Published online Jul 7, 2005. doi: 10.3748/wjg.v11.i25.3830
Revised: January 2, 2005
Accepted: January 5, 2005
Published online: July 7, 2005
AIM: To observe the anti-cancer effect of iNOS selective inhibitor (aminoguanidine, AG) and investigate the relationship between iNOS inhibitor and angiogenesis, infiltration or metastasis in MFC gastric cancer xenografts.
METHODS: Fifty athymic mice xenograft models were established by inoculating gastric cancer cell MFC subcutaneously. Twenty-four hours later, 0.9% sodium chloride solution, mitomycin, low dosage AG, high dosage AG, mitomycin and AG were administered by intraperitoneal injection respectively. Thus these mice were divided into five groups of 10 each randomly: control group, MMC group, AGL group, AGH group, MMC+AGH group. Two weeks later the mice were killed, and the tumor weight, inhibitory rate were evaluated. Greiss assay was used to detect the nitric oxide levels in plasma. HE and immunohistochemistry staining were used to examine microvessel density (MVD) and the expression of iNOS, VEGF, and PCNA. Apoptosis was detected by using TUNEL assay.
RESULTS: The inhibitory rates in MMC+AGH group and AGH group were 52.9% and 47.1% respectively, which is significant statistically compared with that of control group (0). In treatment groups, the cell proliferation index (PI) was lower and apoptosis index was higher than those of control group. Microvessel density, iNOS, and VEGF in MMC+ AGH group were 8.8 ± 2.6, 2.4 ± 1.1, and 2.1 ± 1.4 respectively, which is significant statistically compared with those of control group (68.3 ± 10.6, 11.3 ± 1.3, and 10.3 ± 1.6). The NO level in plasma of MMC+ AGH and AGH group were 12.7 ± 2.1 and 12.9 ± 2.0 μmol/L. Compared with that of control group (46.6 ± 2.3 μmol/L), the difference is statistically significant.
CONCLUSION: AG has anticancer effect on gastric cancer, and it has positive synergistic effect with chemotherapeutic drugs. It may play important inhibitory roles in angiogenesis of gastric cancer. The anticancer effect of iNOS inhibitors may include inducing cell apoptosis, suppressing cell proliferation and reducing angiogenesis.
- Citation: Wang GY, Ji B, Wang X, Gu JH. Anti-cancer effect of iNOS inhibitor and its correlation with angiogenesis in gastric cancer. World J Gastroenterol 2005; 11(25): 3830-3833
- URL: https://www.wjgnet.com/1007-9327/full/v11/i25/3830.htm
- DOI: https://dx.doi.org/10.3748/wjg.v11.i25.3830
The incidence of gastric cancer is high in China, and more than 170 000 people die of it each year[1]. It is significant if certain drugs are found to lower its incidence, even prevent it.
To date, we know that carcinogenesis has intimate correlation with angiogenesis[2-4]. So to reduce angiogenesis probably may inhibit the growth and development of tumors[5]. It is well known that vascular endothelial growth factor (VEGF) is an important angiogenesis factor[6,7]. We have proved that inducible nitric oxide synthase (iNOS) can induce the expression of VEGF in gastric cancer, and microvessel density (MVD) can increase with the enhancement of iNOS and VEGF[8-10]. So VEGF and iNOS both can induce angiogenesis. So if the activity of iNOS is inhibited, tumor angiogenesis may be reduced. As a result the growth and development of tumors will be inhibited. Although the roles that iNOS inhibitors play in various tumors and their mechanisms are being widely studied recently, few people have gone deep into in vivo experiment. Based on in vitro cytologic experiments, this study went further into in vivo experiment to confirm the anticancer effect of iNOS inhibitor (aminoguanidine, AG).
Fifty Kunming mice (25 females, 25 males) weighing 20-25 g were purchased from Experimental Animal Center of Jilin University. Mice were maintained under specific pathogen-free conditions and fed with sterilized food and autoclaved water. Human gastric cancer cell line MFC was purchased from Shanghai Tumor Cell Research Institute.
Greiss reagent and AG were purchased from Sigma (St. Louis, USA). Immunohistochemical S-P kit and polyclonal antibodies to iNOS, VEGF, PCNA, and FVIIIRag were all from Fuzhou Maxim Biotechnical Company (Fuzhou, China).
Each mouse was inoculated with a subcutaneous injection of MFC cells (2×106 in 0.2 mL PBS) into the left hind legs. Then these mice were divided into 5 groups of 10 each according to different agents which were administered to mice by intraperitoneal injection for 14 d. These agents included 0.9% sodium chloride solution (control group), mitomycin (MMC group, twice a week 0.7 mg/kg), low dosage of AG (AGL group, 50 mg/(kg·d)), high dosage of AG (AGH group, 150 mg/(kg·d)) and MMC+AGH group (MMC twice a week 0.7 mg/kg, AG 150 mg/(kg·d)). On the 15th d the mice were killed. The blood was taken from abdominal aorta, then centrifuged for plasma to detect NO level by Greiss assay. All tumors were resected from the body and weighed. The inhibitory rate was deduced according to the formula: inhibitory rate (%) = (1-tumor weight of treatment group/tumor weight of control group) × 100%. Then the tumors were fixed in 40 g/L phosphate-buffered formaldehyde.
Streptavidin-peroxidase (SP) method was used to detect MVD and the expression of iNOS, VEGF and PCNA. The formalin-fixed tissues were embedded in paraffin, and sectioned at a thickness of 4 μm. The sections were deparaffinized and hydrated gradually and examined by histology of HE staining, immunohistochemistry, and TUNEL technique respectively. Sections were heated in a microwave oven for 15 min to retrieve antigens. Endogenous peroxidase was blocked with 3 mL/L hydrogen peroxide methanol for 10 min at room temperature. After washing with phosphate-buffered saline (0.01 g/L, pH 7.4) for 3 min × 5 min, the tumor sections were incubated with normal non-immune serum from bull for 15 min at room temperature to eliminate non-specific staining. The sections were then incubated with the primary antibody against iNOS, VEGF, PCNA, and FVIIIRag (dilution 1/100) for 60 min at room temperature, washed with PBS for 3 min × 5 min, and incubated with the secondary antibody for 15 min followed by avidin-biotin-peroxidase for 15 min at room temperature. Finally, the slides were washed for 3-15min with PBS, visualized with DAB reagent and counterstained with hematoxylin. Negative and positive controls were used simultaneously to ensure specificity and reliability of the staining process. The negative controls were performed by substituting the primary antibody with PBS, and a positive section supplied by the manufacturer of the staining kit was taken as positive control. Sections were observed under microscope after being mounted. High vessel density was found in 100 × sights. Microvessels in 10 hot regions were counted in 400 × sights, and the average of microvessels with FVIIIRag staining in 10 hot regions was calculated as MVD. Positive staining with iNOS, VEGF and PCNA were defined by brown staining of cytoplasm. The staining degree was calculated quantitatively with CIMA-400 Colorful Image Assay System which can calculate the percentages of positive staining region of the whole region.
The percentage of positive cells with PCNA staining in five 400 × sights was counted as proliferation index (PI).
The reagent kit for apoptosis detection, TdT-FragEL DNA fragmentation detection kit was bought from ONCOGENE. Test procedures consisting of the following sections were provided in the brochure of the kit. The specimens were deparaffinized and hydrated gradually, and rinsed with 1×TBS, then incubated with proteinase K (20 μg/mL in 10 mmol/L Tris-HCl) for 20 min. After immersed in 30 mL/L H2O2 at room temperature for 5 min and in TdT labeling reaction mixture at 37°C for 1.5 h, specimens were covered with 1×conjugate for 30 min, visualized by DAB and counter-stained by hematoxylin afterwards. TBS took the place of primary antibodies as a negative control. After being mounted, sections were observed under microscope. The results of staining were analyzed and evaluated with American Image-Pro Plus software. The percentage of positive cells with TUNEL staining in five 400 × sights served as apoptosis index (AI).
All data were presented as mean±SD. The results were compared by one-way analysis of variance (ANOVA). All statistical calculations were performed with the SPSS11.0 software package. A P value less than 0.05 was regarded as statistically significant.
The tumor volume of MMC group on the 7th d was (383.4 ± 179.3) mm3, and on the 10th d those of AGH and MMC+AGH groups were (382.8 ± 132.8) mm3 and (50.0 ± 16.6) mm3 respectively. The tumor cell proliferation was almost completely suppressed. On the 14th d the tumor weight of control group was (1.7 ± 0.5) g, and those of MMC, AGH and MMC+AGH groups were (1.0 ± 0.2), (0.9 ± 0.3) and (0.8 ± 0.2) g, respectively. Compared with control group, the difference was significant statistically (P < 0.01). The NO level of plasma in AGL, AGH and MMC+AGH groups were lower than that of the control group, and there was dose-effect relationship. The difference was significant statistically (P < 0.05, Table 1).
MVD has positive correlation with iNOS and VEGF respectively. The coefficient of product-moment correlation rVEGF = 0.80, riNOS = 0.85, P < 0.05. The linear regression equation is YVEGF = 2.3565 + 0.1087X, n = 46, YiNOS = 1.8485 ± 0.1227X, n = 46. Student’s t test, P < 0.05 (Table 2).
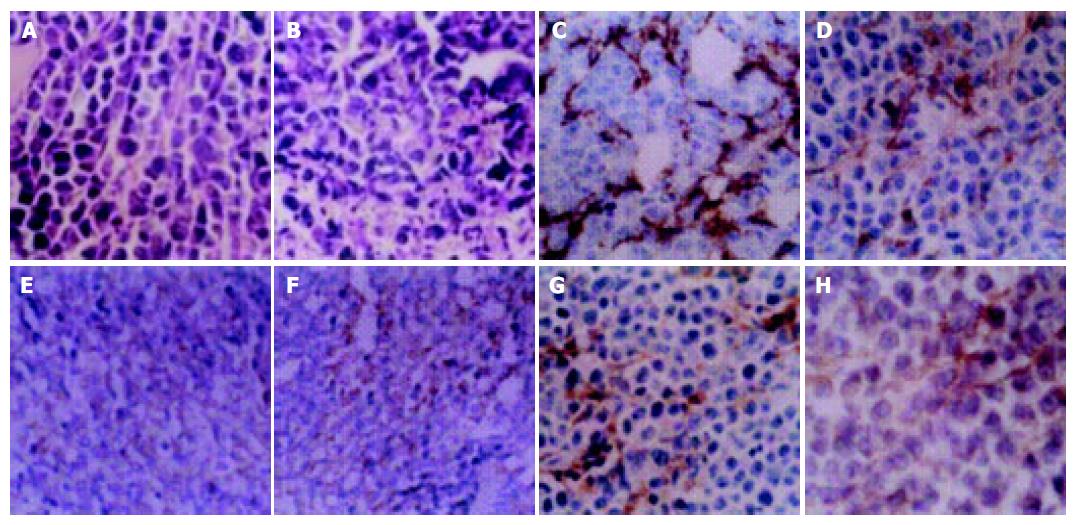
In AG and MMC+AGH groups many necrotic cells were seen and many inflammatory cells were invasive. The tumor tissues were separated by necrosis regions. In MMC group diffusely necrotic tissues could be seen. However, in control group there were a few nuclear mitotic phases in tumor cells and in tumor tissues few muscle fibers could be seen (Figures 1A and B).
MVD and the expression of iNOS and VEGF in AG groups were apparently lower than those in the control (P < 0.01). The difference was significant statistically. This revealed that AG could suppress angiogenesis of MFC xenografts. (Table 2, Figures 1C-H).
PI of control group was significantly higher than that of AG group and MMC+AGH group (P < 0.05), but the difference was not notable between treatment groups. AI in treatment groups was higher than that in the control group (P < 0.05), while there was also no difference between the treatment groups. The AI/PI value was calculated and compared among all groups. Consequently, it was apparently larger in treatment groups (P < 0.01), however no difference was shown between them (data not shown).
NO which has many bilological functions is a cytokine in mammifer[11]. It is synthesized from L-Arginine by iNOS which is the only rate-limiting enzyme[12]. It involves a serial physiological and pathological process, such as carcinogenesis. NO can induce angiogenesis, but the mechanisms are not clear[13-16]. However, several researches have revealed that NO can regulate the roles of VEGF in inducing angiogenesis by stimulating vascular endothelial cell proliferation and migration and improving vascular penetration[17-20]. VEGF can increase the activity of iNOS[6,7]. So iNOS and VEGF have positive correlation[8-10]. It has been observed that iNOS is highly expressed in many human tumors, such as colon cancer, gastric cancer, ovarian cancer, breast cancer, etc.[21]. In our previous study we have observed that the expression of VEGF and iNOS in gastric cancer presents positive correlation[8-10]. This indicates that iNOS plays an important role in the expression and activity of VEGF. We also found that iNOS and VEGF have positive correlation with the clinicopathological characteristics of gastric cancer, such as infiltration, lymphatic or hematogenous metastasis, etc. At the same time MVD was higher with the enhancement of VEGF and iNOS. This revealed that iNOS and VEGF can induce angiogenesis in gastric cancer[8-10,21].
To explore the anticancer mechanisms of iNOS inhibitor (aminoguanidine), in this study we evaluated the effect of AG on tumor cell proliferation and apoptosis in xenografts. The AI/PI, a value reflecting cytokinetics, showed a more significant difference. Finally it is shown that compared with control group, the inhibitory rate of treatment groups are apparently higher. We also observed that AG obviously decreases the expression of iNOS and VEGF, and reduces MVD compared to that of control group. By immunohist-ochemistry (SP method) and Greiss assay we detected the NO level in plasma. The results indicate that the anticancer mechanisms of AG are mainly to inhibit the expression of iNOS and reduce the NO level in plasma. Therefore, the growth of tumor and angiogenesis are inhibited directly, and MVD and the expression of VEGF are suppressed indirectly. In this way the nutrition supply of the tumor is impaired, and further cell proliferation is inhibited and cell apoptosis is improved. These results are consistent with those of Koh et al They observed that AG could inhibit the activity of iNOS in gastric cancer, and suppressed carcinogenesis. At the same time AG and COX-2 selective inhibitors could obviously inhibit the activity of iNOS and COX-2. So, the primitive pathology of colon cancer (aberrant crypt foci, ACF) is prevented.
Compared with traditional anticancer therapy, antian-giogenesis therapy has many superiorities. In this study the effect of MMC group is similar to that of AG group. However, the target of chemotherapeutic drugs is tumor cells which have genetic instability and are apt to mutation and drug-resistance, so the drug-resistance incidence of chemotherapeutic drugs is 30%. And these drugs have many toxic effects. In MMC+AGH group the anticancer effect is the best which indicates that the effect of AG depends on proper treatment opportunity and scheme. MMC and AG have positive synergy. So certain drugs which can reduce the NO level by inhibiting the expression and activity of iNOS at cellular or genetic level probably may regulate the NO level of tumor microvessel to inhibit carcinogenesis and angiogenesis. And if combined with radiotherapy or bioreactive modulators, it will play a more important role in anticancer effect. This is the first part of a serial study, and we will carry out the further study afterwards with molecular biological technology, such as Southern blotting, Northern blotting, RT-PCR, etc.
Science Editor Guo SY Language Editor Elsevier HK
| 1. | Sun XD, Mu R, Zhou YS, Dai XD, Zhang SW, Huangfu XM, Sun J, Li LD, Lu FZ, Qiao YL. Analysis of mortality rate of stomach cancer and its trend in twenty years in China. Zhonghua ZhongLiu ZaZhi. 2004;26:4-9. [PubMed] |
| 2. | Feng CW, Wang LD, Jiao LH, Liu B, Zheng S, Xie XJ. Expression of p53, inducible nitric oxide synthase and vascular endothelial growth factor in gastric precancerous and cancerous lesions: correlation with clinical features. BMC Cancer. 2002;2:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 98] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Koh E, Noh SH, Lee YD, Lee HY, Han JW, Lee HW, Hong S. Differential expression of nitric oxide synthase in human stomach cancer. Cancer Lett. 1999;146:173-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 4. | Doi C, Noguchi Y, Marat D, Saito A, Fukuzawa K, Yoshikawa T, Tsuburaya A, Ito T. Expression of nitric oxide synthase in gastric cancer. Cancer Lett. 1999;144:161-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Eroğlu A, Demirci S, Ayyildiz A, Kocaoğlu H, Akbulut H, Akgül H, Elhan HA. Serum concentrations of vascular endothelial growth factor and nitrite as an estimate of in vivo nitric oxide in patients with gastric cancer. Br J Cancer. 1999;80:1630-1634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 40] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Lee JC, Chow NH, Wang ST, Huang SM. Prognostic value of vascular endothelial growth factor expression in colorectal cancer patients. Eur J Cancer. 2000;36:748-753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 215] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 7. | Mattern J, Koomägi R, Volm M. Coexpression of VEGF and bFGF in human epidermoid lung carcinoma is associated with increased vessel density. Anticancer Res. 1997;17:2249-2252. [PubMed] |
| 8. | Wang GY, Gu JH, Lu GY, Meng XY. Effets of aminoguanidine on transplantation stomach cancer in mice. J Jilin University. 2004;30:409-412. |
| 9. | Wang GY, Wang X. Relationship between tumor angiogen-esis and expression of vascular endothelial growth factor and nitric oxide synthase in human gastric cancer. Zhongguo Puwai Jichu Yu Linchuang Zazhi. 2004;11:55-57. |
| 10. | Wang X, Wang GY. Expression of vascular endothelial growth factor and nitric oxide synthase in human gastric cancer and its relation to angiogenesis and clinical pathology. Zhonghua Weichang Waike Zazhi. 2002;5:145-148. |
| 11. | Farsky SH, Borelli P, Fock RA, Proto SZ, Ferreira JM, Mello SB. Chronic blockade of nitric oxide biosynthesis in rats: effect on leukocyte endothelial interaction and on leukocyte recruitment. Inflamm Res. 2004;53:442-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Tong BC, Barbul A. Cellular and physiological effects of arginine. Mini Rev Med Chem. 2004;4:823-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 107] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Lala PK, Orucevic A. Role of nitric oxide in tumor progression: lessons from experimental tumors. Cancer Metastasis Rev. 1998;17:91-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 138] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 14. | Jenkins DC, Charles IG, Thomsen LL, Moss DW, Holmes LS, Baylis SA, Rhodes P, Westmore K, Emson PC, Moncada S. Roles of nitric oxide in tumor growth. Proc Natl Acad Sci USA. 1995;92:4392-4396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 549] [Cited by in RCA: 563] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 15. | Kitadai Y, Haruma K, Tokutomi T, Tanaka S, Sumii K, Carvalho M, Kuwabara M, Yoshida K, Hirai T, Kajiyama G. Significance of vessel count and vascular endothelial growth factor in human esophageal carcinomas. Clin Cancer Res. 1998;4:2195-2200. [PubMed] |
| 16. | Salvucci O, Carsana M, Bersani I, Tragni G, Anichini A. Antiapoptotic role of endogenous nitric oxide in human melanoma cells. Cancer Res. 2001;61:318-326. [PubMed] |
| 17. | Gallo O, Masini E, Morbidelli L, Franchi A, Fini-Storchi I, Vergari WA, Ziche M. Role of nitric oxide in angiogenesis and tumor progression in head and neck cancer. J Natl Cancer Inst. 1998;90:587-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 316] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 18. | Rieder G, Hofmann JA, Hatz RA, Stolte M, Enders GA. Up-regulation of inducible nitric oxide synthase in Helicobacter pylori-associated gastritis may represent an increased risk factor to develop gastric carcinoma of the intestinal type. Int J Med Microbiol. 2003;293:403-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 49] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 19. | Rao CV, Indranie C, Simi B, Manning PT, Connor JR, Reddy BS. Chemopreventive properties of a selective inducible nitric oxide synthase inhibitor in colon carcinogenesis, administered alone or in combination with celecoxib, a selective cyclooxygenase-2 inhibitor. Cancer Res. 2002;62:165-170. [PubMed] |
| 20. | Jung ID, Yang SY, Park CG, Lee KB, Kim JS, Lee SY, Han JW, Lee HW, Lee HY. 5-Fluorouracil inhibits nitric oxide production through the inactivation of IkappaB kinase in stomach cancer cells. Biochem Pharmacol. 2002;64:1439-1445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Chen T, Nines RG, Peschke SM, Kresty LA, Stoner GD. Chemopreventive effects of a selective nitric oxide synthase inhibitor on carcinogen-induced rat esophageal tumorigenesis. Cancer Res. 2004;64:3714-3717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.0] [Reference Citation Analysis (0)] |