Published online May 20, 2015. doi: 10.5493/wjem.v5.i2.110
Peer-review started: October 5, 2014
First decision: December 12, 2014
Revised: January 28, 2015
Accepted: February 4, 2015
Article in press: February 9, 2015
Published online: May 20, 2015
Processing time: 228 Days and 21.4 Hours
Cardiovascular (CV) complications are an essential causal element of prospect in diabetes mellitus (DM), with carotid atherosclerosis being a common risk factor for prospective crisis of coronary artery diseases and/or cerebral infarction in DM subjects. From another point of view, asymmetric dimethylarginine (ADMA) has been established as an inhibitor of endogenous nitric oxide synthesis and the relationship between ADMA and arteriosclerosis has been reported. In our study with 87 type 2 DM (T2DM) patients, we have examined whether ADMA and other CV risk factors are the useful predictors of DMCV complications. After the measurement of the respective CV risk factors, we have followed the enrolled T2DM patients for 5 years. We have finally analyzed 77 patients. DMCV complications developed in 15 cases newly within 5 years, and 4 cases recurred. The concentrations of ADMA in plasma were markedly more elevated in 19 DM patients with CV complications than in 58 DM patients without CV complications. Urinary albumin (U-Alb), mean intimal-medial thickness (IMT) and ankle brachial index (ABI) were also higher in patients with CV complications. Multiple regression analyses showed that U-Alb had an influence on the high level of ADMA (standardized β = 6.59, P = 0.00014) independently of age, systolic BP, fibrinogen, mean IMT, plaque score, and ABI. The review indicates what is presently known regarding plasma ADMA that might be a new and meaningful biomarker of CV complications in DM subjects.
Core tip: Asymmetric dimethylarginine (ADMA) is an emerging independent biomarker for prospective cardiovascular (CV) complications. In our study, the results show that the cases with a high level of ADMA could have diabetes mellitus CV (DMCV) complications in the future within five years. Furthermore, not only ADMA but also urinary albumin was associated with DMCV complications in the multiple regression analyses. The clinical acceptation of this parameter will rely on the availability of therapies to immediately reduce ADMA such as incretin-based drugs, which could support the part of ADMA as an etiologic risk factor.
- Citation: Konya H, Miuchi M, Satani K, Matsutani S, Yano Y, Tsunoda T, Ikawa T, Matsuo T, Ochi F, Kusunoki Y, Tokuda M, Katsuno T, Hamaguchi T, Miyagawa JI, Namba M. Asymmetric dimethylarginine, a biomarker of cardiovascular complications in diabetes mellitus. World J Exp Med 2015; 5(2): 110-119
- URL: https://www.wjgnet.com/2220-315X/full/v5/i2/110.htm
- DOI: https://dx.doi.org/10.5493/wjem.v5.i2.110
Diabetes mellitus (DM) is a complex metabolic disorder and one of the common chronic diseases worldwide. The number of people with DM globally was estimated at 382 million in 2013, and is supposed to reach over 592 million by 2035[1]. Close to 5.1 million deceased in adults aged 20-79 years were attributable to DM in 2013, accounting for 8.4% of the global all-cause mortality in this age group[2]. In addition to the effect on the subjects’ life quality, the microvascular [diabetic retinopathy (DR), diabetic nephropathy (DN), and neuropathy] and macrovascular complicating diseases (coronary heart diseases, peripheral artery diseases, and stroke) of DM also increase the global healthcare burden. Approximated planetary healthcare expenditure to care and preclude DM and its complicating diseases are anticipated to total leastwise 548 billion United States dollars (USD) in 2013. By 2035, this number is proposed to surpass some 627 billion USD[3]. Worldwide, DM is probable to be the fifth leading killer[4].
Cardiovascular (CV) complications are an essential causal element of prognosis in type 2 DM (T2DM), with carotid atherosclerosis (CA) being a common risk factor for prospective crisis of coronary artery disease (CAD) and/or cerebral infarction[5,6]. Some molecules, such as high-sensitivity C-reactive protein (CRP), interleukin-18, and hepatocyte growth factor (HGF) would have been presented to be atherosclerotic biomarkers[7-9]. Preclusion of DM and its involvements, early invention of disease stages, and interventions that would act in the presence of hyperglycemia to avoid, retard or inverse the involvements are the principal concerns. Biomarkers have been investigated for understanding the structures of the evolution and progress of DM involvements[10]. This review presents what is currently known regarding plasma asymmetric dimethylarginine (ADMA) level that might be a new and meaningful biomarker of DMCV complications.
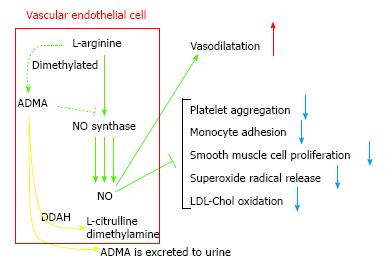
Endothelial dysfunction is distinguished as afflicted nitric oxide (NO)-mediated vascular reaction and is related to maturation of arteriosclerosis and DMCV involvements[11]. NO produced by the vascular endothelium is related to the regulatory mechanisms of the CV system[12]. Since NO is a molecule distributed to important anti-atherosclerotic properties, reduced NO availability may be deemed a crucial risk factor for atherothrombosis and acute CV events. NO is compounded by stereospecific oxidization of the terminal guanidino nitrogen of the amino acid L-arginine by the activity of a group of enzymes known as NO synthases (NOSs)[13]. The major isoform of NOS existing in endothelial cells, eNOS, is constitutively aggressive and transforms the amino acid L-arginine into NO and citrulline. Synthesis of NO can be selectively inhibited by guanidino-substituted analogues of L-arginine which blockade the action site of NOS in competition. In animals, NG-monomethyl L-arginine (L-NMMA) and NG-nitro L-arginine methyl ester have been used as pharmacological tools to reduce NO availability and induce experimental hypertension[14-16]. L-arginine analogues were identified in human plasma and urine[17]. The two L-arginine analogues identified as endogenous inhibitors of NOS were L-NMMA and NG, NG-dimethyl L-arginine (asymmetric dimethylarginine, ADMA). L-NMMA and ADMA were equipotent as NOS inhibitors[17]. Another endogenous arginine analogue (symmetric dimethylarginine, SDMA) was unable to inhibit NOS[17]. As human plasma ADMA concentrations are 10-fold higher than those of L-NMMA[17], ADMA might be regarded as the major endogenous NOS inhibitor. Increased plasma concentrations of ADMA caused impaired NO synthesis, leading to endothelial dysfunction, atherogenesis, and CV disease processes. ADMA and SDMA have been demonstrated to collect in renal failure patients[17].
Though ADMA is somewhat eliminated by the nephros, the predominant metabolism process is decomposition by the dimethylarginine dimethylaminohydrolases (DDAHs) 1 and 2 into dimethylamine and L-citrulline[18,19] (Figure 1). High blood sugar disorders DDAH efficacy in the endothelia and blood vessel involuntary muscle cells in vitro, whereby empowering raised concentrations of ADMA among DM subjects[20]. Chronic ADMA increase in animals induces arteriosclerotic involvements and nephropathy as an outcome of decreased NO production[21]. This indicates a crucial ADMA function in excusing the correlation between vascular endothelial functional impairment, atherogenesis, and DN. It has been demonstrated that ADMA is augmented in conditions such as renal impairment[17,22], DM[23], hypertension[24], and DN[25]. Moreover, it has been indicated that the concentrations of ADMA are prognosticative of CVD and all-cause death rate in preponderantly non-diabetic chronic kidney disease (CKD)[26], end-stage renal disease[27], and CAD[27-29].
Vallance et al[17] showed that during continuous 3 mg/kg per hour ADMA infusate in Cavia porcellus, systolic blood pressure (BP) rose by nearly 15%, as plasma ADMA concentrations rose approximately nine-fold. In the same animal model, bolus injections of 1-30 mg/kg ADMA resulted in a dose-dependent mean arterial BP increment[17]. Gardiner et al[30] confirmed in rats the dose-dependent pressor effects of ADMA. De Gennaro Colonna et al[31] administered ADMA 10 mg/kg per day subcutaneously via an osmotic minipump for 14 d to male adult rats. ADMA-treated rats indicated increased systolic BP, reduced plasma nitrite/nitrate levels, NO-stable end-products and a lower vasorelaxant reaction of the aortic tissues to accumulative acetylcholine concentrations. Isolated perfused cardiac specimens from ADMA-cared rats demonstrated a deterioration of post-ischemic ventricular functional impairment[31]. In humans (healthy male volunteers), an infusion of ADMA at rates of 0.0125 and 0.025 mg/kg per minute increased plasma ADMA concentrations between 2 and 10 μmol/L and remarkably reduced plasma cyclic guanosine monophosphate (cGMP) (the main second messenger of NO) concentrations[32]. The infusion of a higher ADMA dose (0.10 mg/kg per min) importantly reduced stroke volume and heartbeat and raised systemic blood vessel resistance[32]. In another study by Achan et al[19], an intravenous injection of 3.0 mg/kg ADMA in healthy volunteers reduced heart rate and cardiac output and elevated BP and systemic vascular resistance. The effects of a suppressor dose of ADMA (0.25 mg/kg per min) were studied by Kielstein et al[33] in healthy normotensive male patients. ADMA infusion reduced plasma cGMP concentrations (indicating reduced NO production), decreased effective renal plasma flow, and elevated renovascular resistance. On the other hand, ADMA did not alter plasma renin and noradrenalin levels[33], suggesting the facility of this arginine substance to regulate renal function without impacting the renin-angiotensin and sympathetic systems. Moreover, the effects of ADMA on cerebral vascular tone in humans were researched[34]. An infusion of 0.10 mg/kg ADMA per minute in healthy males elevated vascular stiffness and reduced total cerebral perfusion[34], demonstrating that ADMA might also be related to the pathogenesis of cerebrovascular disease. In a word, respective studies demonstrate that ADMA systemic application in humans suppresses NO production and spoils endothelial action in several regions, i.e., the heart, kidney, and brain.
Respective studies have indicated a prognosticative count of ADMA for CV outcomes. The incidence rate of CV endpoints in high risk subjects has been established to be straight and severally connected with elevated concentrations of ADMA in subjects with CAD[28], peripheral arterial occlusive disease[35], T2DM[36], type 1 DM (T1DM)[37] and chronic heart failure[38]. A specific potent relationship between ADMA and hemodynamic factors alike clinical result has been detected in pulmonary arterial hypertension subjects[39]. These forecasting information from experimental studies only reports statistic correlation and does not approve to form the consequence that ADMA is inevitable for prospective CV outcomes. It seems potential that increased ADMA levels are exclusively an epiphenomenon in parallel with other transformations. Nevertheless, animal study outcomes intimate that ADMA indicates not merely a risk biomarker but a risk factor for CV outcomes. It was demonstrated that continuous ADMA infusate for 4 wk resulted in the microangiopathy generation in murine coronary arteries[21]. Overexpression of the ADMA breakdown enzyme DDAH decreased murine ADMA and graft CAD[40]. Moreover, overexpression of DDAH avoided progress of nephropathy by blocking decrease of periductal capillary vessels and tubulointerstitium fibrosis in CKD rats[41]. Konishi et al[42] indicated that transgenic mice overexpressing DDAH displayed augmented endothelium rebirth and neointimas after vessel trauma. These discoveries mean that ADMA might straightly endow angiopathy. Nevertheless, it remains to be decided if a proximate ADMA level alteration can decrease CV risk in humans.
A particular pharmacologic intervention to impact ADMA is not usable yet. It was demonstrated that other steps to decrease CV hazard inclusive of survival training in T1DM subjects[43] and in increased CV hazard subjects[44] or weight reduction in pathologically corpulent subjects[45] are able to reduce the levels of circulating ADMA. It was presented that a few agents (e.g., pravastatin, telmisartan or pioglitazone) are able to elevate the action or the generation of the enzyme DDAH and whereby decrease ADMA via in vitro experiments[46-48]. In addition, clinical studies indicated that intervention with metformin, ACE inhibitors, angiotensin receptor blockers or alpha-lipoic acid would be able to reduce the levels of circulating ADMA[49-55]. The bibliography on the efficacy of statins on ADMA is contentious[20,56-61]. Meanwhile, one clinical study demonstrated an ADMA decrease during intervention with rosiglitazone, and this was not recognized in a discrete age group[62,63]. However, it remains wondering whether regulation of ADMA by these interventions will straightly affect CV hazard additionally.
DM subjects have an untoward CV hazard character. Increased ADMA levels have been reported in subjects with T2DM and T1DM[23,25]. High blood sugar intrinsically may elevate ADMA levels due to decreased metabolic process. It was indicated that increased blood sugar concentrations can suppress DDAH action in cultivated vascular endothelial cells via an in vitro study[20]. In addition, clinical studies in subjects demonstrate that ADMA is straightly relevant to blood sugar concentrations[23,64]. Yasuda et al[65] reported that rigorous diabetic control might affect anti-atherogenicity outcomes via decreasing ADMA concentrations in T2DM subjects. Furthermore, there is proof that insulin resistance is related with elevated ADMA levels[50,62]. This is established by the fact that transgenic mice that overexpress DDAH have decreased ADMA levels and ameliorated insulin sensitivity measured against wild type animals[66]. Several lines of proof suggested that increased levels of ADMA were related with blood glucose control in distinct cohort which applies to the supposition that ADMA might act as a hazard predictive factor for CV outcomes[67,68]. In addition to relations between ADMA and metabolic control, the levels of ADMA might play an important part for the generation of DM involvements as well. ADMA is increased in subjects with T1DM, T2DM or DN with micro- or macroalbuminuria[25,69]. ADMA is correlated with the generation of kidney disorder and might consequently have possible harmful actions in DN subjects[26,70]. In addition, increased ADMA concentrations have been described in T2DM subjects with retinopathy[71]. Thus, it is alluring to hypothesize that ADMA might serve as a pathophysiologic suitable agent for DM involvements. Notwithstanding, high blood sugar remains a principal reason for both elevated ADMA and the generation of DM complicating disease which makes the reading of data more complicated.
Augmented intima-media thickness (IMT) has been demonstrated to be a substitute biomarker for prognosticating CV hazard[72]. In a study by Miyazaki et al[73], stepwise regression analysis indicated plasma concentrations of ADMA to be remarkably related to carotid IMT. In an epidemiologic survey of 712 people, plasma concentrations of ADMA were assayed along with carotid IMT. On multiple stepwise regression analysis, carotid IMT was conspicuously related with the concentrations of ADMA[74] and the generation of carotid IMT, over a 6-year period, was associated with serum concentrations of ADMA[75]. In the PREVENCION study of 922 grown-up subjects, ADMA remarkably prognosticated carotid IMT even after adaptation for CV hazard parameters, CRP, and kidney function, but did not prefigure carotid-femoral pulse wave velocity, BP, or hemodynamic abnormality[76]. Kocak et al[77] determined higher ADMA concentrations in people without already-known arteriosclerotic disease who were on continuous ambulatory peritoneal dialysis and revealed a remarkable positive correlational statistics between the concentrations of ADMA and carotid IMT in these subjects.
We carried out a clinical study to examine the correlation between the plasma ADMA concentrations and the stage of CA in T2DM subjects[78,79].
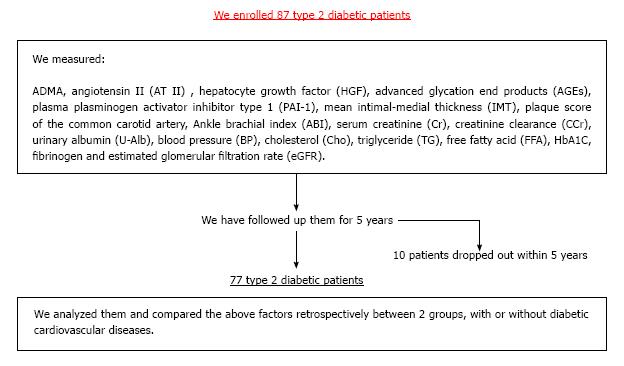
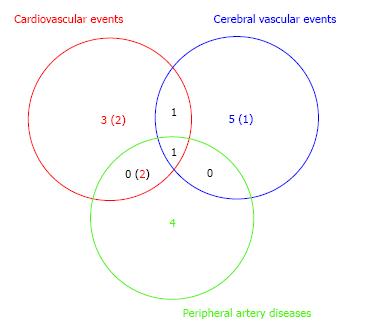
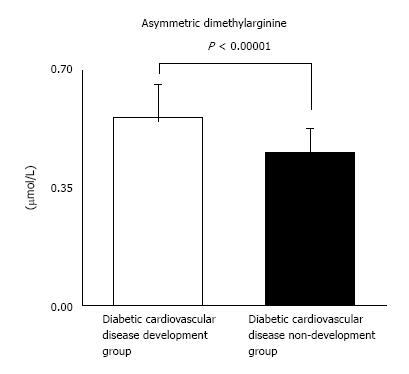
In our study with 87 T2DM patients (Table 1), we have examined whether ADMA and other CV risk factors are the useful predictors of DMCV complications. After the measurement of the respective CV risk factors, we have followed the enrolled T2DM patients for 5 years (Figure 2). We investigated the risk factors as follows: ADMA, angiotensin II (AT II), HGF, advanced glycation end products (AGEs), plasma plasminogen activator inhibitor type 1 (PAI-1), mean IMT, plaque score of the common artery and ankle brachial index (ABI). Furthermore, we measured serum creatinine (Cr), creatinine clearance (CCr), urinary albumin (U-Alb), BP, cholesterol (Cho), triglyceride (TG), free fatty acid (FFA), HbA1C, fibrinogen and eGFR. We compared retrospectively the above factors between two groups with or without CV complications. In addition, we showed results in form of the average ± SD. We have finally analyzed 77 patients. DMCV complications developed in 15 cases (CV events: 5 cases; cerebral vascular events: 7 cases; peripheral artery diseases: 7 cases) newly, and 4 cases (CV events: 4 cases; cerebral vascular events: 1 case; peripheral artery diseases: none case) recurred within 5 years (Figure 3). The concentrations of ADMA in plasma were conspicuously higher in 19 DM subjects with CV complications than in 58 DM subjects without (0.56 ± 0.09 μmol/L vs 0.45 ± 0.07 μmol/L, P < 0.00001) (Figure 4). U-Alb (319.9 ± 522.6 μg/min vs 83.5 ± 199.4 μg/min, P = 0.008) and the value of mean IMT (1.39 ± 0.33 mm vs 1.16 ± 0.30 mm, P = 0.006) were also higher in patients with CV complications. The value of ABI (1.0 ± 0.2 vs 1.1 ± 0.2, P = 0.046) was lower in patients with CV complications. In the study, the other risk factors (AT II, HGF, AGEs, PAI-1, plaque score, Cr, CCr, BP, Cho, TG, FFA, HbA1C, fibrinogen and eGFR) were not associated with the development of the DMCV complications within 5 years. A relative risk of the DMCV complication development reached the highest level (6.81) when the level of ADMA was over 0.54 μmol/L. Multiple regression analyses showed that U-Alb had an influence on the high level of ADMA (standardized β = 6.59, P = 0.00014) independently of age, systolic BP, fibrinogen, mean IMT, plaque score, and ABI. Increased levels of ADMA were found in the generation of CA in patients with T2DM[80,81] or gestational DM[82]. Kanazawa et al[80] demonstrated that serum ADMA would be a predictive factor of arteriosclerosis and linked to the existence of CV complications in Japanese T2DM subjects, but serum SDMA, a structural isomer of ADMA, was correlated neither with factors for arteriosclerosis nor with the existence of CV complications.
| Number of patients | 87 |
| Male/female | 47/40 |
| Age (yr) | 62.6 ± 10.4 |
| BMI (kg/m2) | 23.4 ± 4.2 |
| HbA1C (%) | 9.4 ± 2.2 |
| Duration of diabetes (yr) | 13.5 ± 10.6 |
| With hypertension | 44 |
| With dyslipidemia | 32 |
As well as our results, Celik et al[83] showed that the concentration of ADMA was higher in diabetic subjects with CV complications compared to diabetic patients without complications. They also reported that the levels of fundamental determinants of ADMA were evaluated in diabetic patients with macrovascular complications, using ADMA as a dependent variable in multiple regression analysis. It was found that the most fundamental determinant of ADMA is total homocysteine (tHcy). Moreover, Krzyzanowska et al[84] showed that ADMA is related to clinical CV atherosclerotic disease diagnosis in T2DM, and also concluded that ADMA is associated with total tHcy, U-Alb, Cr, and GFR and that tHcy correlates with age, ADMA, Cr, GFR, and LDL[84]. Research has also shown that increased tHcy concentration in T2DM is associated with increased CV complications[85,86]. Thus, ADMA may be used to predict the likelihood of developing CV complications in DM patients.
According to these studies, the cases with a high level of ADMA, particularly those complicated with nephropathy, should be followed more carefully to prevent new developments and/or recurrences of the DMCV complications.
Lately, a novel treatment strategy for the intervention of T2DM that directs the incretin hormones has been generated. These peptide hormones, i.e., glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic peptide, would be secreted from the bowel after a repast and induce insulin secretion in a glucose-dependent manner[87]. Nevertheless, their activity is restricted by prompt deactivation via the enzyme dipeptidyl peptidase (DPP)-4. Furthermore, T2DM subjects normally do not react well to glucose-dependent insulinotropic peptide and GLP-1[88,89]. Suppression of DPP-4 will elevate levels of active incretins, so DPP-4 has turned out to be a marker in diabetic control[90-92]. Incretin-based therapy was first made available for the treatment of T2DM in the United States in 2006 and in Japan in 2009[93]. Up to now, seven DPP-4 inhibitors are usable in Japan, including sitagliptin, vildagliptin, alogliptin, linagliptin, anagliptin, teneligliptin, and saxagliptin[93-95]. The elevated intrinsic plasma level of GLP-1 is considered to show protective outcomes on the CV system[96]. Ojima et al[97] showed that GLP-1 receptor agonist would inhibit ADMA development in the kidney of streptozotocin-induced DM rats. In addition, serum concentration of ADMA was sought with the DPP-4 inhibitor saxagliptin in an animal experiment[98]. The discoveries of that study indicate that the DPP-4 inhibitors could impact serum concentrations of ADMA. In fact, several DPP-4 inhibitors might reduce ADMA levels in T2DM subjects[99,100].
Incretin-based drugs were found to decrease serum concentrations of ADMA in T2DM subjects. This discovery ought to be backed up with larger-scale prospective randomized trials such as Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus-Thrombolysis in Myocardial Infarction 53 study[101,102] and EXamination of cArdiovascular outcoMes: AlogliptIN vs standard of carE in patients with type 2 diabetes mellitus and acute coronary syndrome trial[103] to conclude that incretin-based drugs provide CV protection along with DM regulation.
ADMA is a rising distinct biomarker for prospective CV accidents. The clinical adoption of this factor will rely on the accessibility of therapies to straightly lower ADMA such as incretin-based drugs, which could support the function of ADMA as a prolific risk factor. Additional studies would be guaranteed in DM patients especially concerning the possible effects of ADMA on DMCV complicating diseases.
P- Reviewer: Buzas GM, Paraskevas KI, Sertoglu E S- Editor: Ji FF L- Editor: Wang TQ E- Editor: Lu YJ
| 1. | International Diabetes Association. The global burden, Diabetes and impaired glucose tolerance, IDF Diabetes Atlas. Available from: http: //www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf. |
| 2. | International Diabetes Association. The global burden, Mortality. IDF Diabetes Atlas. Available from: http: //www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf. |
| 3. | International Diabetes Association. The global burden, Health expenditure, IDF Diabetes Atlas. Available from: http: //www.idf.org/sites/default/files/EN_6E_Atlas_Full_0.pdf. |
| 4. | Roglic G, Unwin N, Bennett PH, Mathers C, Tuomilehto J, Nag S, Connolly V, King H. The burden of mortality attributable to diabetes: realistic estimates for the year 2000. Diabetes Care. 2005;28:2130-2135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 524] [Cited by in RCA: 501] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 5. | Folsom AR, Chambless LE, Duncan BB, Gilbert AC, Pankow JS. Prediction of coronary heart disease in middle-aged adults with diabetes. Diabetes Care. 2003;26:2777-2784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 93] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 6. | Shinoda-Tagawa T, Yamasaki Y, Yoshida S, Kajimoto Y, Tsujino T, Hakui N, Matsumoto M, Hori M. A phosphodiesterase inhibitor, cilostazol, prevents the onset of silent brain infarction in Japanese subjects with Type II diabetes. Diabetologia. 2002;45:188-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 61] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 7. | Aso Y, Okumura K, Takebayashi K, Wakabayashi S, Inukai T. Relationships of plasma interleukin-18 concentrations to hyperhomocysteinemia and carotid intimal-media wall thickness in patients with type 2 diabetes. Diabetes Care. 2003;26:2622-2627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 90] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 8. | Ridker PM, Buring JE, Shih J, Matias M, Hennekens CH. Prospective study of C-reactive protein and the risk of future cardiovascular events among apparently healthy women. Circulation. 1998;98:731-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1037] [Cited by in RCA: 1016] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 9. | Konya H, Miuchi M, Satani K, Matsutani S, Tsunoda T, Yano Y, Katsuno T, Hamaguchi T, Miyagawa J, Namba M. Hepatocyte growth factor, a biomarker of macroangiopathy in diabetes mellitus. World J Diabetes. 2014;5:678-688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Aslan D. Biomarkers for diabetic complications: The results of several clinical studies. J Med Biochem. 2011;30:207-212. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 11. | Cooke JP, Dzau VJ. Nitric oxide synthase: role in the genesis of vascular disease. Annu Rev Med. 1997;48:489-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 518] [Cited by in RCA: 500] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 12. | Moncada S, Palmer RM, Higgs EA. Nitric oxide: physiology, pathophysiology, and pharmacology. Pharmacol Rev. 1991;43:109-142. [PubMed] |
| 13. | Moncada S, Higgs A. The L-arginine-nitric oxide pathway. N Engl J Med. 1993;329:2002-2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4289] [Cited by in RCA: 4163] [Article Influence: 130.1] [Reference Citation Analysis (0)] |
| 14. | Whittle BJ, Lopez-Belmonte J, Rees DD. Modulation of the vasodepressor actions of acetylcholine, bradykinin, substance P and endothelin in the rat by a specific inhibitor of nitric oxide formation. Br J Pharmacol. 1989;98:646-652. [PubMed] |
| 15. | Ribeiro MO, Antunes E, de Nucci G, Lovisolo SM, Zatz R. Chronic inhibition of nitric oxide synthesis. A new model of arterial hypertension. Hypertension. 1992;20:298-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 550] [Cited by in RCA: 549] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 16. | De Gennaro Colonna V, Rossoni G, Rigamonti A, Bonomo S, Manfredi B, Berti F, Muller E. Enalapril and quinapril improve endothelial vasodilator function and aortic eNOS gene expression in L-NAME-treated rats. Eur J Pharmacol. 2002;450:61-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 17. | Vallance P, Leone A, Calver A, Collier J, Moncada S. Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet. 1992;339:572-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1547] [Cited by in RCA: 1540] [Article Influence: 46.7] [Reference Citation Analysis (0)] |
| 18. | MacAllister RJ, Parry H, Kimoto M, Ogawa T, Russell RJ, Hodson H, Whitley GS, Vallance P. Regulation of nitric oxide synthesis by dimethylarginine dimethylaminohydrolase. Br J Pharmacol. 1996;119:1533-1540. [PubMed] |
| 19. | Achan V, Broadhead M, Malaki M, Whitley G, Leiper J, MacAllister R, Vallance P. Asymmetric dimethylarginine causes hypertension and cardiac dysfunction in humans and is actively metabolized by dimethylarginine dimethylaminohydrolase. Arterioscler Thromb Vasc Biol. 2003;23:1455-1459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 435] [Cited by in RCA: 449] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 20. | Lin KY, Ito A, Asagami T, Tsao PS, Adimoolam S, Kimoto M, Tsuji H, Reaven GM, Cooke JP. Impaired nitric oxide synthase pathway in diabetes mellitus: role of asymmetric dimethylarginine and dimethylarginine dimethylaminohydrolase. Circulation. 2002;106:987-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 508] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 21. | Suda O, Tsutsui M, Morishita T, Tasaki H, Ueno S, Nakata S, Tsujimoto T, Toyohira Y, Hayashida Y, Sasaguri Y. Asymmetric dimethylarginine produces vascular lesions in endothelial nitric oxide synthase-deficient mice: involvement of renin-angiotensin system and oxidative stress. Arterioscler Thromb Vasc Biol. 2004;24:1682-1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 149] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 22. | Zoccali C, Bode-Böger S, Mallamaci F, Benedetto F, Tripepi G, Malatino L, Cataliotti A, Bellanuova I, Fermo I, Frölich J. Plasma concentration of asymmetrical dimethylarginine and mortality in patients with end-stage renal disease: a prospective study. Lancet. 2001;358:2113-2117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 799] [Cited by in RCA: 797] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 23. | Abbasi F, Asagmi T, Cooke JP, Lamendola C, McLaughlin T, Reaven GM, Stuehlinger M, Tsao PS. Plasma concentrations of asymmetric dimethylarginine are increased in patients with type 2 diabetes mellitus. Am J Cardiol. 2001;88:1201-1203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 258] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 24. | Surdacki A, Nowicki M, Sandmann J, Tsikas D, Boeger RH, Bode-Boeger SM, Kruszelnicka-Kwiatkowska O, Kokot F, Dubiel JS, Froelich JC. Reduced urinary excretion of nitric oxide metabolites and increased plasma levels of asymmetric dimethylarginine in men with essential hypertension. J Cardiovasc Pharmacol. 1999;33:652-658. [PubMed] |
| 25. | Tarnow L, Hovind P, Teerlink T, Stehouwer CD, Parving HH. Elevated plasma asymmetric dimethylarginine as a marker of cardiovascular morbidity in early diabetic nephropathy in type 1 diabetes. Diabetes Care. 2004;27:765-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 137] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 26. | Ravani P, Tripepi G, Malberti F, Testa S, Mallamaci F, Zoccali C. Asymmetrical dimethylarginine predicts progression to dialysis and death in patients with chronic kidney disease: a competing risks modeling approach. J Am Soc Nephrol. 2005;16:2449-2455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 284] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 27. | Lu TM, Ding YA, Lin SJ, Lee WS, Tai HC. Plasma levels of asymmetrical dimethylarginine and adverse cardiovascular events after percutaneous coronary intervention. Eur Heart J. 2003;24:1912-1919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 132] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 28. | Schnabel R, Blankenberg S, Lubos E, Lackner KJ, Rupprecht HJ, Espinola-Klein C, Jachmann N, Post F, Peetz D, Bickel C. Asymmetric dimethylarginine and the risk of cardiovascular events and death in patients with coronary artery disease: results from the AtheroGene Study. Circ Res. 2005;97:e53-e59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 286] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 29. | Valkonen VP, Päivä H, Salonen JT, Lakka TA, Lehtimäki T, Laakso J, Laaksonen R. Risk of acute coronary events and serum concentration of asymmetrical dimethylarginine. Lancet. 2001;358:2127-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 438] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 30. | Gardiner SM, Kemp PA, Bennett T, Palmer RM, Moncada S. Regional and cardiac haemodynamic effects of NG, NG,dimethyl-L-arginine and their reversibility by vasodilators in conscious rats. Br J Pharmacol. 1993;110:1457-1464. [PubMed] |
| 31. | De Gennaro Colonna V, Bonomo S, Ferrario P, Bianchi M, Berti M, Guazzi M, Manfredi B, Muller EE, Berti F, Rossoni G. Asymmetric dimethylarginine (ADMA) induces vascular endothelium impairment and aggravates post-ischemic ventricular dysfunction in rats. Eur J Pharmacol. 2007;557:178-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 32. | Kielstein JT, Impraim B, Simmel S, Bode-Böger SM, Tsikas D, Frölich JC, Hoeper MM, Haller H, Fliser D. Cardiovascular effects of systemic nitric oxide synthase inhibition with asymmetrical dimethylarginine in humans. Circulation. 2004;109:172-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 292] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 33. | Kielstein JT, Simmel S, Bode-Böger SM, Roth HJ, Schmidt-Gayk H, Haller H, Fliser D. Subpressor dose asymmetric dimethylarginine modulates renal function in humans through nitric oxide synthase inhibition. Kidney Blood Press Res. 2004;27:143-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 34. | Kielstein JT, Donnerstag F, Gasper S, Menne J, Kielstein A, Martens-Lobenhoffer J, Scalera F, Cooke JP, Fliser D, Bode-Böger SM. ADMA increases arterial stiffness and decreases cerebral blood flow in humans. Stroke. 2006;37:2024-2029. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 165] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 35. | Mittermayer F, Krzyzanowska K, Exner M, Mlekusch W, Amighi J, Sabeti S, Minar E, Müller M, Wolzt M, Schillinger M. Asymmetric dimethylarginine predicts major adverse cardiovascular events in patients with advanced peripheral artery disease. Arterioscler Thromb Vasc Biol. 2006;26:2536-2540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 112] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 36. | Krzyzanowska K, Mittermayer F, Wolzt M, Schernthaner G. Asymmetric dimethylarginine predicts cardiovascular events in patients with type 2 diabetes. Diabetes Care. 2007;30:1834-1839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 37. | Lajer M, Tarnow L, Jorsal A, Teerlink T, Parving HH, Rossing P. Plasma concentration of asymmetric dimethylarginine (ADMA) predicts cardiovascular morbidity and mortality in type 1 diabetic patients with diabetic nephropathy. Diabetes Care. 2008;31:747-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 38. | Dückelmann C, Mittermayer F, Haider DG, Altenberger J, Eichinger J, Wolzt M. Asymmetric dimethylarginine enhances cardiovascular risk prediction in patients with chronic heart failure. Arterioscler Thromb Vasc Biol. 2007;27:2037-2042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 39. | Skoro-Sajer N, Mittermayer F, Panzenboeck A, Bonderman D, Sadushi R, Hitsch R, Jakowitsch J, Klepetko W, Kneussl MP, Wolzt M. Asymmetric dimethylarginine is increased in chronic thromboembolic pulmonary hypertension. Am J Respir Crit Care Med. 2007;176:1154-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 40. | Tanaka M, Sydow K, Gunawan F, Jacobi J, Tsao PS, Robbins RC, Cooke JP. Dimethylarginine dimethylaminohydrolase overexpression suppresses graft coronary artery disease. Circulation. 2005;112:1549-1556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 78] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 41. | Matsumoto Y, Ueda S, Yamagishi S, Matsuguma K, Shibata R, Fukami K, Matsuoka H, Imaizumi T, Okuda S. Dimethylarginine dimethylaminohydrolase prevents progression of renal dysfunction by inhibiting loss of peritubular capillaries and tubulointerstitial fibrosis in a rat model of chronic kidney disease. J Am Soc Nephrol. 2007;18:1525-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 42. | Konishi H, Sydow K, Cooke JP. Dimethylarginine dimethylaminohydrolase promotes endothelial repair after vascular injury. J Am Coll Cardiol. 2007;49:1099-1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 63] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 43. | Mittermayer F, Pleiner J, Krzyzanowska K, Wiesinger GF, Francesconi M, Wolzt M. Regular physical exercise normalizes elevated asymmetrical dimethylarginine concentrations in patients with type 1 diabetes mellitus. Wien Klin Wochenschr. 2005;117:816-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 44. | Richter B, Niessner A, Penka M, Grdić M, Steiner S, Strasser B, Ziegler S, Zorn G, Maurer G, Simeon-Rudolf V. Endurance training reduces circulating asymmetric dimethylarginine and myeloperoxidase levels in persons at risk of coronary events. Thromb Haemost. 2005;94:1306-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Krzyzanowska K, Mittermayer F, Kopp HP, Wolzt M, Schernthaner G. Weight loss reduces circulating asymmetrical dimethylarginine concentrations in morbidly obese women. J Clin Endocrinol Metab. 2004;89:6277-6281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 46. | Yin QF, Xiong Y. Pravastatin restores DDAH activity and endothelium-dependent relaxation of rat aorta after exposure to glycated protein. J Cardiovasc Pharmacol. 2005;45:525-532. [PubMed] |
| 47. | Scalera F, Martens-Lobenhoffer J, Bukowska A, Lendeckel U, Täger M, Bode-Böger SM. Effect of telmisartan on nitric oxide--asymmetrical dimethylarginine system: role of angiotensin II type 1 receptor gamma and peroxisome proliferator activated receptor gamma signaling during endothelial aging. Hypertension. 2008;51:696-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 48. | Wakino S, Hayashi K, Tatematsu S, Hasegawa K, Takamatsu I, Kanda T, Homma K, Yoshioka K, Sugano N, Saruta T. Pioglitazone lowers systemic asymmetric dimethylarginine by inducing dimethylarginine dimethylaminohydrolase in rats. Hypertens Res. 2005;28:255-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 81] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 49. | Chang JW, Lee EK, Kim TH, Min WK, Chun S, Lee KU, Kim SB, Park JS. Effects of alpha-lipoic acid on the plasma levels of asymmetric dimethylarginine in diabetic end-stage renal disease patients on hemodialysis: a pilot study. Am J Nephrol. 2007;27:70-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 60] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 50. | Heutling D, Schulz H, Nickel I, Kleinstein J, Kaltwasser P, Westphal S, Mittermayer F, Wolzt M, Krzyzanowska K, Randeva H. Asymmetrical dimethylarginine, inflammatory and metabolic parameters in women with polycystic ovary syndrome before and after metformin treatment. J Clin Endocrinol Metab. 2008;93:82-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 91] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 51. | Asagami T, Abbasi F, Stuelinger M, Lamendola C, McLaughlin T, Cooke JP, Reaven GM, Tsao PS. Metformin treatment lowers asymmetric dimethylarginine concentrations in patients with type 2 diabetes. Metabolism. 2002;51:843-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 137] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 52. | Chen JW, Hsu NW, Wu TC, Lin SJ, Chang MS. Long-term angiotensin-converting enzyme inhibition reduces plasma asymmetric dimethylarginine and improves endothelial nitric oxide bioavailability and coronary microvascular function in patients with syndrome X. Am J Cardiol. 2002;90:974-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 157] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 53. | Napoli C, Sica V, de Nigris F, Pignalosa O, Condorelli M, Ignarro LJ, Liguori A. Sulfhydryl angiotensin-converting enzyme inhibition induces sustained reduction of systemic oxidative stress and improves the nitric oxide pathway in patients with essential hypertension. Am Heart J. 2004;148:e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 122] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 54. | Ito A, Egashira K, Narishige T, Muramatsu K, Takeshita A. Renin-angiotensin system is involved in the mechanism of increased serum asymmetric dimethylarginine in essential hypertension. Jpn Circ J. 2001;65:775-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 80] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 55. | Kawata T, Daimon M, Hasegawa R, Teramoto K, Toyoda T, Sekine T, Yamamoto K, Uchida D, Himi T, Yoshida K. Effect of angiotensin-converting enzyme inhibitor on serum asymmetric dimethylarginine and coronary circulation in patients with type 2 diabetes mellitus. Int J Cardiol. 2009;132:286-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 56. | Päivä H, Laakso J, Lehtimäki T, Isomustajärvi M, Ruokonen I, Laaksonen R. Effect of high-dose statin treatment on plasma concentrations of endogenous nitric oxide synthase inhibitors. J Cardiovasc Pharmacol. 2003;41:219-222. [PubMed] |
| 57. | Valkonen VP, Laakso J, Päivä H, Lehtimäki T, Lakka TA, Isomustajärvi M, Ruokonen I, Salonen JT, Laaksonen R. Asymmetrical dimethylarginine (ADMA) and risk of acute coronary events. Does statin treatment influence plasma ADMA levels? Atheroscler Suppl. 2003;4:19-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 58. | Shinohara K, Shoji T, Kimoto E, Yokoyama H, Fujiwara S, Hatsuda S, Maeno T, Shoji T, Fukumoto S, Emoto M. Effect of atorvastatin on regional arterial stiffness in patients with type 2 diabetes mellitus. J Atheroscler Thromb. 2005;12:205-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 59. | Eid HM, Eritsland J, Larsen J, Arnesen H, Seljeflot I. Increased levels of asymmetric dimethylarginine in populations at risk for atherosclerotic disease. Effects of pravastatin. Atherosclerosis. 2003;166:279-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 91] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 60. | Pereira EC, Bertolami MC, Faludi AA, Salem M, Bersch D, Abdalla DS. Effects of simvastatin and L-arginine on vasodilation, nitric oxide metabolites and endogenous NOS inhibitors in hypercholesterolemic subjects. Free Radic Res. 2003;37:529-536. [PubMed] |
| 61. | Lu TM, Ding YA, Leu HB, Yin WH, Sheu WH, Chu KM. Effect of rosuvastatin on plasma levels of asymmetric dimethylarginine in patients with hypercholesterolemia. Am J Cardiol. 2004;94:157-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 130] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 62. | Stühlinger MC, Abbasi F, Chu JW, Lamendola C, McLaughlin TL, Cooke JP, Reaven GM, Tsao PS. Relationship between insulin resistance and an endogenous nitric oxide synthase inhibitor. JAMA. 2002;287:1420-1426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 429] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 63. | Kelly AS, Thelen AM, Kaiser DR, Gonzalez-Campoy JM, Bank AJ. Rosiglitazone improves endothelial function and inflammation but not asymmetric dimethylarginine or oxidative stress in patients with type 2 diabetes mellitus. Vasc Med. 2007;12:311-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 49] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 64. | Worthley MI, Holmes AS, Willoughby SR, Kucia AM, Heresztyn T, Stewart S, Chirkov YY, Zeitz CJ, Horowitz JD. The deleterious effects of hyperglycemia on platelet function in diabetic patients with acute coronary syndromes mediation by superoxide production, resolution with intensive insulin administration. J Am Coll Cardiol. 2007;49:304-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 125] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 65. | Yasuda S, Miyazaki S, Kanda M, Goto Y, Suzuki M, Harano Y, Nonogi H. Intensive treatment of risk factors in patients with type-2 diabetes mellitus is associated with improvement of endothelial function coupled with a reduction in the levels of plasma asymmetric dimethylarginine and endogenous inhibitor of nitric oxide synthase. Eur Heart J. 2006;27:1159-1165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 66. | Sydow K, Mondon CE, Schrader J, Konishi H, Cooke JP. Dimethylarginine dimethylaminohydrolase overexpression enhances insulin sensitivity. Arterioscler Thromb Vasc Biol. 2008;28:692-697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 81] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 67. | Anderson JL, Carlquist JF, Roberts WL, Horne BD, May HT, Schwarz EL, Pasquali M, Nielson R, Kushnir MM, Rockwood AL. Asymmetric dimethylarginine, cortisol/cortisone ratio, and C-peptide: markers for diabetes and cardiovascular risk? Am Heart J. 2007;153:67-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 68. | Mittermayer F, Kautzky-Willer A, Winzer C, Krzyzanowska K, Prikoszovich T, Demehri S, Wagner O, Wolzt M. Elevated concentrations of asymmetric dimethylarginine are associated with deterioration of glucose tolerance in women with previous gestational diabetes mellitus. J Intern Med. 2007;261:392-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 69. | Krzyzanowska K, Mittermayer F, Shnawa N, Hofer M, Schnabler J, Etmüller Y, Kapiotis S, Wolzt M, Schernthaner G. Asymmetrical dimethylarginine is related to renal function, chronic inflammation and macroangiopathy in patients with Type 2 diabetes and albuminuria. Diabet Med. 2007;24:81-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 42] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 70. | Ueda S, Yamagishi S, Matsumoto Y, Fukami K, Okuda S. Asymmetric dimethylarginine (ADMA) is a novel emerging risk factor for cardiovascular disease and the development of renal injury in chronic kidney disease. Clin Exp Nephrol. 2007;11:115-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 71. | Malecki MT, Undas A, Cyganek K, Mirkiewicz-Sieradzka B, Wolkow P, Osmenda G, Walus-Miarka M, Guzik TJ, Sieradzki J. Plasma asymmetric dimethylarginine (ADMA) is associated with retinopathy in type 2 diabetes. Diabetes Care. 2007;30:2899-2901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 72. | Satani K, Konya H, Hamaguchi T, Umehara A, Katsuno T, Ishikawa T, Kohri K, Hasegawa Y, Suehiro A, Kakishita E. Clinical significance of circulating hepatocyte growth factor, a new risk marker of carotid atherosclerosis in patients with Type 2 diabetes. Diabet Med. 2006;23:617-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 73. | Miyazaki H, Matsuoka H, Cooke JP, Usui M, Ueda S, Okuda S, Imaizumi T. Endogenous nitric oxide synthase inhibitor: a novel marker of atherosclerosis. Circulation. 1999;99:1141-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 497] [Cited by in RCA: 500] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 74. | Furuki K, Adachi H, Matsuoka H, Enomoto M, Satoh A, Hino A, Hirai Y, Imaizumi T. Plasma levels of asymmetric dimethylarginine (ADMA) are related to intima-media thickness of the carotid artery: an epidemiological study. Atherosclerosis. 2007;191:206-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 75. | Furuki K, Adachi H, Enomoto M, Otsuka M, Fukami A, Kumagae S, Matsuoka H, Nanjo Y, Kakuma T, Imaizumi T. Plasma level of asymmetric dimethylarginine (ADMA) as a predictor of carotid intima-media thickness progression: six-year prospective study using carotid ultrasonography. Hypertens Res. 2008;31:1185-1189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 76. | Chirinos JA, David R, Bralley JA, Zea-Díaz H, Muñoz-Atahualpa E, Corrales-Medina F, Cuba-Bustinza C, Chirinos-Pacheco J, Medina-Lezama J. Endogenous nitric oxide synthase inhibitors, arterial hemodynamics, and subclinical vascular disease: the PREVENCION Study. Hypertension. 2008;52:1051-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 48] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 77. | Kocak H, Gumuslu S, Ermis C, Mahsereci E, Sahin E, Gocmen AY, Ersoy F, Suleymanlar G, Yakupoglu G, Tuncer M. Oxidative stress and asymmetric dimethylarginine is independently associated with carotid intima media thickness in peritoneal dialysis patients. Am J Nephrol. 2008;28:91-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 78. | Miuchi M, Konya H, Ida S, Kataoka S, Konishi K, Nagai E, Tokuda M, Murai K, Katsuno T, Hamaguchi T. Is asymmetric dimethylarginine (ADMA) a predictor of the diabetic cardiovascular diseases? IDF. 2009;20th World Congress Abstract Book Available from: http: //www.eubirod.eu/documents/papers/ IDFMontreal09abstractbook.pdf. |
| 79. | Konya H, Miuchi M, Ueyama T, Konishi K, Nagai E, Kusunoki Y, Tokuda M, Murai K, Ida S, Katsuno T. Asymmetric dimetylarginine (ADMA) could affect on cardiovascular complications in type 2 diabetes, particularly complicated with nephropathy. Endocr J. 2010;57:S378, P1-12-3. [DOI] [Full Text] |
| 80. | Kanazawa I, Yano S, Notsu Y, Yamaguchi T, Nabika T, Sugimoto T. Asymmetric dimethylarginine as a risk factor for cardiovascular disease in Japanese patients with type 2 diabetes mellitus. Clin Endocrinol (Oxf). 2011;74:467-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 81. | Xia W, Shao Y, Wang Y, Wang X, Chi Y. Asymmetric dimethylarginine and carotid atherosclerosis in Type 2 diabetes mellitus. J Endocrinol Invest. 2012;35:824-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 82. | Xia W, Li D, Zhang C, Xu L, Xu W, Shao Y. Asymmetric dimethylarginine is associated with high-sensitivity C-reactive protein and early carotid atherosclerosis in women with previous gestational diabetes mellitus. Endocrine. 2015;48:528-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 83. | Celik M, Cerrah S, Arabul M, Akalin A. Relation of asymmetric dimethylarginine levels to macrovascular disease and inflammation markers in type 2 diabetic patients. J Diabetes Res. 2014;2014:139215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 84. | Krzyzanowska K, Mittermayer F, Krugluger W, Schnack C, Hofer M, Wolzt M, Schernthaner G. Asymmetric dimethylarginine is associated with macrovascular disease and total homocysteine in patients with type 2 diabetes. Atherosclerosis. 2006;189:236-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 85. | Clarke R, Daly L, Robinson K, Naughten E, Cahalane S, Fowler B, Graham I. Hyperhomocysteinemia: an independent risk factor for vascular disease. N Engl J Med. 1991;324:1149-1155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1491] [Cited by in RCA: 1422] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 86. | Perry IJ, Refsum H, Morris RW, Ebrahim SB, Ueland PM, Shaper AG. Prospective study of serum total homocysteine concentration and risk of stroke in middle-aged British men. Lancet. 1995;346:1395-1398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 603] [Cited by in RCA: 553] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 87. | Holz GG, Kühtreiber WM, Habener JF. Pancreatic beta-cells are rendered glucose-competent by the insulinotropic hormone glucagon-like peptide-1(7-37). Nature. 1993;361:362-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 412] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 88. | Freeman JS. The pathophysiologic role of incretins. J Am Osteopath Assoc. 2007;107 Suppl:S6-S9. [PubMed] |
| 89. | Nauck MA, Baller B, Meier JJ. Gastric inhibitory polypeptide and glucagon-like peptide-1 in the pathogenesis of type 2 diabetes. Diabetes. 2004;53 Suppl 3:S190-S196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 149] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 90. | Ahrén B, Foley JE. The islet enhancer vildagliptin: mechanisms of improved glucose metabolism. Int J Clin Pract Suppl. 2008;159:8-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 49] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 91. | Banerjee M, Younis N, Soran H. Vildagliptin in clinical practice: a review of literature. Expert Opin Pharmacother. 2009;10:2745-2757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 92. | Palalau AI, Tahrani AA, Piya MK, Barnett AH. DPP-4 inhibitors in clinical practice. Postgrad Med. 2009;121:70-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 93. | Namba M, Katsuno T, Kusunoki Y, Matsuo T, Miuchi M, Miyagawa J. New strategy for the treatment of type 2 diabetes mellitus with incretin-based therapy. Clin Exp Nephrol. 2013;17:10-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 94. | Konya H, Tsunoda T, Kamitani M. Add-on effect of sitagliptin in an obese type 2 diabetic patient with glimepiride and metformin on blood glucose control and incretin response. J Diabetes Invest. 2012;3 Suppl 1:S206. PCS-21-5. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 95. | Konya H, Yano Y, Matsutani S, Tsunoda T, Ikawa T, Kusunoki Y, Matsuo T, Miuchi M, Katsuno T, Hamaguchi T. Profile of saxagliptin in the treatment of type 2 diabetes: focus on Japanese patients. Ther Clin Risk Manag. 2014;10:547-558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 96. | Poornima I, Brown SB, Bhashyam S, Parikh P, Bolukoglu H, Shannon RP. Chronic glucagon-like peptide-1 infusion sustains left ventricular systolic function and prolongs survival in the spontaneously hypertensive, heart failure-prone rat. Circ Heart Fail. 2008;1:153-160. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 148] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 97. | Ojima A, Ishibashi Y, Matsui T, Maeda S, Nishino Y, Takeuchi M, Fukami K, Yamagishi S. Glucagon-like peptide-1 receptor agonist inhibits asymmetric dimethylarginine generation in the kidney of streptozotocin-induced diabetic rats by blocking advanced glycation end product-induced protein arginine methyltranferase-1 expression. Am J Pathol. 2013;182:132-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 123] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 98. | Mason RP, Jacob RF, Kubant R, Walter MF, Bellamine A, Jacoby A, Mizuno Y, Malinski T. Effect of enhanced glycemic control with saxagliptin on endothelial nitric oxide release and CD40 levels in obese rats. J Atheroscler Thromb. 2011;18:774-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 99. | Kubota Y, Miyamoto M, Takagi G, Ikeda T, Kirinoki-Ichikawa S, Tanaka K, Mizuno K. The dipeptidyl peptidase-4 inhibitor sitagliptin improves vascular endothelial function in type 2 diabetes. J Korean Med Sci. 2012;27:1364-1370. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 100. | Cakirca M, Karatoprak C, Zorlu M, Kiskac M, Kanat M, Cikrikcioglu MA, Soysal P, Hursitoglu M, Camli AA, Erkoc R. Effect of vildagliptin add-on treatment to metformin on plasma asymmetric dimethylarginine in type 2 diabetes mellitus patients. Drug Des Devel Ther. 2014;8:239-243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 101. | Mosenzon O, Raz I, Scirica BM, Hirshberg B, Stahre CI, Steg PG, Davidson J, Ohman P, Price DL, Frederich B. Baseline characteristics of the patient population in the Saxagliptin Assessment of Vascular Outcomes Recorded in patients with diabetes mellitus (SAVOR)-TIMI 53 trial. Diabetes Metab Res Rev. 2013;29:417-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 102. | Scirica BM, Bhatt DL, Braunwald E, Steg PG, Davidson J, Hirshberg B, Ohman P, Frederich R, Wiviott SD, Hoffman EB. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med. 2013;369:1317-1326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2804] [Cited by in RCA: 2570] [Article Influence: 214.2] [Reference Citation Analysis (0)] |
| 103. | White WB, Cannon CP, Heller SR, Nissen SE, Bergenstal RM, Bakris GL, Perez AT, Fleck PR, Mehta CR, Kupfer S. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med. 2013;369:1327-1335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1927] [Cited by in RCA: 1894] [Article Influence: 157.8] [Reference Citation Analysis (0)] |