CASE REPORT
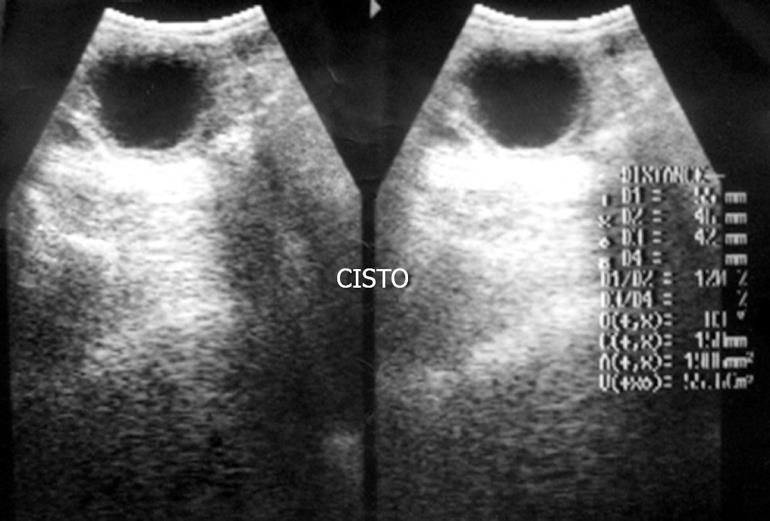
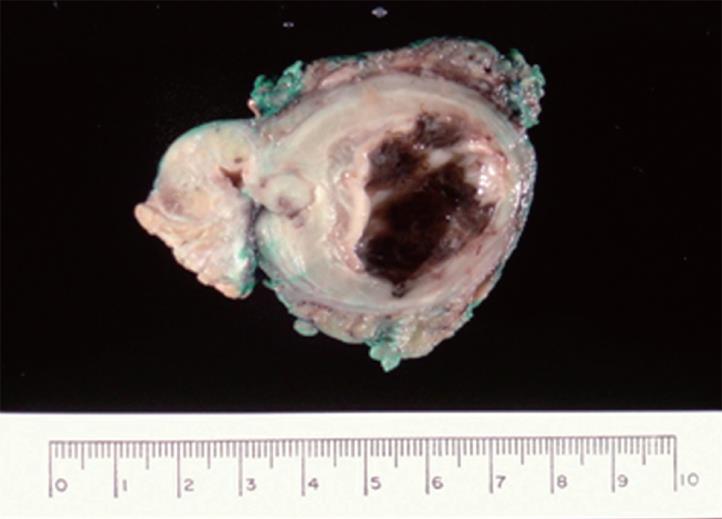
A 43-year-old woman was admitted with a 2-mo history of abdominal pain and mass. Ultrasonography (US) and computerized tomography (CT) showed a cystic mass (Figure 1) in left upper abdomen. In laboratory studies, serum amylase level was elevated at 332 U/L (normal, 0-125 U/L). The preoperative diagnosis was pancreatic tumor and fine needle aspiration biopsy was not performed preoperatively. At laparotomy, a mass was found in the mesenteric border of the transverse colon attached to the colon and pancreas. A distal pancreatectomy was performed with resection of the cyst. On gross examination, the cyst was 6 cm in diameter and contained a serous fluid (Figure 2). Microscopically, the cyst was lined by ulcerated gastric mucosa, submucosa and muscular wall (Figure 3); pancreatic tissue was normal. The patient’s pain improved following surgical resection of the mass.
Figure 1 Ultrasonography demonstrating a cystic lesion.
Figure 2 Surgical specimen including distal pancreas and the cyst.
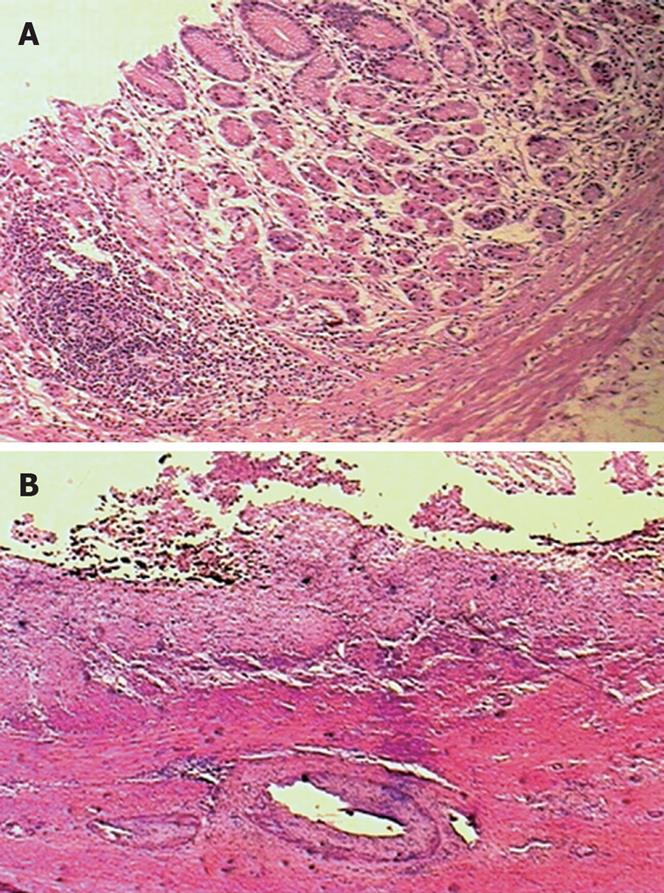
Figure 3 Histological section of the cyst wall.
A: Ectopic gastric mucosa with gastritis (HE, × 100); B: Ectopic gastric mucosa with ulceration (HE, × 25).
DISCUSSION
Duplications of the alimentary tract are characterized by attachment to at least one part of the tract, a layer of smooth muscle and epithelial lining resembling some part of the tract[2]. These malformations are formed before differentiation of the lining epithelium, and therefore, are named depending on the associated part of the bowel and not according to the mucosal lining[3]. Gastric mucosa, for example, is frequently observed in the wall of duplications, irrespective of their site of origin in the alimentary tract[4]. The duplications are located on the mesenteric border of the associated native bowel, vary in size and shape (cystic or tubular) and communicate or not with the lumen of the adjacent bowel; the duplication of cystic type usually does not communicate with the lumen of the adjacent bowel and contains serous fluid[1]. In the present case, the duplication was cystic and there was no communication with lumen of the colon.
The diagnosis of intestinal duplication may be difficult and usually is not made preoperatively. The patient may remain asymptomatic or present with a variety of nonspecific signs and symptoms[5]. Similar to our case, these signs and symptoms usually include abdominal mass and pain, often confusing colonic duplication with other diagnoses. Intestinal obstruction[6], bleeding[7] and malignant transformation[8] are all recognized complications. Ulceration, perforation and peritonitis may also occur in cases with ectopic gastric mucosa[9], which is probably the cause of ulceration in our case. In addition, various urogenital and vertebral malformations have been reported but, in the present report, no associated congenital abnormality was observed[5].
In our case, elevated serum amylase, probably owing to compression of the pancreatic duct, associated with the findings of US and CT were suggestive of pancreatic cyst, but some image findings may be helpful in establishing preoperative diagnosis of intestinal duplication. On US, the duplication is characterized by “gut signature” which refers to the hiperechoic inner layer produced by the mucosa surrounded by a hypoechoic outer layer caused by smooth muscle[10]. Peristalsis of the cyst wall has also been noted on ultrasound scan[11]. Contrast barium enema may show communication between the duplication and the normal bowel, with opacification of the two colons[12]; the communication between the duplication cyst and main pancreatic duct may be seen by endoscopic retrograde cholangiopancreatography[13]. Besides this, technetium scan may demonstrate ectopic gastric mucosa in duplication[14].
Previous reports[15–17] have described gastrointestinal duplications mimicking pancreatic tumor. Among them, a case of transverse colon duplication in a child, like in our case, was not suspected before surgery[18]. The differential diagnosis includes in these cases, all neoplastic and non-neoplastic cystic lesions of the pancreas as intraductal papillary-mucinous neoplasm and pseudocyst[19].
The recommended treatment for duplication is complete resection. Sometimes resection of normal intestine must accompany removal of the lesion because of the intimate attachment of the common wall or because isolated resection would compromise blood flow to the adjacent intestine segment[20]. Generally, duplications of the cystic type are easily removed from their attachments to the surrounding tissues[21]. During surgery, diagnosis may be made based on the frozen sections examination of the wall of the duplication[22] but, in our case, it was not performed.
On histopathological examination, ectopic gastric mucosa showed chronic inflammation, lymphoid follicles and ulceration but no H pylori was identified in haematoxilin-eosin and Warthin-Starry silver stains. Previous reports about the role of H pylori in complications of the ectopic gastric mucosa in intestinal duplication were not found. But in most cases of ectopic gastric mucosa in Meckel’s Diverticulum, H pylori also is not identified and seems that its presence is not essential to induce “heterotopic gastritis” and its complications[23–25]. In our case, the endoscopic examination was normal, and gastric biopsy was not performed.
In conclusion, we reported a case of cystic colonic duplication in an adult and illustrated the difficulties in making this rare diagnosis that should be considered in differential diagnosis of abdominal mass.