Published online Oct 7, 2010. doi: 10.3748/wjg.v16.i37.4697
Revised: July 6, 2010
Accepted: July 13, 2010
Published online: October 7, 2010
AIM: To determine whether the serum level of matrix metalloproteinase-7 (MMP7) has the potential to diagnosis cholangiocarcinoma from benign biliary tract diseases.
METHODS: This study was performed according to the PRoBE (a prospective-specimen-collection, retrospective-blinded-evaluation) design. A total of 187 patients with obstructive jaundice were consecutively enrolled. After the diagnostic status of these patients was ascertained, their levels of serum MMP7 were assayed and compared with serum carbohydrate antigen 19-9 (CA19-9). This was conducted in a blinded case (cholangiocarcinoma)-control (benign biliary tract disease) setup.
RESULTS: MMP7 and CA19-9 serum levels were significantly elevated in cholangiocarcinoma patients (P < 0.001). The area under the curve (AUC) from a receiver operating characteristic (ROC) curve analysis for the diagnosis of cholangiocarcinoma, using MMP7 was more accurate than CA19-9 (AUC = 0.84, 95% CI: 0.778-0.903 for MMP7 and AUC = 0.79, 95% CI: 0.708-0.868 for CA19-9). The sensitivity and specificity of serum MMP7 (cut-off value of 5.5 ng/mL) was 75% and 78%, respectively, while the sensitivity and specificity of serum CA19-9 (cut-off value of 100 U/mL) was 68% and 87%, respectively.
CONCLUSION: Serum values of MMP7 and CA19-9 appear to be useful biomarkers for differentiating cholangiocarcinoma from benign biliary tract obstructive diseases.
- Citation: Leelawat K, Narong S, Wannaprasert J, Ratanashu-ek T. Prospective study of MMP7 serum levels in the diagnosis of cholangiocarcinoma. World J Gastroenterol 2010; 16(37): 4697-4703
- URL: https://www.wjgnet.com/1007-9327/full/v16/i37/4697.htm
- DOI: https://dx.doi.org/10.3748/wjg.v16.i37.4697
Cholangiocarcinoma is one of the most common causes of cancer-related mortality in Thailand[1]. The high mortality rate of cholangiocarcinoma is due to the aggressiveness of tumors that are often discovered at a late-stage of disease progression[2]. To improve the survival rate, the diagnosis and treatment of these patients should be performed as soon as possible. Bile duct obstruction is the focal symptom that the vast majority of patients with cholangiocarcinoma present with at hospital. However, there are many cases of benign biliary tract diseases including common bile duct stone and bile duct stricture. These other conditions often present with clinical symptoms similar to those of patients with cholangiocarcinoma. In addition, it is very difficult to obtain pathological tissue for the diagnosis of cholangiocarcinoma due to both the desmoplastic reaction and the tumor location[3,4]. Brush cytology has a sensitivity of only 62.5% for detecting cholangiocarcinoma[5]. Owing to the differences in treatment and prognosis between cholangiocarcinoma and benign biliary tract diseases, the most important issue is to obtain a reliable method to differentially diagnosis patients with cholangiocarcinoma from those with benign biliary tract diseases. Identification of tumor markers in the serum would aid in the accurate diagnosis of cholangiocarcinoma.
To date, carbohydrate antigen 19-9 (CA19-9) is used as a tumor marker for detecting cholangiocarcinoma. The sensitivity and specificity of CA19-9 in diagnosing cholangiocarcinoma were shown to be 53%-89% and 80.5%-86%, respectively[6-9]. Unfortunately, elevated serum levels of CA19-9 have also been found in patients with benign obstructive jaundice[10,11]. Due to this, CA19-9 is a not a reliable marker for differentiating cholangiocarcinoma from benign obstructive jaundice.
Previous studies demonstrated that high expression of matrix metalloproteinase-7 (MMP7) could be readily detected in cholangiocarcinoma specimens[12-14]. We previously performed research to study the serum levels of CA19-9, CEA, MMP9 and MMP7 in patients with obstructive jaundice[6]. This previous study was performed using a case-control design and serum collected from a serum bank. The results showed that only the level of MMP7 was significantly higher in patients with cholangiocarcinoma compared to patients suffering from benign biliary tract disease. In addition, when comparing the areas under the curve of the receiver operating characteristic (ROC) for CEA, CA19-9 and MMP9, a ROC curve analysis demonstrated that the detection of MMP7 in serum was the most accurate for differentiating cholangiocarcinoma from benign biliary tract disease. This finding indicated that serum MMP7 should be used as a tumor marker for the diagnosis of cholangiocarcinoma in patients with obstructive jaundice.
According to the study of biomarker use, it is now widely appreciated that the evaluation of biomarker performance must be separated from biomarker discovery. In discovery research, its performance in samples may be biased in an overoptimistic direction. To estimate performance without bias, an independent dataset should be investigated[15-18]. Therefore, the aim of the present study was to evaluate the performance of serum MMP7 and CA19-9 for their potential in the diagnosis of cholangiocarcinoma. We used a new and independent dataset of prospective consecutive cases with evidence of bile duct obstruction due to various etiologies. This study was performed according to the PRoBE (a prospective-specimen-collection, retrospective-blinded-evaluation) design[15]. We collected serum from a cohort that was representative of the target population (consecutive cases of obstructive jaundice who had undergone ERCP, PTBD or bile duct surgery). After the diagnostic status of these patients was ascertained, the levels of serum MMP7 and CA19-9 were assayed in a fashion that blinded the analysis to a case-control status. In addition, we implemented STARD statements (STAndards for the Reporting of Diagnostic accuracy studies)[16-18] to ensure standardization and transparency of our study.
This study was conducted within the Rajavithi Hospital Surgery Department located in Bangkok, Thailand. The local ethics committee approved the study protocol. Sample size was calculated on the basis of an expected area under the ROC curve of MMP7 serum levels (= 0.70) for the diagnosis of cholangiocarcinoma[6]. Using a significance level of 0.05 (two-sided) and a power of 0.95, a sample of 50 cholangiocarcinoma patients was required for the study[19]. From previous data, the prevalence of cholangiocarcinoma detection in patients with obstructive jaundice treated at our department was shown to be in the range of 27%-30%. Therefore, we prospectively included consecutive patients with symptoms of obstructive jaundice who had undergone ERCP, PTBD or bile duct surgery during the period from June of 2008 to July of 2009. Exclusion criteria included presence of other cancers, age less than 20 years and the presence of severe pulmonary fibrosis[20]. All patients gave written informed consent. The diagnosis of cholangiocarcinoma was carried out using one of the following tests[6]: (1) tissue biopsy; and (2) cytology plus radiological (helical CT scan or MRI) and clinical observation to identify tumor progression at a follow-up of at least two months. Patients with an inconclusive diagnosis were excluded from this study.
Five-milliliter samples of fasting peripheral venous blood were collected from the patients before ERCP, PTBD, or bile duct surgery were carried out, and their serum was separated and stored at -78°C within 2 h. Serum biochemical tests including albumin, globulin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), total and direct bilirubin, alkaline phosphatase (ALP) and CA19-9 were measured using routine automated methods in Rajavithi Hospital Pathological Laboratory.
The serum levels of MMP7 were measured using an enzyme-linked immunosorbent assay (ELISA) kit (R&D Systems, Minneapolis, MN, USA), as previously described[6]. Briefly, the diluted serum samples were added in duplicate to 96-well plates coated with MMP7 antibody and then incubated at room temperature for 2 h. After washing, the conjugated secondary antibody was added, and the plate was further incubated for another 2 h. Plates were washed again prior to incubation with the substrate solution for 1 h. Following termination of the reaction with the stop solution (1 N sulfuric acid), the optical density was measured at 490 nm using a spectrophotometric microplate reader. The concentration of MMP7 in each sample was calculated from a standard curve. The scientist examining these serum samples was unaware of the patient’s diagnosis. In addition, the MMP7 test results had no influence on the clinical diagnosis of the patients in the study.
Data are presented as the mean ± SD, unless otherwise mentioned. Comparisons between the quantitative variables were performed using Mann-Whitney U or Student’s t-test, as appropriate. One-way analysis of variance (ANOVA) with multiple comparisons by the Post HOC Scheffe method or Kruskal Wallis test was used to compare each value (MMP7, CA19-9) to the control early and late stage cholangiocarcinoma groups. Qualitative variables were reported as counts, and comparisons between independent groups were performed using Pearson Chi-square tests. Correlations between MMP7 levels and other parameters were examined using the Pearson correlation coefficient. A ROC curve was generated by plotting the sensitivity against 1-specificity, and the area under the curve with 95% confidence intervals (CI) was calculated. The optimal cutoff points for MMP7 were selected based on the ROC curve analysis. Sensitivity, specificity, positive predictive value and negative predictive values were calculated using a 2 × 2 table of the collected data. The data on various blood chemistries and levels of CA19-9 and MMP7 that were significantly different between the control and cholangiocarcinoma groups were analyzed by multiple logistic regression analysis.
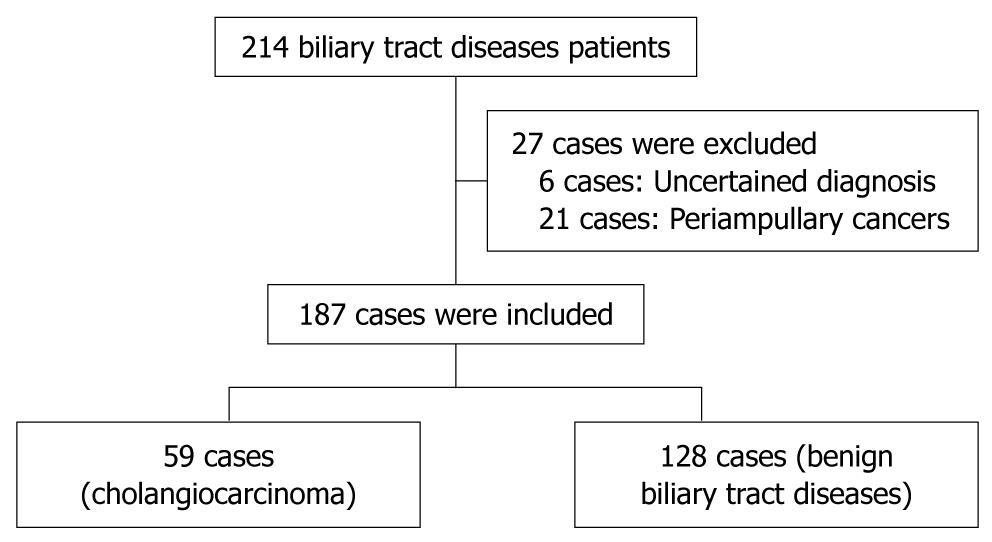
A total of 230 patients with obstructive jaundice were consecutively enrolled. Twenty-four cases were excluded according to their diagnosis of ampullary cancer (7 cases), pancreatic cancer (9 cases), gall bladder cancer (3 cases), duodenum cancer (2 cases), metastatic cancer from ovarian cancer (1 case) and hepatocellular carcinoma (2 cases). In addition, nineteen cases were excluded according to their uncertain diagnosis. The 187 subjects studied included 128 patients with benign biliary tract diseases (control group) which included intra-hepatic duct stones, common bile duct stones, and benign bile duct strictures, and a total of 59 patients with cholangiocarcinoma. For cholangiocarcinoma, 40 cases were diagnosed as perihilar-cholangiocarcinoma, 16 cases were diagnosed as intrahepatic cholangiocarcinoma and 3 cases were diagnosed as distal common bile duct cholangiocarcinoma (Figure 1).
As shown in Table 1, no statistically significant differences in gender, age, serum globulin and ALT levels were identified among the data from the control patients when compared to the cholangiocarcinoma patients. However, the level of serum albumin, AST, bilirubin and ALP were significantly higher in cholangiocarcinoma patients than in the control patients (Mann-Whitney U test, P < 0.05).
| Control | Cholangiocarcinoma | P | |
| n = 128 | n = 59 | ||
| Age (yr) | 61 ± 7 | 67 ± 5 | 0.451 |
| Sex (M:F) | 62:66 | 36:23 | 0.118 |
| Albumin (mg/dL) | 3.9 ± 0.67 | 3.1 ± 0.59 | < 0.001 |
| Globulin (mg/dL) | 3.9 ± 0.72 | 4.1 ± 0.91 | 0.073 |
| Total bilirubin (mg/dL) | 3.3 ± 3.71 | 12.0 ± 11.35 | < 0.001 |
| AST (U/L) | 73.4 ± 78.14 | 91.2 ± 75.91 | 0.003 |
| ALT (U/L) | 76.3 ± 83.32 | 52.2 ± 43.58 | 0.884 |
| ALP (U/L) | 320.3 ± 230.03 | 380.5 ± 314.52 | < 0.001 |
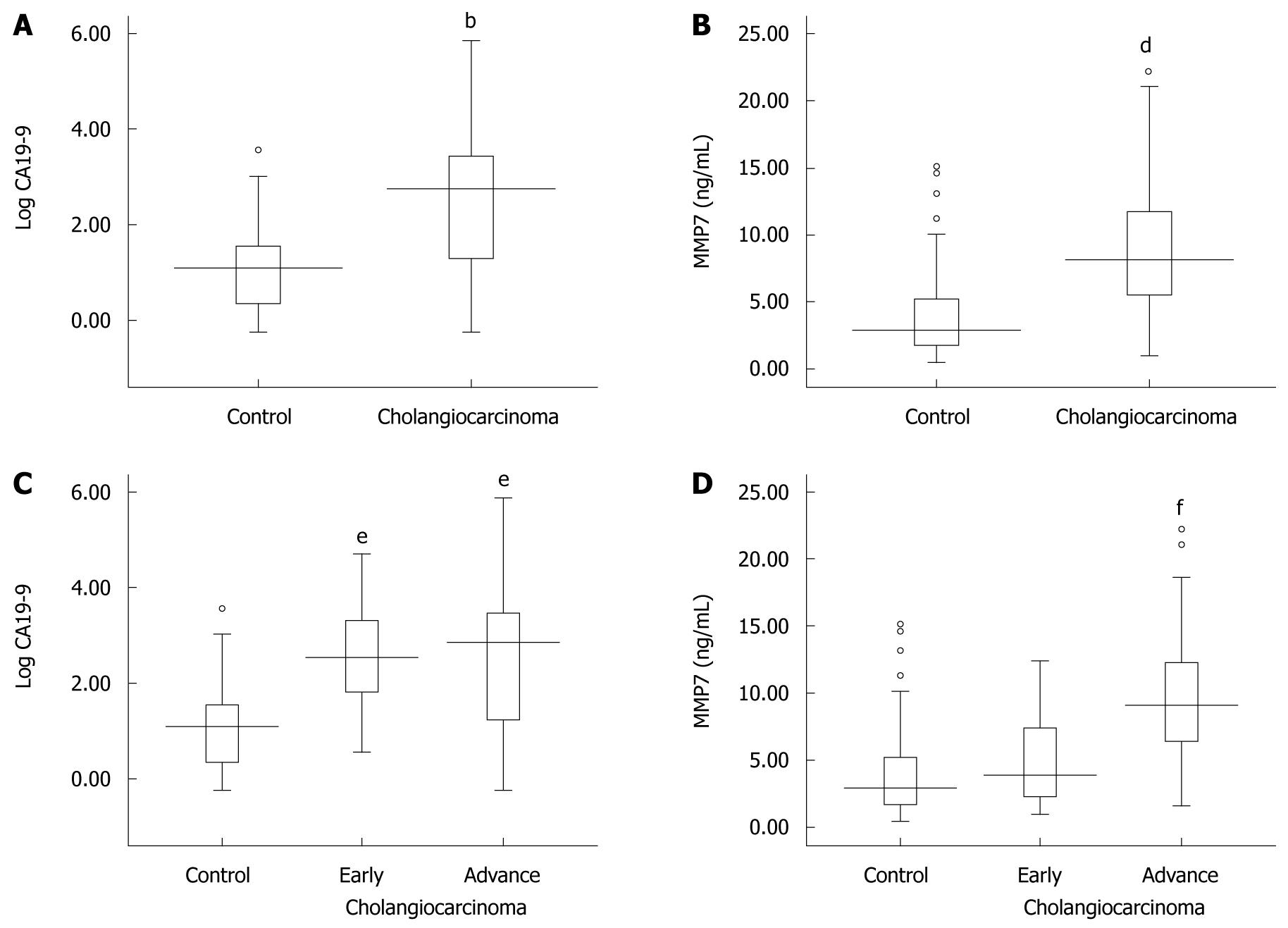
The serum CA19-9 and MMP7 levels were compared among disease groups. The median values of serum CA19-9 levels were 20.43 U/mL (range: 0.6-71 000 U/mL) in the control group and 571.2 U/mL (range: 0.6-71 000 U/mL) in the cholangiocarcinoma group. The mean values of serum MMP7 levels were 3.7 ± 2.81 ng/mL in the control group and 8.7 ± 4.56 ng/mL in the cholangiocarcinoma group. As shown in Figure 2A and B, serum CA19-9 and MMP7 values were significantly higher in cholangiocarcinoma cases when compared to the control patients (CA19-9: Mann-Whitney U test, P < 0.001 and MMP7: Student’s t-test, P < 0.001).
Moreover, we also classified cholangiocarcinoma patients into two groups: early (TNM stage I and II; 11 patients) and advanced (TNM stage III and IV; 48 patients) stages. Although the serum CA19-9 values in the early and late stages of cholangiocarcinoma were significantly higher than in the controls (Kruskal Wallis test, P < 0.001), the values were not significantly different between the early and late stages of cholangiocarcinoma (Figure 2C). The data shown in Figure 2D demonstrates that the MMP7 levels tended to increase according to the progression of cholangiocarcinoma. The serum MMP7 levels were significantly different between early and late stages of cholangiocarcinoma (ANOVA, P < 0.001). However, the serum MMP7 levels in early stage cholangiocarcinoma were not significantly different from the serum MMP7 levels in benign control patients (ANOVA, P = 0.47).
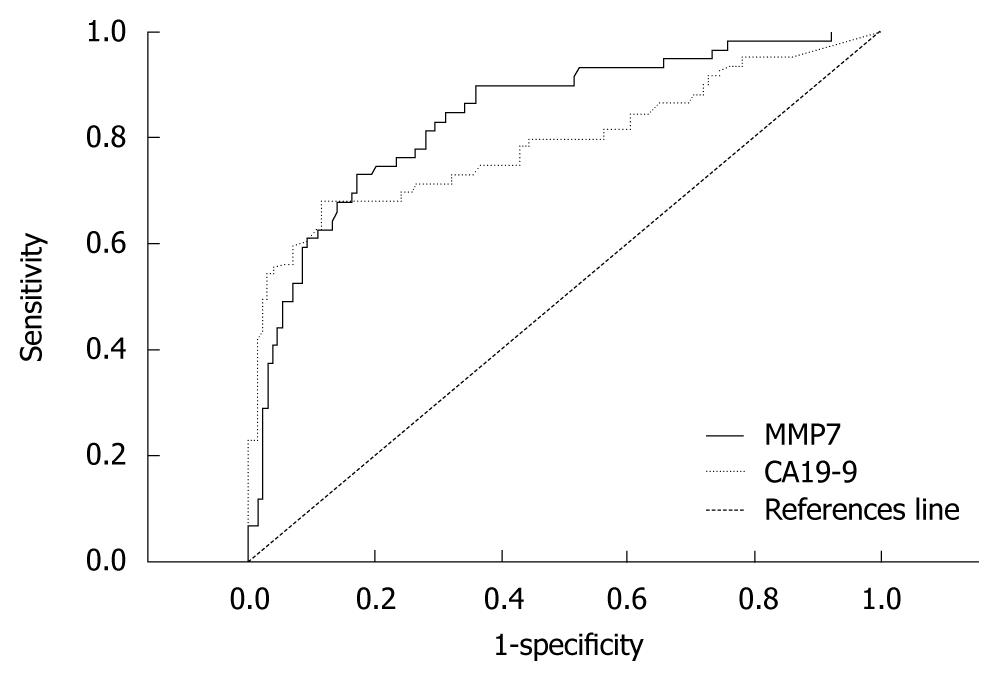
To determine the diagnostic accuracy of serum CA19-9 and MMP7 levels for differentiating cholangiocarcinoma from benign bile duct diseases, a ROC curve analysis was applied to calculate the area under the curve (AUC). These levels were determined to be 0.79 (95% CI: 0.708-0.868) and 0.84 (95% CI: 0.778-0.903) for CA19-9 and MMP7, respectively (Figure 3). The sensitivity, specificity, positive and negative predictive values for selected cut-off points of CA19-9 and MMP7 are presented in Table 2.
| Tumor markers (cut-off value) | Sensitivity | Specificity | PPV | NPV | LR+ | LR- |
| MMP7 (5.5 ng/mL) | 75 (63-86) | 78 (71-85) | 61 (50-72) | 87 (81-93) | 3.41 (2.38-4.89) | 0.33 (0.21-0.51) |
| MMP7 (6.5 ng/mL) | 63 (50-75) | 87 (81-93) | 69 (56-81) | 83 (77-90) | 4.72 (2.91-7.66) | 0.43 (0.31-0.60) |
| MMP7 (7.5 ng/mL) | 53 (40-65) | 92 (88-97) | 76 (62-89) | 81 (74-87) | 6.73 (3.54-12.70) | 0.51 (0.39-0.68) |
| CA19-9 (35 U/mL) | 71 (60-83) | 73 (66-81) | 55 (44-66) | 85 (78-91) | 2.68 (1.93-3.73) | 0.39 (0.26-0.59) |
| CA19-9 (100 U/mL) | 68 (56-80) | 87 (81-93) | 70 (58-82) | 85 (79-91) | 5.1 (3.17-8.22) | 0.37 (0.25-0.54) |
| CA19-9 (200 U/mL) | 59 (47-72) | 93 (89-97) | 80 (68-91) | 83 (77-89) | 8.44 (4.34-16.40) | 0.44 (0.32-0.60) |
When the cut-off value of serum MMP7 was set at 5.5 ng/mL and serum CA19-9 values were set at 100 U/mL, the predictive probabilities for the diagnosis of cholangiocarcinoma could then be calculated from logistic regression analysis. As shown in Table 3, if the patients had a serum MMP7 and CA19-9 level higher than the cut-off values, the probability of a diagnosis of cholangiocarcinoma was equal to 86.12%. In addition, if the patients had a serum MMP7 and serum CA19-9 level less than the cut-off values, the probability of a positive diagnosis of cholangiocarcinoma was very low (< 6.4%).
| CA19-9 (> 100 U/mL) | MMP7 (> 5.5 ng/mL) | Predicted probability (%) |
| - | - | 6.40 |
| - | + | 36.10 |
| + | - | 42.84 |
| + | + | 86.12 |
The correlations between the levels of serum albumin, AST, ALT, ALP, total bilirubin, CA19-9, and MMP7 were investigated. As presented in Table 4, the level of serum MMP7 was significantly correlated with serum albumin, AST, ALP, total bilirubin and CA19-9, although none of these parameters had a high Pearson correlation coefficient value (> 0.7). We suggest that the significant correlation of these blood chemistries with serum MMP7 was caused by the high number of samples analyzed in this study.
To determine whether the levels of serum CA19-9 and MMP7 were predictive of cholangiocarcinoma independent of the other blood chemistry levels that were significantly different between control and cholangiocarcinoma patients, we carried out a logistical regression analysis. In a multivariable model using CA19-9 (cut-off value = 100 ng/mL), MMP7 (cut-off value = 5.5 ng/mL), total bilirubin (cut-off value = 5 mg/dL), albumin (cut-off value = 4 mg/dL), AST (cut-off value = 100 U/L) and ALP (cut-off value = 200 U/L), CA19-9, MMP7 and albumin were shown to be independent predictors for cholangiocarcinoma. None of the other parameters (total bilirubin, AST and ALP) reached statistical significance (Table 5).
| Variables | OR (95% CI) | P |
| CA19-9 | 15.2 (5.20-44.56) | < 0.001 |
| MMP7 | 5.5 (1.87-16.03) | 0.002 |
| Albumin | 0.015 (0.01-0.15) | < 0.001 |
| Total bilirubin | 2.4 (0.81-7.20) | 0.115 |
| AST | 1.2 (0.37-4.12) | 0.738 |
| ALP | 0.3 (0.09-1.05) | 0.060 |
Our study demonstrates that serum MMP7 levels are significantly elevated in patients with a diagnosis of cholangiocarcinoma when compared to patients suffering from benign bile duct diseases. When we compared MMP7 to CA19-9, which is a common clinically-used biomarker of cholangiocarcinoma, the value of AUC from the ROC curve demonstrated that serum levels of MMP7 are better than CA19-9 for the diagnosis of cholangiocarcinoma. These results are consistent with our previous study, in which serum MMP7 was higher in cholangiocarcinoma than in benign obstructive jaundice patients[6]. This suggested that serum MMP7 has the potential to be a tumor marker for cholangiocarcinoma in patients with obstructive jaundice.
Previous studies have demonstrated that MMP7 plays a key role in the mechanism of cancer invasion via proteolytic cleavage of the extracellular matrix tissues. It has also been shown to activate other MMPs, such as proMMP-2 and proMMP-9[21], and inhibit E-cadherin function by ectodomain shedding of E-cadherin[22]. The results of several recent studies indicate that MMP7 is over-expressed in a variety of epithelial tumors including those of the esophagus[23], colon[24,25], pancreas[26], and cholangiocarcinoma tumors[12]. In addition, several studies have shown that MMP7 could be detected in the serum of cancer patients, including patients with ovarian[27], colorectal[28] and gastric cancer[29]. This finding suggests that high levels of serum MMP7 are not specific to cholangiocarcinoma. It can be detected in many types of cancer. Therefore, it should be used with other diagnostic modalities (clinical presentation and imaging study) before making a diagnosis.
In this study, the levels of blood chemistry markers were shown to be significantly different between control and cholangiocarcinoma groups. Although several differences were observed, serum CA19-9 and MMP7 levels were shown to be predictors of cholangiocarcinoma, independent of other blood chemistry values. In addition, the present study is the first to demonstrate the probability of a diagnosis of cholangiocarcinoma using the combination of serum MMP7 and CA19-9 levels (Table 3). We suggest that the combination of these markers will aid the physician to identify cholangiocarcinoma from benign obstructive jaundice.
The values of AUC from the ROC curve for MMP7 and CA19-9 in this study were shown to be much higher than those observed in our previous study[6]. The differences in the designs of these studies should be considered. Our previous study was designed as a retrospective case-control study for diagnostic accuracy. Therefore, some bias from the selection of samples may have occurred. A strength of the present study was the implementation of the strategies of the PRoBE designs to avoid the problems of bias that may affect the studies of the diagnostic test[15]. We collected serum from all obstructive jaundice patients before the diagnosis of cholangiocarcinoma or benign biliary tract diseases was determined. This procedure assured that biases related to differences in sample collection and handling would be avoided[30]. Limitations of this design include the fact that the majority of the study participants were in advanced stages of cholangiocarcinoma. The number of patients with early-stage cholangiocarcinoma was small (n = 11), and this number of patients would not have had the statistical power to detect a difference in mean value between these early stages of cholangiocarcinoma and the control group. Further studies, which should include an increased number of early-stage cholangiocarcinoma cases, need to be carried out before using MMP7 as a screening test for the detection of early stage cholangiocarcinoma. In addition, this study was performed in a referral center, which has a high prevalence of cholangiocarcinoma. As a result, the findings may not be broadly applicable to other hospitals that typically have a low volume of cholangiocarcinoma.
In conclusion, this study demonstrated that serum MMP7 levels are significantly elevated in cholangiocarcinoma patients. This marker has the potential to be used as a new tumor marker for discriminating cholangiocarcinoma patients from benign biliary tract disease patients.
To date, carbohydrate antigen 19-9 (CA19-9) is used as a tumor marker for detecting cholangiocarcinoma. Unfortunately, elevated serum levels of CA19-9 have also been found in patients with benign obstructive jaundice. Previous studies demonstrated that cholangiocarcinoma cells express a high level of matrix metalloproteinase (MMP)-7.
High expression of MMP7 was detected in cholangiocarcinoma specimens. In addition, the authors’ previous nonconsecutive case-control study demonstrated that the serum level of MMP7 is higher in cholangiocarcinoma than in benign biliary tract disease patients. However, a prospective consecutive study of the evaluation of serum MMP7 as a diagnostic marker for cholangiocarcinoma has not been established. In this study, the authors collected a new and independent dataset of prospective consecutive cases with evidence of bile duct obstruction due to various etiologies, and demonstrated that the serum level of MMP7 could be a potential tumor marker for differentiating cholangiocarcinoma from benign biliary tract obstruction.
This is the first consecutive prospective study to report that the serum level of MMP7 was significantly higher in patients with cholangiocarcinoma than in those with benign biliary tract obstruction. The authors suggest that the serum level of MMP7 may be a potential tumor marker for differentiating cholangiocarcinoma from benign biliary tract obstruction.
This study may represent a future strategy for diagnosing patients with cholangiocarcinoma by the detection of serum level of MMP7 and CA19-9.
This is a prospective-specimen-collection and retrospective-blinded-evaluation study of 187 patients with obstructive jaundice where a novel serum marker, MMP7, for the diagnosis of cholangiocarcinoma was investigated. In general, it’s a nicely designed and accomplished study with sound conclusion, hopefully of interest for a wide range of readers and researchers.
Peer reviewers: Giedrius Barauskas, Professor, Department of Surgery, Kaunas University of Medicine, Eiveniu str. 2, Kaunas, LT-50009, Lithuania; Wen-Hsin Huang, MD, Division of Hepatogastroenterology, Department of Internal Medicine, China Medical University Hospital, No 2, Yuh-Der Road, Taichung 404, Taiwan, China
S- Editor Wang JL L- Editor Webster JR E- Editor Ma WH
| 1. | Sripa B, Pairojkul C. Cholangiocarcinoma: lessons from Thailand. Curr Opin Gastroenterol. 2008;24:349-356. [Cited in This Article: ] |
| 2. | Bridgewater J, Imber C. New advances in the management of biliary tract cancer. HPB (Oxford). 2007;9:104-111. [Cited in This Article: ] |
| 3. | Blechacz BR, Gores GJ. Cholangiocarcinoma. Clin Liver Dis. 2008;12:131-150, ix. [Cited in This Article: ] |
| 4. | Byrnes V, Afdhal N. Cholangiocarcinoma of the Hepatic Hilum (Klatskin Tumor). Curr Treat Options Gastroenterol. 2002;5:87-94. [Cited in This Article: ] |
| 5. | Furmanczyk PS, Grieco VS, Agoff SN. Biliary brush cytology and the detection of cholangiocarcinoma in primary sclerosing cholangitis: evaluation of specific cytomorphologic features and CA19-9 levels. Am J Clin Pathol. 2005;124:355-360. [Cited in This Article: ] |
| 6. | Leelawat K, Sakchinabut S, Narong S, Wannaprasert J. Detection of serum MMP-7 and MMP-9 in cholangiocarcinoma patients: evaluation of diagnostic accuracy. BMC Gastroenterol. 2009;9:30. [Cited in This Article: ] |
| 7. | Nichols JC, Gores GJ, LaRusso NF, Wiesner RH, Nagorney DM, Ritts RE Jr. Diagnostic role of serum CA 19-9 for cholangiocarcinoma in patients with primary sclerosing cholangitis. Mayo Clin Proc. 1993;68:874-879. [Cited in This Article: ] |
| 8. | Leelawat K, Leelawat S, Ratanachu-Ek T, Trubwongchareon S, Wannaprasert J, Tripongkaruna S, Chantawibul S, Tepaksorn P. Circulating hTERT mRNA as a tumor marker in cholangiocarcinoma patients. World J Gastroenterol. 2006;12:4195-4198. [Cited in This Article: ] |
| 9. | Patel AH, Harnois DM, Klee GG, LaRusso NF, Gores GJ. The utility of CA 19-9 in the diagnoses of cholangiocarcinoma in patients without primary sclerosing cholangitis. Am J Gastroenterol. 2000;95:204-207. [Cited in This Article: ] |
| 10. | Ong SL, Sachdeva A, Garcea G, Gravante G, Metcalfe MS, Lloyd DM, Berry DP, Dennison AR. Elevation of carbohydrate antigen 19.9 in benign hepatobiliary conditions and its correlation with serum bilirubin concentration. Dig Dis Sci. 2008;53:3213-3217. [Cited in This Article: ] |
| 11. | Principe A, Del Gaudio M, Grazi GL, Paolucci U, Cavallari A. Mirizzi syndrome with cholecysto-choledocal fistula with a high CA19-9 level mimicking biliary malignancies: a case report. Hepatogastroenterology. 2003;50:1259-1262. [Cited in This Article: ] |
| 12. | Itatsu K, Zen Y, Yamaguchi J, Ohira S, Ishikawa A, Ikeda H, Sato Y, Harada K, Sasaki M, Sasaki M. Expression of matrix metalloproteinase 7 is an unfavorable postoperative prognostic factor in cholangiocarcinoma of the perihilar, hilar, and extrahepatic bile ducts. Hum Pathol. 2008;39:710-719. [Cited in This Article: ] |
| 13. | Itatsu K, Zen Y, Ohira S, Ishikawa A, Sato Y, Harada K, Ikeda H, Sasaki M, Nimura Y, Nakanuma Y. Immunohistochemical analysis of the progression of flat and papillary preneoplastic lesions in intrahepatic cholangiocarcinogenesis in hepatolithiasis. Liver Int. 2007;27:1174-1184. [Cited in This Article: ] |
| 14. | Miwa S, Miyagawa S, Soeda J, Kawasaki S. Matrix metalloproteinase-7 expression and biologic aggressiveness of cholangiocellular carcinoma. Cancer. 2002;94:428-434. [Cited in This Article: ] |
| 15. | Pepe MS, Feng Z, Janes H, Bossuyt PM, Potter JD. Pivotal evaluation of the accuracy of a biomarker used for classification or prediction: standards for study design. J Natl Cancer Inst. 2008;100:1432-1438. [Cited in This Article: ] |
| 17. | Bruns DE. The STARD initiative and the reporting of studies of diagnostic accuracy. Clin Chem. 2003;49:19-20. [Cited in This Article: ] |
| 18. | Bossuyt PM. STARD statement: still room for improvement in the reporting of diagnostic accuracy studies. Radiology. 2008;248:713-714. [Cited in This Article: ] |
| 19. | Hanley JA, McNeil BJ. A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology. 1983;148:839-843. [Cited in This Article: ] |
| 20. | Huh JW, Kim DS, Oh YM, Shim TS, Lim CM, Lee SD, Koh Y, Kim WS, Kim WD, Kim KR. Is metalloproteinase-7 specific for idiopathic pulmonary fibrosis? Chest. 2008;133:1101-1106. [Cited in This Article: ] |
| 21. | Ii M, Yamamoto H, Adachi Y, Maruyama Y, Shinomura Y. Role of matrix metalloproteinase-7 (matrilysin) in human cancer invasion, apoptosis, growth, and angiogenesis. Exp Biol Med (Maywood). 2006;231:20-27. [Cited in This Article: ] |
| 22. | Noë V, Fingleton B, Jacobs K, Crawford HC, Vermeulen S, Steelant W, Bruyneel E, Matrisian LM, Mareel M. Release of an invasion promoter E-cadherin fragment by matrilysin and stromelysin-1. J Cell Sci. 2001;114:111-118. [Cited in This Article: ] |
| 23. | Yamamoto H, Adachi Y, Itoh F, Iku S, Matsuno K, Kusano M, Arimura Y, Endo T, Hinoda Y, Hosokawa M. Association of matrilysin expression with recurrence and poor prognosis in human esophageal squamous cell carcinoma. Cancer Res. 1999;59:3313-3316. [Cited in This Article: ] |
| 24. | Adachi Y, Yamamoto H, Itoh F, Hinoda Y, Okada Y, Imai K. Contribution of matrilysin (MMP-7) to the metastatic pathway of human colorectal cancers. Gut. 1999;45:252-258. [Cited in This Article: ] |
| 25. | Yamamoto H, Iku S, Adachi Y, Imsumran A, Taniguchi H, Nosho K, Min Y, Horiuchi S, Yoshida M, Itoh F. Association of trypsin expression with tumour progression and matrilysin expression in human colorectal cancer. J Pathol. 2003;199:176-184. [Cited in This Article: ] |
| 26. | Jones LE, Humphreys MJ, Campbell F, Neoptolemos JP, Boyd MT. Comprehensive analysis of matrix metalloproteinase and tissue inhibitor expression in pancreatic cancer: increased expression of matrix metalloproteinase-7 predicts poor survival. Clin Cancer Res. 2004;10:2832-2845. [Cited in This Article: ] |
| 27. | Palmer C, Duan X, Hawley S, Scholler N, Thorpe JD, Sahota RA, Wong MQ, Wray A, Bergan LA, Drescher CW. Systematic evaluation of candidate blood markers for detecting ovarian cancer. PLoS One. 2008;3:e2633. [Cited in This Article: ] |
| 28. | Maurel J, Nadal C, Garcia-Albeniz X, Gallego R, Carcereny E, Almendro V, Mármol M, Gallardo E, Maria Augé J, Longarón R. Serum matrix metalloproteinase 7 levels identifies poor prognosis advanced colorectal cancer patients. Int J Cancer. 2007;121:1066-1071. [Cited in This Article: ] |
| 29. | Yeh YC, Sheu BS, Cheng HC, Wang YL, Yang HB, Wu JJ. Elevated serum matrix metalloproteinase-3 and -7 in H. pylori-related gastric cancer can be biomarkers correlating with a poor survival. Dig Dis Sci. 2010;55:1649-1657. [Cited in This Article: ] |
| 30. | Biesheuvel CJ, Vergouwe Y, Oudega R, Hoes AW, Grobbee DE, Moons KG. Advantages of the nested case-control design in diagnostic research. BMC Med Res Methodol. 2008;8:48. [Cited in This Article: ] |