Published online Nov 20, 2019. doi: 10.5500/wjt.v9.i7.145
Peer-review started: March 26, 2019
First decision: March 26, 2019
Revised: October 16, 2019
Accepted: November 6, 2019
Article in press: November 6, 2019
Published online: November 20, 2019
Processing time: 238 Days and 13.5 Hours
The Karnofsky Performance Status (KPS) scale has been widely validated for clinical practice for over 60 years.
To examine the extent to which poor pre-transplant functional status, assessed using the KPS scale, is associated with increased risk of mortality and/or graft failure at 1-year post-transplantation.
This study included 38278 United States adults who underwent first, non-urgent, liver-only transplantation from 2005 to 2014 (Scientific Registry of Transplant Recipients). Functional impairment/disability was categorized as severe, moderate, or none/normal. Analyses were conducted using multivariable-adjusted Cox survival regression models.
The median age was 56 years, 31% were women, median pre-transplant Model for End-Stage for Liver Disease score was 18. Functional impairment was present in 70%; one-quarter of the sample was severely disabled. After controlling for key recipient and donor factors, moderately and severely disabled patients had a 1-year mortality rate of 1.32 [confidence interval (CI): 1.21-1.44] and 1.73 (95%CI: 1.56-1.91) compared to patients with no impairment, respectively. Subjects with moderate and severe disability also had a multivariable-adjusted 1-year graft failure rate of 1.13 (CI: 1.02-1.24) and 1.16 (CI: 1.02-1.31), respectively.
Pre-transplant functional status is a useful prognostic indicator for 1-year post-transplant patient and graft survival.
Core tip: Poor functional status, as defined by The Karnofsky Performance Status scale, is a strong predictor of worse 1-year post-transplant outcomes (patient and graft survival) in a national United States liver transplant population.
- Citation: Dolgin NH, Movahedi B, Anderson FA, Brüggenwirth IM, Martins PN, Bozorgzadeh A. Impact of recipient functional status on 1-year liver transplant outcomes. World J Transplant 2019; 9(7): 145-157
- URL: https://www.wjgnet.com/2220-3230/full/v9/i7/145.htm
- DOI: https://dx.doi.org/10.5500/wjt.v9.i7.145
Due to increasing organ shortages in the United States, patients on the liver transplant waiting list are older and sicker than ever before, while wait time continues to climb[1-3]. Among the 15000 patients with End-Stage Liver Disease on the transplant waiting list in 2013, 20% were over 65 years old, 20% had been waiting for at least 5 years already, 20% died while awaiting transplant; fewer than 6000 patients received an organ[2]. In 2002, in response to increasing shortages and waitlist mortality, the liver allocation system was reorganized to prioritize patients according to urgency[4]. “Urgency” was defined according to risk of 3-month mortality, calculated using 3 objective laboratory values (creatinine, bilirubin, international normalized ratio) to create individualized Model for End-Stage for Liver Disease (MELD) scores used to rank patients. Although this system successfully lowered population-level waitlist mortality rates, it is an insufficient summary measure for describing global health status[5,6], and has recently been shown to underestimate mortality risk among subgroups of “frail” patients[6-8].
Frailty is increasingly recognized as an important predictor of outcomes after major surgical procedures including liver transplantation[9-11]. Frailty syndrome is defined by a cluster of signs and symptoms that are hallmark sequelae of liver disease, malnutrition, sarcopenia, functional impairment/disability, which ultimately lead to increased vulnerability to stressors due to depleted physiologic reserve[12]. However, there is no gold standard measure of frailty[13].
Liver and lung transplant centers in the United States are mandated to submit Karnofsky Performance Status (KPS) functional status data on all patients, with other clinical data, to the Organ Procurement and Transplantation Network (OPTN) each quarter. This frailty measure has been widely used in clinical practice and research for over 60 years and has been extensively validated across a wide range of disease groups including transplant, liver disease, and End-Stage Renal Disease populations[14-27]. However, though several studies have used KPS as a predictor of liver transplant outcomes, the majority of studies were limited in generalizability as they were either single-center studies[28,29], conducted outside of the United States[11,30], limited to the early post-transplant period[11,30,31], and/or took place before MELD implementation, at which point the transplant recipient population shifted dramatically[28,32]. In 2013, the Liver and Intestinal Transplant Committee of OPTN publicly asked for researchers to fill this gap in the literature regarding the utility of the KPS scale in a national liver transplant population[18].
To our knowledge, this will be the first study to evaluate a standardized, validated measure of functional status as a predictor of 1-year post-transplant outcomes in a national United States liver transplant population. Using data from the only comprehensive nationwide transplant database, the United Network for Organ Sharing (UNOS) Scientific Registry of Transplant Recipients (SRTR), we assessed the clinical utility of the KPS scale for the prediction of 1-year post-liver transplant patient and graft survival.
This retrospective cohort study used data from the SRTR. The SRTR is contracted to UNOS by the United States Department of Health and Human Services to manage data collected via government-mandated reporting by all United States transplant centers. This study was deemed exempt by the University of Massachusetts Medical School Institutional Review Board.
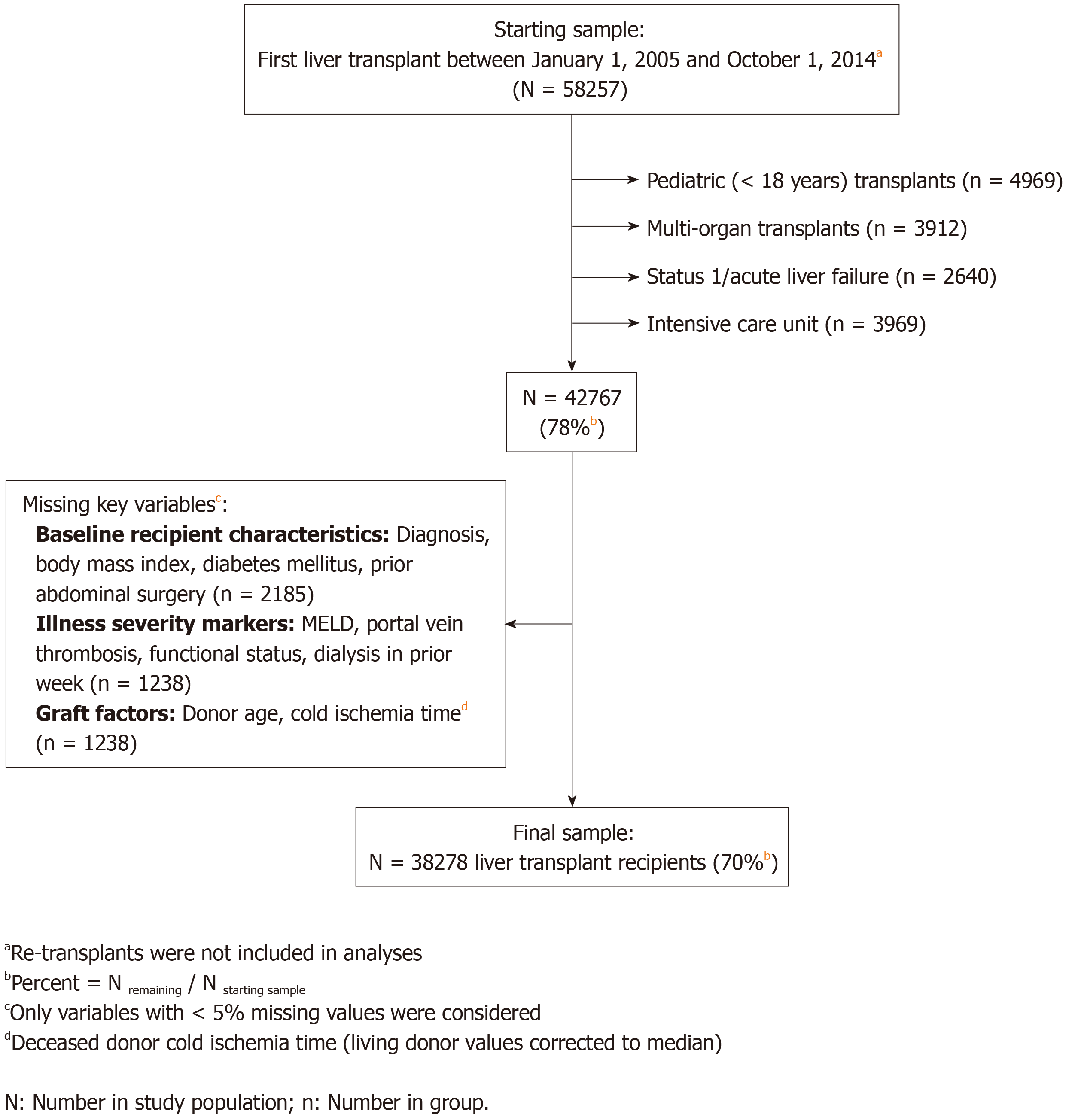
The study population included patients that underwent a first liver transplant between January 1, 2005 and October 1, 2014 (Figure 1). Exclusion criteria consisted of the following: (1) Pediatric transplant (< 18 years); (2) Multi-organ transplant; (3) UNOS Status 1 or acute liver failure; (4) ICU pre-transplant; or (5) Subjects with missing data in any of the key variables of interest (variables with ≥ 5% missing values were not used in this study). Pediatric and multi-organ transplants were excluded because the organ allocation systems for these patients are separate, and represent a distinct set of indications and disease courses, than the general United States liver transplant population. In addition, the risk of complications and graft loss are higher in these groups. We excluded urgent (Status 1 or acute hepatic necrosis) and ICU-admitted patients. This was done because these are often patients who rapidly decline due to an inciting event (e.g., infection) and may, therefore, be categorized as being of poor functional status due to the event as opposed to being “frail”, which is conceptualized as a chronic process leading to depletion of physiologic reserve.
Exposure variable: The primary exposure of interest was provider assessment of preoperative (“pre-transplant”) functional status using the KPS scale (Table 1). The KPS defines functional status on an 11-point scale from 100% (normal, no complaints, no evidence of disease) to 0% (dead) in 10% increments, with 3 corresponding tiers. We used the 3-tiered version of the scale based on higher inter-rater reliability scores[33]. We assigned labels to the categories with respect to level of functional impairment/disability as follows: Subjects with minimal or no symptoms of disease (80%-100%) were labeled “(A) None/Normal [function]”; subjects needing varying levels of assistance in daily activities (50%-70%) were labeled “(B) Moderate [impairment in function]”; and subjects who were disabled and/or hospitalization indicated and/or moribund (10%-40%) were labeled “(C) Severe [functional impairment/disability]”.
| Conditiona | % | Rating criteria |
| A (“None/Normal”) Able to carry on normal activity and to work; no special care needed | 100 | Normal, no complaints; no evidence of disease |
| 90 | Able to carry on normal activity; minor signs or symptoms of disease | |
| 80 | Normal activity with effort; some signs or symptoms of disease | |
| B (“Moderate”) Unable to work; able to live at home and care for most personal needs; varying amount of assistance needed | 70 | Cares for self; unable to carry on normal activity or to do active work |
| 60 | Requires occasional assistance, but is able to care for most of his personal needs | |
| 50 | Requires considerable assistance and frequent medical care | |
| C (“Severe”) Unable to care for self; requires equivalent of institutional or hospital care; disease may be progressing rapidly | 40 | Disabled; requires special care and assistance |
| 30 | Severely disabled; hospital admission is indicated although death not imminent | |
| 20 | Very sick; hospital admission necessary; active supportive treatment necessary | |
| 10 | Moribund; fatal processes progressing rapidly | |
| 0 | Dead |
Study end points: The primary outcome of interest was 1-year all-cause mortality (Social Security Death Master File/Organ Procurement and Transplant Network data). As mortality after transplantation is highest in the early postoperative period and likely related to operative risks and complications that may become less relevant for long-term outcomes, the importance of functional status may change with time and context[34]. Therefore, we also examined death rates during the 1-month postoperative period (day 0-30) as compared to residual risk during the remaining 11 months of the year (day 31-365). The secondary outcome of interest was 1-year graft failure.
Lastly, among patients who did not experience either adverse outcome (death or graft failure), we describe the proportion that was able to return to “Normal” functional status during the first year post-transplant. Transplant centers must report follow-up data on transplant recipients at 6-month post-transplant, 1-year, and annually thereafter; follow-up records from day 0 to 395 (365 + 30 d) with functional status data available (< 5% of recipients were missing follow-up functional status) were analyzed (but counted once per patient).
Potential confounding variables: Potential confounders were identified from a priori clinical knowledge, literature review, and variables included in the SRTR risk-adjustment models, available at srtr.org. Potential confounders included recipient sociodemographic and medical/surgical history factors (i.e., information known at least 2 weeks before transplant, e.g., primary liver diagnosis; Table 2), pre-transplant illness severity markers (e.g., last-calculated laboratory MELD; Table 3), and all Donor Risk Index factors (e.g., cause of death; Table 4)[34,35]. Every variable evaluated as a potential confounder was categorized and is described in Tables 2-4 (exceptions: Baseline functional status, time on the waitlist, and MELD component labs are listed for descriptive purposes only).
| Characteristicb | Functional impairment/disability | ||
| A: None (n = 11674) | B: Moderate (n = 17530) | C: Severe (n = 9074) | |
| Sociodemographics | |||
| Age in years | |||
| 18-44 | 11.6 | 10.3 | 12.7 |
| 45-54 | 28.9 | 30.2 | 32.1 |
| 55-64 | 44.9 | 45.3 | 43.2 |
| ≥ 65 | 14.5 | 14.3 | 12.0 |
| Women | 26.8 | 32.1 | 33.4 |
| Race/ethnicity | |||
| White | 73.1 | 74.3 | 70.4 |
| Hispanic/Latino | 10.6 | 12.5 | 15.9 |
| Black | 8.9 | 8.4 | 9.1 |
| Health insurance | |||
| Private | 68.1 | 55.3 | 54.2 |
| Medicare | 18.5 | 26.5 | 24.6 |
| Medicaid | 8.8 | 14.0 | 17.0 |
| Medical/Surgical History | |||
| Functional impairment at registration | |||
| None | 74.6 | 33.4 | 22.4 |
| Moderate | 19.9 | 57.0 | 35.4 |
| Severe | 2.5 | 6.6 | 37.0 |
| Primary cause of liver disease | |||
| Non-Cholestatic | 56.2 | 64.7 | 75.8 |
| Cholestatic | 9.5 | 8.1 | 7.8 |
| Malignancy | 30.1 | 23.0 | 12.5 |
| Hepatitis C | 44.4 | 45.5 | 42.1 |
| Diabetesc | 23.2 | 25.8 | 25.3 |
| Previous Abdominal Surgery | 46.2 | 51.6 | 50.3 |
| Characteristica | Functional impairment/disability | ||
| A: None (n = 11674) | B: Moderate (n = 17530) | C: Severe (n = 9074) | |
| Waitlist time, mo | 4.0 (1.3-10.2) | 3.7 (1.1-10.3) | 2.1 (0.4-8.4) |
| Weight loss ≥ 5%b | 19.7 | 24.2 | 27.0 |
| BMI, kg/m2 | |||
| Underweight (< 18.5) | 1.6 | 1.6 | 2.4 |
| Normal (18.5-25) | 28.3 | 27.5 | 27.1 |
| Overweight (25-30) | 37.0 | 36.0 | 34.0 |
| Obese (≥ 30) | 33.2 | 35.0 | 36.5 |
| MELD | |||
| < 15 | 49.9 | 34.9 | 14.3 |
| 15-29 | 45.9 | 56.5 | 45.9 |
| ≥ 30 | 4.2 | 8.6 | 39.8 |
| Total bilirubin | 2.3 (1.2-4.4) | 3.0 (1.6-6.1) | 6.6 (2.9-17.2) |
| International normalized ratio | 1.4 (1.2-1.7) | 1.5 (1.3-1.9) | 2.0 (1.5-2.6) |
| Serum creatinine | 0.9 (0.7-1.2) | 1.0 (0.8-1.3) | 1.4 (0.9-2.3) |
| Serum sodium | 137 (134-140) | 136 (133-139) | 135 (132-139) |
| Child Pugh score | |||
| A (Good) | 23.8 | 11.8 | 4.1 |
| B (Fair) | 38.7 | 33.8 | 18.5 |
| C (Poor) | 37.5 | 54.4 | 77.4 |
| Ascitesc | |||
| None | 36.9 | 23.0 | 14.0 |
| Mild/Moderate | 48.4 | 52.4 | 43.5 |
| Severe | 14.8 | 24.6 | 42.6 |
| Encephalopathyd | |||
| None | 52.7 | 36.6 | 26.0 |
| Grade 1-2 | 44.4 | 57.5 | 61.4 |
| Grade 3-4 | 2.9 | 5.9 | 12.6 |
| Albumin | |||
| > 3.5 | 26.7 | 19.3 | 22.8 |
| 2.8-3.5 | 41.7 | 41.2 | 39.0 |
| < 2.8 | 31.6 | 39.6 | 38.2 |
| Dialysise | 0.9 | 1.6 | 10.9 |
| Portal Vein Thrombosis | 6.5 | 9.7 | 12.4 |
| Characteristica | Functional impairment/disability | P value | ||
| A: None (n = 11674) | B: Moderate (n = 17530) | C: Severe (n = 9074) | ||
| Transplant type: Living donor | 5.5 | 5.0 | 2.3 | < 0.001 |
| Donor Risk Indexb | 1.6 (1.3-1.9) | 1.6 (1.3-1.9) | 1.5 (1.2-1.9) | < 0.001 |
| Age in years | ||||
| 18-39 | 37.3 | 37.1 | 39.9 | |
| 40-49 | 19.7 | 19.7 | 19.7 | |
| 50-59 | 19.8 | 20.4 | 20.7 | |
| 60-69 | 11.7 | 12.7 | 10.8 | |
| ≥ 70 | 5.5 | 4.9 | 3.9 | < 0.001 |
| Women | 40.5 | 41.4 | 40.9 | 0.35 |
| Race/ethnicity | ||||
| White | 68.2 | 68.1 | 65.6 | |
| Black | 17.2 | 17.9 | 17.5 | |
| Other | 14.6 | 14.0 | 16.9 | < 0.001 |
| BMI, kg/m2 | ||||
| Underweight (< 18.5) | 2.9 | 2.9 | 2.6 | |
| Normal (18.5-25) | 36.5 | 35.9 | 36.2 | |
| Overweight (25-30) | 33.5 | 34.2 | 33.4 | |
| Obese (≥ 30) | 27.1 | 27.0 | 27.9 | 0.43 |
| Cause of death | ||||
| Trauma | 34.3 | 34.1 | 34.1 | |
| Anoxia | 21.5 | 22.5 | 24 | |
| Cardiovascular accident | 41.4 | 40.9 | 39.4 | < 0.01 |
| Donation after cardiac death | 12.6 | 13.2 | 12.1 | 0.04 |
| Split/Partial liver | 1.5 | 1.4 | 1.3 | 0.38 |
| Allocation type | ||||
| Regional | 17.8 | 19.9 | 23.3 | |
| National | 6.6 | 5.0 | 3.8 | < 0.001 |
| Cold ischemia time ≥ 8 h | 30.9 | 28.9 | 27.9 | < 0.001 |
We explored bivariate relationships between the primary exposure (functional status) and potential confounders of interest using contingency table analyses [chi-squared tests for categorical variables, ANOVA for continuous variables, and Spearman’s rho (rs) for ordered-variable correlations, using expanded KPS, continuous MELD]. Relationships between variables and post-transplant time were explored graphically. One-year cumulative failure rates were estimated using the Kaplan Meier method.
To quantify the extent to which impaired functional status was associated with increased risk of 1-year all-cause mortality and 1-year graft failure, we developed separate Cox survival regression models for each outcome. We applied a manual forward approach, sequentially adding conceptually meaningful groups of variables to the model. With the exception of recipient age, sex, race/ethnicity, and MELD, variables (and variable interactions) with p-values of > 0.05 were excluded from the final model. Goodness-of-fit and proportionality of hazards were tested using the omnibus Gronnesby and Borgan test and martingale residuals and confirmed non-significant (no evidence of poor fit) for all models reported[36].
Results are reported as hazard ratios (HR) with 95% confidence interval (CI); P-values ≤ 0.05 were considered statistically significant. All analyses were conducted using Stata version 13 (StataCorp LP, College Station, TX, United States).
The final study sample included 38278 liver transplant recipients (Figure 1). The median [interquartile range (IQR)] age was 56 (51-61) years and MELD was 18 (12-25). Women represented 31% of the sample and the largest ethnic minority was Hispanic/Latino (12.7%). Median follow-up time was 3.3 (1.5-6.0) years after transplant.
At pre-transplant assessment, approximately 70% of the sample had some degree of functional impairment or disability. Approximately one-quarter (23.7%) had “Severe” functional impairment/disability (≤ 40% function), 45.8% were “Moderately” impaired, and the remaining 30.5% had no functional impairments (≥ 80% function). The median (IQR) pre-transplant functional performance status score was 60% (50%-80%) and the mean (standard deviation) was 61% (21%).
Table 2 describes baseline characteristics of the sample by category of pre-transplant functional status. Subjects who were of worse functional status pre-transplant were more likely to be female, of Hispanic/Latino race/ethnicity, and/or have Medicaid insurance. Primary diagnosis of hepatic malignancy was associated with better physical function. Baseline and pre-transplant functional status were moderately correlated (rs = 0.42, P < 0.001). Sixty-percent of recipients maintained the same level of function over their waitlist course while 30% declined from a higher level of function at baseline.
Table 3 describes recipient clinical characteristics pre-transplant by category of pre-transplant functional status. Significant weight loss (≥ 5% of baseline weight) over the waitlist period was more common among transplant recipients who were impaired/disabled pre-transplant, and the weight loss occurred more rapidly. Poor functional status was moderately correlated with worse (higher) MELD scores (rs = -0.49; P < 0.001). However, only 64% of subjects with MELD scores ≥ 30 were “Severely” impaired/disabled, and less than half (44%) of patients with MELD scores < 15 were of “Normal” functional status. Cirrhosis severity according to Child-Pugh class was associated with severity of functional status. Around 10% of severely impaired subjects were on dialysis pre-transplant, compared to < 1% of Normal functional status subjects.
Table 4 describes donor characteristics by categories of pre-transplant functional status. Donor characteristics were mostly comparable across functional status categories. Only 8.3% of living donor liver transplant recipients was Severely impaired/disabled (n = 729). Functionally impaired patients were slightly less likely to receive higher risk organs (e.g., donor ≥ 70 years, nationally allocated or with prolonged cold ischemia time).
Death within one year was observed in 3595 (9.4%) transplant recipients. The mortality rate was directly related to functional status. Among patients that were severely impaired/disabled, 12.8% died compared with 9.3% of those with moderate functional limitations and 6.9% of those with normal functional status at the time of transplant.
Table 5 describes the results of unadjusted and adjusted Cox regression models for 1-year mortality. Subjects with severe or moderate functional impairment pre-transplant were at significantly increased risk of dying within one year post-transplant. After multivariable adjustment, severely and moderately impaired patients had 1-year mortality rates that were 1.73 (CI: 1.56-1.91) and 1.32 (CI: 1.21-1.44) times greater than the hazard for subjects without any functional impairment, respectively.
| Functional impairment/disability | Hazard Ratio (95%CI) | |||||
| 1-Yr | Day 0-30c | Day 31-365d | ||||
| Unadjusted | Adjustedb | Unadjusted | Adjustedb | Unadjusted | Adjustedb | |
| Severe | 1.94 (1.77-2.13) | 1.73 (1.56-1.91) | 2.40 (1.99-2.89) | 2.10 (1.71-2.59) | 1.82 (1.64-2.02) | 1.62 (1.45-1.82) |
| Moderate | 1.38 (1.27-1.51) | 1.32 (1.21-1.44) | 1.60 (1.34-1.92) | 1.53 (1.27-1.83) | 1.33 (1.20-1.46) | 1.26 (1.15-1.40) |
| None | Referent | Referent | Referent | |||
Mortality risks were greatest in the immediate postoperative period (day 0-30) when 881 (2.3%) deaths were observed in a single month. The adjusted 30-d mortality risk for Severely impaired/disabled patients was more than double (HR: 2.10; CI: 1.71-2.59) that of patients of “Normal” functional status, after adjusting for all variables controlled for in the full 1-year survival model (Table 5). Approximately three-quarters (n = 2714) of all one-year deaths occurred during the remaining 11 mo of the postoperative year (day 31-365); HRs were comparable to estimates for overall one-year mortality (< 10% relative difference).
Graft failure was observed in 2214 of the study population within one year of transplant. The estimated failure rate on day 365 was 6.2% (cumulative failure or death rate on day 365: 12.7%). Approximately half (53.8%) received a second transplant within the first post-transplant year, of which 75.9% (n = 905) survived the year; 98.6% (n = 1008) of those who did not undergo retransplantation within the first postoperative year did not survive to 1-year post-transplant.
Table 6 describes the results of unadjusted and adjusted Cox regression models for 1-year graft failure. Subjects with severe and moderate impairment/disability had multivariable-adjusted 1-year graft failure rates that were 1.16 (CI: 1.02-1.31) and 1.13 (CI: 1.02-1.24) times higher than patients with normal function, respectively.
Among the 33764 (88.2%) transplant recipients who experienced neither outcome (death or graft failure within a year), 95% (n = 32004) had at least 1 follow-up functional status assessment within a year. The majority (86.3%) recovered from transplant and reached “Normal” functional status within 1 year. Of the 7258 recipients in this subsample that were severely impaired/disabled pre-transplant, 81% (n = 5861) recovered full physical function (“Normal” functional status) within 1 year of transplant.
Almost 1 in 4 patients included in this national study of 38278 United States adults that underwent non-urgent liver transplantation between 2005 and 2014 had severe functional impairment/disability at the time of transplant. This group of patients was found to have a markedly increased hazard of dying and/or having graft failure at 1 year compared to Normal functional status patients. This increased hazard was observed in both unadjusted and multivariable-adjusted regression analyses controlling for a variety of potentially confounding factors of prognostic importance. Approximately 86% of recipients who did not experience 1-year death or graft failure (and had follow-up data available) recovered from transplant and reached “Normal” functional status within 1-year.
We present data from the first national study illustrating the role of pre-transplant functional status as a predictor of one-year survival among liver transplant recipients. Our results are in agreement with the findings from earlier studies that evaluated Poor functional status as a predictor of adverse transplant outcomes[11,28-30]. Two such studies, each with approximately 4000 United Kingdom recipients of a liver transplant, reported a near 2-fold increased risk of post-transplant mortality at 90 d for the worst functioning group relative to the highest functioning group[11,30]. Studies have also shown that objective measures of physical function, such as walking distance or speed and grip strength, are also strong predictors of adverse liver transplant outcomes regardless of recipient age, size, or cause/severity of liver disease[29].
Insight into a transplant patient’s global health status guides day-to-day clinical management, as well as transplant decisions, particularly in the face of contradictory laboratory or otherwise objective measures of pathological disease progression (e.g., MELD score). Capturing such insight through the use of a quantitative physical health scale may help transplant teams to strategize and communicate complex medical and surgical management decisions with patients, families, and the many other members of multidisciplinary transplant teams that provide longitudinal care for liver transplant patients.
Knowledge of a patient’s functional status before transplant may practically assist transplant teams to anticipate, communicate, and coordinate resources for postoperative critical care, rehabilitation after discharge, and potentially longer-term occupational therapy to help patients recover physical health and quality of life[37]. Many well-established risk factors for adverse outcomes among patients undergoing liver transplant may be unpredictable or sudden (spontaneous bacterial peritonitis), unavoidable (older age), and/or untreatable (portal vein thrombosis). Furthermore, many of the strongest predictors of adverse outcomes are present in a relatively small percentage of the liver transplant population. Many risk factors are unknown until very close to transplant time (e.g., life support, cold ischemia time), whereas functional impairment can present very early and progress insidiously in end-stage liver disease patients over the course of waiting for an organ. All patients can also be assigned a value for functional status at baseline, which can be used as a reference point to assess change over time. While this scale is an all-encompassing global physical function measure and a patient can fall anywhere on the continuous scale, many risk factors considered in transplant decisions are individual dichotomous variables, which are usually assessed in combination with other risk factors that can take time to accumulate. Thus, as functional status is a harbinger of adverse outcomes and may present early, it may be a useful clinical tracking tool that can be used for strategic care management.
Promising interventional studies have also shown that “prehabilitation”, physical therapy (e.g., strength training) and nutritional support, designed to improve functional status (or slow decline) in anticipation of a physiologic stressor such as surgery[38,39], is effective at improving postoperative recovery and outcomes after major abdominal surgery[40-44]. Although none of these studies focused on liver transplant patients, several included cohorts that similarly have a high likelihood of becoming frail due malnutrition, inflammation, and sarcopenia (e.g., cancer patients[41,42] and older populations[43,44]). Prehabilitation has the potential for providing clinicians with a way to not only recognize, but also slow or prevent decline to the point of “Severe” impairment/disability. However, more research on prehabilitation specific to a liver disease population is warranted. Frailty due to liver failure may not respond to the same interventions that have successfully slowed progression of frailty due to aging as there may be fundamental differences in etiology and pathogenesis between these populations that may limit their effectiveness[45].
Strengths of this study include its use of the SRTR with complete capture of every solid organ transplant in the United States since 1987, including waitlist, donor, follow-up, and external data file linkages (e.g., Social Security Death Master File). Mandated reporting of KPS providing more than a decade of nationally representative data on functional status is also a major strength of our investigation.
This study is limited by reliance on the less-than-ideal KPS scale as it is the only available measure of functional status in the SRTR. The KPS uses multiple domains in the assessment of function and concern may arise that it is less objective or standardized than direct measures of frailty such as grip strength or walking distance. However, the KPS has been extensively validated across a wide range of diseases over the last 60 years, and the simplest 3-tiered scale version used in this analysis has shown excellent inter-rater reliability regardless of provider type or setting[33]. The KPS has in fact been validated in transplant populations specifically, compared with the Short Form survey and other physical function scales for liver disease patients[46], and extensively validated in end-stage renal disease populations[14,19].
Several different measures of physical function and composite frailty scores have been used in the literature and were effective predictors of waitlist and transplant outcomes. However, such direct measures of frailty would demand more resources (time, training, materials) from transplant centers than the KPS alone. Furthermore, their advantages over the use of the KPS should be evaluated in terms of predictive value (area under the curve) before major investment in resource-intensive measures that may already capture similar predictive information.
In summary, we have demonstrated that there is substantial value in using a simple 3-point functional status scale for predicting one-year liver transplant outcomes. We highlight areas where future research may further the validity, and ultimately, the clinical utility of the Karnofsky Functional Performance scale in a liver transplant population. It is important to continue to develop objective measures for describing global health status and illness severity to help in the allocation of organs and waitlist management, patient health improvement, and accurate adjustment for transplant center case-mix for transplant reimbursement.
Frailty is increasingly recognized as an important predictor of outcomes after major surgical procedures including liver transplantation. The Karnofsky Performance Status (KPS) scale has been widely validated for clinical practice for over 60 years.
To investigate the impact of frailty on liver transplant outcomes.
We wanted to determine the extent to which poor pre-transplant functional status, assessed using the KPS scale, is associated with increased risk of mortality and/or graft failure at 1-year post-transplantation. This would give clinicians some objective assessment and help on the decision to allocate livers for high-risk recipients.
This study included 38278 United States adults who underwent first, non-urgent, liver-only transplantation from 2005 to 2014 (Scientific Registry of Transplant Recipients). Functional impairment/disability was categorized as severe (10%-40% of optimal function), moderate (50%-70%), or none/normal (80%-100%). Analyses were conducted using multivariable-adjusted Cox survival regression models. We explored bivariate relationships between the primary exposure (functional status) and potential confounders of interest using contingency table analyses [chi-squared tests for categorical variables, ANOVA for continuous variables, and Spearman's rho (rs) for ordered-variable correlations, using expanded KPS, continuous Model for End-Stage for Liver Disease (MELD)]. Relationships between variables and post-transplant time were explored graphically. One-year cumulative failure rates were estimated using the Kaplan Meier method.
The median age was 56 years, 31% were women, median pre-transplant MELD was 18. Functional impairment was present in 70%; one-quarter of the sample was severely disabled. After controlling for key recipient and donor factors, moderately and severely disabled patients had a 1-year mortality rate of 1.32 [confidence interval (CI): 1.21-1.44] and 1.73 (95%CI: 1.56-1.91) compared to patients with no impairment, respectively. Subjects with moderate and severe disability also had a multivariable-adjusted 1-year graft failure rate of 1.13 (CI: 1.02-1.24) and 1.16 (CI: 1.02-1.31), respectively.
Pre-transplant functional status is a useful prognostic indicator for 1-year post-transplant patient and graft survival. It is important to continue to develop objective measures for describing global health status and illness severity to help in the allocation of organs and waitlist management, patient health improvement, and accurate adjustment for transplant center case-mix for transplant reimbursement. We present data from the first national study illustrating the role of pre-transplant functional status as a predictor of one-year survival among liver transplant recipients. Our results are in agreement with the findings from earlier studies that evaluated Poor functional status as a predictor of adverse transplant outcomes. We have not proposed any theory. The index of frailty - The KPS scale - can be used as a predictor of 1-year post-transplant outcomes (patient and graft survival) in a national United States liver transplant population. Poor functional status predicts 1-year post-liver transplant outcomes. No new methods were proposed. We used very well-established methods of outcome research. It will help to determine objective measures for describing frailty and overall clinical status to help in the allocation of organs and waitlist management, patient health improvement, transplant reimbursement, and policy changes.
That patients’ pre-transplant frailty plays a critical role in transplant outcomes and the transplant community need to study it with more detail. To perform prospective and randomized studies associating frailty index with other biomarkers and correlate them with transplant outcomes. Associating frailty indexes with other biomarkers and correlate them with transplant outcomes. To measure the impact of interventions to improve frailty pre-transplant and correlate it with outcomes.
Manuscript source: Invited manuscript
Specialty type: Transplantation
Country of origin: United States
Peer-review report classification
Grade A (Excellent): A, A
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Hanna RM, Nacif L S-Editor: Dou Y L-Editor: A E-Editor: Qi LL
| 1. | Saidi RF, Markmann JF, Jabbour N, Li Y, Shah SA, Cosimi AB, Bozorgzadeh A. The faltering solid organ donor pool in the United States (2001-2010). World J Surg. 2012;36:2909-2913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 2. | Kim WR, Lake JR, Smith JM, Skeans MA, Schladt DP, Edwards EB, Harper AM, Wainright JL, Snyder JJ, Israni AK, Kasiske BL. OPTN/SRTR 2013 Annual Data Report: liver. Am J Transplant. 2015;15 Suppl 2:1-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 250] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 3. | Dolgin NH, Movahedi B, Martins PN, Goldberg R, Lapane KL, Anderson FA, Bozorgzadeh A. Decade-Long Trends in Liver Transplant Waitlist Removal Due to Illness Severity: The Impact of Centers for Medicare and Medicaid Services Policy. J Am Coll Surg. 2016;222:1054-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 4. | Austin MT, Poulose BK, Ray WA, Arbogast PG, Feurer ID, Pinson CW. Model for end-stage liver disease: did the new liver allocation policy affect waiting list mortality? Arch Surg. 2007;142:1079-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 52] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Cholongitas E, Burroughs AK. The evolution in the prioritization for liver transplantation. Ann Gastroenterol. 2012;25:6-13. [PubMed] |
| 6. | Gotthardt D, Weiss KH, Baumgärtner M, Zahn A, Stremmel W, Schmidt J, Bruckner T, Sauer P. Limitations of the MELD score in predicting mortality or need for removal from waiting list in patients awaiting liver transplantation. BMC Gastroenterol. 2009;9:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 54] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 7. | Lai JC, Feng S, Terrault NA, Lizaola B, Hayssen H, Covinsky K. Frailty predicts waitlist mortality in liver transplant candidates. Am J Transplant. 2014;14:1870-1879. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 384] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 8. | Lai JC, Covinsky KE, Hayssen H, Lizaola B, Dodge JL, Roberts JP, Terrault NA, Feng S. Clinician assessments of health status predict mortality in patients with end-stage liver disease awaiting liver transplantation. Liver Int. 2015;35:2167-2173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 9. | Hubbard RE, Story DA. Patient frailty: the elephant in the operating room. Anaesthesia. 2014;69 Suppl 1:26-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 10. | McIsaac DI, Bryson GL, van Walraven C. Association of Frailty and 1-Year Postoperative Mortality Following Major Elective Noncardiac Surgery: A Population-Based Cohort Study. JAMA Surg. 2016;151:538-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 240] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 11. | Lewsey JD, Dawwas M, Copley LP, Gimson A, Van der Meulen JH. Developing a prognostic model for 90-day mortality after liver transplantation based on pretransplant recipient factors. Transplantation. 2006;82:898-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 12. | van de Mortel TF. Faking it: Social desirability response bias in selfreport research. Aust J Adv Nurs. 2008;25:40-48. |
| 13. | Theou O, Brothers TD, Mitnitski A, Rockwood K. Operationalization of frailty using eight commonly used scales and comparison of their ability to predict all-cause mortality. J Am Geriatr Soc. 2013;61:1537-1551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 386] [Cited by in RCA: 473] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 14. | Arogundade FA, Zayed B, Daba M, Barsoum RS. Correlation between Karnofsky Performance Status Scale and Short-Form Health Survey in patients on maintenance hemodialysis. J Natl Med Assoc. 2004;96:1661-1667. [PubMed] |
| 15. | Kelly DM, Bennett R, Brown N, McCoy J, Boerner D, Yu C, Eghtesad B, Barsoum W, Fung JJ, Kattan MW. Predicting the discharge status after liver transplantation at a single center: a new approach for a new era. Liver Transpl. 2012;18:796-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | van Diepen M, Schroijen MA, Dekkers OM, Rotmans JI, Krediet RT, Boeschoten EW, Dekker FW. Predicting mortality in patients with diabetes starting dialysis. PLoS One. 2014;9:e89744. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 17. | Yates JW, Chalmer B, McKegney FP. Evaluation of patients with advanced cancer using the Karnofsky performance status. Cancer. 1980;45:2220-2224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 18. | OPTN/UNOS. Liver and Intestinal Organ Transplantation Committee: Item for Board Consideration [Internet]. Atlanta, GA; November 11, 2013. Available from: https://unos.org/wp-content/uploads/unos/Board_11-2013_Liver_Committee_Action.pdf. |
| 19. | Edgell ET, Coons SJ, Carter WB, Kallich JD, Mapes D, Damush TM, Hays RD. A review of health-related quality-of-life measures used in end-stage renal disease. Clin Ther. 1996;18:887-938. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 89] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 20. | Moore D, Feurer I, Speroff T, Shaffer D, Nylander W, Kizilisik T, Butler J, Awad J, Gorden DL, Chari R, Wright JK, Pinson CW. Survival and quality of life after organ transplantation in veterans and nonveterans. Am J Surg. 2003;186:476-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 21. | Moore DE, Feurer ID, Speroff T, Gorden DL, Wright JK, Chari RS, Pinson CW. Impact of donor, technical, and recipient risk factors on survival and quality of life after liver transplantation. Arch Surg. 2005;140:273-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 84] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 22. | Mor V, Laliberte L, Morris JN, Wiemann M. The Karnofsky Performance Status Scale. An examination of its reliability and validity in a research setting. Cancer. 1984;53:2002-2007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 23. | Pinson CW, Feurer ID, Payne JL, Wise PE, Shockley S, Speroff T. Health-related quality of life after different types of solid organ transplantation. Ann Surg. 2000;232:597-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 148] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 24. | Schaafsma J, Osoba D. The Karnofsky Performance Status Scale re-examined: a cross-validation with the EORTC-C30. Qual Life Res. 1994;3:413-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 86] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 25. | Schag CC, Heinrich RL, Ganz PA. Karnofsky performance status revisited: reliability, validity, and guidelines. J Clin Oncol. 1984;2:187-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 969] [Cited by in RCA: 1077] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 26. | Terret C, Albrand G, Moncenix G, Droz JP. Karnofsky Performance Scale (KPS) or Physical Performance Test (PPT)? That is the question. Crit Rev Oncol Hematol. 2011;77:142-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 27. | Terwey TH, Hemmati PG, Martus P, Dietz E, Vuong LG, Massenkeil G, Dörken B, Arnold R. A modified EBMT risk score and the hematopoietic cell transplantation-specific comorbidity index for pre-transplant risk assessment in adult acute lymphoblastic leukemia. Haematologica. 2010;95:810-818. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 28. | Geevarghese SK, Bradley AE, Wright JK, Chapman WC, Feurer I, Payne JL, Hunter EB, Pinson CW. Outcomes analysis in 100 liver transplantation patients. Am J Surg. 1998;175:348-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Sonnenday CJ, Volk M, Englesbe MJ. PBC, PSC, and autoimmune disease. Hepatology. 2013;249A-253A. [DOI] [Full Text] |
| 30. | Jacob M, Copley LP, Lewsey JD, Gimson A, Rela M, van der Meulen JH; UK and Ireland Liver Transplant Audit. Functional status of patients before liver transplantation as a predictor of posttransplant mortality. Transplantation. 2005;80:52-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 31. | Dolgin NH, Martins PN, Movahedi B, Lapane KL, Anderson FA, Bozorgzadeh A. Functional status predicts postoperative mortality after liver transplantation. Clin Transplant. 2016;30:1403-1410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 62] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 32. | Ricci P, Therneau TM, Malinchoc M, Benson JT, Petz JL, Klintmalm GB, Crippin JS, Wiesner RH, Steers JL, Rakela J, Starzl TE, Dickson ER. A prognostic model for the outcome of liver transplantation in patients with cholestatic liver disease. Hepatology. 1997;25:672-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 63] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Péus D, Newcomb N, Hofer S. Appraisal of the Karnofsky Performance Status and proposal of a simple algorithmic system for its evaluation. BMC Med Inform Decis Mak. 2013;13:72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 260] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 34. | Rana A, Kaplan B, Jie T, Porubsky M, Habib S, Rilo H, Gruessner AC, Gruessner RW. A critical analysis of early death after adult liver transplants. Clin Transplant. 2013;27:E448-E453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 35. | Feng S, Goodrich NP, Bragg-Gresham JL, Dykstra DM, Punch JD, DebRoy MA, Greenstein SM, Merion RM. Characteristics associated with liver graft failure: the concept of a donor risk index. Am J Transplant. 2006;6:783-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1435] [Cited by in RCA: 1488] [Article Influence: 78.3] [Reference Citation Analysis (0)] |
| 36. | Grønnesby JK, Borgan O. A method for checking regression models in survival analysis based on the risk score. Lifetime Data Anal. 1996;2:315-328. [PubMed] |
| 37. | Scott PJ. Occupational therapy services to enable liver patients to thrive following transplantation. Occup Ther Health Care. 2011;25:240-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 38. | Topp R, Ditmyer M, King K, Doherty K, Hornyak J. The effect of bed rest and potential of prehabilitation on patients in the intensive care unit. AACN Clin Issues. 2002;13:263-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 206] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 39. | Ditmyer MM, Topp R, Pifer M. Prehabilitation in preparation for orthopaedic surgery. Orthop Nurs. 2002;21:43-51; quiz 52-4. [PubMed] |
| 40. | Santa Mina D, Clarke H, Ritvo P, Leung YW, Matthew AG, Katz J, Trachtenberg J, Alibhai SM. Effect of total-body prehabilitation on postoperative outcomes: a systematic review and meta-analysis. Physiotherapy. 2014;100:196-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 262] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 41. | Gillis C, Li C, Lee L, Awasthi R, Augustin B, Gamsa A, Liberman AS, Stein B, Charlebois P, Feldman LS, Carli F. Prehabilitation versus rehabilitation: a randomized control trial in patients undergoing colorectal resection for cancer. Anesthesiology. 2014;121:937-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 482] [Cited by in RCA: 563] [Article Influence: 51.2] [Reference Citation Analysis (0)] |
| 42. | Li C, Carli F, Lee L, Charlebois P, Stein B, Liberman AS, Kaneva P, Augustin B, Wongyingsinn M, Gamsa A, Kim DJ, Vassiliou MC, Feldman LS. Impact of a trimodal prehabilitation program on functional recovery after colorectal cancer surgery: a pilot study. Surg Endosc. 2013;27:1072-1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 342] [Cited by in RCA: 394] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 43. | Mayo NE, Feldman L, Scott S, Zavorsky G, Kim DJ, Charlebois P, Stein B, Carli F. Impact of preoperative change in physical function on postoperative recovery: argument supporting prehabilitation for colorectal surgery. Surgery. 2011;150:505-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 303] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 44. | Carli F, Zavorsky GS. Optimizing functional exercise capacity in the elderly surgical population. Curr Opin Clin Nutr Metab Care. 2005;8:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 270] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 45. | Tessari P. Protein metabolism in liver cirrhosis: from albumin to muscle myofibrils. Curr Opin Clin Nutr Metab Care. 2003;6:79-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 46. | Feurer ID, Moore DE, Speroff T, Liu H, Payne J, Harrison C, Pinson CW. Refining a health-related quality of life assessment strategy for solid organ transplant patients. Clin Transplant. 2004;18 Suppl 12:39-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |