Published online Feb 4, 2017. doi: 10.5492/wjccm.v6.i1.13
Peer-review started: September 7, 2016
First decision: September 29, 2016
Revised: October 30, 2016
Accepted: December 7, 2016
Article in press: December 9, 2016
Published online: February 4, 2017
Processing time: 141 Days and 14.9 Hours
Artificial nutrition (AN) is necessary to meet the nutritional requirements of critically ill patients at nutrition risk because undernutrition determines a poorer prognosis in these patients. There is debate over which route of delivery of AN provides better outcomes and lesser complications. This review describes the management of parenteral nutrition (PN) in critically ill patients. The first aim is to discuss what should be done in order that the PN is safe. The second aim is to dispel “myths” about PN-related complications and show how prevention and monitoring are able to reach the goal of “near zero” PN complications. Finally, in this review is discussed the controversial issue of the route for delivering AN in critically ill patients. The fighting against PN complications should consider: (1) an appropriate blood glucose control; (2) the use of olive oil- and fish oil-based lipid emulsions alternative to soybean oil-based ones; (3) the adoption of insertion and care bundles for central venous access devices; and (4) the implementation of a policy of targeting “near zero” catheter-related bloodstream infections. Adopting all these strategies, the goal of “near zero” PN complications is achievable. If accurately managed, PN can be safely provided for most critically ill patients without expecting a relevant incidence of PN-related complications. Moreover, the use of protocols for the management of nutritional support and the presence of nutrition support teams may decrease PN-related complications. In conclusion, the key messages about the management of PN in critically ill patients are two. First, the dangers of PN-related complications have been exaggerated because complications are uncommon; moreover, infectious complications, as mechanical complications, are more properly catheter-related and not PN-related complications. Second, when enteral nutrition is not feasible or tolerated, PN is as effective and safe as enteral nutrition.
Core tip: The goal of parenteral nutrition (PN) is to complete the therapy without complications. But the goal of “near zero” PN-related complications is achievable if appropriate prevention and monitoring procedures for reducing PN complications are instituted. The key message of this review is the strong recommendation for the development and implementation of protocols for the safe management of PN in critically ill patients, in which each healthcare professional will be actively engaged. If accurately managed, PN can be safely provided for most critically ill patients without expecting a relevant incidence of PN-related complications.
- Citation: Cotogni P. Management of parenteral nutrition in critically ill patients. World J Crit Care Med 2017; 6(1): 13-20
- URL: https://www.wjgnet.com/2220-3141/full/v6/i1/13.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v6.i1.13
Nowadays, the debate on the use of artificial nutrition (AN) in critically ill patients is a hot topic. In fact, many controversies still remain on several aspects of nutritional support of these patients, e.g., timing, quality of macronutrients, safety, incidence of complications, and route of delivery[1,2].
This review has not the purpose to extensively discuss the indications for parenteral nutrition (PN) that are clearly stated in the guidelines, but describes the management of PN in critically ill patients. The first aim is to discuss what should be done in order that the PN is safe. The second aim is to dispel “myths” about PN-related complications and show how prevention and monitoring are able to reach the goal of “near zero” PN complications. Finally, in this review is discussed the controversial issue of the route for delivering AN in critically ill patients.
Generally speaking, guidelines[3-5] recommend in critically ill patients to initiate enteral nutrition (EN) if oral food intake is insufficient, and PN if EN is not sufficient or feasible. But, the key question is: When to use PN in critically ill patients? Nowadays, the role of PN in critically ill patients is one of the most controversial topic in debate. In fact, there are conflicting recommendations released by the American Society of Parenteral and Enteral Nutrition (ASPEN)[3] and the European Society for Clinical Nutrition and Metabolism (ESPEN)[4] regarding the use of PN in this population. For this reason, the indications for PN in critically ill patients were copied from the guidelines and “pasted” in an Appendix enclosed to this review.
PN is a complex prescription therapy associated as every other therapy with the potential risk of adverse effects. To prescribe, compound, and dispense PN in critically ill patients are three key components of a multifaceted process involving different healthcare professionals representing the discipline of medicine, nursing, pharmacy, and dietetics, frequently working together in a Nutrition Support Team[6].
Specifically, the physician prescribing PN should have some competences in indications for PN, infection control, peripheral and central vascular access devices (VADs) and all their related complications. As a matter of fact, PN complications can occur both because of the PN itself and as the result of the PN process.
Moreover, lapses, mistakes, and errors may occur during all the phases of PN. Examples of lapses in the verification process include PN administration: (1) to the wrong patient; (2) by the wrong route, infusing a central mixture via a peripheral vein or through an incorrect tubing connection; and (3) at the wrong infusion rate. Mistakes concerning wrong infusion rates are among the most frequent errors quoted by literature. These errors pose the risk for patient harm due to potential severe metabolic alterations such as hyperglycemia or fat overload.
A proper management of the PN can get full advantage from its beneficial impact on the patient’s condition and lessen the potential adverse effects. Actually, the ASPEN continuously releases clinical guidelines and consensus recommendations about PN safety[7].
The introduction in the daily practice of the “all-in-one” bags (i.e., the commercially available premade multichambered bags) without doubt has improved the PN safety. The ASPEN guidelines[7], comparing the compounded with the multichambered PN formulations, state that the latter have many clinical advantages and better meet the needs of patients. In fact, the “all-in-one” bags allow continuous and stable infusion of all macronutrients. In particular, optimal nitrogen utilization has been found to be achieved when the administration of all macronutrients is performed simultaneously.
Since 2000, Pichard et al[8] demonstrated that the use of 3-compartment bags for PN was less expensive than separate bottles and hospital compounded bags. Moreover, these authors demonstrated that standard formulas satisfied the needs of more than 95% of the adult patients. Pontes-Arruda et al[9] documented that in critically ill patients the delivery of PN through compounded bags was associated with a significantly higher rate of bloodstream infections (BSIs) and central line-associated bloodstream infections (CLABSIs). These authors suggested that the use of multichamber bags may play a role in decreasing the rate of BSIs in critically ill patients receiving PN.
Another crucial issue about PN safety is the Y-site compatibility of intravenous (IV) drugs with PN. Administering PN and medications at the Y-site is not recommended, but unfortunately cannot be avoided when a large number of IV drugs have to be co-infused like in critically ill patients. In an interesting study[10], the physicochemical compatibility of the contact 1:1 between many medications and PN was evaluated in vitro after 1 and 4 h. The authors found an incompatibility after 1 h among PN and the following medications at the tested concentrations: Albumin (200 mg/mL), esomeprazole (0.8 mg/mL), pantoprazole (0.8 mg/mL), tropisetron (1 mg/mL), and fluorouracil (25 and 50 mg/mL). Moreover, they reported an incompatibility after 4 h among PN and the following antibiotics: Cefepime (100 mg/mL) and amoxicillin (50 mg/mL) plus acid clavulanic (10 mg/mL).
The ASPEN recommends the use of in-line filters for PN delivery to reduce potential harms due to microorganisms, air emboli, particulates, and microprecipitates[7]. In-line filters are required for at increased risk critically ill patients (immune-suppressed or infants, neonates, and children), but their use is controversial in not at-risk ones.
To reduce the rate of PN-related complications the first recommendation is to know well them. Every healthcare professional involved in the management of critically ill patient should be prepared to recognize early the onset of a PN complication and intervene for managing it. The PN-related complications can be classified as: (1) metabolic; (2) infectious; and (3) mechanical.
The metabolic complications that can occur acutely are altered hydration status, electrolyte disturbances, and hyperglycemia. These complications are common in critically ill patients, but fairly easy to manage. On the contrary, other severe metabolic complications such as PN-associated liver dysfunctions (i.e., steatosis, cholestasis, and gallstones) and metabolic bone disease may be caused by long-term PN use (i.e., years). Indeed, this is not the case for critically ill patients that usually are on PN for weeks and rarely for few months.
To decrease the incidence of metabolic complications is very important to identify them early by monitoring. The latter should be clinical - and laboratory-based. It is strongly recommended the monitoring of fluid balance targeting “near zero”, as well as the daily check for edema and fluid retention. Moreover, a well-scheduled laboratory monitoring should be designed for checking electrolytes (particularly, phosphate, magnesium, and calcium), renal (particularly, Estimated Glomerular Filtration Rate) and liver function (transaminases, bilirubin, and gamma-glutamyl transferase). In case of prolonged PN (i.e., weeks or months), the laboratory monitoring should be designed for checking trace elements deficiencies (selenium, zinc, and copper), as well as potential anemia causes (vitamin B12, folic acid, iron, and copper).
However, the most important recommendation to reduce the incidence of metabolic complications is to prevent them. At the beginning of the use of PN, the administration of high doses of glucose frequently caused hyperglycemia. Hyperglycemia (i.e., glucose > 10 mmol/L or > 180 mg/dL) contributes to severe infections, organ dysfunctions, and death in critically ill patients and therefore should be carefully avoided. Currently, glucose-induced abnormalities can be prevented by choosing PN formulations with a reduced glucose amount. In hospitalized patients with hyperglycemia, glycemic control is usually easily achieved by the IV pump-driven infusion of short-acting insulin or by the addition of short-acting insulin into the bag (1-2 U/10 g of dextrose).
In 2001, Van den Berghe et al[11] demonstrated in a randomized controlled trial (RCT) in critically ill patients a remarkable positive impact of an intensive insulin therapy on mortality and several other outcomes in case of hyperglycemia. However, many authors subsequently reported that there was a higher incidence of severe hypoglycemia (i.e., 2.2 mmol/L or 40 mg/dL) in patients treated to the tighter limits (i.e., 4.4 mmol/L or 80 mg/dL). Therefore, in our hospital we recommend to use for blood glucose control a cut off of 8.3 mmol/L (or 150 mg/dL). From a practical point of view, an appropriate blood glucose monitoring, based on clinical conditions and infusions scheduled, is able to reduce the risk of both hyperglycemia and hypoglycemia.
Lipid-induced abnormalities arise very rarely in critically ill patients on PN. When these alterations occur generally are more frequent related to liver dysfunction/failure than to PN. When triglyceride levels become greater than 5 mmol/dL (or > 400 mg/dL), we recommend to reduce the fat provision (e.g., reducing the opening of the lipid compartment of the bag). Specifically, we suggest a frequency of lipid administration of 1 to 4 times per week in proportion to the triglyceride levels, although evidence-based data supporting this policy are lacking.
Another hot topic in the debate over the nutritional support of critically ill patients is the issue of the quality of lipid therapy. This issue is still controversial due to, at least in part, inconclusive or contradicting results in several clinical trials using IV lipid emulsions alternative to soybean oil-based IV lipid emulsions. In 2013, Manzanares et al[12] concluded that alternative oil-based lipid emulsions may be associated with reductions in length of stay in intensive care unit (ICU), duration of mechanical ventilation, and mortality in critically ill patients, but lack of statistical precision prevents any recommendations until further studies confirm these positive effects.
In 2014, in a prospective multicenter study was compared the use of different lipid emulsions (i.e., soybean oil, olive oil, and fish oil) in critically ill patients and found that patients receiving olive oil and fish oil had a shorter time to termination of mechanical ventilation alive and a shorter time to ICU discharge alive[13].
High doses of protein intake may lead to high levels of nitrogen-containing compounds such as urea and creatinine, metabolic acidosis, and hypertonic dehydration. However, these are very rare PN complications.
Clinical features of deficiencies or excesses of micronutrients (i.e., vitamins and trace elements) during PN can be avoided simply with the addition of these micronutrients according to a time schedule. Specifically, the regular provision through commercial parenteral vitamins and trace elements preparations avoids deficiencies. For example, thiamine supplements (i.e., 100-300 mg/d) should be administered during the first 3 d in patients with possible thiamine deficiency (i.e., in case of severe malnutrition, anorexia nervosa or alcohol abuse) to prevent neurological side effects associated with glucose delivery during PN.
The optimal intake of macronutrients both energy and protein is largely undefined and the prospective trials have given controversial results[2]. The gold standard to avoid overfeeding should be the use of indirect calorimetry for measuring energy expenditure (EE)[1]. The main limit of EE measurements is that caloric needs may change during the ICU stay. If indirect calorimetry is unavailable, a feeding protocol may significantly reduce the risk of overfeeding. In fact, as suggested since many years in ICU where the patient is often metabolic instable, a protocol for management of PN may markedly decrease the incidence of PN-related complications.
The most feared metabolic complication of PN is the refeeding syndrome (RS). This syndrome can occur as a consequence of administration of nutrients to a patient with a severe undernutrition (i.e., with anorexia nervosa or after a long-standing starvation). The clinical picture of RS includes severe and life-threatening electrolyte abnormalities (hypophosphatemia, hypokalemia, and hypomagnesemia), as well as sodium and fluid retention potentially leading to respiratory failure, heart failure, and consequently death. RS can be prevented through a stepwise and patient’s tailored feeding protocol[14].
The key points for preventing RS is to provide an optimal management and a daily monitoring of serum electrolyte levels, fluid balance, and organ functions. In patients at risk of RS is of pivotal importance to provide a prophylactic supplement of phosphate, as well as to strictly monitor the serum phosphate levels. Besides, sodium and IV fluids administration should be restricted to maintain zero balance[14].
In patients receiving PN, the most feared and relevant infectious complications are catheter-related bloodstream infections (CRBSIs) and CLABSIs[15]. However, these infectious complications are not PN-related but more properly catheter-related complications.
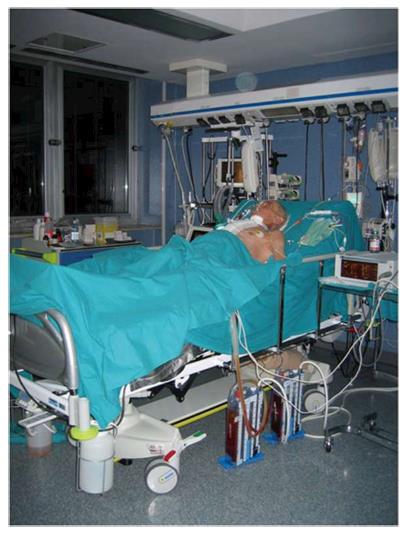
The central VAD is of key importance for quite all critically ill patients in different clinical scenarios (ICU, emergency department, and surgical ward) (Figure 1) for the treatment of different disease states (sepsis, shock, organ dysfunction/failure, major trauma, burns, and postoperative complications) and for a variety of purposes (antibiotic therapy, PN, fluids or medications infusion, procedures of dialysis/apheresis, and hemodynamic monitoring)[16].
However, healthcare providers are frequently worried about the potential complications (mainly, BSIs and thrombosis) related to the use of a central VAD, both centrally inserted central catheters (CICCs) and peripherally inserted central catheters (PICCs). The choice between CICCs and PICCs in critically ill patients is a controversial issue, but many authors suggest that PICCs have many advantages over standard CICCs[17,18]. For a complete review on choice of the VAD (indications for PICCs and comparison between CICCs and PICCs), insertion techniques (site of insertion, ultrasound guidance, single vs multiple-lumen, stabilization, and tip position), and care of the VAD (dressing of vascular access exit site, administration set replacement, and catheter flushing and locking) in all critically ill patients (adults, neonates, infants, and children)[19].
Since 2006, Pronovost et al[20] demonstrated that it is possible to decrease CRBSI in ICU by introducing some interventions and recently seems that the goal of “near zero” CRBSI could become a reality[21]. In 2013, a group of experts included in the top patient safety strategies that can be strongly encouraged for immediate adoption bundles that have checklists to prevent CLASBIs[22].
The first important strategy to decrease the rate of infectious complications is to apply a bundle for the insertion of central VADs, including accurate hand hygiene, skin disinfection with 2% chlorhexidine, maximal sterile barrier precautions, and ultrasound guidance for the catheter insertion[23].
Since 2007, the evidence-based guidelines for preventing healthcare associated infections[24] stated that the use of ultrasound may indirectly reduce infectious complications by facilitating an insertion without immediate mechanical complications[25]. Indeed, nowadays is not justified not using ultrasound guidance for central VAD insertion[26].
The second relevant strategy to decrease the incidence of infectious complications is to apply a bundle for the care of central VADs, including the use of biopatch, semipermeable transparent dressing, 2% chlorhexidine for skin disinfection, and sutureless devices for the catheter care.
Another important strategy to decrease the rate of CRBSI is the selection of the exit site[27]. Many authors suggest the importance to move the exit site of the central VAD from the neck or the supraclavicular area to the infraclavicular area for CICCs or the upper mid-arm for PICCs.
The mechanical complications that can more frequently occur are: Lumen occlusion, catheter dislocation, rupture of external tract, and the most feared venous thrombosis. However, these complications, as infectious complications, are catheter-related and not PN-related complications. According to the ESPEN guidelines, central catheter-related venous thrombosis may be prevented by: (1) the use at insertion of the ultrasound guidance; (2) the use of a VAD with the smallest caliber compatible with the patient’s need; and (3) the position of the tip of the central VAD between the superior vena cava and the right atrium (at or near the so-called atrio-caval junction)[23].
Based on the experience of Dudrick et al[28], the use of PN was introduced in the late 1960s. Since then, without any doubt PN helped greatly many critically ill patients to recover from previously life-limiting clinical conditions. However, the diffuse use of this therapy in all patients, even with extensive indications, brought reservations regarding its benefits and increased the role of EN in the subsequent years.
In the late 1980s emerging evidence from animal studies supported the concept that EN promotes gut function and prevents the translocation of intestinal bacteria. Therefore, total PN was considered to be a “dangerous” form of therapy (e.g., “more harm than good” or “a poison”) and this belief resulted in EN becoming the new standard of care in AN.
The PN is also criticized because it is more expensive than EN. Indeed, both EN and PN are relatively cheap treatments, especially if compared to other therapies that the critically ill patients need to survive.
All together, these concerns influenced the decision-making of physicians about the choice between EN and PN for the nutrition support of critically ill patients.
In fact, in 2000 Heyland[29] questioned if PN in critically ill patients was more harm than good because there were studies comparing EN with PN suggesting that PN was associated with increased complications and mortality in some subgroups of these patients. On the contrary, Jeejeebhoy[30] in 2001 had an absolutely opposite judgment regarding PN and stated that the rate of PN-related complications have been overstated.
In critically ill patients, EN is the recommended method of nutritional support when the patient is unable to have an adequate oral intake of nutrients to meet his/her nutritional requirements and the gastrointestinal tract is functional. The enteral route is efficient and cost-effective, however it is not always as easy as it looks.
Also EN may be the cause of complications that can be classified as: (1) metabolic (e.g., RS may occur also with EN); (2) gastrointestinal (e.g., early satiety, nausea, vomiting, and diarrhea); and (3) mechanical or tube-related (e.g., malposition, dislodgement, and clogging, both in case of nasogastric tube and percutaneous endoscopic gastrostomy). Actually, the most feared complication of EN is pulmonary aspiration because it can be a life-threatening condition.
The debate over the topic of the route for delivering AN in critically ill patients are relevant and attractive. Since several years, meta-analysis and RCTs comparing EN and PN found conflicting results as regards the benefits of EN vs PN in critically ill patients. In my opinion, there is a great misunderstanding in this debate; in particular, that EN and PN are competitors. On the contrary, the selection of EN or PN for delivering nutritional support should be tailored on an individual basis.
In fact, the route chosen for providing AN should be appropriate to the patient’s clinical conditions and should frequently be evaluated for persistent appropriateness, as well as for its adequacy in meeting nutritional requirements of the patient. Not infrequently, in critically ill patients the gastrointestinal tract is not able to tolerate the administration of the prescribed amount of EN formula. If a patient develops intestinal dysfunction/failure due to his/her critical illnesses, PN is more efficient to meet patient’s nutritional needs and better tolerated than EN.
In 2014, Harvey et al[31] reported the results of a RCT evaluating the route of early nutrition support in 2400 adult critically ill patients. The trial compared early NE with early PN and demonstrated that: (1) the PN and the EN groups did not have significant differences in rates of adverse events, treated infectious complications, and other 14 secondary outcomes; and (2) the target intake was not achieved in most patients although the caloric intake was comparable between the groups. Therefore, the authors concluded that: (1) there was no association between 30-d mortality and the route for delivering the early nutritional support; and (2) early PN, as it is generally administered, is neither more beneficial nor more harmful than EN in critically ill patients.
The PN-related complications are catheter-related or metabolic complications. In 2005, Beghetto et al[32] reported that PN was an independent risk factor for central venous catheter-related infection in nonselected hospitalized adult patients. In the past years, the rate of catheter-related complications varied from 1.5 to 4.9 episodes per 1000 catheter days, depending on the inhospital patient population, severity of illness, and the type of central VAD[33]. However, after the Pronovost paper[20], the widespread diffusion of guidelines on care, diagnosis, and therapy of complications of central venous catheters access in PN patients[23], and the introduction of nutrition support teams[6], the incidence of PN-related complications is markedly reduced.
In fact, in the RCT of Harvey et al[31] the mean number of infectious complications was 0.22 in the parenteral group. Moreover, many studies demonstrated that the goal of “near zero” CRBSI[21] has been achieved; actually, in the recent years the incidence of catheter-related infections in ICU patients varied from 0[18,34-36] to 2.4[37] episodes per 1000 catheter days.
Moreover, the optimization of energy provision with supplemental PN in critically ill patients could reduce nosocomial infections, antibiotic usage, and time on mechanical ventilation, and consequently overall health-care costs[38].
It is well known that hepatic dysfunctions are common PN-related metabolic complications in patients receiving long-term PN[39], but this is not the case for critically ill patients that generally receive PN for weeks.
PN is effective and safe when EN is not feasible or tolerated. However, in these patients receiving PN, endogenous infection is more significant than exogenous infection. Indeed, the lack of EN significantly disrupts the usual gastrointestinal microbiota, leads to mucosal gut atrophy and impaired intestinal barrier function. This may determine an impaired immunity favoring endotoxin absorption, macrophage-mediated and cytokine-mediated inflammation, and dangerous bacterial translocation[33].
The question is: How to decrease the incidence of CRBSIs in this condition? The suggestion is that all patients receiving PN, because of EN was initially not tolerated or feasible, should be assessed every day for tolerance of EN and a combined administration of PN and EN should be initiated as soon as feasible.
An emerging and intriguing issue is the use of probiotics, alone or in combination with prebiotics, in critically ill patients to restore the balance of gastrointestinal microbiota with a beneficial impact on intestinal permeability and bacterial translocation. However, further studies in this field are needed before a clear recommendation can be released on the possible therapeutic use of pre- and probiotics for the protection of the gut in ICU patients[40].
The AN is necessary to meet the nutritional requirements of critically ill patients at nutrition risk because undernutrition determines a poorer prognosis in these patients. There is debate over which route of delivery of AN provides better outcomes and lesser complications in critically ill patients.
The fighting against PN complications should consider: (1) an appropriate blood glucose control; (2) the use of olive oil- and fish oil-based lipid emulsions alternative to soybean oil-based ones; (3) the adoption of insertion and care central-line bundles; and (4) the implementation of a policy of targeting “near zero” CRBSIs. Adopting all these strategies, the goal of “near zero” PN complications is achievable.
If accurately managed, PN can be safely provided for most critically ill patients without expecting a relevant incidence of PN-related complications. Moreover, the use of protocols for the management of nutritional support and the presence of nutrition support teams may decrease PN-related complications.
In conclusion, the key messages about the management of PN in critically ill patients are two. First, the dangers of PN-related complications have been exaggerated because complications are uncommon; moreover, infectious complications, as mechanical complications, are more properly catheter-related and not PN-related complications. Second, when EN is not feasible or tolerated, PN is as effective and safe as EN.
Manuscript source: Invited manuscript
Specialty type: Critical care medicine
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Dunser MW, Ren HT, Yu WK S- Editor: Qiu S L- Editor: A E- Editor: Li D
| 1. | Berger MM, Pichard C. Development and current use of parenteral nutrition in critical care - an opinion paper. Crit Care. 2014;18:478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Preiser JC, van Zanten AR, Berger MM, Biolo G, Casaer MP, Doig GS, Griffiths RD, Heyland DK, Hiesmayr M, Iapichino G. Metabolic and nutritional support of critically ill patients: consensus and controversies. Crit Care. 2015;19:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 247] [Cited by in RCA: 263] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 3. | Taylor BE, McClave SA, Martindale RG, Warren MM, Johnson DR, Braunschweig C, McCarthy MS, Davanos E, Rice TW, Cresci GA. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Adult Critically Ill Patient: Society of Critical Care Medicine (SCCM) and American Society for Parenteral and Enteral Nutrition (A.S.P.E.N.). Crit Care Med. 2016;44:390-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 428] [Article Influence: 47.6] [Reference Citation Analysis (0)] |
| 4. | Singer P, Berger MM, Van den Berghe G, Biolo G, Calder P, Forbes A, Griffiths R, Kreyman G, Leverve X, Pichard C. ESPEN Guidelines on Parenteral Nutrition: intensive care. Clin Nutr. 2009;28:387-400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 858] [Cited by in RCA: 691] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 5. | Kreymann KG, Berger MM, Deutz NE, Hiesmayr M, Jolliet P, Kazandjiev G, Nitenberg G, van den Berghe G, Wernerman J, Ebner C. ESPEN Guidelines on Enteral Nutrition: Intensive care. Clin Nutr. 2006;25:210-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 987] [Cited by in RCA: 820] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 6. | Hvas CL, Farrer K, Donaldson E, Blackett B, Lloyd H, Forde C, Garside G, Paine P, Lal S. Quality and safety impact on the provision of parenteral nutrition through introduction of a nutrition support team. Eur J Clin Nutr. 2014;68:1294-1299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Boullata JI, Gilbert K, Sacks G, Labossiere RJ, Crill C, Goday P, Kumpf VJ, Mattox TW, Plogsted S, Holcombe B. A.S.P.E.N. clinical guidelines: parenteral nutrition ordering, order review, compounding, labeling, and dispensing. JPEN J Parenter Enteral Nutr. 2014;38:334-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 217] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 8. | Pichard C, Schwarz G, Frei A, Kyle U, Jolliet P, Morel P, Romand JA, Sierro C. Economic investigation of the use of three-compartment total parenteral nutrition bag: prospective randomized unblinded controlled study. Clin Nutr. 2000;19:245-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 9. | Pontes-Arruda A, Dos Santos MC, Martins LF, González ER, Kliger RG, Maia M, Magnan GB. Influence of parenteral nutrition delivery system on the development of bloodstream infections in critically ill patients: an international, multicenter, prospective, open-label, controlled study--EPICOS study. JPEN J Parenter Enteral Nutr. 2012;36:574-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 10. | Bouchoud L, Fonzo-Christe C, Klingmüller M, Bonnabry P. Compatibility of intravenous medications with parenteral nutrition: in vitro evaluation. JPEN J Parenter Enteral Nutr. 2013;37:416-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 11. | van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345:1359-1367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7077] [Cited by in RCA: 6180] [Article Influence: 257.5] [Reference Citation Analysis (2)] |
| 12. | Manzanares W, Dhaliwal R, Jurewitsch B, Stapleton RD, Jeejeebhoy KN, Heyland DK. Alternative lipid emulsions in the critically ill: a systematic review of the evidence. Intensive Care Med. 2013;39:1683-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 13. | Edmunds CE, Brody RA, Parrott JS, Stankorb SM, Heyland DK. The effects of different IV fat emulsions on clinical outcomes in critically ill patients. Crit Care Med. 2014;42:1168-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Khan LU, Ahmed J, Khan S, Macfie J. Refeeding syndrome: a literature review. Gastroenterol Res Pract. 2011;2011:pii: 410971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 83] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 15. | Lorente L, Villegas J, Martín MM, Jiménez A, Mora ML. Catheter-related infection in critically ill patients. Intensive Care Med. 2004;30:1681-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Palmer D, McFie J. Venous access for parenteral nutrition. In: Payne James J, Grimble GK, Silk, DBA. Artificial nutrition support in clinical practice. London, UK 2001; 379-400. |
| 17. | Griffiths VR, Philpot P. Peripherally inserted central catheters (PICCs): do they have a role in the care of the critically ill patient? Intensive Crit Care Nurs. 2002;18:37-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Pittiruti M, Brutti A, Celentano D, Pomponi M, Biasucci DG, Annetta MG, Scoppettuolo G. Clinical experience with power-injectable PICCs in intensive care patients. Crit Care. 2012;16:R21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 19. | Cotogni P, Pittiruti M. Focus on peripherally inserted central catheters in critically ill patients. World J Crit Care Med. 2014;3:80-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 83] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 20. | Pronovost P, Needham D, Berenholtz S, Sinopoli D, Chu H, Cosgrove S, Sexton B, Hyzy R, Welsh R, Roth G. An intervention to decrease catheter-related bloodstream infections in the ICU. N Engl J Med. 2006;355:2725-2732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3000] [Cited by in RCA: 2758] [Article Influence: 145.2] [Reference Citation Analysis (0)] |
| 21. | Ruiz-Santana S, Saavedra P, León C. “Near zero” catheter-related bloodstream infections: turning dreams into reality*. Crit Care Med. 2012;40:3083-3084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Shekelle PG, Pronovost PJ, Wachter RM, McDonald KM, Schoelles K, Dy SM, Shojania K, Reston JT, Adams AS, Angood PB. The top patient safety strategies that can be encouraged for adoption now. Ann Intern Med. 2013;158:365-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 197] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 23. | Pittiruti M, Hamilton H, Biffi R, MacFie J, Pertkiewicz M. ESPEN Guidelines on Parenteral Nutrition: central venous catheters (access, care, diagnosis and therapy of complications). Clin Nutr. 2009;28:365-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 24. | Loveday HP, Wilson JA, Pratt RJ, Golsorkhi M, Tingle A, Bak A, Browne J, Prieto J, Wilcox M. epic3: national evidence-based guidelines for preventing healthcare-associated infections in NHS hospitals in England. J Hosp Infect. 2014;86 Suppl 1:S1-S70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 817] [Cited by in RCA: 716] [Article Influence: 65.1] [Reference Citation Analysis (0)] |
| 25. | Lamperti M, Bodenham AR, Pittiruti M, Blaivas M, Augoustides JG, Elbarbary M, Pirotte T, Karakitsos D, Ledonne J, Doniger S. International evidence-based recommendations on ultrasound-guided vascular access. Intensive Care Med. 2012;38:1105-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 528] [Article Influence: 40.6] [Reference Citation Analysis (0)] |
| 26. | Bodenham AR. Can you justify not using ultrasound guidance for central venous access? Crit Care. 2006;10:175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Pittiruti M, Migliorini I, Emoli A, Dolcetti L, Pomponi M, Scoppettuolo G, LaGreca A. Preventing central venous catheter related infections: catheter site selection and insertion technique significantly affect the chances of adequate catheter site care. Intensive Care Med. 2007;33 Suppl:S13. |
| 28. | Dudrick SJ, Wilmore DW, Vars HM, Rhoads JE. Long-term total parenteral nutrition with growth, development, and positive nitrogen balance. Surgery. 1968;64:134-142. [PubMed] |
| 29. | Heyland DK. Parenteral nutrition in the critically-ill patient: more harm than good? Proc Nutr Soc. 2000;59:457-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 30. | Jeejeebhoy KN. Total parenteral nutrition: potion or poison? Am J Clin Nutr. 2001;74:160-163. [PubMed] |
| 31. | Harvey SE, Parrott F, Harrison DA, Bear DE, Segaran E, Beale R, Bellingan G, Leonard R, Mythen MG, Rowan KM. Trial of the route of early nutritional support in critically ill adults. N Engl J Med. 2014;371:1673-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 381] [Cited by in RCA: 385] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 32. | Beghetto MG, Victorino J, Teixeira L, de Azevedo MJ. Parenteral nutrition as a risk factor for central venous catheter-related infection. JPEN J Parenter Enteral Nutr. 2005;29:367-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 33. | Mundi MS, Nystrom EM, Hurley DL, McMahon MM. Management of Parenteral Nutrition in Hospitalized Adult Patients. JPEN J Parenter Enteral Nutr. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 34. | Berenholtz SM, Pronovost PJ, Lipsett PA, Hobson D, Earsing K, Farley JE, Milanovich S, Garrett-Mayer E, Winters BD, Rubin HR. Eliminating catheter-related bloodstream infections in the intensive care unit. Crit Care Med. 2004;32:2014-2020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 695] [Cited by in RCA: 627] [Article Influence: 29.9] [Reference Citation Analysis (0)] |
| 35. | Pronovost PJ, Goeschel CA, Colantuoni E, Watson S, Lubomski LH, Berenholtz SM, Thompson DA, Sinopoli DJ, Cosgrove S, Sexton JB. Sustaining reductions in catheter related bloodstream infections in Michigan intensive care units: observational study. BMJ. 2010;340:c309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 348] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 36. | DePalo VA, McNicoll L, Cornell M, Rocha JM, Adams L, Pronovost PJ. The Rhode Island ICU collaborative: a model for reducing central line-associated bloodstream infection and ventilator-associated pneumonia statewide. Qual Saf Health Care. 2010;19:555-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 37. | Peredo R, Sabatier C, Villagrá A, González J, Hernández C, Pérez F, Suárez D, Vallés J. Reduction in catheter-related bloodstream infections in critically ill patients through a multiple system intervention. Eur J Clin Microbiol Infect Dis. 2010;29:1173-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 38. | Heidegger CP, Berger MM, Graf S, Zingg W, Darmon P, Costanza MC, Thibault R, Pichard C. Optimisation of energy provision with supplemental parenteral nutrition in critically ill patients: a randomised controlled clinical trial. Lancet. 2013;381:385-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 537] [Cited by in RCA: 522] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 39. | Raman M, Allard JP. Parenteral nutrition related hepato-biliary disease in adults. Appl Physiol Nutr Metab. 2007;32:646-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 40. | Manzanares W, Lemieux M, Langlois PL, Wischmeyer PE. Probiotic and synbiotic therapy in critical illness: a systematic review and meta-analysis. Crit Care. 2016;19:262. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 200] [Article Influence: 22.2] [Reference Citation Analysis (0)] |