Published online Oct 14, 2009. doi: 10.3748/wjg.15.4799
Revised: August 18, 2009
Accepted: August 25, 2009
Published online: October 14, 2009
AIM: To observe the effects of traditional antiinflammatory medicine Lonicerae Flos (LF) on rat reflux esophagitis (RE) induced by pylorus and forestomach ligation compared with the well-known proton antioxidant, α-tocopherol.
METHODS: Rats were pretreated with three different dosages of LF (500, 250 and 125 mg/kg) orally, once a day for 14 d before pylorus and forestomach ligation. Nine hours after pylorus and forestomach ligation, changes to the stomach and esophagus lesion areas, gastric volumes, acid and pepsin outputs, antioxidant effects, esophageal lipid peroxidation, superoxide dismutase (SOD), catalase (CAT), malondialdehyde (MDA), myeloperoxidase and glutathione (GSH) levels, and collagen contents (marker of flexibility) were observed on the esophageal and fundic histopathology. The results were compared with an α-tocopherol (once orally, 1 h before operation, 30 mg/kg) treated group in which the effects on RE were already confirmed.
RESULTS: Pylorus and forestomach ligations caused marked increases of gross esophageal and gastric mucosa lesion areas, which corresponded with histopathological changes. In addition, increases of esophageal lipid peroxidation, decreases of SOD, CAT, and GSH-free radical scavengers, increases of collagen were observed. However, these pylorus and forestomach ligation induced RE were dose-dependently inhibited by treatment of 500, 250 and 125 mg/kg of LF extract, mediated by antioxidant effects. RE at 250 mg/kg showed similar effects α-tocopherol.
CONCLUSION: The results suggest that antioxidant effects of LF could attenuate the severity of RE and prevent the esophageal mucosal damage, and validate its therapeutic use in esophageal reflux disease.
- Citation: Ku SK, Seo BI, Park JH, Park GY, Seo YB, Kim JS, Lee HS, Roh SS. Effect of Lonicerae Flos extracts on reflux esophagitis with antioxidant activity. World J Gastroenterol 2009; 15(38): 4799-4805
- URL: https://www.wjgnet.com/1007-9327/full/v15/i38/4799.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.4799
| Group | Damage protecting percentages (%) | Inflammatory cells infiltration in esophagus (%) | Thickness of mucosa in esophagus (μm) | Hemorrhage depth in stomach (mm) |
| Controls | ||||
| Sham | 1.23 ± 0.13 | 0.86 ± 0.7 | 261.89 ± 23.67 | 0.003 ± 0.005 |
| RE | 12.72 ± 3.81b | 94.80 ± 4.07b | 20.81 ± 46.53b | 0.963 ± 0.343 |
| α-tocopherol | 29.15 ± 10.23bc | 72.53 ± 1.17bd | 57.12 ± 43.83b | 0.19 ± 0.11bd |
| LF extracts (mg/kg) | ||||
| 125 | 24.86 ± 4.8bd | 78.92 ± 10.42bc | 138.56 ± 48.8ad | 0.13 ± 0.08bd |
| 250 | 26.56 ± 8.8bc | 68.31 ± 10.79bd | 99.75 ± 85.6b | 0.18 ± 0.08bd |
| 500 | 46.41 ± 17.77bc | 56.11 ± 4.05bd | 118.64 ± 80.38ac | 0.25 ± 0.18bd |
| Group | MDA (nmol/g tissue) | GSH (μmol/g tissue) | SOD (U/g protein) | CAT (U/g tissue) |
| Controls | ||||
| Sham | 10.11 ± 1.21 | 1.61 ± 0.29 | 64.83 ± 6.80 | 4.95 ± 0.69 |
| RE | 16.39 ± 0.75b | 0.91 ± 0.22b | 42.67 ± 6.70b | 3.19 ± 0.56b |
| α-tocopherol | 11.92 ± 0.94ac | 1.18 ± 0.13bc | 54.08 ± 6.79bd | 4.08 ± 0.50ac |
| LF extracts (mg/kg) | ||||
| 125 | 15.05 ± 1.14b | 1.03 ± 0.15b | 49.04 ± 5.36b | 3.78 ± 0.81b |
| 250 | 12.11 ± 1.61bd | 1.19 ± 0.17bc | 53.58 ± 4.80bd | 4.04 ± 0.28ac |
| 500 | 10.54 ± 1.54d | 1.31 ± 0.20ad | 60.26 ± 5.22d | 4.41 ± 0.46d |
Lonicerae Flos (LF), also called Jinyinhua, is a widely used herb prescribed in many Chinese formulas. It has latent-heat-clearing, antipyretic, detoxicant, and antiinflammatory actions[1].
It has been prescribed to treat fever due to common cold, febrile disease, dysentery, carbuncles, and virulent swellings, in Chinese medicine. Many previous reports have shown that LF is an effective antioxidant[2,3]. Chlorogenic acid, one of the major components in LF, has been widely adopted to control the quality of LF, owing to its high content and antibiotic property. Chlorogenic acid was revealed as having an effective activity in the 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging assay and ferric reducing antioxidant power assay. In addition, 14 compounds in LF were found to possess a potential antioxidant activity. They were identified as chlorogenic acid, 1-O-caffeoylquinic acid (CQA), caffeic acid, 4-O-CQA, rutin, isoquercitrin, luteolin-7-O-glucoside, lonicerin, 4,5-O-dicaffeoylquinic acid, 3,5-O-diCQA, 1,3-O-diCQA, 3,4-O-diCQA, 1,4-O-diCQA, and luteolin[4] and the antioxidant capacity of LF extracts in water, methanol and 70% ethanol to scavenge DPPH radical and reduce Fe3+ to Fe2+ was evaluated[1].
Esophageal reflux disease has gained clinical and surgical importance over the past decade. It is one of the most common complaints, affecting approximately 10% of the population[5].
More recent investigations confirmed that the pathogenesis of esophageal reflux seems to be multifactorial and the gastric contents refluxing into the esophagus contain complex components[6,7].
Acid secreted from gastric parietal cells is a potentially damaging factor in the gastric lumen, and oxidative stress also plays an important role in depletion of the adherent mucus layer and damage the esophageal mucosa from the mechanical forces associated with digestion. Reflux of caustic gastric contents, reactive oxygen species such as superoxide radical and hydroxyl radical, and release of lysosomal enzymes, is known to directly or indirectly cause symptoms such as heartburn and nausea[8,9].
Therapy that merely suppresses gastric acid secretion does not improve the function of the lower esophageal sphincter and oxidative stress is an added important factor in the pathogenesis of reflux esophagitis (RE) in rats[8].
LF has oxidant and antiinflammatory activities[1-4,8-10]; therefore, in the present study, we determined if an extract of LF could be useful for treating RE in rats, by detecting mucosal damages in an RE rat model.
Ninety-six female SD rats (6-wk old upon receipt, SLC, Japan) were used after acclimatization for 7 d. The animals were allocated five per polycarbonate cage in a temperature (20-25°C) and humidity (40%-45%) controlled room. The light/dark cycle was 12 h/12 h, and feed and water were supplied ad libitum. The experimental protocols were carried out in accordance with internationally accepted principles for laboratory animal use and care, as stated in the US guidelines.
A sample of LF was purchased from Omniherb Co. (Daegu, South Korea) in 2008. The plant was identified by Professor Seongsoo Roh and a reference specimen (Dipsacales) was deposited in our laboratory. Plant material (200g) was extracted three times with distilled water. The extract was filtered and evaporated on a rotary evaporator (Buchi, Switzerland), dried in a freeze drier (Eyela FDU-540, Tokyo, Japan). The yield (w/w) of the extract was about 12%. α-tocopherol was purchased from Sigma (MO, USA).
RE was induced in fasted rats after 24 h under 25 mg/kg of Zoletile mixture (Vibrac, France). After anesthesia, the abdomen of the animal was opened by a median incision of about 2 cm, the transitional region between the forestomach and corpus was ligated with a silk thread (2-0), and the contiguous pylorus portion was ligated. A longitudinal cardiomyotomy of about 1 cm length across the gastro-esophageal junction was performed to enhance reflux from the stomach contents into the esophageal body. The incised regions were immediately sutured and the animals were kept in a recovery chamber before being returned to their home cages. 500, 250, and 125 mg/kg of LF were orally administered, once a day for 14 d before the operation, using distilled water as vehicle. α-tocopherol [diluted in corn oil (Sigma, MO, USA)] was administered once orally at doses of 25 mg/kg, 1h before ligation. In sham and vehicle control rats, only distilled water was orally administered, once a day, for 14 d.
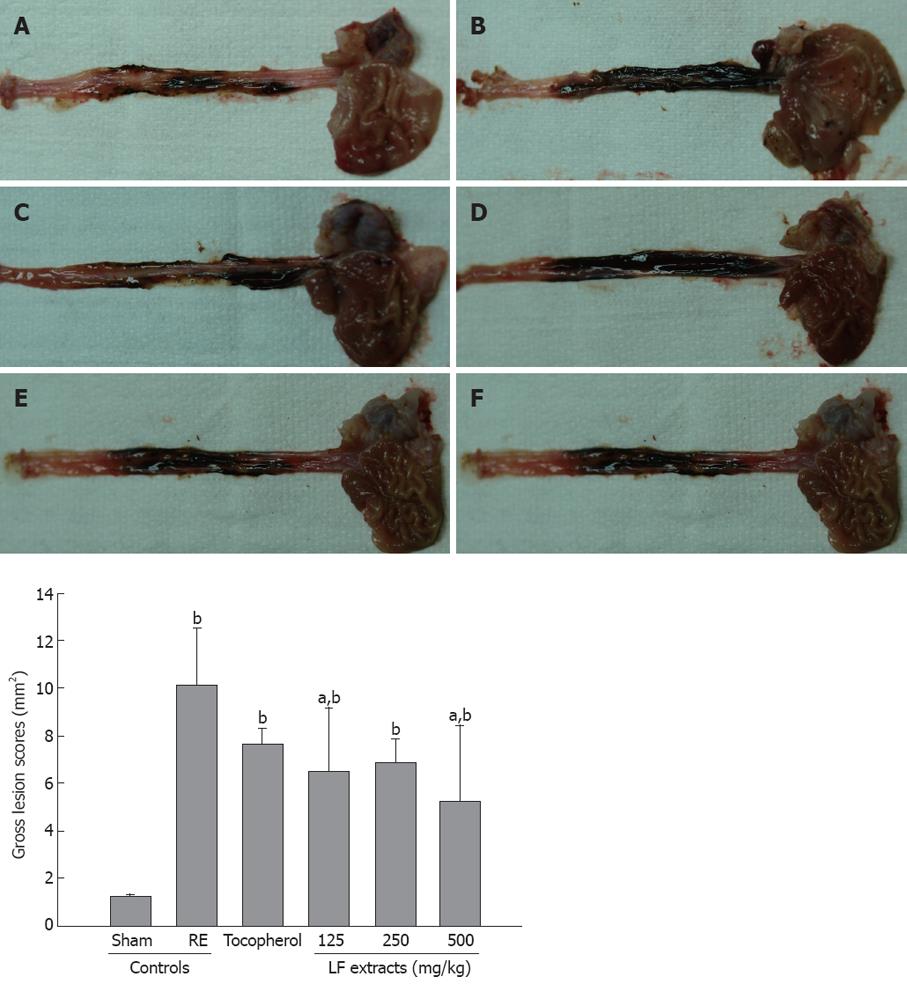
After nine hours, the animals were sacrificed and the esophagus and stomach were removed. The organs were opened along the greater curvature of the stomach, and the esophagus was longitudinally dissected out. The tissues were washed with physiological saline and examined for ulceration under a dissecting microscope (Nikon, Japan) according to a method described by Nagahama et al[11]. Photographs were taken of specified areas of damage and the width of the damaged esophagitis area (mm2) was determined and named the lesion score.
Stomach samples were homogenized in ice-cold 150 mmol/L KCl for determination of MDA and GSH levels. The MDA levels were assayed for products of lipid peroxidation[12]. Results were expressed as nmol MDA/g tissue. GSH was determined by a spectrophotometric method using Ellman’s reagent[13]. Results were expressed as μmol GSH/g tissue.
SOD was determined by the modified version from the method of Minami and Yoshikawa[14]. Briefly, 15 μL of gastric homogenate were mixed with 450 μL of cold deionized water, 125 μL of chloroform, and 250 μL of ethanol. The mixture was then, centrifuged at 8000 g for 2 min at 4°C. 500 μL of the extracts were added to a reaction mixture containing 500 μL of 72.4 mmol/L triscacodylate buffer with 3.5 mmol/L diethylene pentaacetic acid (pH 8.2; Sigma, MO, USA), 100 μL of 16% Triton X-100, and 250 μL of 0.9 mmol/L nitroblue tetrazolium (Sigma, MO, USA). The reaction mixture was incubated for 5 min at 37°C before adding 10 μL of 9 mmol/L of pyrogallol (Sigma, MO, USA) dissolved in 10 mmol/L HCl. The reaction was incubated for exactly 5 min at 37°C, before being stopped by the addition of 300 μL of 2 mol/L formic buffer (pH 3.5) containing 16% Triton X-100. The absorbance was measured at 540 nm in a spectrophotometer. One unit of SOD enzymatic activity is equal to the amount of enzyme that diminishes the initial absorbance of nitroblue tetrazolium by 50%.
CAT was determined according to the method of Rice Evans and Diplock[15]. Homogenate of rat gastric mucosa was diluted with buffer, as described before, to obtain an adequate dilution of the enzyme. Then, 2 mL of the enzyme dilution were added to a cuvette and mixed with 1 mL of 30 mmol/L H2O2, and the absorbance at 240 nm was measured for 100 s. Initial absorbance of the reaction mixture should be around 0.5. The enzyme activity (U/g of tissue) is expressed as the first order constant that describes the decomposition of H2O2 at room temperature.
The tissue samples (about 0.2 g) were homogenized in 10 volumes of ice-cold potassium phosphate buffer (50 mmol/L K2HPO4, pH 6.0; Sigma, MO, USA) containing hexadecyltrimethyl-ammonium bromide (HETAB; 0.5% w/v; Sigma, MO, USA). The homogenate was centrifuged at 12 000 g for 10 min at 4°C, and the supernatant was discarded. The pellet was then re-homogenized with an equivalent volume of 50 mmol/L K2HPO4 containing 0.5% (w/v) HETAB and 10 mmol/L EDTA (Sigma, MO, USA). MPO activity was assessed by measuring the H2O2-dependent oxidation of o-dianisidine-2 HCl. One unit (U) of enzyme activity was defined as the amount of the MPO present/g tissue weight that caused a change in absorbance of 1.0/min at 460 nm and 37°C[16].
Tissue samples were fixed in 10% formalin in 0.1 mol/L phosphate buffer (pH 7.2) in paraffin and 15 μm thick sections were obtained. Collagen content was measured according to a method described by López-De León et al[17]. The method is based on selective binding of the dyes Sirius Red (Sigma, MO, USA) and Fast Green FCF (Sigma, MO, USA) to collagen and non-collagenous components respectively. Both dyes were eluted readily and simultaneously by using 0.1 mol/L NaOH-methanol (1:1). Finally, the absorbances at 540 and 605 nm were used to determine the amount of collagen and protein respectively.
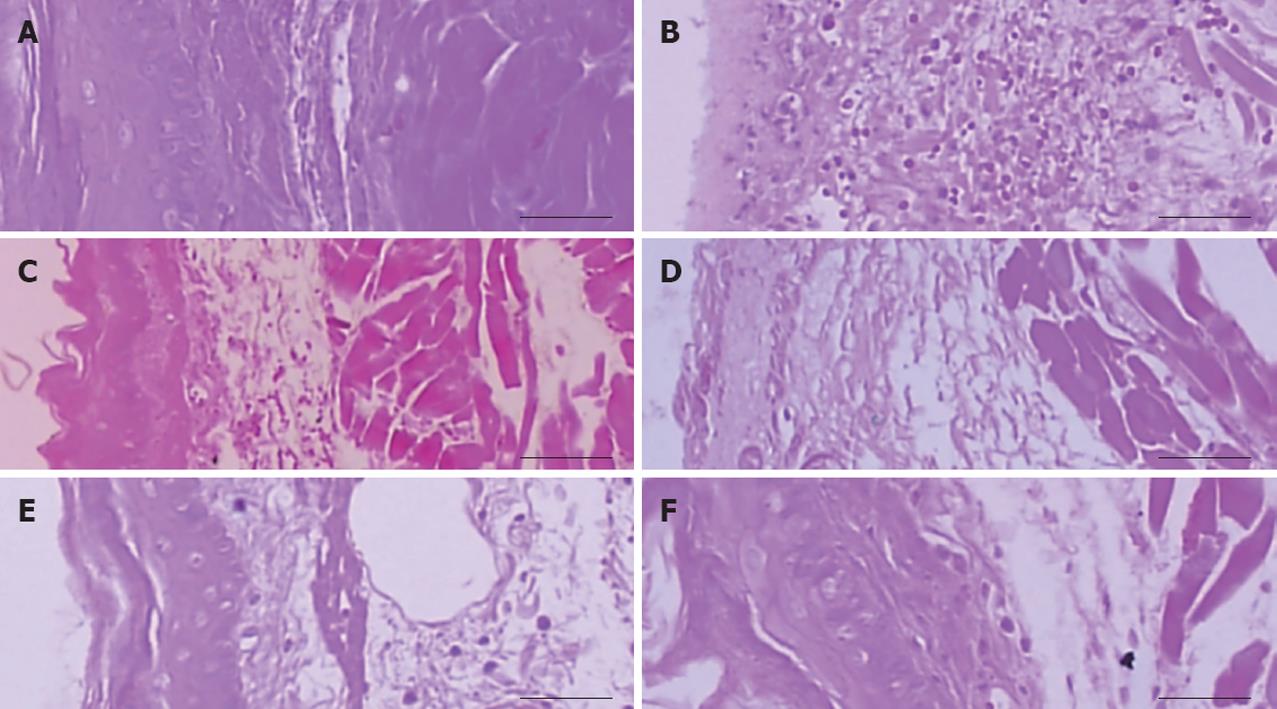
Nine hours after the operations of pylorus and forestomach ligation, the junction area from the esophagus to the cardia (about 5 cm) and a part of the fundus tissue were separated and fixed in 10% neutral buffered formalin, after paraffin embedding, 3 μm serial sections were prepared and stained with hematoxylin and eosin. Thickness of mucosa, submucosa in the esophagus, and full thickness of esophagus were measured in each prepared specimens using a CCD image analyzer (DMI-300, DMI, South Korea) as mm/crossly trimmed tissues. The invasive percentages of lesions in the fundus and percentage of mucosal damage of the esophagus were enumerated as follows: Invasive percentages of lesions (%) = (Length of lesions on the crossly trimmed esophageal or fundic walls/total thickness of crossly trimmed esophageal walls) × 100; Mucosal damage protecting percentages (%) = (Length of lesions on the crossly trimmed esophageal mucosa/total length of crossly trimmed esophageal mucosa) × 100.
Multiple comparison tests for different dose groups were conducted. Variance homogeneity was examined using the Levene test. If the Levene test indicated no significant deviations from variance homogeneity, the obtain data were analyzed by one way ANOVA test, followed by least-significant differences (LSD) multi-comparison test to determine which pairs of group comparison were significantly different. In cases where significant deviations from variance homogeneity were observed using the Levene test, a non-parametric comparison test, the Kruskal-Wallis H test, was conducted. When a significant difference was observed in the Kruskal-Wallis H test, the Mann-Whitney U-Wilcoxon Rank Sum W test was conducted to determine the specific pairs of group comparison that are significantly different. Statistical analyses were conducted using SPSS for Windows (Release 12.0K SPSS Inc., IL, USA).
A significant (P < 0.01) increase of esophageal lesion scores was detected in RE control as compared with sham control. However, these increases of esophageal lesion scores were markedly decreased by treatment with all three dosages of LF and α-tocopherol, as compared with the RE control (Figure 1).
We examined hole sizes of mucosa on histological images, measured damaged sizes of the mucosa, and calculated esophagus damage protecting percentages. Lesions on the mucosa in the sham are not shown. Lesions in the RE control, α-tocopherol, and LF groups were significantly (P < 0.01) increased compared to the sham. However, lesions in the α-tocopherol and LF groups were significantly (P < 0.05 and P < 0.01) decreased compared to the RE control. Infiltrations of inflammatory cells in esophagus tissue of the α-tocopherol and LF groups were significantly (P < 0.05 and P < 0.01) decreased compared to the RE control. In addition, the hemorrhage depth in the stomach of the α-tocopherol and LF groups was decreased compared to the RE control. Thicknesses of esophagus tissue in the RE control, α-tocopherol and LF groups were increased compared to the sham, but the thicknesses of esophagus tissue of α-tocopherol and LF groups were significantly (P < 0.05 and P < 0.01) decreased compared to the RE control. Mucosal thicknesses in the RE control, α-tocopherol and LF groups were significantly (P < 0.05 and P < 0.01) decreased compared to the sham, but mucosal thickness of the α-tocopherol and LF groups were significantly (P < 0.05 and P < 0.01) increased compared to the RE control (Table 1 and Figure 2).
Malondialdehyde (MDA) content increased significantly in the gastric mucosa after the induction of RE. α-tocopherol and all three dosages of LF, significantly (P < 0.01) inhibited MDA production as compared with the RE control (Table 2). GSH levels increased with esophageal mucosal damage. Significant differences were found between rats with gastroesophageal reflux as compared to normal esophagus. Gastric GSH contents of all experiment group were significantly (P < 0.01) lower than in normal esophageal mucosa (Table 2). However, those of the α-tocopherol and LF groups were significantly (P < 0.05, P < 0.01) higher than in RE control esophageal mucosa (Table 2). SOD content showed a significant decrease in biopsies taken from RE control group with erosive esophagitis, when compared to that of sham rats. However, of the SOD levels in the α-tocopherol and LF groups were significantly (P < 0.05, P < 0.01) higher than in the RE control group (Table 2). Gastric catalase content decreased in the RE control group compared to sham rats. However, of the catalase activities of the α-tocopherol and LF groups were significantly (P < 0.05, P < 0.01) higher than in the RE control group (Table 2).
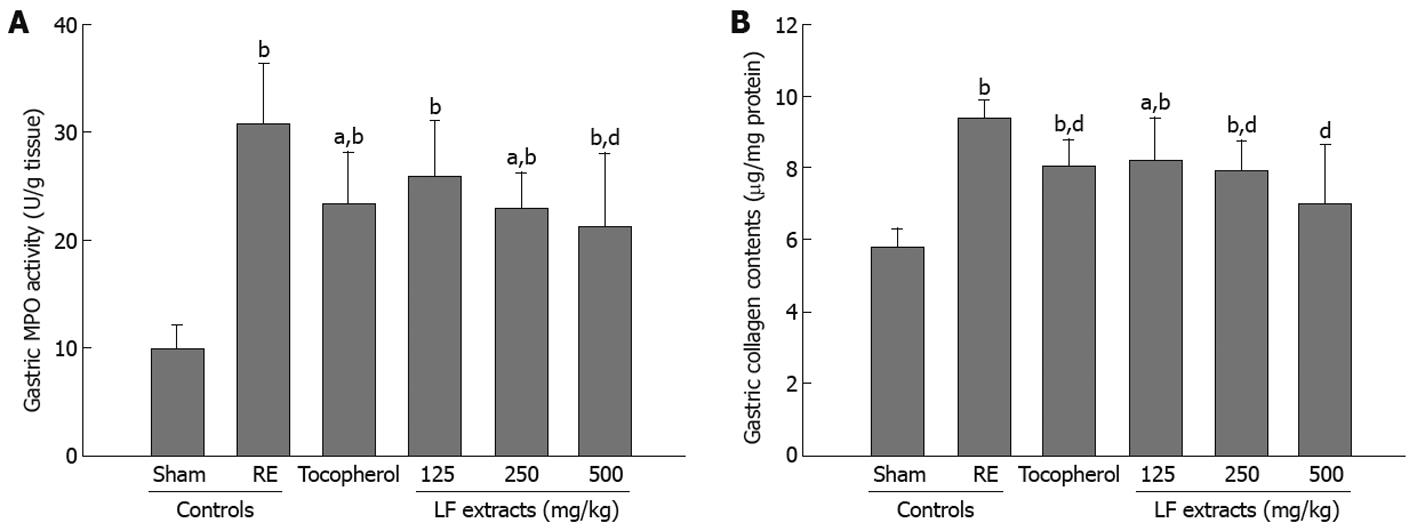
Accumulation of polymorphonucleocytes (PMNs) in the surgically induced reflux esophageal tissue is considered one of the primary contributory mechanisms to esophageal injury. The myeloperoxidase (MPO) activity in the esophageal tissue was measured as a marker for PMN accumulation (Figure 3A). The MPO activity was low in sham rats. However, the surgically induced esophagitis rats showed a large increase in esophageal MPO activity (P < 0.01). Treatment of esophagitis rats with α-tocopherol and LF significantly attenuated esophageal MPO activity in comparison with untreated rats (P < 0.05, P < 0.01), indicating that α-tocopherol and LF retarded the accumulation of PMNs in the RE model.
The collagen contents rose significantly (P < 0.01) in the RE-induced experiment groups; however, α-tocopherol and LF significantly (P < 0.05, P < 0.01) attenuated the elevated levels of collagen in esophageal tissue compared to the RE control (Figure 3B).
Many researches have reported that LF had an antioxidant effect[1,2,4,18], and the protection provided by the dose of α-tocopherol was chosen according to an earlier report[19] in which 16 mg/kg prevented oxidative stress in gastric ulcers. α-tocopherol is the most potent antioxidant that can break the propagation of the free radical chain reaction in the lipid part of the biological membrane. Chandana and Madhavan[20] reported that α-tocopherol has efficacy to esophagitis. Thus, we attempted to determine any anti-esophagitis effects of LF in a rat model of RE induced by pylorus and forestomach ligation. The RE control group showed induced esophageal inflammation, edema, and ulcer. Histology revealed increasing of thickness, damage to the mucosa, and hemorrhages in esophagus tissues. However, the experimental groups treated with α-tocopherol (30 mg/kg) and LF (at doses of 500, 250 and 125 mg/kg) showed decreased lesions of esophagitis and reduced edema, damage to the mucosa and hemorrhages in the stomach (Table 1 and Figure 1).
It has been suggested that lipid peroxidation, a sensitive marker of membrane damage caused by free radicals, is involved in reflux oesophagitis[21]. Antioxidants constitute the foremost defense system for limiting the toxicity associated with free radicals. Under physiological conditions, oxygen radicals are part of normal regulatory circuits, and the cellular redox state is very sensitive to antioxidants[22]. In recent years, there has been an upsurge in research on medicinal herbs and plant derived compounds, especially in combating oxidative stress and free radical damage[23]. GSH, SOD, and catalase are important radical superoxide scavengers that provide major protection by participating in the cellular defense systems against oxidative damage[8,9,24]. The gastric mucosa contains high levels of glutathione, which is important for maintenance of mucosal integrity, because depletion of GSH from the gastric mucosa by electrophilic compounds induces macroscopic mucosal ulceration[25]. In the present study, marked increases in lipid peroxidation were detected in the gastric mucosa of the RE control with decreases of endogenous antioxidants, GSH, SOD, and catalase, respectively (Table 2).
Many studies have focused on the role of neutrophil infiltration in the development of gastric mucosal injury[26-28]. Besides their direct damaging effects on tissues, it is well established that oxygen metabolites play a role in the recruitment of neutrophils, preferentially PMNs, into injured tissues[29]. Activated PMNs are also a potential source of oxygen metabolites[30] and MPO an activating cytotoxic enzyme released from PMNs[31]. In the present study, marked increases of gastric MPO activities were detected in the RE control compared with the intact control. We examined infiltration percentages of inflammatory cells in the esophagus and stomach. Inflammation cells infiltrations in esophageal tissue of α-tocopherol and LF-treated groups were decreased compared to the RE group (P < 0.05 and P < 0.01) (Table 1 and Figure 1). Figure 3A shows the changes to the levels of in gastric MPO.
The severity of RE impairs the strength and flexibility of collagen fibers. Collagen constitutes the major structural protein in the extracellular matrix, providing mechanical strength and structural integrity to the various connective tissues of the body[32]. In the present study, gastric collagen contents were determined as a marker of oxidant-induced fibrosis[31], and marked increases of gastric collagen contents were observed in the RE control as compared with the sham control (Figure 3B). Collagen contents rose significantly in the esophagitis-induced experiment groups; however, α-tocopherol and LF significantly attenuated the elevated levels of collagen in esophageal tissue as that of RE control (Figure 3B).
In conclusion, LF has an effective antioxidant activity and the clinical administration of LF could attenuate the severity of RE and prevent direct esophageal mucosal damage. The present study validates the potential therapeutic use of LF for gastroesophageal reflux disease.
Reflux esophagitis (RE) is occurs widely in the world and is generally treated with antacids, prokinetics, histamine type 2 receptor antagonists, and proton pump inhibitors (PPI). Especially, antioxidants having antiinflammatory activity are useful for treating RE. Lonicerae Flos (LF) might be effective for RE-induced by pylorus and forestomach ligation through its antioxidant activity.
The research field of this research is RE, which is one of the most common complaints, affecting approximately 10% of the population. An important focus of the authors’ research is that effective antioxidants in Chinese medicine have potential for use in RE.
Omeprazol is a representative PPI drug that also has antioxidant activity. As alternatives, natural medicines having antioxidant effects and no side effects could be potential remedies. This article might lead a researcher of natural products to study drug treatments for RE.
By examining which components of LF are more effective against RE, a natural new drug could be manufactured. The ability to easily obtain LF from the environment will encourage patients with RE to take folk remedies as medicines to cure RE.
In RE, the acid secreted from gastric parietal cells is a potentially damaging factor in the gastric lumen and oxidative stress also plays an important role in depletion of the adherent mucus layer. Damage to the esophageal mucosa from the mechanical forces associated with digestion might also occur. Reflux of caustic gastric contents, reactive oxygen species such as superoxide radicals and hydroxyl radical, and release of lysosomal enzymes can also cause damage.
In this article, the authors determined that the antioxidant effects of LF could attenuate the severity of RE and prevent esophageal mucosal damage. α-tocopherol is a major antioxidant, a previous report explained that it was useful in RE of pylorus and forestomach ligation. LF is an antioxidant and anti-inflammatory agent, and has effective activity on RE in the pylorus and forestomach ligation rat model.
Peer reviewer: Tomohiko Shimatani, Assistant Professor, Department of General Medicine, Hiroshima University Hospital, 1-2-3 Kasumi, Minami-ku, Hiroshima 7348551, Japan
S- Editor Tian L L- Editor Stewart GJ E- Editor Lin YP
| 1. | Lan W, Zhaojun Z, Zesheng Z. Characterization of antioxidant activity of extracts from Flos Lonicerae. Drug Dev Ind Pharm. 2007;33:841-847. |
| 2. | Choi CW, Jung HA, Kang SS, Choi JS. Antioxidant constituents and a new triterpenoid glycoside from Flos Lonicerae. Arch Pharm Res. 2007;30:1-7. |
| 3. | Liu JH, Ho SC, Lai TH, Liu TH, Chi PY, Wu RY. Protective effects of Chinese herbs on D-galactose-induced oxidative damage. Methods Find Exp Clin Pharmacol. 2003;25:447-452. |
| 4. | Tang D, Li HJ, Chen J, Guo CW, Li P. Rapid and simple method for screening of natural antioxidants from Chinese herb Flos Lonicerae Japonicae by DPPH-HPLC-DAD-TOF/MS. J Sep Sci. 2008;31:3519-3526. |
| 5. | Nasi A, de Moraes-Filho JP, Zilberstein B, Cecconello I, Gama-Rodrigues J. [Gastroesophageal reflux disease: comparison between patients with and without esophagitis, concerning age, gender and symptoms]. Arq Gastroenterol. 2001;38:109-115. |
| 6. | Malfertheiner P, Hallerbäck B. Clinical manifestations and complications of gastroesophageal reflux disease (GERD). Int J Clin Pract. 2005;59:346-355. |
| 7. | Gouvea A, Costa MS, Aldighieri FC, Oliveira MA, Pereira JC, Duarte PS. [Evaluation of the usefulness of assessing pulmonary aspiration in a gastroesophageal reflux scintigraphy study]. Rev Assoc Med Bras. 2007;53:257-260. |
| 8. | Oh TY, Lee JS, Ahn BO, Cho H, Kim WB, Kim YB, Surh YJ, Cho SW, Hahm KB. Oxidative damages are critical in pathogenesis of reflux esophagitis: implication of antioxidants in its treatment. Free Radic Biol Med. 2001;30:905-915. |
| 9. | Li Y, Wo JM, Ellis S, Ray MB, Jones W, Martin RC. A novel external esophageal perfusion model for reflux esophageal injury. Dig Dis Sci. 2006;51:527-532. |
| 10. | Sun Y, Wang Y, Guan X, Feng Y, Zhao Y. [Antimicrobial properties of Flos Lonicerae against oral pathogens]. Zhongguo Zhongyao Zazhi. 1996;21:242-243 inside backcover. |
| 11. | Nagahama K, Yamato M, Kato S, Takeuchi K. Protective effect of lafutidine, a novel H2-receptor antagonist, on reflux esophagitis in rats through capsaicin-sensitive afferent neurons. J Pharmacol Sci. 2003;93:55-61. |
| 12. | Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol. 1978;52:302-310. |
| 13. | Beutler E. Glutathione in red blood cell metabolism. A Manual of Biochemical Methods. New York: Grune & Stratton 1975; 112-114. |
| 14. | Minami M, Yoshikawa H. A simplified assay method of superoxide dismutase activity for clinical use. Clin Chim Acta. 1979;92:337-342. |
| 15. | Evans RC, Diplock AT, Laboratory techniques in biochemistry and molecular biology. In: Burtin RH, Knippenberg PH, eds. Techniques in free radical research. The Netherlands, Amsterdam: Elsevier 1991; 199-201. |
| 16. | Bradley PP, Priebat DA, Christensen RD, Rothstein G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol. 1982;78:206-209. |
| 17. | López-De León A, Rojkind M. A simple micromethod for collagen and total protein determination in formalin-fixed paraffin-embedded sections. J Histochem Cytochem. 1985;33:737-743. |
| 18. | Wu L. Effect of chlorogenic acid on antioxidant activity of Flos Lonicerae extracts. J Zhejiang Univ Sci B. 2007;8:673-679. |
| 19. | Suzuki Y, Ishihara M, Segami T, Ito M. Anti-ulcer effects of antioxidants, quercetin, alpha-tocopherol, nifedipine and tetracycline in rats. Jpn J Pharmacol. 1998;78:435-441. |
| 20. | Rao CV, Vijayakumar M. Effect of quercetin, flavonoids and alpha-tocopherol, an antioxidant vitamin, on experimental reflux oesophagitis in rats. Eur J Pharmacol. 2008;589:233-238. |
| 21. | Farhadi A, Fields J, Banan A, Keshavarzian A. Reactive oxygen species: are they involved in the pathogenesis of GERD, Barrett's esophagus, and the latter's progression toward esophageal cancer? Am J Gastroenterol. 2002;97:22-26. |
| 22. | Das D, Banerjee RK. Effect of stress on the antioxidant enzymes and gastric ulceration. Mol Cell Biochem. 1993;125:115-125. |
| 23. | Wiseman H, Halliwell B. Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem J. 1996;313:17-29. |
| 24. | Ross D. Glutathione, free radicals and chemotherapeutic agents. Mechanisms of free-radical induced toxicity and glutathione-dependent protection. Pharmacol Ther. 1988;37:231-249. |
| 25. | Maity S, Vedasiromoni JR, Ganguly DK. Role of glutathione in the antiulcer effect of hot water extract of black tea (Camellia sinensis). Jpn J Pharmacol. 1998;78:285-292. |
| 26. | Ichikawa H, Naito Y, Takagi T, Tomatsuri N, Yoshida N, Yoshikawa T. A specific peroxisome proliferator-activated receptor-gamma (PPAR-gamma) ligand, pioglitazone, ameliorates gastric mucosal damage induced by ischemia and reperfusion in rats. Redox Rep. 2002;7:343-346. |
| 27. | Jiménez MD, Martín MJ, Alarcón de la Lastra C, Bruseghini L, Esteras A, Herrerías JM, Motilva V. Role of L-arginine in ibuprofen-induced oxidative stress and neutrophil infiltration in gastric mucosa. Free Radic Res. 2004;38:903-911. |
| 28. | Sener G, Paskaloglu K, Kapucu C, Cetinel S, Contuk G, Ayanoğlu-Dülger G. Octreotide ameliorates alendronate-induced gastric injury. Peptides. 2004;25:115-121. |
| 29. | Zimmerman BJ, Grisham MB, Granger DN. Role of oxidants in ischemia/reperfusion-induced granulocyte infiltration. Am J Physiol. 1990;258:G185-G190. |
| 30. | Sullivan GW, Sarembock IJ, Linden J. The role of inflammation in vascular diseases. J Leukoc Biol. 2000;67:591-602. |
| 31. | Işeri SO, Sener G, Yüksel M, Contuk G, Cetinel S, Gedik N, Yegen BC. Ghrelin against alendronate-induced gastric damage in rats. J Endocrinol. 2005;187:399-406. |
| 32. | Hendel L. Hydroxyproline in the oesophageal mucosa of patients with progressive systemic sclerosis during omeprazole-induced healing of reflux oesophagitis. Aliment Pharmacol Ther. 1991;5:471-480. |