Published online Jun 6, 2011. doi: 10.4292/wjgpt.v2.i3.17
Revised: May 25, 2011
Accepted: June 2, 2011
Published online: June 6, 2011
Over the past two decades, proton pump inhibitors (PPIs) have emerged as highly effective and relatively safe agents for the treatment of a variety of gastrointestinal disorders. Unfortunately, this desirable pharmacological profile has also contributed to superfluous and widespread use in both the inpatient and outpatient settings. While generally well-tolerated, research published over the last decade has associated these agents with increased risks of Clostridium difficile disease, fractures likely due to calcium malabsorption and both community-acquired (CAP) and hospital-acquired pneumonias (HAP). The mechanism behind PPI-associated pneumonia may be multifactorial, but is thought to stem from compromising the stomach’s “acid mantle” against gastric colonization of acid-labile pathogenic bacteria which then may be aspirated. A secondary postulate is that PPIs, through their inhibition of extra-gastric H+/K+-ATPase enzymes, may reduce the acidity of the upper aerodigestive tract, thus resulting in increased bacterial colonization of the larynx, esophagus and lungs. To date, several retrospective case control studies have been published looking at the association between PPI use and CAP. Some studies found a temporal relationship between PPI exposure and the incidence of pneumonia, but only two could define a dose-response relationship. Furthermore, other studies found an inverse correlation between duration of PPI use and risk of CAP. In terms of HAP, we reviewed two retrospective cohort studies and one prospective study. One retrospective study in a medical ICU found no increased association of HAP in PPI-exposed patients compared to no acid-lowering therapy, while the other in cardiothoracic surgery patients showed a markedly increased risk compared to those receiving H2RAs. The one prospective study in ICU patients showed an increased risk of HAP with PPIs, but not with H2RAs. In conclusion, the current literature shows a slight trend toward an association between PPI use and pneumonia and an increased risk with PPIs over H2RAs, but the findings are not consistent across all studies. Larger controlled trials still need to be done to better identify the risk that PPIs impart towards patients contracting CAP or HAP. Until these are completed, we will have to continue to extrapolate across smaller controlled trials to predict the associated risks in our respective patient populations. In the interim, it appears prudent to limit the use of PPIs to situations where they are clinically indicated and, in such cases, use them at the lowest effective dose. In the case of prescribing for stress ulcer prophylaxis in ICU patients, perhaps H2RAs should be used as the preferred agents over PPIs.
- Citation: Fohl AL, Regal RE. Proton pump inhibitor-associated pneumonia: Not a breath of fresh air after all? World J Gastrointest Pharmacol Ther 2011; 2(3): 17-26
- URL: https://www.wjgnet.com/2150-5349/full/v2/i3/17.htm
- DOI: https://dx.doi.org/10.4292/wjgpt.v2.i3.17
In 2001, expenditure of acid-suppressive medications, most commonly PPIs, totaled $ 10.8 billion, ranking second overall in US retail sales[1]. Since 2001, the annual growth rate for PPI sales has increased by approximately 2%[2]. In 2008, $ 13.9 billion were spent on PPIs, ranking third in US sales by therapeutic class with 113.4 million prescriptions dispensed[2]. The popularity of these medications is not restricted to the USA, making PPIs among the most frequently prescribed drugs in the world[3].
These burgeoning sales volumes stem not only from superfluous prescribing in the outpatient arena, but also from inappropriate initiation of anti-secretory therapy during hospitalization. For example, in three different studies performed on inpatients, 40% to 71.4% received acid-suppressive therapy with either an H2RA or a PPI during hospitalization. According to each author’s criteria, 65% to 70% of these patients had no true indication for these acid-lowering drugs. Furthermore, 50% to 55% of the patients without an indication were continued on acid-suppressive therapy in the outpatient setting[4-6].
Although proton pump inhibitors (PPIs) are generally well-tolerated and clearly the most effective available agents used in the treatment of gastroesophageal reflux disease (GERD), peptic ulcer disease (PUD), Helicobacter Pylori infections and other hypersecretory conditions, their use does not come without risks. For example, long term use of PPIs has been associated with increased risk of hip fractures[7-10], Clostridium difficile infections[11,12] and, as is the focus of this paper, pneumonia. However, before engaging in a discussion on the potential risk of pneumonia associated with PPI exposure based on the current literature, this review first provides some relevant background on the basic pharmacology of PPIs.
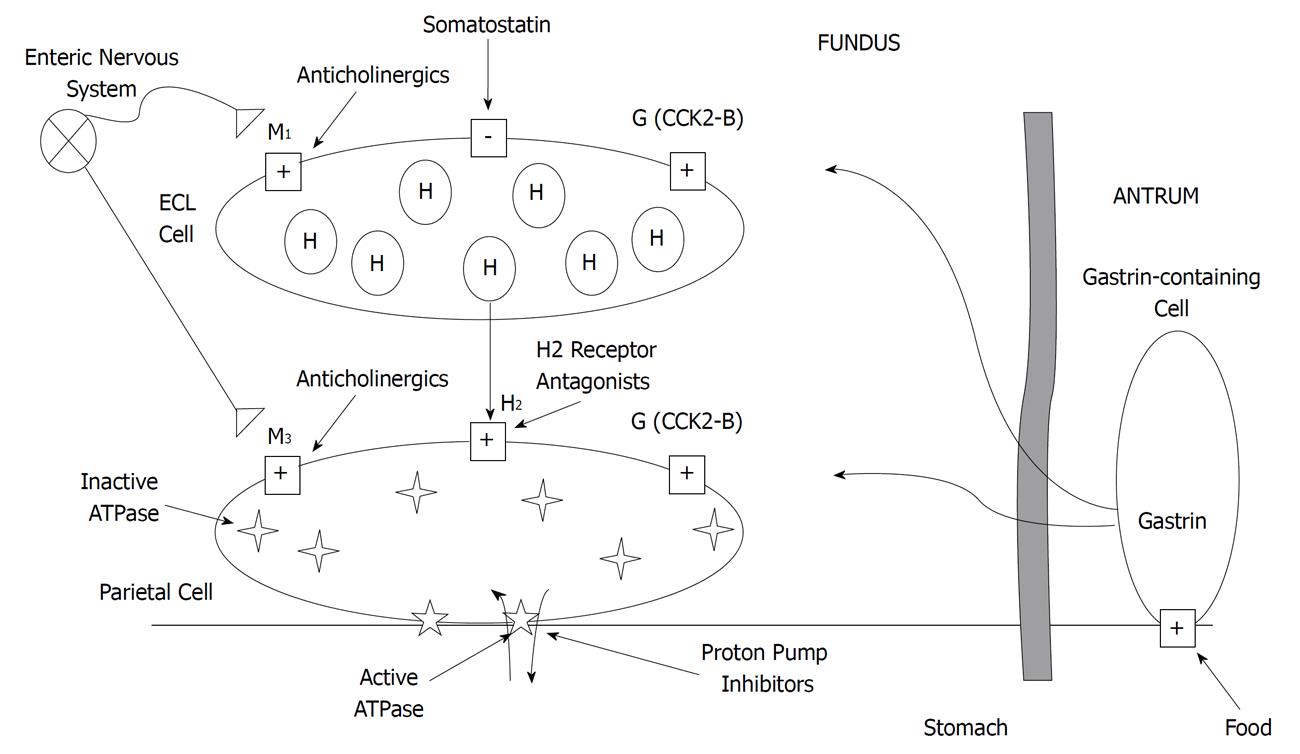
Gastric acid secretion is regulated by a redundant neurohormonal pathway involving gastrin, acetylcholine and histamine (Figure 1). Gastrin is secreted from gastrin-containing cells located in the antrum of the stomach. These cells release gastrin in response to food-induced increases in pH. Acetylcholine is released from the axon terminals of enteric neurons. Like gastrin, acetylcholine is released in response to food-induced increases in pH but can also be triggered by the sight and smell of food. Binding of gastrin and acetylcholine to the gastric parietal cell increases intracellular calcium concentrations. The intracellular calcium activates protein kinases which ultimately activate the proton/potassium adenosine triphosphatase (H+/K+-ATPase) pump located in the secretory canaliculus[13,14].
Histamine, on the other hand, is less involved with meal-stimulated acid secretion and plays a larger role in nocturnal acid secretion. Histamine is released from enterochromaffin-like (ECL) cells, which are activated by gastrin and acetylcholine and inhibited by somatostatin. Histamine activates adenylyl cyclase which increases intracellular cyclic adenosine monophosphate (cAMP) concentrations. The intracellular cAMP activates protein kinases which then activate the H+/K+-ATPase pump[13-15].
Despite the different actions of each of the neurohormones, the final converging step towards gastric acid production is through the activation of the H+/K+-ATPase pump[16]. The H+/K+-ATPase pump releases hydrogen ions into the gastric lumen in exchange for potassium ions[13,15]. These pumps generate a pH of 0.8 to 1.0 within the parietal cell[15]. As a result, the parietal cell is the only membrane-enclosed space within the human body that achieves a pH below 4.0[14].
PPIs are the most effective acid suppressive agents because they target the common step in the redundant gastric acid secretory pathway. By forming irreversible disulfide bonds with the H+/K+-ATPase pump, PPIs effectively limit gastric acid secretion without a bypass mechanism. It is interesting to note that most, but not all, proton pumps are inhibited with the first dose of the PPI and up to seven doses may be needed to maximally inhibit gastric acid secretion at a given dose level[17,18]
The gastric lumen normally maintains a pH of less than 2. However, a gastric pH above 4 is recommended to effectively treat GERD, PUD and H pylori infections[13]. With a single daily administration, PPIs can maintain an intragastric pH above 4 for 10.5 h to 16.8 h in a 24-h period[15,19]. The median 24-hour intragastric pH can vary from 3.6 to 4.9, depending on the particular PPI and milligram dose administered.
In contrast to the PPIs, antacids effectively neutralize gastric acid for up to two hours, while histamine-2 receptor antagonists (H2RAs) can achieve this goal for up to eight hours. Furthermore, with H2RAs, tachyphylaxis can begin to occur within the first few days of initiation. This is because, while H2RAs prevent histamine-mediated stimulation of gastric parietal cells, these agents fail to block the activation of the proton pump by gastrin and acetylcholine. These two pathways tend to compensate by increasing activity once histamine blockade is elicited by the H2RA[13,20]. Since gastric acid secretion is stimulated by redundant pathways, inhibition of the proton pump, the final step in the secretory pathway, is the most effective approach. Thus, as previously mentioned, unlike other acid-suppressive agents, PPIs avoid the body’s compensatory mechanisms by targeting this final step in gastric acid secretion[13,15,20].
While PPIs are used to reduce gastric acidity, most are actually acid-labile prodrugs. To protect the acid-labile prodrug from activation in the acidic environment of the stomach, PPIs are compounded as either enteric-coated formulations or buffered suspensions. The enteric-coated formulations should be swallowed whole and, therefore, should not be chewed. The importance of this acid-lability is demonstrated when comparing the oral bioavailability of PPIs when taken on an empty stomach versus when they are taken with meals. When taken on an empty stomach, the oral bioavailability of PPIs ranges from 30% to 90% depending on the PPI and the number of doses administered. However, when taken with food, the oral bioavailability may decrease by as much as 50% with some formulations[13-15].
Once the PPI formulation has navigated the pyloric sphincter and entered the alkaline intestinal lumen, the enteric coating dissolves and releases the prodrug. Upon dissolution in the intestinal lumen, the prodrug, a lipophilic weak base with a pKa between 4 and 5, readily crosses the lipid membrane of the intestine to enter the systemic circulation[14]. From the systemic circulation, the prodrug is eventually distributed to the gastric parietal cells where it exerts its pharmacological effect.
Within the acidic environment of the gastric parietal cell, the prodrug undergoes protonation to convert the inactive prodrug to the active thiophilic sulfonamide cation. Once protonated to the thiophilic sulfonamide cation, PPIs are less membrane permeable and accumulate in the parietal cell. The thiophilic sulfonamide is unable to form the inactivating disulfide bond with the cysteine sulfhydryl moiety of the H+/K+-ATPase pump when the pump is inactive[14]. However, upon activation, the pump undergoes a conformational change in order to exchange protons for potassium across the gastric mucosa. This conformational change exposes the cysteine sulfhydryl moiety to the activated sulfonamide cation, resulting in disulfide bond formation. The formation of this disulfide bond results in the irreversible inactivation of the proton pump[14].
Since the activity of the PPI depends on the conformational change of the active H+/K+-ATPase pump, PPIs should be present in the parietal cell while the proton pumps are activated. Ideally, the peak serum concentration of the PPI should coincide with maximal proton pump activation. Since food initiates the pathway to activate the proton pumps, PPIs should be taken 30 min to 60 min prior to meals. The 30 min to 60 min delay between drug administration and food consumption allows the PPI to be absorbed from the gastrointestinal tract and distributed to the parietal cells in the gastric mucosa. The timing of the drug administration relative to meals is further reinforced by the half-life of the drug. Most PPIs have a half-life of 30 min to 90 min. This short half-life suggests that the drug is rapidly cleared from the systemic circulation[13,15]. However, literature suggests that PPIs may effectively control gastric acid secretion for up to 24 h. The extended duration of activity is due to the PPI’s accumulation in the parietal cell and the irreversible inhibition of the H+/K+-ATPase. Once irreversibly bound to the H+/K+-ATPase through a covalent interaction, the duration of action is limited by the replacement of inactivated proton pumps. Inactivated proton pumps are replaced approximately every 50 h[13,15]. Consequently, PPIs may be administered once daily despite the short elimination half-life.
Despite the optimal pharmacokinetic and pharmacodynamic parameters, new PPI formulations are being created. Potassium-competitive acid blockers (P-CABs), which specifically inhibit the potassium-binding region of the H+/K+-ATPases, have been considered the next generation of gastric acid suppressants. Like PPIs, P-CABs are lipophilic, weak bases which are rapidly protonated in an acidic environment. Although studies suggest P-CABs increase the intragastric pH more rapidly than PPIs while maintaining similar antisecretory effects, P-CABs may have the same theoretical absorption challenges as PPIs. The clinical benefits of the theoretical pharmacodynamic advantages have yet to be demonstrated[21,22].
Although PPIs are relatively safe, and can in fact be administered throughout pregnancy, some patients may experience adverse effects[15]. The most commonly reported side effects in clinical studies have been diarrhea, headache and abdominal pain, with an incidence of about 1% to 5%, comparable to the incidence with placebo. Other than these usually self-limiting side effects, PPIs may have the potential to cause nutritional deficits. Gastric acid releases vitamin B12 from ingested food and subtherapeutic B12 concentrations have been observed with prolonged PPI therapy. Gastric acid also promotes the release of food-bound minerals including iron, zinc and calcium. The consistent administration of PPIs for at least one year may lead to low levels of circulating divalent cations, including magnesium and copper[23,24]. Although malabsorption has been suspected, PPI-induced iron, zinc and B12 deficiencies have not been shown to be clinically significant[10,13,15,25-27].
Calcium malabsorption, however, may be attributed to the increased incidence of hip fractures in patients with no other risk factors. One nested case-control study determined the adjusted odds ratio (AOR) for hip fractures with the administration of PPIs increased with the duration of therapy. The AOR for one year of therapy was 1.22 and increased to 1.41 with the second year. After three years, the AOR increased to 1.51. With four years of PPI therapy, the AOR was determined to be 1.59[7-10]. Calcium supplementation could be achieved with either calcium carbonate or calcium citrate; however, the relative bioavailability of the calcium salts in less acidic gastric environments needs to be considered. At a neutral pH, calcium carbonate is relatively insoluble but calcium citrate is unaffected by the increase in gastric pH[20,28]. When compared with placebo in one study, the bioavailability of calcium carbonate in patients taking omeprazole decreased by 41% after seven days[29]. Consequently, supplementation with calcium citrate should be recommended in patients who are on long-term PPI therapy [30].
Apart from the potential nutritional maladies imparted by PPI therapy, the inhibition of gastric acid may also increase the risk of enteric infections. Gastric acid is an important barrier to colonization and infections by invading pathogens[31,32]. There may be an increased incidence of Clostridium difficile infections with PPI administration[12]. Studies suggest that the administration of PPIs doubled the incidence of Clostridium difficile infections[11,33-35]. In one study, the incidence of Clostridium difficile infections doubled from 4.4% in patients not receiving PPIs to 9.3% in patients receiving PPIs, resulting in a statistically significant relative risk ratio of 2.1[33]. In a second study, the incidence of Clostridium difficile infections increased from nearly 37% to approximately 63% with PPI exposure[10]. Furthermore, in a retrospective study, administration of PPIs resulted in 1.44 cases of Clostridium difficile infections per 100 patients compared to 0.74 cases per 100 patients not exposed to PPIs[35]. The length of hospital stays increased from 7.3 d to 12.9 d in patients with Clostridium difficile infections who were also receiving PPI therapy[36]. For patients who were on PPIs for longer than 6 mo, the incidence of Clostridium difficile infections increased from 5% to 23%[35]. In a study by Cadle focusing on patients treated with oral metronidazole for 10-14 d, the Clostridium difficile infection cure rates decreased by 50% and the incidence of recurrent infections doubled in patients treated with PPIs relative to patients not receiving PPIs[37]. Consequently exposure to PPIs resulted in increased incidence of Clostridium difficile infections.
Pneumonia has also been associated with patient exposure to PPI therapy. Salivary gland secretions, gastric acid and well synchronized gastric motility are innate mechanisms to restrict bacterial colonization in the gastric lumen. A gastric pH of less than 2 effectively limits bacterial colonization from ingested microbes[15]. Since PPIs effectively increase the gastric pH above 4 for the majority of a 24-h period, this defense mechanism against the colonization of ingested bacteria is compromised[15]. Consequently, due to the reduced gastric acidity, the bacterial load increases in the stomach. Some studies suggest there may be a dose-response relationship in the administration of PPIs and the risk of pneumonia[1,38]. Perhaps providing some means to explain this association, single or repeated administration of PPIs may in fact lead to delayed gastric emptying, increased gastric contents, increased bacterial load and increased pressure on the lower esophageal sphincter[15]. The increased pressure against the lower esophageal sphincter may lead to retrograde movement of gastric contents up the esophagus. This reflux may then increase the risk of subsequent aspiration of both the gastric contents and the bacteria[15].
Of further interest in explaining a dose-related association between PPI use and pneumonias were several in vitro studies conducted by Altman[39-41] which identified the presence of proton pumps in extragastric sites including the larynx and the lungs. The clinical significance of their presence was not assessed. Since proton pump inhibitors are absorbed and distributed throughout the systemic circulation, PPIs may reduce the acidity of the upper aerodigestive tract, thus resulting in increased bacterial colonization of the larynx, esophagus and lungs. The increased bacterial colonization of the larynx and esophagus may therefore contribute to an increased incidence of pneumonia.
A study conducted by Eckley[42] took this concept one step further. Twenty three patients with either GERD or larygnopharyngeal reflux were enrolled in a 12 wk study to assess the change in the salivary pH when administered omeprazole 20 mg twice daily by mouth. Prior to beginning therapy, the average salivary pH was 7.15, but this increased to 7.58 following 12 wk of therapy[42]. Although this was a statistically significant increase in pH, the physiological pH of salivary acid is generally about 8.0. Consequently, the patient’s exposure to PPIs returned the pH towards normal physiological pH and the increase in salivary pH was thought to be clinically insignificant.
This study was unable to conclude if the increase in salivary pH was actually due to inhibition of the proton pumps located in the larynx or lungs or if the increase in salivary pH was related to the regurgitation of less acid contents from the gastric lumen due to the inhibition of the gastric proton pumps. Although in vitro studies have confirmed the presence of extragastric pumps, in vivo evidence assessing the clinical impact of PPIs on the extragastric pumps appears to be lacking.
Is it possible that PPIs actually potentiate infections by hindering the activity of white blood cells? The humoral defense mechanism against bacterial colonization is mediated by the white blood cells. In vitro studies[43-45] have assessed the cytotoxicity of natural killer (NK) cells when exposed to target cells in varying ratios and in different solution media. Not surprisingly, as the ratio of NK cells to target cells increased, the percentage of target cells lysed increased, suggesting a positive correlation. The varying ratios of NK cells to target cells were then exposed to media with an omeprazole concentration ranging between 0 µmol/L and 288 µmol/L[43,44]. The cytotoxic potential of the NK cells decreased with increasing concentrations of omeprazole in the solution media, suggesting an inverse correlation.
Due to the inverse correlation between NK cell cytotoxicity and omeprazole exposure, a study was conducted to determine the extent of the potential drug-immune system interaction which may exist between omeprazole and NK cells. Without omeprazole exposure, NK cells lysed 48.7% of the target cells. The NK cells were then exposed to a 288 µM concentration of omeprazole before the target cells were introduced to the media. The cytotoxic potential of the NK cells decreased to 4.2%, confirming the inverse correlation previously identified. To determine if this interaction was reversible or irreversible, the NK cells were exposed to a 288 µmol/L concentration of omeprazole for one hour, rinsed of the omeprazole solution and then the target cells were introduced to the NK cell media. The NK cells recovered 75% of their cytotoxicity when the media solution was rinsed of omeprazole. By rechallenging the rinsed NK cells with a second omeprazole solution, the cytotoxic activity of the NK cells was again reduced[43]. The reversible interaction between the omeprazole solution and the NK cell’s cytotoxicity appeared to be dependent on continuous exposure to the omeprazole.
In vivo studies assessing the clinical significance of omeprazole-induced inhibition of NK cell cytotoxicity are currently lacking but it is thought to be clinically insignificant for two reasons. First, the inhibition of cytotoxic activity started to significantly decrease when the omeprazole concentration was approximately 72 µmol/L. The therapeutic concentration for omeprazole ranges from 0.19 µmol/L to 19.4 µmol/L. Consequently, the physiological concentration achieved in the blood would only minimally inhibit the cytotoxic potential of the NK cells. Secondly, 75% of the cytotoxic inhibitory effect was washed out when the solution was rinsed of the omeprazole. Knowing the plasma half-life of the PPIs ranges between 30 min and 90 min, the NK cell exposure to PPIs would be limited as the PPI would be readily cleared from the systemic circulation[43].
In a nested case-control study conducted by Laheij[38], 5551 new cases of documented pneumonia were observed from a total of 364 683 individuals enrolled in a general practice research database in the Netherlands. Patients were required to have one year of medical history documented in the database prior to being enrolled in the study and the pneumonia diagnosis was determined both objectively and subjectively. The objective assessments included thorax radiography and microbiological culture results; while subjective assessments included determining whether the patient’s clinical symptoms were consistent with a pneumonia diagnosis.
In this study, patients were approximately four times more likely to acquire pneumonia when using PPIs compared to non-users, regardless of the appropriateness of the therapy. Furthermore, when adjusted for individuals currently using PPIs and those who stopped using PPIs, the relative risk of developing pneumonia was nearly 1.89 (95% CI: 1.36-2.62) with current use. In an attempt to make the study more conservative, patients were excluded if they did not have an objective laboratory measure of pneumonia. Patients were still approximately two times more likely to acquire pneumonia following PPI administration.
This study identified a positive correlation between the incidence of pneumonia and the PPI dose administered. Based on the defined daily dose (DDD), the dose of the specific PPI administered could be compared across the class. As the DDD increased, the incidence of pneumonia also increased. Relative to patients who had stopped taking PPIs 30 d to 180 d prior to their pneumonia diagnosis, the increased incidence of pneumonia achieved statistical significance at a DDD of at least one (Table 1).
This study also identified an inverse correlation between the incidence of pneumonia and the duration of PPI use prior to the pneumonia diagnosis. Although all the durations of exposure achieved statistical significance, there was a trend in the odds ration. As the duration of PPI use prior to the pneumonia diagnosis increased, the incidence of pneumonia decreased (Table 1).
PPI administration was attributed to an incidence of 1.05 cases of pneumonia per 100 person-years of PPI exposure. Assuming the average duration of use for PPIs is 0.42 years (approximately 5.5 mo), 1 patient would acquire pneumonia out of 226 patients treated with PPIs.
Thinking that the association to pneumonia incidence may be a class effect, the study also assessed which PPI was associated with an increased incidence of pneumonia.Unfortunately, this study was not powered to detect a difference in the incidence of pneumonia based on the agent administered.
An editorial by Gregor[46] introduced potential limitations of Laheij’s study[38]. In order to confirm the correlation between PPI use and pneumonia incidence, a trial of 9000 individuals would have needed to be conducted with pneumonia as the primary outcome measure, rather than the 5551 used in the study. Additionally, he suggested that the individuals with more severe reflux were more apt to develop pneumonia as they were unable to discontinue the PPI due to the severity of their condition. Consequently, these individuals would be at an increased risk for chemical aspiration and the subsequent complication of infectious pneumonia.
In a case-control study conducted by Gulmez[47], 7642 patients with their first diagnosis of community-acquired pneumonia (CAP) were compared with 34 176 healthy controls matched by age and gender from 2000 to 2004. PPI exposure was assessed by determining if the patients had redeemed a prescription for a PPI during the past 90 d. If a prescription had not been redeemed in the past 90 d, the patients were considered to be past users of PPIs and patients who had redeemed a PPI prescription within 90 d were assumed to be current users. Of the 2401 patients who were deemed to be current users of PPIs in this study, 818 patients were diagnosed with CAP for the first time.
The Gulmez study[45] also identified a temporal relationship between PPI treatment and pneumonia. The highest incidence of pneumonia was associated with PPI administration within seven days of diagnosis, with an odds ratio of 5.0 (95% CI: 2.1-11.7). This temporal relationship declined to an odds ratio of 1.3 (95% CI: 1.2-1.4) when PPI therapy was started more than 84 d before the diagnosis of pneumonia. This correlation was contrary to the independent variable belief, which would suggest an increased incidence of pneumonia with continued administration and increased pH. Previous use of PPIs was not associated with an increased incidence of pneumonia. Nor was a dose-response relationship between PPIs and pneumonia found.
Despite the current dogma suggesting that older patients are more susceptible to acquiring pneumonia, the strongest association of the first-time CAP diagnosis with PPI administration appeared to be in younger patients and those without hospital contacts. Among current PPI users, an inverse correlation was found between the patient’s age and the incidence of pneumonia. The incidence of pneumonia appeared to decrease as the patient aged (Table 2).
The Gulmez study[47] suggested that young, healthy patients who have not had a previous hospital admission and have not been administered antibiotics in the past 90 d are actually at an increased risk of contracting community-acquired pneumonia (Table 2). Unfortunately, this study only included patients who have never had a prior diagnosis of pneumonia.
A nested-case control study by Dublin et al[48] evaluated the risk of pneumonia in community-dwelling adults between the age of 65 and 94 years. Pneumonia was identified based on ICD-9 codes and PPI use was determined from computerized pharmacy records. PPIs were being administered to 12% (132/1125) of the population who acquired pneumonia. Meanwhile, PPIs were administered to 7% (160/2235) of the population who remained pneumonia-free. There was no difference detected between these PPI exposure rates, suggesting PPIs were not contributing to the incidence of pneumonia in this study.
However, in a study conducted by Roughead et al[49], an increased incidence of pneumonia was identified in an elderly population. Of the 185 533 veterans enrolled in this study, 58% were men with an average age of 79.4 years. There was a 16% increase in hospitalization for pneumonia in the population receiving PPIs. This study also evaluated the association of bacterial pneumonia in patients administered antibiotics. The incidence of bacterial pneumonia was similar but patients prescribed antibiotics were also likely to be prescribed a PPI.
Myles et al[50] identified 3709 cases of pneumonia and 22 174 controls to evaluate the risk of pneumonia in patients taking PPIs. Patients were at least 40 years of age but the majority of the cases were at least 70 years of age. This study found the risk of pneumonia increased by 55% in patients who were taking PPIs. However, patients diagnosed with pneumonia were more likely to be smokers, have greater heart disease and chronic lung disease compared to the general population.
Rodriguez et al[51] found an increased association of pneumonia in patients between 20 and 79 years of age enrolled in the Health Improvement Network database. Patients taking PPIs had a 16% increase in the incidence of pneumonia relative to the matched controls. Although this was only a slight increase in incidence, this study found that the association occurred only in the first 12 mo of treatment with PPIs.
An additional study, like Laheij, also assessed the impact of dose and duration of PPI exposure was conducted by Sarkar[1]. This study further demonstrated the temporal relationship between PPI administration and the first diagnosis of CAP. This nested case-control study included patients who were enrolled in the general practice research database from the United Kingdom.
Just like the Laheij study[38], a dose-dependent correlation between the administration of PPI therapy and the incidence of pneumonia was detected. This study[1] divided the population into two dosage categories. Only the patients who received greater than 1.5 DDDs of a PPI achieved statistical significance. This further suggested a dose-dependent association between PPI therapy and the first incidence of pneumonia.
Sakar et al further confirmed the inverse correlation between the duration of PPI use and the incidence of CAP realized by Laheij[38]. The association of recent PPI initiation with the incidence of pneumonia determined the risk for pneumonia progressively increased as PPIs were started 14 d, 7 d and even 2 d prior to the onset of pneumonia symptoms (Table 3). As long as the PPI exposure was within 30 d of symptom presentation, the increased incidence of CAP achieved statistical significance. The risk of pneumonia significantly decreased beyond 30 d of use. PPI exposure between 30 d and 180 d was not statistically significant.
| PPI exposure | Case patients | Control participants | Odds Ratio | P value |
| No PPI exposure | 73 187 | 777 626 | 1 | - |
| No PPI exposure in 30 d | 3424 | 19 215 | 0.95 | 0.05 |
| Current PPI exposure | 3455 | 10 031 | 1.02 | 0.48 |
| < 1.5 DDD | 3056 | 9126 | 1 | 0.94 |
| > 1.5 DDD | 399 | 905 | 1.23 | 0.01 |
| Duration of PPI Use (d) | ||||
| < 2 | 64 | 54 | 6.53 | < 0.001 |
| < 7 | 124 | 148 | 3.8 | < 0.001 |
| < 14 | 204 | 288 | 3.16 | < 0.001 |
| < 30 | 469 | 1100 | 1.74 | < 0.001 |
| 30-180 | 940 | 2447 | 1.09 | 0.11 |
| >180 | 2046 | 6484 | 0.91 | 0.01 |
A meta-analysis conducted by Johnstone et al[52] included six studies evaluating nearly 1 million patients. Despite the heterogeneity of the studies included in the meta-analysis, there was a significant increase in the incidence of pneumonia in patients taking PPIs. This increased incidence appeared to be limited to the short-term exposure, specifically the first 30 d. There was no difference in the risk of pneumonia in patients chronically exposed to PPIs.
A second meta-analysis conducted by Eom et al[53] expanded their analysis to include 18 studies. Similar to the Johstone meta-analysis, there was significant heterogeneity in the study population and PPIs use was associated with a 27% increase in the incidence of pneumonia.
A retrospective cohort study by Beaulieu[54] was conducted in a medical intensive care unit in a 554 bed, university-affiliated, academic medical center. The patients included in the study were admitted to the MICU of L,Hopital du Sacre-Coeur de Montreal between March 14, 2002 and May 31, 2004. Nosocomial pneumonia was defined as a pneumonia diagnosis made after the first 48 h of their ICU admission. Of the 787 patients included, 104 patients developed nosocomial pneumonia and 24 of these died within 30 d of being diagnosed. During this study, 37.1% (292 patients) of the patients received a PPI with a mean duration of use of 6.6 d. Unfortunately, there was no information regarding the number of patients who received PPI therapy in each group and if the patients who died had received PPIs. There was no statistically significant difference in the risk of pneumonia between patients who received PPIs relative to non-exposed patients.
Although this study did not find an association between the administration of PPI therapy and the incidence of pneumonia, it did find three statistically significant associations pertaining specifically to the ICU patient. Statistical significance was achieved with the administration of sedatives or neuromuscular antagonists for two or more days, an APACHE II score greater than 15 and the presence of a central venous catheter.
Herzig[5] conducted a prospective study which included 63 878 admissions to a large, urban, academic medical center in Boston. Patients were hospitalized for at least three days without admission to the ICU between January 1, 2004 and December 31, 2007. The three day inclusion criterion was consistent with the Infectious Diseases Society of America guidelines[55,56]. PPI exposure was defined as a prescription written during the patient’s hospitalization.
The Herzig study[5] assessed the incidence of HAP in the general inpatient population. The total incidence of HAP in the 2219 study population was 3.5%. The patients exposed to PPIs within 48 h of admission had a higher unadjusted odds ratio of 2.8 (95% CI: 2.5-3.1) relative to the unexposed patients (5.3% vs 2.0% respectively). The odds ratio declined to 1.3 (95% CI: 1.1-1.4) when adjusted for co-morbidities but the difference was still statistically significant (Table 4). In contrast to PPIs, H2RAs demonstrated an unadjusted odds ratio of 1.6 (95% CI: 1.3-1.9) but this fell to an adjusted rate of 1.2 (95% CI: 0.98-1.4) which failed to meet statistical significance.
| Characteristic | Acid-suppressive exposure | No acid-suppressive exposure | Unadjusted OR (95% CI) | Adjusted OR (95% CI) |
| PPI | ||||
| Total admissions | 25 374 | 30 956 | 56 330 | 56 330 |
| HAP (%) | 1340 (5.3) | 610 (2.0) | 2.8 (2.5-3.1) | 1.3 (1.1-1.4) |
| H2RA | ||||
| Total admissions | 5686 | 30 956 | 36 642 | 36 642 |
| HAP (%) | 176 (3.1) | 610 (2.0) | 1.6 (1.3-1.9) | 1.2 (0.98-1.4) |
Although the association of PPI administration and pneumonia remained significant when subdivided into aspiration pneumonia and non-aspiration pneumonia, there was a stronger association with aspiration pneumonia (1.4, 95% CI: 1.1-1.8) than non-aspiration pneumonia (1.2, 95% CI: 1.1-1.4) (Table 5). This may suggest that some of the increased incidence of pneumonia when patients are co-administered PPIs may be due to the risk of aspiration pneumonia. However, only 22% of the cases of HAP in this population were attributed to aspiration pneumonia.Unfortunately, more studies need to be conducted to determine the significance of aspiration pneumonia with PPI administration.
| Outcome | Acid-suppressive exposure (%) | No acid-suppressive exposure (%) | Adjusted OR (95% CI) | Propensity-matched |
| Number of patients | 32 922 | 30 956 | 63 878 | 32 792 |
| HAP | 1609 (4.9) | 610 (2.0) | 1.3 (1.1-1.4) | 1.3 (1.1-1.4) |
| Aspiration PNA | 361 (1.1) | 112 (0.4) | 1.4 (1.1-1.8) | 1.4 (1.1-1.8) |
| Non-Aspiration PNA | 1262 (3.8) | 501 (1.6) | 1.2 (1.1-1.4) | 1.2 (1.1-1.4) |
The initial baseline characteristics revealed significant differences between the groups exposed versus not exposed to PPIs. A majority of these differences were negated once the groups were assessed in a propensity-matched analysis. The propensity-matched analysis paired 16 396 patients from each group and reassessed the significance of the PPI exposure. The baseline characteristics were more similar; however, a statistically significant association between PPI exposure and pneumonia incidence was seen. This association was also maintained when comparing aspiration and non-aspiration pneumonia with PPI exposure. The odds ratio was still 1.3 with a 95% confidence interval between 1.1 and 1.4.
The Herzig study[5] also conducted a sensitivity analysis to determine the reliability of the pneumonia diagnoses made for this study. The study significance would still be maintained despite a 5% miscalculation rate of pneumonia diagnosis between the two groups. In order to determine the misdiagnosis rate in this study, 100 patient charts were independently screened which were previously deemed not to have pneumonia and only one misdiagnosis was detected. This rendered a 1% error with an upper bound confidence interval of 2.95%. This 2.95% error rate was less than the 5% error rate required to change the significance of the findings.
This study extrapolated the risk factors of PPI administration. With 40 million discharges per year and 50% of these patients receiving PPIs, there are 20 million discharged patients exposed to PPIs annually. With an odds ratio of 1.3, the incidence of HAP increases by 30% with PPI administration. Based on the Herzig study[5], knowing 3.5% of patients acquire HAP and a 52% PPI exposure rate, the total risk of acquiring HAP from PPI exposure is 0.9% (NNH = 111). 0.9% correlates to 180 000 cases of HAP annually due to PPI exposure. With a HAP mortality of 18%, nearly 33 000 deaths per year may be prevented with limited PPI administration[5].
A retrospective cohort study by Miano[57] included 834 patients in a cardiothoracic surgery database who received stress ulcer prophylaxis with either pantoprazole or ranitidine. The patients included in this study were at least 18 years of age and were admitted to the Wake Forest University Baptist Medical Center cardiothoracic surgery service between January 2004 and March 2007. The admitting physician determined whether the patient received pantoprazole or ranitidine but, for inclusion in this study, the patient could not switch between the two agents during their first admission. Patients were excluded if they had a diagnosis of pneumonia within three months of their first admission or if they had documented aspiration throughout their hospitalization. Initially, the baseline characteristics were statistically significantly different for age, incidence of reflux disease, New York Heart Association class IV heart failure, duration of mechanical ventilation, use of blood products and need for tracheostomy. This was adjusted for using a propensity matching score.
This study determined nosocomial pneumonia occurred in 35/377 (9.3%) of patients with pantoprazole compared to just 7/457 (1.5%) in the ranitidine treated population (OR = 6.6, 95% CI: 2.9-14.9). In the pantoprazole treated population, 31 of the 35 (88.5%) pneumonia cases were classified as ventilator-associated pneumonia. After propensity was adjusted, multivariable logistic regression, pantoprazole was found to be an independent risk factor for nosocomial pneumonia (OR = 2.7, 95% CI: 1.1 to 6.7, P = 0.034).
Larger controlled trials still need to be done to better identify the risk that PPIs impart towards patients contracting community and/or hospital-acquired pneumonia. Until these are completed, we will have to continue to use smaller controlled trials to predict the associated risks in our respective patient populations. Studies which can provide more definitive differentiation between the diagnosis of pneumonia versus asthma, COPD or indigestion need to be conducted to fine-tune the association. In the interim, it appears prudent to limit the use of PPIs to situations where they are clinically indicated and, in such cases, use them for the shortest possible length of time and at the lowest effective dose. In the case of prescribing for stress ulcer prophylaxis in ICU patients, perhaps H2RAs should be used as the preferred agents over PPIs.
Peer reviewers: Dr. Andreas Marc Palmer, PhD, Department of Medicinal Chemistry, Nycomed GmbH, Byk Gulden Str. 2, Konstanz 78467, Germany; Mark Anthony A De Lusong, MD, Associate Professor, Section of Gastroenterology, University of the Philippines-Philippine General Hospital, Taft Avenue, Ermita, Manila 1200, Philippines
S- Editor Wang JL L- Editor Roemmele A E- Editor Zhang L
| 1. | Sarkar M, Hennessy S, Yang YX. Proton-pump inhibitor use and the risk for community-acquired pneumonia. Ann Intern Med. 2008;149:391-398. [PubMed] [Cited in This Article: ] |
| 2. | Available from: http: //www.marketingcharts.com/wp/wp-content/uploads/2009/03/ims-top-15-us-pharmaceutical-products-sales-2008.jpg. [Cited in This Article: ] |
| 3. | Forgacs I, Loganayagam A. Overprescribing proton pump inhibitors. BMJ. 2008;336:2-3. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 338] [Cited by in F6Publishing: 349] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 4. | Nardino RJ, Vender RJ, Herbert PN. Overuse of acid-suppressive therapy in hospitalized patients. Am J Gastroenterol. 2000;95:3118-3122. [PubMed] [Cited in This Article: ] |
| 5. | Herzig SJ, Howell MD, Ngo LH, Marcantonio ER. Acid-suppressive medication use and the risk for hospital-acquired pneumonia. JAMA. 2009;301:2120-2128. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 319] [Cited by in F6Publishing: 284] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 6. | Pham CQ, Regal RE, Bostwick TR, Knauf KS. Acid suppressive therapy use on an inpatient internal medicine service. Ann Pharmacother. 2006;40:1261-1266. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 100] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 7. | Yang YX, Lewis JD, Epstein S, Metz DC. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947-2953. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 845] [Cited by in F6Publishing: 777] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 8. | Fournier MR, Targownik LE, Leslie WD. Proton pump inhibitors, osteoporosis, and osteoporosis-related fractures. Maturitas. 2009;64:9-13. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 9. | Kaye JA, Jick H. Proton pump inhibitor use and risk of hip fractures in patients without major risk factors. Pharmacotherapy. 2008;28:951-959. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 133] [Cited by in F6Publishing: 143] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 10. | Wright MJ, Proctor DD, Insogna KL, Kerstetter JE. Proton pump-inhibiting drugs, calcium homeostasis, and bone health. Nutr Rev. 2008;66:103-108. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Aseeri M, Schroeder T, Kramer J, Zackula R. Gastric acid suppression by proton pump inhibitors as a risk factor for clostridium difficile-associated diarrhea in hospitalized patients. Am J Gastroenterol. 2008;103:2308-2313. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 176] [Cited by in F6Publishing: 165] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 12. | Cote G, Howden C. Potential adverse effects of proton pump inhibitors. Curr Gastroenterol Rep. 2008;10:208-214. [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Pham C, Sadowski-Hayes L, Regal R. Prevalent Prescribing of Proton Pump Inhibitors: Prudent or Pernicious? P&T. 2006;31:159-167. [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 14. | Sachs G, Shin J, Howden C. Review article: the clinical pharmacology of proton pump inhibitors. Aliment Pharmacol Ther. 2006;23 Suppl 2:2-8. [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 41] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Savarino V, Di Mario F, Scarpignato C. Proton pump inhibitors in GORD An overview of their pharmacology, efficacy and safety. Pharmacol Res. 2009;59:135-153. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 127] [Cited by in F6Publishing: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 16. | Schubert ML. Gastric secretion. Curr Opin Gastroenterol. 2010;26:598-603. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 17. | Bartlett JG, Dowell SF, Mandell LA, File Jr TM, Musher DM, Fine MJ. Practice guidelines for the management of community-acquired pneumonia in adults. Infectious Diseases Society of America. Clin Infect Dis. 2000;31:347-382. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1078] [Cited by in F6Publishing: 1002] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 18. | Hartmann M, Theiss U, Huber R, Lühmann R, Bliesath H, Wurst W, Lücker PW. Twenty-four-hour intragastric pH profiles and pharmacokinetics following single and repeated oral administration of the proton pump inhibitor pantoprazole in comparison to omeprazole. Aliment Pharmacol Ther. 1996;10:359-366. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 62] [Cited by in F6Publishing: 64] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 19. | Hunt RH, Armstrong D, James C, Chowdhury SK, Yuan Y, Fiorentini P, Taccoen A, Cohen P. Effect on intragastric pH of a PPI with a prolonged plasma half-life: comparison between tenatoprazole and esomeprazole on the duration of acid suppression in healthy male volunteers. Am J Gastroenterol. 2005;100:1949-1956. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 20. | Ivanovich P, Fellows H, Rich C. The absorption of calcium carbonate. Ann Intern Med. 1967;66:917-923. [PubMed] [Cited in This Article: ] |
| 21. | Scarpignato C, Hunt RH. Proton pump inhibitors: the beginning of the end or the end of the beginning? Curr Opin Pharmacol. 2008;8:677-684. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 22. | DeVault K, Talley N. Insights into the future of gastric acid suppression. Nat Rev Gastroenterol Hepatol. 2009;6:524-532. [DOI] [Cited in This Article: ] [Cited by in Crossref: 54] [Cited by in F6Publishing: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 23. | Hoorn EJ, van der Hoek J, de Man RA, Kuipers EJ, Bolwerk C, Zietse R. A case series of proton pump inhibitor-induced hypomagnesemia. Am J Kidney Dis. 2010;56:112-116. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 141] [Cited by in F6Publishing: 154] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 24. | Furlanetto T, Faulhaber G. Hypomagnesemia and Proton Pump Inhibitors: Below the Tip of the Iceberg. Arch Intern Med. 2011;(Epub ahead of print). [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Mullin JM, Gabello M, Murray LJ, Farrell CP, Bellows J, Wolov KR, Kearney KR, Rudolph D, Thornton JJ. Proton pump inhibitors: actions and reactions. Drug Discov Today. 2009;14:647-660. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 26. | Vesper BJ, Jawdi A, Altman KW, Haines GK, Tao L, Radosevich JA. The effect of proton pump inhibitors on the human microbiota. Curr Drug Metab. 2009;10:84-89. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 105] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 27. | Yang YX, Metz DC. Safety of proton pump inhibitor exposure. Gastroenterology. 2010;139:1115-1127. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 152] [Cited by in F6Publishing: 152] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 28. | Recker RR. Calcium absorption and achlorhydria. N Engl J Med. 1985;313:70-73. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 377] [Cited by in F6Publishing: 290] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 29. | O’Connell MB, Madden DM, Murray AM, Heaney RP, Kerzner LJ. Effects of proton pump inhibitors on calcium carbonate absorption in women: a randomized crossover trial. Am J Med. 2005;118:778-781. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 292] [Cited by in F6Publishing: 301] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 30. | Hollis I, Regal R. Calcium and vitamin D in the prevention and treatment of osteoporosis. P&T. 2007;32:502-513. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 292] [Cited by in F6Publishing: 301] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 31. | Richards MJ, Edwards JR, Culver DH, Gaynes RP. Nosocomial infections in medical intensive care units in the United States. National Nosocomial Infections Surveillance System. Crit Care Med. 1999;27:887-892. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1053] [Cited by in F6Publishing: 1119] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 32. | Thorens J, Froehlich F, Schwizer W, Saraga E, Bille J, Gyr K, Duroux P, Nicolet M, Pignatelli B, Blum AL. Bacterial overgrowth during treatment with omeprazole compared with cimetidine: a prospective randomised double blind study. Gut. 1996;39:54-59. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 239] [Cited by in F6Publishing: 242] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 33. | Dial S, Alrasadi K, Manoukian C, Huang A, Menzies D. Risk of Clostridium difficile diarrhea among hospital inpatients prescribed proton pump inhibitors: cohort and case-control studies. CMAJ. 2004;171:33-38. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 418] [Cited by in F6Publishing: 437] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 34. | Choudhry M, Soran H, Ziglam H. Overuse and inappropriate prescribing of proton pump inhibitors in patients with Clostridium difficile-associated disease. Q J Med. 2008;101:445-448. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 418] [Cited by in F6Publishing: 437] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 35. | Dalton B, Lye-Maccannall T, Henderson E, Maccannell D, Louie T. Proton pump inhibitors increase significantly the risk of clostridium difficile infection in a low-endemicity, non-outbreak hospital setting. Aliment Pharmacol Ther. 2009;29:626-634. [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 36. | Nachnani JS, Bulchandani D, Allen MJ. Proton pump inhibitors are an independent risk factor for an increased length of hospital stay in patients with Clostridium difficile infection. Indian J Gastroenterol. 2008;27:171-172. [PubMed] [Cited in This Article: ] |
| 37. | Cadle RM, Mansouri MD, Logan N, Kudva DR, Musher DM. Association of proton-pump inhibitors with outcomes in Clostridium difficile colitis. Am J Health Syst Pharm. 2007;64:2359-2363. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 78] [Cited by in F6Publishing: 80] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 38. | Laheij RJ, Sturkenboom MC, Hassing RJ, Dieleman J, Stricker BH, Jansen JB. Risk of community-acquired pneumonia and use of gastric acid-suppressive drugs. JAMA. 2004;292:1955-1960. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 625] [Cited by in F6Publishing: 652] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 39. | Altman K, Haines G, Hammer N, Radosevich J. The H /K -ATPase (proton) pump is expressed in human laryngeal submucosal glands. Larygnoscope. 2003;113:1927-1930. [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 40. | Altman KW, Waltonen JD, Hammer ND, Radosevich JA, Haines GK. Proton pump (H+/K+-ATPase) expression in human laryngeal seromucinous glands. Otolaryngol Head Neck Surg. 2005;133:718-724. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 41. | Altman KW, Waltonen JD, Tarjan G, Radosevich JA, Haines GK. Human lung mucous glands manifest evidence of the H+/K+-ATPase proton pump. Ann Otol Rhinol Laryngol. 2007;116:229-234. [PubMed] [Cited in This Article: ] |
| 42. | Eckley C, Costa H. Comparative study of salivary pH and volume in adults with chronic laryngopharyngitis by gastroesophageal reflux disease before and after treatment. Rev Bras Otorrinolaringol. 2006;71:55-60. [PubMed] [Cited in This Article: ] |
| 43. | Aybay C, Imir T, Okur H. The effect of omeprazole on human natural killer cell activity. Gen Pharmac. 1995;26:1416-1418. [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 42] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 44. | Zedtwitz-Liebenstein K, Wenisch C, Patruta S, Parschalk B, Daxböck F, Graninger W. Omeprazole treatment diminishes intra- and extracellular neutrophil reactive oxygen production and bactericidal activity. Crit Care Med. 2002;30:1118-1122. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 150] [Cited by in F6Publishing: 149] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 45. | Capodicasa E, De Bellis F, Pelli MA. Effect of lansoprazole on human leukocyte function. Immunopharmacol Immunotoxicol. 1999;21:357-377. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 46. | Gregor JC. Acid suppression and pneumonia: a clinical indication for rational prescribing. JAMA. 2004;292:2012-2013. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.2] [Reference Citation Analysis (1)] |
| 47. | Gulmez SE, Holm A, Frederiksen H, Jensen TG, Pedersen C, Hallas J. Use of proton pump inhibitors and the risk of community-acquired pneumonia: a population-based case-control study. Arch Intern Med. 2007;167:950-955. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 242] [Cited by in F6Publishing: 257] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 48. | Dublin S, Walker RL, Jackson ML, Nelson JC, Weiss NS, Jackson LA. Use of proton pump inhibitors and H2 blockers and risk of pneumonia in older adults: a population-based case-control study. Pharmacoepidemiol Drug Saf. 2010;19:792-802. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 49. | Roughead EE, Ramsay EN, Pratt NL, Ryan P, Gilbert AL. Proton-pump inhibitors and the risk of antibiotic use and hospitalisation for pneumonia. Med J Aust. 2009;190:114-116. [PubMed] [Cited in This Article: ] |
| 50. | Myles PR, Hubbard RB, McKeever TM, Pogson Z, Smith CJ, Gibson JE. Risk of community-acquired pneumonia and the use of statins, ace inhibitors and gastric acid suppressants: a population-based case-control study. Pharmacoepidemiol Drug Saf. 2009;18:269-275. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 77] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 51. | Rodríguez LA, Ruigómez A, Wallander MA, Johansson S. Acid-suppressive drugs and community-acquired pneumonia. Epidemiology. 2009;20:800-806. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 53] [Cited by in F6Publishing: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 52. | Johnstone J, Nerenberg K, Loeb M. Meta-analysis: proton pump inhibitor use and the risk of community-acquired pneumonia. Aliment Pharmacol Ther. 2010;31:1165-1177. [DOI] [Cited in This Article: ] [Cited by in Crossref: 117] [Cited by in F6Publishing: 110] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 53. | Eom CS, Jeon CY, Lim JW, Cho EG, Park SM, Lee KS. Use of acid-suppressive drugs and risk of pneumonia: a systematic review and meta-analysis. CMAJ. 2011;183:310-319. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 272] [Cited by in F6Publishing: 286] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 54. | Beaulieu M, Williamson D, Sirois C, Lachaine J. Do proton-pump inhibitors increase the risk for nosocomial pneumonia in a medical intensive care unit? J Crit Care. 2008;23:513-518. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 55. | Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171:388-416. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4266] [Cited by in F6Publishing: 4099] [Article Influence: 215.7] [Reference Citation Analysis (0)] |
| 56. | Mandell LA, Wunderink RG, Anzueto A, Bartlett JG, Campbell GD, Dean NC, Dowell SF, File TM, Musher DM, Niederman MS. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44 Suppl 2:S27-S72. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3978] [Cited by in F6Publishing: 4053] [Article Influence: 238.4] [Reference Citation Analysis (0)] |
| 57. | Miano TA, Reichert MG, Houle TT, MacGregor DA, Kincaid EH, Bowton DL. Nosocomial pneumonia risk and stress ulcer prophylaxis: a comparison of pantoprazole vs ranitidine in cardiothoracic surgery patients. Chest. 2009;136:440-447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 69] [Article Influence: 4.6] [Reference Citation Analysis (0)] |