INTRODUCTION
Chronic gastritis is an inflammatory condition of the gastric mucosa characterized by elementary lesions whose type, extent, and distribution are related to their etiology and modulated by host responses and environmental factors[1]. Infection with H pylori, which affects an estimated three to four billion persons worldwide, is by far the most common cause of chronic active gastritis; chemical agents, autoimmune phenomena, and other infections account for a very small proportion of chronic, usually non-active gastritides. H pylori-gastritis is epidemiologically and biologically linked to the development of gastric cancer[2] and H pylori has been listed as a class I carcinogen[3]. Epidemiological and pathological data suggest that extent, intensity, and distribution patterns of gastric inflammation and atrophy are consistently related to the incidence of gastric cancer in a population[4-7]. Although odd-ratios for gastric cancer and peptic ulcer risk in relationship with the type of gastritis have been estimated, most often retrospectively, only in small series and in few populations[5,8-10], it is widely accepted that the accurate histopathological assessment of the gastric mucosa could serve as a reasonably good predictor of cancer risk in an individual patient. In fact, most recent classifications of gastritis have contained the implicit aim of providing a clinico-pathological correlation that could be both synchronous (that is, at the time of the sampling) and, more usefully, diachronic.
When appropriate sampling is available, the histo-pathological features of the gastric mucosa recognized as being part of the neoplastic process and broadly referred to as “pre-neoplastic lesions” (atrophy, pyloric and intestinal metaplasia, epithelial dysplasia) can be accurately evaluated by the microscopic examination of mucosal biopsies. Although classification systems such as the Sydney System[11], its Houston-updated version[12], and the more recent guidelines for the evaluation of atrophy[13] suggest strategies for the formulation of histopathological reports, we still lack a way to translate the pathological information into a standardized report that would convey comprehensive information on the gastric condition while lending itself to a straightforward analysis of cancer risk.
The purpose of this article is to explore ways for pathologists to maximize the predictive value of the gastric evaluation by: (1) streamlining the histopathological report of gastric biopsies, and (2) integrating relevant laboratory information with pathological data.
GASTRIC MUCOSAL CHANGES RELATED TO GASTRIC CANCER
As a result of seminal field studies conducted by Max Siurala in Finland and Estonia[14-17] and Pelayo Correa in Colombia[4,18-19], as well as the crucial body of knowledge derived from decades of Japanese studies[20,21], the separate entities of chronic superficial gastritis, atrophy, metaplasia, dysplasia and carcinoma were integrated into a hypothetical sequence known as Correa’s cascade[22]. Increasingly well-documented by patho-epidemiological studies, the 1984 multi-step hypothesis of gastric carcinogenesis still lacked an etiological initiator. The missing first step was discovered in the same year[23] and H pylori found its place at the top of the cascade[24].
The histopathological lesions broadly regarded as preneoplastic are chronic gastritis, atrophy, intestinal metaplasia, dysplasia, and neoplasia. Their evolution in a cohort can be viewed as a pyramid with a very large base representing the entire H pylori-infected population; a segment of these subjects (generally larger in developing than in industrialized areas) will progress to atrophic gastritis, mostly accompanied by intestinal metaplasia. A very small minority will progress further to dysplasia with some eventually developing adenocarcinoma. The closer a lesion is to neoplasia, the more likely it will progress into it. Thus, whereas chronic gastritis is a remote and uncertain precursor of gastric cancer that could be better called a “predisposing condition,” high-grade dysplasia is considered already a neoplastic lesion[25,26]. If pathologists could make a reliable assessment of the risk that each patient has, based on a staging of the disease, effective strategies could be developed to detect the early, curable phase of gastric cancer and prevent its progression.
Chronic gastritis
The risk of gastric cancer for a patient with simple, non-atrophic H pylori gastritis is negligible, thus, the decision to treat the infection is based, in most cases, on other considerations. There is, however, one exception. Gastric cancer and atrophic gastritis associated with it have at least some familial predisposition[27-30]; therefore, it would seem wise to treat H pylori infection as early as possible in direct relatives of patients with gastric cancer. This is one of the rare circumstances in which H pylori would be eradicated for the specific purpose of preventing gastric carcinoma in an individual patient.
Atrophy
Gastric atrophy is defined as the loss of appropriate glands in a given gastric compartment[13,31]. This purely histopathological definition indicates that the glands expected to be present in the portion of gastric mucosa under examination (for example, oxyntic glands in the mucosa of the corpus) are no longer there, and have been replaced by something else that does not belong to that area. This “something else” may be extracellular matrix, fibroblasts and collagen, or other glands that normally are not there (e.g., intestinal-type glands or pseudo-pyloric glands). Any of these replacements prevents that portion of gastric mucosa from performing its normal functions (e.g., to secrete acid). Thus, the functional correlate of atrophy is strictly related to its extension.
Atrophic gastritis is a condition characterized by the presence of significant areas of atrophy. Its two most common underlying causes are chronic infection with H pylori and the autoimmune gastritis that may become associated with pernicious anemia. In the Updated Sydney System, the term “atrophic gastritis” is used in contrast to “non-atrophic gastritis” or simply “gastritis,” a condition usually more severe in the antrum (hence the term “antral-predominant”) found in most subjects infected with H pylori in the Western industrialized world.
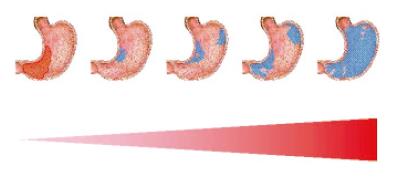
The stomach affected by atrophic gastritis shows a decrease or absence of appropriate glands, an expansion of the antral-type mucosa into the corpus (“antralization” or pseudo-pyloric metaplasia) and usually extensive areas of intestinal metaplasia. This condition has been known for several decades to represent a significant epidemiological risk factor for gastric adenocarcinoma[14,17,24,32-36]; as schematically depicted in Figure 1, its prognostic implications in the individual patient seem to be related to the extent and distribution of the atrophic areas[10,37].
Figure 1 Schematic representation of the progression of atrophy, from absent in the case of antrum-predominant non-atrophic gastritis depicted on the left to the almost generalized metaplastic atrophy depicted on the right.
The increased extension of atrophy corresponds to an increased cancer risk, indicated as an expanding triangle. The extension of atrophy can also be reported as a stage from 0 to 4.
Intestinal metaplasia
Intestinal metaplasia is the replacement of the normal gastric mucosa with an epithelium similar to that of the intestine. Attempts to classify the different types of intestinal metaplasia have resulted in a confusing terminology (complete vs incomplete, type 1, 2a and 2b, etc.); the classification currently used was proposed by Jass and Filipe[38,39]: Type I (brush border and no sulfomucins); Type II (no brush border, rare sulfomucins); and Type III (no brush border, cellular disarray, abundant sulfomucins). Type I intestinal metaplasia has been often said to pose little increased risk of developing carcinoma, whereas type III has been considered as an already dysplastic lesion[10,40-42]. The classification of the three types of metaplasia requires relatively sophisticated histochemical techniques and is far from being standardized. Furthermore, the data suggesting different cancer risks for the different types of intestinal metaplasia are not unequivocal[43]. Therefore, immunohistochemical sub-typing of intestinal metaplasia should be limited to the clinical research setting and not a part of the routine evaluation of patients with intestinal metaplasia.
Dysplasia
Malignancy is the final step of progressive genetic and phenotypic changes that modify the original cellular morphology, eventually generating a biologically new cell characterized by uncontrolled growth and the potential to migrate and implant in locations beyond its original fixed site. This biological process has been called multi-step or step-wise oncogenesis. In epithelial tissues (for example, the squamous lining of the uterine cervix or the columnar lining of the colon) the first of step visible to an observer using a light microscope is a change in the morphology of the cells that form the epithelium. Nuclei are larger, nucleoli may be prominent and the chromatin may be clumped or granular; compared to the larger nucleus, the cytoplasm appear smaller, a phenomenon referred to as “increased nucleo-cytoplasmic ratio”. Various degrees of disarray of the orderly structure of the normal epithelium usually accompanying these changes. Epithelial alterations of this kind occur in two situations: when the epithelium has been injured and is undergoing repair, and when genetic alterations have transformed the cells in a neoplastic growth. It is generally agreed by pathologists that in the former instance one refers to the phenomenon as “regenerative atypia”, whereas in the latter case the term “dysplasia” is used[44].
The importance of recognizing and correctly identifying dysplasia is self-evident: while regenerative atypia is the desired response to epithelial injury and an essential part of an organism’s homeostasis, dysplasia is the harbinger of cancer and requires immediate action. However, the morphological differences between atypia and dysplasia are not always apparent, and significant areas of phenotypic overlap exist between the two. Pathologists have tried for years to standardize the criteria for the diagnosis and grading of dysplasia in tissues accessible to biopsy sampling. Without getting into the complex historical details of the process, for the purpose of this review we say only that, through the efforts of pioneers such as the late Rodger Haggitt, Robert Riddell, Brian Reid, and others, a satisfactory level of agreement has been reached for dysplasia of the colon and of Barrett’s epithelium[44-46]. Gastric dysplasia has received less attention in the past, with only one major consensus article addressing the issue before 1996[47].
In the last decade, the discovery of H pylori and its relationship with gastric cancer has stimulated increasing attention to the preneoplastic lesions of the stomach. The possibility that curing this infection could prevent or even cause the regression of such lesions has highlighted the need for uniform and rigorous definitions and diagnostic criteria. However, unlike metaplasia, whose recognition has always been largely free of major disputes, or atrophy, which has been the focus of major conceptual disagreements among pathologists, dysplasia exposed a novel angle of controversy: a pathological schism between East and West, or, more accurately, between Japan and the West[48].
Japan is one of the countries with the highest incidence of adenocarcinoma of the stomach in the world; at the same time, it also has the world’s best survival rates for gastric cancer. Although the effective early detection programs, innovative endoscopic techniques, and daring and successful therapeutic endoscopists have been invoked to explain the Japanese success in this area, another explanation has been suggested, mostly in a veiled or oblique manner. To state it simply, it has been implied that, to have such good survival rates the Japanese must call cancer what others call dysplasia. The question has been propelled into the international scientific forum only recently, through the efforts of RJ Schlemper, who in 1996 organized a workshop to address the issue. This workshop resulted in a seminal paper entitled “Differences in diagnostic criteria for gastric carcinoma between Japanese and Western pathologists,” published in the Lancet in 1997[49]. Following the workshop and publication of its findings, several other groups have formed to tackle the problem in the traditional pathologists’ fashion: by trying to measure the level of agreement (or disagreement) amongst observers. These groups included various proportions of Japanese and Western pathologists, and the ultimate aim was to reach a consensus that classification, if used globally, would allow comparative studies performed in different countries. As a result, new issues have emerged and new classifications have been proposed. The classification currently accepted by the World Health Organization[50] is largely modeled on the consensus agreement reached in Padua, Italy, in 1998[26], and summarizes one of the most recent proposals for an integrated therapeutic and pathological approach[51].
The Padova model is based: (1) on the definition of dysplasia as pre-invasive neoplasia; and (2) on a five-category classification of gastric neoplasia which includes: 1, negative for dysplasia; 2, indefinite for dysplasia; 3, non-invasive neoplasia; 4, suspicious for invasive cancer; 5, gastric cancer. The numerical prefix assigned to each diagnostic category essentially corresponds to the diagnostic categories of the Japanese Classification for Gastric Cancer[52]. Within each category one or more sub-categories are hierarchically ordered to cover the spectrum of epithelial alterations.
THE IMPORTANCE OF GOOD SAMPLING
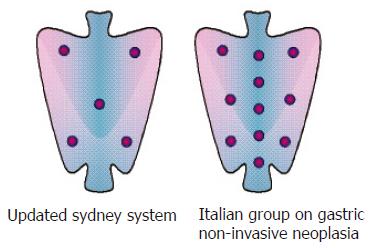
The topographic distribution of inflammatory infiltrates, lymphoid follicles, atrophy, and metaplasia is an essential determinant used for all classifications of gastritis. These changes may be patchily distributed and their relative intensity in different parts of the stomach may be highly variable. Furthermore, the inflammatory and atrophic processes have different phenotypical expressions in different regions of the stomach. Therefore, to obtain an accurate picture of gastritis, pathologists must have a set of specimens representative of each gastric compartment. Each specimen is examined according to uniform criteria, a general impression of the intensity of the features of gastritis is extrapolated from the various specimens from each compartment, and finally this information is amalgamated in a topographical diagnosis. The location of the biopsy specimens recommended by the Updated Sydney System[12] is depicted in the left panel of Figure 2. A suggestion has been made to replace the original sites with others, purportedly more likely to yield information about the extension of intestinal metaplasia[53], but in the absence of independent testing no proposal in this sense has been presented.
Figure 2 Two different biopsy protocols.
Irrespective of the protocol used, gastroenterologists must keep in mind that the predictive information they can get from their pathologist is only as good as the biopsy sampling submitted for examination. The Sydney System 5-biopsy protocol is a compromise between what is practically doable in routine practice and the ideal need for maximal topographic information. As depicted in Figure 2, right panel, in special situations such as the diagnosis and follow-up of gastric mucosa-associated lymphoid tissue B-cell lymphomas or the diachronic investigation of dysplasia much more extensive sampling protocols need to be applied[48,54-56].
VIRTUAL HISTOPATHOLOGY
The determination of serum pepsinogens I (PG I) and II(PGII), gastrin-17 (G-17) and IgG anti-H pylori antibodies by ELISA has been proposed as an array of non-invasive markers for the assessment of both morphological and functional status of the gastric mucosa[57]. The rationale for this approach, described by its enthusiastic supporters as the “serological biopsy,” rests on the fact that PGI is exclusively secreted by oxyntic glands and represents an excellent marker of the secretory ability of the gastric corpus. In contrast, PG II is produced by all types of gastric and duodenal glands and its production is influenced by gastric inflammation[58,59]. Although these molecules are secreted into the gastric lumen, small amounts seep out into the bloodstream and can be measured. Gastrin-17 (G-17), produced in the antrum and secreted directly into the blood, is a specific marker of G cell function[60]. Several studies have now shown that serum levels of PG I, PG II and G-17 are high in subjects with H pylori non-atrophic chronic gastritis[61]. Both PG I and PGII concentrations are found to decrease significantly two months after the eradication of H pylori[62,63]. Furthermore, the ratio of PGI and G-17 levels have been found to correlate well with the histopathological diagnosis of atrophic body gastritis and, in some studies, to be associated with the presence of gastric cancer[64-66].
In a recent study, De Mario and his colleagues[67] demonstrated that the analysis of serum pepsinogens, G-17 and anti-H pylori IgG levels provide consistent and reproducible information regarding gastric atrophy and its association with H pylori. The authors suggest that dyspeptic patients with normal PG I, PGII, G-17 and a negative serological test for H pylori can be reassured that they are unlikely to have peptic ulcer disease and can be treated symptomatically. In contrast, patients with panel test results indicating H pylori-related chronic gastritis, with or without atrophy, could either be treated for H pylori or referred for endoscopy, depending on the type and severity of their manifestations.
GENERATING A CLINICALLY USEFUL HISTOPATHOLOGY REPORT
The article reporting the Updated Sydney System, published in October 1996, has recently passed the 1000-citation milestone[68], suggesting that the semi-quantitative scoring system it advocated remains a useful tool for clinical research. Nevertheless, the same pathologists who use it when assessing biopsies for clinical studies find it too cumbersome to use in their routine diagnostic activities.
Using the framework provided by the Sydney System’s and the Atrophy Club’s analytic approach, we have recently put forward a proposal for a grading and staging scheme that integrates the relevant histopathological data gathered and interpreted by the pathologist and delivers them in the form of a simple, yet information-rich report[69]. We have suggested that the method is both feasible and practical, and that staging and grading (preceded by a description of the histological findings in the biopsy samples) could represent the concluding message of the histological report. This scheme could be do for chronic gastritis what the grading and staging system introduced by the International Group of Hepatologists in 1995 did for chronic hepatitis: make prognostically significant and reproducible information immediately available in the clinical practice[70,71].
Briefly, the proposal consists of summarizing the combined intensity of mononuclear and scoring granulocytic inflammation in both antral and oxyntic biopsy samples in a grade from 0 (no inflammation) to 4 (a very dense infiltrate in all the biopsy samples). The extent of atrophy, with or without intestinal metaplasia, would be reported as a stage from 0 (no atrophy) to 4 (pan-atrophy involving all antral and oxyntic samples). The latter would convey information on the anatomical extent of the atrophic-metaplastic changes related to cancer risk. Figure 1 shows the progression from stage 0 (left) to stage 4 (right).
A pathologist who had access to the results of the “serological biopsy” and applied the grading and staging principles outlined in this scheme could generate a comprehensive informative integrated report that could be used by clinicians as a solid base for the management of patients with gastric conditions.
This proposal has been discussed at an international consensus group of gastroenterologists and pathologists (Operative Link for Gastritis Assessment-OLGA) that gathered in Parma, Italy, in April 2005. The group included Massimo Rugge, Padova, Italy; Pelayo Correa, New Orleans, Louisiana, USA; Francesco Di Mario, Parma, Italy; Emad El-Omar, Aberdeen, Scotland, UK; Roberto Fiocca, Genova, Italy; Karel Geboes, Leuven, Belgium; David Y Graham, Houston, Texas, USA; Takanori Hattori, Shiga, Japan; Peter Malfertheiner, Magdeburg, Germany; Pentti Sipponen, Espoo, Finland; Joseph Sung, Hong Kong, China; Wilfred Weinstein, Los Angeles, California, USA; Michael Vieth, Bayreuth, Germany; and Robert M Genta, Geneva, Switzerland.
After deliberations that led to a number of modifi-cations of the original proposal, the OLGA group has agreed that an international staging method is needed to advance research in gastritis and is preparing to test its feasibility and reproducibility both in retrospective and prospective multi-center studies.
S- Editor Wang J L- Editor Ma JY E- Editor Bai SH