Published online Mar 21, 2006. doi: 10.3748/wjg.v12.i11.1770
Revised: July 25, 2005
Accepted: August 20, 2005
Published online: March 21, 2006
AIM: To study the effects of drug treatment on hyperplastic gastric polyps infected with Helicobacter pylori (H pylori).
METHODS: Forty-eight patients with hyperplastic gastric polyps (3-10 mm in diameter) infected with H pylori were randomly assigned to a treatment group (n = 24) which received proton-pump inhibitor (omeprazole or lansoprazole), clarithromycin, bismuth citrate and tinidazole, and a control group (n = 24) which received protective agent of gastric mucosa(tepretone). Patients underwent endoscopy and H pylori examination regularly before enrollment and 1-12 mo after treatment.
RESULTS: Twenty-two patients in the treatment group and 21 in the control group completed the entire test protocol. In the treatment group, polyps disappeared 1-12 mo (average, 6.5 ± 1.1 mo) after the treatment in 15 of 22 patients (68.2%) and H pylori infection was eradicated in 19 of the 22 patients (86.4%). However, 12 months after the study, no change in polyps or H pylori status was seen in any controls (bP < 0.01).
CONCLUSION: Most hyperplastic gastric polyps disappear after eradication of H pylori.
- Citation: Ji F, Wang ZW, Ning JW, Wang QY, Chen JY, Li YM. Effect of drug treatment on hyperplastic gastric polyps infected with Helicobacter pylori: A randomized, controlled trial. World J Gastroenterol 2006; 12(11): 1770-1773
- URL: https://www.wjgnet.com/1007-9327/full/v12/i11/1770.htm
- DOI: https://dx.doi.org/10.3748/wjg.v12.i11.1770
The risk of conversion of hyperplastic gastric polyps is very low, only accounting for 1.5%-3% [1]. Patients with gastric polyps may present with bleeding of the upper gastrointestinal tract, abdominal pain, or gastric outlet obstruction. Large gastric polyps or polyps associated with complications can be removed endoscopically or surgically. Helicobacter pylori (H pylori) infection is closely associated with hyperplastic gastric polyps and H pylori is present in 100% of hyperplastic gastric polyps[2-4]. Hyperplastic gastric polyps may disappear in 40-71% patients after eradication of H pylori [5,6]. Since the pathogenicity of H pylori is different in various regions, we conducted this randomized, controlled trial to see whether hyperplastic gastric polyps disappear after eradication of H pylori.
Forty-eight patients were from the Gastroenterology Department, First Affiliated Hospital of Zhejiang University and Red Cross Hospital of Hangzhou (26 men, 22 women, age range from 21 to 73 years, average 47 years). All patients did not receive antibiotics, bismuth, steroid and non-steroid drugs before their enrollment in the study. Hyperplastic gastric polyps (3-10 mm in diameter) were diagnosed on the basis of the examination results at least three histological samples. Patients were considered to have multiple gastric polyps (at least 5) when they were easily cut. Our criteria for hyperplastic gastric polyps included hyperplasia of the foveolar epithelium on histologic examination and infiltration of inflammatory cells into the stroma in biopsy specimens [7,8]. Hyperplastic gastric polyps were diagnosed by two blinded pathologists. Patients with adenomatous gastric polyps, Peutz-Jegher syndrome and juvenile polyps were excluded. The diagnosis of H pylori infection was based the positive results of staining with Giemsa and 14C-urea breath test. The patients were randomly assigned to two groups and sequentially numbered. In the treatment group (n = 24), patients received proton-pump inhibitor (omeprazole 20mg/d or lansoprazole 30 mg/d), clarithromycin (1g/d), bismuth citrate (440 mg/d) and tinidazole (1g/d), 2 weeks a course. In the control group (n = 24), patients had endoscopic examination and received protective agent of gastric mucosa (tepretone 150 mg/d). Two patients of the treatment group did not take the drugs for one course. In the control group, 2 patients lost follow-up and 1 patient took both proton-pump inhibitor and amoxicillin. These 5 patients exited from our study. If polyps progressed and were accompanied with malignant transformation, the study was stopped and the polyps were removed endoscopically. After completion of our study, endoscopic removal of polyps or eradication of H pylori was proposed for those who failed in H pylori eradication.
Patients in the treatment group underwent endoscopy every 3 months after the treatment. On each occasion, biopsy specimens were taken from the same areas (three from the antrum and three from the body) for histologic examination. If patients without eradication of H pylori were not adapted to endoscopic examination very well, controls underwent endoscopy every 3 months after enrollment. Biopsy specimens for histologic examination were stained with Giemsa and evaluated for the presence of H pylori. Histologic diagnosis of the biopsied mucosa of the antrum and body was made by two blinded pathologists. The severity of activity, inflammation, atrophy, and metaplasia was graded on a scale from 1 to 4 and expressed as the histologic index according to the updated Sydney System: 1: normal, 2: mild, 3: moderate, and 4: marked [9]. Eradication of H pylori was confirmed by the negative results of these two tests 1-3 months after the treatment and endoscopic examination. The size and number of polyps were measured at each endoscopic examination using biopsy forceps (GIF XQ240 or GIF140, Olympus) placed near the polyp (open size: 6 mm in diameter; closed size: 2 mm in diameter). The endoscopic data on the disappearance and regression of polyps were reviewed independently by two blinded endoscopists.
All data were analyzed by unpaired t test (for age), Wilcoxon rank-sum test and Fisher’s exact test. P < 0.05 was considered statistically significant. All data were coordinated by SPSS RDS and statistical analyses were done by SPSS software.
Twenty-two patients in the treatment group and 21 patients in the control group completed the entire study. The two groups were similar with respect to the number, age, sex, coexisting disease, as well as the number, size and distribution of polyps, histologic findings (Table 1). The two groups were comparable.
| Characteristics | Treatment groups(n = 22) | Control groups(n = 21) |
| Mean age(mean±SD,yr) | 49 ± 9 | 47 ± 8 |
| Men, n(%) | 13(59.1) | 11(52.4) |
| Coexisting disease, n(%) | ||
| Chronic atrophic gastritis | 10(45.4) | 12(57.1) |
| Duodenal ulcer | 2(9.1) | 2(9.5) |
| Gastric ulcer | 0(0) | 1(4.8) |
| Mean number of polyps | 5.6 | 4.2 |
| Mean size of polyps(mm) | 6.6 | 7.8 |
| Distribution of polyps, n (%) | ||
| Body | 79(64.2) | 61(69.3) |
| Antrum | 19(15.4) | 15(17.0) |
| Angle | 8(6.5) | 5(5.7) |
| Fundus | 9(7.3) | 4(4.5) |
| Cardia | 8(6.5) | 3(3.4) |
| Histologic findings | ||
| Inflammation | 2.7, 2.4 | 2.6, 2.4 |
| Activity | 2.5, 2.5 | 2.5, 2.3 |
| Atrophy | 2.4, 2.6 | 2.5, 2.4 |
| Metaplasia | 0.9, 0.6 | 1.0, 0.6 |
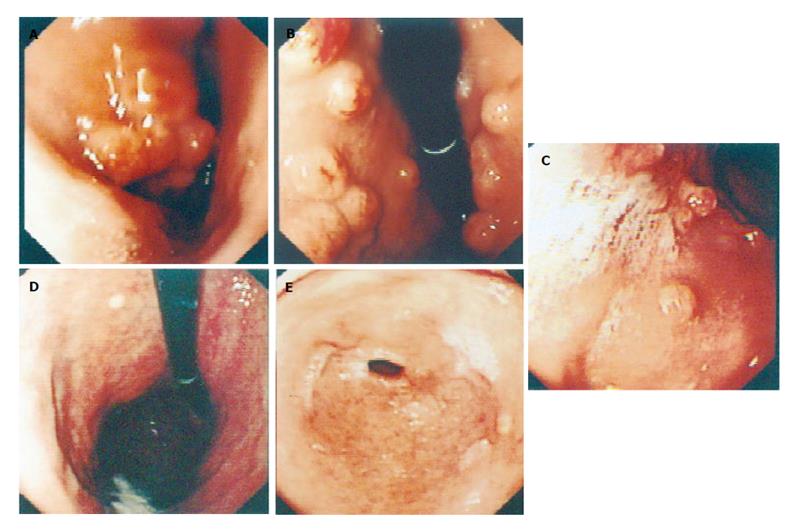
In the treatment group, H pylori was eradicated without serious side effects in 19 of 22 patients (86.3% [95% CI, 63%-99%]), and polyps disappeared in 15 of 22 patients (68.2% [95% CI, 54%-91%]) 1-12 months after treatment (Figure 1). Hyperplastic gastric polyps in the other 4 patients with successful H pylori eradication regressed to a certain extent, decreasing in size or number. However, in the remaining 3 patients without H pylori eradication, no polyps showed regression and no diminution of inflammation in the gastric mucosa 12 months after the treatment. In the control group, all patients showed no change of H pylori infection (0% [95% CI, 0%-21%]), no hyperplastic gastric polyp regression or disappearance (0% [95% CI, 0%-21%]) and no significant diminution of inflammation (Table 2). Polyps were enlarged or increased in number in 5 of the 21 patients. The rates of eradication of H pylori and disappearance of polyps in the treatment group were significantly higher than those in the control group (P < 0.01).
| Variable | Treatment groups(n = 22) | Control groups(n = 21) | P |
| Histologic findings | |||
| Inflammation | 1.6, 1.5 | 2.4, 2.3 | <0.01 |
| Activity | 1.1, 1.3 | 2.4, 2.5 | <0.01 |
| Atrophy | 2.2, 2.1 | 2.4, 2.4 | >0.05 |
| Metaplasia | 0.8, 0.6 | 0.9, 0.7 | >0.05 |
In our study, H pylori was successfully eradicated in 19 of the 22 patients. The regression or disappearance of hyperplastic gastric polyps was seen in the 19 patients. The polyps disappeared in 15 of the 19 patients 1-12 months (average, 6.5 ± 1.1 mo) after the treatment. However, none of the polyps in any of the controls or in patients without H pylori eradication showed regression. These results strongly suggest that eradication of H pylori leads to regression and disappearance of hyperplastic gastric polyps. H pylori is the main cause of chronic active gastritis and can produce multi-virulence agents, damage the gastric mucosa, stimulate gastric body to release inflammation medium, activate various cytokines and promote inflammation reaction. H pylori infection damages the gastric mucosa and glandular cells, stimulates crypt epithelia and muscularis mucosa hyperplasia with eminent mucus. If the damage factors continue their existence, the pathological changes can progress to intestinal metaplasia or atypical hyperplasia and even carcinoma. Yasunaga et al [10] reported that increased production of interleukin-1 beta and hepatocyte growth factor due to H pylori infection may contribute to thickening of the stomach by stimulating epithelial cell proliferation and foveolar hyperplasia in patients with enlarged fold gastritis, leading to formation of hyperplastic polyps.
Although hyperplastic gastric polyps did not accompany malignant transformation during the 9-12 month follow-up period in our study, H pylori infection is closely associated with gastric carcinoma. We therefore recommend that when hyperplastic gastric polyps are detected during endoscopy, serologic and pathologic tests should be done to detect and eradicatw H pylori .
Though the risk of hyperplastic gastric polyps conversing into cancer is very low, large hyperplastic gastric polyps should be snared and removed completely. Because of the malignant potential, all gastric polyps (0.5 cm or larger in diameter) should be removed. In addition, about 1 year after eradication of H pylori (4 patients in this study), any remaining hyperplastic polyps should be removed endoscopically because of the potential for development of cancer.
In conclusion, H pylori infection is related with hyperplastic gastric polyps and inflammatory cell infiltration, eradication of H pylori can prevent formation of hyperplastic gastric polyps.
S- Editor Guo SY L- Editor Wang XL E- Editor Zhang Y
| 1. | Daibo M, Itabashi M, Hirota T. Malignant transformation of gastric hyperplastic polyps. Am J Gastroenterol. 1987;82:1016-1025. [PubMed] |
| 2. | Nishi Y, Isomoto H, Mukae H, Ishimoto H, Wen CY, Wada A, Ohnita K, Mizuta Y, Murata I, Hirayama T. Concentrations of alpha- and beta-defensins in gastric juice of patients with various gastroduodenal diseases. World J Gastroenterol. 2005;11:99-103. [PubMed] |
| 3. | Veereman Wauters G, Ferrell L, Ostroff JW, Heyman MB. Hyperplastic gastric polyps associated with persistent Helicobacter pylori infection and active gastritis. Am J Gastroenterol. 1990;85:1395-1397. [PubMed] |
| 4. | Ljubicić N, Kujundzić M, Roić G, Banić M, Cupić H, Doko M, Zovak M. Benign epithelial gastric polyps--frequency, location, and age and sex distribution. Coll Antropol. 2002;26:55-60. [PubMed] |
| 5. | Ohkusa T, Takashimizu I, Fujiki K, Suzuki S, Shimoi K, Horiuchi T, Sakurazawa T, Ariake K, Ishii K, Kumagai J. Disappearance of hyperplastic polyps in the stomach after eradication of Helicobacter pylori. A randomized, clinical trial. Ann Intern Med. 1998;129:712-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 86] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 6. | Ljubicic N, Banic M, Kujundzic M, Antic Z, Vrkljan M, Kovacevic I, Hrabar D, Doko M, Zovak M, Mihatov S. The effect of eradicating Helicobacter pylori infection on the course of adenomatous and hyperplastic gastric polyps. Eur J Gastroenterol Hepatol. 1999;11:727-730. [RCA] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 7. | Laxen F, Sipponen P, Ihamaki T, Hakkiluoto A, Dortscheva Z. Gastric polyps; their morphological and endoscopical characteristics and relation to gastric carcinoma. CActa Pathol Microbiol Immunol Scand. 1982;90:221-228. |
| 8. | Gencosmanoglu R, Sen-Oran E, Kurtkaya-Yapicier O, Avsar E, Sav A, Tozun N. Gastric polypoid lesions: analysis of 150 endoscopic polypectomy specimens from 91 patients. World J Gastroenterol. 2003;9:2236-2239. [PubMed] |
| 9. | Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Patho. 1996;20:1161-1181. [PubMed] |
| 10. | Yasunaga Y, Shinomura Y, Kanayama S, Higashimoto Y, Yabu M, Miyazaki Y, Kondo S, Murayama Y, Nishibayashi H, Kitamura S. Increased production of interleukin 1 beta and hepatocyte growth factor may contribute to foveolar hyperplasia in enlarged fold gastritis. Gut. 1996;39:787-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 58] [Article Influence: 2.0] [Reference Citation Analysis (0)] |