Peer-review started: August 29, 2016
First decision: November 14, 2016
Revised: November 19, 2016
Accepted: December 7, 2016
Article in press: December 9, 2016
Published online: February 20, 2017
Processing time: 174 Days and 0.7 Hours
Pluripotent stem cells have the remarkable self-renewal ability and are capable of differentiating into multiple diverse cells. There is increasing evidence that the aging process can have adverse effects on stem cells. As stem cells age, their renewal ability deteriorates and their ability to differentiate into the various cell types is altered. Accordingly, it is suggested aging-induced deterioration of stem cell functions may play a key role in the pathophysiology of the various aging-associated disorders. Understanding the role of the aging process in deterioration of stem cell function is crucial, not only in understanding the pathophysiology of aging-associated disorders, but also in future development of novel effective stem cell-based therapies to treat aging-associated diseases. This review article first focuses on the basis of the various aging disease-related stem cell dysfunction. It then addresses the several concepts on the potential mechanism that causes aging-related stem cell dysfunction. It also briefly discusses the current potential therapies under development for aging-associated stem cell defects.
Core tip: Stem cells have the remarkable self-renewal capability and the amazing ability to differentiate into all cell types. It is generally believe that stem cells are the main source that provides cells to repair and regenerate damaged tissues and organs. However, there is now compelling evidence that the aging process has a deleterious effect on stem cells, and that the aging effects on stem cells may have play essential roles in the pathophysiology of the various aging-associated diseases. This review discusses briefly the relationship of aging-associated stem cell dysfunction and the various aging-associated ailments, and several proposed concepts on the molecular mechanism of aging-related stem cell dysfunction.
- Citation: Ahmed ASI, Sheng MH, Wasnik S, Baylink DJ, Lau KHW. Effect of aging on stem cells. World J Exp Med 2017; 7(1): 1-10
- URL: https://www.wjgnet.com/2220-315X/full/v7/i1/1.htm
- DOI: https://dx.doi.org/10.5493/wjem.v7.i1.1
Aging is an unavoidable physiological consequence of the living animals. Mammalian aging is mediated by the complex cellular and organismal processes, driven by diverse acquired and genetic factors[1]. Aging is among the greatest known risk factors for most human diseases[2-5], and of roughly 150000 people who die each day across the globe, about two thirds die from age-related causes[6].
In modern era, one of the emerging fields in treating human diseases is the “stem cells” research, as stem cells have the remarkable potential for use to treat a wide range of diseases. Accordingly, stem cells research has become a focal point of biomedical research since 1998, when Dr. James Alexander Thomson made the scientific breakthrough of successful generation of several embryonic stem cell lines from human blastocysts[7,8]. Stem cells are undifferentiated pluripotent cells that can give rise to all tissue types and serve as a sort of internal repair system[9]. Until the recent advance in development of induced pluripotent stem cells (iPSCs), scientists primarily worked with two kinds of pluripotent stem cells from animals and humans: Embryonic stem cells, which are isolated from the inner cell mass of blastocysts, and non-embryonic “somatic” or “adult” stem cells, which are found in various tissues[10]. Because of potential ethical issues, “adult” stem cells have become the primary target.
Although stem cell science promises to offer revolutionary new ways of treating diseases, it is identified that aging affect the ability of stem (and progenitor) cells to function properly, which ultimately can lead to cell death (apoptosis), senescence (loss of a cell’s power of division and growth), or loss of regenerative potential[11,12]. Aging may also shift gene functions, as reported for some genes such as, p53 and mammalian target of rapamycin (mTOR), which are beneficial in early life, but becomes detrimental later in life[13-15]. In this regard, a novel theory, namely “stem cell theory of aging”, has been formulated, and it assumes that inability of various types of pluripotent stem cells to continue to replenish the tissues of an organism with sufficient numbers of appropriate functional differentiated cell types capable of maintaining that tissue’s (or organ’s) original function is in large part responsible for the aging process[1]. In addition, aging also compromises the therapeutic potentials of stem cells, including cells isolated from aged individuals or cells that had been cultured for many passages in vitro. Nevertheless, in either case, understanding the molecular mechanism involved in aging and deterioration of stem cell function is crucial in developing effective new therapies for aging- as well as stem cell malfunction-related diseases. In fact, given the importance of the aging-associated diseases, scientists have developed a keen interest in understanding the aging process as well as attempting to define the role of dysfunctional stem cells in the aging process.
In this review, we will first focus on the various aging disease-related stem cell dysfunction and then address the several concepts on potential mechanisms that cause aging-related stem cell dysfunction. We will also discuss current strategies for reversing age-related stem cell dysfunction. Finally, we will discuss up-to-date therapies for aging-associated stem cell defects, available-drugs, growth factors, etc.
Adult stem cells, also known as somatic stem cells, are found throughout the body in every tissues and organs after development and function as self-renewing cell pools to replenish dying cells and regenerate damaged tissues throughout life[16]. However, adult stem cells appear to age with the person. As stem cells age, their functional ability also deteriorates[12,17]. Specifically, this regenerative power appears to decline with age, as injuries in older individuals heal more slowly than in childhood. For example, healing of a fractured bone takes much longer time in elderly than in young individuals[18-21]. There is a substantial amount of evidence showing that deterioration of adult stem cells in the adult phase can become an important player in the initiation of several diseases in aging[22,23]. The following is some of the examples of aging-associated effects on stem cells.
Neural stem cells (NSCs) are multipotent and self-renewing cells and located primarily in the neural tissues. In response to a complex combination of signaling pathways, NSCs differentiate into various specific cell types locally in the central nervous system (CNS), like neurons, astrocytes, and oligodendrocytes[24]. NSCs in humans maintain brain homeostasis and it continuously replenishes new neurons, which are important for cognitive functions[25,26]. However, there is now strong evidence for the aging-associated cognitive deficits, such as olfactory dysfunction, spatial memory deficits, and neurodegenerative disorders, which are caused by deterioration of NSC proliferation and differentiation and enhanced NSC senescence as a consequence of aging[27,28].
Mesenchymal stem cells (MSCs) are multipotent stromal cells that can differentiate into cells of mesenchyme tissues, including osteoblasts (bone cells)[29], chondrocytes (cartilage cells)[30], myocytes (muscle cells)[31] and adipocytes (fat cells)[32]. MSCs were first isolated from the bone marrow of guinea pigs in 1970’s and after that it was isolated from almost every organ in mice including fat, liver, spleen, pancreas, kidney, lung, muscle, and brain[32]. Human MSCs have also been isolated from umbilical cord tissue and cord blood, placenta, bone and joints[33]. However, the major sources of MSCs are the bone marrow-derived MSCs (BM-MSCs) and the adipose tissue-derived MSCs (A-MSCs); and they are currently the most studied MSCs[32,34]. Aging also affects MSCs in humans and in animal models as indicated by the decrease in the bone marrow MSC pool and also shifts their lineage differentiation from one that usually favors osteoblastic differentiation to one that prefers adipogenic differentiation[35], which is largely responsible for the gradual and aging-associated shift of hematopoietic (red) marrows to fatty (yellow) marrows, and which also contributes significantly to the etiology of senile osteoporosis. It is also evident that with increasing donor age, MSCs from both bone marrow and adipose tissues have been shown to have reduced capacity to handle oxidative stress[36-38]. During the aging process, oxidative stress leads to hyperactivity of pro-growth pathways, such as insulin/IGF-1 and mTOR pathways, and the subsequent accumulation of toxic aggregates and cellular debris ultimately lead to apoptosis, necrosis, or autophagy[39]. In addition, in some non-skeletal tissues, particularly the hematopoietic system, MSCs is a key niche component for hematopoietic cells. Aging of MSCs has been shown to be detrimental with respect to this important function[35].
Adult skeletal muscle stem cells (satellite cells) have a remarkable capacity to regenerate[40,41]. Similarly, their regeneration capacity declines with aging, although it is not clear whether this is due to extrinsic changes in the environment and/or to cell-intrinsic mechanisms associated to aging. This impaired regenerative capacity of skeletal muscle during aging is due to accumulation of the altered progeny, which leads to progressive deterioration of tissue structure and function, manifesting after injury or in response to the depletion of memory B cells and naive T cells in the hematopoietic system in the elderly[41-44].
Hematopoietic stem cells (HSCs) are the blood-forming stem cells through the process of hematopoiesis[45]. They are located in the red bone marrow within marrow cavity of most bones. HSCs also produce immune cells of the body. Since blood cells are responsible for constant maintenance and immune protection of every cell type of the body, the constant production of billions of new blood cells each day by HSCs is very important for mammal life. HSC-derived monocytes can give rise to osteoclasts, macrophage and granulocyte. Osteoclasts are giant cells with numerous nuclei that work in synergy with osteoblasts through complicated bone coupling mechanisms to maintain bone homeostasis[35,46]. All these activities of HSCs are carefully modulated by a complex interplay between cell-intrinsic mechanisms and cell-extrinsic factors produced by the microenvironment; and aging altered this fine-tuned regulatory network, leading to aberrant HSC cell cycle regulation, degraded HSC function, and hematological malignancy[47].
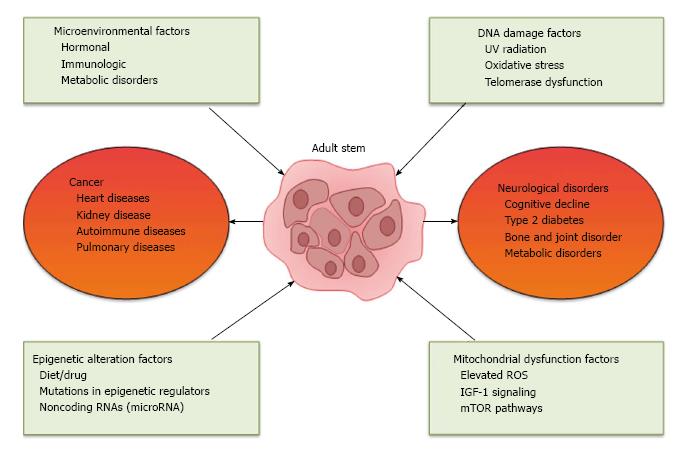
There are several potential mechanisms that are believed to contribute to the aging-associated stem cell dysfunction; and they probably are in part responsible for many aging-associated diseases. Figure 1 proposes some of the contributing factors/mechanisms that could be responsible for the aging-induced deterioration of stem cell functions and aging-associated diseases. This section summarizes some of these contributing factors/mechanisms and their potential roles in the aging effect on stem cells.
Aging is characterized by common environmental conditions, such as hormonal, immunologic, and metabolic disorders[48-50] and these are considered as the critical microenvironmental factors affecting stem cell functions. Changes in these microenvironmental factors in response to aging are believed to be responsible for the changes in stem cell function with aging[51]. It has been shown that potentially underlying aging-related tissue degeneration, such as osteoporosis, could be due to impaired MSCs by surrounding micro-environmental pathologic factors[52,53]. It has also been shown that in mammals, metabolic alterations of hyperglycemia and hyperinsulinemia are important pathologic factors in aging and in MSC dysfunction[54]. However, the molecular mechanism in mediating stem cells dysfunction by microenvironmental signals is not yet fully understood.
Cells produce soluble (endocrine or paracrine) factors necessary for information exchange among cells of distant tissues and/or within the same organ[51]. Aging cells can influence an organ or tissue by secreting soluble endocrine or paracrine factors. Accordingly, aging of the endocrine glands has known to result in hormonal disturbances[50,55], which ultimately affects normal function and or differentiation of the stem cells. In humans, sex hormones, especially estrogen, are the most prominent endocrine factors that change with aging, and sex hormones discordance often leads to several significant diseases. Estrogen insufficiency also induces the biased differentiation of MSCs to adipocytes over osteoblasts[50,56,57]. Aging-related elevation in circulating levels of proinflammatory cytokines, such as interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α), can also cause differentiation disorders of MSCs[58,59].
In mammals, spontaneous and extrinsic mutational events occur on DNA on daily basis. While most of the damaged DNAs are repaired by normal DNA repair mechanism, some of the mutated DNAs appear to escape from the repair mechanism and accumulate over time. Accordingly, there would be a significant accumulation of mutated or damaged DNAs in aging cells compared to young cells. The accumulation of damaged DNA may in part be responsible for the various cellular events of the aging process. In fact, this “mutational theory” is one of the earliest theories of the aging process[15]. DNA damage can be caused by environmental factors, like UV irradiation, and also can be the consequence of the cell’s own metabolic processes [e.g., generating reactive oxygen species (ROS)] that tend to accumulate with time[60]. DNA damage impaired stem-cell function in aging, which has been documented by the study that HSCs derived from aged mice harbored significant alterations in their DNA repair response[1,17]. DNA-repair proteins, such as FANCD1[61], MSH2[62] or ERCC1[63], are found to be deficient in adult mice with significant functional defects of HSCs and the dysfunction of MSCs in aging led to leukemia and aging-associated remodeling[64]. In addition, measures of DNA damage in HSCs, such as histone H2AX phosphorylation and comet tails, were also found to be increased with advancing age[65,66]. In satellite cells, H2AX phosphorylation was also accumulated with increasing age[67].
Premature aging can be resulted from defects in the DNA repair and telomerase pathway components in humans and mice[68]. In aging diseases, there has been significant interest in the telomere shortening that is now being used as a hallmark of aging, to which even stem cells are not immune[1,17]. A telomere is a region of repetitive nucleotide sequences at each end of a chromosome. It protects genome from nucleolytic degradation, unnecessary recombination, repair, or fusion with neighboring chromosomes[69]. Although stem cells express telomerase, the telomeres of HSCs, MSCs, NSCs, HFSCs and GSCs do shorten with age[70-72]. When telomeres become critically short, the cell becomes senescent, it ceases to divide and may undergo apoptosis. In fact, many aging-associated diseases, like the increased cancer risk[73,74], coronary heart disease[75-77], heart failure[78], diabetes[79], and osteoporosis[80], are caused by accelerated telomere shortening. Despite considerable evidence that telomere shortening causes reduction in life span, the telomere shortening concept of aging is still somewhat controversial, since laboratory mice lacking telomerase RNA component (TERC) showed no obvious abnormal phenotypes even after five generations[81,82].
Mitochondria are ubiquitous intracellular organelles in mammals and are the main source of cellular adenosine triphosphate (ATP) that plays a central role in a variety of cellular processes. As mitochondria produce about 90% of cellular energy, the aging-related ROS generation, disruption in Ca2+ homeostasis, and increased cell apoptosis are three causes of mitochondria dysfunction that directly affects aging-related diseases[83]. In fact, there have been many studies suggesting a direct relationship between mitochondrial dysfunction and human stem cell aging[84-87]. Accordingly, in several cell systems, mitochondrial dysfunction has been shown to lead to respiratory chain dysfunction, which may be the result of the accumulation of mutations in mitochondrial DNA (mtDNA)[88]. The elevated ROS in aging is mainly due to mtDNA mutation, as mitochondria is the primary cellular sources of ROS[89]. In addition, it has been confirmed that mitochondrial aging interact with other cellular pathways of aging, such as the IGF-1 signaling and the mTOR pathways, which presumed to play a major role in aging[90,91].
Epigenetics refer to changes in gene expression, which are heritable through modifications without affecting the DNA sequence. It has also been defined more broadly as the dynamic regulation of gene expression by sequence-independent mechanisms, including but not limited to changes in DNA methylation and histone modifications[92-94]. Epigenetic marks in stem cells are transmitted heritably to their daughter cells, priming lineage-specific loci for modification in downstream progenies[95]. Stem cell fates are regulated by epigenetic modifications of DNA that establish the memory of active and silent gene states[96,97]. Aberrant epigenetic regulation affects the organismal aging[98], age-associated dysfunction of stem cells, and predisposition to hematological cancers development[99]. For instance, DNA methylation specific to regions of the genome that are important for lineage-specific gene expression increased in aging HSCs[100] and the perturbations of their histone modifications (H3K4me3) may impair its self-renewal genes[101]. It has also been reported that mutations in epigenetic regulators, such as DNMT3a, TET2, and ASXL1, are frequently found in myeloid neoplasia[102]. Since most of the chromatin changes are intrinsically reversible, epigenetic alterations are therefore considered good therapeutic targets for molecular effectors and thereby are potential therapies for certain distinct pathologies[103,104]. Therefore, there has been immense interest in understanding these genome-scale regulatory mechanisms that lead to impaired gene expression, and that contribute to the decline of stem cell and tissue function with age.
MicroRNAs (miRNAs) are another key class of epigenetic mediators of stem cell dysfunction. They are a class of small noncoding RNAs composed of 18- to 25-bp nucleotides[105] that functions in RNA silencing and post-transcriptional regulation of gene expression[106-109]. It plays an important role in regulating stem cell self-renewal and differentiation by repressing the translation of selected mRNAs in stem cells and differentiating daughter cells[110]. In fact, non-coding RNA-mediated regulatory events as a part of the epigenetic mechanism to modulate mRNA degradation and/or protein translation that play important role in development and disease state[111]. MiRNAs, such as miR-17, regulates osteoblast differentiation of MSCs[112-114]. MiR-290–295 cluster seems to promote embryonic stem cell differentiation, self-renewal, and maintenance of pluripotency[110,115]. Moreover, recent findings show the involvement of miRNAs in senescence manipulation. These findings have led to the suggested use of these miRNAs as clinical biomarkers of stem cell senescence and their potentiality[116].
In recent years with increasing understanding of stem cell behavior in different niche of the body offers promise for the development of potential therapeutic approaches to treat aging-associated dysregulation of adult stem cells and aging-related diseases. Some of the potential therapeutic approaches for the treatment of age-related stem cell dysfunction are discussed below.
The concept of parabiosis is not new; however, in the past decade its role in reversing the effects of aging and enhancing rejuvenation has gathered substantial momentum. Recent findings suggest that aging-related cellular dysfunctions can be repaired successfully by modulating the molecular architecture of the tissue environment rather than inducing cell intrinsic changes alone[117]. Therefore, the effects of aging in an old individual can be modulated or reversed by the circulatory or systemic factors derived from the young blood through anatomical joining, parabiosis[40]. The fascinating results of parabiosis have been reported to rejenuvate brain[118], muscles[67], and liver tissues in the aged animals[119]. In skeletal muscle regeneration, serum derived from young mice activated the Notch signaling pathway and regulated the satellite cells proliferation of old mice in vitro[119]. In aged mice, through the parabiosis approach, systemic factors from young mice successfully reversed inefficient CNS remyelination, a regenerative process of CNS that produces new myelin sheaths from adult stem cells[118]. Despite the promising outcomes in animal models, there is persistence of contradiction in functions of factors identified in prominent parabiosis studies, rendering the concept highly controversial for use in humans. For instance, growth differentiation factor 11 (GDF-11) has been reported to show both positive[67] and negative corelations[120] with stem cell aging.
Retrotansposons are mobile DNA elements that can induce genetic instability and have been reported to be a cause of cellular dysfunction during aging[121]. The long interspaced nuclear elements (L1) are 6-kb long retrotransposons that code for RNA binding protein and endonuclease protein. There are 500000 copies of L1 elements in the human genome, and approximately 100 of such active elements replicated to induce genomic instabilities and to increase the risk of DNA damage. Elevated activity of L1 has been reported in aging-related pathological conditions[122]. The link between SIRT-6 (an important marker of longevity) and L1 offered more direct evidence for the role of L1 in aging-related genomic complications. SIRT6 are known to repress the activity of L1 retrotransposons[123]. DNA damage-induced mobilization of SIRT6 to the site of repair and subsequent repression of L1 have been contemplated in the development of therapeutics for age-related neurological pathologies, such as dementia and cancer[124]. Suppression of L1 activity by overexpression of SIRT6 in senescence cells delayed the onset of L1-induced pathological conditions. High caloric diet activated the SIRT1 activity and has been reported to protect the animal from premature aging in Cockayne syndrome[125], whereas in the case of the mouse Alzheimer’s disease model the caloric restriction slowed down the disease progression[126]. Other than modulation of SIRT6 expression, inhibition of reverse transcriptase (a critical enzyme for the L1 replication) is another way to attenuate L1 activity[127]. Several small non-coding RNAs, such as pi-RNAs, si-RNAs and L1 specific small RNAs, have also been reported to regulate the silencing of retrotransposons element activity in mouse germ cells and in aging human somatic cells[127].
iPSCs are a type of pluripotent stem cell that can be generated directly from adult cells and the recent advances in this area have opened up many gateways for the research in cell-based therapeutics[128]. Cellular reprogramming of aged somatic cells towards iPSC enables the editing and resetting of the cellular clock by removing the characteristic feature of aging. The ability to derive iPSCs from aging-related pathological cells have enabled investigators to develop recombination-based therapeutic approaches to edit genetic defects responsible of premature and accelerated aging. The reprogramming of aged somatic cells to target stem can be used as an alternative source to get cells for transplantation and for genetic editing. Recent studies show encouraging effects of reprogramming in rejuvenation of senescent cells, as evident by elongated telomeres and reduced oxidative stress[129]. Human iPSC-based models for aging-related degenerative diseases have been tested to understand the disease dynamics in Parkinson’s disease,
Alzheimer’s disease and in progeroid laminopathies[130]. Valuable information from these studies has resulted in the first clinical trial for progeroid patients[131]. In a mouse model of skeletal defect, human iPSC designed to express PAX7 were able to be differentiated into muscle progenitor cells that engrafted and repaired the defective dystrophin-positive myofibers formation. In case of Hutchinson-Gilford progeria syndrome (HGPS), reprogramming of HGPS fibroblasts by transduction with vectors expressing Oct4, Sox2, Klf4 and c-Myc has been reported to revert aging-associated markers, such as Lamin, to a “young” state[132].
As discussed above, the telomere length is inversely linked to the chronical age, and thus it is believed that increasing the length of telomere may increase life span. Many advanced approaches are being developed to efficiently increase the telomere length and to protect cells from chromosome shortening. In in vitro cultured human cells, the delivery of RNA coding for telomere-extending protein has been reported to increase the cell proliferation rate[133]. In telomere-deficient mice, genetic editing to reactivate telomerase activity has been reported to reverse the aging symptoms[134]. Telomerase activation drugs and telomerase gene therapy are also alternative approaches that aim to increase the telomere length to protect the cells from premature aging[135,136].
From the various advances in stem cell research, it is clear that we grow old partly because our stem cells grow old with us. The functions of aged stem cells become impaired as the result of cell-intrinsic pathways and surrounding environmental changes. With the sharp rise in the aging-associated diseases, the need for effective regenerative medicine strategies for the aged is more important than ever. Fortunately, rapid advances in stem cell and regenerative medicine technologies continue to provide us with a better understanding of the diseases that allows us to develop more effective therapies and diagnostic technologies to better treat aged patients. However, there is a big ethical concern regarding the use of human embryos to procure embryonic stem cells and many countries already currently restrict experiments on embryos to the first 14 d. Additionally, the International Society for Stem Cell Research has issued guidelines advising researchers across the globe to stick with this 14-d window. Nevertheless, it seems that the human stem cell research in the next decade will likely bring enormous progress in the aging-associated disease therapies but may also reach a step closer to the edge of ethical concern of creation of “Frankenstein”.
Manuscript source: Invited manuscript
Specialty type: Medicine, research and experimental
Country of origin: United States
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Jun Y, Kiselev SL, Zaminy A S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Sharpless NE, DePinho RA. How stem cells age and why this makes us grow old. Nat Rev Mol Cell Biol. 2007;8:703-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 654] [Cited by in RCA: 626] [Article Influence: 34.8] [Reference Citation Analysis (1)] |
| 2. | Dillin A, Gottschling DE, Nyström T. The good and the bad of being connected: the integrons of aging. Curr Opin Cell Biol. 2014;26:107-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 3. | Shane Anderson A, Loeser RF. Why is osteoarthritis an age-related disease? Best Pract Res Clin Rheumatol. 2010;24:15-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 418] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 4. | Reeve A, Simcox E, Turnbull D. Ageing and Parkinson’s disease: why is advancing age the biggest risk factor? Ageing Res Rev. 2014;14:19-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 489] [Cited by in RCA: 607] [Article Influence: 55.2] [Reference Citation Analysis (0)] |
| 5. | Niccoli T, Partridge L. Ageing as a risk factor for disease. Curr Biol. 2012;22:R741-R752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 852] [Cited by in RCA: 1138] [Article Influence: 94.8] [Reference Citation Analysis (0)] |
| 6. | De Grey AD. Life span extension research and public debate: societal considerations. Studies in Ethics, Law, and Technology. 2007;1:1-10. [RCA] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 7. | Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145-1147. [PubMed] |
| 8. | Vogel G. Breakthrough of the year. Capturing the promise of youth. Science. 1999;286:2238-2239. [PubMed] |
| 9. | Biehl JK, Russell B. Introduction to stem cell therapy. J Cardiovasc Nurs. 2009;24:98-103; quiz 104-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 165] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 10. | Marchetto MC, Gage FH. Your brain under the microscope: the promise of stem cells. Cerebrum. 2014;2014:1. [PubMed] |
| 11. | Jones DL, Rando TA. Emerging models and paradigms for stem cell ageing. Nat Cell Biol. 2011;13:506-512. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 177] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 12. | Oh J, Lee YD, Wagers AJ. Stem cell aging: mechanisms, regulators and therapeutic opportunities. Nat Med. 2014;20:870-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 554] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 13. | Kirkwood TB. Understanding the odd science of aging. Cell. 2005;120:437-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1261] [Cited by in RCA: 1253] [Article Influence: 62.7] [Reference Citation Analysis (0)] |
| 14. | Blagosklonny MV. Revisiting the antagonistic pleiotropy theory of aging: TOR-driven program and quasi-program. Cell Cycle. 2010;9:3151-3156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 119] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 15. | Medawar P. An Unsolved Problem in Biology. Lewis, London. Reprinted in Medawar PB (1981). The Uniqueness of the Individual. New York: Dover 1952; . |
| 16. | Boyette LB, Tuan RS. Adult Stem Cells and Diseases of Aging. J Clin Med. 2014;3:88-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 17. | Schultz MB, Sinclair DA. When stem cells grow old: phenotypes and mechanisms of stem cell aging. Development. 2016;143:3-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 217] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 18. | Ho AD, Wagner W, Mahlknecht U. Stem cells and ageing. The potential of stem cells to overcome age-related deteriorations of the body in regenerative medicine. EMBO Rep. 2005;6 Spec No:S35-S38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 60] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Sousounis K, Baddour JA, Tsonis PA. Aging and regeneration in vertebrates. Curr Top Dev Biol. 2014;108:217-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 20. | Paxson JA, Gruntman A, Parkin CD, Mazan MR, Davis A, Ingenito EP, Hoffman AM. Age-dependent decline in mouse lung regeneration with loss of lung fibroblast clonogenicity and increased myofibroblastic differentiation. PLoS One. 2011;6:e23232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 21. | Keller K, Engelhardt M. Strength and muscle mass loss with aging process. Age and strength loss. Muscles Ligaments Tendons J. 2013;3:346-350. [PubMed] |
| 22. | Wagner W, Bork S, Horn P, Krunic D, Walenda T, Diehlmann A, Benes V, Blake J, Huber FX, Eckstein V. Aging and replicative senescence have related effects on human stem and progenitor cells. PLoS One. 2009;4:e5846. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 332] [Cited by in RCA: 375] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 23. | Mansilla E, Díaz Aquino V, Zambón D, Marin GH, Mártire K, Roque G, Ichim T, Riordan NH, Patel A, Sturla F. Could metabolic syndrome, lipodystrophy, and aging be mesenchymal stem cell exhaustion syndromes? Stem Cells Int. 2011;2011:943216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 24. | Alenzi FQ, Bahkali AH. Stem cells: Biology and clinical potential. Afr J Biotechnol. 2011;10:19929-19940. |
| 25. | Zhu L, Dong C, Sun C, Ma R, Yang D, Zhu H, Xu J. Rejuvenation of MPTP-induced human neural precursor cell senescence by activating autophagy. Biochem Biophys Res Commun. 2015;464:526-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Winner B, Kohl Z, Gage FH. Neurodegenerative disease and adult neurogenesis. Eur J Neurosci. 2011;33:1139-1151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 328] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 27. | Enwere E, Shingo T, Gregg C, Fujikawa H, Ohta S, Weiss S. Aging results in reduced epidermal growth factor receptor signaling, diminished olfactory neurogenesis, and deficits in fine olfactory discrimination. J Neurosci. 2004;24:8354-8365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 389] [Cited by in RCA: 427] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 28. | Ming GL, Song H. Adult neurogenesis in the mammalian brain: significant answers and significant questions. Neuron. 2011;70:687-702. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2119] [Cited by in RCA: 1971] [Article Influence: 140.8] [Reference Citation Analysis (0)] |
| 29. | Brighton CT, Hunt RM. Early histological and ultrastructural changes in medullary fracture callus. J Bone Joint Surg Am. 1991;73:832-847. [PubMed] |
| 30. | Brighton CT, Hunt RM. Early histologic and ultrastructural changes in microvessels of periosteal callus. J Orthop Trauma. 1997;11:244-253. [PubMed] |
| 31. | Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143-147. [PubMed] |
| 32. | Lin F. Adipose tissue-derived mesenchymal stem cells: a fat chance of curing kidney disease? Kidney Int. 2012;82:731-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 33. | Ma L, Aijima R, Hoshino Y, Yamaza H, Tomoda E, Tanaka Y, Sonoda S, Song G, Zhao W, Nonaka K. Transplantation of mesenchymal stem cells ameliorates secondary osteoporosis through interleukin-17-impaired functions of recipient bone marrow mesenchymal stem cells in MRL/lpr mice. Stem Cell Res Ther. 2015;6:104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 34. | Jones E, Schäfer R. Where is the common ground between bone marrow mesenchymal stem/stromal cells from different donors and species? Stem Cell Res Ther. 2015;6:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 35. | Liu H, Xia X, Li B. Mesenchymal stem cell aging: Mechanisms and influences on skeletal and non-skeletal tissues. Exp Biol Med (Maywood). 2015;240:1099-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 71] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 36. | De Barros S, Dehez S, Arnaud E, Barreau C, Cazavet A, Perez G, Galinier A, Casteilla L, Planat-Bénard V. Aging-related decrease of human ASC angiogenic potential is reversed by hypoxia preconditioning through ROS production. Mol Ther. 2013;21:399-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 37. | Sethe S, Scutt A, Stolzing A. Aging of mesenchymal stem cells. Ageing Res Rev. 2006;5:91-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 469] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 38. | Stolzing A, Jones E, McGonagle D, Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mech Ageing Dev. 2008;129:163-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 821] [Cited by in RCA: 943] [Article Influence: 52.4] [Reference Citation Analysis (0)] |
| 39. | Haines DD, Juhasz B, Tosaki A. Management of multicellular senescence and oxidative stress. J Cell Mol Med. 2013;17:936-957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 40. | Brack AS, Muñoz-Cánoves P. The ins and outs of muscle stem cell aging. Skelet Muscle. 2016;6:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 109] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 41. | García-Prat L, Sousa-Victor P, Muñoz-Cánoves P. Functional dysregulation of stem cells during aging: a focus on skeletal muscle stem cells. FEBS J. 2013;280:4051-4062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 108] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 42. | Shefer G, Wleklinski-Lee M, Yablonka-Reuveni Z. Skeletal muscle satellite cells can spontaneously enter an alternative mesenchymal pathway. J Cell Sci. 2004;117:5393-5404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 232] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 43. | Taylor-Jones JM, McGehee RE, Rando TA, Lecka-Czernik B, Lipschitz DA, Peterson CA. Activation of an adipogenic program in adult myoblasts with age. Mech Ageing Dev. 2002;123:649-661. [PubMed] |
| 44. | Brack AS, Conboy MJ, Roy S, Lee M, Kuo CJ, Keller C, Rando TA. Increased Wnt signaling during aging alters muscle stem cell fate and increases fibrosis. Science. 2007;317:807-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1084] [Cited by in RCA: 1162] [Article Influence: 64.6] [Reference Citation Analysis (0)] |
| 45. | Birbrair A, Frenette PS. Niche heterogeneity in the bone marrow. Ann N Y Acad Sci. 2016;1370:82-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 209] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 46. | Henriksen K, Karsdal MA, Martin TJ. Osteoclast-derived coupling factors in bone remodeling. Calcif Tissue Int. 2014;94:88-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 109] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 47. | Pietras EM, Warr MR, Passegué E. Cell cycle regulation in hematopoietic stem cells. J Cell Biol. 2011;195:709-720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 324] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 48. | Fontana L, Partridge L, Longo VD. Extending healthy life span--from yeast to humans. Science. 2010;328:321-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2249] [Cited by in RCA: 2179] [Article Influence: 145.3] [Reference Citation Analysis (0)] |
| 49. | Abdelmagid SA, Clarke SE, Roke K, Nielsen DE, Badawi A, El-Sohemy A, Mutch DM, Ma DW. Ethnicity, sex, FADS genetic variation, and hormonal contraceptive use influence delta-5- and delta-6-desaturase indices and plasma docosahexaenoic acid concentration in young Canadian adults: a cross-sectional study. Nutr Metab (Lond). 2015;12:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 50. | Benayoun BA, Pollina EA, Brunet A. Epigenetic regulation of ageing: linking environmental inputs to genomic stability. Nat Rev Mol Cell Biol. 2015;16:593-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 394] [Cited by in RCA: 413] [Article Influence: 41.3] [Reference Citation Analysis (0)] |
| 51. | Sui BD, Hu CH, Zheng CX, Jin Y. Microenvironmental Views on Mesenchymal Stem Cell Differentiation in Aging. J Dent Res. 2016;pii:0022034516653589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 88] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 52. | Kfoury Y, Scadden DT. Mesenchymal cell contributions to the stem cell niche. Cell Stem Cell. 2015;16:239-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 402] [Article Influence: 44.7] [Reference Citation Analysis (0)] |
| 53. | Li CY, Wu XY, Tong JB, Yang XX, Zhao JL, Zheng QF, Zhao GB, Ma ZJ. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Res Ther. 2015;6:55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 230] [Cited by in RCA: 295] [Article Influence: 29.5] [Reference Citation Analysis (0)] |
| 54. | Anisimov VN, Bartke A. The key role of growth hormone-insulin-IGF-1 signaling in aging and cancer. Crit Rev Oncol Hematol. 2013;87:201-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 156] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 55. | Straub RH, Cutolo M, Zietz B, Schölmerich J. The process of aging changes the interplay of the immune, endocrine and nervous systems. Mech Ageing Dev. 2001;122:1591-1611. [PubMed] |
| 56. | Emmerson E, Hardman MJ. The role of estrogen deficiency in skin ageing and wound healing. Biogerontology. 2012;13:3-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 57. | Liao L, Yang X, Su X, Hu C, Zhu X, Yang N, Chen X, Shi S, Shi S, Jin Y. Redundant miR-3077-5p and miR-705 mediate the shift of mesenchymal stem cell lineage commitment to adipocyte in osteoporosis bone marrow. Cell Death Dis. 2013;4:e600. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 107] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 58. | Abdelmagid SM, Barbe MF, Safadi FF. Role of inflammation in the aging bones. Life Sci. 2015;123:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 93] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 59. | Pawelec G, Goldeck D, Derhovanessian E. Inflammation, ageing and chronic disease. Curr Opin Immunol. 2014;29:23-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 293] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 60. | Lombard DB, Chua KF, Mostoslavsky R, Franco S, Gostissa M, Alt FW. DNA repair, genome stability, and aging. Cell. 2005;120:497-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 647] [Cited by in RCA: 683] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 61. | Navarro S, Meza NW, Quintana-Bustamante O, Casado JA, Jacome A, McAllister K, Puerto S, Surrallés J, Segovia JC, Bueren JA. Hematopoietic dysfunction in a mouse model for Fanconi anemia group D1. Mol Ther. 2006;14:525-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 62. | Reese JS, Liu L, Gerson SL. Repopulating defect of mismatch repair-deficient hematopoietic stem cells. Blood. 2003;102:1626-1633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 72] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 63. | Prasher JM, Lalai AS, Heijmans-Antonissen C, Ploemacher RE, Hoeijmakers JH, Touw IP, Niedernhofer LJ. Reduced hematopoietic reserves in DNA interstrand crosslink repair-deficient Ercc1-/- mice. EMBO J. 2005;24:861-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 109] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 64. | Moehrle BM, Geiger H. Aging of hematopoietic stem cells: DNA damage and mutations? Exp Hematol. 2016;44:895-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 65] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 65. | Beerman I, Seita J, Inlay MA, Weissman IL, Rossi DJ. Quiescent hematopoietic stem cells accumulate DNA damage during aging that is repaired upon entry into cell cycle. Cell Stem Cell. 2014;15:37-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 345] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 66. | Rübe CE, Fricke A, Widmann TA, Fürst T, Madry H, Pfreundschuh M, Rübe C. Accumulation of DNA damage in hematopoietic stem and progenitor cells during human aging. PLoS One. 2011;6:e17487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 227] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 67. | Sinha M, Jang YC, Oh J, Khong D, Wu EY, Manohar R, Miller C, Regalado SG, Loffredo FS, Pancoast JR. Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science. 2014;344:649-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 575] [Cited by in RCA: 663] [Article Influence: 60.3] [Reference Citation Analysis (0)] |
| 68. | Schumacher B, Garinis GA, Hoeijmakers JH. Age to survive: DNA damage and aging. Trends Genet. 2008;24:77-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 186] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 69. | Shammas MA. Telomeres, lifestyle, cancer, and aging. Curr Opin Clin Nutr Metab Care. 2011;14:28-34. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 518] [Cited by in RCA: 435] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 70. | Bonab MM, Alimoghaddam K, Talebian F, Ghaffari SH, Ghavamzadeh A, Nikbin B. Aging of mesenchymal stem cell in vitro. BMC Cell Biol. 2006;7:14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 71. | Ferrón SR, Marqués-Torrejón MA, Mira H, Flores I, Taylor K, Blasco MA, Fariñas I. Telomere shortening in neural stem cells disrupts neuronal differentiation and neuritogenesis. J Neurosci. 2009;29:14394-14407. [PubMed] [DOI] [Full Text] |
| 72. | Flores I, Canela A, Vera E, Tejera A, Cotsarelis G, Blasco MA. The longest telomeres: a general signature of adult stem cell compartments. Genes Dev. 2008;22:654-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 268] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 73. | Wu X, Amos CI, Zhu Y, Zhao H, Grossman BH, Shay JW, Luo S, Hong WK, Spitz MR. Telomere dysfunction: a potential cancer predisposition factor. J Natl Cancer Inst. 2003;95:1211-1218. [PubMed] |
| 74. | McGrath M, Wong JY, Michaud D, Hunter DJ, De Vivo I. Telomere length, cigarette smoking, and bladder cancer risk in men and women. Cancer Epidemiol Biomarkers Prev. 2007;16:815-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 275] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 75. | Fitzpatrick AL, Kronmal RA, Gardner JP, Psaty BM, Jenny NS, Tracy RP, Walston J, Kimura M, Aviv A. Leukocyte telomere length and cardiovascular disease in the cardiovascular health study. Am J Epidemiol. 2007;165:14-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 590] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 76. | Brouilette SW, Moore JS, McMahon AD, Thompson JR, Ford I, Shepherd J, Packard CJ, Samani NJ. Telomere length, risk of coronary heart disease, and statin treatment in the West of Scotland Primary Prevention Study: a nested case-control study. Lancet. 2007;369:107-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 556] [Cited by in RCA: 562] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 77. | Zee RY, Michaud SE, Germer S, Ridker PM. Association of shorter mean telomere length with risk of incident myocardial infarction: a prospective, nested case-control approach. Clin Chim Acta. 2009;403:139-141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 68] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 78. | van der Harst P, van der Steege G, de Boer RA, Voors AA, Hall AS, Mulder MJ, van Gilst WH, van Veldhuisen DJ. Telomere length of circulating leukocytes is decreased in patients with chronic heart failure. J Am Coll Cardiol. 2007;49:1459-1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 211] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 79. | Sampson MJ, Winterbone MS, Hughes JC, Dozio N, Hughes DA. Monocyte telomere shortening and oxidative DNA damage in type 2 diabetes. Diabetes Care. 2006;29:283-289. [PubMed] |
| 80. | Valdes AM, Richards JB, Gardner JP, Swaminathan R, Kimura M, Xiaobin L, Aviv A, Spector TD. Telomere length in leukocytes correlates with bone mineral density and is shorter in women with osteoporosis. Osteoporos Int. 2007;18:1203-1210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 120] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 81. | Ju Z, Jiang H, Jaworski M, Rathinam C, Gompf A, Klein C, Trumpp A, Rudolph KL. Telomere dysfunction induces environmental alterations limiting hematopoietic stem cell function and engraftment. Nat Med. 2007;13:742-747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 219] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 82. | Lee HW, Blasco MA, Gottlieb GJ, Horner JW, Greider CW, DePinho RA. Essential role of mouse telomerase in highly proliferative organs. Nature. 1998;392:569-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 963] [Cited by in RCA: 958] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 83. | Kujoth GC, Hiona A, Pugh TD, Someya S, Panzer K, Wohlgemuth SE, Hofer T, Seo AY, Sullivan R, Jobling WA. Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science. 2005;309:481-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1492] [Cited by in RCA: 1611] [Article Influence: 80.6] [Reference Citation Analysis (0)] |
| 84. | Bratic A, Larsson NG. The role of mitochondria in aging. J Clin Invest. 2013;123:951-957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 672] [Cited by in RCA: 780] [Article Influence: 65.0] [Reference Citation Analysis (0)] |
| 85. | Taylor RW, Barron MJ, Borthwick GM, Gospel A, Chinnery PF, Samuels DC, Taylor GA, Plusa SM, Needham SJ, Greaves LC. Mitochondrial DNA mutations in human colonic crypt stem cells. J Clin Invest. 2003;112:1351-1360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 380] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 86. | McDonald SA, Greaves LC, Gutierrez-Gonzalez L, Rodriguez-Justo M, Deheragoda M, Leedham SJ, Taylor RW, Lee CY, Preston SL, Lovell M. Mechanisms of field cancerization in the human stomach: the expansion and spread of mutated gastric stem cells. Gastroenterology. 2008;134:500-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 192] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 87. | Fellous TG, Islam S, Tadrous PJ, Elia G, Kocher HM, Bhattacharya S, Mears L, Turnbull DM, Taylor RW, Greaves LC. Locating the stem cell niche and tracing hepatocyte lineages in human liver. Hepatology. 2009;49:1655-1663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 88. | Miquel J, Economos AC, Fleming J, Johnson JE. Mitochondrial role in cell aging. Exp Gerontol. 1980;15:575-591. [PubMed] |
| 89. | Pervaiz S, Taneja R, Ghaffari S. Oxidative stress regulation of stem and progenitor cells. Antioxid Redox Signal. 2009;11:2777-2789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 142] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 90. | Bonawitz ND, Chatenay-Lapointe M, Pan Y, Shadel GS. Reduced TOR signaling extends chronological life span via increased respiration and upregulation of mitochondrial gene expression. Cell Metab. 2007;5:265-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 327] [Cited by in RCA: 333] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 91. | Choi CS, Befroy DE, Codella R, Kim S, Reznick RM, Hwang YJ, Liu ZX, Lee HY, Distefano A, Samuel VT. Paradoxical effects of increased expression of PGC-1alpha on muscle mitochondrial function and insulin-stimulated muscle glucose metabolism. Proc Natl Acad Sci USA. 2008;105:19926-19931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 249] [Cited by in RCA: 232] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 92. | Jaenisch R, Bird A. Epigenetic regulation of gene expression: how the genome integrates intrinsic and environmental signals. Nat Genet. 2003;33 Suppl:245-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4367] [Cited by in RCA: 4245] [Article Influence: 193.0] [Reference Citation Analysis (0)] |
| 93. | Vaquero A, Loyola A, Reinberg D. The constantly changing face of chromatin. Sci Aging Knowledge Environ. 2003;2003:RE4. [PubMed] |
| 94. | Ma DK, Marchetto MC, Guo JU, Ming GL, Gage FH, Song H. Epigenetic choreographers of neurogenesis in the adult mammalian brain. Nat Neurosci. 2010;13:1338-1344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 281] [Cited by in RCA: 251] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 95. | Beerman I, Rossi DJ. Epigenetic Control of Stem Cell Potential during Homeostasis, Aging, and Disease. Cell Stem Cell. 2015;16:613-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 138] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 96. | Borrelli E, Nestler EJ, Allis CD, Sassone-Corsi P. Decoding the epigenetic language of neuronal plasticity. Neuron. 2008;60:961-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 419] [Cited by in RCA: 373] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 97. | Orkin SH, Hochedlinger K. Chromatin connections to pluripotency and cellular reprogramming. Cell. 2011;145:835-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 312] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 98. | Goodell MA, Rando TA. Stem cells and healthy aging. Science. 2015;350:1199-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 203] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 99. | Buscarlet M, Tessier A, Provost S, Mollica L, Busque L. Human blood cell levels of 5-hydroxymethylcytosine (5hmC) decline with age, partly related to acquired mutations in TET2. Exp Hematol. 2016;44:1072-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 100. | Beerman I, Bock C, Garrison BS, Smith ZD, Gu H, Meissner A, Rossi DJ. Proliferation-dependent alterations of the DNA methylation landscape underlie hematopoietic stem cell aging. Cell Stem Cell. 2013;12:413-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 324] [Cited by in RCA: 358] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 101. | Sun D, Luo M, Jeong M, Rodriguez B, Xia Z, Hannah R, Wang H, Le T, Faull KF, Chen R. Epigenomic profiling of young and aged HSCs reveals concerted changes during aging that reinforce self-renewal. Cell Stem Cell. 2014;14:673-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 427] [Cited by in RCA: 503] [Article Influence: 45.7] [Reference Citation Analysis (0)] |
| 102. | Shih AH, Abdel-Wahab O, Patel JP, Levine RL. The role of mutations in epigenetic regulators in myeloid malignancies. Nat Rev Cancer. 2012;12:599-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 512] [Cited by in RCA: 553] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 103. | Zhou Y, Kim J, Yuan X, Braun T. Epigenetic modifications of stem cells: a paradigm for the control of cardiac progenitor cells. Circ Res. 2011;109:1067-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 104. | García-Prat L, Muñoz-Cánoves P. Aging, metabolism and stem cells: Spotlight on muscle stem cells. Mol Cell Endocrinol. 2016;pii:S0303-7207(16)30315-X. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 105. | Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 106. | Ambros V. The functions of animal microRNAs. Nature. 2004;431:350-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7919] [Cited by in RCA: 8601] [Article Influence: 409.6] [Reference Citation Analysis (0)] |
| 107. | Xu F, Ahmed AS, Kang X, Hu G, Liu F, Zhang W, Zhou J. MicroRNA-15b/16 Attenuates Vascular Neointima Formation by Promoting the Contractile Phenotype of Vascular Smooth Muscle Through Targeting YAP. Arterioscler Thromb Vasc Biol. 2015;35:2145-2152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 69] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 108. | Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281-297. [PubMed] |
| 109. | Heinrich EM, Dimmeler S. MicroRNAs and stem cells: control of pluripotency, reprogramming, and lineage commitment. Circ Res. 2012;110:1014-1022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 110. | Gangaraju VK, Lin H. MicroRNAs: key regulators of stem cells. Nat Rev Mol Cell Biol. 2009;10:116-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 525] [Cited by in RCA: 559] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 111. | Dimmeler S, Losordo D. Stem cells review series: an introduction. Circ Res. 2011;109:907-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 112. | Liu Y, Liu W, Hu C, Xue Z, Wang G, Ding B, Luo H, Tang L, Kong X, Chen X. MiR-17 modulates osteogenic differentiation through a coherent feed-forward loop in mesenchymal stem cells isolated from periodontal ligaments of patients with periodontitis. Stem Cells. 2011;29:1804-1816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 149] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 113. | Jia J, Feng X, Xu W, Yang S, Zhang Q, Liu X, Feng Y, Dai Z. MiR-17-5p modulates osteoblastic differentiation and cell proliferation by targeting SMAD7 in non-traumatic osteonecrosis. Exp Mol Med. 2014;46:e107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 91] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 114. | Liu W, Qi M, Konermann A, Zhang L, Jin F, Jin Y. The p53/miR-17/Smurf1 pathway mediates skeletal deformities in an age-related model via inhibiting the function of mesenchymal stem cells. Aging (Albany NY). 2015;7:205-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 115. | Houbaviy HB, Murray MF, Sharp PA. Embryonic stem cell-specific MicroRNAs. Dev Cell. 2003;5:351-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 835] [Cited by in RCA: 824] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 116. | Bilsland AE, Revie J, Keith W. MicroRNA and senescence: the senectome, integration and distributed control. Crit Rev Oncog. 2013;18:373-390. [PubMed] [DOI] [Full Text] |
| 117. | Eggel A, Wyss-Coray T. A revival of parabiosis in biomedical research. Swiss Med Wkly. 2014;144:w13914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 118. | Ruckh JM, Zhao JW, Shadrach JL, van Wijngaarden P, Rao TN, Wagers AJ, Franklin RJ. Rejuvenation of regeneration in the aging central nervous system. Cell Stem Cell. 2012;10:96-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 525] [Article Influence: 40.4] [Reference Citation Analysis (0)] |
| 119. | Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature. 2005;433:760-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1544] [Cited by in RCA: 1639] [Article Influence: 82.0] [Reference Citation Analysis (0)] |
| 120. | Egerman MA, Cadena SM, Gilbert JA, Meyer A, Nelson HN, Swalley SE, Mallozzi C, Jacobi C, Jennings LL, Clay I. GDF11 Increases with Age and Inhibits Skeletal Muscle Regeneration. Cell Metab. 2015;22:164-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 429] [Article Influence: 42.9] [Reference Citation Analysis (0)] |
| 121. | De Cecco M, Criscione SW, Peterson AL, Neretti N, Sedivy JM, Kreiling JA. Transposable elements become active and mobile in the genomes of aging mammalian somatic tissues. Aging (Albany NY). 2013;5:867-883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 249] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 122. | St Laurent G, Hammell N, McCaffrey TA. A LINE-1 component to human aging: do LINE elements exact a longevity cost for evolutionary advantage? Mech Ageing Dev. 2010;131:299-305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 123. | Van Meter M, Kashyap M, Rezazadeh S, Geneva AJ, Morello TD, Seluanov A, Gorbunova V. SIRT6 represses LINE1 retrotransposons by ribosylating KAP1 but this repression fails with stress and age. Nat Commun. 2014;5:5011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 257] [Cited by in RCA: 291] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 124. | Coufal NG, Garcia-Perez JL, Peng GE, Yeo GW, Mu Y, Lovci MT, Morell M, O’Shea KS, Moran JV, Gage FH. L1 retrotransposition in human neural progenitor cells. Nature. 2009;460:1127-1131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 711] [Cited by in RCA: 637] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 125. | Scheibye-Knudsen M, Mitchell SJ, Fang EF, Iyama T, Ward T, Wang J, Dunn CA, Singh N, Veith S, Hasan-Olive MM. A high-fat diet and NAD(+) activate Sirt1 to rescue premature aging in cockayne syndrome. Cell Metab. 2014;20:840-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 259] [Cited by in RCA: 291] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 126. | Braidy N, Jayasena T, Poljak A, Sachdev PS. Sirtuins in cognitive ageing and Alzheimer‘s disease. Curr Opin Psychiatry. 2012;25:226-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 127. | Goodier JL. Restricting retrotransposons: a review. Mob DNA. 2016;7:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 325] [Cited by in RCA: 290] [Article Influence: 32.2] [Reference Citation Analysis (0)] |
| 128. | Singh VK, Kalsan M, Kumar N, Saini A, Chandra R. Induced pluripotent stem cells: applications in regenerative medicine, disease modeling, and drug discovery. Front Cell Dev Biol. 2015;3:2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 233] [Cited by in RCA: 267] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 129. | Freije JM, López-Otín C. Reprogramming aging and progeria. Curr Opin Cell Biol. 2012;24:757-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 130. | Liu GH, Barkho BZ, Ruiz S, Diep D, Qu J, Yang SL, Panopoulos AD, Suzuki K, Kurian L, Walsh C. Recapitulation of premature ageing with iPSCs from Hutchinson-Gilford progeria syndrome. Nature. 2011;472:221-225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 486] [Cited by in RCA: 442] [Article Influence: 31.6] [Reference Citation Analysis (0)] |
| 131. | Gordon LB, Rothman FG, López-Otín C, Misteli T. Progeria: a paradigm for translational medicine. Cell. 2014;156:400-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 206] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 132. | Miller JD, Ganat YM, Kishinevsky S, Bowman RL, Liu B, Tu EY, Mandal PK, Vera E, Shim JW, Kriks S. Human iPSC-based modeling of late-onset disease via progerin-induced aging. Cell Stem Cell. 2013;13:691-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 614] [Cited by in RCA: 576] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 133. | Ramunas J, Yakubov E, Brady JJ, Corbel SY, Holbrook C, Brandt M, Stein J, Santiago JG, Cooke JP, Blau HM. Transient delivery of modified mRNA encoding TERT rapidly extends telomeres in human cells. FASEB J. 2015;29:1930-1939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 134. | Jaskelioff M, Muller FL, Paik JH, Thomas E, Jiang S, Adams AC, Sahin E, Kost-Alimova M, Protopopov A, Cadiñanos J. Telomerase reactivation reverses tissue degeneration in aged telomerase-deficient mice. Nature. 2011;469:102-106. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 650] [Cited by in RCA: 570] [Article Influence: 40.7] [Reference Citation Analysis (0)] |
| 135. | Bernardes de Jesus B, Schneeberger K, Vera E, Tejera A, Harley CB, Blasco MA. The telomerase activator TA-65 elongates short telomeres and increases health span of adult/old mice without increasing cancer incidence. Aging Cell. 2011;10:604-621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 227] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 136. | Bernardes de Jesus B, Vera E, Schneeberger K, Tejera AM, Ayuso E, Bosch F, Blasco MA. Telomerase gene therapy in adult and old mice delays aging and increases longevity without increasing cancer. EMBO Mol Med. 2012;4:691-704. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 337] [Article Influence: 25.9] [Reference Citation Analysis (0)] |