Published online Jul 29, 2020. doi: 10.5500/wjt.v10.i7.206
Peer-review started: January 16, 2020
First decision: February 25, 2020
Revised: March 26, 2020
Accepted: June 14, 2020
Article in press: June 14, 2020
Published online: July 29, 2020
Processing time: 188 Days and 9 Hours
Despite technical refinements, early pancreas graft loss due to thrombosis continues to occur. Conventional coagulation tests (CCT) do not detect hypercoagulability and hence the hypercoagulable state due to diabetes is left untreated. Thromboelastogram (TEG) is an in-vitro diagnostic test which is used in liver transplantation, and in various intensive care settings to guide anticoagulation. TEG is better than CCT because it is dynamic and provides a global hemostatic profile including fibrinolysis.
To compare the outcomes between TEG and CCT (prothrombin time, activated partial thromboplastin time and international normalized ratio) directed anticoagulation in simultaneous pancreas and kidney (SPK) transplant recipients.
A single center retrospective analysis comparing the outcomes between TEG and CCT-directed anticoagulation in SPK recipients, who were matched for donor age and graft type (donors after brainstem death and donors after circulatory death). Anticoagulation consisted of intravenous (IV) heparin titrated up to a maximum of 500 IU/h based on CCT in conjunction with various clinical parameters or directed by TEG results. Graft loss due to thrombosis, anticoagulation related bleeding, radiological incidence of partial thrombi in the pancreas graft, thrombus resolution rate after anticoagulation dose escalation, length of the hospital stays and, 1-year pancreas and kidney graft survival between the two groups were compared.
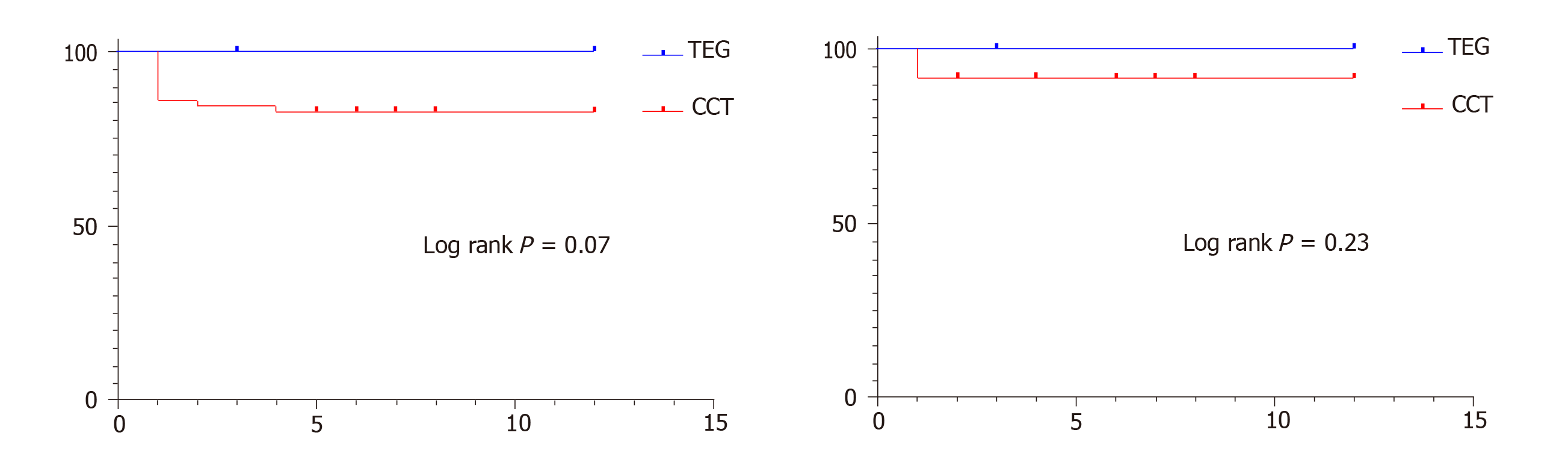
Seventeen patients who received TEG-directed anticoagulation were compared against 51 contemporaneous SPK recipients (ratio of 1: 3) who were anticoagulated based on CCT. No graft losses occurred in the TEG group, whereas 11 grafts (7 pancreases and 4 kidneys) were lost due to thrombosis in the CCT group (P = 0.06, Fisher’s exact test). The overall incidence of anticoagulation related bleeding (hematoma/ gastrointestinal bleeding/ hematuria/ nose bleeding/ re-exploration for bleeding/ post-operative blood transfusion) was 17.65% in the TEG group and 45.10% in the CCT group (P = 0.05, Fisher’s exact test). The incidence of radiologically confirmed partial thrombus in pancreas allograft was 41.18% in the TEG and 25.50% in the CCT group (P = 0.23, Fisher’s exact test). All recipients with partial thrombi detected in computed tomography (CT) scan had an anticoagulation dose escalation. The thrombus resolution rates in subsequent scan were 85.71% and 63.64% in the TEG group vs the CCT group (P = 0.59, Fisher’s exact test). The TEG group had reduced blood product usage {10 packed red blood cell (PRBC) and 2 fresh frozen plasma (FFP)} compared to the CCT group (71 PRBC/ 10 FFP/ 2 cryoprecipitate and 2 platelets). The proportion of patients requiring transfusion in the TEG group was 17.65% vs 39.25% in the CCT group (P = 0.14, Fisher’s exact test). The median length of hospital stay was 18 days in the TEG group vs 31 days in the CCT group (P = 0.03, Mann Whitney test). The 1-year pancreas graft survival was 100% in the TEG group vs 82.35% in the CCT group (P = 0.07, log rank test) and, the 1-year kidney graft survival was 100% in the TEG group vs 92.15% in the CCT group (P = 0.23, log tank test).
TEG is a promising tool in guiding judicious use of anticoagulation with concomitant prevention of graft loss due to thrombosis, and reduces the length of hospital stay.
Core tip: Early post-operative graft thrombosis and graft loss are the Achilles heel of pancreas transplantation and routine prophylactic anticoagulation seems to be the logical remedy. Do all patients need the same dose of anticoagulation? We have compared the outcomes of thromboelastogram directed and conventional coagulation test based anticoagulation in simultaneous pancreas and kidney (SPK) transplant recipients and highlighted the needs and advantages of individualized anticoagulation based on thromboelastogram.
- Citation: Gopal JP, Dor FJ, Crane JS, Herbert PE, Papalois VE, Muthusamy AS. Anticoagulation in simultaneous pancreas kidney transplantation - On what basis? World J Transplant 2020; 10(7): 206-214
- URL: https://www.wjgnet.com/2220-3230/full/v10/i7/206.htm
- DOI: https://dx.doi.org/10.5500/wjt.v10.i7.206
In spite of technical refinements, pancreas allograft thrombosis remains the most common non-immunological cause of early graft loss in pancreas transplantation[1,2]. The exact incidence of pancreas graft loss due to thrombosis varies between 1 and 40%, but has been reported to be as high as 29% in the first 6 months after transplantation[3,4]. The etiology is multifactorial encompassing donor and recipient factors. In contrast to other forms of solid organ transplantation, a hypercoagulable state due to diabetes and alteration in the venous flow dynamics (in a low flow organ) leading to stasis are additional risk factors for thrombosis inherent for pancreas transplantation. In this context, most of the centers have adopted routine prophylactic anticoagulation. Majority of the centers anticoagulate their recipients based on conventional coagulation test (CCT) and only fewer centers utilize point of care (POC) testing like TEG (Thromboelastography)/ ROTEM (Rotational thromboelastometry) to optimize anticoagulation. The existing literature about TEG in pancreas transplantation has suggested that perioperative TEG can identify high risk recipients at risk of graft thrombosis and also has presented the argument for individualized anticoagulation[3,5]. So far, there is no clear consensus on the basis for anticoagulation and to the best of our knowledge no one has attempted to compare the outcomes between TEG and CCT-directed anticoagulation.
Following institutional audit committee approval, a retrospective analysis of 127 pancreas transplants performed between 2008 and 2019 was done. Data was collected from a prospectively maintained database. After excluding isolated pancreas transplants (Pancreas after kidney and pancreas transplant alone), re-transplants, recipients with a known thrombophilic disorder and, grafts from pediatric donors, sixty-eight SPK transplant recipients were included in the study. The recipients in both the groups were matched for donor age and graft type [donors after brainstem death (DBD)/donors after circulatory death (DCD)].
According to our center’s protocol, all the DBD donors were less than 65-years old and all the DCD donors were less than 55-years old. The body mass index (BMI) cut off was 30 kg/m2. All the DCD donors had a functional warm ischemia time (systolic blood pressure < 50 mmHg and/or oxygen saturation of 70%) of less than 60 min and the downtime of less than 30 min.
All of the recipients had a standard arterial reconstruction using the donor iliac artery bifurcation (Y graft) anastomosed to the splenic and superior mesenteric artery (SMA). Portal vein extension was used on an individual case basis. University of Wisconsin solution was used for organ preservation. Pancreas graft was implanted on the right side either extra-peritoneally (74.50%) into the common or external iliac vessels or intra-peritoneally (25.50%) with inflow from the common iliac artery and venous drainage to the inferior vena cava. All of the kidneys were implanted into the external iliac vessels on the contralateral side except for one, which was implanted on the same side. The choice of implantation was based on clinical consideration and surgeon’s preference. All of the recipients had enteric exocrine drainage. Immunosuppression consisted of induction with intravenous alemtuzumab 30 mgs (single dose) and methylprednisolone 500 mgs. Maintenance immunosuppression was with tacrolimus, mycophenolate mofetil and a short course of steroids (7 days).
Thromboelastogram (TEG) was developed by Dr Helmut Hartert[6] in 1948 and is being used extensively in trauma, cardiac surgery and liver transplantation[7-9]. TEG HaemoneticsR is an in-vitro diagnostic test in which a plastic pin attached to a torsion wire is immersed into a small cuvette of blood and the cuvette is rotated through an arc of approximately 4.75 degrees, 6 times per minute to simulate sluggish flow and to activate coagulation. The kinetic changes transmitted by the torsion wire is analyzed by the analyzer. The variables of interest are: reaction time (R, measured in seconds) the time from the start of the test until initiation of fibrin formation; clot kinetics (K, measured in seconds) time from R until clot reaches 20mm; angle (α) angle from the tangential line drawn to meet the TEG tracing from R; maximum amplitude (MA, measured in mm) a reflection of clot strength and coagulation index (CI, measured in dynes/second) which is a culmination of all the above parameters. The R time indicates the concentration of soluble clotting factors in plasma and correlates with prothrombin time (PT) results. The K correlates positively with PT/ activated partial thromboplastin time (aPTT) results and inversely with fibrinogen levels. The angle (α) indicates the rapidity of fibrin build up and cross-linking, and is a dynamic measure that is unique to thromboelastography. The α and MA correlates positively with fibrinogen and platelet levels in circulation; high fibrinogen levels or thrombocytosis results in increased α and MA.
TEG was done at the following time points: at the time of anesthetic induction; before clamping of vessels; on return to anesthesia recovery; twice daily for the first 48 hours; 24 hours after any major alteration in anticoagulation dose; repeated as and when required until discharge. Kaolin tracing was used for clinical decision making. The target CI was between -3 and +3.
Prophylactic anticoagulation consisted of intravenous heparin initially started at 100 IU/hour once clinically stable and titrated up to a maximum of 500 IU/hour directed by TEG results in the TEG group/ by a combination of clinical and laboratory parameters in the CCT group (presence or absence of hematuria, character and quantity of drain output, hemoglobin and platelet trend and, aPTT results). Intravenous heparin was subsequently switched to subcutaneous heparin 2500 or 5000 IU twice daily and then to Enoxaparin 20 mgs once daily/ Tinzaparin 3500 IU once daily at discharge. Low molecular weight heparin (LMWH) was continued until 6 weeks post-transplant. After 6 weeks LMWH was stopped and Aspirin 75 mgs once daily was continued indefinitely.
Therapeutic anticoagulation consisted of either enoxaparin 1.5 mgs/kg body weight or tinzaparin 175 IU/kg body weight or warfarin dosing adjusted to aim for an international normalized ratio between 2 and 2.5, andcontinued for three months.
Post-operative blood or blood component transfusion, gross and significant hematuria, upper or lower gastrointestinal bleeding, intracranial bleeding, nose bleeding, bleeding or hematoma in the injection sites, hematoma identified in computed tomography (CT) scan and re-exploration where no source of bleeding was identified were considered anticoagulation related bleeding.
Intraoperative blood product usage and re-explorations where specific bleeding source was identified were considered surgical bleeding and excluded. Bleeding after initiation of therapeutic dose of anticoagulation was excluded.
A triple phase contrast-enhanced CT scan of the abdomen and pelvis was done for the following indications: Sudden onset of severe abdominal pain; consistently increasing amylase; new onset hyperglycemia after a period of insulin independence; concerns regarding perfusion in the ultrasound scan. All the CT scans were interpreted by two independent radiologists and finally reported.
Pancreas and kidney graft loss due to thrombosis, incidence of anticoagulation related bleeding, blood and blood product usage, proportion of patients requiring transfusion, radiological incidence of partial thrombus, thrombus resolution rate after anticoagulation dose escalation and length of hospital stay were compared between the two groups.
Categorical variables are expressed as frequency (%) and continuous variables as median. Difference between the categorical variables were assessed by using Fisher’s exact test and difference between the continuous variables were assessed by using Mann Whitney test. Survival analysis was done by using Kaplan-Meir survival plots. All the statistical analyses were performed using Prism software (Version 8).
Seventeen SPK recipients received TEG-directed anticoagulation and were compared against 51 contemporaneous SPK recipients who were anticoagulated based on CCT. The two groups were comparable in terms of risk factors for graft thrombosis (Table 1). The peak value of serum amylase and C-reactive protein within the first week after transplantation, and prolonged cold ischemia time for the pancreas (more than 12 hours) were utilized as surrogate markers for graft pancreatitis.
| Parameters | TEG group | CCT group | Statistical significance |
| Median donor age | 49 yr (n = 17) | 38 yr (n = 51) | P = 0.16 Mann-Whitney test |
| Proportion of DCD grafts, n (%) | 4 (23.53) | 12 (23.53) | P > 0.99 Fisher’s exact test |
| Proportion of recipients with portal vein extension, n (%) | 3 (17.65) | 7 (13.73) | P = 0.70 Fisher’s exact test |
| Proportion of recipients with prolonged cold ischemic time(> 12 h) for the pancreas graft, n (%) | 7 (41.18) | 27 (52.94) | P = 0.58 Fisher’s exact test |
| Median peak serum amylase in the first two weeks after transplant | 191 IU/L | 312 IU/L | P = 0.09 Mann-Whitney test |
| Median peak C-reactive protein in the first two weeks after transplant | 148.6 | 160.6 | P = 0.54 Mann-Whitney test |
There was no thrombotic graft loss in the TEG group whereas 10.78% of grafts (7 Pancreases and 4 kidneys) were lost due to thrombus in the CCT group (P = 0.06, Fisher’s exact test). Out of the 11 grafts that were lost, 7 were explanted on table (4 pancreases and 3 kidneys) and the rest of them were explanted within the first week after transplant. 2 of them were from DCD donors and the rest of the 9 grafts were from DBD donors. As depicted in Figure 1, the one-year pancreas graft survival was 100% in the TEG group vs 82.35% in the CCT group (P = 0.07, Log-rank test) and the one-year kidney graft survival was 100% in the TEG group vs 92.15% in the CCT group (P = 0.23, log rank test).
The incidence of anticoagulation related bleeding was 17.65% (3/17) in the TEG group vs 45.10% (23/51) in the CCT group (P = 0.05, Fisher’s exact test). Two patients in TEG group and 4 patients in the CCT group required re-exploration and no source of bleeding was identified. In the CCT group 8 patients had hematoma (peri-nephric-4/ peri-pancreatic-4) identified in the CT scan, one patient had significant hematuria, one patient had GI bleeding and 9 patients required blood transfusion; whereas 1 patient in the TEG group had peri-nephric hematoma.
The overall blood product usage was 12 in TEG group {10 packed red blood cells (PRBC) and 2 fresh frozen plasma (FFP)} vs 85 in the CCT group (71 PRBC/ 10 Cryoprecipitate/ 2 FFP and 2 platelet).
The proportion of patients requiring transfusion in TEG group was 17.65% vs 39.25% in the CCT group (P = 0.14, Fisher’s exact test).
The radiological incidence of partial thrombus in the pancreas graft vasculature in TEG group was 41.18% (7/17) vs 25.50% (13/51) in CCT group (P = 0.23, Fisher’s exact test). The non-occlusive thrombi were identified in the distal splenic artery (n = 7, 4 in the TEG group vs 3 in the CCT group); in distal splenic vein (n = 2, 2 in the CCT group); in both the distal splenic artery and splenic vein (n = 6, 1 in the TEG group vs 5 in the CCT group); in the SMA (n = 1, 1 in the CCT group); in the distal splenic artery and SMA (n = 2, 2 in the TEG group) and in the graft portal vein (n = 3, 1 in the TEG group vs 2 in the CCT group). In the TEG group 2/7 grafts with partial thrombi were from DCD donors while 4/13 grafts with partial thrombi in the CCT group were from DCD donors. All the patients with partial thrombus in TEG group had a kaolin coagulation index (CI) of more than +3 in the intraoperative TEG, indicating a hypercoagulable state. The indications for CT scan were hyperglycemia (n = 5), hyperamylasemia (n = 9), severe abdomen pain (n = 3), hemoglobin drop (n = 2), recurrent hypoglycemia(n = 1), and reduced flow in doppler (n = 1)
Among the patients with partial thrombus, all patients had anticoagulation dose escalation in the TEG group while 9/11 patients had anticoagulation dose escalation in the CCT group and the remaining 2 patients in the CCT group received therapeutic dose of anticoagulation for 3 months due to associated iliofemoral deep vein thrombosis. The thrombus resolution rate after anticoagulation dose escalation was 85.71% (6/7) in TEG group vs 63.64% (7/11) in the CCT group (P = 0.59, fisher’s exact test). All 5 patients with persistent thrombus had functioning pancreas allografts.
The median length of hospital stay was 18 days in TEG group vs 31 days in CCT group (P = 0.03, Mann Whitney test).
Graft thrombosis is still the most common non-immunological cause for early graft loss in pancreas transplantation and the etiology is multifactorial. Anticoagulation is the key solution to prevent graft thrombosis. Although CCT are widely available to guide anticoagulation, they are time consuming and don’t reliably measure hypercoagulability and fibrinolysis. Moreover, these tests are done using plasma rather than whole blood and hence the contribution of platelets towards clot formation and clot strength is not measured.
The thromboelastography is an in-vitro assessment of the thrombodynamic properties of blood as it is induced to clot under a low shear environment, aimed to resemble sluggish venous flow. TEG is a dynamic and real-time measure of coagulation and is more accurate than CCT as it provides a comprehensive hemostatic profile including fibrinolysis. The results of TEG are available much faster than CCT. Titrating anticoagulation in the early post-operative period is very crucial in pancreas transplantation and the rapid accessibility and reproducibility of TEG makes it more suitable than CCT to drive anticoagulation.
The case series by Vaidya et al[5] has already reported that TEG serves to identify the cohort of patients at risk for graft thrombosis and thereby enabling safe therapeutic anticoagulation with minimum morbidity and mortality. For the first time, we have compared CCT vs TEG-directed anticoagulation in pancreas transplantation and have re-iterated the advantages of TEG-directed anticoagulation. Although the difference in the percentage of graft loss between the two groups was not statistically significant, in our view, the two groups were comparable in terms of risk factors for graft thrombosis and no thrombotic graft loss in the TEG group in this setting has a definite clinical impact while a greater number of patients are needed to achieve statistical significance. A pre-operative TEG identifies the subgroup of patients at higher risk of thrombotic graft loss thereby guiding judicious use of anticoagulation. The anticoagulation related bleeding was less in the TEG group as evidenced by lower rates of re-exploration, reduced blood product usage and transfusion requirements. This is because only those patients towards the hypercoagulable spectrum in the TEG (CI > +3) had higher than conventional doses of anticoagulation. The shorter length of hospital stay in the TEG group is an added advantage. The reasons for prolonged hospital admission in CCT group were, the need for re-exploration (n = 8) and peripancreatic collection with infection (n = 14). The delayed graft function rates were comparable between the two groups (17.65% in the TEG group vs 21.56% in the CCT group)
The other important finding in this study is that, apart from the two patients who had thrombi in the pancreas graft vasculature coexisting with iliofemoral deep vein thrombosis, none of the remaining patients with partial venous or arterial thrombi were therapeutically anticoagulated in both the groups. Irrespective of thrombus resolution, all of the patients with partial thrombi in the pancreas graft vasculature had a functioning pancreas allograft. The inference is that not all patients with partial thrombi in the pancreas allograft need therapeutic anticoagulation. The different strategies reported for the management of partial thrombi are: therapeutic anticoagulation, early re-exploration and endovascular thrombectomy and thrombolysis[10-12]. In our study, 16/19 patients had distal non-occlusive thrombi and hence anticoagulation dose escalation was sufficient. Even with the 3 patients who had non-occlusive thrombi in the main portal vein, anticoagulation dose escalation was adequate. Our results concur with the study published by Hakeem et al[13], although it is not supported by a CT grading system.
The optimal regimen for prophylactic anticoagulation still remains a topic of debate. The anticoagulation protocols are center specific, and is often a mix of heparin, antiplatelet agents, dextran and warfarin[3,5,14,15]. Most of our recipients were on intravenous heparin in the immediate post-operative period due to the feasibility of urgent reversal in the event of bleeding and, subsequently switched to LMWH, that was continued until 6 weeks. LMWH was not commenced from the beginning as there are several studies[14,16] reporting that early post-operative use of low dose unfractionated heparin prevents early graft loss due to thrombosis without increased risk of bleeding and also due to the different pharmacokinetics of LMWH that hampers their safe usage in the early post-transplant period. The incidence of pancreas graft loss due to thrombosis in our study was 10.30% (7 patients), which is similar to that reported in other studies[3,5,14,15]. Another approach would be to use platelet function assays and fibrinolysis to guide anticoagulation. Raveh et al[17] has reported a higher graft thrombosis rate (61%) in patients with pre-operative platelet dysfunction, although the thrombosis rate was not significantly different between normal and abnormal fibrinolysis phenotypes. This might help in deciding the addition or dose modification of antiplatelets in the anticoagulation regimen. With the widespread utilization of alemtuzumab for induction therapy, the associated thrombocytopenia with a reported incidence of 14%[18] and subsequent platelet reconstitution also needs to be accounted for when deciding anticoagulation and hence dynamic anticoagulation monitoring such as the TEG is crucial.
With the rising organ scarcity and shifting donor demographics, the pancreas transplant community has obviously expanded the donor acceptance criteria. This together with increasing DCD donation in many countries can potentially increase the incidence of pancreas graft thrombosis[19-21]. TEG can be a promising tool that can aid to push the boundary more effectively thereby translating into more successful clinical outcomes.
The benefits of TEG-directed anticoagulation do not stop with pancreas component of the SPK. As evidenced from the study none of the kidneys in the TEG group were lost to thrombosis. Transplant renal vein thrombosis occurs early after the transplantation with a reported incidence of 0.1%-4.2% and diabetes in the recipient is one among the other risk factors for graft thrombosis[22,23]. TEG-directed anticoagulation in diabetic patients needing kidney transplantation is another potential area for application.
Limitations of the study are the retrospective nature, and a relatively small number of patients. The surgical techniques for pancreas transplantation have been evolving and transplants with caval venous drainage has been reported to have lower risk of graft thrombosis predominantly due to higher blood flow in the inferior vena cava[24,25]. The majority of the transplants in the TEG group were performed intra-peritoneally with caval venous drainage (52.94%) compared to the CCT group (5.88%) and it could potentially be a confounding variable. This reflects a change in practice in our center due to the expanding surgical team. It is also crucial to note that none of the grafts were lost to thrombosis even in the remaining 8 recipients with iliac venous drainage in the TEG group. In our opinion, the results of this study pertaining to graft loss confirm the existing literature about TEG in pancreas transplantation and add new insights on several other benefits of TEG-directed anticoagulation.
In conclusion, this is the first study to compare the outcomes between TEG and CCT-directed anticoagulation in SPK transplantation. It is clearly evident that TEG-directed anticoagulation prevents thrombotic graft loss without concurrent increase in anticoagulation related bleeding and also reduces the length of hospital stay. Future larger studies with cost benefit analyses would be relevant for increasing the utilization of TEG in pancreas transplantation.
Pancreas allograft thrombosis is the most common non-immunological cause for early graft loss. Hence, prophylactic anticoagulation has become the routine practice. Conventional coagulation tests (CCT) are slow in titrating anticoagulation especially in the early post-operative period and also don’t detect hypercoagulable state that is inherent to diabetes and is left unaddressed. Thromboelastogram (TEG) is a dynamic, rapid and reliable tool that provides a complete picture of coagulation. TEG based anticoagulation in pancreas transplantation has been proven to identify patients at risk of thrombotic graft loss thereby enabling safe anticoagulation with least morbidity and mortality.
Despite these studies, there is no clear consensus for the basis of anticoagulation. Therefore, we aimed to compare the outcomes between TEG and CCT based anticoagulation in simultaneous pancreas and kidney (SPK) transplantation.
A single center retrospective analysis comparing the outcomes between TEG and CCT-directed anticoagulation in SPK recipients, who were matched for donor age and graft type (Donors after brainstem death and donors after circulatory death). Anticoagulation consisted of intravenous (IV) heparin titrated up to a maximum of 500 IU/hour based on CCT in conjunction with various clinical parameters or directed by TEG results. Graft loss due to thrombosis, anticoagulation related bleeding, radiological incidence of partial thrombi in the pancreas graft, thrombus resolution rate after anticoagulation dose escalation, length of the hospital stays and, 1-year pancreas and kidney graft survival between the two groups were compared.
For the first time we have compared TEG and CCT directed anticoagulation in pancreas transplantation. There were no thrombotic graft losses in the TEG group whereas 7 pancreases and 4 kidneys were lost in the CCT group. The incidence of anticoagulation related bleeding was less (17.65% TEG vs 45.10%CCT, P = 0.05) and also the median length of hospital stay was reduced (18 days TEG vs 31 days CCT, P = 0.03) in TEG group compared to the CCT group.
TEG based anticoagulation prevents thrombotic graft loss without concomitant increase in the incidence of anticoagulation related bleeding and also reduces the length of hospital stay. Hereby our findings re-confirm the published literature.
Future prospective studies with more patient numbers will be more beneficial for generating a robust evidence base.
We would like to acknowledge our nephrology and anesthesia colleagues of Imperial College Renal and Transplant Center for their contribution towards patient management.
Manuscript source: Invited manuscript
Specialty type: Transplantation
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Grignani G, Rituerto DC S-Editor: Ma YJ L-Editor: A E-Editor: Wu YXJ
| 1. | Troppmann C, Gruessner AC, Benedetti E, Papalois BE, Dunn DL, Najarian JS, Sutherland DE, Gruessner RW. Vascular graft thrombosis after pancreatic transplantation: univariate and multivariate operative and nonoperative risk factor analysis. J Am Coll Surg. 1996;182:285-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 2. | Humar A, Ramcharan T, Kandaswamy R, Gruessner RW, Gruessner AC, Sutherland DE. Technical failures after pancreas transplants: why grafts fail and the risk factors--a multivariate analysis. Transplantation. 2004;78:1188-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 201] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 3. | Burke GW, Ciancio G, Figueiro J, Buigas R, Olson L, Roth D, Kupin W, Miller J. Hypercoagulable state associated with kidney-pancreas transplantation. Thromboelastogram-directed anti-coagulation and implications for future therapy. Clin Transplant. 2004;18:423-428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 4. | Muthusamy AS, Giangrande PL, Friend PJ. Pancreas allograft thrombosis. Transplantation. 2010;90:705-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 5. | Vaidya A, Muthusamy AS, Hadjianastassiou VG, Roy D, Elker DE, Moustafellos P, Muktadir A, Sinha S, Friend PJ. Simultaneous pancreas--kidney transplantation: to anticoagulate or not? Is that a question? Clin Transplant. 2007;21:554-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 6. | Hartert, H. Blutgerinnungsstudien mit der Thrombelastographie, einem neuen Untersuchungsverfahren. Klin Wochenschr. 1948;26:577–583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 394] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 7. | Krenn CG, De Wolf AM. Current approach to intraoperative monitoring in liver transplantation. Curr Opin Organ Transplant. 2008;13:285-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 8. | Shore-Lesserson L, Manspeizer HE, DePerio M, Francis S, Vela-Cantos F, Ergin MA. Thromboelastography-guided transfusion algorithm reduces transfusions in complex cardiac surgery. Anesth Analg. 1999;88:312-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 427] [Cited by in RCA: 378] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 9. | Ives C, Inaba K, Branco BC, Okoye O, Schochl H, Talving P, Lam L, Shulman I, Nelson J, Demetriades D. Hyperfibrinolysis elicited via thromboelastography predicts mortality in trauma. J Am Coll Surg. 2012;215:496-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 121] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 10. | Harbell JW, Morgan T, Feldstein VA, Roll GR, Posselt A, Kang SM, Feng S, Hirose R, Freise CE, Stock P. Splenic Vein Thrombosis Following Pancreas Transplantation: Identification of Factors That Support Conservative Management. Am J Transplant. 2017;17:2955-2962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 11. | Fridell JA, Mangus RS, Mull AB, Taber TE, Sanders CE, Slisher RC, Goble ML, Powelson JA. Early reexploration for suspected thrombosis after pancreas transplantation. Transplantation. 2011;91:902-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 12. | Barrufet M, Burrel M, Angeles García-Criado M, Montañà X, Real MI, Ferrer J, Fernández-Cruz L, Gilabert R. Pancreas transplants venous graft thrombosis: endovascular thrombolysis for graft rescue. Cardiovasc Intervent Radiol. 2014;37:1226-1234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Hakeem A, Chen J, Iype S, Clatworthy MR, Watson CJE, Godfrey EM, Upponi S, Saeb-Parsy K. Pancreatic allograft thrombosis: Suggestion for a CT grading system and management algorithm. Am J Transplant. 2018;18:163-179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 37] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 14. | Scheffert JL, Taber DJ, Pilch NA, Chavin KD, Baliga PK, Bratton CF. Clinical outcomes associated with the early postoperative use of heparin in pancreas transplantation. Transplantation. 2014;97:681-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Schenker P, Vonend O, Ertas N, Wunsch A, Schaeffer M, Rump LC, Viebahn R. Incidence of pancreas graft thrombosis using low-molecular-weight heparin. Clin Transplant. 2009;23:407-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Humar A, Kandaswamy R, Granger D, Gruessner RW, Gruessner AC, Sutherland DE. Decreased surgical risks of pancreas transplantation in the modern era. Ann Surg. 2000;231:269-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 191] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 17. | Raveh Y, Ciancio G, Burke GW, Figueiro J, Chen L, Morsi M, Namias N, Singh BP, Lindsay M, Alfahel W, Sleem MS, Nicolau-Raducu R. Susceptibility-directed anticoagulation after pancreas transplantation: A single-center retrospective study. Clin Transplant. 2019;33:e13619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Muthusamy AS, Vaidya AC, Sinha S, Roy D, Elker DE, Friend PJ. Alemtuzumab induction and steroid-free maintenance immunosuppression in pancreas transplantation. Am J Transplant. 2008;8:2126-2131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 19. | Farney AC, Rogers J, Stratta RJ. Pancreas graft thrombosis: causes, prevention, diagnosis, and intervention. Curr Opin Organ Transplant. 2012;17:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 20. | Muthusamy AS, Mumford L, Hudson A, Fuggle SV, Friend PJ. Pancreas transplantation from donors after circulatory death from the United Kingdom. Am J Transplant. 2012;12:2150-2156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Mittal S, Gilbert J, Friend PJ. Donors after circulatory death pancreas transplantation. Curr Opin Organ Transplant. 2017;22:372-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 22. | Parajuli S, Lockridge JB, Langewisch ED, Norman DJ, Kujovich JL. Hypercoagulability in Kidney Transplant Recipients. Transplantation. 2016;100:719-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 23. | El Zorkany K, Bridson JM, Sharma A, Halawa A. Transplant Renal Vein Thrombosis. Exp Clin Transplant. 2017;15:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Laftavi MR, Gruessner A, Gruessner R. Surgery of pancreas transplantation. Curr Opin Organ Transplant. 2017;22:389-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Laftavi MR, Pankewycz O, Kohli R, Feng L, Said M, Sharma R, Patel S. Short and long-term outcomes of systemic drainage to IVC: a new technique for pancreas transplantation. Transplant Proc. 2014;46:1900-1904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |