Published online Jun 21, 2013. doi: 10.3748/wjg.v19.i23.3703
Revised: February 27, 2013
Accepted: April 3, 2013
Published online: June 21, 2013
Processing time: 184 Days and 0.1 Hours
Increased risk due to nonsteroidal anti-inflammatory drugs (NSAIDs) therapy has been observed in patients. Although diaphragm-like stricture in the small bowel and colon induced by NSAIDs therapy has been rarely reported, gastric body diaphragm-like stricture has not been reported. We describe the first case of gastric body diaphragm-like stricture due to NSAIDs in a 44-year-old male patient who was successfully treated by an endoscopic approach to avoid complicated surgery. This case highlights new insight into the disadvantages of NSAIDs and provides new data for future clinical studies.
Core tip: The major disadvantage of nonsteroidal anti-inflammatory drugs (NSAIDs) therapy is the potential to induce adverse gastrointestinal effects. However, diaphragm disease is a rare complication of long-term NSAIDs use. In this study, the first case of NSAIDs-induced diaphragm-like stricture in the gastric body is reported which was successfully treated by an endoscopic approach to avoid a complicated surgical intervention.
- Citation: Wu LL, Yang YS, Cai FC, Wang SF. Gastric body diaphragm-like stricture as a rare complication of nonsteroidal anti-inflammatory drugs. World J Gastroenterol 2013; 19(23): 3703-3706
- URL: https://www.wjgnet.com/1007-9327/full/v19/i23/3703.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i23.3703
Nonsteroidal anti-inflammatory drugs (NSAIDs) are known to cause erosion, ulceration, occult bleeding and subsequent stricture formation in the gastrointestinal tract. A rare NSAIDs-induced complication is the formation of diaphragm-like strictures in the intestine[1-4]. Herein, we report, to our knowledge, the first case of NSAIDs-induced diaphragm-like stricture in the gastric body successfully treated by an endoscopic approach to avoid a complicated surgical intervention.
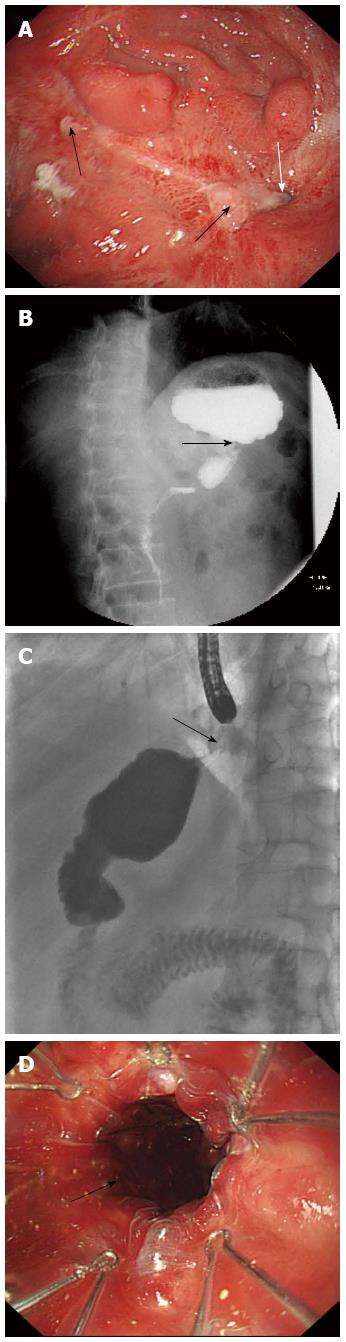
A 44-year-old male patient presented with a 2-mo history of abdominal distention and vomiting. There was temporary relief after vomiting and the vomitus was composed of gastric contents. The patient had taken more than 15 g of compound aminopyrine phenacetin tablets by mistake whilst drunk three months previously. On physical examination, his abdomen was tender without any other relevant physical findings. Routine blood and biochemical tests were normal, and tumor markers were within the normal ranges. Gastroscopy showed multiple erosions, ulcers and nodular changes in the proximal gastric body, in which the largest ulcer was about 1.0 cm × 1.5 cm (Figure 1A, black arrow). Deformation and stricture of the gastric body was noted, and the gastroscope was unable to pass through the stricture (Figure 1A, white arrow). Biopsy pathology at the ulcer edges revealed inflammation and other benign changes. Organic iodine solution radiography of the upper gastrointestinal tract showed a very thin stricture in the gastric body (Figure 1B). Subsequently, a pediatric gastroscope was used, but also failed to pass through the stricture. Repeat biopsy pathology confirmed the previous histology of benign inflammation, and immunohistochemistry showed weakly positive results for CD3, CD43, CD20, Ki-67 and CK, which were consistent with the drug-induced gastric body benign ulcers and stricture. Endoscopic transcatheter radiography was then performed which demonstrated that the gastric body stricture was a diaphragm-like stricture (Figure 1C). The patient underwent balloon dilation and placement of a metal stent (Figure 1D); his symptoms resolved after these endoscopic procedures. On follow-up 2 mo later, the patient had no symptoms.
NSAIDs, such as aspirin, indomethacin, diclofenac, and compound aminopyrine phenacetin, are the most commonly prescribed drugs for inflammation, arthritis and cardiovascular protection. However, a major disadvantage of NSAIDs therapy is the potential to induce adverse gastrointestinal effects, particularly in the stomach and duodenum. It has been reported that as many as 25% of chronic NSAIDs users may develop ulcer disease, and 2%-4% of these ulcers may bleed or perforate[5,6]. NSAIDs are absorbed into enterocytes and then uncouple mitochondrial oxidative phosphorylation, resulting in the dysfunction of tight intracellular junctions and intestinal permeability. Enterocytes are thereby exposed to luminal aggressive contents, leading to inflammation and ulceration[7]. Recent clinical research shed light on NSAID-induced small intestinal mucosal damage including erosions and ulcerations, which occur more often than previously expected[8]. Graham et al[9] reported that 70% of patients who took NSAIDs for > 3 mo had small intestinal ulcers and erosions shown by capsule endoscopy.
Diaphragm disease induced by NSAIDs, first described by Lang et al[1] in 1988, is a rare and severe complication of long-term NSAIDs use, especially in elderly patients[2]. Although diaphragm-like stricture of the small bowel was not associated with the use of NSAIDs in recent reports[10], diaphragm disease was thought at one time to be a unique form of intestinal pathology associated with NSAIDs administration. Disease may occur in 2% of NSAIDs users in the small bowel[3], commonly in the ileum[2,4], or in the duodenum in some cases[11,12]. In the past decade, diaphragm-like strictures in the large intestine due to adverse effects of NSAIDs have been increasingly reported[13,14].
The precise pathogenesis of diaphragm disease is unclear, however, the main histological abnormalities include thickening and chaotic arrangement of muscular bundles in the muscularis mucosae, fibrosis of the lamina propriae and mucosal ulceration[15]. Therefore, affected patients frequently present with gastrointestinal obstructive symptoms and often require surgical treatment.
Although diaphragm-like strictures have been reported in the small bowel and colon, strictures in the gastric body have not been documented in the literature. This may be because the gastric body not only has a wider lumen or space, but also has a thick muscularis. We recently experienced a rare case which occurred after ingestion of a large quantity of NSAIDs. It is known that endoscopic dilation, surgical resection and suspension of NSAIDs administration are common treatment options depending on the position, length and severity of the stricture. In the present middle-age patient, we successfully used the minimally invasive treatment modalities endoscopic dilation and placement of a metal stent, which avoided complicated surgical management.
More and more cases induced by NSAIDs have been reported in the literature, including gastrocolic fistula[16], Brar et al[17] reported a case of perforation, and even a case of Crohn’s disease was reported which was endoscopically and histologically misinterpreted[18]. Due to the increasing world-wide use of NSAIDs, NSAID-related gastrointestinal complications still continue to be a major concern and require more therapeutic strategies[19-24]. This study, for the first time, reports a rare case of diaphragm-like gastric body stricture which was successfully treated by an endoscopic approach. The endoscope is a useful tool for the diagnosis and treatment of suspected NSAIDs-related gastrointestinal complications.
P- Reviewers Higuchi K, Slomiany BL S- Editor Zhai HH L- Editor Webster JR E- Editor Zhang DN
| 1. | Lang J, Price AB, Levi AJ, Burke M, Gumpel JM, Bjarnason I. Diaphragm disease: pathology of disease of the small intestine induced by non-steroidal anti-inflammatory drugs. J Clin Pathol. 1988;41:516-526. [PubMed] |
| 2. | Puri A, Agarwal AK, Garg S, Tyagi P, Sakhuja P. Diaphragm disease of the ileum: a case report. Trop Gastroenterol. 2006;27:46-47. [PubMed] |
| 3. | Maiden L, Thjodleifsson B, Seigal A, Bjarnason II, Scott D, Birgisson S, Bjarnason I. Long-term effects of nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 selective agents on the small bowel: a cross-sectional capsule enteroscopy study. Clin Gastroenterol Hepatol. 2007;5:1040-1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 190] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 4. | Chernolesskiy A, Lanzon-Miller S, Hill F, Al-Mishlab T, Thway Y. Subacute small bowel obstruction due to diaphragm disease. Clin Med. 2010;10:296-298. [PubMed] |
| 5. | Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, Makuch R, Eisen G, Agrawal NM, Stenson WF. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: A randomized controlled trial. Celecoxib Long-term Arthritis Safety Study. JAMA. 2000;284:1247-1255. [PubMed] |
| 6. | Lanza FL, Chan FK, Quigley EM. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol. 2009;104:728-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 421] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 7. | Matsui H, Shimokawa O, Kaneko T, Nagano Y, Rai K, Hyodo I. The pathophysiology of non-steroidal anti-inflammatory drug (NSAID)-induced mucosal injuries in stomach and small intestine. J Clin Biochem Nutr. 2011;48:107-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 175] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 8. | Higuchi K, Umegaki E, Watanabe T, Yoda Y, Morita E, Murano M, Tokioka S, Arakawa T. Present status and strategy of NSAIDs-induced small bowel injury. J Gastroenterol. 2009;44:879-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 187] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 9. | Graham DY, Opekun AR, Willingham FF, Qureshi WA. Visible small-intestinal mucosal injury in chronic NSAID users. Clin Gastroenterol Hepatol. 2005;3:55-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Wang ML, Miao F, Tang YH, Zhao XS, Zhong J, Yuan F. Special diaphragm-like strictures of small bowel unrelated to non-steroidal anti-inflammatory drugs. World J Gastroenterol. 2011;17:3596-3604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 11. | Blinder GH, Hautekeete ML, Holvoet JP, Kockx MM, Hubens HK. Duodenal diaphragmlike stricture induced by acetylsalicylic acid. Dig Dis Sci. 1994;39:1365-1369. [PubMed] |
| 12. | Ammori BJ. Laparoscopic pancreas-preserving distal duodenectomy for duodenal stricture related to nonsteroidal antiinflammatory drugs (NSAIDs). Surg Endosc. 2002;16:1362-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Kurahara K, Matsumoto T, Iida M, Honda K, Yao T, Fujishima M. Clinical and endoscopic features of nonsteroidal anti-inflammatory drug-induced colonic ulcerations. Am J Gastroenterol. 2001;96:473-480. [PubMed] |
| 14. | Klein M, Linnemann D, Rosenberg J. Non-steroidal anti-inflammatory drug-induced colopathy. BMJ Case Rep. 2011;2011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | De Petris G, López JI. Histopathology of diaphragm disease of the small intestine: a study of 10 cases from a single institution. Am J Clin Pathol. 2008;130:518-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Yarze JC. Gastrocolic fistula related to NSAID-induced gastric ulcer. Gastrointest Endosc. 2011;74:687-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 17. | Brar AS, Gill RS, Gill SS, Wang H. NSAID-Associated Perforation of a Meckels Diverticulum: A Case Report. J Clin Med Res. 2011;3:96-98. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Stolte M, Hartmann FO. Misinterpretation of NSAID-induced Colopathy as Crohn’s disease. Z Gastroenterol. 2010;48:472-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 19. | Bardou M, Barkun AN. Preventing the gastrointestinal adverse effects of nonsteroidal anti-inflammatory drugs: from risk factor identification to risk factor intervention. Joint Bone Spine. 2010;77:6-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 20. | Scarpignato C, Hunt RH. Nonsteroidal antiinflammatory drug-related injury to the gastrointestinal tract: clinical picture, pathogenesis, and prevention. Gastroenterol Clin North Am. 2010;39:433-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 164] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 21. | Uc A, Zhu X, Wagner BA, Buettner GR, Berg DJ. Heme oxygenase-1 is protective against nonsteroidal anti-inflammatory drug-induced gastric ulcers. J Pediatr Gastroenterol Nutr. 2012;54:471-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 22. | Lanas A. Gastrointestinal bleeding associated with low-dose aspirin use: relevance and management in clinical practice. Expert Opin Drug Saf. 2011;10:45-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 23. | Ng FH, Wong SY, Lam KF, Chu WM, Chan P, Ling YH, Kng C, Yuen WC, Lau YK, Kwan A. Famotidine is inferior to pantoprazole in preventing recurrence of aspirin-related peptic ulcers or erosions. Gastroenterology. 2010;138:82-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 24. | Sugano K, Kontani T, Katsuo S, Takei Y, Sakaki N, Ashida K, Mizokami Y, Asaka M, Matsui S, Kanto T. Lansoprazole for secondary prevention of gastric or duodenal ulcers associated with long-term non-steroidal anti-inflammatory drug (NSAID) therapy: results of a prospective, multicenter, double-blind, randomized, double-dummy, active-controlled trial. J Gastroenterol. 2012;47:540-552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |