious complications and perioperative mortality[271,272]. These findings were confirmed by a recent multicenter randomized controlled study from the Netherlands which showed that the rates of serious complications were 39% for patients who underwent early surgical resection in comparison to 74% in the group that underwent pre-operative biliary decompression (P < 0.001)[264]. Similarly, surgical complications occurred in 37% of patients undergoing early resection in comparison to 47% for individuals who had preoperative biliary decompression. Although the difference did not reach statistical significance (P = 0.14), the overall mortality and hospital stay were comparable between the two groups[273].
During the last decade, there has been an increasing interest in treating patients with neo-adjuvant chemoradiotherapy to improve disease-free and overall survival in patients undergoing surgery. Although there are still no phase III randomized controlled studies to support the use of this strategy, several phase II randomized trials have shown that neo-adjuvant chemo and chemo-radiation therapy are relatively well tolerated, do not reduce the resectability rate and seem to increase the percentage of patients who undergo R0 resections[274-283]. For jaundiced PC patients, candidates for neo-adjuvant therapy must undergo biliary decompression to prevent liver decompensation and stent patency is required for several months. Currently, the only study assessing the outcome of patients undergoing chemo-radiation therapy prior to PD has shown that plastic stents do not provide patency of the biliary system for long enough to complete the preoperative protocols. In fact, 55% of cases required unplanned repeat ERCP with stent exchange for recurrence of jaundice or ascending cholangitis[284]. For these patients, self expanding metallic stents should be used as the direct costs associated with repeating ERCP and hospital admissions for recurrent biliary obstruction and ascending cholangitis appear to be superior to the initial higher cost of using metallic stents[285].
Standard vs pylorus preserving PD: Walter Kausch first described PD in 1912[286], and Allan Whipple later popularized the procedure that bears his name[287]. The classic Whipple (CW) operation consists of an en-bloc removal of the pancreatic head, the duodenum, the common bile duct, the gall bladder and the distal portion of the stomach together with the adjacent lymph nodes[288]. This operation can lead to specific long-term complications such as early and late dumping syndrome, post-operative weight loss[289] and post-operative acid and bile reflux[290].
Pylorus preserving PD (PPPD) was first introduced by Watson in 1942[291], and the procedure was popularized by Traverso and Longmire in 1978[292]. Although it was originally described for the treatment of periampullary tumors, many surgeons nowadays perform PPPD for PC in the head of the pancreas. In order to retain a functioning pylorus, the stomach and the first 2 cm of the duodenum are preserved along with their neurovascular supply. The rationale behind preservation of the stomach is to improve long-term gastrointestinal function[293]. There is still some controversy as to which is the best surgical treatment for PC of the head of the pancreas. In comparison to CW, PPPD has the advantages of reduced operative time[294], less blood loss, better access to the biliary anastomosis for post-operative endoscopy in patients with recurrent biliary obstruction, improvement of post-operative weight gain and quality of life[295]. On the other hand, some series have reported that PPPD has a higher incidence of delayed gastric emptying[296,297]. Moreover, it has not been unequivocally shown that PPPD is oncologically equivalent to CW[298]. A number of RCTs and meta-analyses have demonstrated that both perioperative morbidity and long-term outcome are equal in CW and PPPD[263,299,300].
Pancreatic reconstruction: The most significant cause of morbidity and mortality after PD is the development of complications caused by leakage of pancreatic secretions and pancreatic fistulae observed in up to 20% in specialized centers[301,302]. The meticulous reconstruction of pancreatico-enteric continuity is the key to preventing pancreatic fistulae[303]. Pancreatico-jejunostomy and pancreatico-gastrostomy (PG) are the most commonly employed techniques for pancreaticoenteric reconstruction. PG was believed to be an easier technique and less prone to ischemia as a result of the close proximity between the stomach and the pancreatic stump and the presence of a better vascular supply in the stomach in comparison to the jejunum. However, RCTs have not demonstrated superiority of one technique over the other in terms of post-operative complication rates or incidence of pancreatic fistulae[304,305].
Distal pancreatectomy: Distal pancreatectomy is the surgical procedure of choice for PC of the body and tail of the pancreas. It entails resection of the portion of the pancreas extending to the left of the superior mesenteric vessels and not including the duodenum and the distal bile duct[306]. The spleen is conventionally removed in an en-bloc fashion[307]. However, splenic preservation could be accomplished without an increased rate of complications, operative time or the duration of post-operative hospital stay[295,308]. Several closure techniques have been introduced for the pancreatic remnant in an attempt to reduce pancreatic fistulae. They include hand-sewn suture techniques, staple closure techniques or a combination of both[309-312], ultrasonic dissection devices[313], pancreatico-enteric anastomosis[314], application of meshes, seromuscular[315] and gastric serosal patches[316], or sealing the pancreatic stump with fibrin glue[199].
Cancers of the body and tail of pancreas usually present at a later stage of the disease in comparison to PC of the head due to lack of early symptoms[317]. There are no survival differences between resections for equal TNM stage tumors of the head vs tumors of the body and tail as shown by a retrospective study that reported a 5-year survival of 17% after resection of the pancreatic head vs 15% for left-sided tumors in stage I cancers[318].
Laparoscopic pancreatic resection: Laparoscopic pancreatic surgery represents one of the most challenging abdominal operations[319,320]. Gagner and Pomp were the first to describe a laparoscopic duodeno-pancreatectomy in 1994[321]. Since then, the total number of laparoscopic duodeno-pancreatectomies has remained small due to technical difficulties associated with this operation[322]. A recent study from the Mayo clinic with 65 patients who underwent total laparoscopic PD (TLPD) outlined that TLPD is safe, feasible and its results appear to be comparable to the open approach[323] (Table 10).
Nevertheless, larger prospective studies are required in order to better assess the advantages of TLPD.
Laparoscopic distal pancreatic resection is currently the most frequently performed laparoscopic pancreatic surgery[327]. Most of the studies on distal laparoscopic pancreatectomy are case series with a relatively small number of patients[328]. Although recent studies have shown that laparoscopic distal pancreatectomy is feasible and safe[329-331], the morbidity, mortality and hospital stay are similar to those after open surgery[332]. This is probably due to the fact that morbidity after pancreatic surgery results from retroperitoneal dissection, length of the operation and pancreatic fistulae rather than the incision. In addition, a recent prospective observational study comparing 85 open vs 27 laparoscopic distal pancreatectomies has shown that the number of lymph nodes removed during the minimally invasive procedure was significantly inferior (mean number: 5.2) in comparison to the open approach (mean number: 9.4)[333]. These findings suggest that at this time there is a lack of evidence to support oncological equipoise between laparoscopic and open resections for PC.
Total pancreatectomy: Total pancreatectomy has been employed in selected patients with chronic pancreatitis[334], multifocal islet cell tumors or diffuse IPMN[335]. Total pancreatectomy for PC was initially proposed to avoid the risk of pancreatico-enteric leaks and to remove potential undetectable synchronous disease in other parts of the gland[336]. However, the indication of total pancreatectomy to avoid the risks of pancreatic fistulae is still controversial[337]. Improvement in operative techniques, advances in nutritional support, critical care and interventional radiology have significantly decreased the incidence of life-threatening sequels of pancreaticoenteric leaks[338]. In addition, the permanent endocrine insufficiency associated with total pancreatectomy impacts enormously on the quality of life and long-term outcome of these patients[339]. Some studies have demonstrated a significant increased risk of perioperative morbidity and mortality associated with total pancreatectomy compared with PD[318]. A recent study by Reddy et al[335] showed that long-term survival rates were equivalent after total pancreatectomy and PD (19.9% vs 18.5%), supporting the fact that there is no oncological benefit of total pancreatectomy vs a more limited resection in PC. Currently, total pancreatectomy should be performed in patients with PC if it is the only oncologically sound treatment option[335].
Vascular resections and extended lymphadenectomy: With the advancement in operative techniques and peri-operative management of patients with PC, more radical surgical procedures with vascular resection and extended lymphadenectomy have been proposed for selected cases[340]. The results of extended vascular and lymphatic resections remain controversial.
The principal use of venous resection and reconstruction is to allow complete tumor clearance when precluded by tumor involvement of the superior mesenteric or portal vein, and when the surgeon expects to achieve a negative resection margin[341]. Post-operative morbidity and mortality rates following portal or superior mesenteric vein resections seem to be similar to those of patients with standard PD (42%-48.4% vs 47.1%, 3.2%-5.9% vs 2.5%, respectively)[342,343]. Another study showed that patients undergoing pancreatic resection with venous reconstruction (VR) had a median survival of 22 mo compared to 20 mo for those who had classic PD (P = 0.25)[344]. In another study, a slight survival benefit was noted in patients who did not require VR (33.5%) compared to those with VR (20%, P = 0.18), although this did not reach statistical significance[345].
Pancreatectomies with major arterial resections (common hepatic artery/celiac axis and superior mesenteric artery) have been reported in recent years with acceptable outcomes. Nevertheless, arterial reconstruction during pancreatectomies remains a challenging procedure with increased risk of complications compared to classic PD and PD with VR. In addition, most PCs with arterial invasion are for the majority, advanced tumors with distant lymph node involvement and metastases, and therefore indicated only in a very select group of patients[346]. Recent data on pancreatectomies requiring arterial resections at high volume tertiary centers have shown operative mortality rates of 4.3%[346], peri-operative mortality rates (60 d) of 17%[347], morbidity rates of 48%[348] and 3-year survival rates of 17%-23.1%, which are much higher than for classic PD[346,347].
It has been noted that lymph node involvement outside the standard PD specimens occurs in more than 30% of cases[349]. This has led to the evaluation of the need for a more extended lymph node dissection (ELND) in the surgical management of PC. To date, the definitions of a standard lymphadenectomy as well as ELND are still not very clear[341]. A number of Japanese studies have shown an increased survival rate in patients who have undergone ELND compared to conventional PD[350-352]. However, these studies were not randomized and their data were not validated by other centers[353].
The first RCT comparing standard PD and ELND was reported by Pedrazzoli et al[354] in 1998. In this study, standard lymph node dissection was defined as the removal of lymph nodes from the anterior and posterior pancreatoduodenal region, pyloric region, biliary duct, superior and inferior pancreatic head and body. In addition to the above, ELND included removal of lymph nodes from the hepatic hilum and along the aorta from the diaphragmatic hiatus to the inferior mesenteric artery and laterally to both renal hila, with circumferential clearance of the origin of the celiac trunk and SMA. This study showed no difference in morbidity, mortality or 4-year survival rates between the two groups.
Recently, a meta-analysis on standard PD and PD + ELND for PC patients showed comparable morbidity and mortality rates with a trend towards higher rates of delayed gastric emptying in the ELND group. The weighted mean log hazard ratio for overall survival was 0.93 (CI: 0.77-1.13), revealing no significant outcome differences between the standard and extended procedure (P = 0.480) suggesting that ELND does not benefit overall survival and has a trend towards increased morbidity[355].
During the last two decades, several large observational studies in the U.S., Canada and the Netherlands have shown that the institutional volume of pancreatic resections affects patients’ outcomes. Higher perioperative morbidity, mortality and decreased use of multimodality therapy have been observed more frequently in low volume centers[356-363]. In 1993, Edge and colleagues reported that case load did not correlate with mortality after pancreatic resection[364]. However, surgeons who performed fewer than 4 resections per year had more complications. Recent studies have shown significant improvements in perioperative morbidity and mortality in patients undergoing pancreatic resections in high volume centers. For example, investigators at Memorial Sloan Kettering Cancer Center found that in a cohort of 1972 patients, high-volume centers defined as performing more than 40 cases per year in New York State had significantly less mortality (4% vs 12.3%) than low volume centers[356].
The definition of high and low volume varied among all these studies, but the findings were consistent and were confirmed by Birkmeyer et al[365] who showed that very low volume centers (0-1 procedure per year), low volume hospitals (1-2 procedures per year) and higher volume hospitals (more than 5 procedures per year) had significantly different mortality rates (16% and 12% vs 4% respectively; P < 0.001). The largest difference in operative mortality between very low volume (17.6%) and high volume (3.8%) centers is even more significant for PD when compared to other major surgeries as shown in a retrospective analysis of data from the national Medicare claims database and the Nationwide Inpatient Sample[257].
A recent study involving 301 033 patients with PC included in the National Cancer Database evaluated the treatment patterns of 1667 hospitals over a 19-year period[366]. During that time the pancreatectomy rate as well as the use of multimodality adjuvant therapy for patients with stage I and II disease increased significantly (pancreatectomy rate increased from 39.6% to 49.3%; P < 0.001, and the use of multimodality therapy increased from 26.8% to 38.7%; P < 0.001). Furthermore, patients were more likely to receive multimodality therapy at academic institutions, particularly those considered to be high volume hospitals. Despite these important advances, it appears that there is still a high percentage (71.4%) of patients with potentially resectable disease who are still not referred for surgical therapy as reported by Bilimoria et al[367]. These findings would suggest that a persistent nihilism of clinicians towards PC amd pancreatectomy may be the most significant correctable factor that contributes to the current poor long-term outcomes of PC.
Several single agent chemotherapeutic agents have been tried in the treatment of PC. 5-FU has been used in PC for more than 25 years with response rates of 8%-15%[368]. The addition of Leucovorin to 5-FU doubled the response rate to 26%, however, it showed no benefit in terms of survival[369]. The only chemotherapeutic agent that demonstrated prolonged survival in comparison to 5-FU and Leucovorin was Gemcitabine[370].
After pancreatic resection, the 5 year survival rate is only 20% or less as PC has a high loco-regional recurrence rate and a tendency towards early liver metastasis (Table 11)[258,371-376].
| Author | Yr | Resection (n) | R0 resection (n) | Overall 5-yr survival (%) | R0 5-yr survival (%) | Median survival (mo) |
| Fatima et al[371] | 2010 | 617 | 468 | 17.4 | 20 | 18 |
| Kato et al[376] | 2009 | 138 | 115 | 9.9 | 13.2 | 12.3 |
| Raut et al[373] | 2007 | 360 | 300 | NA | NA | 24.9 |
| Cameron et al[258] | 2006 | 1000 | NA | 18 | 23 | 33 |
| Shimada et al[372] | 2006 | 88 | 66 | 19 | 26 | 22 |
| Howard et al[375] | 2006 | 126 | 158 | 4 | 67 | 18 |
| Moon et al[374] | 2003 | 81 | 20 | 10.8 | 67.8 | 11.8 |
Based on these observations it appears necessary to employ adjuvant therapy in combination with surgical resection in order to improve survival. Only a few years ago there was no valid data on adjuvant chemoradiation therapy after curative surgical resection[377].
The first RCT that showed benefit from adjuvant chemoradiation therapy in comparison to surgery alone was the Gastrointestinal Tumor Study Group (GITSG) trial, where patients receiving 40 cGy followed by 5-FU showed a mean survival of 18 mo in comparison to 11 mo for those who received surgery alone (P = 0.05). The two- and five-year survival rates of the two groups were 43% vs 18% and 19% vs 0%, respectively[378].
The EORTC (European Organization for Research and Treatment of Cancer) study showed that patients undergoing chemoradiation therapy (5-FU protocol) had a median survival of 17.1 mo compared to 12.6 mo for the controls (P = 0.099). The two- and five-year overall survival rates were 37% and 20% for the experimental arm and 23% and 10% for the control arm (P = NS)[379].
The European Study Group for PC 1 trial (ESPAC-1) compared four groups of patients who underwent pancreatic resection; (1) surgery alone; (2) 5-FU and Leucovorin adjuvant chemotherapy; (3) combination of adjuvant radiation therapy and 5-FU chemotherapy; and (4) adjuvant chemoradiation followed by chemotherapy[380]. In this study, the five-year survival rate for patients who received adjuvant chemotherapy was 21% compared to 8% for patients who did not (P = 0.009). Patients who underwent chemoradiation therapy had an inferior five-year survival rate (10% vs 20%) in comparison to patients who did not receive radiation (P = 0.05).
In 2006, the Radiation Therapy Oncology Group trial compared patients receiving adjuvant chemoradiation (5040 cGy in combination with continuous 5-FU) followed by 5-FU vs similar chemoradiation therapy followed by Gemcitabine. For patients affected by PC of the head, the arm treated with Gemcitabine had a superior median (18.8 mo vs 16.7 mo) and overall survival at 3 years [31% vs 21% (P = 0.047)], but with a higher incidence of toxicity (80% vs 60%)[381].
In 2007, a RCT conducted in Germany and Austria (CONKO-1 [Charite Onkologie Clinical Studies in GI Cancer 001]) compared patients undergoing R0 or R1 pancreatic resection alone vs resection followed by Gemcitabine-based chemotherapy. The median disease-free survival for patients treated with Gemcitabine was 13.9 mo vs 6.9 mo in the observation arm (P < 0.001), although there was no difference in the overall survival between the two groups (22 mo vs 20 mo)[382]. From the results of these studies, adjuvant chemotherapy has become the standard of care for patients who can tolerate the treatment after surgical resection.
Neoadjuvant therapy is defined as the preoperative intervention aiming to convert unresectable PCs to resectable tumors or to increase the probability of complete microscopic tumor resection[383]. One of the limitations of the role of neoadjuvant therapy for PC is the fact that there is no standardized definition for tumor resectability and there is no data from randomized phase three trials on the benefit of neoadjuvant therapy. In addition, data from prospective and retrospective studies have several biases due to heterogeneity of inclusion and exclusion criteria, preoperative quality of imaging tests, and surgical pathology reports on lymph node involvement and resection margin status.
A recent systematic review[383] evaluating retrospective and prospective studies on neoadjuvant chemo and radiation therapy from 1966 to 2009 included a total of 111 studies and 4,394 patients. The results of this meta-analysis showed that the majority of patients were treated with Gemcitabine, 5-FU or oral analogue Mitomycin-c, and Platinum compounds. Patients undergoing neoadjuvant treatment received radiotherapy in the range of 24-63 Gy. The analysis showed that neoadjuvant treatment in patients with unresectable tumor was able to convert 33.2% of patients to resectable candidates, providing a median survival of 20.5 mo which was equivalent to patients undergoing resection followed by adjuvant therapy who had median survival of 20.1 to 23.6 mo. On the other hand, neoadjuvant therapy for patients with resectable cancer did not seem to improve overall outcome.
Persistent loco-regional disease after pancreatic surgery is a major determinant of recurrence[384]. Although there is supportive evidence for the use of adjuvant chemotherapy[380,385], the role of adjuvant radiation remains unresolved. Generally it is believed that external-beam radiotherapy (EBRT) alone is a suboptimal treatment for locally advanced PC as most patients will die of systemic disease[386].
In the Mayo clinic clinical trial and the GITSG trial, patients who were randomized to receive EBRT only had a median survival of 5.3-6.3 mo which was inferior to EBRT plus 5-FU[387,388].
Among 210 patients who underwent surgical resection for PC [PD (73%), total and/or distal pancreatectomy (25%), Appleby procedure (2%)] followed by intraoperative electron beam radiotherapy (IOERT), some patients received a single fraction of IOERT alone (25 Gy), whereas others (30%) received additional EBRT and 54% received various forms of adjuvant chemotherapy. The study demonstrated excellent local control with the addition of IOERT (75%). Despite the benefit in local control, the overall median survival was similar to other studies with adjuvant chemotherapy or chemoradiation (19 mo)[389]. A combined study of extended resection and intraoperative radiation therapy (IORT) concluded that IORT contributed to local control; however, it provided no overall survival benefits (14.6% 5-year survival)[390].
In the United States, chemoradiation with concurrent 5-FU followed by Gemcitabine continues to represent the standard for adjuvant therapy of tumor of the pancreatic head. A direct comparison of chemo-radiation therapy and chemotherapy alone seems to be difficult to achieve and additive chemotherapy before or after chemo-radiation-therapy will have to be tested in randomized studies in order to determine the optimal sequencing[391].
Palliative treatment of patients with PC plays a very important role as 80% to 90% of newly diagnosed tumors are not resectable due to local invasion or presence of distal metastatic disease[392]. Median survival for patients with unresectable PC located in the head and body of the gland is approximately 7 mo, while for PC located in the tail median survival is significantly less [3 mo (P = 0.0002)], as they are usually diagnosed in more advanced stages[393]. For these patients, relief of symptoms secondary to gastric outlet obstruction, jaundice and pain are essential to improve their quality of life and overall survival. In the past, surgical palliation was more common as the diagnosis of unresectable disease was frequently done in the operating room and patients underwent one or more of the following procedures: gastric bypass, hepatico-enteric decompression and celiac plexus neurolysis for pain relief during the same surgery. With the improvement in diagnostic imaging tests, the role of surgical staging has decreased as the vast majority of patients can be currently classified as suffering from unresectable disease by non-invasive modalities such as CT and MRI or by endoscopic US. Nevertheless, there are still controversies on the best palliative strategies for these patients as there is a lack of randomized controlled trials and abundant contrasting data from observational studies.
There is still some controversy on the use of routine gastro-intestinal bypass for PC diagnosed as unresectable at the time of exploratory laparoscopy or laparotomy.
In a large observational study of 155 patients with unresectable PC staged by extended laparoscopy at the Memorial Sloan Kettering Cancer Center, only 4% of patients required surgical intervention for gastric outlet obstruction before their death: 2 patients required open gastro-jejunal anastomosis alone and 1 patient underwent a combined gastro and hepatico-jejunostomy a few days after laparoscopy[393]. In addition, 1 patient required a percutaneous endoscopic gastrostomy for palliation of gastric outlet obstruction a few weeks before demise. The authors concluded that the routine use of gastric bypass in patients with unresectable PC is not indicated. On the other hand, several other retrospective studies[394,395] have suggested that up to 25% of patients with unresectable PC would develop gastric outlet obstruction requiring surgical intervention.
A recent prospective randomized trial compared 44 patients who were found unresectable at the time of surgery and who underwent a retrocolic gastro-jejunostomy to 43 patients who did not[396]. The two groups had similar morbidity (32% vs 33%), mortality (0%) and hospital stay. On the other hand, patients who had gastric bypass did not develop any gastric outlet obstruction, while 19% of patients in the control group did (P < 0.01). Although this study would suggest that gastric bypass should be performed in all patients found unresectable at the time of surgery, the introduction of metallic self-expanding intestinal stents has changed the options for palliation.
A prospective multicenter cohort study of 51 patients with malignant gastric outlet obstruction treated with self-expandable metallic stents showed that in 98% of cases the stent was successfully deployed and that the median duration of patency was 10 mo. Only 14% of patients had stent dysfunction, and migration was observed in only 2% of cases[397]. Similar results were reported by another study from South Korea which showed a median stent patency of 385 d, and only 1% serious complications (gastrointestinal bleeding or perforation)[398]. Other observational studies have shown that compared with palliative surgery, stent placement provides a shorter hospital stay, earlier resumption of oral intake, fewer complications and lower hospital costs[399,400]. The only randomized controlled study that compared duodenal stent and laparoscopic gastrojejunostomy favored endoscopic therapy as it was associated with less discomfort, shorter hospital stay and improved physical health scores at 1 mo[401]. In this small study, only a third of patients were alive at 1 year and no cases of stent occlusion were observed. The two groups had similar overall survival supporting equipoise between endoscopic and surgical palliation. Nevertheless, surgical palliation can still play an important role when patients have a long life-expectancy, need biliary and gastric bypass in combination with celiac neurolysis for pain control.
The majority of PCs occur in the head of the pancreas and obstructive jaundice is one of the early symptoms for 50%-80% of patients[396]. In the past, staging laparotomy and biliary bypass were frequently performed for unresectable PC of the head[402,403]. During the last decades, the development of interventional radiology and endoscopy has allowed palliation of obstructive jaundice by the insertion of percutaneous or endoluminal stents with minimal morbidity and mortality. Currently, endoscopic biliary stenting is the treatment of choice for unresectable PC with obstructive jaundice. Percutaneous transhepatic stenting is reserved only for patients in whom endoscopic stenting has failed as it is associated with a higher complication rate than endoscopic palliation (61% vs 35%)[404,405]. High risk surgical patients are best managed by biliary stenting, however, it is still unclear whether palliative surgical biliary decompression is superior to other interventions for patients who are fit for surgery or who have a longer life expectancy. A European randomized controlled study comparing surgical biliary decompression vs endoscopic plastic stenting showed that both interventions were equally successful in palliating jaundice (95% vs 94%, respectively) and provided equal overall survival. Nevertheless, major complications (29% vs 11%) and procedure-related mortality (14% vs 3%) were significantly higher for surgical patients[406]. In addition, surgical decompression was more expensive than stenting, although recurrent biliary obstructions and late gastric bypasses were more common in patients undergoing endoscopic treatment even if that did not reach statistical significance. Similar results were reported in a more recent Brazilian study which found that endoscopic therapy with self-expandable metallic stents was more cost-effective than surgical decompression (US$2832 vs US$3821, P = 0.031) and provided better quality of life at 30 (P = 0.04) and 60 d (P = 0.05)[407]. The only available meta-analysis of randomized controlled studies comparing surgery with endoscopic stenting included only 3 studies where none tested the use of metallic self-expanding stents[408]. Although the reintervention rate was 3% (0%-16%) in surgically treated patients compared with 36% (28%-43%) in stented patients, because of the limited number of studies with a relatively small group of patients and heterogeneous quality, the authors concluded that they could not identify which treatment was preferable.
The patency of biliary stents has greatly improved with the introduction of expandable metallic stents (EMS) as they offer a larger diameter for drainage and are associated with a lower occlusion rate than plastic stents[409,410]. The concurrent use of chemotherapeutic agents in patients palliated with SEMS was thought to increase the risk for ascending cholangitis. However, a Japanese retrospective study has demonstrated that the combination of SEMS and palliative chemotherapy for unresectable PC did not change the incidence of biliary infectious complications[411]. In patients with combined biliary and duodenal obstructions, concomitant biliary and duodenal stenting is now feasible and justified as the need to repeat endoscopic therapies is rarely required even in long-term survival patients[412].
Currently, surgical biliary bypass is advocated only for patients with obstructive jaundice who fail endoscopic or percutaneous stent placement.
About 70% of patients with unresectable PC develop clinically important pain during their lives[413]. Pain is the main cause of the significant drop in quality and quantity of life of these patients and good palliation is necessary as pain incidence and severity increases with disease progression[414].
For the majority of patients, pain from PC can be managed with opioid analgesics. However, approximately one third of patients experience inadequate control of pain with oral analgesics alone[415]. For these patients, radiation therapy, chemotherapy and celiac plexus neurolysis have been used. Percutaneous neurolytic celiac plexus block with injection of 50%-100% ethyl alcohol under radiological guidance has become the most commonly recognized method of splanchnicectomy with a 70%-96% success rate[416]. The celiac plexus block has several advantages as it has been proven to ease pain without the side effects of opioids and can be administered intraoperatively, percutaneously, or by endoscopic ultrasonography. Recent studies have shown that endoscopic ultrasonography-guided neurolysis is effective and has minimal risk of the potentially serious complications associated with surgical or percutaneous approaches[417,418].
A recent double-blind randomized controlled study comparing patients treated with celiac plexus block vs systemic analgesic therapy showed that splanchnic neurolysis provided superior pain relief and quality of life scores, but overall opioid consumption, frequency of opioid adverse effects and overall survival did not reach statistical significance between the two groups[419]. For the majority of PC patients, pain is still controlled pharmacologically even if other modalities such as surgical thoracoscopic splanchnicectomy, epidural anesthesia, subcutaneous injection with octreotide, hypofractionated-accelerated radiotherapy and more recently photodynamic therapy have shown some temporary success[414,420-423].
The median survival of patients with unresectable PC is 33 wk and for advanced metastatic disease is only 10 wk[424]. About 90% of patients with PC have significant weight loss at the time of diagnosis and all of them develop progressive cachexia due to neoplastic metabolic derangements. Secondary events such as pancreatic exocrine insufficiency due to pancreatic duct obstruction, fat malabsorption due to biliary obstruction and poor oral caloric intake caused by nausea or gastric outlet obstruction are also responsible for the progressive weight loss. Even if weight loss has been found to have a prognostic effect on survival, most of the palliative care interventions for PC are directed at correcting biliary obstruction, gastric outlet obstruction and pain, and relatively little attention has been paid to interventions that can prevent or reduce the progressive weight loss of these patients[425]. Recently, a placebo-controlled trial comparing patients receiving enteric coated pancreatic enzyme supplements vs placebo showed that after 2 mo, patients receiving pancreatin had gained 1.2% of their body weight in comparison to controls who lost 3.7% (P = 0.02), and that they had higher daily total energy intake (8.4 MJ vs 6.6 MJ, P = 0.04)[424]. Although the Karnofsky performance status between the two groups was not different and survival analysis was not performed to determine if body weight gain translates into better prognosis, this study was the first to show an effective palliative strategy able to increase the intestinal absorptive function of patients who suffer from steatorrhea.
In recent decades, diagnostic modalities, and the surgical and palliative treatments of PC have clearly progressed although the overall prognosis has barely changed. The management of patients affected by PC is complex and requires expertise in many fields. Multidisciplinary teams are necessary to optimize the overall care, and palliative techniques have to be mastered as the majority of PCs are diagnosed in advanced stages. Better outcomes are reached if PC patients are appropriately referred to tertiary centers for assessment by surgical, medical and radiation oncologists, gastroenterologists, palliative care specialists and other dedicated health care providers. Despite recent progress, there is still a very limited ability to detect PC at an early stage, and there is a need for more studies to better understand genetic predisposing factors and to discover new markers that could assist physicians in this task. Randomized controlled studies are necessary to explore the role of neo-adjuvant therapies and new protocols for adjuvant strategies in patients undergoing pancreatic resection.
Peer reviewer: Hiroyuki Uehara, MD, PhD, Chief, Division of Pancreatology, Department of Gastroenterology, Osaka Medical Center for Cancer and Cardiovascular Diseases, 1-3-3 Nakamichi, Higashinari, Osaka 537-8511, Japan
S- Editor Tian L L- Editor Webster JR E- Editor Lin YP
| 1. | Cubilla AL, Fitzgerald PJ. Morphological lesions associated with human primary invasive nonendocrine pancreas cancer. Cancer Res. 1976;36:2690-2698. |
| 2. | Sener SF, Fremgen A, Menck HR, Winchester DP. Pancreatic cancer: a report of treatment and survival trends for 100,313 patients diagnosed from 1985-1995, using the National Cancer Database. J Am Coll Surg. 1999;189:1-7. |
| 3. | Porta M, Fabregat X, Malats N, Guarner L, Carrato A, de Miguel A, Ruiz L, Jariod M, Costafreda S, Coll S. Exocrine pancreatic cancer: symptoms at presentation and their relation to tumour site and stage. Clin Transl Oncol. 2005;7:189-197. |
| 4. | Kullavanijaya P, Treeprasertsuk S, Thong-Ngam D, Kladcharoen N, Mahachai V, Suwanagool P. Adenocarcinoma of the pancreas: the clinical experience of 45 histopathologically proven patients, a 6 year study. J Med Assoc Thai. 2001;84:640-647. |
| 5. | Elli M, Piazza E, Franzone PC, Isabella L, Poliziani D, Taschieri AM. Considerations on early diagnosis of carcinoma of the pancreas. Hepatogastroenterology. 2003;50:2205-2207. |
| 6. | Cook DJ, Mulrow CD, Haynes RB. Systematic reviews: synthesis of best evidence for clinical decisions. Ann Intern Med. 1997;126:376-380. |
| 7. | Mulrow CD, Oxman AD. Cochrane Collaboration Handbook. Oxford: Update Software 1997; 1. |
| 8. | Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52:377-384. |
| 9. | Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225-249. |
| 10. | Shaib YH, Davila JA, El-Serag HB. The epidemiology of pancreatic cancer in the United States: changes below the surface. Aliment Pharmacol Ther. 2006;24:87-94. |
| 11. | Ghadirian P, Lynch HT, Krewski D. Epidemiology of pancreatic cancer: an overview. Cancer Detect Prev. 2003;27:87-93. |
| 12. | Lowenfels AB, Maisonneuve P. Epidemiology and risk factors for pancreatic cancer. Best Pract Res Clin Gastroenterol. 2006;20:197-209. |
| 13. | Neugut AI, Ahsan H, Robinson E. Pancreas cancer as a second primary malignancy. A population-based study. Cancer. 1995;76:589-592. |
| 14. | Lynch SM, Vrieling A, Lubin JH, Kraft P, Mendelsohn JB, Hartge P, Canzian F, Steplowski E, Arslan AA, Gross M. Cigarette smoking and pancreatic cancer: a pooled analysis from the pancreatic cancer cohort consortium. Am J Epidemiol. 2009;170:403-413. |
| 15. | Brand RE, Greer JB, Zolotarevsky E, Brand R, Du H, Simeone D, Zisman A, Gorchow A, Lee SY, Roy HK. Pancreatic cancer patients who smoke and drink are diagnosed at younger ages. Clin Gastroenterol Hepatol. 2009;7:1007-1012. |
| 16. | Iodice S, Gandini S, Maisonneuve P, Lowenfels AB. Tobacco and the risk of pancreatic cancer: a review and meta-analysis. Langenbecks Arch Surg. 2008;393:535-545. |
| 17. | Hassan MM, Bondy ML, Wolff RA, Abbruzzese JL, Vauthey JN, Pisters PW, Evans DB, Khan R, Chou TH, Lenzi R. Risk factors for pancreatic cancer: case-control study. Am J Gastroenterol. 2007;102:2696-2707. |
| 18. | Permert J, Ihse I, Jorfeldt L, von Schenck H, Arnqvist HJ, Larsson J. Pancreatic cancer is associated with impaired glucose metabolism. Eur J Surg. 1993;159:101-107. |
| 19. | Gullo L, Pezzilli R, Morselli-Labate AM. Diabetes and the risk of pancreatic cancer. N Engl J Med. 1994;331:81-84. |
| 20. | Everhart J, Wright D. Diabetes mellitus as a risk factor for pancreatic cancer. A meta-analysis. JAMA. 1995;273:1605-1609. |
| 21. | Wideroff L, Gridley G, Mellemkjaer L, Chow WH, Linet M, Keehn S, Borch-Johnsen K, Olsen JH. Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J Natl Cancer Inst. 1997;89:1360-1365. |
| 22. | Gapstur SM, Gann PH, Lowe W, Liu K, Colangelo L, Dyer A. Abnormal glucose metabolism and pancreatic cancer mortality. JAMA. 2000;283:2552-2558. |
| 23. | Chari ST, Leibson CL, Rabe KG, Ransom J, de Andrade M, Petersen GM. Probability of pancreatic cancer following diabetes: a population-based study. Gastroenterology. 2005;129:504-511. |
| 24. | Ogawa Y, Tanaka M, Inoue K, Yamaguchi K, Chijiiwa K, Mizumoto K, Tsutsu N, Nakamura Y. A prospective pancreatographic study of the prevalence of pancreatic carcinoma in patients with diabetes mellitus. Cancer. 2002;94:2344-2349. |
| 25. | Huxley R, Ansary-Moghaddam A, Berrington de González A, Barzi F, Woodward M. Type-II diabetes and pancreatic cancer: a meta-analysis of 36 studies. Br J Cancer. 2005;92:2076-2083. |
| 26. | Yalniz M, Pour PM. Diabetes mellitus: a risk factor for pancreatic cancer? Langenbecks Arch Surg. 2005;390:66-72. |
| 27. | Silverman DT, Schiffman M, Everhart J, Goldstein A, Lillemoe KD, Swanson GM, Schwartz AG, Brown LM, Greenberg RS, Schoenberg JB. Diabetes mellitus, other medical conditions and familial history of cancer as risk factors for pancreatic cancer. Br J Cancer. 1999;80:1830-1837. |
| 28. | Perrin MC, Terry MB, Kleinhaus K, Deutsch L, Yanetz R, Tiram E, Calderon R, Friedlander Y, Paltiel O, Harlap S. Gestational diabetes as a risk factor for pancreatic cancer: a prospective cohort study. BMC Med. 2007;5:25. |
| 29. | Fisher WE, Boros LG, Schirmer WJ. Insulin promotes pancreatic cancer: evidence for endocrine influence on exocrine pancreatic tumors. J Surg Res. 1996;63:310-313. |
| 30. | Lowenfels AB, Maisonneuve P, Cavallini G, Ammann RW, Lankisch PG, Andersen JR, Dimagno EP, Andrén-Sandberg A, Domellöf L. Pancreatitis and the risk of pancreatic cancer. International Pancreatitis Study Group. N Engl J Med. 1993;328:1433-1437. |
| 31. | Jiao L, Silverman DT, Schairer C, Thiébaut AC, Hollenbeck AR, Leitzmann MF, Schatzkin A, Stolzenberg-Solomon RZ. Alcohol use and risk of pancreatic cancer: the NIH-AARP Diet and Health Study. Am J Epidemiol. 2009;169:1043-1051. |
| 32. | Genkinger JM, Spiegelman D, Anderson KE, Bergkvist L, Bernstein L, van den Brandt PA, English DR, Freudenheim JL, Fuchs CS, Giles GG. Alcohol intake and pancreatic cancer risk: a pooled analysis of fourteen cohort studies. Cancer Epidemiol Biomarkers Prev. 2009;18:765-776. |
| 33. | Rohrmann S, Linseisen J, Vrieling A, Boffetta P, Stolzenberg-Solomon RZ, Lowenfels AB, Jensen MK, Overvad K, Olsen A, Tjonneland A. Ethanol intake and the risk of pancreatic cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC). Cancer Causes Control. 2009;20:785-794. |
| 34. | Fernandez E, La Vecchia C, Porta M, Negri E, d'Avanzo B, Boyle P. Pancreatitis and the risk of pancreatic cancer. Pancreas. 1995;11:185-189. |
| 35. | Silverman DT, Brown LM, Hoover RN, Schiffman M, Lillemoe KD, Schoenberg JB, Swanson GM, Hayes RB, Greenberg RS, Benichou J. Alcohol and pancreatic cancer in blacks and whites in the United States. Cancer Res. 1995;55:4899-4905. |
| 36. | Bansal P, Sonnenberg A. Pancreatitis is a risk factor for pancreatic cancer. Gastroenterology. 1995;109:247-251. |
| 37. | Karlson BM, Ekbom A, Josefsson S, McLaughlin JK, Fraumeni JF Jr, Nyrén O. The risk of pancreatic cancer following pancreatitis: an association due to confounding? Gastroenterology. 1997;113:587-592. |
| 38. | Farrow B, Evers BM. Inflammation and the development of pancreatic cancer. Surg Oncol. 2002;10:153-169. |
| 39. | Whitcomb DC, Applebaum S, Martin SP. Hereditary pancreatitis and pancreatic carcinoma. Ann N Y Acad Sci. 1999;880:201-209. |
| 40. | Lowenfels AB, Maisonneuve P, DiMagno EP, Elitsur Y, Gates LK Jr, Perrault J, Whitcomb DC. Hereditary pancreatitis and the risk of pancreatic cancer. International Hereditary Pancreatitis Study Group. J Natl Cancer Inst. 1997;89:442-446. |
| 41. | Hruban RH, Petersen GM, Goggins M, Tersmette AC, Offerhaus GJ, Falatko F, Yeo CJ, Kern SE. Familial pancreatic cancer. Ann Oncol. 1999;10 Suppl 4:69-73. |
| 42. | O'Brien JM. Environmental and heritable factors in the causation of cancer: analyses of cohorts of twins from Sweden, Denmark, and Finland, by P. Lichtenstein, N.V. Holm, P.K. Verkasalo, A. Iliadou, J. Kaprio, M. Koskenvuo, E. Pukkala, A. Skytthe, and K. Hemminki. N Engl J Med 343:78-84, 2000. Surv Ophthalmol. 2000;45:167-168. |
| 43. | Lilley M, Gilchrist D. The hereditary spectrum of pancreatic cancer: the Edmonton experience. Can J Gastroenterol. 2004;18:17-21. |
| 44. | Klein AP, Brune KA, Petersen GM, Goggins M, Tersmette AC, Offerhaus GJ, Griffin C, Cameron JL, Yeo CJ, Kern S. Prospective risk of pancreatic cancer in familial pancreatic cancer kindreds. Cancer Res. 2004;64:2634-2638. |
| 45. | Brentnall TA, Bronner MP, Byrd DR, Haggitt RC, Kimmey MB. Early diagnosis and treatment of pancreatic dysplasia in patients with a family history of pancreatic cancer. Ann Intern Med. 1999;131:247-255. |
| 46. | Meckler KA, Brentnall TA, Haggitt RC, Crispin D, Byrd DR, Kimmey MB, Bronner MP. Familial fibrocystic pancreatic atrophy with endocrine cell hyperplasia and pancreatic carcinoma. Am J Surg Pathol. 2001;25:1047-1053. |
| 47. | Zogopoulos G, Rothenmund H, Eppel A, Ash C, Akbari MR, Hedley D, Narod SA, Gallinger S. The P239S palladin variant does not account for a significant fraction of hereditary or early onset pancreas cancer. Hum Genet. 2007;121:635-637. |
| 48. | Lynch HT, Fitzsimmons ML, Smyrk TC, Lanspa SJ, Watson P, McClellan J, Lynch JF. Familial pancreatic cancer: clinicopathologic study of 18 nuclear families. Am J Gastroenterol. 1990;85:54-60. |
| 49. | Goldstein AM, Fraser MC, Struewing JP, Hussussian CJ, Ranade K, Zametkin DP, Fontaine LS, Organic SM, Dracopoli NC, Clark WH Jr. Increased risk of pancreatic cancer in melanoma-prone kindreds with p16INK4 mutations. N Engl J Med. 1995;333:970-974. |
| 50. | Goggins M, Schutte M, Lu J, Moskaluk CA, Weinstein CL, Petersen GM, Yeo CJ, Jackson CE, Lynch HT, Hruban RH. Germline BRCA2 gene mutations in patients with apparently sporadic pancreatic carcinomas. Cancer Res. 1996;56:5360-5364. |
| 51. | Hahn SA, Greenhalf B, Ellis I, Sina-Frey M, Rieder H, Korte B, Gerdes B, Kress R, Ziegler A, Raeburn JA. BRCA2 germline mutations in familial pancreatic carcinoma. J Natl Cancer Inst. 2003;95:214-221. |
| 52. | Murphy KM, Brune KA, Griffin C, Sollenberger JE, Petersen GM, Bansal R, Hruban RH, Kern SE. Evaluation of candidate genes MAP2K4, MADH4, ACVR1B, and BRCA2 in familial pancreatic cancer: deleterious BRCA2 mutations in 17%. Cancer Res. 2002;62:3789-3793. |
| 53. | Su GH, Hruban RH, Bansal RK, Bova GS, Tang DJ, Shekher MC, Westerman AM, Entius MM, Goggins M, Yeo CJ. Germline and somatic mutations of the STK11/LKB1 Peutz-Jeghers gene in pancreatic and biliary cancers. Am J Pathol. 1999;154:1835-1840. |
| 54. | Permuth-Wey J, Egan KM. Family history is a significant risk factor for pancreatic cancer: results from a systematic review and meta-analysis. Fam Cancer. 2009;8:109-117. |
| 55. | Shi C, Hruban RH, Klein AP. Familial pancreatic cancer. Arch Pathol Lab Med. 2009;133:365-374. |
| 56. | Ghadirian P, Liu G, Gallinger S, Schmocker B, Paradis AJ, Lal G, Brunet JS, Foulkes WD, Narod SA. Risk of pancreatic cancer among individuals with a family history of cancer of the pancreas. Int J Cancer. 2002;97:807-810. |
| 57. | Bartsch DK, Kress R, Sina-Frey M, Grützmann R, Gerdes B, Pilarsky C, Heise JW, Schulte KM, Colombo-Benkmann M, Schleicher C. Prevalence of familial pancreatic cancer in Germany. Int J Cancer. 2004;110:902-906. |
| 59. | Landi S. Genetic predisposition and environmental risk factors to pancreatic cancer: A review of the literature. Mutat Res. 2009;681:299-307. |
| 60. | van der Heijden MS, Yeo CJ, Hruban RH, Kern SE. Fanconi anemia gene mutations in young-onset pancreatic cancer. Cancer Res. 2003;63:2585-2588. |
| 61. | Rieder H, Sina-Frey M, Ziegler A, Hahn SA, Przypadlo E, Kress R, Gerdes B, Colombo Benkmann M, Eberl T, Grützmann R. German national case collection of familial pancreatic cancer - clinical-genetic analysis of the first 21 families. Onkologie. 2002;25:262-266. |
| 62. | Hruban RH, Canto MI, Yeo CJ. Prevention of pancreatic cancer and strategies for management of familial pancreatic cancer. Dig Dis. 2001;19:76-84. |
| 63. | Petersen GM, de Andrade M, Goggins M, Hruban RH, Bondy M, Korczak JF, Gallinger S, Lynch HT, Syngal S, Rabe KG. Pancreatic cancer genetic epidemiology consortium. Cancer Epidemiol Biomarkers Prev. 2006;15:704-710. |
| 64. | Wang W, Chen S, Brune KA, Hruban RH, Parmigiani G, Klein AP. PancPRO: risk assessment for individuals with a family history of pancreatic cancer. J Clin Oncol. 2007;25:1417-1422. |
| 65. | Li D, Morris JS, Liu J, Hassan MM, Day RS, Bondy ML, Abbruzzese JL. Body mass index and risk, age of onset, and survival in patients with pancreatic cancer. JAMA. 2009;301:2553-2562. |
| 66. | Patel AV, Rodriguez C, Bernstein L, Chao A, Thun MJ, Calle EE. Obesity, recreational physical activity, and risk of pancreatic cancer in a large U.S. Cohort. Cancer Epidemiol Biomarkers Prev. 2005;14:459-466. |
| 67. | Larsson SC, Permert J, Håkansson N, Näslund I, Bergkvist L, Wolk A. Overall obesity, abdominal adiposity, diabetes and cigarette smoking in relation to the risk of pancreatic cancer in two Swedish population-based cohorts. Br J Cancer. 2005;93:1310-1315. |
| 68. | Berrington de González A, Spencer EA, Bueno-de-Mesquita HB, Roddam A, Stolzenberg-Solomon R, Halkjaer J, Tjønneland A, Overvad K, Clavel-Chapelon F, Boutron-Ruault MC. Anthropometry, physical activity, and the risk of pancreatic cancer in the European prospective investigation into cancer and nutrition. Cancer Epidemiol Biomarkers Prev. 2006;15:879-885. |
| 69. | Fitzgerald TL, Hickner ZJ, Schmitz M, Kort EJ. Changing incidence of pancreatic neoplasms: a 16-year review of statewide tumor registry. Pancreas. 2008;37:134-138. |
| 70. | Conlon KC, Klimstra DS, Brennan MF. Long-term survival after curative resection for pancreatic ductal adenocarcinoma. Clinicopathologic analysis of 5-year survivors. Ann Surg. 1996;223:273-279. |
| 71. | Winter JM, Cameron JL, Campbell KA, Arnold MA, Chang DC, Coleman J, Hodgin MB, Sauter PK, Hruban RH, Riall TS. 1423 pancreaticoduodenectomies for pancreatic cancer: A single-institution experience. J Gastrointest Surg. 2006;10:1199-1210; discussion 1210-1211. |
| 72. | Poultsides GA, Reddy S, Cameron JL, Hruban RH, Pawlik TM, Ahuja N, Jain A, Edil BH, Iacobuzio-Donahue CA, Schulick RD. Histopathologic basis for the favorable survival after resection of intraductal papillary mucinous neoplasm-associated invasive adenocarcinoma of the pancreas. Ann Surg. 2010;251:470-476. |
| 73. | Mortenson MM, Katz MH, Tamm EP, Bhutani MS, Wang H, Evans DB, Fleming JB. Current diagnosis and management of unusual pancreatic tumors. Am J Surg. 2008;196:100-113. |
| 74. | Papavramidis T, Papavramidis S. Solid pseudopapillary tumors of the pancreas: review of 718 patients reported in English literature. J Am Coll Surg. 2005;200:965-972. |
| 75. | Chadwick B, Willmore-Payne C, Tripp S, Layfield LJ, Hirschowitz S, Holden J. Histologic, immunohistochemical, and molecular classification of 52 IPMNs of the pancreas. Appl Immunohistochem Mol Morphol. 2009;17:31-39. |
| 76. | Shin SH, Han DJ, Park KT, Kim YH, Park JB, Kim SC. Validating a simple scoring system to predict malignancy and invasiveness of intraductal papillary mucinous neoplasms of the pancreas. World J Surg. 2010;34:776-783. |
| 77. | Fan F, Lai EC, Xie F, Yang JM, Xu F, Kan T, Shen RX, Yang XY, Lau Wan Y, Wu MC. Intraductal papillary mucinous neoplasms of the pancreas--predictors of malignancy. Hepatogastroenterology. 2010;57:635-639. |
| 78. | Sohn TA, Yeo CJ, Cameron JL, Hruban RH, Fukushima N, Campbell KA, Lillemoe KD. Intraductal papillary mucinous neoplasms of the pancreas: an updated experience. Ann Surg. 2004;239:788-797; discussion 797-799. |
| 80. | Dhebri AR, Connor S, Campbell F, Ghaneh P, Sutton R, Neoptolemos JP. Diagnosis, treatment and outcome of pancreatoblastoma. Pancreatology. 2004;4:441-451; discussion 452-453. |
| 81. | Chen J, Baithun SI. Morphological study of 391 cases of exocrine pancreatic tumours with special reference to the classification of exocrine pancreatic carcinoma. J Pathol. 1985;146:17-29. |
| 82. | Paal E, Thompson LD, Frommelt RA, Przygodzki RM, Heffess CS. A clinicopathologic and immunohistochemical study of 35 anaplastic carcinomas of the pancreas with a review of the literature. Ann Diagn Pathol. 2001;5:129-140. |
| 83. | Connolly MM, Dawson PJ, Michelassi F, Moossa AR, Lowenstein F. Survival in 1001 patients with carcinoma of the pancreas. Ann Surg. 1987;206:366-373. |
| 84. | Wilentz RE, Goggins M, Redston M, Marcus VA, Adsay NV, Sohn TA, Kadkol SS, Yeo CJ, Choti M, Zahurak M. Genetic, immunohistochemical, and clinical features of medullary carcinoma of the pancreas: A newly described and characterized entity. Am J Pathol. 2000;156:1641-1651. |
| 85. | Ridder GJ, Maschek H, Klempnauer J. Favourable prognosis of cystadeno- over adenocarcinoma of the pancreas after curative resection. Eur J Surg Oncol. 1996;22:232-236. |
| 86. | Madura JA, Jarman BT, Doherty MG, Yum MN, Howard TJ. Adenosquamous carcinoma of the pancreas. Arch Surg. 1999;134:599-603. |
| 87. | Mulkeen AL, Yoo PS, Cha C. Less common neoplasms of the pancreas. World J Gastroenterol. 2006;12:3180-3185. |
| 88. | Holen KD, Klimstra DS, Hummer A, Gonen M, Conlon K, Brennan M, Saltz LB. Clinical characteristics and outcomes from an institutional series of acinar cell carcinoma of the pancreas and related tumors. J Clin Oncol. 2002;20:4673-4678. |
| 89. | Shi C, Daniels JA, Hruban RH. Molecular characterization of pancreatic neoplasms. Adv Anat Pathol. 2008;15:185-195. |
| 90. | Liu F. SMAD4/DPC4 and pancreatic cancer survival. Commentary re: M. Tascilar et al., The SMAD4 protein and prognosis of pancreatic ductal adenocarcinoma. Clin. Cancer Res., 7: 4115-4121, 2001. Clin Cancer Res. 2001;7:3853-3856. |
| 91. | Hurban RH, Pitman MB, Klimstra DS. Tumors of the pancreas. Atlas of tumor pathology. Washington DC: American Registry of Pathology and Armed forces Institute of Patholgy 2007; . |
| 92. | Klimstra DS, Wenig BM, Heffess CS. Solid-pseudopapillary tumor of the pancreas: a typically cystic carcinoma of low malignant potential. Semin Diagn Pathol. 2000;17:66-80. |
| 93. | Abraham SC, Klimstra DS, Wilentz RE, Yeo CJ, Conlon K, Brennan M, Cameron JL, Wu TT, Hruban RH. Solid-pseudopapillary tumors of the pancreas are genetically distinct from pancreatic ductal adenocarcinomas and almost always harbor beta-catenin mutations. Am J Pathol. 2002;160:1361-1369. |
| 94. | Tanaka Y, Kato K, Notohara K, Hojo H, Ijiri R, Miyake T, Nagahara N, Sasaki F, Kitagawa N, Nakatani Y. Frequent beta-catenin mutation and cytoplasmic/nuclear accumulation in pancreatic solid-pseudopapillary neoplasm. Cancer Res. 2001;61:8401-8404. |
| 95. | El-Bahrawy MA, Rowan A, Horncastle D, Tomlinson I, Theis BA, Russell RC, Stamp G. E-cadherin/catenin complex status in solid pseudopapillary tumor of the pancreas. Am J Surg Pathol. 2008;32:1-7. |
| 96. | Laffan TA, Horton KM, Klein AP, Berlanstein B, Siegelman SS, Kawamoto S, Johnson PT, Fishman EK, Hruban RH. Prevalence of unsuspected pancreatic cysts on MDCT. AJR Am J Roentgenol. 2008;191:802-807. |
| 97. | Tanaka M, Chari S, Adsay V, Fernandez-del Castillo C, Falconi M, Shimizu M, Yamaguchi K, Yamao K, Matsuno S. International consensus guidelines for management of intraductal papillary mucinous neoplasms and mucinous cystic neoplasms of the pancreas. Pancreatology. 2006;6:17-32. |
| 98. | Schönleben F, Qiu W, Ciau NT, Ho DJ, Li X, Allendorf JD, Remotti HE, Su GH. PIK3CA mutations in intraductal papillary mucinous neoplasm/carcinoma of the pancreas. Clin Cancer Res. 2006;12:3851-3855. |
| 99. | Sato N, Rosty C, Jansen M, Fukushima N, Ueki T, Yeo CJ, Cameron JL, Iacobuzio-Donahue CA, Hruban RH, Goggins M. STK11/LKB1 Peutz-Jeghers gene inactivation in intraductal papillary-mucinous neoplasms of the pancreas. Am J Pathol. 2001;159:2017-2022. |
| 100. | Iacobuzio-Donahue CA, Klimstra DS, Adsay NV, Wilentz RE, Argani P, Sohn TA, Yeo CJ, Cameron JL, Kern SE, Hruban RH. Dpc-4 protein is expressed in virtually all human intraductal papillary mucinous neoplasms of the pancreas: comparison with conventional ductal adenocarcinomas. Am J Pathol. 2000;157:755-761. |
| 101. | Hruban RH, Adsay NV, Albores-Saavedra J, Compton C, Garrett ES, Goodman SN, Kern SE, Klimstra DS, Klöppel G, Longnecker DS. Pancreatic intraepithelial neoplasia: a new nomenclature and classification system for pancreatic duct lesions. Am J Surg Pathol. 2001;25:579-586. |
| 102. | Wilentz RE, Iacobuzio-Donahue CA, Argani P, McCarthy DM, Parsons JL, Yeo CJ, Kern SE, Hruban RH. Loss of expression of Dpc4 in pancreatic intraepithelial neoplasia: evidence that DPC4 inactivation occurs late in neoplastic progression. Cancer Res. 2000;60:2002-2006. |
| 103. | Maitra A, Hruban RH. Pancreatic cancer. Annu Rev Pathol. 2008;3:157-188. |
| 104. | Canto MI, Goggins M, Hruban RH, Petersen GM, Giardiello FM, Yeo C, Fishman EK, Brune K, Axilbund J, Griffin C. Screening for early pancreatic neoplasia in high-risk individuals: a prospective controlled study. Clin Gastroenterol Hepatol. 2006;4:766-781; quiz 665. |
| 105. | Klimstra DS, Longnecker DS. Acinar cell carcinoma. World Health Organization Classifiaction of tumors. Lyon: IARC Press 2000; 241-243. |
| 106. | Kerr NJ, Chun YH, Yun K, Heathcott RW, Reeve AE, Sullivan MJ. Pancreatoblastoma is associated with chromosome 11p loss of heterozygosity and IGF2 overexpression. Med Pediatr Oncol. 2002;39:52-54. |
| 107. | Abraham SC, Wu TT, Klimstra DS, Finn LS, Lee JH, Yeo CJ, Cameron JL, Hruban RH. Distinctive molecular genetic alterations in sporadic and familial adenomatous polyposis-associated pancreatoblastomas : frequent alterations in the APC/beta-catenin pathway and chromosome 11p. Am J Pathol. 2001;159:1619-1627. |
| 108. | Hoorens A, Prenzel K, Lemoine NR, Klöppel G. Undifferentiated carcinoma of the pancreas: analysis of intermediate filament profile and Ki-ras mutations provides evidence of a ductal origin. J Pathol. 1998;185:53-60. |
| 109. | Goggins M, Offerhaus GJ, Hilgers W, Griffin CA, Shekher M, Tang D, Sohn TA, Yeo CJ, Kern SE, Hruban RH. Pancreatic adenocarcinomas with DNA replication errors (RER+) are associated with wild-type K-ras and characteristic histopathology. Poor differentiation, a syncytial growth pattern, and pushing borders suggest RER+. Am J Pathol. 1998;152:1501-1507. |
| 110. | Yamamoto H, Itoh F, Nakamura H, Fukushima H, Sasaki S, Perucho M, Imai K. Genetic and clinical features of human pancreatic ductal adenocarcinomas with widespread microsatellite instability. Cancer Res. 2001;61:3139-3144. |
| 111. | Calhoun ES, Jones JB, Ashfaq R, Adsay V, Baker SJ, Valentine V, Hempen PM, Hilgers W, Yeo CJ, Hruban RH. BRAF and FBXW7 (CDC4, FBW7, AGO, SEL10) mutations in distinct subsets of pancreatic cancer: potential therapeutic targets. Am J Pathol. 2003;163:1255-1260. |
| 112. | Ribic CM, Sargent DJ, Moore MJ, Thibodeau SN, French AJ, Goldberg RM, Hamilton SR, Laurent-Puig P, Gryfe R, Shepherd LE. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med. 2003;349:247-257. |
| 113. | Becker WF, Welsh RA, Pratt HS. Cystadenoma and cystadenocarcinoma of the pancreas. Ann Surg. 1965;161:845-863. |
| 114. | King JC, Ng TT, White SC, Cortina G, Reber HA, Hines OJ. Pancreatic serous cystadenocarcinoma: a case report and review of the literature. J Gastrointest Surg. 2009;13:1864-1868. |
| 115. | Roggin KK, Chennat J, Oto A, Noffsinger A, Briggs A, Matthews JB. Pancreatic cystic neoplasm. Curr Probl Surg. 2010;47:459-510. |
| 116. | Wenig BM, Albores-Saavedra J, Buetow PC, Heffess CS. Pancreatic mucinous cystic neoplasm with sarcomatous stroma: a report of three cases. Am J Surg Pathol. 1997;21:70-80. |
| 117. | Posen JA. Giant cell tumor of the pancreas of the osteoclastic type associated with a mucous secreting cystadenocarcinoma. Hum Pathol. 1981;12:944-947. |
| 118. | Sarnaik AA, Saad AG, Mutema GK, Martin SP, Attar A, Lowy AM. Osteoclast-like giant cell tumor of the pancreas associated with a mucinous cystadenocarcinoma. Surgery. 2003;133:700-701. |
| 119. | Campman SC, Fajardo MA, Rippon MB, Kraegel SA, Ruebner BH. Adenosquamous carcinoma arising in a mucinous cystadenoma of the pancreas. J Surg Oncol. 1997;64:159-162. |
| 120. | Fukushima N, Mukai K, Kanai Y, Hasebe T, Shimada K, Ozaki H, Kinoshita T, Kosuge T. Intraductal papillary tumors and mucinous cystic tumors of the pancreas: clinicopathologic study of 38 cases. Hum Pathol. 1997;28:1010-1017. |
| 121. | Crippa S, Salvia R, Warshaw AL, Domínguez I, Bassi C, Falconi M, Thayer SP, Zamboni G, Lauwers GY, Mino-Kenudson M. Mucinous cystic neoplasm of the pancreas is not an aggressive entity: lessons from 163 resected patients. Ann Surg. 2008;247:571-579. |
| 122. | Wood D, Silberman AW, Heifetz L, Memsic L, Shabot MM. Cystadenocarcinoma of the pancreas: neo-adjuvant therapy and CEA monitoring. J Surg Oncol. 1990;43:56-60. |
| 123. | Bradley EL 3rd, Satchidanand SK. Is there a role for chemoradiation in the management of cystadenocarcinoma of the pancreas? Pancreas. 2005;31:101-103. |
| 124. | Brown HA, Dotto J, Robert M, Salem RR. Squamous cell carcinoma of the pancreas. J Clin Gastroenterol. 2005;39:915-919. |
| 125. | Stelow EB, Shaco-Levy R, Bao F, Garcia J, Klimstra DS. Pancreatic acinar cell carcinomas with prominent ductal differentiation: Mixed acinar ductal carcinoma and mixed acinar endocrine ductal carcinoma. Am J Surg Pathol. 2010;34:510-518. |
| 126. | Caruso RA, Inferrera A, Tuccari G, Barresi G. Acinar cell carcinoma of the pancreas. A histologic, immunocytochemical and ultrastructural study. Histol Histopathol. 1994;9:53-58. |
| 127. | Halder SL, McBeth J, Silman AJ, Thompson DG, Macfarlane GJ. Psychosocial risk factors for the onset of abdominal pain. Results from a large prospective population-based study. Int J Epidemiol. 2002;31:1219-1225; discussion 1225-1226. |
| 128. | Fernandez E, Porta M, Malats N, Belloc J, Gallén M. Symptom-to-diagnosis interval and survival in cancers of the digestive tract. Dig Dis Sci. 2002;47:2434-2440. |
| 129. | Ridder GJ, Klempnauer J. Back pain in patients with ductal pancreatic cancer. Its impact on resectability and prognosis after resection. Scand J Gastroenterol. 1995;30:1216-1220. |
| 130. | La Seta F, Cottone M, Marcenò MP, Maringhini A, Sciarrino E, Pagliaro L. [Echography in patients with clinically suspected pancreatic carcinoma. Analysis of a prospective study]. Radiol Med. 1983;69:538-540. |
| 131. | Krech RL, Walsh D. Symptoms of pancreatic cancer. J Pain Symptom Manage. 1991;6:360-367. |
| 132. | Permert J, Larsson J, Westermark GT, Herrington MK, Christmanson L, Pour PM, Westermark P, Adrian TE. Islet amyloid polypeptide in patients with pancreatic cancer and diabetes. N Engl J Med. 1994;330:313-318. |
| 133. | Schwarts SS, Zeidler A, Moossa AR, Kuku SF, Rubenstein AH. A prospective study of glucose tolerance, insulin, C-peptide, and glucagon responses in patients with pancreatic carcinoma. Am J Dig Dis. 1978;23:1107-1114. |
| 134. | Tarpila E, Borch K, Kullman E, Liedberg G. Pancreatic cancer. Ann Chir Gynaecol. 1986;75:146-150. |
| 135. | Grahm AL, Andrén-Sandberg A. Prospective evaluation of pain in exocrine pancreatic cancer. Digestion. 1997;58:542-549. |
| 136. | Nicolson GL. Cancer metastasis. Organ colonization and the cell-surface properties of malignant cells. Biochim Biophys Acta. 1982;695:113-176. |
| 137. | Hakomori S. Aberrant glycosylation in tumors and tumor-associated carbohydrate antigens. Adv Cancer Res. 1989;52:257-331. |
| 138. | Kleene R, Berger EG. The molecular and cell biology of glycosyltransferases. Biochim Biophys Acta. 1993;1154:283-325. |
| 139. | Magnani JL, Nilsson B, Brockhaus M, Zopf D, Steplewski Z, Koprowski H, Ginsburg V. A monoclonal antibody-defined antigen associated with gastrointestinal cancer is a ganglioside containing sialylated lacto-N-fucopentaose II. J Biol Chem. 1982;257:14365-14369. |
| 140. | Tanaka N, Okada S, Ueno H, Okusaka T, Ikeda M. The usefulness of serial changes in serum CA19-9 levels in the diagnosis of pancreatic cancer. Pancreas. 2000;20:378-381. |
| 141. | Boeck S, Stieber P, Holdenrieder S, Wilkowski R, Heinemann V. Prognostic and therapeutic significance of carbohydrate antigen 19-9 as tumor marker in patients with pancreatic cancer. Oncology. 2006;70:255-264. |
| 142. | Ni XG, Bai XF, Mao YL, Shao YF, Wu JX, Shan Y, Wang CF, Wang J, Tian YT, Liu Q. The clinical value of serum CEA, CA19-9, and CA242 in the diagnosis and prognosis of pancreatic cancer. Eur J Surg Oncol. 2005;31:164-169. |
| 143. | Steinberg W. The clinical utility of the CA 19-9 tumor-associated antigen. Am J Gastroenterol. 1990;85:350-355. |
| 144. | Safi F, Schlosser W, Kolb G, Beger HG. Diagnostic value of CA 19-9 in patients with pancreatic cancer and nonspecific gastrointestinal symptoms. J Gastrointest Surg. 1997;1:106-112. |
| 145. | Goonetilleke KS, Siriwardena AK. Systematic review of carbohydrate antigen (CA 19-9) as a biochemical marker in the diagnosis of pancreatic cancer. Eur J Surg Oncol. 2007;33:266-270. |
| 146. | Lundin J, Roberts PJ, Kuusela P, Haglund C. The prognostic value of preoperative serum levels of CA 19-9 and CEA in patients with pancreatic cancer. Br J Cancer. 1994;69:515-519. |
| 147. | Sperti C, Pasquali C, Catalini S, Cappellazzo F, Bonadimani B, Behboo R, Pedrazzoli S. CA 19-9 as a prognostic index after resection for pancreatic cancer. J Surg Oncol. 1993;52:137-141. |
| 148. | Glenn J, Steinberg WM, Kurtzman SH, Steinberg SM, Sindelar WF. Evaluation of the utility of a radioimmunoassay for serum CA 19-9 levels in patients before and after treatment of carcinoma of the pancreas. J Clin Oncol. 1988;6:462-468. |
| 149. | Safi F, Schlosser W, Falkenreck S, Beger HG. Prognostic value of CA 19-9 serum course in pancreatic cancer. Hepatogastroenterology. 1998;45:253-259. |
| 150. | Tian F, Appert HE, Myles J, Howard JM. Prognostic value of serum CA 19-9 levels in pancreatic adenocarcinoma. Ann Surg. 1992;215:350-355. |
| 151. | Basso D, Fabris C, Del Favero G, Angonese C, Meggiato T, Infantino A, Plebani M, Piccoli A, Leandro G, Burlina A. Serum carcinoembryonic antigen in the differential diagnosis of pancreatic cancer: influence of tumour spread, liver impairment, and age. Dis Markers. 1988;6:203-207. |
| 152. | Chevinsky AH. CEA in tumors of other than colorectal origin. Semin Surg Oncol. 1991;7:162-166. |
| 153. | Ona FV, Zamcheck N, Dhar P, Moore T, Kupchik HZ. Carcinoembryonic antigen (CEA) in the diagnosis of pancreatic cancer. Cancer. 1973;31:324-327. |
| 154. | Futakawa N, Kimura W, Yamagata S, Zhao B, Ilsoo H, Inoue T, Sata N, Kawaguchi Y, Kubota Y, Muto T. Significance of K-ras mutation and CEA level in pancreatic juice in the diagnosis of pancreatic cancer. J Hepatobiliary Pancreat Surg. 2000;7:63-71. |
| 155. | Ozkan H, Kaya M, Cengiz A. Comparison of tumor marker CA 242 with CA 19-9 and carcinoembryonic antigen (CEA) in pancreatic cancer. Hepatogastroenterology. 2003;50:1669-1674. |
| 156. | Gattani AM, Mandeli J, Bruckner HW. Tumor markers in patients with pancreatic carcinoma. Cancer. 1996;78:57-62. |
| 157. | Pasanen PA, Eskelinen M, Partanen K, Pikkarainen P, Penttilä I, Alhava E. A prospective study of serum tumour markers carcinoembryonic antigen, carbohydrate antigens 50 and 242, tissue polypeptide antigen and tissue polypeptide specific antigen in the diagnosis of pancreatic cancer with special reference to multivariate diagnostic score. Br J Cancer. 1994;69:562-565. |
| 158. | Jiang XT, Tao HQ, Zou SC. Detection of serum tumor markers in the diagnosis and treatment of patients with pancreatic cancer. Hepatobiliary Pancreat Dis Int. 2004;3:464-468. |
| 159. | Lindholm L, Johansson C, Jansson EL, Hallberg C, Nilsson O. An immunoradiometric assay (IRMA) for the CA 50 antigen. Tumor Maker Antigens. Lund: Studentlitteratur 1985; 122-133. |
| 160. | Nilsson O, Johansson C, Glimelius B, Persson B, Nørgaard-Pedersen B, Andrén-Sandberg A, Lindholm L. Sensitivity and specificity of CA242 in gastro-intestinal cancer. A comparison with CEA, CA50 and CA 19-9. Br J Cancer. 1992;65:215-221. |
| 161. | Lundin J, Roberts PJ, Kuusela P, Haglund C. Prognostic significance of serum CA 242 in pancreatic cancer. A comparison with CA 19-9. Anticancer Res. 1995;15:2181-2186. |
| 162. | Mu DQ, Wang GF, Peng SY. p53 protein expression and CA19.9 values in differential cytological diagnosis of pancreatic cancer complicated with chronic pancreatitis and chronic pancreatitis. World J Gastroenterol. 2003;9:1815-1818. |
| 163. | Ma ZH, Wang Z. An evaluation of the diagnostic value of CA 19-9 and CEA levels in patients with pancreatic cancer. Nanjing Yike Daxue Xuebao. 2009;23:199-202. |
| 164. | Röthlin MA, Joller H, Largiadèr F. CA 242 is a new tumor marker for pancreatic cancer. Cancer. 1993;71:701-707. |
| 165. | Carpelan-Holmström M, Louhimo J, Stenman UH, Alfthan H, Haglund C. CEA, CA 19-9 and CA 72-4 improve the diagnostic accuracy in gastrointestinal cancers. Anticancer Res. 2002;22:2311-2316. |
| 166. | Pålsson B, Masson P, Andrén-Sandberg A. The influence of cholestasis on CA 50 and CA 242 in pancreatic cancer and benign biliopancreatic diseases. Scand J Gastroenterol. 1993;28:981-987. |
| 167. | Hall NR, Finan PJ, Stephenson BM, Purves DA, Cooper EH. The role of CA-242 and CEA in surveillance following curative resection for colorectal cancer. Br J Cancer. 1994;70:549-553. |
| 168. | Friess H, Büchler M, Auerbach B, Weber A, Malfertheiner P, Hammer K, Madry N, Greiner S, Bosslet K, Beger HG. CA 494--a new tumor marker for the diagnosis of pancreatic cancer. Int J Cancer. 1993;53:759-763. |
| 169. | Simeone DM, Ji B, Banerjee M, Arumugam T, Li D, Anderson MA, Bamberger AM, Greenson J, Brand RE, Ramachandran V. CEACAM1, a novel serum biomarker for pancreatic cancer. Pancreas. 2007;34:436-443. |
| 170. | Bouvet M, Nardin SR, Burton DW, Lee NC, Yang M, Wang X, Baranov E, Behling C, Moossa AR, Hoffman RM. Parathyroid hormone-related protein as a novel tumor marker in pancreatic adenocarcinoma. Pancreas. 2002;24:284-290. |
| 171. | Kumar Y, Gurusamy K, Pamecha V, Davidson BR. Tumor M2-pyruvate kinase as tumor marker in exocrine pancreatic cancer a meta-analysis. Pancreas. 2007;35:114-119. |
| 172. | Gansauge F, Gansauge S, Parker N, Beger MI, Poch B, Link KH, Safi F, Beger HG. CAM 17.1--a new diagnostic marker in pancreatic cancer. Br J Cancer. 1996;74:1997-2002. |
| 173. | Louhimo J, Alfthan H, Stenman UH, Haglund C. Serum HCG beta and CA 72-4 are stronger prognostic factors than CEA, CA 19-9 and CA 242 in pancreatic cancer. Oncology. 2004;66:126-131. |
| 174. | Tamm EP, Silverman PM, Charnsangavej C, Evans DB. Diagnosis, staging, and surveillance of pancreatic cancer. AJR Am J Roentgenol. 2003;180:1311-1323. |
| 175. | Gandolfi L, Torresan F, Solmi L, Puccetti A. The role of ultrasound in biliary and pancreatic diseases. Eur J Ultrasound. 2003;16:141-159. |
| 176. | Giovannini M, Seitz JF. Endoscopic ultrasonography with a linear-type echoendoscope in the evaluation of 94 patients with pancreatobiliary disease. Endoscopy. 1994;26:579-585. |
| 177. | Böttger TC, Boddin J, Düber C, Heintz A, Küchle R, Junginger T. Diagnosing and staging of pancreatic carcinoma-what is necessary? Oncology. 1998;55:122-129. |
| 178. | Rösch T, Lorenz R, Braig C, Feuerbach S, Siewert JR, Schusdziarra V, Classen M. Endoscopic ultrasound in pancreatic tumor diagnosis. Gastrointest Endosc. 1991;37:347-352. |
| 179. | Niederau C, Grendell JH. Diagnosis of pancreatic carcinoma. Imaging techniques and tumor markers. Pancreas. 1992;7:66-86. |
| 180. | Palazzo L, Roseau G, Gayet B, Vilgrain V, Belghiti J, Fékéte F, Paolaggi JA. Endoscopic ultrasonography in the diagnosis and staging of pancreatic adenocarcinoma. Results of a prospective study with comparison to ultrasonography and CT scan. Endoscopy. 1993;25:143-150. |
| 181. | Karlson BM, Ekbom A, Lindgren PG, Källskog V, Rastad J. Abdominal US for diagnosis of pancreatic tumor: prospective cohort analysis. Radiology. 1999;213:107-111. |
| 182. | Shapiro RS, Wagreich J, Parsons RB, Stancato-Pasik A, Yeh HC, Lao R. Tissue harmonic imaging sonography: evaluation of image quality compared with conventional sonography. AJR Am J Roentgenol. 1998;171:1203-1206. |
| 183. | Baarir N, Amouyal G, Faintuch JM, Houry S, Huguier M. [Comparison of color Doppler ultrasonography and endoscopic ultrasonography for preoperative evaluation of the mesenteric-portal axis in pancreatic lesions]. Chirurgie. 1998;123:445-449. |
| 184. | Casadei R, Ghigi G, Gullo L, Moretti CC, Greco VM, Salizzoni E, Canini R, Marrano D. Role of color Doppler ultrasonography in the preoperative staging of pancreatic cancer. Pancreas. 1998;16:26-30. |
| 185. | Dietrich CF, Braden B, Hocke M, Ott M, Ignee A. Improved characterisation of solitary solid pancreatic tumours using contrast enhanced transabdominal ultrasound. J Cancer Res Clin Oncol. 2008;134:635-643. |
| 186. | Sørensen MB, Weibull AS, Haubek A, Rokkjaer M, Jørgensen J. [Resectability of papillary and pancreatic cancer assessed by ultrasonography and computer tomography]. Ugeskr Laeger. 1997;159:743-747. |
| 187. | Sahani DV, Shah ZK, Catalano OA, Boland GW, Brugge WR. Radiology of pancreatic adenocarcinoma: current status of imaging. J Gastroenterol Hepatol. 2008;23:23-33. |
| 188. | DeWitt J, Devereaux B, Chriswell M, McGreevy K, Howard T, Imperiale TF, Ciaccia D, Lane KA, Maglinte D, Kopecky K. Comparison of endoscopic ultrasonography and multidetector computed tomography for detecting and staging pancreatic cancer. Ann Intern Med. 2004;141:753-763. |
| 189. | Klapman JB, Chang KJ, Lee JG, Nguyen P. Negative predictive value of endoscopic ultrasound in a large series of patients with a clinical suspicion of pancreatic cancer. Am J Gastroenterol. 2005;100:2658-2661. |
| 190. | Brugge WR, Lee MJ, Kelsey PB, Schapiro RH, Warshaw AL. The use of EUS to diagnose malignant portal venous system invasion by pancreatic cancer. Gastrointest Endosc. 1996;43:561-567. |
| 191. | Faigel DO, Kochman ML. The role of endoscopic ultrasound in the preoperative staging of pancreatic malignancies. Gastrointest Endosc. 1996;43:626-628. |
| 192. | Aslanian H, Salem R, Lee J, Andersen D, Robert M, Topazian M. EUS diagnosis of vascular invasion in pancreatic cancer: surgical and histologic correlates. Am J Gastroenterol. 2005;100:1381-1385. |
| 193. | Dietrich CF, Ignee A, Braden B, Barreiros AP, Ott M, Hocke M. Improved differentiation of pancreatic tumors using contrast-enhanced endoscopic ultrasound. Clin Gastroenterol Hepatol. 2008;6:590-597.e1. |
| 194. | Eloubeidi MA, Tamhane A, Varadarajulu S, Wilcox CM. Frequency of major complications after EUS-guided FNA of solid pancreatic masses: a prospective evaluation. Gastrointest Endosc. 2006;63:622-629. |
| 195. | Choi BI, Chung MJ, Han JK, Han MC, Yoon YB. Detection of pancreatic adenocarcinoma: relative value of arterial and late phases of spiral CT. Abdom Imaging. 1997;22:199-203. |
| 196. | Bronstein YL, Loyer EM, Kaur H, Choi H, David C, DuBrow RA, Broemeling LD, Cleary KR, Charnsangavej C. Detection of small pancreatic tumors with multiphasic helical CT. AJR Am J Roentgenol. 2004;182:619-623. |
| 197. | Megibow AJ, Zhou XH, Rotterdam H, Francis IR, Zerhouni EA, Balfe DM, Weinreb JC, Aisen A, Kuhlman J, Heiken JP. Pancreatic adenocarcinoma: CT versus MR imaging in the evaluation of resectability--report of the Radiology Diagnostic Oncology Group. Radiology. 1995;195:327-332. |
| 198. | Gangi S, Fletcher JG, Nathan MA, Christensen JA, Harmsen WS, Crownhart BS, Chari ST. Time interval between abnormalities seen on CT and the clinical diagnosis of pancreatic cancer: retrospective review of CT scans obtained before diagnosis. AJR Am J Roentgenol. 2004;182:897-903. |
| 199. | Ohwada S, Ogawa T, Tanahashi Y, Nakamura S, Takeyoshi I, Ohya T, Ikeya T, Kawashima K, Kawashima Y, Morishita Y. Fibrin glue sandwich prevents pancreatic fistula following distal pancreatectomy. World J Surg. 1998;22:494-498. |
| 200. | Baum U, Lell M, Nömayr A, Wolf H, Brunner T, Greess H, Bautz W. [Multiplanar spiral CT in the diagnosis of pancreatic tumors]. Radiologe. 1999;39:958-964. |
| 201. | Fletcher JG, Wiersema MJ, Farrell MA, Fidler JL, Burgart LJ, Koyama T, Johnson CD, Stephens DH, Ward EM, Harmsen WS. Pancreatic malignancy: value of arterial, pancreatic, and hepatic phase imaging with multi-detector row CT. Radiology. 2003;229:81-90. |
| 202. | Vargas R, Nino-Murcia M, Trueblood W, Jeffrey RB Jr. MDCT in Pancreatic adenocarcinoma: prediction of vascular invasion and resectability using a multiphasic technique with curved planar reformations. AJR Am J Roentgenol. 2004;182:419-425. |
| 203. | Diehl SJ, Lehmann KJ, Sadick M, Lachmann R, Georgi M. Pancreatic cancer: value of dual-phase helical CT in assessing resectability. Radiology. 1998;206:373-378. |
| 204. | Lu DS, Reber HA, Krasny RM, Kadell BM, Sayre J. Local staging of pancreatic cancer: criteria for unresectability of major vessels as revealed by pancreatic-phase, thin-section helical CT. AJR Am J Roentgenol. 1997;168:1439-1443. |
| 205. | Ahn SS, Kim MJ, Choi JY, Hong HS, Chung YE, Lim JS. Indicative findings of pancreatic cancer in prediagnostic CT. Eur Radiol. 2009;19:2448-2455. |
| 206. | Johnson PT, Outwater EK. Pancreatic carcinoma versus chronic pancreatitis: dynamic MR imaging. Radiology. 1999;212:213-218. |
| 207. | Bluemke DA, Cameron JL, Hruban RH, Pitt HA, Siegelman SS, Soyer P, Fishman EK. Potentially resectable pancreatic adenocarcinoma: spiral CT assessment with surgical and pathologic correlation. Radiology. 1995;197:381-385. |
| 208. | Schima W, Függer R, Schober E, Oettl C, Wamser P, Grabenwöger F, Ryan JM, Novacek G. Diagnosis and staging of pancreatic cancer: comparison of mangafodipir trisodium-enhanced MR imaging and contrast-enhanced helical hydro-CT. AJR Am J Roentgenol. 2002;179:717-724. |
| 209. | Ichikawa T, Haradome H, Hachiya J, Nitatori T, Ohtomo K, Kinoshita T, Araki T. Pancreatic ductal adenocarcinoma: preoperative assessment with helical CT versus dynamic MR imaging. Radiology. 1997;202:655-662. |
| 210. | Irie H, Honda H, Kaneko K, Kuroiwa T, Yoshimitsu K, Masuda K. Comparison of helical CT and MR imaging in detecting and staging small pancreatic adenocarcinoma. Abdom Imaging. 1997;22:429-433. |
| 211. | Romijn MG, Stoker J, van Eijck CH, van Muiswinkel JM, Torres CG, Laméris JS. MRI with mangafodipir trisodium in the detection and staging of pancreatic cancer. J Magn Reson Imaging. 2000;12:261-268. |
| 212. | Andersson M, Kostic S, Johansson M, Lundell L, Asztély M, Hellström M. MRI combined with MR cholangiopancreatography versus helical CT in the evaluation of patients with suspected periampullary tumors: a prospective comparative study. Acta Radiol. 2005;46:16-27. |
| 213. | Hänninen EL, Ricke J, Amthauer H, Röttgen R, Böhmig M, Langrehr J, Pech M, Denecke T, Rosewicz S, Felix R. Magnetic resonance cholangiopancreatography: image quality, ductal morphology, and value of additional T2- and T1-weighted sequences for the assessment of suspected pancreatic cancer. Acta Radiol. 2005;46:117-125. |
| 214. | Park HS, Lee JM, Choi HK, Hong SH, Han JK, Choi BI. Preoperative evaluation of pancreatic cancer: comparison of gadolinium-enhanced dynamic MRI with MR cholangiopancreatography versus MDCT. J Magn Reson Imaging. 2009;30:586-595. |
| 215. | Grenacher L, Klauss M, Dukic L, Delorme S, Knaebel HP, Düx M, Kauczor HU, Büchler MW, Kauffmann GW, Richter GM. [Diagnosis and staging of pancreatic carcinoma: MRI versus multislice-CT -- a prospective study]. Rofo. 2004;176:1624-1633. |
| 216. | Bipat S, Phoa SS, van Delden OM, Bossuyt PM, Gouma DJ, Laméris JS, Stoker J. Ultrasonography, computed tomography and magnetic resonance imaging for diagnosis and determining resectability of pancreatic adenocarcinoma: a meta-analysis. J Comput Assist Tomogr. 2005;29:438-445. |
| 217. | Maemura K, Takao S, Shinchi H, Noma H, Mataki Y, Kurahara H, Jinnouchi S, Aikou T. Role of positron emission tomography in decisions on treatment strategies for pancreatic cancer. J Hepatobiliary Pancreat Surg. 2006;13:435-441. |
| 218. | Higashi T, Saga T, Nakamoto Y, Ishimori T, Fujimoto K, Doi R, Imamura M, Konishi J. Diagnosis of pancreatic cancer using fluorine-18 fluorodeoxyglucose positron emission tomography (FDG PET) --usefulness and limitations in "clinical reality". Ann Nucl Med. 2003;17:261-279. |
| 219. | Wakabayashi H, Nishiyama Y, Otani T, Sano T, Yachida S, Okano K, Izuishi K, Suzuki Y. Role of 18F-fluorodeoxyglucose positron emission tomography imaging in surgery for pancreatic cancer. World J Gastroenterol. 2008;14:64-69. |
| 220. | Schima W, Függer R. Evaluation of focal pancreatic masses: comparison of mangafodipir-enhanced MR imaging and contrast-enhanced helical CT. Eur Radiol. 2002;12:2998-3008. |
| 221. | Delbeke D, Rose DM, Chapman WC, Pinson CW, Wright JK, Beauchamp RD, Shyr Y, Leach SD. Optimal interpretation of FDG PET in the diagnosis, staging and management of pancreatic carcinoma. J Nucl Med. 1999;40:1784-1791. |
| 222. | Heinrich S, Goerres GW, Schäfer M, Sagmeister M, Bauerfeind P, Pestalozzi BC, Hany TF, von Schulthess GK, Clavien PA. Positron emission tomography/computed tomography influences on the management of resectable pancreatic cancer and its cost-effectiveness. Ann Surg. 2005;242:235-243. |
| 223. | Lee TY, Kim MH, Park do H, Seo DW, Lee SK, Kim JS, Lee KT. Utility of 18F-FDG PET/CT for differentiation of autoimmune pancreatitis with atypical pancreatic imaging findings from pancreatic cancer. AJR Am J Roentgenol. 2009;193:343-348. |
| 224. | Zhao C, Chen Z, Ye X, Zhang Y, Zhan H, Aburano T, Tian M, Zhang H. Imaging a pancreatic carcinoma xenograft model with 11C-acetate: a comparison study with 18F-FDG. Nucl Med Commun. 2009;30:971-977. |
| 225. | Nakata B, Nishimura S, Ishikawa T, Ohira M, Nishino H, Kawabe J, Ochi H, Hirakawa K. Prognostic predictive value of 18F-fluorodeoxyglucose positron emission tomography for patients with pancreatic cancer. Int J Oncol. 2001;19:53-58. |
| 226. | Yoshioka M, Sato T, Furuya T, Shibata S, Andoh H, Asanuma Y, Hatazawa J, Shimosegawa E, Koyama K, Yamamoto Y. Role of positron emission tomography with 2-deoxy-2-[18F]fluoro-D-glucose in evaluating the effects of arterial infusion chemotherapy and radiotherapy on pancreatic cancer. J Gastroenterol. 2004;39:50-55. |
| 227. | Klever P, Bares R, Fass J, Büll U, Schumpelick V. PET with fluorine-18 deoxyglucose for pancreatic disease. Lancet. 1992;340:1158-1159. |
| 228. | Nakamoto Y, Higashi T, Sakahara H, Tamaki N, Kogire M, Imamura M, Konishi J. Contribution of PET in the detection of liver metastases from pancreatic tumours. Clin Radiol. 1999;54:248-252. |
| 229. | Berberat P, Friess H, Kashiwagi M, Beger HG, Büchler MW. Diagnosis and staging of pancreatic cancer by positron emission tomography. World J Surg. 1999;23:882-887. |
| 230. | Zimny M, Fass J, Bares R, Cremerius U, Sabri O, Buechin P, Schumpelick V, Buell U. Fluorodeoxyglucose positron emission tomography and the prognosis of pancreatic carcinoma. Scand J Gastroenterol. 2000;35:883-888. |
| 231. | Tanaka S, Kitamra T, Yamamoto K, Fujikawa S, Imaoka T, Nishikawa S, Nakaizumi A, Uehara H, Ishikawa O, Ohigashi H. Evaluation of routine sonography for early detection of pancreatic cancer. Jpn J Clin Oncol. 1996;26:422-427. |
| 232. | Candiani F, Meduri F, Norberto L, Calderone M. [Contrast media in ultrasonography. Venous involvement in tumors of the head of the pancreas]. Radiol Med. 1998;95:29-33. |
| 233. | Calculli L, Casadei R, Amore B, Albini Riccioli L, Minni F, Caputo M, Marrano D, Gavelli G. The usefulness of spiral Computed Tomography and colour-Doppler ultrasonography to predict portal-mesenteric trunk involvement in pancreatic cancer. Radiol Med. 2002;104:307-315. |
| 234. | Akahoshi K, Chijiiwa Y, Nakano I, Nawata H, Ogawa Y, Tanaka M, Nagai E, Tsuneyoshi M. Diagnosis and staging of pancreatic cancer by endoscopic ultrasound. Br J Radiol. 1998;71:492-496. |
| 235. | Legmann P, Vignaux O, Dousset B, Baraza AJ, Palazzo L, Dumontier I, Coste J, Louvel A, Roseau G, Couturier D. Pancreatic tumors: comparison of dual-phase helical CT and endoscopic sonography. AJR Am J Roentgenol. 1998;170:1315-1322. |
| 236. | Bilimoria KY, Bentrem DJ, Ko CY, Ritchey J, Stewart AK, Winchester DP, Talamonti MS. Validation of the 6th edition AJCC Pancreatic Cancer Staging System: report from the National Cancer Database. Cancer. 2007;110:738-744. |
| 237. | AJCC Cancer Staging Handbook. 7th edition. New York: Springer-Verlag 2010; . |
| 238. | Warshaw AL, Fernández-del Castillo C. Pancreatic carcinoma. N Engl J Med. 1992;326:455-465. |
| 239. | Soriano A, Castells A, Ayuso C, Ayuso JR, de Caralt MT, Ginès MA, Real MI, Gilabert R, Quintó L, Trilla A. Preoperative staging and tumor resectability assessment of pancreatic cancer: prospective study comparing endoscopic ultrasonography, helical computed tomography, magnetic resonance imaging, and angiography. Am J Gastroenterol. 2004;99:492-501. |
| 240. | Cuschieri A. Laparoscopy for pancreatic cancer: does it benefit the patient? Eur J Surg Oncol. 1988;14:41-44. |
| 241. | Warshaw AL, Tepper JE, Shipley WU. Laparoscopy in the staging and planning of therapy for pancreatic cancer. Am J Surg. 1986;151:76-80. |
| 242. | Warshaw AL, Gu ZY, Wittenberg J, Waltman AC. Preoperative staging and assessment of resectability of pancreatic cancer. Arch Surg. 1990;125:230-233. |
| 243. | Camacho D, Reichenbach D, Duerr GD, Venema TL, Sweeney JF, Fisher WE. Value of laparoscopy in the staging of pancreatic cancer. JOP. 2005;6:552-561. |
| 244. | Ellsmere J, Mortele K, Sahani D, Maher M, Cantisani V, Wells W, Brooks D, Rattner D. Does multidetector-row CT eliminate the role of diagnostic laparoscopy in assessing the resectability of pancreatic head adenocarcinoma? Surg Endosc. 2005;19:369-373. |
| 245. | Morganti AG, Brizi MG, Macchia G, Sallustio G, Costamagna G, Alfieri S, Mattiucci GC, Valentini V, Natale L, Deodato F. The prognostic effect of clinical staging in pancreatic adenocarcinoma. Ann Surg Oncol. 2005;12:145-151. |
| 246. | Nieveen van Dijkum EJ, Romijn MG, Terwee CB, de Wit LT, van der Meulen JH, Lameris HS, Rauws EA, Obertop H, van Eyck CH, Bossuyt PM. Laparoscopic staging and subsequent palliation in patients with peripancreatic carcinoma. Ann Surg. 2003;237:66-73. |
| 247. | Conlon KC, Dougherty E, Klimstra DS, Coit DG, Turnbull AD, Brennan MF. The value of minimal access surgery in the staging of patients with potentially resectable peripancreatic malignancy. Ann Surg. 1996;223:134-140. |
| 248. | Tilleman EH, Busch OR, Bemelman WA, van Gulik TM, Obertop H, Gouma DJ. Diagnostic laparoscopy in staging pancreatic carcinoma: developments during the past decade. J Hepatobiliary Pancreat Surg. 2004;11:11-16. |
| 249. | Pisters PW, Lee JE, Vauthey JN, Charnsangavej C, Evans DB. Laparoscopy in the staging of pancreatic cancer. Br J Surg. 2001;88:325-337. |
| 250. | Kwon AH, Inui H, Kamiyama Y. Preoperative laparoscopic examination using surgical manipulation and ultrasonography for pancreatic lesions. Endoscopy. 2002;34:464-468. |
| 251. | Catheline JM, Turner R, Rizk N, Barrat C, Champault G. The use of diagnostic laparoscopy supported by laparoscopic ultrasonography in the assessment of pancreatic cancer. Surg Endosc. 1999;13:239-245. |
| 252. | White R, Winston C, Gonen M, D'Angelica M, Jarnagin W, Fong Y, Conlon K, Brennan M, Allen P. Current utility of staging laparoscopy for pancreatic and peripancreatic neoplasms. J Am Coll Surg. 2008;206:445-450. |
| 253. | National Comprehensive Cancer Network. Practice Guidelines for Pancreatic Cancer. Accessed on October 20, 2010. . |
| 254. | Chang DK, Merrett ND, Biankin AV. Improving outcomes for operable pancreatic cancer: is access to safer surgery the problem? J Gastroenterol Hepatol. 2008;23:1036-1045. |
| 255. | Freelove R, Walling AD. Pancreatic cancer: diagnosis and management. Am Fam Physician. 2006;73:485-492. |
| 256. | Büchler MW, Wagner M, Schmied BM, Uhl W, Friess H, Z'graggen K. Changes in morbidity after pancreatic resection: toward the end of completion pancreatectomy. Arch Surg. 2003;138:1310-1314; discussion 1315. |
| 257. | Birkmeyer JD, Siewers AE, Finlayson EV, Stukel TA, Lucas FL, Batista I, Welch HG, Wennberg DE. Hospital volume and surgical mortality in the United States. N Engl J Med. 2002;346:1128-1137. |
| 258. | Cameron JL, Riall TS, Coleman J, Belcher KA. One thousand consecutive pancreaticoduodenectomies. Ann Surg. 2006;244:10-15. |
| 259. | The US National Cancer Institute. Surveillance Epidemiology and End Results (SEER) data base. 2007. Available from: http://seer.cancer.gov/. |
| 260. | Zuckerman DS, Ryan DP. Adjuvant therapy for pancreatic cancer: a review. Cancer. 2008;112:243-249. |
| 261. | Takhar AS, Palaniappan P, Dhingsa R, Lobo DN. Recent developments in diagnosis of pancreatic cancer. BMJ. 2004;329:668-673. |
| 263. | Barugola G, Partelli S, Marcucci S, Sartori N, Capelli P, Bassi C, Pederzoli P, Falconi M. Resectable pancreatic cancer: who really benefits from resection? Ann Surg Oncol. 2009;16:3316-3322. |
| 264. | van der Gaag NA, Kloek JJ, de Castro SM, Busch OR, van Gulik TM, Gouma DJ. Preoperative biliary drainage in patients with obstructive jaundice: history and current status. J Gastrointest Surg. 2009;13:814-820. |
| 265. | Klinkenbijl JH, Jeekel J, Schmitz PI, Rombout PA, Nix GA, Bruining HA, van Blankenstein M. Carcinoma of the pancreas and periampullary region: palliation versus cure. Br J Surg. 1993;80:1575-1578. |
| 266. | Kimmings AN, van Deventer SJ, Obertop H, Rauws EA, Huibregtse K, Gouma DJ. Endotoxin, cytokines, and endotoxin binding proteins in obstructive jaundice and after preoperative biliary drainage. Gut. 2000;46:725-731. |
| 267. | Jethwa P, Breuning E, Bhati C, Buckles J, Mirza D, Bramhall S. The microbiological impact of pre-operative biliary drainage on patients undergoing hepato-biliary-pancreatic (HPB) surgery. Aliment Pharmacol Ther. 2007;25:1175-1180. |
| 268. | Limongelli P, Pai M, Bansi D, Thiallinagram A, Tait P, Jackson J, Habib NA, Williamson RC, Jiao LR. Correlation between preoperative biliary drainage, bile duct contamination, and postoperative outcomes for pancreatic surgery. Surgery. 2007;142:313-318. |
| 269. | Coates JM, Beal SH, Russo JE, Vanderveen KA, Chen SL, Bold RJ, Canter RJ. Negligible effect of selective preoperative biliary drainage on perioperative resuscitation, morbidity, and mortality in patients undergoing pancreaticoduodenectomy. Arch Surg. 2009;144:841-847. |
| 270. | Mezhir JJ, Brennan MF, Baser RE, D'Angelica MI, Fong Y, DeMatteo RP, Jarnagin WR, Allen PJ. A matched case-control study of preoperative biliary drainage in patients with pancreatic adenocarcinoma: routine drainage is not justified. J Gastrointest Surg. 2009;13:2163-2169. |
| 271. | Wang Q, Gurusamy KS, Lin H, Xie X, Wang C. Preoperative biliary drainage for obstructive jaundice. Cochrane Database Syst Rev. 2008;CD005444. |
| 272. | Sewnath ME, Karsten TM, Prins MH, Rauws EJ, Obertop H, Gouma DJ. A meta-analysis on the efficacy of preoperative biliary drainage for tumors causing obstructive jaundice. Ann Surg. 2002;236:17-27. |
| 273. | van der Gaag NA, Rauws EA, van Eijck CH, Bruno MJ, van der Harst E, Kubben FJ, Gerritsen JJ, Greve JW, Gerhards MF, de Hingh IH. Preoperative biliary drainage for cancer of the head of the pancreas. N Engl J Med. 2010;362:129-137. |
| 274. | Landry J, Catalano PJ, Staley C, Harris W, Hoffman J, Talamonti M, Xu N, Cooper H, Benson AB 3rd. Randomized phase II study of gemcitabine plus radiotherapy versus gemcitabine, 5-fluorouracil, and cisplatin followed by radiotherapy and 5-fluorouracil for patients with locally advanced, potentially resectable pancreatic adenocarcinoma. J Surg Oncol. 2010;101:587-592. |
| 275. | Lee S, Oh SY, Kim BG, Kwon HC, Kim SH, Rho MH, Kim YH, Rho MS, Jeong JS, Kim HJ. Second-line treatment with a combination of continuous 5-fluorouracil, doxorubicin, and mitomycin-C (conti-FAM) in gemcitabine-pretreated pancreatic and biliary tract cancer. Am J Clin Oncol. 2009;32:348-352. |
| 276. | Le Scodan R, Mornex F, Girard N, Mercier C, Valette PJ, Ychou M, Bibeau F, Roy P, Scoazec JY, Partensky C. Preoperative chemoradiation in potentially resectable pancreatic adenocarcinoma: feasibility, treatment effect evaluation and prognostic factors, analysis of the SFRO-FFCD 9704 trial and literature review. Ann Oncol. 2009;20:1387-1396. |
| 277. | Heinrich S, Schäfer M, Weber A, Hany TF, Bhure U, Pestalozzi BC, Clavien PA. Neoadjuvant chemotherapy generates a significant tumor response in resectable pancreatic cancer without increasing morbidity: results of a prospective phase II trial. Ann Surg. 2008;248:1014-1022. |
| 278. | Marti JL, Hochster HS, Hiotis SP, Donahue B, Ryan T, Newman E. Phase I/II trial of induction chemotherapy followed by concurrent chemoradiotherapy and surgery for locoregionally advanced pancreatic cancer. Ann Surg Oncol. 2008;15:3521-3531. |
| 279. | Le Scodan R, Mornex F, Partensky C, Mercier C, Valette PJ, Ychou M, Roy P, Scoazec JY. Histopathological response to preoperative chemoradiation for resectable pancreatic adenocarcinoma: the French Phase II FFCD 9704-SFRO Trial. Am J Clin Oncol. 2008;31:545-552. |
| 280. | Lind PA, Isaksson B, Almström M, Johnsson A, Albiin N, Byström P, Permert J. Efficacy of preoperative radiochemotherapy in patients with locally advanced pancreatic carcinoma. Acta Oncol. 2008;47:413-420. |
| 281. | Evans DB, Varadhachary GR, Crane CH, Sun CC, Lee JE, Pisters PW, Vauthey JN, Wang H, Cleary KR, Staerkel GA. Preoperative gemcitabine-based chemoradiation for patients with resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3496-3502. |
| 282. | Varadhachary GR, Wolff RA, Crane CH, Sun CC, Lee JE, Pisters PW, Vauthey JN, Abdalla E, Wang H, Staerkel GA. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:3487-3495. |
| 283. | Heinrich S, Pestalozzi BC, Schäfer M, Weber A, Bauerfeind P, Knuth A, Clavien PA. Prospective phase II trial of neoadjuvant chemotherapy with gemcitabine and cisplatin for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26:2526-2531. |
| 284. | Boulay BR, Gardner TB, Gordon SR. Occlusion rate and complications of plastic biliary stent placement in patients undergoing neoadjuvant chemoradiotherapy for pancreatic cancer with malignant biliary obstruction. J Clin Gastroenterol. 2010;44:452-455. |
| 285. | Wasan SM, Ross WA, Staerkel GA, Lee JH. Use of expandable metallic biliary stents in resectable pancreatic cancer. Am J Gastroenterol. 2005;100:2056-2061. |
| 286. | Kausch W. Das carcinom der papilla duodeni und seine radikale entfernung. Beitr Klin Chir. 1912;78:439-451. |
| 287. | Whipple A. Present-day surgery of the pancreas. N Engl J Med. 1942;226:515-518. |
| 288. | Trede M. Surgery of the pancreas. Edinburgh: Churchill Livingstone 1993; . |
| 289. | Seiler CA, Wagner M, Sadowski C, Kulli C, Büchler MW. Randomized prospective trial of pylorus-preserving vs. Classic duodenopancreatectomy (Whipple procedure): initial clinical results. J Gastrointest Surg. 2000;4:443-452. |
| 290. | Williamson RC, Bliouras N, Cooper MJ, Davies ER. Gastric emptying and enterogastric reflux after conservative and conventional pancreatoduodenectomy. Surgery. 1993;114:82-86. |
| 291. | Watson K. Carcinoma of the ampulla of Vater. Successful radical resection. Br J Surg. 1944;31:368. |
| 292. | Traverso LW, Longmire WP Jr. Preservation of the pylorus in pancreaticoduodenectomy. Surg Gynecol Obstet. 1978;146:959-962. |
| 293. | Loos M, Kleeff J, Friess H, Buchler MW. Surgical treatment of pancreatic cancer. Ann N Y Acad Sci. 2008;1138:169-180. |
| 294. | Horstmann O, Markus PM, Ghadimi MB, Becker H. Pylorus preservation has no impact on delayed gastric emptying after pancreatic head resection. Pancreas. 2004;28:69-74. |
| 295. | Wenger FA, Jacobi CA, Haubold K, Zieren HU, Müller JM. [Gastrointestinal quality of life after duodenopancreatectomy in pancreatic carcinoma. Preliminary results of a prospective randomized study: pancreatoduodenectomy or pylorus-preserving pancreatoduodenectomy]. Chirurg. 1999;70:1454-1459. |
| 296. | Park YC, Kim SW, Jang JY, Ahn YJ, Park YH. Factors influencing delayed gastric emptying after pylorus-preserving pancreatoduodenectomy. J Am Coll Surg. 2003;196:859-865. |
| 297. | Roder JD, Stein HJ, Hüttl W, Siewert JR. Pylorus-preserving versus standard pancreatico-duodenectomy: an analysis of 110 pancreatic and periampullary carcinomas. Br J Surg. 1992;79:152-155. |
| 298. | Diener MK, Knaebel HP, Heukaufer C, Antes G, Büchler MW, Seiler CM. A systematic review and meta-analysis of pylorus-preserving versus classical pancreaticoduodenectomy for surgical treatment of periampullary and pancreatic carcinoma. Ann Surg. 2007;245:187-200. |
| 299. | Seiler CA, Wagner M, Bachmann T, Redaelli CA, Schmied B, Uhl W, Friess H, Büchler MW. Randomized clinical trial of pylorus-preserving duodenopancreatectomy versus classical Whipple resection-long term results. Br J Surg. 2005;92:547-556. |
| 300. | Lin PW, Shan YS, Lin YJ, Hung CJ. Pancreaticoduodenectomy for pancreatic head cancer: PPPD versus Whipple procedure. Hepatogastroenterology. 2005;52:1601-1604. |
| 301. | Schmidt CM, Powell ES, Yiannoutsos CT, Howard TJ, Wiebke EA, Wiesenauer CA, Baumgardner JA, Cummings OW, Jacobson LE, Broadie TA. Pancreaticoduodenectomy: a 20-year experience in 516 patients. Arch Surg. 2004;139:718-725; discussion 725-727. |
| 302. | Bassi C, Falconi M, Salvia R, Mascetta G, Molinari E, Pederzoli P. Management of complications after pancreaticoduodenectomy in a high volume centre: results on 150 consecutive patients. Dig Surg. 2001;18:453-457; discussion 458. |
| 303. | Shrikhande SV, D'Souza MA. Pancreatic fistula after pancreatectomy: evolving definitions, preventive strategies and modern management. World J Gastroenterol. 2008;14:5789-5796. |
| 304. | Dixon E, Fingerhut A, Bassi C, Sutherland F, McKay A. Meta-analysis of pancreaticojejunostomy versus pancreaticogastrostomy reconstruction after pancreaticoduodenectomy: authors' comment (Br J Surg 2006; 93: 929-936). Br J Surg. 2006;93:1435. |
| 305. | Wente MN, Shrikhande SV, Müller MW, Diener MK, Seiler CM, Friess H, Büchler MW. Pancreaticojejunostomy versus pancreaticogastrostomy: systematic review and meta-analysis. Am J Surg. 2007;193:171-183. |
| 306. | Andrén-Sandberg A, Wagner M, Tihanyi T, Löfgren P, Friess H. Technical aspects of left-sided pancreatic resection for cancer. Dig Surg. 1999;16:305-312. |
| 307. | Shoup M, Brennan MF, McWhite K, Leung DH, Klimstra D, Conlon KC. The value of splenic preservation with distal pancreatectomy. Arch Surg. 2002;137:164-168. |
| 308. | Warshaw AL. Conservation of the spleen with distal pancreatectomy. Arch Surg. 1988;123:550-553. |
| 309. | Bilimoria MM, Cormier JN, Mun Y, Lee JE, Evans DB, Pisters PW. Pancreatic leak after left pancreatectomy is reduced following main pancreatic duct ligation. Br J Surg. 2003;90:190-196. |
| 310. | Sheehan MK, Beck K, Creech S, Pickleman J, Aranha GV. Distal pancreatectomy: does the method of closure influence fistula formation? Am Surg. 2002;68:264-267; discussion 267-268. |
| 311. | Takeuchi K, Tsuzuki Y, Ando T, Sekihara M, Hara T, Kori T, Nakajima H, Kuwano H. Distal pancreatectomy: is staple closure beneficial? ANZ J Surg. 2003;73:922-925. |
| 312. | Balzano G, Zerbi A, Cristallo M, Di Carlo V. The unsolved problem of fistula after left pancreatectomy: the benefit of cautious drain management. J Gastrointest Surg. 2005;9:837-842. |
| 313. | Suzuki Y, Fujino Y, Tanioka Y, Hori Y, Ueda T, Takeyama Y, Tominaga M, Ku Y, Yamamoto YM, Kuroda Y. Randomized clinical trial of ultrasonic dissector or conventional division in distal pancreatectomy for non-fibrotic pancreas. Br J Surg. 1999;86:608-611. |
| 314. | Adam U, Makowiec F, Riediger H, Trzeczak S, Benz S, Hopt UT. [Distal pancreatic resection--indications, techniques and complications]. Zentralbl Chir. 2001;126:908-912. |
| 315. | Moriura S, Kimura A, Ikeda S, Iwatsuka Y, Ikezawa T, Naiki K. Closure of the distal pancreatic stump with a seromuscular flap. Surg Today. 1995;25:992-994. |
| 316. | Kluger Y, Alfici R, Abbley B, Soffer D, Aladgem D. Gastric serosal patch in distal pancreatectomy for injury: a neglected technique. Injury. 1997;28:127-129. |
| 317. | Brennan MF, Moccia RD, Klimstra D. Management of adenocarcinoma of the body and tail of the pancreas. Ann Surg. 1996;223:506-511; discussion 511-512. |
| 318. | Sohn TA, Yeo CJ, Cameron JL, Koniaris L, Kaushal S, Abrams RA, Sauter PK, Coleman J, Hruban RH, Lillemoe KD. Resected adenocarcinoma of the pancreas-616 patients: results, outcomes, and prognostic indicators. J Gastrointest Surg. 2000;4:567-579. |
| 319. | Mori T, Abe N, Sugiyama M, Atomi Y. Laparoscopic pancreatic surgery. J Hepatobiliary Pancreat Surg. 2005;12:451-455. |
| 320. | Olivié D, Lepanto L, Billiard JS, Audet P, Lavallée JM. Predicting resectability of pancreatic head cancer with multi-detector CT. Surgical and pathologic correlation. JOP. 2007;8:753-758. |
| 321. | Gagner M, Pomp A. Laparoscopic pylorus-preserving pancreatoduodenectomy. Surg Endosc. 1994;8:408-410. |
| 322. | Gagner M, Palermo M. Laparoscopic Whipple procedure: review of the literature. J Hepatobiliary Pancreat Surg. 2009;16:726-730. |
| 323. | Kendrick ML, Cusati D. Total laparoscopic pancreaticoduodenectomy: feasibility and outcome in an early experience. Arch Surg. 2010;145:19-23. |
| 324. | Palanivelu C, Jani K, Senthilnathan P, Parthasarathi R, Rajapandian S, Madhankumar MV. Laparoscopic pancreaticoduodenectomy: technique and outcomes. J Am Coll Surg. 2007;205:222-230. |
| 325. | Dulucq JL, Wintringer P, Mahajna A. Laparoscopic pancreaticoduodenectomy for benign and malignant diseases. Surg Endosc. 2006;20:1045-1050. |
| 326. | Pugliese R, Scandroglio I, Sansonna F, Maggioni D, Costanzi A, Citterio D, Ferrari GC, Di Lernia S, Magistro C. Laparoscopic pancreaticoduodenectomy: a retrospective review of 19 cases. Surg Laparosc Endosc Percutan Tech. 2008;18:13-18. |
| 327. | Sahm M, Pross M, Schubert D, Lippert H. Laparoscopic distal pancreatic resection: our own experience in the treatment of solid tumors. Surg Today. 2009;39:1103-1108. |
| 328. | Al-Taan OS, Stephenson JA, Briggs C, Pollard C, Metcalfe MS, Dennison AR. Laparoscopic pancreatic surgery: a review of present results and future prospects. HPB (Oxford). 2010;12:239-243. |
| 329. | Kooby DA, Gillespie T, Bentrem D, Nakeeb A, Schmidt MC, Merchant NB, Parikh AA, Martin RC 2nd, Scoggins CR, Ahmad S. Left-sided pancreatectomy: a multicenter comparison of laparoscopic and open approaches. Ann Surg. 2008;248:438-446. |
| 330. | Mabrut JY, Fernandez-Cruz L, Azagra JS, Bassi C, Delvaux G, Weerts J, Fabre JM, Boulez J, Baulieux J, Peix JL. Laparoscopic pancreatic resection: results of a multicenter European study of 127 patients. Surgery. 2005;137:597-605. |
| 331. | Fernández-Cruz L, Cosa R, Blanco L, Levi S, López-Boado MA, Navarro S. Curative laparoscopic resection for pancreatic neoplasms: a critical analysis from a single institution. J Gastrointest Surg. 2007;11:1607-1621; discussion 1621-1622. |
| 332. | Briggs CD, Mann CD, Irving GR, Neal CP, Peterson M, Cameron IC, Berry DP. Systematic review of minimally invasive pancreatic resection. J Gastrointest Surg. 2009;13:1129-1137. |
| 333. | Baker MS, Bentrem DJ, Ujiki MB, Stocker S, Talamonti MS. A prospective single institution comparison of peri-operative outcomes for laparoscopic and open distal pancreatectomy. Surgery. 2009;146:635-643; discussion 643-645. |
| 334. | Sohn TA, Campbell KA, Pitt HA, Sauter PK, Coleman JA, Lillemo KD, Yeo CJ, Cameron JL. Quality of life and long-term survival after surgery for chronic pancreatitis. J Gastrointest Surg. 2000;4:355-364; discussion 364-365. |
| 335. | Reddy S, Wolfgang CL, Cameron JL, Eckhauser F, Choti MA, Schulick RD, Edil BH, Pawlik TM. Total pancreatectomy for pancreatic adenocarcinoma: evaluation of morbidity and long-term survival. Ann Surg. 2009;250:282-287. |
| 337. | Bahra M, Neuhaus P. Pancreas: Is there still a role for total pancreatectomy? Nat Rev Gastroenterol Hepatol. 2010;7:72-74. |
| 338. | Yeo CJ, Cameron JL, Sohn TA, Lillemoe KD, Pitt HA, Talamini MA, Hruban RH, Ord SE, Sauter PK, Coleman J. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg. 1997;226:248-257; discussion 257-260. |
| 339. | Pezzilli R. Diabetic control after total pancreatectomy. Dig Liver Dis. 2006;38:420-422. |
| 340. | Hartwig W, Hackert T, Hinz U, Hassenpflug M, Strobel O, Büchler MW, Werner J. Multivisceral resection for pancreatic malignancies: risk-analysis and long-term outcome. Ann Surg. 2009;250:81-87. |
| 341. | Adams RB, Allen PJ. Surgical treatment of resectable and borderline resectable pancreatic cancer: expert consensus statement by Evans et al. Ann Surg Oncol. 2009;16:1745-1750. |
| 342. | Bachellier P, Nakano H, Oussoultzoglou PD, Weber JC, Boudjema K, Wolf PD, Jaeck D. Is pancreaticoduodenectomy with mesentericoportal venous resection safe and worthwhile? Am J Surg. 2001;182:120-129. |
| 343. | Siriwardana HP, Siriwardena AK. Systematic review of outcome of synchronous portal-superior mesenteric vein resection during pancreatectomy for cancer. Br J Surg. 2006;93:662-673. |
| 344. | Leach SD, Lee JE, Charnsangavej C, Cleary KR, Lowy AM, Fenoglio CJ, Pisters PW, Evans DB. Survival following pancreaticoduodenectomy with resection of the superior mesenteric-portal vein confluence for adenocarcinoma of the pancreatic head. Br J Surg. 1998;85:611-617. |
| 345. | Al-Haddad M, Martin JK, Nguyen J, Pungpapong S, Raimondo M, Woodward T, Kim G, Noh K, Wallace MB. Vascular resection and reconstruction for pancreatic malignancy: a single center survival study. J Gastrointest Surg. 2007;11:1168-1174. |
| 346. | Amano H, Miura F, Toyota N, Wada K, Katoh K, Hayano K, Kadowaki S, Shibuya M, Maeno S, Eguchi T. Is pancreatectomy with arterial reconstruction a safe and useful procedure for locally advanced pancreatic cancer? J Hepatobiliary Pancreat Surg. 2009;16:850-857. |
| 347. | Stitzenberg KB, Watson JC, Roberts A, Kagan SA, Cohen SJ, Konski AA, Hoffman JP. Survival after pancreatectomy with major arterial resection and reconstruction. Ann Surg Oncol. 2008;15:1399-1406. |
| 348. | Hirano S, Kondo S, Hara T, Ambo Y, Tanaka E, Shichinohe T, Suzuki O, Hazama K. Distal pancreatectomy with en bloc celiac axis resection for locally advanced pancreatic body cancer: long-term results. Ann Surg. 2007;246:46-51. |
| 349. | Cubilla AL, Fortner J, Fitzgerald PJ. Lymph node involvement in carcinoma of the head of the pancreas area. Cancer. 1978;41:880-887. |
| 350. | Ishikawa O, Ohhigashi H, Sasaki Y, Kabuto T, Fukuda I, Furukawa H, Imaoka S, Iwanaga T. Practical usefulness of lymphatic and connective tissue clearance for the carcinoma of the pancreas head. Ann Surg. 1988;208:215-220. |
| 351. | Manabe T, Ohshio G, Baba N, Miyashita T, Asano N, Tamura K, Yamaki K, Nonaka A, Tobe T. Radical pancreatectomy for ductal cell carcinoma of the head of the pancreas. Cancer. 1989;64:1132-1137. |
| 352. | Kawarada Y, Yokoi H, Isaji S, Naganuma T, Tabata M, Machishi H, Das BC, Takahashi K, Murabayashi K. Modified standard pancreaticoduodenectomy for the treatment of pancreatic head cancer. Digestion. 1999;60 Suppl 1:120-125. |
| 353. | Samra JS, Gananadha S, Hugh TJ. Surgical management of carcinoma of the head of pancreas: extended lymphadenectomy or modified en bloc resection? ANZ J Surg. 2008;78:228-236. |
| 354. | Pedrazzoli S, DiCarlo V, Dionigi R, Mosca F, Pederzoli P, Pasquali C, Klöppel G, Dhaene K, Michelassi F. Standard versus extended lymphadenectomy associated with pancreatoduodenectomy in the surgical treatment of adenocarcinoma of the head of the pancreas: a multicenter, prospective, randomized study. Lymphadenectomy Study Group. Ann Surg. 1998;228:508-517. |
| 355. | Michalski CW, Kleeff J, Wente MN, Diener MK, Büchler MW, Friess H. Systematic review and meta-analysis of standard and extended lymphadenectomy in pancreaticoduodenectomy for pancreatic cancer. Br J Surg. 2007;94:265-273. |
| 356. | Lieberman MD, Kilburn H, Lindsey M, Brennan MF. Relation of perioperative deaths to hospital volume among patients undergoing pancreatic resection for malignancy. Ann Surg. 1995;222:638-645. |
| 357. | Imperato PJ, Nenner RP, Starr HA, Will TO, Rosenberg CR, Dearie MB. The effects of regionalization on clinical outcomes for a high risk surgical procedure: a study of the Whipple procedure in New York State. Am J Med Qual. 1996;11:193-197. |
| 358. | Rosemurgy AS, Bloomston M, Serafini FM, Coon B, Murr MM, Carey LC. Frequency with which surgeons undertake pancreaticoduodenectomy determines length of stay, hospital charges, and in-hospital mortality. J Gastrointest Surg. 2001;5:21-26. |
| 359. | Sosa JA, Bowman HM, Gordon TA, Bass EB, Yeo CJ, Lillemoe KD, Pitt HA, Tielsch JM, Cameron JL. Importance of hospital volume in the overall management of pancreatic cancer. Ann Surg. 1998;228:429-438. |
| 360. | Ho V, Heslin MJ. Effect of hospital volume and experience on in-hospital mortality for pancreaticoduodenectomy. Ann Surg. 2003;237:509-514. |
| 361. | Simunovic M, To T, Theriault M, Langer B. Relation between hospital surgical volume and outcome for pancreatic resection for neoplasm in a publicly funded health care system. CMAJ. 1999;160:643-648. |
| 362. | Gouma DJ, van Geenen RC, van Gulik TM, de Haan RJ, de Wit LT, Busch OR, Obertop H. Rates of complications and death after pancreaticoduodenectomy: risk factors and the impact of hospital volume. Ann Surg. 2000;232:786-795. |
| 363. | Nienhuijs SW, Rutten HJ, Luiten EJ, van Driel OJ, Reemst PH, Lemmens VE, de Hingh IH. Reduction of in-hospital mortality following regionalisation of pancreatic surgery in the south-east of the Netherlands. Eur J Surg Oncol. 2010;36:652-656. |
| 364. | Edge SB, Schmieg RE Jr, Rosenlof LK, Wilhelm MC. Pancreas cancer resection outcome in American University centers in 1989-1990. Cancer. 1993;71:3502-3508. |
| 365. | Birkmeyer JD, Finlayson SR, Tosteson AN, Sharp SM, Warshaw AL, Fisher ES. Effect of hospital volume on in-hospital mortality with pancreaticoduodenectomy. Surgery. 1999;125:250-256. |
| 366. | Bilimoria KY, Bentrem DJ, Ko CY, Tomlinson JS, Stewart AK, Winchester DP, Talamonti MS. Multimodality therapy for pancreatic cancer in the U.S. : utilization, outcomes, and the effect of hospital volume. Cancer. 2007;110:1227-1234. |
| 367. | Bilimoria KY, Bentrem DJ, Ko CY, Stewart AK, Winchester DP, Talamonti MS. National failure to operate on early stage pancreatic cancer. Ann Surg. 2007;246:173-180. |
| 368. | Sherman WH, Fine RL. Combination gemcitabine and docetaxel therapy in advanced adenocarcinoma of the pancreas. Oncology. 2001;60:316-321. |
| 369. | Moertel CG. Chemotherapy of gastrointestinal cancer. Clin Gastroenterol. 1976;5:777-793. |
| 370. | Burris HA 3rd, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR, Cripps MC, Portenoy RK, Storniolo AM, Tarassoff P. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol. 1997;15:2403-2413. |
| 371. | Fatima J, Schnelldorfer T, Barton J, Wood CM, Wiste HJ, Smyrk TC, Zhang L, Sarr MG, Nagorney DM, Farnell MB. Pancreatoduodenectomy for ductal adenocarcinoma: implications of positive margin on survival. Arch Surg. 2010;145:167-172. |
| 372. | Shimada K, Sakamoto Y, Sano T, Kosuge T. Prognostic factors after distal pancreatectomy with extended lymphadenectomy for invasive pancreatic adenocarcinoma of the body and tail. Surgery. 2006;139:288-295. |
| 373. | Raut CP, Tseng JF, Sun CC, Wang H, Wolff RA, Crane CH, Hwang R, Vauthey JN, Abdalla EK, Lee JE. Impact of resection status on pattern of failure and survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Ann Surg. 2007;246:52-60. |
| 374. | Moon HJ, Jang KT, Heo JS, Choi SH, Joh JW, Kim YI. [A result of surgical treatment for ductal adenocarcinoma of the head of the pancreas]. Korean J Gastroenterol. 2003;42:156-163. |
| 375. | Howard TJ, Krug JE, Yu J, Zyromski NJ, Schmidt CM, Jacobson LE, Madura JA, Wiebke EA, Lillemoe KD. A margin-negative R0 resection accomplished with minimal postoperative complications is the surgeon's contribution to long-term survival in pancreatic cancer. J Gastrointest Surg. 2006;10:1338-1345; discussion 1345-1346. |
| 376. | Kato K, Yamada S, Sugimoto H, Kanazumi N, Nomoto S, Takeda S, Kodera Y, Morita S, Nakao A. Prognostic factors for survival after extended pancreatectomy for pancreatic head cancer: influence of resection margin status on survival. Pancreas. 2009;38:605-612. |
| 377. | Sorg C, Schmidt J, Büchler MW, Edler L, Märten A. Examination of external validity in randomized controlled trials for adjuvant treatment of pancreatic adenocarcinoma. Pancreas. 2009;38:542-550. |
| 378. | Further evidence of effective adjuvant combined radiation and chemotherapy following curative resection of pancreatic cancer. Gastrointestinal Tumor Study Group. Cancer. 1987;59:2006-2010. |
| 379. | Klinkenbijl JH, Jeekel J, Sahmoud T, van Pel R, Couvreur ML, Veenhof CH, Arnaud JP, Gonzalez DG, de Wit LT, Hennipman A. Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann Surg. 1999;230:776-782; discussion 782-784. |
| 380. | Neoptolemos JP, Stocken DD, Friess H, Bassi C, Dunn JA, Hickey H, Beger H, Fernandez-Cruz L, Dervenis C, Lacaine F. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med. 2004;350:1200-1210. |
| 381. | Crane CH, Winter K, Regine WF, Safran H, Rich TA, Curran W, Wolff RA, Willett CG. Phase II study of bevacizumab with concurrent capecitabine and radiation followed by maintenance gemcitabine and bevacizumab for locally advanced pancreatic cancer: Radiation Therapy Oncology Group RTOG 0411. J Clin Oncol. 2009;27:4096-4102. |
| 382. | Oettle H, Post S, Neuhaus P, Gellert K, Langrehr J, Ridwelski K, Schramm H, Fahlke J, Zuelke C, Burkart C. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA. 2007;297:267-277. |
| 383. | Gillen S, Schuster T, Meyer Zum Büschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7:e1000267. |
| 384. | Shah AP, Strauss JB, Abrams RA. Review and commentary on the role of radiation therapy in the adjuvant management of pancreatic cancer. Am J Clin Oncol. 2010;33:101-106. |
| 385. | Stocken DD, Büchler MW, Dervenis C, Bassi C, Jeekel H, Klinkenbijl JH, Bakkevold KE, Takada T, Amano H, Neoptolemos JP. Meta-analysis of randomised adjuvant therapy trials for pancreatic cancer. Br J Cancer. 2005;92:1372-1381. |
| 386. | Kim R, Saif MW. Is there an optimal neoadjuvant therapy for locally advanced pancreatic cancer? JOP. 2007;8:279-288. |
| 387. | Moertel CG, Childs DS Jr, Reitemeier RJ, Colby MY Jr, Holbrook MA. Combined 5-fluorouracil and supervoltage radiation therapy of locally unresectable gastrointestinal cancer. Lancet. 1969;2:865-867. |
| 388. | Moertel CG, Frytak S, Hahn RG, O'Connell MJ, Reitemeier RJ, Rubin J, Schutt AJ, Weiland LH, Childs DS, Holbrook MA. Therapy of locally unresectable pancreatic carcinoma: a randomized comparison of high dose (6000 rads) radiation alone, moderate dose radiation (4000 rads + 5-fluorouracil), and high dose radiation + 5-fluorouracil: The Gastrointestinal Tumor Study Group. Cancer. 1981;48:1705-1710. |
| 389. | Ogawa K, Karasawa K, Ito Y, Ogawa Y, Jingu K, Onishi H, Aoki S, Wada H, Kokubo M, Etoh H. Intraoperative radiotherapy for resected pancreatic cancer: a multi-institutional retrospective analysis of 210 patients. Int J Radiat Oncol Biol Phys. 2010;77:734-742. |
| 390. | Takamori H, Hiraoka T, Kanemitsu K, Tsuji T, Tanaka H, Chikamoto A, Horino K, Beppu T, Hirota M, Baba H. Long-term outcomes of extended radical resection combined with intraoperative radiation therapy for pancreatic cancer. J Hepatobiliary Pancreat Surg. 2008;15:603-607. |
| 391. | Klautke G, Brunner TB. Radiotherapy in pancreatic cancer. Strahlenther Onkol. 2008;184:557-564. |
| 393. | Espat NJ, Brennan MF, Conlon KC. Patients with laparoscopically staged unresectable pancreatic adenocarcinoma do not require subsequent surgical biliary or gastric bypass. J Am Coll Surg. 1999;188:649-655; discussion 655-657. |
| 394. | Warshaw AL, Swanson RS. Pancreatic cancer in 1988. Possibilities and probabilities. Ann Surg. 1988;208:541-553. |
| 395. | Singh SM, Longmire WP Jr, Reber HA. Surgical palliation for pancreatic cancer. The UCLA experience. Ann Surg. 1990;212:132-139. |
| 396. | Lillemoe KD, Cameron JL, Hardacre JM, Sohn TA, Sauter PK, Coleman J, Pitt HA, Yeo CJ. Is prophylactic gastrojejunostomy indicated for unresectable periampullary cancer? A prospective randomized trial. Ann Surg. 1999;230:322-328; discussion 328-330. |
| 397. | van Hooft JE, Uitdehaag MJ, Bruno MJ, Timmer R, Siersema PD, Dijkgraaf MG, Fockens P. Efficacy and safety of the new WallFlex enteral stent in palliative treatment of malignant gastric outlet obstruction (DUOFLEX study): a prospective multicenter study. Gastrointest Endosc. 2009;69:1059-1066. |
| 398. | Kim JH, Song HY, Shin JH, Hu HT, Lee SK, Jung HY, Yook JH. Metallic stent placement in the palliative treatment of malignant gastric outlet obstructions: primary gastric carcinoma versus pancreatic carcinoma. AJR Am J Roentgenol. 2009;193:241-247. |
| 399. | Maetani I, Tada T, Ukita T, Inoue H, Sakai Y, Nagao J. Comparison of duodenal stent placement with surgical gastrojejunostomy for palliation in patients with duodenal obstructions caused by pancreaticobiliary malignancies. Endoscopy. 2004;36:73-78. |
| 400. | Maetani I, Akatsuka S, Ikeda M, Tada T, Ukita T, Nakamura Y, Nagao J, Sakai Y. Self-expandable metallic stent placement for palliation in gastric outlet obstructions caused by gastric cancer: a comparison with surgical gastrojejunostomy. J Gastroenterol. 2005;40:932-937. |
| 401. | Mehta S, Hindmarsh A, Cheong E, Cockburn J, Saada J, Tighe R, Lewis MP, Rhodes M. Prospective randomized trial of laparoscopic gastrojejunostomy versus duodenal stenting for malignant gastric outflow obstruction. Surg Endosc. 2006;20:239-242. |
| 402. | Watanapa P, Williamson RC. Surgical palliation for pancreatic cancer: developments during the past two decades. Br J Surg. 1992;79:8-20. |
| 403. | Neuberger TJ, Wade TP, Swope TJ, Virgo KS, Johnson FE. Palliative operations for pancreatic cancer in the hospitals of the U.S. Department of Veterans Affairs from 1987 to 1991. Am J Surg. 1993;166:632-636; discussion 636-637. |
| 404. | Piñol V, Castells A, Bordas JM, Real MI, Llach J, Montañà X, Feu F, Navarro S. Percutaneous self-expanding metal stents versus endoscopic polyethylene endoprostheses for treating malignant biliary obstruction: randomized clinical trial. Radiology. 2002;225:27-34. |
| 405. | Maosheng D, Ohtsuka T, Ohuchida J, Inoue K, Yokohata K, Yamaguchi K, Chijiiwa K, Tanaka M. Surgical bypass versus metallic stent for unresectable pancreatic cancer. J Hepatobiliary Pancreat Surg. 2001;8:367-373. |
| 406. | Smith AC, Dowsett JF, Russell RC, Hatfield AR, Cotton PB. Randomised trial of endoscopic stenting versus surgical bypass in malignant low bileduct obstruction. Lancet. 1994;344:1655-1660. |
| 407. | Artifon EL, Sakai P, Cunha JE, Dupont A, Filho FM, Hondo FY, Ishioka S, Raju GS. Surgery or endoscopy for palliation of biliary obstruction due to metastatic pancreatic cancer. Am J Gastroenterol. 2006;101:2031-2037. |
| 408. | Taylor MC, McLeod RS, Langer B. Biliary stenting versus bypass surgery for the palliation of malignant distal bile duct obstruction: a meta-analysis. Liver Transpl. 2000;6:302-308. |
| 409. | Knyrim K, Wagner HJ, Pausch J, Vakil N. A prospective, randomized, controlled trial of metal stents for malignant obstruction of the common bile duct. Endoscopy. 1993;25:207-212. |
| 410. | Davids PH, Groen AK, Rauws EA, Tytgat GN, Huibregtse K. Randomised trial of self-expanding metal stents versus polyethylene stents for distal malignant biliary obstruction. Lancet. 1992;340:1488-1492. |
| 411. | Nakai Y, Isayama H, Kawabe T, Tsujino T, Yoshida H, Sasaki T, Tada M, Arizumi T, Yagioka H, Kogure H. Efficacy and safety of metallic stents in patients with unresectable pancreatic cancer receiving gemcitabine. Pancreas. 2008;37:405-410. |
| 412. | Maire F, Hammel P, Ponsot P, Aubert A, O'Toole D, Hentic O, Levy P, Ruszniewski P. Long-term outcome of biliary and duodenal stents in palliative treatment of patients with unresectable adenocarcinoma of the head of pancreas. Am J Gastroenterol. 2006;101:735-742. |
| 413. | Wang T, Tian FZ, Cai ZH, Li X, Cheng T, Shi L, Cheng Q. Ultrasonic interventional analgesia in pancreatic carcinoma with chemical destruction of celiac ganglion. World J Gastroenterol. 2006;12:3288-3291. |
| 414. | Caraceni A, Portenoy RK. Pain management in patients with pancreatic carcinoma. Cancer. 1996;78:639-653. |
| 415. | Shulman M, Harris JE, Lubenow TR, Nath HA, Ivankovich AD. Comparison of epidural butamben to celiac plexus neurolytic block for the treatment of the pain of pancreatic cancer. Clin J Pain. 2000;16:304-309. |
| 416. | Eisenberg E, Carr DB, Chalmers TC. Neurolytic celiac plexus block for treatment of cancer pain: a meta-analysis. Anesth Analg. 1995;80:290-295. |
| 417. | Gunaratnam NT, Sarma AV, Norton ID, Wiersema MJ. A prospective study of EUS-guided celiac plexus neurolysis for pancreatic cancer pain. Gastrointest Endosc. 2001;54:316-324. |
| 418. | Puli SR, Reddy JB, Bechtold ML, Antillon MR, Brugge WR. EUS-guided celiac plexus neurolysis for pain due to chronic pancreatitis or pancreatic cancer pain: a meta-analysis and systematic review. Dig Dis Sci. 2009;54:2330-2337. |
| 419. | Wong GY, Schroeder DR, Carns PE, Wilson JL, Martin DP, Kinney MO, Mantilla CB, Warner DO. Effect of neurolytic celiac plexus block on pain relief, quality of life, and survival in patients with unresectable pancreatic cancer: a randomized controlled trial. JAMA. 2004;291:1092-1099. |
| 421. | Bown SG, Rogowska AZ, Whitelaw DE, Lees WR, Lovat LB, Ripley P, Jones L, Wyld P, Gillams A, Hatfield AW. Photodynamic therapy for cancer of the pancreas. Gut. 2002;50:549-557. |
| 422. | Morganti AG, Trodella L, Valentini V, Barbi S, Macchia G, Mantini G, Turriziani A, Cellini N. Pain relief with short-term irradiation in locally advanced carcinoma of the pancreas. J Palliat Care. 2003;19:258-262. |
| 423. | De Conno F, Saita L, Ripamonti C, Ventafridda V. Subcutaneous octreotide in the treatment of pain in advanced cancer patients. J Pain Symptom Manage. 1994;9:34-38. |