Published online Nov 6, 2014. doi: 10.5527/wjn.v3.i4.220
Revised: June 22, 2014
Accepted: September 6, 2014
Published online: November 6, 2014
Processing time: 171 Days and 3.5 Hours
Erectile dysfunction (ED) is a common condition among male chronic kidney disease (CKD) patients. Its prevalence is estimated to be approximately 80% among these patients. It has been well established that the production of nitric oxide from the cavernous nerve and vascular endothelium and the subsequent production of cyclic GMP are critically important in initiating and maintaining erection. Factors affecting these pathways can induce ED. The etiology of ED in CKD patients is multifactorial. Factors including abnormalities in gonadal-pituitary system, disturbance in autonomic nervous system, endothelial dysfunction, anemia (and erythropoietin deficiency), secondary hyperparathyroidism, drugs, zinc deficiency, and psychological problems are implicated in the occurrence of ED. An improvement of general conditions is the first step of treatment. Sufficient dialysis and adequate nutritional intake are necessary. In addition, control of anemia and secondary hyperparathyroidism is required. Changes of drugs that potentially affect erectile function may be necessary. Further, zinc supplementation may be necessary when zinc deficiency is suspected. Phosphodiesterase type 5 inhibitors (PDE5Is) are commonly used for treating ED in CKD patients, and their efficacy was confirmed by many studies. Testosterone replacement therapy in addition to PDE5Is may be useful, particularly for CKD patients with hypogonadism. Renal transplantation may restore erectile function. ED is an early marker of cardiovascular disease (CVD), which it frequently precedes; therefore, it is crucial to examine the presence of ED in CKD patients not only for the improvement of the quality of life but also for the prevention of CVD attack.
Core tip: Erectile dysfunction (ED) is a common condition in chronic kidney disease (CKD) patients. The etiology is multifactorial. Phosphodiesterase type 5 inhibitors are commonly used for the initial treatment. ED has gained attention as an early marker for cardiovascular disease (CVD), which it frequently precedes. Therefore, it is pivotal to examine the presence of ED in CKD patients not only for the improvement of quality of life but also for the prevention of CVD attack. The pathophysiology of erection, which most nephrologists are not familiar with, is also discussed.
- Citation: Suzuki E, Nishimatsu H, Oba S, Takahashi M, Homma Y. Chronic kidney disease and erectile dysfunction. World J Nephrol 2014; 3(4): 220-229
- URL: https://www.wjgnet.com/2220-6124/full/v3/i4/220.htm
- DOI: https://dx.doi.org/10.5527/wjn.v3.i4.220
Erectile dysfunction (ED) is defined as an inability to attain and/or maintain penile erection sufficient for satisfactory sexual performance. It is now a common condition and approximately 150 million males worldwide are estimated to suffer from ED[1]. The prevalence of ED in 2025 is projected to be approximately 300 million worldwide[2]. It is well known that age, metabolic disorders (hypertension, diabetes, and hyperlipidemia), and smoking are major risk factors for ED. Recently, chronic kidney disease (CKD) has also gained attention as a risk factor for ED. Although CKD causes sexual dysfunction in both genders, this review article focuses on the role of CKD in the development of ED. We discuss the etiology and treatment of ED in CKD patients.
The prevalence of ED in the United States male population aged > 50 years (Participants: 31,742 men, age 53-90 years) was reported to be 33%[3], whereas that in the Turkish male population aged > 40 years (Participants: 2158 men) was 69.2%[4]. However, the prevalence was 36% when mild ED cases were excluded. Navaneethan et al[5] reported in their meta-analysis study that the prevalence of ED in CKD patients was 70% on average. Furthermore, Mesquita et al[6] reported that the prevalence of ED in CKD outpatients with stages 3, 4, and 5 was 72.3%, 81.5%, and 85.7%, respectively. Nassir reported that the prevalence of ED in patients just entering dialysis programs was 82.7%[7]. Thus, it is observed that ED frequently occurs in CKD patients.
The blood supply to the penis originates predominantly from the internal pudendal artery, which branches into the penile artery. The penile artery then branches into the cavernous arteries. The cavernous artery enters the cavernous body and subsequently divides into many branches called the helicine arteries, which open into the cavernous sinuses. Blood in the cavernous sinuses is drained by the subtunical veins that form the venous plexuses just beneath the tunica albuginea and then returns to the circulation via 3 sets of veins; the superficial, intermediate and deep veins.
Penile erection and detumescence are regulated by relaxation and contraction, respectively, of the smooth muscle located in the arteries and the cavernous body. In the flaccid state, the sympathetic nervous system is dominant, and the arterial and corporal smooth muscle is tonically contracted. As a result, only a minimal amount of blood flows through the cavernous artery into the cavernous body. After sexual stimulation, parasympathetic activity causes a decrease in the peripheral resistance due to vasodilatation, and the blood flow through the cavernous and helicine arteries increases. The intracavernous pressure increases without any increase in the systemic pressure. In the full erectile state, increased blood volume in the cavernous body and the following compression of the subtunical drainage veins against the rigid tunica albuginea lead to a reduction in the venous outflow (referred to as the veno-occlusive mechanism), and therefore, high intracavernous pressure is maintained. However, when the corporal smooth muscle is unable to relax sufficiently and/or the corporal tissue loses its normal compliance, the increased intracavernous pressure during erection cannot adequately compress the subtunical veins, resulting in the leakage of blood out of the cavernous body during erection. This is a major cause of ED and is referred to as the corporal veno-occlusive dysfunction (CVOD). CVOD occurs when the smooth muscle content decreases and/or when the collagen content increases in the cavernous body[8]. Therefore, the ratio of the smooth muscle content to the collagen content in the cavernous body decreases in CVOD.
Detumescence of the penis is predominantly mediated by adrenergic nerve terminals whose neurotransmitter, norepinephrine, activates adrenergic receptors on the penile smooth muscle. The contraction of penile arteries and trabecular smooth muscle is largely mediated by α-1 adrenergic receptors[9,10]. Other vasoconstrictors including endothelin-1, prostaglandin F2α, thromboxane A2 and angiotensin II are also implicated in the contraction of smooth muscle in the penis[11-13].
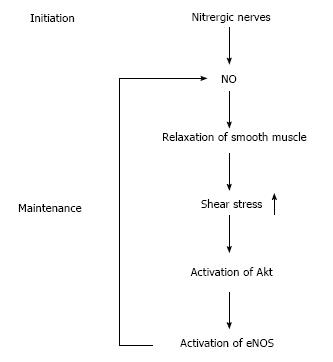
Dilatation of the cavernous artery and helicine arteries is the first event in the development of an erection. The blood flow and pressure increase in the cavernous sinuses, and subsequently, smooth muscles surrounding the trabeculae relax, resulting in further expansion and accumulation of blood in the cavernous body. It is now well established that nitric oxide (NO) plays a pivotal role in the initiation and maintenance of erection. NO acts through the stimulation of the soluble guanylate cyclase, which mediates the subsequent formation of cyclic-GMP (cGMP). cGMP activates protein kinase G (PKG), and PKG is implicated in the relaxation of smooth muscle. cGMP is inactivated by phosphodiesterase type 5 (PDE5), which is predominantly located in the cavernous smooth muscle and is the target of PDE5 inhibitors (PDE5Is) such as sildenafil and vardenafil. NO synthase (NOS) uses the amino acid L-arginine and molecular oxygen to produce NO. Three distinct isoforms of NOS have been identified. Two constitutive forms, neuronal NOS (nNOS) and endothelial NOS (eNOS), are present in the nervous system and vascular endothelial cells, respectively. A third isoform, inducible NOS (iNOS) is expressed in a variety of cells in response to inflammatory mediators and bacterial products. The isoforms nNOS and eNOS are expressed in the autonomic nerves and endothelium of the penis, respectively[14-17]. Under physiological conditions, iNOS is not expressed in the penis. Postganglionic parasympathetic nerves, which express nNOS and release NO as a cotransmitter with acetylcholine, are now termed nitrergic nerves[17,18]. The stimulation of the cavernous nerve activates nitrergic nerve fibers and elicits NO release at the nerve terminals, which causes relaxation of penile smooth muscle. The functional role of NO released from the nitrergic nerve termini during the relaxation of penile smooth muscle has been demonstrated in many studies in which penile erection induced by stimulation of the cavernous nerves or the spinal cord can be inhibited by NOS inhibitors[14,19-21]. The role of eNOS in erection has also been studied. One possibility was that acetylcholine released from postganglionic cholinergic nerves evoked the release of NO from the endothelium to induce endothelium-dependent relaxation of the penile smooth muscle. However, atropine, a competitive inhibitor of the muscarinic effect of acetylcholine, did not inhibit cavernous nerve-induced penile erection[14]. Furthermore, neurogenic relaxation of the cavernous body does not require a functional endothelium[22,23], suggesting that acetylcholine-induced endothelium-dependent relaxation of the smooth muscle is not required for cavernous nerve-induced penile erection. A second possibility was the activation of eNOS by shear stress. During erection, an increased blood flow on the luminal surface of the penile artery and cavernous sinuses can cause shear stress, which may lead to the activation of protein kinase Akt (also known as Protein kinase B) and subsequent phosphorylation and activation of eNOS, facilitating NO release from the endothelium. Hurt et al[24] demonstrated that both electrical stimulation of the cavernous nerve and direct intracavernosal injection of a vasorelaxant drug, papaverine, caused a rapid increase in the phosphorylation and activation of Akt and eNOS. The authors also showed that penile erection elicited by papaverine is significantly reduced in eNOS gene knockout mice. They proposed a model in which the rapid, brief activation of nNOS initiates the erectile response, whereas Akt-dependent phosphorylation and activation of eNOS are necessary for sustained NO production and maximal erection (Figure 1).
Most studies in this field have been performed using dialysis patients and renal transplant recipients. Little data exist on the etiology and treatment of ED in CKD patients before entering a dialysis program.
Chronic renal failure (CRF) is associated with impaired spermatogenesis, and it often results in infertility[25]. In addition, testes develop endocrine dysfunction. Total and free testosterone levels are typically reduced, although the binding capacity and concentration of sex hormone-binding globulin are normal[26-28]. Serum luteinizing hormone (LH) level increases in CRF patients, and testosterone secretion in response to acute administration of human chorionic gonadotropin (HCG), a compound with LH-like actions, shows a blunted response, suggesting that the testosterone-producing Leydig cells have low responsiveness to LH and that this is the primary cause of low testosterone levels in CRF[29]. Interestingly, a factor capable of blocking the LH receptor in vitro has been identified in uremic serum, providing an explanation for the blunted response of Leydig cells to infusion of HCG. This blocking activity is inversely correlated with GFR and almost disappears after renal transplantation[30]. In addition, follicle-stimulating hormone (FSH) secretion increases in men with CRF. FSH release from the pituitary gland is negatively regulated by inhibin, a peptide product of Sertoli cells that are located in the convoluted seminiferous tubules. FSH concentration appears to increase in uremic patients because of the damage to seminiferous tubules, resulting in the suppression of inhibit production[31].
Testosterone is required not only for libido but also for the maintenance of the normal morphology and function of the penis. Testosterone deficiency leads to the loss of smooth muscle in the cavernous body and its replacement with collagen fibers[32,33]. This may result in CVOD. It has also been demonstrated that the activity of nNOS and PDE5 are positively regulated by testosterone[32].
Elevated plasma prolactin levels are commonly found in CRF[34]. Increased production is the main cause because the kidney plays little, if any, role in its catabolism. Secondary hyperparathyroidism may be implicated in the increased prolactin secretion in CRF because an infusion of parathyroid hormone (PTH) in healthy men enhances prolactin release[35]. Depletion of zinc reserves may also play a role in uremic hyperprolactinemia[36]. Hyperprolactinemia induces the loss of libido and low serum testosterone levels[37], which may cause ED.
It is now well known that CKD is a risk factor for cardiovascular disease (CVD)[38,39]. Endothelial dysfunction is an early marker of CVD, and has also been reported to occur in CKD patients[40-42]. In addition, endothelial dysfunction is a cause of ED, because NO production from the endothelium decreases in this state. Therefore, it is not surprising that ED frequently occurs in CKD patients. Furthermore, CKD patients often suffer from metabolic diseases such as hypertension, hyperlipidemia, and diabetes. Diabetes is a major cause of CKD. These metabolic diseases also cause endothelial dysfunction and are risk factors for ED. Therefore, in addition to the concomitant metabolic diseases, CKD per se appears, at least in some part, to cause ED via the induction of endothelial dysfunction.
Autonomic neuropathy occurs in end-stage renal disease and can be a cause of ED[43,44]. It is well known that autonomic neuropathy is a common complication of diabetes, and it can be a cause of ED in CKD patients.
Erythropoietin (Epo) has been widely used to treat anemia in uremic patients. Several reports have demonstrated that treatment with Epo improved erectile function in dialysis patients[45-47], suggesting that anemia and/or Epo deficiency are implicated in ED. The mechanism by which Epo restores erectile function remains unclear. Epo normalized the increased serum prolactin level in early studies[45,48], but this finding was not confirmed by other studies[49-51]. Moreover, Epo increased serum testosterone levels in some studies[51,52]; however, this finding was again not confirmed by other studies[45,46,49,50]. Allaf et al[53] examined the effects of Epo on the recovery of erectile function in a rat model of cavernous nerve injury and found that Epo restored erectile function. They also found that Epo stimulated axonal regeneration of the injured cavernous nerve. Therefore, Epo may stimulate the regeneration of the cavernous nerve. Epo reportedly has protective effects against ischemic damages via its anti-apoptotic activity[54-59]. Therefore, Epo may protect the cavernous body against injuries via its anti-apoptotic activity. Furthermore, the receptor for Epo is expressed on vascular endothelial cells (VECs) and Epo stimulates the proliferation and migration of VECs[60,61]. Epo is also capable of mobilizing endothelial progenitor cells (EPCs) from the bone marrow[62,63]. EPCs were originally isolated from human peripheral blood[64]. EPCs are progenitor cells whose differentiation potential is restricted to VECs. They were incorporated in the capillaries and small arteries of ischemic tissues in vivo and expressed markers for VECs such as CD31 when introduced into the circulation using a hindlimb ischemia model[64], suggesting their involvement in the stimulation of angiogenesis. Several studies have reported that the number of circulating EPCs decreased in ED patients[65-67]. These data suggest that Epo may restore erectile function via its proangiogenic activity. In summary, Epo has nerve-protective, anti-apoptotic, and proangiogenic activities, at least in animal models, and these activities may be implicated in Epo-induced restoration of erectile function. It is likely that Epo restores erectile function via interaction with its receptors on cells such as nerves and VECs rather than on red blood cells with a resultant improvement in anemia.
Although no conclusive data have been published, Massry et al[68] reported that a decline in serum PTH concentration by treatment with 1,25(OH)2 vitamin D3 correlated with the recovery of erectile function in dialysis patients. It was also reported that PTH administration increased serum prolactin concentration[35]. Therefore, it is possible that secondary hyperparathyroidism is implicated in erectile dysfunction in dialysis patients.
Many drugs used for CKD patients potentially cause ED. Common examples are anti-hypertensive drugs including diuretics, agonists for α-2 adrenergic receptors, and beta-blockers. Other examples are cimetidine, tricyclic antidepressants, and metoclopramide.
The prevalence of depression among dialysis patients has been estimated to be 20%-30%[69-71]. Several studies demonstrated that depression is an independent risk factor for ED[72,73].
Several reports demonstrated that oral zinc supplementation restored erectile function, which was associated with an increase in serum testosterone concentration[74,75]; however, some negative effects of zinc supplementation on erectile function were also reported[76]. Possible causes of ED in CKD patients are summarized in Table 1.
| Abnormalities in the gonadal and pituitary systems |
| Testosterone↓ |
| LH↑, FSH↑ |
| Prolactin↑ |
| Endothelial dysfunction |
| Hypertension, diabetes, hyperlipidemia |
| Autonomic neuropathy |
| Anemia (Erythropoietin↓) |
| Secondary hyperparathyroidism |
| Drugs |
| Diuretics |
| Agonists for α-2 adrenergic receptors and β-blockers |
| Cimetidine |
| Tricyclic antidepressants |
| Depression |
| Zinc deficiency |
Because of the high prevalence of ED among CVD patients, ED was traditionally regarded as a secondary complication of CVD. Recently, ED has gained attention as an early marker of CVD, because ED often precedes the occurrence of CVD. The Prostate Cancer Prevention Trial was a prospective, randomized, and placebo-controlled trial to assess whether finasteride decreased the prevalence of prostate cancer[77]. Finasteride is an inhibitor of 5α-reductase, and inhibits the conversion of testosterone to dihydrotestosterone, which is the primary androgen in the prostate. Participants were regularly monitored for overall health, including cardiovascular events and sexual function. Data from 9457 men randomized to the placebo group in this trial were analyzed to assess the hypothesis that ED is an early marker of patients with occult CVD[78]. At entry to the study, 8063 (85%) men had no CVD; of these men, 3816 (47%) patients reported some level of ED. Among the 4247 men without ED at study entry, 2420 men (57%) reported an incident ED after 5 years, and this incidence increased to 65% at 7 years. Incidents of ED were significantly associated with subsequent angina, myocardial infarction, or stroke; hazard ratio after adjustment was 1.25. Several other studies also confirmed this finding that ED often precedes the onset of CVD[79-81]. Furthermore, ED has been recognized as an early marker for silent coronary artery disease (CAD). Gazzaruso et al[82] examined the prevalence of ED in 133 uncomplicated type 2 diabetic men with angiographically verified silent CAD and in 127 diabetic men without myocardial ischemia[82]. The groups were comparable for age and diabetes duration. The prevalence of ED was significantly higher in patients with silent CAD than in those without silent CAD (33.8% vs 4.7%, P = 0.000). Significant risk factors for silent CAD were identified using multiple logistic regression analysis. These risk factors included ED, apolipoprotein (a) polymorphism, smoking, microalbuminuria, HDL, and LDL. Interestingly, among these risk factors, ED was the strongest predictor of silent CAD (odds ratio 14.8). García-Malpartida et al[83] also examined the association between ED and silent myocardial ischemia (SMI) in 154 type 2 diabetic patients without a clinical evidence of CVD and demonstrated that ED was significantly associated with SMI (18.1% in patients with ED vs 4.1% in patients without ED, P = 0.018). Therefore, ED should be examined carefully in CKD patients not only for the improvement of their quality of life but also for the prevention of CVD.
Sufficient dialysis and adequate nutritional intake are necessary to improve the general condition of uremic patients. In addition, control of anemia using Epo and control of secondary hyperparathyroidism using phosphate binders, an active form of vitamin D and/or cinacalcet hydrochloride are required. Zinc supplementation may be necessary when zinc deficiency is suspected. If a psychological problem is suspected, psychotherapy and/or antidepressant medications may be necessary.
PDE5Is are inhibitors of PDE5 and suppress the degradation of cGMP, thereby stimulating the relaxation of smooth muscle in the cavernous body. Many studies have demonstrated the efficacy of PDE5Is for the treatment of ED in dialysis patients and in renal transplant recipients[84-90]. Although headache, flushing, and dyspepsia are the most common adverse effects[91], PDE5Is were well tolerated among dialysis patients in these studies. Among PDE5Is, sildenafil without dose adjustment has been used to treat ED in dialysis patients in several studies. However, it may be safer to start with half the dose (25 mg) and subsequently increase it up to 100 mg, depending on the patients’ responses. Special care should be taken when PDE5Is are administered to patients with cardiovascular or hepatic diseases.
Although testosterone replacement therapy is generally effective for patients with low circulating levels of testosterone when causes of ED are other than CKD, the administration of testosterone to uremic men usually fails to restore libido or potency, despite increased testosterone levels[92,93]. However, one pilot study demonstrated that treatment with testosterone gel improved erectile function in hypogonadal hemodialysis patients[94]. Testosterone stimulates an increase in NO production and degradation of cGMP, because it reportedly increases the activities of nNOS and PDE5 simultaneously[32,95,96]. Thus, the stimulatory effect of testosterone on NO production may be negated by its stimulatory effect on PDE5 activity. In this regard, combination therapy of testosterone and PDE5Is may be more effective than treatment with either testosterone or PDE5Is alone. Indeed, several reports demonstrated the efficacy of combination therapy on erectile function in hypogonadal men who did not respond to PDE5Is[97-100]. The efficacy of the combination therapy was also reported in dialysis patients and renal transplant recipients[101]. However, a recent randomized, double-blind, placebo-controlled trial did not show a significant effect of the addition of testosterone to sildenafil therapy on erectile function[102]. Therefore, the efficacy of the combination therapy is still controversial.
Other options for the treatment of ED include injecting prostaglandin E1 into the shaft of the penis, vacuum constriction devices and constriction bands, and penile prostheses. These treatments are beyond the scope of this review, and have not been discussed in detail.
It is well recognized that dialysis therapy does not improve sexual function[103,104]. Several reports demonstrated the improvement of erectile function after renal transplantation[104-106]. Nassir performed a prospective study in which the erectile function of 52 patients undergoing dialysis therapy was analyzed before and after renal transplantation[104]. No improvement of erectile function was observed in patients during dialysis therapy, whereas renal transplantation significantly improved erectile function. Akbari et al[107] examined the effect of renal transplantation on sperm quality and sex hormone levels. The authors found that sperm motility significantly improved, although morphology and sperm count did not change significantly. They also found that the level of testosterone significantly increased, whereas levels of FSH, LH and prolactin significantly decreased after renal transplantation. Furthermore, erectile function was compared between patients on dialysis therapy and renal transplant recipients in several studies, and erectile function was reportedly better in renal transplant recipients[108-110]. However, ED is still common in renal transplant recipients (approximately 50%)[111,112], and the prevention of the occurrence of CVD seems necessary in these patients to maintain erectile function[113,114].
Most studies on this topic collect information from patients on dialysis therapy and renal transplant recipients. Little reliable data exist with regard to the prevalence, etiology, and treatment of ED in CKD patients before starting dialysis therapy. Future studies are required to elucidate these points.
ED is a very common disease in CKD patients, and it is a multifactorial disease whose causes include hormonal, metabolic, nutritional, and psychological factors. PDE5Is are commonly used during treatment. Testosterone replacement therapy together with PDE5Is may be useful, particularly for CKD patients with hypogonadism. Renal transplantation may restore erectile function, particularly for young patients. ED is an early marker for CVD and it precedes the occurrence of CVD; therefore, ED should be examined carefully in CKD patients to avoid occurrence of CVD.
P- Reviewer: Bernieh B, Chung FT S- Editor: Song XX L- Editor: A E- Editor: Lu YJ
| 1. | Seftel AD, Sun P, Swindle R. The prevalence of hypertension, hyperlipidemia, diabetes mellitus and depression in men with erectile dysfunction. J Urol. 2004;171:2341-2345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 261] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 2. | Ayta IA, McKinlay JB, Krane RJ. The likely worldwide increase in erectile dysfunction between 1995 and 2025 and some possible policy consequences. BJU Int. 1999;84:50-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 776] [Cited by in RCA: 853] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 3. | Bacon CG, Mittleman MA, Kawachi I, Giovannucci E, Glasser DB, Rimm EB. Sexual function in men older than 50 years of age: results from the health professionals follow-up study. Ann Intern Med. 2003;139:161-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 522] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 4. | Akkus E, Kadioglu A, Esen A, Doran S, Ergen A, Anafarta K, Hattat H; Turkish Erectile Dysfunction Prevalence Study G. Prevalence and correlates of erectile dysfunction in Turkey: a population-based study. Eur Urol. 2002;41:298-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 164] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 5. | Navaneethan SD, Vecchio M, Johnson DW, Saglimbene V, Graziano G, Pellegrini F, Lucisano G, Craig JC, Ruospo M, Gentile G. Prevalence and correlates of self-reported sexual dysfunction in CKD: a meta-analysis of observational studies. Am J Kidney Dis. 2010;56:670-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 117] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 6. | Mesquita JF, Ramos TF, Mesquita FP, Bastos Netto JM, Bastos MG, Figueiredo AA. Prevalence of erectile dysfunction in chronic renal disease patients on conservative treatment. Clinics (Sao Paulo). 2012;67:181-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 7. | Nassir A. Erectile dysfunction risk factors for patients entering dialysis programme. Andrologia. 2010;42:41-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 8. | Gonzalez-Cadavid NF, Rajfer J. Molecular pathophysiology and gene therapy of aging-related erectile dysfunction. Exp Gerontol. 2004;39:1705-1712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Hedlund H, Andersson KE. Comparison of the responses to drugs acting on adrenoreceptors and muscarinic receptors in human isolated corpus cavernosum and cavernous artery. J Auton Pharmacol. 1985;5:81-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 88] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 10. | Saenz de Tejada I, Kim N, Lagan I, Krane RJ, Goldstein I. Regulation of adrenergic activity in penile corpus cavernosum. J Urol. 1989;142:1117-1121. [PubMed] |
| 11. | Saenz de Tejada I, Carson MP, de las Morenas A, Goldstein I, Traish AM. Endothelin: localization, synthesis, activity, and receptor types in human penile corpus cavernosum. Am J Physiol. 1991;261:H1078-H1085. [PubMed] |
| 12. | Kifor I, Williams GH, Vickers MA, Sullivan MP, Jodbert P, Dluhy RG. Tissue angiotensin II as a modulator of erectile function. I. Angiotensin peptide content, secretion and effects in the corpus cavernosum. J Urol. 1997;157:1920-1925. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 107] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 13. | Angulo J, Cuevas P, La Fuente JM, Pomerol JM, Ruiz-Castañé E, Puigvert A, Gabancho S, Fernández A, Ney P, Sáenz De Tejada I. Regulation of human penile smooth muscle tone by prostanoid receptors. Br J Pharmacol. 2002;136:23-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 62] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Burnett AL, Lowenstein CJ, Bredt DS, Chang TS, Snyder SH. Nitric oxide: a physiologic mediator of penile erection. Science. 1992;257:401-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 896] [Cited by in RCA: 852] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 15. | Burnett AL, Tillman SL, Chang TS, Epstein JI, Lowenstein CJ, Bredt DS, Snyder SH, Walsh PC. Immunohistochemical localization of nitric oxide synthase in the autonomic innervation of the human penis. J Urol. 1993;150:73-76. [PubMed] |
| 16. | Dail WG, Barba V, Leyba L, Galindo R. Neural and endothelial nitric oxide synthase activity in rat penile erectile tissue. Cell Tissue Res. 1995;282:109-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Hedlund P, Alm P, Andersson KE. NO synthase in cholinergic nerves and NO-induced relaxation in the rat isolated corpus cavernosum. Br J Pharmacol. 1999;127:349-360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 68] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Moncada S, Higgs A, Furchgott R. International Union of Pharmacology Nomenclature in Nitric Oxide Research. Pharmacol Rev. 1997;49:137-142. [PubMed] |
| 19. | Holmquist F, Stief CG, Jonas U, Andersson KE. Effects of the nitric oxide synthase inhibitor NG-nitro-L-arginine on the erectile response to cavernous nerve stimulation in the rabbit. Acta Physiol Scand. 1991;143:299-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 103] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Finberg JP, Levy S, Vardi Y. Inhibition of nerve stimulation-induced vasodilatation in corpora cavernosa of the pithed rat by blockade of nitric oxide synthase. Br J Pharmacol. 1993;108:1038-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 21. | Trigo-Rocha F, Aronson WJ, Hohenfellner M, Ignarro LJ, Rajfer J, Lue TF. Nitric oxide and cGMP: mediators of pelvic nerve-stimulated erection in dogs. Am J Physiol. 1993;264:H419-H422. [PubMed] |
| 22. | Kim N, Azadzoi KM, Goldstein I, Saenz de Tejada I. A nitric oxide-like factor mediates nonadrenergic-noncholinergic neurogenic relaxation of penile corpus cavernosum smooth muscle. J Clin Invest. 1991;88:112-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 299] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 23. | Okamura T, Ayajiki K, Fujioka H, Toda M, Fujimiya M, Toda N. Effects of endothelial impairment by saponin on the responses to vasodilators and nitrergic nerve stimulation in isolated canine corpus cavernosum. Br J Pharmacol. 1999;127:802-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Hurt KJ, Musicki B, Palese MA, Crone JK, Becker RE, Moriarity JL, Snyder SH, Burnett AL. Akt-dependent phosphorylation of endothelial nitric-oxide synthase mediates penile erection. Proc Natl Acad Sci USA. 2002;99:4061-4066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 282] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 25. | Prem AR, Punekar SV, Kalpana M, Kelkar AR, Acharya VN. Male reproductive function in uraemia: efficacy of haemodialysis and renal transplantation. Br J Urol. 1996;78:635-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 68] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 26. | Lim VS, Fang VS. Restoration of plasma testosterone levels in uremic men with clomiphene citrate. J Clin Endocrinol Metab. 1976;43:1370-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 62] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 27. | de Vries CP, Gooren LJ, Oe PL. Haemodialysis and testicular function. Int J Androl. 1984;7:97-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 28. | Levitan D, Moser SA, Goldstein DA, Kletzky OA, Lobo RA, Massry SG. Disturbances in the hypothalamic-pituitary-gonadal axis in male patients with acute renal failure. Am J Nephrol. 1984;4:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 26] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Stewart-Bentley M, Gans D, Horton R. Regulation of gonadal function in uremia. Metabolism. 1974;23:1065-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 83] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 30. | Dunkel L, Raivio T, Laine J, Holmberg C. Circulating luteinizing hormone receptor inhibitor(s) in boys with chronic renal failure. Kidney Int. 1997;51:777-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 72] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Chryssicopoulos A, Koutsikos D, Kapetanaki A, Agroyannis B, Tzanatos H, Rammos G, Fourtounas C, Kopelias I, Bossiolis B, Darema M. Evaluation of the hypothalamic-pituitary axis in uremic males using dynamic tests. The possible role of testicular inhibin: a preliminary report. Ren Fail. 1996;18:911-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 32. | Traish AM, Park K, Dhir V, Kim NN, Moreland RB, Goldstein I. Effects of castration and androgen replacement on erectile function in a rabbit model. Endocrinology. 1999;140:1861-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 171] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 33. | Shen ZJ, Zhou XL, Lu YL, Chen ZD. Effect of androgen deprivation on penile ultrastructure. Asian J Androl. 2003;5:33-36. [PubMed] |
| 34. | Gómez F, de la Cueva R, Wauters JP, Lemarchand-Béraud T. Endocrine abnormalities in patients undergoing long-term hemodialysis. The role of prolactin. Am J Med. 1980;68:522-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 101] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 35. | Isaac R, Merceron RE, Caillens G, Raymond JP, Ardaillou R. Effect of parathyroid hormone on plasma prolactin in man. J Clin Endocrinol Metab. 1978;47:18-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 36. | Caticha O, Norato DY, Tambascia MA, Santana A, Stephanou A, Sarlis NJ. Total body zinc depletion and its relationship to the development of hyperprolactinemia in chronic renal insufficiency. J Endocrinol Invest. 1996;19:441-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 37. | Maggi M, Buvat J, Corona G, Guay A, Torres LO. Hormonal causes of male sexual dysfunctions and their management (hyperprolactinemia, thyroid disorders, GH disorders, and DHEA). J Sex Med. 2013;10:661-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 38. | Sarnak MJ, Levey AS, Schoolwerth AC, Coresh J, Culleton B, Hamm LL, McCullough PA, Kasiske BL, Kelepouris E, Klag MJ. Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Hypertension. 2003;42:1050-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 786] [Cited by in RCA: 827] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 39. | Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7995] [Cited by in RCA: 8513] [Article Influence: 405.4] [Reference Citation Analysis (0)] |
| 40. | Thambyrajah J, Landray MJ, McGlynn FJ, Jones HJ, Wheeler DC, Townend JN. Abnormalities of endothelial function in patients with predialysis renal failure. Heart. 2000;83:205-209. |
| 41. | Bolton CH, Downs LG, Victory JG, Dwight JF, Tomson CR, Mackness MI, Pinkney JH. Endothelial dysfunction in chronic renal failure: roles of lipoprotein oxidation and pro-inflammatory cytokines. Nephrol Dial Transplant. 2001;16:1189-1197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 194] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 42. | Yilmaz MI, Stenvinkel P, Sonmez A, Saglam M, Yaman H, Kilic S, Eyileten T, Caglar K, Oguz Y, Vural A. Vascular health, systemic inflammation and progressive reduction in kidney function; clinical determinants and impact on cardiovascular outcomes. Nephrol Dial Transplant. 2011;26:3537-3543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 104] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 43. | Campese VM, Procci WR, Levitan D, Romoff MS, Goldstein DA, Massry SG. Autonomic nervous system dysfunction and impotence in uremia. Am J Nephrol. 1982;2:140-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 44. | Zucchelli P, Sturani A, Zuccalà A, Santoro A, Degli Esposti E, Chiarini C. Dysfunction of the autonomic nervous system in patients with end-stage renal failure. Contrib Nephrol. 1985;45:69-81. [PubMed] |
| 45. | Schaefer RM, Kokot F, Wernze H, Geiger H, Heidland A. Improved sexual function in hemodialysis patients on recombinant erythropoietin: a possible role for prolactin. Clin Nephrol. 1989;31:1-5. [PubMed] |
| 46. | Bommer J, Kugel M, Schwöbel B, Ritz E, Barth HP, Seelig R. Improved sexual function during recombinant human erythropoietin therapy. Nephrol Dial Transplant. 1990;5:204-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 47. | Evans RW, Rader B, Manninen DL. The quality of life of hemodialysis recipients treated with recombinant human erythropoietin. Cooperative Multicenter EPO Clinical Trial Group. JAMA. 1990;263:825-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 48. | Schaefer RM, Kokot F, Kuerner B, Zech M, Heidland A. Normalization of serum prolactin levels in hemodialysis patients on recombinant human erythropoietin. Int J Artif Organs. 1989;12:445-449. [PubMed] |
| 49. | Watschinger B, Watzinger U, Templ H, Spona J, Graf H, Luger A. Effect of recombinant human erythropoietin on anterior pituitary function in patients on chronic hemodialysis. Horm Res. 1991;36:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 50. | Steffensen G, Aunsholt NA. Does erythropoietin cause hormonal changes in haemodialysis patients? Nephrol Dial Transplant. 1993;8:1215-1218. [PubMed] |
| 51. | Schaefer F, van Kaick B, Veldhuis JD, Stein G, Schärer K, Robertson WR, Ritz E. Changes in the kinetics and biopotency of luteinizing hormone in hemodialyzed men during treatment with recombinant human erythropoietin. J Am Soc Nephrol. 1994;5:1208-1215. [PubMed] |
| 52. | Kokot F, Wiecek A, Grzeszczak W, Klin M. Influence of erythropoietin treatment on follitropin and lutropin response to luliberin and plasma testosterone levels in haemodialyzed patients. Nephron. 1990;56:126-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 53. | Allaf ME, Hoke A, Burnett AL. Erythropoietin promotes the recovery of erectile function following cavernous nerve injury. J Urol. 2005;174:2060-2064. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 48] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 54. | Sakanaka M, Wen TC, Matsuda S, Masuda S, Morishita E, Nagao M, Sasaki R. In vivo evidence that erythropoietin protects neurons from ischemic damage. Proc Natl Acad Sci USA. 1998;95:4635-4640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 715] [Cited by in RCA: 738] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 55. | Sirén AL, Fratelli M, Brines M, Goemans C, Casagrande S, Lewczuk P, Keenan S, Gleiter C, Pasquali C, Capobianco A. Erythropoietin prevents neuronal apoptosis after cerebral ischemia and metabolic stress. Proc Natl Acad Sci USA. 2001;98:4044-4049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 776] [Cited by in RCA: 747] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 56. | Moon C, Krawczyk M, Ahn D, Ahmet I, Paik D, Lakatta EG, Talan MI. Erythropoietin reduces myocardial infarction and left ventricular functional decline after coronary artery ligation in rats. Proc Natl Acad Sci USA. 2003;100:11612-11617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 231] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 57. | Parsa CJ, Matsumoto A, Kim J, Riel RU, Pascal LS, Walton GB, Thompson RB, Petrofski JA, Annex BH, Stamler JS. A novel protective effect of erythropoietin in the infarcted heart. J Clin Invest. 2003;112:999-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 371] [Cited by in RCA: 378] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 58. | Yang CW, Li C, Jung JY, Shin SJ, Choi BS, Lim SW, Sun BK, Kim YS, Kim J, Chang YS. Preconditioning with erythropoietin protects against subsequent ischemia-reperfusion injury in rat kidney. FASEB J. 2003;17:1754-1755. [PubMed] |
| 59. | Sharples EJ, Patel N, Brown P, Stewart K, Mota-Philipe H, Sheaff M, Kieswich J, Allen D, Harwood S, Raftery M. Erythropoietin protects the kidney against the injury and dysfunction caused by ischemia-reperfusion. J Am Soc Nephrol. 2004;15:2115-2124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 301] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 60. | Anagnostou A, Lee ES, Kessimian N, Levinson R, Steiner M. Erythropoietin has a mitogenic and positive chemotactic effect on endothelial cells. Proc Natl Acad Sci USA. 1990;87:5978-5982. [PubMed] |
| 61. | Anagnostou A, Liu Z, Steiner M, Chin K, Lee ES, Kessimian N, Noguchi CT. Erythropoietin receptor mRNA expression in human endothelial cells. Proc Natl Acad Sci USA. 1994;91:3974-3978. [PubMed] |
| 62. | Heeschen C, Aicher A, Lehmann R, Fichtlscherer S, Vasa M, Urbich C, Mildner-Rihm C, Martin H, Zeiher AM, Dimmeler S. Erythropoietin is a potent physiologic stimulus for endothelial progenitor cell mobilization. Blood. 2003;102:1340-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 636] [Cited by in RCA: 606] [Article Influence: 27.5] [Reference Citation Analysis (0)] |
| 63. | Bahlmann FH, De Groot K, Spandau JM, Landry AL, Hertel B, Duckert T, Boehm SM, Menne J, Haller H, Fliser D. Erythropoietin regulates endothelial progenitor cells. Blood. 2004;103:921-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 372] [Cited by in RCA: 372] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 64. | Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, Witzenbichler B, Schatteman G, Isner JM. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964-967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6624] [Cited by in RCA: 6340] [Article Influence: 226.4] [Reference Citation Analysis (1)] |
| 65. | Foresta C, Caretta N, Lana A, Cabrelle A, Palù G, Ferlin A. Circulating endothelial progenitor cells in subjects with erectile dysfunction. Int J Impot Res. 2005;17:288-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 72] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 66. | Baumhäkel M, Werner N, Böhm M, Nickenig G. Circulating endothelial progenitor cells correlate with erectile function in patients with coronary heart disease. Eur Heart J. 2006;27:2184-2188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 67. | Esposito K, Ciotola M, Maiorino MI, Giugliano F, Autorino R, De Sio M, Jannini E, Lenzi A, Giugliano D. Circulating CD34+ KDR+ endothelial progenitor cells correlate with erectile function and endothelial function in overweight men. J Sex Med. 2009;6:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 68. | Massry SG, Goldstein DA, Procci WR, Kletzky OA. Impotence in patients with uremia: a possible role for parathyroid hormone. Nephron. 1977;19:305-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 68] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 69. | Kimmel PL. Psychosocial factors in dialysis patients. Kidney Int. 2001;59:1599-1613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 226] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 70. | Lopes AA, Bragg J, Young E, Goodkin D, Mapes D, Combe C, Piera L, Held P, Gillespie B, Port FK. Depression as a predictor of mortality and hospitalization among hemodialysis patients in the United States and Europe. Kidney Int. 2002;62:199-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 366] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 71. | Chilcot J, Wellsted D, Da Silva-Gane M, Farrington K. Depression on dialysis. Nephron Clin Pract. 2008;108:c256-c264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 129] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 72. | Peng YS, Chiang CK, Hung KY, Chiang SS, Lu CS, Yang CS, Wu KD, Yang CC, Lin RP, Chang CJ. The association of higher depressive symptoms and sexual dysfunction in male haemodialysis patients. Nephrol Dial Transplant. 2007;22:857-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 73. | Fernandes GV, dos Santos RR, Soares W, de Lima LG, de Macêdo BS, da Fonte JE, de Carvalho BS, Coelho SN, Calado AA. The impact of erectile dysfunction on the quality of life of men undergoing hemodialysis and its association with depression. J Sex Med. 2010;7:4003-4010. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 74. | Antoniou LD, Shalhoub RJ, Sudhakar T, Smith JC. Reversal of uraemic impotence by zinc. Lancet. 1977;2:895-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 132] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 75. | Mahajan SK, Abbasi AA, Prasad AS, Rabbani P, Briggs WA, McDonald FD. Effect of oral zinc therapy on gonadal function in hemodialysis patients. A double-blind study. Ann Intern Med. 1982;97:357-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 86] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 76. | Rodger RS, Sheldon WL, Watson MJ, Dewar JH, Wilkinson R, Ward MK, Kerr DN. Zinc deficiency and hyperprolactinaemia are not reversible causes of sexual dysfunction in uraemia. Nephrol Dial Transplant. 1989;4:888-892. [PubMed] |
| 77. | Thompson IM, Goodman PJ, Tangen CM, Lucia MS, Miller GJ, Ford LG, Lieber MM, Cespedes RD, Atkins JN, Lippman SM. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349:215-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2044] [Cited by in RCA: 1882] [Article Influence: 85.5] [Reference Citation Analysis (0)] |
| 78. | Thompson IM, Tangen CM, Goodman PJ, Probstfield JL, Moinpour CM, Coltman CA. Erectile dysfunction and subsequent cardiovascular disease. JAMA. 2005;294:2996-3002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 642] [Cited by in RCA: 627] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 79. | Montorsi F, Briganti A, Salonia A, Rigatti P, Margonato A, Macchi A, Galli S, Ravagnani PM, Montorsi P. Erectile dysfunction prevalence, time of onset and association with risk factors in 300 consecutive patients with acute chest pain and angiographically documented coronary artery disease. Eur Urol. 2003;44:360-364; discussion 360-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 422] [Cited by in RCA: 399] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 80. | Baumhäkel M, Böhm M. Erectile dysfunction correlates with left ventricular function and precedes cardiovascular events in cardiovascular high-risk patients. Int J Clin Pract. 2007;61:361-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 53] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 81. | Kumar J, Bhatia T, Kapoor A, Ranjan P, Srivastava A, Sinha A, Kumar S, Garg N, Tewari S, Kapoor R. Erectile dysfunction precedes and is associated with severity of coronary artery disease among Asian Indians. J Sex Med. 2013;10:1372-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 82. | Gazzaruso C, Giordanetti S, De Amici E, Bertone G, Falcone C, Geroldi D, Fratino P, Solerte SB, Garzaniti A. Relationship between erectile dysfunction and silent myocardial ischemia in apparently uncomplicated type 2 diabetic patients. Circulation. 2004;110:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 228] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 83. | García-Malpartida K, Mármol R, Jover A, Gómez-Martínez MJ, Solá-Izquierdo E, Victor VM, Rocha M, Sanmiguel D, Hernández-Mijares A. Relationship between erectile dysfunction and silent myocardial ischemia in type 2 diabetic patients with no known macrovascular complications. J Sex Med. 2011;8:2606-2616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 84. | Chen J, Mabjeesh NJ, Greenstein A, Nadu A, Matzkin H. Clinical efficacy of sildenafil in patients on chronic dialysis. J Urol. 2001;165:819-821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 85. | Rosas SE, Wasserstein A, Kobrin S, Feldman HI. Preliminary observations of sildenafil treatment for erectile dysfunction in dialysis patients. Am J Kidney Dis. 2001;37:134-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 86. | Seibel I, Poli De Figueiredo CE, Telöken C, Moraes JF. Efficacy of oral sildenafil in hemodialysis patients with erectile dysfunction. J Am Soc Nephrol. 2002;13:2770-2775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 87. | YeniçerioGlu Y, Kefi A, Aslan G, Cavdar C, Esen AA, Camsari T, Celebi I. Efficacy and safety of sildenafil for treating erectile dysfunction in patients on dialysis. BJU Int. 2002;90:442-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 88. | Barrou B, Cuzin B, Malavaud B, Petit J, Pariente JL, Buchler M, Cormier L, Benoit G, Costa P. Early experience with sildenafil for the treatment of erectile dysfunction in renal transplant recipients. Nephrol Dial Transplant. 2003;18:411-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 89. | Sharma RK, Prasad N, Gupta A, Kapoor R. Treatment of erectile dysfunction with sildenafil citrate in renal allograft recipients: a randomized, double-blind, placebo-controlled, crossover trial. Am J Kidney Dis. 2006;48:128-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 90. | Vecchio M, Navaneethan SD, Johnson DW, Lucisano G, Graziano G, Querques M, Saglimbene V, Ruospo M, Bonifati C, Jannini EA. Treatment options for sexual dysfunction in patients with chronic kidney disease: a systematic review of randomized controlled trials. Clin J Am Soc Nephrol. 2010;5:985-995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 91. | Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med. 1998;338:1397-1404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1644] [Cited by in RCA: 1537] [Article Influence: 56.9] [Reference Citation Analysis (0)] |
| 92. | Lawrence IG, Price DE, Howlett TA, Harris KP, Feehally J, Walls J. Correcting impotence in the male dialysis patient: experience with testosterone replacement and vacuum tumescence therapy. Am J Kidney Dis. 1998;31:313-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 47] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 93. | Brockenbrough AT, Dittrich MO, Page ST, Smith T, Stivelman JC, Bremner WJ. Transdermal androgen therapy to augment EPO in the treatment of anemia of chronic renal disease. Am J Kidney Dis. 2006;47:251-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 94. | Cangüven O, Aykose G, Albayrak S, Goktas C, Horuz R, Yencilek F. Efficacy of testosterone gel in the treatment of erectile dysfunction in hypogonadal hemodialysis patients: a pilot study. Int J Impot Res. 2010;22:140-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 95. | Morelli A, Filippi S, Mancina R, Luconi M, Vignozzi L, Marini M, Orlando C, Vannelli GB, Aversa A, Natali A. Androgens regulate phosphodiesterase type 5 expression and functional activity in corpora cavernosa. Endocrinology. 2004;145:2253-2263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 243] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 96. | Zhang XH, Morelli A, Luconi M, Vignozzi L, Filippi S, Marini M, Vannelli GB, Mancina R, Forti G, Maggi M. Testosterone regulates PDE5 expression and in vivo responsiveness to tadalafil in rat corpus cavernosum. Eur Urol. 2005;47:409-416; discussion 416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 141] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 97. | Aversa A, Isidori AM, Spera G, Lenzi A, Fabbri A. Androgens improve cavernous vasodilation and response to sildenafil in patients with erectile dysfunction. Clin Endocrinol (Oxf). 2003;58:632-638. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 210] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 98. | Shabsigh R, Kaufman JM, Steidle C, Padma-Nathan H. Randomized study of testosterone gel as adjunctive therapy to sildenafil in hypogonadal men with erectile dysfunction who do not respond to sildenafil alone. J Urol. 2004;172:658-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 226] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 99. | Shamloul R, Ghanem H, Fahmy I, El-Meleigy A, Ashoor S, Elnashaar A, Kamel I. Testosterone therapy can enhance erectile function response to sildenafil in patients with PADAM: a pilot study. J Sex Med. 2005;2:559-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 53] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 100. | Buvat J, Montorsi F, Maggi M, Porst H, Kaipia A, Colson MH, Cuzin B, Moncada I, Martin-Morales A, Yassin A. Hypogonadal men nonresponders to the PDE5 inhibitor tadalafil benefit from normalization of testosterone levels with a 1% hydroalcoholic testosterone gel in the treatment of erectile dysfunction (TADTEST study). J Sex Med. 2011;8:284-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 101. | Chatterjee R, Wood S, McGarrigle HH, Lees WR, Ralph DJ, Neild GH. A novel therapy with testosterone and sildenafil for erectile dysfunction in patients on renal dialysis or after renal transplantation. J Fam Plann Reprod Health Care. 2004;30:88-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 102. | Spitzer M, Basaria S, Travison TG, Davda MN, Paley A, Cohen B, Mazer NA, Knapp PE, Hanka S, Lakshman KM. Effect of testosterone replacement on response to sildenafil citrate in men with erectile dysfunction: a parallel, randomized trial. Ann Intern Med. 2012;157:681-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 109] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 103. | Soykan A, Boztas H, Kutlay S, Ince E, Nergizoglu G, Dileköz AY, Berksun O. Do sexual dysfunctions get better during dialysis? Results of a six-month prospective follow-up study from Turkey. Int J Impot Res. 2005;17:359-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 104. | Nassir A. Sexual function in male patients undergoing treatment for renal failure: a prospective view. J Sex Med. 2009;6:3407-3414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 105. | Shamsa A, Motavalli SM, Aghdam B. Erectile function in end-stage renal disease before and after renal transplantation. Transplant Proc. 2005;37:3087-3089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 106. | Ahmad M, Rafiuddin Q, Hassan U, Ahmad A, Husain S. Impact of renal transplantation on erectile dysfunction due to chronic renal failure in male patients. J Ayub Med Coll Abbottabad. 2009;21:69-71. [PubMed] |
| 107. | Akbari F, Alavi M, Esteghamati A, Mehrsai A, Djaladat H, Zohrevand R, Pourmand G. Effect of renal transplantation on sperm quality and sex hormone levels. BJU Int. 2003;92:281-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 77] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 108. | Barroso LV, Miranda EP, Cruz NI, Medeiros MA, Araújo AC, Mota Filho FH, Medeiros FC. Analysis of sexual function in kidney transplanted men. Transplant Proc. 2008;40:3489-3491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 109. | Tavallaii SA, Mirzamani M, Heshmatzade Behzadi A, Assari S, Khoddami Vishteh HR, Hajarizadeh B, Einollahi B. Sexual function: a comparison between male renal transplant recipients and hemodialysis patients. J Sex Med. 2009;6:142-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 110. | Al Khallaf HH. Analysis of sexual functions in male nondiabetic hemodialysis patients and renal transplant recipients. Transpl Int. 2010;23:176-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 111. | Lasaponara F, Paradiso M, Milan MG, Morabito F, Sedigh O, Graziano ME, Abbona A, Piccoli GB, Rossetti M, Mezza E. Erectile dysfunction after kidney transplantation: our 22 years of experience. Transplant Proc. 2004;36:502-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 112. | Espinoza R, Gracida C, Cancino J, Ibarra A. Prevalence of erectile dysfunction in kidney transplant recipients. Transplant Proc. 2006;38:916-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 113. | Diemont WL, Hendriks JC, Lemmens WA, Langen Hv, Berden JH, Meuleman EJ. Prognostic factors for the vascular components of erectile dysfunction in patients on renal replacement therapy. Int J Impot Res. 2003;15:44-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 114. | Rebollo P, Ortega F, Valdés C, Fernández-Vega F, Ortega T, García-Mendoza M, Gómez E. Factors associated with erectile dysfunction in male kidney transplant recipients. Int J Impot Res. 2003;15:433-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.3] [Reference Citation Analysis (0)] |