Published online Nov 25, 2022. doi: 10.5501/wjv.v11.i6.485
Peer-review started: July 28, 2022
First decision: August 22, 2022
Revised: September 1, 2022
Accepted: October 14, 2022
Article in press: October 14, 2022
Published online: November 25, 2022
Processing time: 118 Days and 2.6 Hours
Coronavirus disease 2019 (COVID-19) has crippled humanity since early 2020. Various sequelae of COVID-19 have been reported in different body systems. Musculoskeletal symptoms are widely reported during COVID-19 infection, but musculoskeletal complications in long COVID-19 are underreported. However, post-COVID-19 survivors have reported complaints of persisting or new-onset fatigue, myalgia, arthralgia, arthritis, muscle weakness, etc in clinical practice. The well-known detrimental effects of steroids on the musculoskeletal system coupled with their over-the-counter availability can also be anticipated since they were the cornerstone of life-saving management in this pandemic.
To determine the musculoskeletal complications in long COVID.
We performed a systematic review of ‘systematic reviews and meta-analyses’.
Of the 63 articles screened, 24 articles were included. Two articles specifically discussed children and adolescents. One article discussed rehabilitation intervention. No article addressed rehabilitation of musculoskeletal issues in long COVID-19 in particular. Fatigue was the most common musculoskeletal complication.
Fatigue is found to be very common along with myalgia and arthralgia. There were no studies on rehabilitation intervention in musculoskeletal complications specifically. Considering the lacuna in literature and the needs of the current situation, further studies are warranted to standardize effective rehabilitation interventions in musculoskeletal complications. More homogenous studies are needed. Studies on functional impairment due to musculoskeletal involvement are essential.
Core Tip: Though musculoskeletal involvement is reported in severe acute respiratory syndrome coronavirus 2 infection, the literature is limited for musculoskeletal symptoms in long coronavirus disease 2019 (COVID-19). Moreover, rehabilitation of each musculoskeletal complaint is not addressed in most reviews. We highlighted those keys areas through our review article. Fatigue is the most common musculoskeletal issue in long COVID-19. Considering the gaps in literature and current needs, future studies are warranted to standardize effective rehabilitation interventions in musculoskeletal complications.
- Citation: Swarnakar R, Jenifa S, Wadhwa S. Musculoskeletal complications in long COVID-19: A systematic review. World J Virol 2022; 11(6): 485-495
- URL: https://www.wjgnet.com/2220-3249/full/v11/i6/485.htm
- DOI: https://dx.doi.org/10.5501/wjv.v11.i6.485
Since 2020 the world has witnessed multiple waves of the coronavirus disease 2019 (COVID-19) pandemic caused by different variants of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) at different times and places. As of September 1, 2022, 599 million confirmed cases and more than 6 million deaths have been reported[1]. The loss of lives, superimposed by the deterioration of the quality of life of a significant number of survivors, made this pandemic a huge hurdle for the whole world. A range of long-term effects or complications involving different body systems have been reported. The respiratory sequelae of COVID-19 have been widely investigated, but musculoskeletal complications are underreported. Here we performed a systematic review of systematic reviews and meta-analyses to find musculoskeletal complications caused by long COVID-19 conditions.
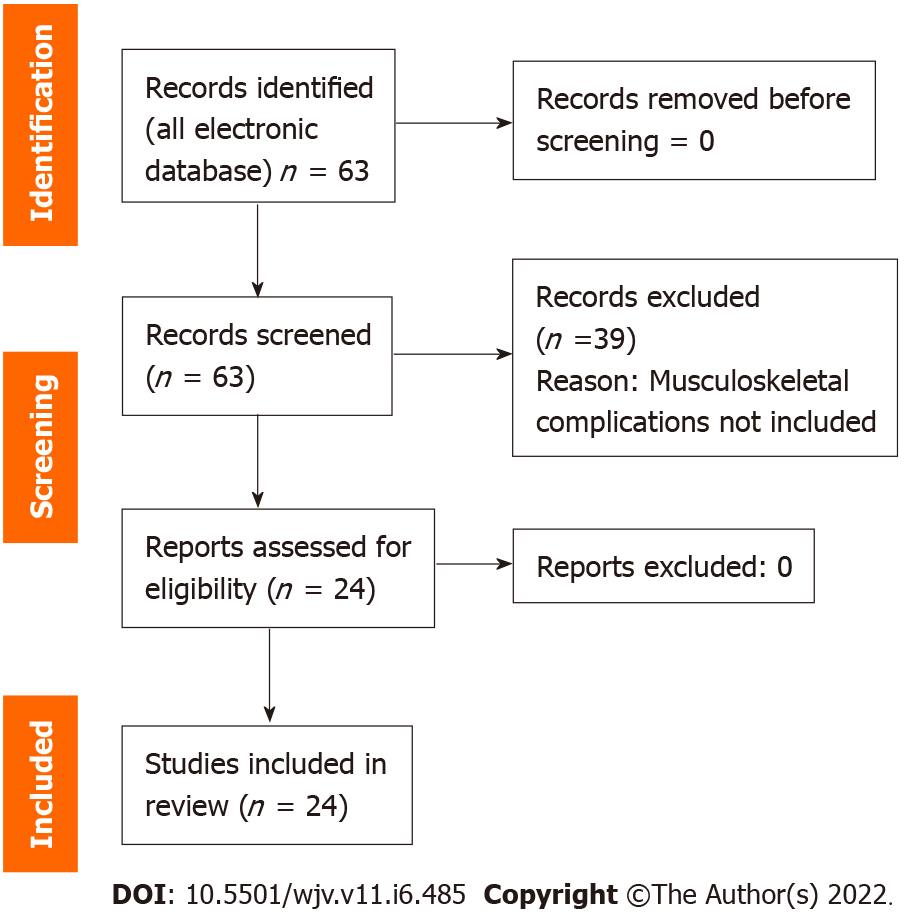
Here a systematic review of systematic reviews and meta-analyses was conducted (Figure 1). We also cited high-quality articles in Reference Citation Analysis (https://www.referencecitationanalysis.com).
PICOS model: (1) Studies that considered patients with long-term COVID-19 symptoms at least > 4 wk of COVID-19 infections (population); (2) Studies where the primary aim was to evaluate long-term COVID-19 symptoms in mild, moderate, severe, and critical patients that have a follow-up of at least 14 d (interventions); (3) Studies with or without a control group (comparisons); (4) Studies that reported the long COVID-19 symptoms (outcomes); and (5) Systematic review and meta-analyses (study designs). From January 2020 to mid-July 2022, any relevant studies that followed the above mentioned PICOS model and that reported musculoskeletal complications in long COVID-19 were eligible for inclusion.
The search was carried out by two independent researchers in all electronic databases, mainly MEDLINE, EMBASE, Web of Science, and Google Scholar with this time period. We combined search terms and key words related to the population (e.g., “COVID-19”, “SARS-CoV-2”, “long Covid-19”, “long Covid”, “long haulers”) and outcomes (e.g., “fatigue”, “pain”, “musculoskeletal”, “myalgia”, “myopathy”, “arthralgia”, “arthritis”, “rheumatic”, “joint”). We additionally filtered study designs “systemic review” and “meta-analyses” in humans.
All the systematic reviews and meta-analyses on long COVID-19 following our above-mentioned PICOS model were included. After the preliminary search, we extracted the musculoskeletal complications that were reported in long COVID-19 studies or in post COVID-19 studies (at least 4 wk after COVID-19 active infection). We excluded any musculoskeletal issues that occurred after any neurological sequelae of long COVID-19 and excluded any myocarditis or issues related to smooth muscle dysfunction.
Titles and abstracts were screened for potentially eligible studies. Following an initial screening, full texts of potentially eligible studies were acquired for detailed evaluation eliminating all duplicates. Manual scanning of key articles and review papers was conducted to identify additional articles missed by the search strategy. Two reviewers assessed the articles independently and in case of any disagreements, the opinion of the third reviewer was consulted.
We performed a descriptive analysis of the included reviews.
Of the 63 articles screened; 24 articles were included[2-25]. Two articles specifically discussed children and adolescents. One article discussed rehabilitation intervention. No article addressed rehabilitation on musculoskeletal issues in long COVID-19 in particular. Details of the selected articles are listed in Table 1.
| Serial no. | Ref. | Reported musculoskeletal complications | Type of study | Types of patients | Rehabilitation intervention |
| 1 | Ludvigsson[2], 2021 | Fatigue, muscle weakness | Systematic review | Children | No |
| 2 | Akbarialiabad et al[3], 2021 | Fatigue (63%), muscle weakness | Systematic scoping review | All age groups | No |
| 3 | Michelen et al[4], 2021 | Weakness (41%; 95%CI: 25%-59%), general malaise (33%; 95%CI: 15%-57%), fatigue (31%; 95%CI: 24%-39%) | Living systematic review | All age groups | No |
| 4 | Iqbal et al[5], 2021 | 48% fatigue in >12 wk | Systematic review and meta-analysis | All age groups | No |
| 5 | Vollbracht and Kraft[6], 2021 | Vitamin C improved in post-COVID-19 fatigue; the IV vitamin C doses administered ranged from 3.5 g to > 75 g/d | A systematic review on intervention | All age groups | No |
| 6 | Jennings et al[7], 2021 | Arthralgia 13% (6%-29%), myalgia 34% (2%-86%), fatigue 44% (10%-71%) | Systematic review | All age groups | No |
| 7 | Fernández-de-Las-Peñas et al[8], 2021 | Fatigue (58%), headache (44%), joint pain (15%-20%) | Systematic review | All age groups | No |
| 8 | Malik et al[9], 2022 | Fatigue (64, 54-73), arthralgia (24.3, 14.0-36.0), headache (21, 3-47) | Systematic review and meta-analysis | All age groups | No |
| 9 | Ceban et al[10], 2022 | Fatigue in 30% of cases | Systematic review and meta-analysis | All age groups | No |
| 10 | Chen et al[11], 2022 | Fatigue prevalence 0.23 (95%CI: 0.17-0.30) | Systematic review and meta-analysis | All age groups | No |
| 11 | van Kessel et al[12], 2022 | Fatigue most common | Systematic review | All age groups | No |
| 12 | Alkodaymi et al[13], 2022 | Fatigue 3-6 mo follow-up 32%, 36% 6-9 mo, 37% 9-12 mo, > 12 mo, 41% | Systematic review | All age groups | No |
| 13 | Fernández-de-Las-Peñas et al[14], 2022 | Prevalence of post-COVID-19 myalgia, joint pain, and chest pain ranged from 5.65% to 18.15%, 4.6% to 12.1%, and 7.8% to 23.6%, respectively, at different follow-up periods during the 1st yr postinfection. Almost 10% of individuals infected by SARS-CoV-2 will suffer from musculoskeletal post-COVID-19 pain symptomatology at some time during the 1st yr after the infection | Systematic review | All age groups | No |
| 14 | Han et al[15], 2022 | Fatigue/weakness (28%, 95%CI: 18%-39%), arthromyalgia (26%, 95%CI: 8%-44%) | Systematic review | All age groups | No |
| 15 | d’Ettorre et al[16], 2022 | 63% of fatigue reported | Systematic review | All age groups | No |
| 16 | Behnood et al[17], 2022 | 47% fatigue, 25% myalgia, 35% headache, females with higher pain symptoms | Systematic review | In children and young people | No |
| 17 | Nguyen et al[18], 2022 | Fatigue (16%-64%), arthralgia (8%-55%), thoracic pain (5%-62%), myalgia (1%-22%), headache (9%-15%) | Systematic review | All age groups | No |
| 18 | Lopez-Leon et al[19], 2022 | Fatigue (9.66%) | Systematic review | Children and adolescents | No |
| 19 | Abdel-Gawad et al[20], 2022 | Fatigue (72.8%) and joint pain (31.4%) | Systematic review | All age groups | No |
| 20 | Almas et al[21], 2022 | Fatigue (54.11%), arthralgia (16.35%), myalgia (5.78%), chest pain (10.37%) | Systematic review | All age groups | No |
| 21 | Maglietta et al[22], 2022 | Fatigue and female sex association statistically significant, with OR = 1.54, 95%CI: 1.32-1.79 | Systematic review | All age groups | No |
| 22 | Healey et al[23], 2022 | fatigue (37%; 95%CI: 23%-55%), myalgia (12%; 95%CI: 5%-25%), headache (7%; 95%CI: 3%-16%), chest pain (3%; 95%CI: 1%-8%) | Systematic review | All age groups | No |
| 23 | de Oliveira Almeida et al[24], 2022 | Fatigue. COVID-19 survivors can have a reduction in physical function, ability to perform activities of daily living and their health-related quality of life 1-6 mo post-infection | Systematic review | All age groups | No |
| 24 | Fugazzaro et al[25], 2022 | Muscle strength, walking capacity, sit-to-stand performance improvements | Systematic review of rehabilitation interventions | All age groups | Yes |
According to the National Institute of Health and Care Excellence guidelines, post-acute COVID-19 and post-COVID-19 syndrome are included in long COVID. Post-acute-COVID-19 means ongoing symptomatic COVID-19 for people who still have symptoms 4 wk and 12 wk after acute COVID-19. On the other hand, post-COVID-19 syndrome means that patients are having persisting symptoms for > 12 wk after acute symptoms[26]. According to the World Health Organization, post-COVID-19 conditions generally occur 3 mo from the onset of COVID-19 with symptoms lasting for at least 2 mo and should be unexplained by any alternative diagnosis[27].
Another definition consists of “not recovering several weeks or months following the start of symptoms that were suggestive of COVID-19, regardless individuals were tested or not”[28]. Common symptoms reported are fatigue, shortness of breath, cognitive dysfunction/attention disorder, hair loss, and dyspnea[29,30]. Musculoskeletal symptoms of skeletal muscle, neurological, bone, and joint disorders have also been reported. The proinflammatory responses can impact nearly every organ system, including the musculoskeletal system. Myalgias, arthralgias, fatigue, exercise, and intolerance are some of the common musculoskeletal sequelae.
SARS-CoV-2 has three structural proteins (membrane protein, spike protein, and envelope protein). Spike glycoprotein through its subunits S1 and S2 helps in entering the host cells[31]. The angiotensin-converting enzyme 2 (ACE2) receptor acts as the entry receptor using the serine protease trans-membrane protease, serine 2 (TMPRSS2) for spike protein priming[32]. Following the binding of the receptor, viral spike protein is broken down by TMPRSS2 proteolytically, which exposes a fusion peptide signal that helps in the fusion of viral and human membranes. It leads to the cytoplasmic release of viral RNA. Interestingly, ACE2 is found in the lung, heart, kidney, liver, gastrointestinal, and musculoskeletal systems.
In humans, endothelial cells, smooth muscle cells, pericytes, muscle stem cells, macrophages, B cells, T cells, natural killer cells, and myonuclei express TMPRSS2. Furthermore, several cells in the synovium including fibroblasts, monocytes, B cells, and T cells express ACE2 and TMPRSS2. However, only smooth muscle cells and pericytes express ACE2. Articular cartilage (proliferative, hypertrophic, and effector chondrocytes) express ACE2, and only homeostatic chondrocytes (which control circadian rhythm in cartilage) express TMPRSS2. In the meniscus, a few cartilage progenitors and regulatory fibrochondrocytes express ACE2 (no TMPRSS2 is detected). ACE2 is also found to be present in composite unenriched cortical and trabecular bone and osteoblast enriched tissues. TMPRSS2 was almost absent in composite bone tissue, and TMPRSS2 was detected in all osteoblast-enriched samples.
The presence of these receptors implies that skeletal muscle, synovium, and cortical bone may serve as potential areas of direct SARS-CoV-2 infection and its probable long-term sequelae[33]. The cytokines and signaling molecules are induced by the infection [C-X-C motif chemokine 10, interferon-gamma, interleukin (IL)-1β, IL-6, IL-8, IL-17, and tumor necrosis factor-alpha (TNF-α)]. They play a crucial role in the pathogenesis of clinical signs and symptoms and long-term sequelae of COVID-19. Interferon-gamma, IL-1β, IL-6, IL-17, and TNF-α show a negative impact on skeletal muscle (fiber proteolysis and decreasing protein synthesis). IL-1β and IL-6 may lead to fibrosis after inducing increased muscle fibroblast activity. IL-1β and TNF-α induce muscle fiber growth by inhibiting the differentiation and proliferation of satellite cells, the progenitor cells[34].
Corticosteroids, a lifesaving medication in the management of COVID-19, has been overused in many cases. Additionally, long-term corticosteroid use has been known to cause a variety of effects on the bone, including osteonecrosis, reduced bone mineral density (BMD), avascular necrosis of the hip joint, and osteoporosis with or without fracture. It implies that steroids might be an important cause of multiple musculoskeletal complications.
Many studies have reported fatigue myalgia and generalized weakness as some of the common persisting complaints in symptomatic infections of the disease[35]. In the previous epidemics of SARS, extensive myalgias and muscle dysfunction were also reported. Direct viral infection and/or the cytokine storm could lead to pathological changes in skeletal muscle tissue in addition to deconditioning due to prolonged disuse during the hospitalization or disease period.
Mayer et al[36] showed that a long intensive care unit stay is linked with a rapid and significant reduction in the volume of the rectus femoris muscle (average: 18.5%), until the 7th d of hospitalization. Carfì et al[37], in a study to follow up the post-COVID-19 patients in a hospital in Italy, found that in recovered patients, 87.4% responded with at least one persistent symptom, especially fatigue. Paneroni et al[38] evaluated the muscle strength of the quadriceps and biceps femoris of patients in post-discharge recovered COVID-19 cases. They found that 86% of cases had quadriceps weakness and 73% had biceps femoris weakness. These findings proved muscle dysfunction in individuals with long COVID-19. Jacobs et al[39] in their study to assess the persistence of symptoms and quality of life at 35 d after hospitalization of COVID-19 infection found fatigue as the most common persisting symptom.
Fatigue was found to be the most common symptom followed by shortness of breath (31%), loss of smell (22%), and muscle ache (21%) by the Office for the National Statistics, census 2021, in the estimates of the prevalence of self-reported long COVID-19 and associated activity limitation using United Kingdom Coronavirus (COVID-19) Infection Survey data[40]. Compared with age-matched healthy controls, approximately 2-3 mo after discharge, moderate to severe cases had a 32% reduction in grip strength and a 13% reduction in the distance walked in 6 min[41].
Aiyegbusi et al[42] did a review on symptoms, complications, and management of long COVID-19 and found that 47% reporting fatigue as the most common, myalgia (muscle pain) in 25%, and joint pain in 20%. Varghese et al[43] found that 54% of the patients reported fatigue as one of the persisting symptoms. Huang et al[44] did a follow-up study from June 16, 2020 to September 3, 2020 to assess 6 mo consequences of COVID-19 in patients discharged from the hospital, and they reported fatigue (63%) and sleep difficulties (26%) as the most common symptoms. Miyazato et al[45] also reported fatigue as one of the prolonged and late-onset symptoms conducted in patients admitted for COVID-19 to the Disease Control and Prevention Center and National Center for Global Health and Medicine from February to June 2020. Daher et al[46] conducted a follow-up study on 33 confirmed COVID-19 positive patients 6 wk post-discharge to assess the pulmonary and extrapulmonary disease sequelae and found a significant tendency among the patients to suffer from fatigue symptoms with significant limitations of their mobility, which was reflected by reduced 6-min walking test distance among the extrapulmonary sequelae. In their study, characterizing long COVID-19 in an international cohort over 7 mo of symptoms and their impact, Davis et al[47] also reported the patients who have had or were suspicious of COVID-19 reported fatigue as the most common persisting symptom even after 6 mo.
Multiple etiologies of fatigue (physical, mental, emotional) could be present. Therefore, fatigue should be researched according to the accompanying symptoms or more specific features[48]. Another sequelae is intolerance to physical activities associated with a chronic fatigue condition and difficulty in returning to normal daily life[49]. Eighteen people living with long COVID-19 in the United Kingdom were interviewed with a semi-structured questionnaire in a qualitative study by Humphrey et al[50] showing people faced reduced physical function, compounded by the cognitive and psychological effects of long COVID-19.
Arthralgia is pain localized to the joints, while myalgia is pain localized to muscle. They are typically present in the early course of the disease and in patients experiencing long-term effects of COVID-19 or a prolonged disease course. Studies have described how SARS-CoV-2 infection induces systemic elevations of cytokines and signaling molecules. This ‘cytokine storm’ is thought to be implicated in musculoskeletal manifestations, among many others. Myalgia and arthralgia are reported as one of the most common persistent symptoms in patients with post-acute sequelae of COVID-19 and are more notable in patients who were prone to being positioned during intensive care unit admission[51].
In a study of 294 patients hospitalized with COVID-19, Hoong et al[52] observed that 30% of patients reported musculoskeletal complaints; 37.5% had myalgia, 5.7% had arthralgia, 6.8% had new-onset backache, and 50% had generalized body aches. Elhiny et al[53] reported that physical decline was the most common symptom reported in musculoskeletal complications. Patients who also had mild to moderate forms of the infection can experience exacerbated muscle and joint pain. Petersen et al[54] in their study of long COVID-19 in a longitudinal study in the Faroe Islands found out arthralgia is one of the most persistent symptoms following fatigue and loss of smell and taste.
Follow-up of adults with non-critical COVID-19 after symptom onset in a study by Carvalho-Schneider et al[55] found that 13% of the patients who never had arthralgia at the onset of the disease reported arthralgia 30 d after discharge and 21% after 60 d. The study by Chopra et al[56] on clinical predictors of long COVID-19 symptoms in patients with mild COVID-19 at 30 d post-discharge (long COVID-19) found myalgia as one of the most common persistent symptoms following fatigue and cough. Stavem et al[57] also reported myalgia as one of the most common persisting symptoms 1.5-6.0 mo after infection in non-hospitalized patients. Ghosn et al[58] in a large prospective cohort study in France among the post-discharge patients at 3 mo and 6 mo observed mostly fatigue, dyspnea, joint pain, and myalgia. COVID-19 has also been found to cause reactive arthritis and new-onset inflammatory arthritis typically occurring within a month after its diagnosis[59].
There were reported cases of reactive arthritis post discharge from COVID-19[60]. Derksen et al[61] in a Dutch study of 5 patients who presented with inflammatory arthritis 6.6 wk post COVID-19 infection, found that 2 patients had strongly positive and another patient had weakly positive anti-CCP antibodies, suggesting post-COVID-19 rheumatoid arthritis development.
C-X-C motif chemokine 10, IL-17, and TNF-α induce osteoclastogenesis and inhibit osteoblast proliferation and differentiation causing increased bone fragility[34]. Berktaş et al[62] assessed the BMD of hospitalized COVID-19 patients at diagnosis and follow-up visits using chest computed tomography. BMD was retrospectively measured by quantitative computed tomography. BMD decreased by a mean of 8.6% (± 10.5%) from diagnosis to follow-up. The osteoporosis ratio increased two-fold after hospitalization for COVID-19 because of this substantial bone loss.
An animal experimental study characterized the effects of SARS-CoV-2 infections on bone metabolism in an established golden Syrian hamster model for COVID-19. SARS-CoV-2 caused significant multifocal loss of bone trabeculae in the long bones and lumbar vertebrae of all infected hamsters implicating the same could happen in humans post-COVID-19. A multicenter study by Kottlor et al[63] showed that COVID-19 patients requiring intensive care had significantly lower BMD than those who were managed in non-intensive care settings.
Researchers at Indiana University School of Medicine discovered that the mouse models infected with the novel coronavirus lost nearly 25% of their bone mass within 2 wk of infection. They also found mouse models with a 63% increase in osteoclasts, the cells that cause the bone to break down.
Musculoskeletal manifestations can be a result of underlying neurological disturbances. The central and peripheral nervous systems control our movements via the spinal motor neurons, which act as the final common pathway to the muscles[64]. Many studies have reported peripheral neuropathy, most commonly Guillain-Barre and related symptoms. Guillain-Barre syndrome and critical illness-induced polyneuropathy/myopathy are two important peripheral neuropathies seen in COVID-19[65].
A follow-up study conducted for 8 mo in Denmark performed electromyography and conventional nerve conduction study of 20 patients with persistent fatigue. They found that all patients with myopathic electromyography reported physical fatigue; 8 patients reported about myalgia while 3 patients without myopathic changes complained about physical fatigue. Long-term COVID-19 does not cause large fiber neuropathy, but myopathic changes were seen[66]. Acute myopathies are reported in acute COVID-19 infection[67], which may have a detrimental effect in the muscle in the post infective stages.
COVID-19 has multisystem effects including physical as well as psychological effects. The wholesome evaluation and rehabilitation of such patients require a multifaceted and interdisciplinary approach to cover all aspects properly. Identification of the pre-existing disabling conditions contributing to the cumulative effect of long COVID-19 is also an important aspect. Reinfection, post-viral bacterial and fungal infections, baseline routine investigations along with C-reactive protein, fibrinogen, D-dimer, troponin, and ferritin can also be considered if clinically indicated. Cardiac function tests (echocardiography) should be done to check cardiopulmonary status before framing the exercise program.
Rehabilitation should be addressed holistically following the domains of the International Classification of Functioning, Disability, and Health. Studies have shown that early mobilization helps in the reduction of the harmful effects of the disease, especially on muscle and cardiopulmonary function, mobility, and function[68], implying rehabilitation of long COVID-19 should start from the beginning. Physical exercise should be individualized specifying intensity, frequency, duration, and type of exercise. Exercise should be gradually increased according to one’s capacity. The patient should be educated with an emphasis on self-management. The patient should respect the pain and their own capabilities. Energy conservation techniques such as simplifying tasks, pacing the activities over time, and taking breaks should be followed. Repeated practice of functional activities and a set of specific actions according to the patient’s priorities, needs, and goals may improve the functional aspects. All such activities need to be evaluated regularly to determine whether they should be continued, changed, or stopped[69].
However, no studies on rehabilitation intervention have been investigated in long COVID-19 for musculoskeletal complications in particular[70]. In our systematic reviews, we did only descriptive analysis. We did not address the individual cases or case series study or any cohort or trials, which may miss the characteristics of the individual cases in particular. However, performing a systematic review of all systematic reviews and meta-analyses provided a stronger evidence-based study.
Musculoskeletal involvement is common during active SARS-CoV-2 infection. Fatigue is very common during this phase. Here we have highlighted the musculoskeletal complications in long COVID-19 syndrome. Again, fatigue is found to be very common along with myalgia and arthralgia. There is a lack of studies on these aspects. Moreover, all the studies are heterogeneous, especially in terms of the duration of post-COVID and the definition of long COVID. There are no studies for rehabilitation intervention in musculoskeletal complications specifically. This study reinforced the gravity of the current situation. Considering the lacuna in literature and the needs of the current situation, further studies are warranted to standardize effective rehabilitation interventions in musculoskeletal complications. More homogenous studies are needed using proper case definition and duration of long COVID. Studies on functional impairment due to musculoskeletal involvement are needed.
Research is lacking in musculoskeletal complications in long coronavirus disease 2019 (COVID-19).
Currently, many long COVID-19 patients are coming to outpatient departments of rehabilitation for musculoskeletal issues.
To find musculoskeletal complications in long COVID-19 and relevant rehabilitation interventions.
A systematic review of systematic reviews and meta-analyses was done.
Among many musculoskeletal issues, fatigue was found to be the most common complication. Rehab intervention is severely lacking in literature.
Rehabilitation need identification is of the utmost importance in musculoskeletal aspects of long COVID. Fatigue was found to be the most common complication.
Identification of rehabilitation needed following identification of musculoskeletal complications is crucial in long COVID-19 cases.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Virology
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Mahmoud MZ, Saudi Arabia; Mehri-Ghahfarrokhi A, Iran S-Editor: Wang JJ L-Editor: Filipodia P-Editor: Wang JJ
| 1. | World Health Organization. Data at WHO. [cited 1 September 2022]. Available from: https://www.who.int/data. |
| 2. | Ludvigsson JF. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID-19. Acta Paediatr. 2021;110:914-921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 261] [Cited by in RCA: 217] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 3. | Akbarialiabad H, Taghrir MH, Abdollahi A, Ghahramani N, Kumar M, Paydar S, Razani B, Mwangi J, Asadi-Pooya AA, Malekmakan L, Bastani B. Long COVID, a comprehensive systematic scoping review. Infection. 2021;49:1163-1186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 212] [Article Influence: 53.0] [Reference Citation Analysis (1)] |
| 4. | Michelen M, Manoharan L, Elkheir N, Cheng V, Dagens A, Hastie C, O'Hara M, Suett J, Dahmash D, Bugaeva P, Rigby I, Munblit D, Harriss E, Burls A, Foote C, Scott J, Carson G, Olliaro P, Sigfrid L, Stavropoulou C. Characterising long COVID: a living systematic review. BMJ Glob Health. 2021;6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 392] [Cited by in RCA: 564] [Article Influence: 141.0] [Reference Citation Analysis (0)] |
| 5. | Iqbal FM, Lam K, Sounderajah V, Clarke JM, Ashrafian H, Darzi A. Characteristics and predictors of acute and chronic post-COVID syndrome: A systematic review and meta-analysis. EClinicalMedicine. 2021;36:100899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 208] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 6. | Vollbracht C, Kraft K. Feasibility of Vitamin C in the Treatment of Post Viral Fatigue with Focus on Long COVID, Based on a Systematic Review of IV Vitamin C on Fatigue. Nutrients. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 52] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 7. | Jennings G, Monaghan A, Xue F, Mockler D, Romero-Ortuño R. A Systematic Review of Persistent Symptoms and Residual Abnormal Functioning following Acute COVID-19: Ongoing Symptomatic Phase vs. Post-COVID-19 Syndrome. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 80] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 8. | Fernández-de-Las-Peñas C, Palacios-Ceña D, Gómez-Mayordomo V, Florencio LL, Cuadrado ML, Plaza-Manzano G, Navarro-Santana M. Prevalence of post-COVID-19 symptoms in hospitalized and non-hospitalized COVID-19 survivors: A systematic review and meta-analysis. Eur J Intern Med. 2021;92:55-70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 346] [Cited by in RCA: 348] [Article Influence: 87.0] [Reference Citation Analysis (0)] |
| 9. | Malik P, Patel K, Pinto C, Jaiswal R, Tirupathi R, Pillai S, Patel U. Post-acute COVID-19 syndrome (PCS) and health-related quality of life (HRQoL)-A systematic review and meta-analysis. J Med Virol. 2022;94:253-262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 366] [Article Influence: 91.5] [Reference Citation Analysis (0)] |
| 10. | Ceban F, Ling S, Lui LMW, Lee Y, Gill H, Teopiz KM, Rodrigues NB, Subramaniapillai M, Di Vincenzo JD, Cao B, Lin K, Mansur RB, Ho RC, Rosenblat JD, Miskowiak KW, Vinberg M, Maletic V, McIntyre RS. Fatigue and cognitive impairment in Post-COVID-19 Syndrome: A systematic review and meta-analysis. Brain Behav Immun. 2022;101:93-135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 841] [Cited by in RCA: 885] [Article Influence: 295.0] [Reference Citation Analysis (0)] |
| 11. | Chen C, Haupert SR, Zimmermann L, Shi X, Fritsche LG, Mukherjee B. Global Prevalence of Post COVID-19 Condition or Long COVID: A Meta-Analysis and Systematic Review. J Infect Dis. 2022;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 789] [Article Influence: 263.0] [Reference Citation Analysis (0)] |
| 12. | van Kessel SAM, Olde Hartman TC, Lucassen PLBJ, van Jaarsveld CHM. Post-acute and long-COVID-19 symptoms in patients with mild diseases: a systematic review. Fam Pract. 2022;39:159-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 210] [Article Influence: 70.0] [Reference Citation Analysis (0)] |
| 13. | Alkodaymi MS, Omrani OA, Fawzy NA, Shaar BA, Almamlouk R, Riaz M, Obeidat M, Obeidat Y, Gerberi D, Taha RM, Kashour Z, Kashour T, Berbari EF, Alkattan K, Tleyjeh IM. Prevalence of post-acute COVID-19 syndrome symptoms at different follow-up periods: a systematic review and meta-analysis. Clin Microbiol Infect. 2022;28:657-666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 377] [Cited by in RCA: 315] [Article Influence: 105.0] [Reference Citation Analysis (0)] |
| 14. | Fernández-de-Las-Peñas C, Navarro-Santana M, Plaza-Manzano G, Palacios-Ceña D, Arendt-Nielsen L. Time course prevalence of post-COVID pain symptoms of musculoskeletal origin in patients who had survived severe acute respiratory syndrome coronavirus 2 infection: a systematic review and meta-analysis. Pain. 2022;163:1220-1231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 87] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 15. | Han Q, Zheng B, Daines L, Sheikh A. Long-Term Sequelae of COVID-19: A Systematic Review and Meta-Analysis of One-Year Follow-Up Studies on Post-COVID Symptoms. Pathogens. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 344] [Article Influence: 114.7] [Reference Citation Analysis (0)] |
| 16. | d'Ettorre G, Gentilini Cacciola E, Santinelli L, De Girolamo G, Spagnolello O, Russo A, Tarsitani L, Ciccozzi M, Mastroianni CM, d'Ettorre G, Ceccarelli G. Covid-19 sequelae in working age patients: A systematic review. J Med Virol. 2022;94:858-868. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 17. | Behnood SA, Shafran R, Bennett SD, Zhang AXD, O'Mahoney LL, Stephenson TJ, Ladhani SN, De Stavola BL, Viner RM, Swann OV. Persistent symptoms following SARS-CoV-2 infection amongst children and young people: A meta-analysis of controlled and uncontrolled studies. J Infect. 2022;84:158-170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 153] [Article Influence: 51.0] [Reference Citation Analysis (0)] |
| 18. | Nguyen NN, Hoang VT, Dao TL, Dudouet P, Eldin C, Gautret P. Clinical patterns of somatic symptoms in patients suffering from post-acute long COVID: a systematic review. Eur J Clin Microbiol Infect Dis. 2022;41:515-545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 59] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 19. | Lopez-Leon S, Wegman-Ostrosky T, Ayuzo Del Valle NC, Perelman C, Sepulveda R, Rebolledo PA, Cuapio A, Villapol S. Long-COVID in children and adolescents: a systematic review and meta-analyses. Sci Rep. 2022;12:9950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 296] [Article Influence: 98.7] [Reference Citation Analysis (0)] |
| 20. | Abdel-Gawad M, Zaghloul MS, Abd-Elsalam S, Hashem M, Lashen SA, Mahros AM, Mohammed AQ, Hassan AM, Bekhit AN, Mohammed W, Alboraie M. Post-COVID-19 Syndrome Clinical Manifestations: A Systematic Review. Antiinflamm Antiallergy Agents Med Chem. 2022;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 21. | Almas T, Malik J, Alsubai AK, Jawad Zaidi SM, Iqbal R, Khan K, Ali M, Ishaq U, Alsufyani M, Hadeed S, Alsufyani R, Ahmed R, Thakur T, Huang H, Antony M, Antony I, Bhullar A, Kotait F, Al-Ani L. Post-acute COVID-19 syndrome and its prolonged effects: An updated systematic review. Ann Med Surg (Lond). 2022;80:103995. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 22. | Maglietta G, Diodati F, Puntoni M, Lazzarelli S, Marcomini B, Patrizi L, Caminiti C. Prognostic Factors for Post-COVID-19 Syndrome: A Systematic Review and Meta-Analysis. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 111] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 23. | Healey Q, Sheikh A, Daines L, Vasileiou E. Symptoms and signs of long COVID: A rapid review and meta-analysis. J Glob Health. 2022;12:05014. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 24. | de Oliveira Almeida K, Nogueira Alves IG, de Queiroz RS, de Castro MR, Gomes VA, Santos Fontoura FC, Brites C, Neto MG. A systematic review on physical function, activities of daily living and health-related quality of life in COVID-19 survivors. Chronic Illn. 2022;17423953221089309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 61] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 25. | Fugazzaro S, Contri A, Esseroukh O, Kaleci S, Croci S, Massari M, Facciolongo NC, Besutti G, Iori M, Salvarani C, Costi S; Reggio Emilia COVID-19 Working Group. Rehabilitation Interventions for Post-Acute COVID-19 Syndrome: A Systematic Review. Int J Environ Res Public Health. 2022;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 121] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 26. | Venkatesan P. NICE guideline on long COVID. Lancet Respir Med. 2021;9:129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 291] [Cited by in RCA: 320] [Article Influence: 80.0] [Reference Citation Analysis (0)] |
| 27. | A clinical case definition of post COVID-19 condition by a Delphi consensus (6 October 2021). [cited 6 July 2022]. Available from: https://reliefweb.int/report/world/clinical-case-definition-post-covid-19-condition-delphi-consensus-6-october-2021. |
| 28. | Nabavi N. Long covid: How to define it and how to manage it. BMJ. 2020;370:m3489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 186] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 29. | Yan Z, Yang M, Lai CL. Long COVID-19 Syndrome: A Comprehensive Review of Its Effect on Various Organ Systems and Recommendation on Rehabilitation Plans. Biomedicines. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 105] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 30. | Soriano JB, Murthy S, Marshall JC, Relan P, Diaz JV; WHO Clinical Case Definition Working Group on Post-COVID-19 Condition. A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect Dis. 2022;22:e102-e107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 1457] [Article Influence: 485.7] [Reference Citation Analysis (0)] |
| 31. | Wang MY, Zhao R, Gao LJ, Gao XF, Wang DP, Cao JM. SARS-CoV-2: Structure, Biology, and Structure-Based Therapeutics Development. Front Cell Infect Microbiol. 2020;10:587269. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 474] [Cited by in RCA: 526] [Article Influence: 105.2] [Reference Citation Analysis (0)] |
| 32. | Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271-280.e8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11946] [Cited by in RCA: 14271] [Article Influence: 2854.2] [Reference Citation Analysis (0)] |
| 33. | Disser NP, De Micheli AJ, Schonk MM, Konnaris MA, Piacentini AN, Edon DL, Toresdahl BG, Rodeo SA, Casey EK, Mendias CL. Musculoskeletal Consequences of COVID-19. J Bone Joint Surg Am. 2020;102:1197-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 257] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 34. | Hasan LK, Deadwiler B, Haratian A, Bolia IK, Weber AE, Petrigliano FA. Effects of COVID-19 on the Musculoskeletal System: Clinician's Guide. Orthop Res Rev. 2021;13:141-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 35. | Tenforde MW, Kim SS, Lindsell CJ, Billig Rose E, Shapiro NI, Files DC, Gibbs KW, Erickson HL, Steingrub JS, Smithline HA, Gong MN, Aboodi MS, Exline MC, Henning DJ, Wilson JG, Khan A, Qadir N, Brown SM, Peltan ID, Rice TW, Hager DN, Ginde AA, Stubblefield WB, Patel MM, Self WH, Feldstein LR; IVY Network Investigators; CDC COVID-19 Response Team; IVY Network Investigators. Symptom Duration and Risk Factors for Delayed Return to Usual Health Among Outpatients with COVID-19 in a Multistate Health Care Systems Network - United States, March-June 2020. MMWR Morb Mortal Wkly Rep. 2020;69:993-998. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 624] [Cited by in RCA: 853] [Article Influence: 170.6] [Reference Citation Analysis (0)] |
| 36. | Mayer KP, Thompson Bastin ML, Montgomery-Yates AA, Pastva AM, Dupont-Versteegden EE, Parry SM, Morris PE. Acute skeletal muscle wasting and dysfunction predict physical disability at hospital discharge in patients with critical illness. Crit Care. 2020;24:637. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 114] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 37. | Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA. 2020;324:603-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2398] [Cited by in RCA: 2810] [Article Influence: 562.0] [Reference Citation Analysis (0)] |
| 38. | Paneroni M, Simonelli C, Saleri M, Bertacchini L, Venturelli M, Troosters T, Ambrosino N, Vitacca M. Muscle Strength and Physical Performance in Patients Without Previous Disabilities Recovering From COVID-19 Pneumonia. Am J Phys Med Rehabil. 2021;100:105-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 140] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 39. | Jacobs LG, Gourna Paleoudis E, Lesky-Di Bari D, Nyirenda T, Friedman T, Gupta A, Rasouli L, Zetkulic M, Balani B, Ogedegbe C, Bawa H, Berrol L, Qureshi N, Aschner JL. Persistence of symptoms and quality of life at 35 days after hospitalization for COVID-19 infection. PLoS One. 2020;15:e0243882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 206] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 40. | Prevalence of ongoing symptoms following coronavirus (COVID-19) infection in the UK - Office for National Statistics [Internet]. [cited 14 July 2022]. Available from: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/bulletins/prevalenceofongoingsymptomsfollowingcoronaviruscovid19infectionintheuk/7july2022. |
| 41. | Lau HM, Lee EW, Wong CN, Ng GY, Jones AY, Hui DS. The impact of severe acute respiratory syndrome on the physical profile and quality of life. Arch Phys Med Rehabil. 2005;86:1134-1140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 65] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 42. | Aiyegbusi OL, Hughes SE, Turner G, Rivera SC, McMullan C, Chandan JS, Haroon S, Price G, Davies EH, Nirantharakumar K, Sapey E, Calvert MJ; TLC Study Group. Symptoms, complications and management of long COVID: a review. J R Soc Med. 2021;114:428-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 530] [Article Influence: 132.5] [Reference Citation Analysis (0)] |
| 43. | Varghese J, Sandmann S, Ochs K, Schrempf IM, Frömmel C, Dugas M, Schmidt HH, Vollenberg R, Tepasse PR. Persistent symptoms and lab abnormalities in patients who recovered from COVID-19. Sci Rep. 2021;11:12775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 44. | Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, Kang L, Guo L, Liu M, Zhou X, Luo J, Huang Z, Tu S, Zhao Y, Chen L, Xu D, Li Y, Li C, Peng L, Xie W, Cui D, Shang L, Fan G, Xu J, Wang G, Zhong J, Wang C, Wang J, Zhang D, Cao B. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397:220-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3187] [Cited by in RCA: 2889] [Article Influence: 722.3] [Reference Citation Analysis (1)] |
| 45. | Miyazato Y, Morioka S, Tsuzuki S, Akashi M, Osanai Y, Tanaka K, Terada M, Suzuki M, Kutsuna S, Saito S, Hayakawa K, Ohmagari N. Prolonged and Late-Onset Symptoms of Coronavirus Disease 2019. Open Forum Infect Dis. 2020;7:ofaa507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 96] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 46. | Daher A, Balfanz P, Cornelissen C, Müller A, Bergs I, Marx N, Müller-Wieland D, Hartmann B, Dreher M, Müller T. Follow up of patients with severe coronavirus disease 2019 (COVID-19): Pulmonary and extrapulmonary disease sequelae. Respir Med. 2020;174:106197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 217] [Article Influence: 43.4] [Reference Citation Analysis (0)] |
| 47. | Davis HE, Assaf GS, McCorkell L, Wei H, Low RJ, Re'em Y, Redfield S, Austin JP, Akrami A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine. 2021;38:101019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1118] [Cited by in RCA: 1572] [Article Influence: 393.0] [Reference Citation Analysis (0)] |
| 48. | Yelin D, Margalit I, Nehme M, Bordas-Martínez J, Pistelli F, Yahav D, Guessous I, Durà-Miralles X, Carrozzi L, Shapira-Lichter I, Vetter P, Peleato-Catalan D, Tiseo G, Wirtheim E, Kaiser L, Gudiol C, Falcone M, Leibovici L; On Behalf Of The LongCOV Research Group. Patterns of Long COVID Symptoms: A Multi-Center Cross Sectional Study. J Clin Med. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 49. | Dos Santos PK, Sigoli E, Bragança LJG, Cornachione AS. The Musculoskeletal Involvement After Mild to Moderate COVID-19 Infection. Front Physiol. 2022;13:813924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 50. | Humphreys H, Kilby L, Kudiersky N, Copeland R. Long COVID and the role of physical activity: a qualitative study. BMJ Open. 2021;11:e047632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 108] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 51. | Cardemil CV, Balachandran N, Kambhampati A, Grytdal S, Dahl RM, Rodriguez-Barradas MC, Vargas B, Beenhouwer DO, Evangelista KV, Marconi VC, Meagley KL, Brown ST, Perea A, Lucero-Obusan C, Holodniy M, Browne H, Gautam R, Bowen MD, Vinjé J, Parashar UD, Hall AJ. Incidence, Etiology, and Severity of Acute Gastroenteritis Among Prospectively Enrolled Patients in 4 Veterans Affairs Hospitals and Outpatient Centers, 2016-2018. Clin Infect Dis. 2021;73:e2729-e2738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 52. | Hoong CWS, Amin MNME, Tan TC, Lee JE. Viral arthralgia a new manifestation of COVID-19 infection? Int J Infect Dis. 2021;104:363-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 53. | Elhiny R, Al-Jumaili AA, Yawuz MJ. What might COVID-19 Patients Experience after Recovery? A Systematic Review. 2021 Preprint. Available from: PPR: PPR358497. [DOI] [Full Text] |
| 54. | Petersen MS, Kristiansen MF, Hanusson KD, Danielsen ME, Á Steig B, Gaini S, Strøm M, Weihe P. Long COVID in the Faroe Islands: A Longitudinal Study Among Nonhospitalized Patients. Clin Infect Dis. 2021;73:e4058-e4063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 237] [Article Influence: 47.4] [Reference Citation Analysis (0)] |
| 55. | Carvalho-Schneider C, Laurent E, Lemaignen A, Beaufils E, Bourbao-Tournois C, Laribi S, Flament T, Ferreira-Maldent N, Bruyère F, Stefic K, Gaudy-Graffin C, Grammatico-Guillon L, Bernard L. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin Microbiol Infect. 2021;27:258-263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 476] [Article Influence: 95.2] [Reference Citation Analysis (0)] |
| 56. | Chopra N, Chowdhury M, Singh AK, Ma K, Kumar A, Ranjan P, Desai D, Wig N. Clinical predictors of long COVID-19 and phenotypes of mild COVID-19 at a tertiary care centre in India. Drug Discov Ther. 2021;15:156-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 57. | Stavem K, Ghanima W, Olsen MK, Gilboe HM, Einvik G. Persistent symptoms 1.5-6 months after COVID-19 in non-hospitalised subjects: a population-based cohort study. Thorax. 2021;76:405-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 118] [Cited by in RCA: 198] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 58. | Ghosn J, Piroth L, Epaulard O, Le Turnier P, Mentré F, Bachelet D, Laouénan C; French COVID cohort study and investigators groups. Persistent COVID-19 symptoms are highly prevalent 6 months after hospitalization: results from a large prospective cohort. Clin Microbiol Infect. 2021;27:1041.e1-1041.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 83] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 59. | Ono K, Kishimoto M, Shimasaki T, Uchida H, Kurai D, Deshpande GA, Komagata Y, Kaname S. Reactive arthritis after COVID-19 infection. RMD Open. 2020;6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 60. | Sapkota HR, Nune A. Long COVID from rheumatology perspective - a narrative review. Clin Rheumatol. 2022;41:337-348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 61. | Derksen VFAM, Kissel T, Lamers-Karnebeek FBG, van der Bijl AE, Venhuizen AC, Huizinga TWJ, Toes REM, Roukens AHE, van der Woude D. Onset of rheumatoid arthritis after COVID-19: coincidence or connected? Ann Rheum Dis. 2021;80:1096-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 62. | Berktaş BM, Gökçek A, Hoca NT, Koyuncu A. COVID-19 illness and treatment decrease bone mineral density of surviving hospitalized patients. Eur Rev Med Pharmacol Sci. 2022;26:3046-3056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 63. | Kottlors J, Große Hokamp N, Fervers P, Bremm J, Fichter F, Persigehl T, Safarov O, Maintz D, Tritt S, Abdullayev N. Early extrapulmonary prognostic features in chest computed tomography in COVID-19 pneumonia: Bone mineral density is a relevant predictor for the clinical outcome - A multicenter feasibility study. Bone. 2021;144:115790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 64. | Kerkman JN, Daffertshofer A, Gollo LL, Breakspear M, Boonstra TW. Network structure of the human musculoskeletal system shapes neural interactions on multiple time scales. Sci Adv. 2018;4:eaat0497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 98] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 65. | Bahouth S, Chuang K, Olson L, Rosenthal D. COVID-19 related muscle denervation atrophy. Skeletal Radiol. 2021;50:1717-1721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 66. | Agergaard J, Leth S, Pedersen TH, Harbo T, Blicher JU, Karlsson P, Østergaard L, Andersen H, Tankisi H. Myopathic changes in patients with long-term fatigue after COVID-19. Clin Neurophysiol. 2021;132:1974-1981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 67. | Islam B, Ahmed M, Islam Z, Begum SM. Severe acute myopathy following SARS-CoV-2 infection: a case report and review of recent literature. Skelet Muscle. 2021;11:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 68. | Bonorino KC, Cani KC. Early mobilization in the time of COVID-19. Rev Bras Ter Intensiva. 2020;32:484-486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 69. | Wade DT. Rehabilitation after COVID-19: an evidence-based approach. Clin Med (Lond). 2020;20:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 103] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 70. | Swarnakar R, Yadav SL. Rehabilitation in long COVID-19: A mini-review. World J Methodol. 2022;12:235-245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (3)] |