Published online Jun 21, 2008. doi: 10.3748/wjg.14.3719
Revised: May 19, 2008
Accepted: May 26, 2008
Published online: June 21, 2008
AIM: To compare the demographic and clinical features of different manometric subsets of ineffective oesophageal motility (IOM; defined as ≥ 30% wet swallows with distal contractile amplitude < 30 mmHg), and to determine whether the prevalence of gastro-oesophageal reflux differs between IOM subsets.
METHODS: Clinical characteristics of manometric subsets were determined in 100 IOM patients (73 female, median age 58 years) and compared to those of 100 age-and gender-matched patient controls with oesophageal symptoms, but normal manometry. Supine oesophageal manometry was performed with an eight-channel DentSleeve water-perfused catheter, and an ambulatory pH study assessed gastro-oesophageal reflux.
RESULTS: Patients in the IOM subset featuring a majority of low-amplitude simultaneous contractions (LASC) experienced less heartburn (prevalence 26%), but more dysphagia (57%) than those in the IOM subset featuring low-amplitude propagated contractions (LAP; heartburn 70%, dysphagia 24%; both P≤ 0.01). LASC patients also experienced less heartburn and more dysphagia than patient controls (heartburn 68%, dysphagia 11%; both P < 0.001). The prevalence of heartburn and dysphagia in IOM patients featuring a majority of non-transmitted sequences (NT) was 54% (P = 0.04 vs LASC) and 36% (P < 0.01 vs controls), respectively. No differences in age and gender distribution, chest pain prevalence, acid exposure time (AET) and symptom/reflux association existed between IOM subsets, or between subsets and controls.
CONCLUSION: IOM patients with LASC exhibit a different symptom profile to those with LAP, but do not differ in gastro-oesophageal reflux prevalence. These findings raise the possibility of different pathophysiological mechanisms in IOM subsets, which warrants further investigation.
- Citation: Haack HG, Hansen RD, Malcolm A, Kellow JE. Ineffective oesophageal motility: Manometric subsets exhibit different symptom profiles. World J Gastroenterol 2008; 14(23): 3719-3724
- URL: https://www.wjgnet.com/1007-9327/full/v14/i23/3719.htm
- DOI: https://dx.doi.org/10.3748/wjg.14.3719
The term ineffective oesophageal motility (IOM), the most common variant of oesophageal dysmotility, was introduced in 1997 to replace the term “non-specific oesophageal motility disorder”[1], as the former term appears to better reflect the functional and clinical relevance of manometric alterations[23]. IOM is defined manometrically as ≥ 30% of swallow sequences with a contractile amplitude < 30 mmHg in the distal oesophagus[4]. Such a definition, however, encompasses three abnormal contractile patterns, namely low-amplitude propagated contractions (LAP), low-amplitude simultaneous contractions (LASC), and non-transmitted contractions. IOM is, therefore, diagnosed if there is a combination of these abnormal contractile patterns.
It is not known if the predominant oesophageal symptom (heartburn, dysphagia, chest pain) experienced by IOM patients is associated with the predominance of one of these ineffective manometric patterns. In addition, although gastro-oesophageal reflux disease (GORD) has been associated with IOM[5–7], it is not known whether particular manometric subsets of IOM display a higher prevalence of GORD.
This study was thus aimed at (1) comparing the demographic and manometric features of IOM patients with different predominant symptoms (heartburn, dysphagia, chest pain); (2) comparing the demographic and clinical features of different manometric subsets of IOM; (3) comparing the clinical features of IOM subsets with those of patients with oesophageal symptoms, but normal oesophageal manometry; and (4) determining whether the prevalence of gastro-oesophageal reflux differs between the IOM manometric subsets.
A total of 100 consecutive patients (73 females, mean ± SD age 56 ± 18 years) were studied. Patients with oesophageal symptoms were referred to the Gastrointestinal Investigation Unit of the Royal North Shore Hospital. All patients fulfilled the current manometric criteria for IOM: ≥ 30% of wet swallows with low-amplitude propagated sequences (distal contractile amplitude < 30 mmHg), LASC, or non-transmitted contractions[4]. Swallow sequences were considered to be simultaneous when propagation velocity was > 8 cm/s between two or more manometric channels, and were considered to be non-transmitted when contractile amplitude was ≤ 10 mmHg at any site. A group of 100 age- and gender-matched patients (73 females, age 56 ± 20 years) referred with oesophageal symptoms, but exhibiting normal oesophageal manometry[8] served as a patient control group. Patients in the two groups had undergone upper gastrointestinal endoscopy and/or barium studies to exclude structural disease of the oesophagus, apart from the presence of reflux oesophagitis and sliding hiatus hernia. Patients with systemic diseases that could alter oesophageal motility, such as diabetes mellitus or scleroderma, were excluded. The study was approved by the Human Research Ethics Committee of the Northern Sydney Area Health Service.
A standardized symptom assessment was completed by all patients. This assessment comprised the Rome II Integrative Questionnaire[9] with an additional evaluation of oesophageal symptoms. This additional evaluation determined the predominant (i.e. most troublesome) oesophageal symptom (heartburn, dysphagia, chest pain, or others)[10] and the time period since its first appearance. The usual intensity, frequency, and duration of all reported symptoms were assessed, and symptom severity scores for heartburn, dysphagia, and chest pain were calculated as the product of these intensity, frequency, and duration data.
All patients were studied after an overnight fast. Oesophageal manometry was performed using an eight-channel DentSleeve water-perfused manometric catheter with an external diameter of 4.5 mm (DentSleeve Pty. Ltd., Belair, SA, Australia) and a computer-based data acquisition and charting system (Acquidata, Neomedix Systems, Warriewood, NSW, Australia). The catheter was introduced transnasally and swallowing was recorded via a port positioned in the pharynx, 25 cm above the proximal end of the hydraulic sleeve segment of the catheter. Oesophageal contractions were measured by ports located 5, 10, 15, and 20 cm above the proximal end of the sleeve and gastric pressure by a side hole 1 cm below the distal end of the 6 cm long sleeve. Each patient made approximately 10 swallows, each of 5 mL water of room temperature, in the seated position to acclimatize to the procedure. The patient was then placed in the supine position, and a minimum of 10 supine water swallows, each of 5 mL water, with at least 30 s in between swallows, were performed. A station pull-through technique was then used to accurately locate the position of the lower oesophageal sphincter (LOS). The LOS function was assessed by measuring mid-respiratory LOS resting pressure and swallow-induced LOS relaxation[8].
A subset of patients (54 IOM, 72 patient controls with normal manometry) underwent an ambulatory oesophageal pH study. A probe with an antimony pH sensor (Flexilog, Oakfield Instruments Ltd., Eynsham, England) was introduced transnasally and the sensor was positioned 5 cm above the upper edge of the LOS, which was determined manometrically as described above. The probe was connected to a data logger (Digitrapper Mark II, Synectics, Sweden), which sampled pH at 5-s intervals for approximately 24 h. Mealtimes, symptom events, and supine periods were recorded in a patient diary. The computerized analysis (Esophagram, Synectics, Sweden) included the percentage of time pH was below 4 (Acid Exposure Time, AET)[11], and the symptom association probability (SAP)[12] was calculated when possible.
Analysis based on predominant symptom: Propor-tions of total abnormal swallows, and proportions of LAP, LASC, and non-transmitted sequences (NT) were calculated for each of the three main predominant symptoms (heartburn, dysphagia, chest pain). Differences in proportions were then determined between symptom subgroups.
Analysis based on predominant contractile abnormality: All IOM manometric studies were further categorized into the following three subsets according to the predominant contractile abnormality contributing to the 30% or more abnormal swallows: those exhibiting a majority of LAP, those exhibiting a majority of LASC, and those exhibiting a majority of NT. Differences in clinical features between these three subsets were then determined.
Statistical analysis: Results are presented as mean ± SE, unless otherwise stated. χ2 and Fisher’s exact tests were used to determine differences in gender distribution between symptom subgroups, and differences in symptom prevalence between manometric subsets. Age differences between subsets, and between IOM patients and patient controls, were determined via Kruskal-Wallis tests with post-hoc Mann-Whitney U comparisons. Similarly, differences between symptom subgroups in proportions of contractile abnormalities and in LOS pressure and AET were determined via one-way analysis of variance (ANOVA) with post-hoc Scheffe tests. Correlation and regression analysis was used to describe relationships between variables. All analyses were performed using the SPSS statistical program (Release 14, SPSS Inc., Chicago, IL), with P < 0.05 considered significant.
Symptom, gender, and age distribution: The prevalence of predominant heartburn, predominant dysphagia, and predominant chest pain in IOM patients was 53%, 36%, and 9%, respectively. The prevalence of heartburn was significantly lower in IOM patients than in patient controls (68%, P = 0.03), and the prevalence of dysphagia was significantly higher in IOM patients than in patient controls (11%, P < 0.001).
There were no significant differences in gender and age distribution, and in time period since symptom onset, between the three symptom subgroups in IOM (Table 1).
| Gender (F:M) | Median age [yr (range)] | Median time since symptom onset (yr) | Total abnormal swallows (mean ± SD)(%) | |
| Heartburn | 37:16 | 57 (26-82) | 6.7 | 69 ± 22 |
| Dysphagia | 28:8 | 63 (19-86) | 2.5 | 68 ± 22 |
| Chest pain | 8:1 | 59 (31-76) | 6.1 | 61 ± 25 |
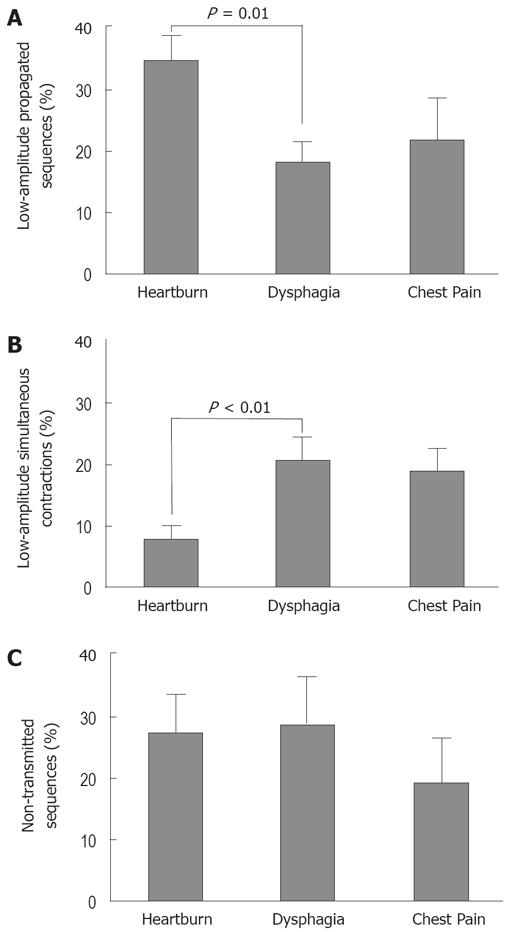
Proportions of abnormal swallows: The mean proportion of LAP was significantly higher in patients with heartburn than in those with dysphagia, and the mean proportion of LASC was significantly higher in patients with dysphagia than in those with heartburn. The mean proportion of NT was similar across the three symptom subgroups (Figure 1; ANOVA and post-hoc tests). The proportion of total abnormal swallows did not differ between the three symptom subgroups (Table 1).
When all reported symptoms were considered (i.e. predominant symptom plus additional symptoms), the relationships between symptom severity scores and proportions of abnormal sequences were examined. There were positive correlations between dysphagia severity score and proportion of LASC (r = 0.2, P = 0.05), and between chest pain severity score and proportion of LAP (r = 0.22, P < 0.05).
LOS mid-respiratory resting pressure: Mean LOS pressure was significantly higher in patients with predominant dysphagia (12.5 ± 1.2 mmHg) than in those with heartburn (9.2 ± 0.6 mmHg; P = 0.03). The mean value for chest pain patients was 11.1 ± 1.0 mmHg.
Ambulatory pH data: The gender and age distribution of the patients who underwent a pH study closely reflected that of the total subject pool, and mean LOS pressures of subgroups were almost identical to those of the total subject pool. There were no significant differences between the three subgroups in terms of AET or proportion of patients with an abnormal AET, or proportion with significant SAP values.
Gender and age distribution: The gender and age distribution for the three IOM manometric subsets and the patient controls is shown in Table 2. There were no statistical differences in gender and age distribution between patient controls and any of the IOM manometric subsets, or between IOM subsets.
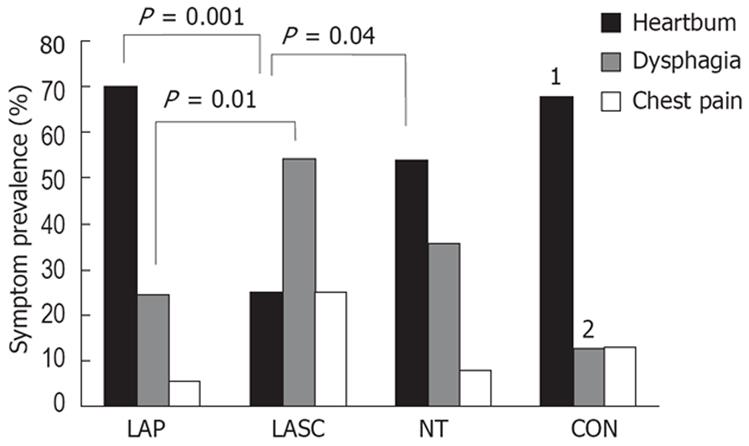
Predominant symptoms: Figure 2 shows the distribution of predominant symptoms for the three manometric subsets and for patient controls. Heartburn was highly prevalent (70%) in the LAP subset, but was significantly less common (26%) in the LASC subset. Conversely, dysphagia was more prevalent in LASC (57%) than in LAP (24%) (χ2 tests). There were no significant differences between IOM subsets, however, in severity scores for heartburn, dysphagia or chest pain, or in time since the onset of the primary symptom.
LOS mid-respiratory resting pressure: There were no statistically significant differences between the mean LOS pressures of the IOM subsets: values for LAP, LASC, and NT were 10.1 ± 0.9, 11.3 ± 1.4, and 10.9 ± 0.8 mmHg, respectively. There was a trend (P = 0.05) for the LAP subset to have a lower value than that of patient controls (12.0 ± 0.5 mmHg).
Ambulatory pH data: There were no significant differences in the prevalence of abnormal AET between IOM subsets. Analysis of mean AET values between the IOM subsets, and compared to patient controls, also revealed no significant differences (Table 2). Similarly, there were no differences in upright and supine AET values between subsets and groups. There were no significant SAP differences between the IOM subsets or in comparison to the control group (Table 2).
Despite almost 10 years of usage of the category IOM, and recent studies evaluating IOM with combined intraluminal impedance and manometry[3], the extent to which the various contractile patterns of IOM differ according to the predominant symptom has remained largely unexplored. Our study in a large patient cohort shows that IOM is indeed a heterogeneous clinical entity with regard to the predominant symptom reported. We focused on the type of contractile abnormality, rather than solely considering the contractile amplitude. The novel finding was that two specific contractile patterns, namely LASC and LAP, were closely related to predominant dysphagia and predominant heartburn, respectively. Additional analyses provided further support for these findings in that as the proportion of LASC increased, the dysphagia severity score also increased. Consistent with these findings, there were notable differences in symptom distribution between IOM manometric subsets, categorized according to the most frequent type of contractile pattern in an individual patient. Differences were particularly prominent between patients with low-amplitude propagated sequences and those with LASC; the latter experienced more dysphagia and less heartburn than the former patients.
We did not find a difference in age distribution between manometric subsets. Others have noted that simultaneous contractions are more likely to occur in older than in younger patients[1314], especially in the presence of severe GORD[15]. The effect of age in relation to the occurrence of simultaneous contractions in healthy volunteers, however, remains unclear, as one study revealed no relationship between simultaneous contractions and age[16], whereas a more recent study did find a direct correlation between the proportion of simultaneous contractions and age[17]. The most recent study revealed differences in muscle thickness between similarly-categorized IOM subsets, and a significant correlation between muscle thickness, and the occurrence of simultaneous contractions[14]. Whether muscle thickness actually plays a causative role in the pathophysiology of simultaneous contractions remains unclear, and this needs to be further evaluated. It is possible that the clinical profile of IOM patients with a majority of LASC is similar to that of patients with diffuse oesophageal spasm (DOS), a disorder that also features intermittent simultaneous (but moderate- to high-amplitude) contractions[4]. Hence, treatments currently considered for DOS patients[18–20] may prove beneficial for selected IOM patients, and this should also be further evaluated.
In the subset of IOM patients with mainly non-transmitted swallow patterns, it is feasible that this pattern could represent an evolving achalasia-like dysmotility, although these patients exhibited only a modest prevalence of dysphagia. As long-term follow-up of IOM patients has scarcely been reported[21], findings regarding disease progression remain inconclusive, and further studies are needed.
Neither the prevalence nor the severity of gastro-oesophageal acid reflux differed between IOM subsets. In addition, we found no significant differences in symptom/reflux association or heartburn severity score between IOM subsets, despite some differences in LOS resting pressure. A low LOS tone and transient LOS relaxation[22] are regarded as the main mechanisms of gastro-oesophageal reflux. As the LOS tone was lower in IOM patients with low-amplitude propagated sequences compared with patient controls, and heartburn prevalence was highest in this IOM subset, these patients could be expected to have the highest acid exposure. This was not the case, and our findings are consistent with those of Lemme et al, who found that the proportion of low-amplitude swallows did not differ between IOM patients with erosive versus non-erosive GORD[23]. It is feasible that the presence of propagating (albeit low-amplitude) sequences equates to more efficient clearance[24] of refluxate in these patients, and novel approaches to stimulating clearance[25] may prove beneficial in this IOM subset. Another explanation for the high heartburn prevalence is that these patients exhibit oesophageal hypersensitivity[26]. Despite the lack of difference in acid exposure between IOM subsets, pooled analyses of all IOM patients showed that a low LOS tone was associated with both high heartburn severity scores and high AETs. These findings indicate the complex interactions between LOS characteristics, oesophageal body dysfunction and symptomatology in IOM patients with gastro-oesophageal reflux, which require further investigation.
The current study focused primarily on oesophageal body dysmotility in IOM. LOS swallow-induced relaxation is assessed via manometry, but does not feature in the diagnosis of IOM. Conchillo et al have recently shown that, in addition to inadequate LOS relaxation, a shorter duration of LOS relaxation could contribute to abnormal bolus transit in IOM patients[27]. This might represent an additional manometric discriminator of dysmotility, and warrants further investigation.
In summary, we have examined subsets of IOM patients based on symptoms and manometrically-determined oesophageal body dysmotility. These subsets differ in regard to symptom profile, but do not differ in acid exposure or symptom/reflux association. Patients with LASC experience more dysphagia, but less heartburn, than IOM patients with low-amplitude, but propagated sequences. These findings raise the possibility of different pathophysiological mechanisms in IOM subsets, and this warrants further investigation.
The term ineffective oesophageal motility (IOM) encompasses a variety of symptoms and three types of abnormal oesophageal body peristalsis.
The pathogenesis of IOM remains unknown, particularly whether it is related to GORD or represents a primary oesophageal motor disorder.
This study raises the possibility of different pathophysiologic mechanisms in the different subsets of IOM.
Further study of the IOM subsets using novel technologies such as impedance and topographic manometry is required.
IOM refers to low-amplitude, simultaneous, or non-transmitted oesophageal body contractions.
The authors compared the demographic and clinical features of different manometric subsets of ineffective oesophageal motility and determined whether the prevalence of gastro-oesophageal reflux differs between IOM subsets. This is an interesting and well-written study.
| 1. | Leite LP, Johnston BT, Barrett J, Castell JA, Castell DO. Ineffective esophageal motility (IEM): the primary finding in patients with nonspecific esophageal motility disorder. Dig Dis Sci. 1997;42:1859-1865. |
| 2. | Kahrilas PJ, Dodds WJ, Hogan WJ. Effect of peristaltic dysfunction on esophageal volume clearance. Gastroenterology. 1988;94:73-80. |
| 3. | Tutuian R, Castell DO. Clarification of the esophageal function defect in patients with manometric ineffective esophageal motility: studies using combined impedance-manometry. Clin Gastroenterol Hepatol. 2004;2:230-236. |
| 4. | Spechler SJ, Castell DO. Classification of oesophageal motility abnormalities. Gut. 2001;49:145-151. |
| 5. | Ho SC, Chang CS, Wu CY, Chen GH. Ineffective esophageal motility is a primary motility disorder in gastroesophageal reflux disease. Dig Dis Sci. 2002;47:652-656. |
| 6. | Fouad YM, Katz PO, Hatlebakk JG, Castell DO. Ineffective esophageal motility: the most common motility abnormality in patients with GERD-associated respiratory symptoms. Am J Gastroenterol. 1999;94:1464-1467. |
| 7. | Diener U, Patti MG, Molena D, Fisichella PM, Way LW. Esophageal dysmotility and gastroesophageal reflux disease. J Gastrointest Surg. 2001;5:260-265. |
| 8. | Kahrilas PJ, Clouse RE, Hogan WJ. American Gastroenterological Association technical review on the clinical use of esophageal manometry. Gastroenterology. 1994;107:1865-1884. |
| 9. | Drossman DA, Corraziari EC, Talley NJ, Thompson WG, Whitehead WE. Rome II integrative questionnaire. Rome II: the functional gastrointestinal disorders. McLean VA: Degnon Associates 2000; 691-710. |
| 10. | Bak YT, Lorang M, Evans PR, Kellow JE, Jones MP, Smith RC. Predictive value of symptom profiles in patients with suspected oesophageal dysmotility. Scand J Gastroenterol. 1994;29:392-397. |
| 11. | Kahrilas PJ, Quigley EM. Clinical esophageal pH recording: a technical review for practice guideline development. Gastroenterology. 1996;110:1982-1996. |
| 12. | Weusten BL, Roelofs JM, Akkermans LM, Van Berge-Henegouwen GP, Smout AJ. The symptom-association probability: an improved method for symptom analysis of 24-hour esophageal pH data. Gastroenterology. 1994;107:1741-1745. |
| 13. | Ribeiro AC, Klingler PJ, Hinder RA, DeVault K. Esophageal manometry: a comparison of findings in younger and older patients. Am J Gastroenterol. 1998;93:706-710. |
| 14. | Kim JH, Rhee PL, Park EH. Evaluation of esophageal muscle thickness in patients with ineffective esophageal motility using a high-frequency intraluminal ultrasound. Gastroenterology. 2005;128:W1551. |
| 15. | Achem AC, Achem SR, Stark ME, DeVault KR. Failure of esophageal peristalsis in older patients: association with esophageal acid exposure. Am J Gastroenterol. 2003;98:35-39. |
| 16. | Richter JE, Wu WC, Johns DN, Blackwell JN, Nelson JL 3rd, Castell JA, Castell DO. Esophageal manometry in 95 healthy adult volunteers. Variability of pressures with age and frequency of "abnormal" contractions. Dig Dis Sci. 1987;32:583-592. |
| 17. | Grande L, Lacima G, Ros E, Pera M, Ascaso C, Visa J, Pera C. Deterioration of esophageal motility with age: a manometric study of 79 healthy subjects. Am J Gastroenterol. 1999;94:1795-1801. |
| 18. | Storr M, Allescher HD, Classen M. Current concepts on pathophysiology, diagnosis and treatment of diffuse oesophageal spasm. Drugs. 2001;61:579-591. |
| 19. | Storr M, Allescher HD, Rosch T, Born P, Weigert N, Classen M. Treatment of symptomatic diffuse esophageal spasm by endoscopic injections of botulinum toxin: a prospective study with long-term follow-up. Gastrointest Endosc. 2001;54:754-759. |
| 20. | Miller LS, Pullela SV, Parkman HP, Schiano TD, Cassidy MJ, Cohen S, Fisher RS. Treatment of chest pain in patients with noncardiac, nonreflux, nonachalasia spastic esophageal motor disorders using botulinum toxin injection into the gastroesophageal junction. Am J Gastroenterol. 2002;97:1640-1646. |
| 21. | Achem SR, Crittenden J, Kolts B, Burton L. Long-term clinical and manometric follow-up of patients with nonspecific esophageal motor disorders. Am J Gastroenterol. 1992;87:825-830. |
| 22. | Sifrim D, Tack J, Lerut T, Janssens J. Transient lower esophageal sphincter relaxations and esophageal body muscular contractile response in reflux esophagitis. Dig Dis Sci. 2000;45:1293-1300. |
| 23. | Lemme EM, Abrahao-Junior LJ, Manhaes Y, Shechter R, Carvalho BB, Alvariz A. Ineffective esophageal motility in gastroesophageal erosive reflux disease and in nonerosive reflux disease: are they different? J Clin Gastroenterol. 2005;39:224-227. |
| 24. | Simren M, Silny J, Holloway R, Tack J, Janssens J, Sifrim D. Relevance of ineffective oesophageal motility during oesophageal acid clearance. Gut. 2003;52:784-790. |
| 25. | Grossi L, Cappello G, Marzio L. Effect of an acute intraluminal administration of capsaicin on oesophageal motor pattern in GORD patients with ineffective oesophageal motility. Neurogastroenterol Motil. 2006;18:632-636. |
| 26. | Fass R, Tougas G. Functional heartburn: the stimulus, the pain, and the brain. Gut. 2002;51:885-892. |
| 27. | Conchillo JM, Nguyen NQ, Samsom M, Holloway RH, Smout AJ. Multichannel intraluminal impedance monitoring in the evaluation of patients with non-obstructive Dysphagia. Am J Gastroenterol. 2005;100:2624-2632. |