Published online May 4, 2016. doi: 10.5492/wjccm.v5.i2.121
Peer-review started: November 30, 2015
First decision: December 28, 2015
Revised: January 11, 2016
Accepted: March 7, 2016
Article in press: March 9, 2016
Published online: May 4, 2016
Processing time: 150 Days and 2 Hours
Acute respiratory distress syndrome (ARDS) is a syndrome with heterogeneous underlying pathological processes. It represents a common clinical problem in intensive care unit patients and it is characterized by high mortality. The mainstay of treatment for ARDS is lung protective ventilation with low tidal volumes and positive end-expiratory pressure sufficient for alveolar recruitment. Prone positioning is a supplementary strategy available in managing patients with ARDS. It was first described 40 years ago and it proves to be in alignment with two major ARDS pathophysiological lung models; the “sponge lung” - and the “shape matching” -model. Current evidence strongly supports that prone positioning has beneficial effects on gas exchange, respiratory mechanics, lung protection and hemodynamics as it redistributes transpulmonary pressure, stress and strain throughout the lung and unloads the right ventricle. The factors that individually influence the time course of alveolar recruitment and the improvement in oxygenation during prone positioning have not been well characterized. Although patients’ response to prone positioning is quite variable and hard to predict, large randomized trials and recent meta-analyses show that prone position in conjunction with a lung-protective strategy, when performed early and in sufficient duration, may improve survival in patients with ARDS. This pathophysiology-based review and recent clinical evidence strongly support the use of prone positioning in the early management of severe ARDS systematically and not as a rescue maneuver or a last-ditch effort.
Core tip: Lung protective ventilation has become the standard treatment strategy for patients with acute respiratory distress syndrome (ARDS). The physiological basis of prone positioning seems to act beneficially in most pathophysiological disorders of ARDS improving hemodynamics, gas exchange and respiratory mechanics. Moreover prone positioning seems to exert an additional beneficial effect against ventilator-induced lung injury. In patients with severe ARDS, early use of prolonged prone positioning in conjunction with lung-protective strategies decreases mortality significantly.
- Citation: Koulouras V, Papathanakos G, Papathanasiou A, Nakos G. Efficacy of prone position in acute respiratory distress syndrome patients: A pathophysiology-based review. World J Crit Care Med 2016; 5(2): 121-136
- URL: https://www.wjgnet.com/2220-3141/full/v5/i2/121.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v5.i2.121
The adult respiratory distress syndrome was first described during Vietnam War in 1960s as a new distinctive clinical entity of hypoxemic respiratory failure affecting both lungs. This term was later modified to acute respiratory distress syndrome (ARDS) characterized by a diffuse inflammatory condition of the lungs, decreased respiratory system compliance, bilateral pulmonary infiltrates and rapid onset of hypoxemic respiratory failure following a variety of lung insults.
ARDS is a clinical syndrome with heterogeneous underlying pathological processes; it can arise from direct (pulmonary) injury to the lung parenchyma or from indirect (extrapulmonary) systemic insults transmitted by circulation. Regardless of the underlying insult, the development of diffuse alveolar damage involves neutrophil activation and endothelial injury, leading to noncardiogenic pulmonary edema and atelectasis.
In 1994, the American and European Consensus Conference (AECC) established specific criteria for acute lung injury (ALI) and ARDS, with ARDS being the most severe form of the syndrome[1,2]. These criteria included acute onset, bilateral lung infiltrates on chest radiograph, no evidence of elevated left atrial pressure and severe hypoxaemia, assessed by the arterial oxygen tension to inspired oxygen fraction (PaO2/FiO2) ratio. According to these guidelines, ARDS existed when the PaO2/FiO2 ratio was ≤ 200 mmHg and ALI when the PaO2/FiO2 ratio was ≤ 300 mmHg. The AECC definition for ARDS remained the basis for enrollment in most of the landmark trials over the past 20 years.
Based on the limitations of diagnostic reliability and stratification of patients with ARDS/ALI according to severity by AECC criteria, the European Society of Intensive Care Medicine proposed the Berlin ARDS definition in 2011 (Table 1). This new “Berlin” definition is not substantially different from the old, but defines the criteria more specifically including timing, chest imaging, origin of edema and oxygenation, and classifies the severity of disease on the basis of the degree of hypoxemia and positive end-expiratory pressure (PEEP) or continuous positive airway pressure (CPAP)[3].
| Timing | Within 1 wk of a known clinical insult or new or worsening respiratory symptoms |
| Chest imaging1 | Bilateral opacities - not fully explained by effusions, lobar/lung collage, or nodules |
| Origin of edema | Respiratory failure not fully explained by cardiac failure of fluid overload. Need objective assessment (e.g., echocardiography) to exclude hydrostatic edema if no risk factor present |
| Oxygenation2 | |
| Mild | 200 mmHg < PaO2/FiO2 ≤ 300 mmHg with PEEP or |
| CPAP ≥ 5 cmH2O3 | |
| Moderate | 100 mmHg < PaO2/FiO2 ≤ 200 mmHg with PEEP or |
| CPAP ≥ 5 cmH2O | |
| Severe | PaO2/FiO2 ≤ 100 mmHg with PEEP or CPAP ≥ 5 cmH2O |
ARDS represents a common clinical problem in intensive care unit patients[4]. It has a varying incidence from 5-7.2 in Europe to 33.8 new cases/100000 population/year in the United States (150000-200000 cases/year)[5-7]. In the ICU setting, 7%-10% of admitted patients and 5%-8% of the mechanically ventilated ones meet criteria for ALI/ARDS[8]. After continued progress in understanding ARDS pathophysiology and the application of lung protective ventilation, mortality rate significantly decreased from a rate of 65%-70% in the early 1980s to 35%-40% to date in RCTs and consistently higher in real word observational studies[7,9,10].
The majority of patients with ARDS will require mechanical ventilation. The goals of mechanical ventilation for ARDS patients are to minimize iatrogenic lung injury [ventilator-induced lung injury (VILI)] while providing acceptable oxygenation and carbon dioxide (CO2) clearance.
Numerous studies provided clear evidence of large mortality benefit when patients with ARDS were ventilated with a lung-protective strategy: Avoidance of alveolar overdistention using tidal volumes of 6 mL/kg predicted body weight, with plateau pressures ≤ 30 cmH2O, and allowing a low pH in order to achieve these targets[11,12].
A major and controversial aspect of mechanical ventilation regards PEEP; the appropriate levels of PEEP and proper method of titration remain controversial[13-17]. Some authors recommend the lowest level (5-10 cm H2O) of PEEP to be used to support oxygenation and maintain FiO2 at or below 0.6. A recent meta-analysis, which included data from ALVEOLI, LOVS, and EXPRESS clinical trials, revealed that higher levels of PEEP were associated with improved survival and oxygenation among patients with moderate to severe ARDS[18,19].
Conceptually, prone position may result to a more uniform distribution of lung stress and strain, leading to improved ventilation-perfusion matching and regional improvement in lung and chest wall mechanics. Prior clinical trials showed that prone positioning improves oxygenation in patients with ARDS, without benefits in terms of survival[20-22]. A recent multicenter prospective controlled trial (the PROSEVA study) showed that prone positioning decreased 28-d and 90-d mortality, increased ventilator-free days and decreased time to extubation[23]. Based on these data, ventilation in the prone position is recommended for the first week in moderate to severe ARDS patients.
Other adjunctive strategies used in the ARDS setting include recruitment maneuvers, conservative fluid strategy[24], neuromuscular blocking agents[25], extracorporeal membrane oxygenation, high-frequency ventilation[26,27], corticosteroids[28], and inhaled pharmacologic agents.
In this review article, we describe the ARDS pathophysiological models supporting the prone position, we highlight the physiological and lung protective effects of prone positioning and we review the most recent clinical trials on prone position in ARDS patients.
The possible benefits of prone positioning were first speculated in 1974 from studies on the effects of sedation and paralysis on the diaphragm. Bryan et al[29] suggested that anaesthetized and paralyzed patients in the prone position should exhibit a better expansion of the dependent (dorsal) lung regions with consistent improvement in oxygenation, indicating prone’s potential beneficial impact on lung mechanics. Two years later, Piehl et al[30] reported dramatic effects on oxygenation improvement by prone position in five patients with ARDS and in the following year Douglas et al[31] reported similar findings in six ARDS patients, confirming that prone positioning could effectively improve oxygenation in this patient group. Although the first reports were very promising, the following years the clinical application of prone positioning in ARDS patients was not very popular. Not until 1986, when Maunder et al[32] with their chest computed tomography scans study challenged the previously commonly held assumption that ARDS is a homogeneous process (as usually shown by anteroposterior radiography), associated with generalized and relatively uniform damage to the alveolar capillary membrane. The same year Gattinoni et al[33] demonstrated that in ARDS, affected areas primarily occur in the dependent portion of the lung parenchyma. This was soon accompanied by the finding that in ARDS, respiratory compliance is also well correlated with the amount of normally aerated (nondependent) tissue and not with the amount of nonaerated (dependent) tissue[34]. ARDS lung is not stiff but “small” (“baby lung”), and the elasticity of the residual inflated lung is nearly normal. At first physicians, believed that “baby lung” was something well defined, constant and anatomically confined in the ventral (nondependent) regions of the lungs. They turned ARDS patients to the prone position, trying to redistribute the blood flow from the posterior unventilated lung to the previously nondependent baby lung, in order to improve lung’s perfusion, to minimize the resulted shunt and to improve the oxygenation[35,36]. Although the physiologic mechanisms leading to improved oxygenation during prone positioning proved to be different as first suggested, and the redistribution concerned the alveolar gas more, the interest in prone positioning remained strong and prone position proved to be beneficial for both oxygenation and outcome of ARDS patients.
From 1988 to 1991 computerized tomograms of ARDS patients being in the prone position revealed an unexpected finding: The disappearance of the posterobasal densities after prone positioning and their redistribution to the new dependent lung regions[37,38]. This finding changed the concept of “baby lung” from an anatomically confined- to a functional entity, and led to the development of an early pathophysiological model known as “sponge lung” model[35,39].
When someone removes a sponge from the water and holds it flat, the water drains from it and then slows to a stop. If the sponge is turned from horizontal to vertical position, the drainage begins again and then slows again to a stop. As it slows, the sponge is not equally wet from top to bottom, with the top having more empty pores than the bottom. This is pretty much what the “sponge lung” model in ARDS patients suggested: Edema increases the lung weight and squeezes the gas out of the dependent lung regions producing alveolar collapse and increasing the CT densities in dependent regions (compression atelectasis)[40,41]; the size of open airway and the amount of gas decreases along the vertical axis. Although in ARDS the edema has a nongravitational distribution and is quite homogeneously distributed throughout the lung parenchyma[40,42], the “sponge lung” model provided, at that time, a satisfying explanation for three different things. Firstly, how the increased lung mass in ARDS patients due to edema and the increased superimposed pressure, including the heart weight squeeze out the gas of the gravity-dependent lung regions leading to loss of lung aeration[35]. Secondly, why the lung densities shift from dorsal to ventral regions during prone position in ARDS lung[37,43]: The superimposed hydrostatic pressure is reversed and the ventral regions, as the result of the gravitational forces, are newly compressed (this can happen within minutes). And thirdly, “sponge lung” model explained the mechanism through which PEEP acts as a counterforce to oppose the collapsing, compressing forces: PEEP greater than the superimposed pressure keeps the most dependent lung regions open[36,41].
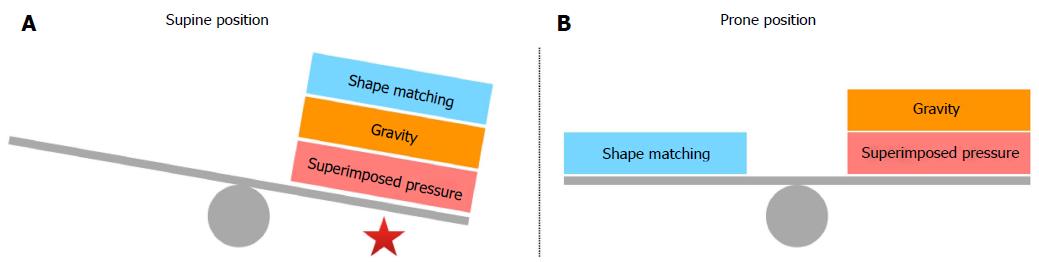
Some years later the “sponge lung” model and the opinion that in ARDS patients the lung edema causes the lung to collapse under its own weight in dependent regions was challenged as a hypothesis by some authors[44,45] and a new supplementary hypothesis was proposed. In ARDS patients in supine position, the dependent areas of the lung collapse not only due to edema and the increased superimposed pressure but also due to the different shape existing between the lung and the chest wall and the resulted nonhomogeneous expansion of alveolar units. The isolated lung normally has a conical shape with the dependent side being bigger than the nondependent side (in supine position). On the other hand, the chest wall has a cylindrical shape and the problem proves to be a shape-matching problem (the fitting of an elastic cone into a rigid cylinder). Because the two structures have the same volume, the lung must expand its upper regions more than the lower ones and this condition results to a greater expansion of the nondependent alveolar units or otherwise to a lesser expansion of the dependent ones[46]. In ARDS patients who are in supine position, the gravitational forces, the increased superimposed pressure, and the shape matching of the lung into the chest cavity act to the same direction having a detrimental effect on dependent alveolar units. On the contrary, in ARDS patients, who are turned in the prone position, shape matching counterbalances gravity and superimposed pressure allowing a more homogeneous inflation of the dependent lung areas (Figure 1). In addition, prone position eliminates compression of the lungs by the heart[47,48] and relieves the dependent lung area from the abdominal pressure[45,49].
The “shape matching” model enlightens two aspects of prone positioning. If lungs would not have a conical shape and were just symmetrical, the degree of shunt and hypoxia would not vary between supine and prone position if perfusion would remain the same. After the rotation of the patient to the prone position the shunt lessens and the oxygenation improves because the recruitment of the dorsal areas overcomes the de-recruitment of the ventral regions due to “shape matching”[44]. Secondly this model takes into account an inherent nonuniform alveolar stress that is not gravitationally determined and explains in part why the application of prone positioning diminishes alveolar hyperinflation and protects the lungs from high shearing forces and eventually from ventilator induced lung injury (VILI)[50].
Oxygenation: It is well known that there is normally a regional difference in intrapleural pressure, being more subatmospheric at the apex and at the nondependent lung areas. This is clearly a gravity dependent phenomenon and results in exponentially regional differences in transpulmonary pressure and thus in the size of alveoli; the transpulmonary pressure, i.e., the distending forces of the lung, decreases along the ventral-to-dorsal axis and the size of the alveolar units decreases toward the dependent areas.
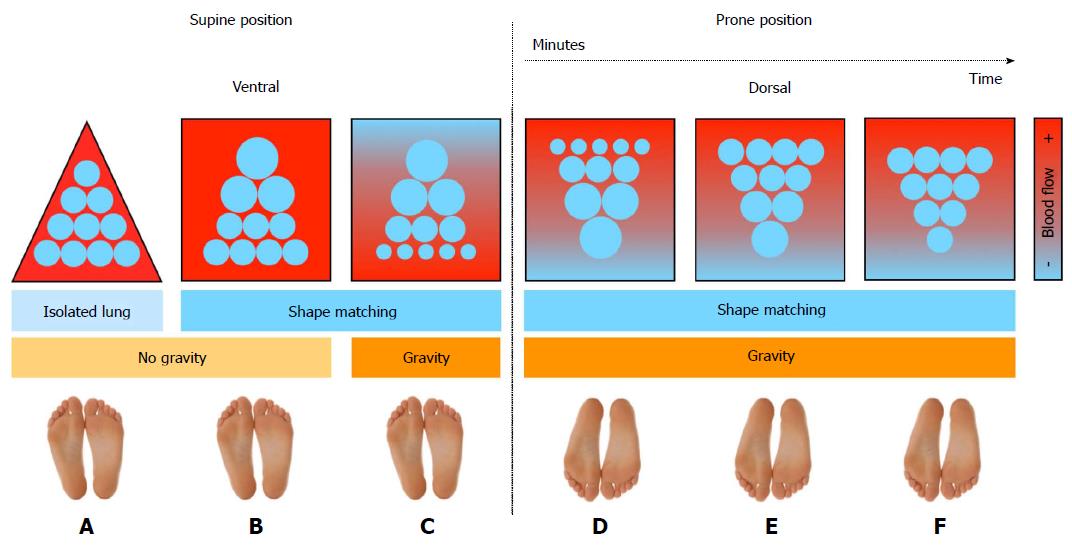
It was found that by turning the patient to the prone position due to thoracic-lung shape modifications the intrapleural pressure becomes less negative in non-dependent and less positive in dependent regions[51,52]. The net effect of prone positioning is not only the increase of regional inflation distribution in dorsal regions and decrease in ventral regions respectively, but intrapleural pressure, transpulmonary pressure and regional inflation distribution become more homogeneous throughout the lung (Figure 2)[53]. It was early suggested that this could be explained by the reversal of lung weight gradients, the direct transmission of the weight of the heart to subjacent regions, direct transmission of the weight of abdominal contents to caudal regions of the dorsal lung and/or regional mechanical properties and shape of the chest wall and lung[54].
In addition, although in healthy lung pulmonary perfusion is distributed along a ventral-dorsal gradient and progressively increases down the lung, data suggest that in the diseased lung, blood flow is being diverted toward the nondependent regions. This is caused by several mechanisms including hypoxic vasoconstriction, vessel obliteration and extrinsic vessel compression[55-57]. Also, human and animal studies have shown that in the conversion from the supine to prone ventilation, pulmonary blood flow in dorsal regions of the lung is maintained unmodified and prevalent in lung dorsal areas (Figure 2)[46,53,58-63]. Besides, in patients with ARDS, the increased lung weight due to serious inflammation and pulmonary edema would act also as hypergravity to squeeze the blood flow as well as ventilation out of the dependent area to the nondependent region[16,64]. Thus, the reduction in intrapulmonary shunt and the increase in oxygenation observed in patients with ARDS who are turned in the prone position mainly results from better ventilated well-perfused lung areas with dorsal recruitment being in parallel greater than ventral de-recruitment (Figure 2)[36,51,53]. Animal data had early suggested that during prone position homogeneity of ventilation increases V/Q as well as the correlation between regional ventilation and perfusion[65]. Very important is the finding that prone position, when combined, is followed by an improved and/or a more sustained response to recruiting maneuvers[50,66-68].
Albert et al[47] in their study determined the fraction of lung that might be subjected to the weight of the heart when patients are in the supine vs the prone position. The study included only non-ARDS patients, but it was found that turning patients to the prone position eliminates the compressive force of the heart on dorsal lung regions redirecting it to only a small portion of the ventral lung regions (Figure 3). This is in agreement with the results of previous studies. In a study conducted by our group it was shown that ARDS patients with congestive heart failure and cardiomegaly after being turned to the prone positioning exhibited a significant, rapid, and persistent improvement in oxygenation. This improvement could be partly due to the decompression of the left lower lobe by the enlarged heart[69]. Wiener et al[70] had early found that patients with cardiomegaly exhibited reduced left mid- and lower zone ventilation in the supine but not in the prone position.
It is worth to mention that the interaction between PEEP and posture on regional distribution of ventilation was recently examined in anesthetized human volunteers. It was found that after the addition of PEEP in the prone position there is a much greater redistribution to ventral areas for blood flow than for ventilation, causing increased V/Q mismatch. In the study of Petersson et al[71], without PEEP, the vertical ventilation-to-perfusion gradient was less in prone postures than in supine, but with PEEP, the gradient was similar. Although this finding supports prior studies, which have shown that lower PEEP is needed to maintain oxygenation in the prone posture than in the supine[66,72], reductions of PEEP are inappropriate, at least when V/Q matching and systemic oxygenation are being evaluated[73,74].
CO2 clearance: During the course of ARDS the CO2 clearance is becoming impaired due to structural changes of the lung[75-77] and the increase in dead space proves to be a prognostic marker of ARDS mortality[78]. Interestingly, turning the ARDS patient to prone position does not always result in decrease in arterial CO2 because the presence of aerated alveoli doesn’t necessarily mean that they are also well ventilated. In fact, it has been suggested that oxygen and carbon dioxide responses to prone position are independent and a decrease in PaCO2 to the first pronation rather than an increase in PaO2/FiO2, is significantly associated with lung recruitability and a better outcome[79,80].
It has been proposed that in PaCO2 nonresponders, the primary mechanism of the PaO2 increase is diversion of the blood flow, whereas in PaCO2 responders the primary mechanism is greater dorsal recruitment in comparison to ventral derecruitment, combined with reduced alveolar overinflation[46,79]. The PaCO2 responders seem to have a higher potential to be recruited with prone positioning than with nonresponders, revealing a difference in underlying lung pathologies[81]. It has also been suggested that when PaO2 increases and PaCO2 does not simultaneously decrease, it is a sign that either cardiac output is lowered or alveolar dead space ventilation is increased by PEEP, reflecting lung overdistention[63].
Total respiratory system mechanics in general are not modified during prone position and it has been shown that respiratory mechanics improve after returning to supine position, suggesting the potential beneficial structural effects of prone positioning[20,54]. Although chest wall compliance decreases[20,40] prone position does not affect total respiratory system compliance[54]. The only exception may be patients with secondary ARDS (nonpulmonary insult), who have shown an increase in respiratory system compliance[22,82,83].
Low tidal volume ventilation[11], high PEEP and recruitment maneuvers are commonly used in the ARDS setting as protective ventilation strategies to minimize lung overdistention and ventilation heterogeneity, but ventilation at low tidal volumes can also cause injury through repetitive opening and closing of the small airways and lung units[84,85], effects on surfactant function[86], and regional hypoxia[87]. Stress is the tension developed in the lungs’ fibrous skeleton when a distending force is applied, and strain is the volume increase caused by the applied force relative to the resting volume of the lungs[88]. The clinical equivalent of stress is transpulmonary pressure (airway pressure minus pleural pressure) and the clinical equivalent of strain is the ratio of volume change (ΔV) to the functional residual capacity (FRC), which is the resting lung volume[89]. Under mechanical ventilation with PEEP, lung strain may be calculated as: Strain = (VT + PEEP volume)/FRC, and stress and strain are linked by the formula: Stress = k x strain, where k is the lung-specific elastance (approximately equal 13 cmH2O in either healthy or acutely injured lungs)[89-91].
As already mentioned, prone positioning results to a more homogeneous distribution of transpulmonary pressure and regional inflation throughout the lung. By favoring such a homogenization, turning ARDS patients to prone position could help eliminate lung overdistention, which proves to be the main component of VILI (volutrauma). In the prone position, air is distributed more homogeneously throughout the lungs, and stress and strain are decreased. Indeed, there are several levels of evidence supporting the preventive effect of prone position on VILI. Animal studies suggest that prone positioning decreases or delays the progression of VILI[92-95], while human studies have confirmed the relevant beneficial effect of prone positioning in the ARDS setting. In a study conducted by our group, Galiatsou et al[50] compared lung CT scans in ARDS patients in supine and prone position and found that prone position is associated with significant alveolar recruitment and less hyperinflation compared to the supine position; this process was more prevalent in lobar than in diffuse ARDS patients. This finding was confirmed and even more extended in the study by Cornejo et al[96]; the authors concluded that prone positioning enchases lung recruitment and decreases tidal hyperinflation, even in those ARDS patients classified as having low potential for lung recruitment. The same study also suggested that prone positioning decreases alveolar instability and cyclic alveolar recruitment/derecruitment. This proves to be particularly important, as intra-airway shear forces due to cyclical airspace opening and closing of airway and pulmonary units result to injury of airway epithelial cells (atelectrauma), which is the second component of VILI pathogenesis[90].
Another component of VILI is biotrauma (lung inflammation). It follows the application of unphysiological mechanical forces to lung tissue, the release of proinflammatory cytokines and the recruitment of white cells; it can lead to multi-organ failure[88,97]. Papazian et al[98] compared neutrophil counts and interleukin-8 levels in the bronchoalveolar lavage fluid of ARDS patients being in the supine and in the prone position and found that prone position reduced lung inflammation in ARDS patients. In an experimental study conducted by our group, we examined whether the prone position of the patients affects histological changes and apoptosis in the lung and “end organs”, including the brain, heart, diaphragm, liver, kidneys and small intestine. We found that prone position appears to reduce the severity and the extent of lung injury, and is associated with decreased apoptosis in the lung and “end organs”[99]. It is also known that mechanical ventilation induces heterogeneous lung injury by mitogen-activated protein kinase (MAPK). Park et al[100] in their experimental study on rodent lungs exposed to injurious ventilation found that the prone positioning has a protective lung effect by increasing the expression of MAPK-phosphatase 1, while the supine position has an opposite effect.
The prone positioning has extra protective lung effects: It improves the mobilization and postural drainage of secretions from the posterior lung segments[54,101-103], and it has been shown to reduce the risk of ventilator-associated pneumonia (VAP)[76,104-108]. Finally, by enchasing oxygenation, proning reduces the need of sustaining high toxic levels of inspired oxygen[109].
Vieillard-Baron et al[110] investigated the short-term effects of prone positioning on right ventricular function in patients with severe ARDS and it was found that proning unloads the right ventricle by decreasing right ventricular enlargement and mean septal dyskinesia. Recently, Jozwiack et al[111] confirmed the beneficial hemodynamic effects of prone positioning in patients with ARDS as it was shown that prone positioning increases cardiac preload, reduces right ventricular afterload and increases left ventricular preload; this resulted to increased cardiac index only in patients with preload reserve. During the prone positioning, pulmonary arterial occlusion pressure is also increased and the transpulmonary pressure gradient (the difference of mean pulmonary arterial pressure relative to pulmonary artery occlusion pressure) is reduced[111]. Elevated transpulmonary pressure gradient defines pulmonary vascular dysfunction and is independently related to increased ARDS mortality[51,112,113]. Besides, the increased pulmonary arterial occlusion pressure transfers some lung regions from West zone 2 to zone 3, thus having the potential to decrease dead space ventilation, another factor independently related to ARDS mortality[51,78,111]. Prone positioning serves both the lung and the “right ventricle protective approach” of mechanical ventilation[112].
Brücken et al[114] investigated the influence of prone positioning on the measurement of transpulmonary thermodilution-derived variables in ARDS patients and found that although extravascular lung water index (EVLWI) and global end-diastolic volume index measurements are possibly influenced by prone positioning, the differences are minor and presumably of no clinical relevance. The positive effect of prone positioning on EVLWI was demonstrated by McAuley et al[115], where an initial transient increase was followed by a statistically significant decrease on EVLWI.
Grensenmann et al[116] investigated the influence of modified prone positioning (135°) on the accuracy of pulse contour-derived calibrated cardiac index and uncalibrated cardiac index in ARDS patient with transpulmonary thermodilution as reference technique. They found that the prone positioning only marginally influences calibrated pulse contour-derived cardiac index measurements, while uncalibrated pulse contour analysis showed a degree of error higher than considered acceptable[116].
Although prone positioning in ARDS setting is generally associated with increased arterial oxygenation, the truth is that patients’ response is quite variable and hard to predict. There are few patients, who after being turned to the prone position show no improvement at all or even a deterioration.
According to arterial blood gas changes after their proning, ARDS patients can be classified as “responders” or not “responders”; “PaO2 responders” are those whose PaO2/FiO2 ratio increases by at least 20% or by ≥ 20 mmHg, whereas PaCO2 responders are those whose PaCO2 decreases by ≥ 1 mmHg. These are the thresholds most selected in previous studies[69,79,102,117-121]. Expected PaCO2 changes are relatively smaller than PaO2 changes because of the different slopes of the content/tension relationship.
ARDS responders can also be classified as “persistent” or “not persistent” based upon whether arterial oxygenation is partially maintained or not respectively when they are turned supine again[37,102]. These patients may exhibit one of three different responses: (1) Display an improved oxygenation compared to prone positioning; (2) maintain a good oxygenation compared to how they were before prone positioning, but not so good as during prone (the majority of the patients); and (3) display a deterioration and return to basal supine oxygenation. The last patients are also called “prone dependent”[122]. When a patient is turned to the prone position repeatedly or the prone position is prolonged, the effect of prone positioning may change with time and be highly variable (during prone position the patient can unpredictably display either improvement or deterioration in oxygenation).
Unfortunately, the factors that influence the time course of alveolar recruitment and the improvement in oxygenation during prone positioning have not been well characterized. These may include the stage of ARDS (early vs late), the cause (pulmonary vs extra-pulmonary), the radiologic pattern (patchy vs diffuse), the severity of hypoxia, the size of initial intrapulmonary shunt, and the patient’s body habitus[22,123-129]. Morphological characteristics from CT scans have also failed to predict the response to prone positioning[130]. Although patient’s response remains still unpredictable, a trial of prone positioning should be performed in all suitable candidates.
Our group has examined the effect of prone positioning in patients with persistent hypoxemia having either hydrostatic pulmonary edema (HPE), ARDS or pulmonary fibrosis[69]. All patients with HPE and 75% of patients with ARDS exhibited improvement of oxygenation when positioned prone. In contrast none of the patients with pulmonary fibrosis responded favorably to prone positioning. We have also found that patients with HPE and early ARDS responded better to prone positioning than patients with late ARDS and pulmonary fibrosis did. This suggests that prone positioning should be applied as early as possible after the onset of the disease when edema, lung recruitability, and absence of structural alterations of the lung are most represented[22,69].
Prone positioning in ARDS patients with refractory hypoxemia has been studied for over three decades and more than 300 articles can be found in PUBMED under the terms “prone position” and “ARDS”. In recent years, several clinical studies have evaluated the safety and efficacy of prone positioning in mechanically ventilated patients with ARDS, but only few were randomized and enrolled an adequate number of patients[23,73,75,76,131]. Former studies on prone positioning had several limitations. Small sample size, initiation of positioning, length of time and type of proning, and the absence of use of lung protective ventilation in conjunction with proning were identified as limitations.
Gattinoni et al[75] studied 304 patients with ARDS and PaO2/FiO2 ratio less than 200 mmHg as an inclusion criterion. Patients were randomized in conventional treatment (in the supine position) and predefined strategy of placing patients in a prone position for six or more hours daily for 10 d. Although prone positioning improved oxygenation of patients, the relative risk of death did not significantly differ between the two study groups: In the prone group as compared with the supine group the relative risk was 0.84 at the end of the study period (95%CI: 0.56 to 1.27), 1.05 at the time of discharge from the intensive care unit (95%CI: 0.84 to 1.32), and 1.06 at six months (95%CI: 0.88 to 1.28). A significant limitation of this study was that no lung protective ventilation protocol was used[75].
Guerin et al[76] included 802 patients with acute respiratory failure and PaO2/FiO2 ratio less than 300 mmHg in a prospective, unblinded, multicenter controlled trial. Patients were randomly assigned to prone positioning, that was applied as early as possible for at least 8 h/d, or to supine positioning. In this study, prone positioning improved oxygenation and reduced the risk of VAP, but no significant difference between the two study groups was evident in regard to mortality. A lung protective ventilation protocol was not used in this study as well[76].
Mancebo et al[132] enrolled 136 patients with severe ARDS (mean PaO2/FiO2 ratio 105 mmHg) and randomized them to prone or supine positioning. In this study, the duration of prone was significantly higher in comparison to the previously mentioned trials. In particular, the prone group was targeted to receive continuous prone ventilation treatment for 20 h/d continuously until the patients were ready for weaning from mechanical ventilation. In addition, a lung protective ventilation protocol was used. According to the results of this trial, prone positioning followed a trend for reduced ICU mortality although the difference was not statistically significant[132].
In The Prone-Supine II study, a multicenter, unblinded, randomized controlled trial, Taccone et al[73] assigned 342 adult patients with ARDS, receiving mechanical ventilation, with a lung protective protocol, to undergo either supine or prone (for 20 h/d) positioning during ventilation. In this study, the long duration of prone positioning failed to demonstrate any benefit on survival in all study patients[73].
The only study to date that showed that prone positioning improves mortality in ARDS patients is the Proning Severe ARDS Patients (PROSEVA) study[23]. The PROSEVA study is a randomized controlled trial designed to determine whether prone-position ventilation, applied early, would improve the outcome in patients with severe ARDS. In this study, 466 patients with severe ARDS (defined as a PaO2/FiO2 ratio < 150 mmHg) underwent either at least 16 h of prone positioning or were left in the supine position after 12 to 24 h of initial conventional mechanical ventilation. The primary outcome that was investigated was the rate of death at 28 d. The unadjusted 28-d mortality rate was 16.0% in the prone group compared with 32.8% in the supine group (P < 0.001)[23]. The distinctively different findings in the PROSEVA study can be attributed to several factors. According to the investigators, the improvement in the outcome found in this study compared to the previous ones was related to the shorter period of enrollment (less than 24 h since ARDS criteria were confirmed), the longer prone-positioning sessions used (the prone position was applied for 73% of the time ascribed to the intervention and was concentrated over a period of a few days) and the lung protective mechanical ventilation protocol than was applied[23].
The findings of the PROSEVA study are in accordance with the conclusions of recent meta-analyses including trials where prone positioning sessions and days of treatment were prolonged together with the use of lung protective ventilation protocol, with or without similar PEEP between the two strategies[12,133-135]. All these meta-analyses showed an overall survival benefit of prone positioning[133,134,136-138]. The benefit on survival in these meta-analyses was mainly evident when prolonged sessions of prone positioning were initiated in combination with small tidal volumes in patients with severe hypoxemia[133,134,136-139]. Finally, in the most recent Cochrane review, Bloomfield et al[140] included a total of nine randomized clinical trials. In this meta-analysis, there was no convincing evidence of benefit nor harm from universal application of prone positioning in adults ARDS patients mechanically ventilated in intensive care units[140]. However, in the same review, in three subgroups, early implementation, prolonged adoption of prone positioning and severe hypoxemia at study entry, prone positioning may confer a statistically significant mortality advantage[140]. The basic characteristics of these meta-analyses are shown in Table 2.
| Meta-analysis | No. of studies included | Total number of patients | Main findings |
| Sud et al[138] | 10 | 1867 | Prone ventilation reduces mortality in patients with severe hypoxemia |
| Gattinoni et al[136] | 4 | 1573 | The individual patient meta-analysis of the four major clinical trials available clearly shows that with prone positioning, the absolute mortality of severely hypoxemic ARDS patients may be reduced by approximately 10% |
| Lee et al[137] | 11 | 2246 | Ventilation in the prone position significantly reduced overall mortality in patients with severe acute respiratory distress syndrome. Sufficient duration of prone positioning was statistically significant in associated with a reduction in overall mortality |
| Beitler et al[134] | 7 | 2119 | Prone positioning was associated with a significant decrease in RR of death only among studies with low baseline tidal volume |
| Tonelli et al[133] | 159 | 20671 | Limited supportive evidence that specific interventions can decrease mortality in ARDS, while low tidal volumes and prone positioning in severe ARDS seem effective |
| (93 with overall mortality reported) | |||
| (44 trials reported mortality as a primary outcome) | |||
| Park et al[139] | 8 | 2141 | Prone positioning tends to reduce the mortality rates in ARDS patients, especially when used in conjunction with a lung protective strategy and longer prone position durations. Prone positioning for ARDS patients should be prioritized over other invasive procedures because related life-threatening complications are rare |
| Bloomfield et al[140] | 9 | 2165 | No convincing evidence of benefit nor harm from universal application of prone positioning in adults with hypoxaemia mechanically ventilated in intensive care units Three subgroups (early implementation of prone positioning, prolonged adoption of prone positioning and severe hypoxaemia at study entry) suggested that prone positioning may confer a statistically significant mortality advantage |
Clinicians intending to use prone positioning therapy face the question of optimal duration of prone positioning sessions, which still remains controversial. Early studies were characterized by short prone positioning session of no more than 10 h, ranging between 1-10 h in the majority of the patients[75,76,134]. Later studies used prolonged session of prone positioning, usually more than 12 h[23,72-75,137,139] showing better results on mortality or morbidity but the majority of them did not achieve statistical significance. In their meta-analysis, Beitler et al[134] stratified analysis by high (≥ 12 h/d) or low (< 12 h/d) proning dose and demonstrated a significant reduction in mortality with high doses (RR = 0.71; 95%CI: 0.56-0.90; P = 0.004) but not low doses (RR = 1.05; 95%CI: 0.92-1.19; P = 0.472)[134]. Lee et al[137] showed a negative trend for overall mortality when the actual duration of prone positioning was longer, but the effect of the duration of prone positioning on mortality did not achieve statistical significance (RC = -0.037; 95%CI: -0.089 to 0.013; P = 0.130)[137].
Thus, although data regarding optimal exact duration of prone positioning is far from being sufficient, it seems that periods of more than 12 h of prone positioning are needed in order to improve outcome. According to our experience and the findings of the PROSEVA study, prolonged duration of proning even more than 24-36 h, or a protocol of short period (i.e., 1-2 h) of supine positioning for daily nursing care between 24-h prone sessions for 3-5 d are safe and seems to improve outcome in patients with severe ARDS under lung protective mechanical ventilation (unpublished preliminary data).
In summary, despite former small non-randomized observational studies not showing any beneficial outcome in regard to prone position in ARDS patients, newer large randomized trials and recent meta-analyses show that prone position, when performed early and in sufficient duration, may improve survival in patients with severe hypoxemia and in patients ventilated with a restrict lung protective ventilation protocol characterized by small tidal volumes.
There are only few absolute contraindications to prone positioning, such as unstable vertebral fractures and unmonitored or significantly increased intracranial pressure. Hemodynamic and cardiac rhythm disturbances are strong relative contraindications, since immediate access for cardiopulmonary resuscitation is limited. Except for conditions that would make proning impractical (e.g., the presence of external fixators), for other relative contraindications (Table 3) one should take into account the team expertise, and potential complications should be weighed against the feasibility of recruiting a potentially life-saving treatment[23,46,81,97,141-146].
| Absolute |
| Unmonitored or significantly increased intracranial pressure |
| Unstable vertebral fractures |
| Relative |
| Difficult airway management |
| Tracheal surgery or sternotomy during the previous 15 d |
| New tracheostomy (less than 24 h) |
| Single anterior chest tube with air leaks |
| Serious facial trauma or facial surgery during the previous 15 d |
| Increased intraocular pressure |
| Hemodynamic instability or recent cardiopulmonary arrest |
| Cardiac pacemaker inserted in the last 2 d |
| Ventricular assist device |
| Intra-aortic balloon pump |
| Deep venous thrombosis treated for less than 2 d |
| Massive hemoptysis requiring an immediate surgical or interventional radiology procedure |
| Continuous dialysis |
| Severe chest wall lesions ± rib fractures |
| Recent cardiothoracic surgery/unstable mediastinum or open chest |
| Multiple trauma with unstabilized fractures |
| Femur, or pelvic fractures ± external pelvic fixation |
| Pregnant women |
| Recent abdominal surgery or stoma formation |
| Kyphoscoliosis |
| Advanced osteoarthritis or rheumatoid arthritis |
| Body weight greater than 135 kg |
Although the safety of proning has long been a concern because of the risk of serious complications (Table 4), data from clinical studies indicate that the maneuver is safe and has a minimal risk profile when performed by skilled personnel and in well-selected patients[23,46,53,54,73,75,76,97,104,117,121,131,132,134,137,139,143-145,147-159]. The use of special devices and beds (e.g., Vollman Proning Device or RotoProne™. Therapy System) can facilitate the mechanics of safe proning[81,142,158]. Manual prone positioning proves to be cost-effective since it can be achieved with a sheet or an assistive device (e.g., Vollman Proning Device). It is a simple technique and allows full access to the patient. The main disadvantage of the method is that it requires additional highly skilled nursing resources. The patient’s size and the number of lines will eventually determine the number of people required for the turn; it can take four or more staff members to accomplish safely. On the other hand, automated prone-positioning needs one man, minimizes risk during turning and can provide continuous rotation if required according to patient’s needs and responses. Unfortunately, the cost of automated prone-positioning beds is very high. Besides, quick access to the patient and abdomen release during mechanical ventilation in prone position are also a concern. To the best of our knowledge, in the literature there are no studies comparing manual and automated prone positioning and the user experience for automated prone positioning remains limited.
| Edema (facial, airway, limbs, thorax) |
| Pressure sores |
| Conjunctival hemorrhage |
| Compression of nerves and retinal vessels |
| Endotracheal tube dislocation (main stem intubation or non-scheduled extubation), obstruction or kinking |
| Airway suctioning difficulty |
| Transient hypotension or oxygen desaturation |
| Worsening gas exchange |
| Pneumothorax |
| Thoracic drain kinking or obstruction |
| Cardiac events |
| Inadvertent dislodging of Swan-Ganz catheter |
| Vascular catheter kinking or removal |
| Vascular catheter malfunction during continuous veno-venous hemofiltration |
| Deep venous thrombosis |
| Urinary bladder catheter or nasogastric feeding tube displacement |
| Enteral nutrition intolerance; vomiting; feeding complications |
| Need for increased sedation or muscle paralysis |
| Difficulty in instituting cardiopulmonary resuscitation |
This review strongly supports the use of prone positioning in the early management of ARDS systematically and not as a rescue maneuver or a last-ditch effort. Large randomized trials and recent meta-analyses show that prone position, when performed early and in sufficient duration, may improve survival in patients with severe ARDS and in patients ventilated with a restrict lung protective ventilation protocol characterized by small tidal volumes. The physiological basis of prone positioning seems to act beneficially in most pathophysiological disorders of ARDS improving hemodynamics, gas exchange and respiratory mechanics. Moreover prone positioning seems to exert an additional beneficial effect against ventilator-induced lung injury. The mechanisms by which prone positioning improves with survival, are likely related to its physiologic effects.
P- Reviewer: Chen XL, Qiu HB S- Editor: Qiu S L- Editor: A E- Editor: Jiao XK
| 1. | Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, Legall JR, Morris A, Spragg R. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4145] [Cited by in RCA: 4151] [Article Influence: 133.9] [Reference Citation Analysis (0)] |
| 2. | Li G, Malinchoc M, Cartin-Ceba R, Venkata CV, Kor DJ, Peters SG, Hubmayr RD, Gajic O. Eight-year trend of acute respiratory distress syndrome: a population-based study in Olmsted County, Minnesota. Am J Respir Crit Care Med. 2011;183:59-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 213] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 3. | Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, Fan E, Camporota L, Slutsky AS. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526-2533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1812] [Cited by in RCA: 4281] [Article Influence: 329.3] [Reference Citation Analysis (0)] |
| 4. | Ashbaugh DG, Bigelow DB, Petty TL, Levine BE. Acute respiratory distress in adults. Lancet. 1967;2:319-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2410] [Cited by in RCA: 2233] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 5. | Linko R, Okkonen M, Pettilä V, Perttilä J, Parviainen I, Ruokonen E, Tenhunen J, Ala-Kokko T, Varpula T. Acute respiratory failure in intensive care units. FINNALI: a prospective cohort study. Intensive Care Med. 2009;35:1352-1361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 97] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 6. | Villar J, Blanco J, Añón JM, Santos-Bouza A, Blanch L, Ambrós A, Gandía F, Carriedo D, Mosteiro F, Basaldúa S. The ALIEN study: incidence and outcome of acute respiratory distress syndrome in the era of lung protective ventilation. Intensive Care Med. 2011;37:1932-1941. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 376] [Cited by in RCA: 416] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 7. | Sigurdsson MI, Sigvaldason K, Gunnarsson TS, Moller A, Sigurdsson GH. Acute respiratory distress syndrome: nationwide changes in incidence, treatment and mortality over 23 years. Acta Anaesthesiol Scand. 2013;57:37-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 8. | Esteban A, Ferguson ND, Meade MO, Frutos-Vivar F, Apezteguia C, Brochard L, Raymondos K, Nin N, Hurtado J, Tomicic V. Evolution of mechanical ventilation in response to clinical research. Am J Respir Crit Care Med. 2008;177:170-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 430] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 9. | Phua J, Badia JR, Adhikari NK, Friedrich JO, Fowler RA, Singh JM, Scales DC, Stather DR, Li A, Jones A. Has mortality from acute respiratory distress syndrome decreased over time?: A systematic review. Am J Respir Crit Care Med. 2009;179:220-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 517] [Cited by in RCA: 533] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 10. | Villar J, Sulemanji D, Kacmarek RM. The acute respiratory distress syndrome: incidence and mortality, has it changed? Curr Opin Crit Care. 2014;20:3-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 135] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 11. | Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. The Acute Respiratory Distress Syndrome Network. N Engl J Med. 2000;342:1301-1308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8487] [Cited by in RCA: 8319] [Article Influence: 332.8] [Reference Citation Analysis (3)] |
| 12. | Putensen C, Theuerkauf N, Zinserling J, Wrigge H, Pelosi P. Meta-analysis: ventilation strategies and outcomes of the acute respiratory distress syndrome and acute lung injury. Ann Intern Med. 2009;151:566-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 240] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 13. | Fish E, Novack V, Banner-Goodspeed VM, Sarge T, Loring S, Talmor D. The Esophageal Pressure-Guided Ventilation 2 (EPVent2) trial protocol: a multicentre, randomised clinical trial of mechanical ventilation guided by transpulmonary pressure. BMJ Open. 2014;4:e006356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 14. | Talmor DS, Fessler HE. Are esophageal pressure measurements important in clinical decision-making in mechanically ventilated patients? Respir Care. 2010;55:162-172; discussion 172-174. [PubMed] |
| 15. | Hedenstierna G. Esophageal pressure: benefit and limitations. Minerva Anestesiol. 2012;78:959-966. [PubMed] |
| 16. | Ochiai R. Mechanical ventilation of acute respiratory distress syndrome. J Intensive Care. 2015;3:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 17. | Amato MB, Meade MO, Slutsky AS, Brochard L, Costa EL, Schoenfeld DA, Stewart TE, Briel M, Talmor D, Mercat A. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372:747-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1397] [Cited by in RCA: 1596] [Article Influence: 159.6] [Reference Citation Analysis (2)] |
| 18. | Briel M, Meade M, Mercat A, Brower RG, Talmor D, Walter SD, Slutsky AS, Pullenayegum E, Zhou Q, Cook D. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA. 2010;303:865-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1140] [Cited by in RCA: 933] [Article Influence: 62.2] [Reference Citation Analysis (0)] |
| 19. | Santa Cruz R, Rojas JI, Nervi R, Heredia R, Ciapponi A. High versus low positive end-expiratory pressure (PEEP) levels for mechanically ventilated adult patients with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev. 2013;6:CD009098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 20. | Pelosi P, Tubiolo D, Mascheroni D, Vicardi P, Crotti S, Valenza F, Gattinoni L. Effects of the prone position on respiratory mechanics and gas exchange during acute lung injury. Am J Respir Crit Care Med. 1998;157:387-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 296] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 21. | Mure M, Martling CR, Lindahl SG. Dramatic effect on oxygenation in patients with severe acute lung insufficiency treated in the prone position. Crit Care Med. 1997;25:1539-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 120] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 22. | Blanch L, Mancebo J, Perez M, Martinez M, Mas A, Betbese AJ, Joseph D, Ballús J, Lucangelo U, Bak E. Short-term effects of prone position in critically ill patients with acute respiratory distress syndrome. Intensive Care Med. 1997;23:1033-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 121] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Guérin C, Reignier J, Richard JC, Beuret P, Gacouin A, Boulain T, Mercier E, Badet M, Mercat A, Baudin O. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368:2159-2168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2391] [Cited by in RCA: 2537] [Article Influence: 211.4] [Reference Citation Analysis (0)] |
| 24. | Wiedemann HP, Wheeler AP, Bernard GR, Thompson BT, Hayden D, deBoisblanc B, Connors AF, Hite RD, Harabin AL. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354:2564-2575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2310] [Cited by in RCA: 2303] [Article Influence: 121.2] [Reference Citation Analysis (0)] |
| 25. | Papazian L, Forel JM, Gacouin A, Penot-Ragon C, Perrin G, Loundou A, Jaber S, Arnal JM, Perez D, Seghboyan JM. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363:1107-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1562] [Cited by in RCA: 1551] [Article Influence: 103.4] [Reference Citation Analysis (0)] |
| 26. | Ferguson ND, Cook DJ, Guyatt GH, Mehta S, Hand L, Austin P, Zhou Q, Matte A, Walter SD, Lamontagne F. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med. 2013;368:795-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 574] [Cited by in RCA: 537] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 27. | Young D, Lamb SE, Shah S, MacKenzie I, Tunnicliffe W, Lall R, Rowan K, Cuthbertson BH. High-frequency oscillation for acute respiratory distress syndrome. N Engl J Med. 2013;368:806-813. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 402] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 28. | Meduri GU, Marik PE, Chrousos GP, Pastores SM, Arlt W, Beishuizen A, Bokhari F, Zaloga G, Annane D. Steroid treatment in ARDS: a critical appraisal of the ARDS network trial and the recent literature. Intensive Care Med. 2008;34:61-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 116] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 29. | Bryan AC. Conference on the scientific basis of respiratory therapy. Pulmonary physiotherapy in the pediatric age group. Comments of a devil’s advocate. Am Rev Respir Dis. 1974;110:143-144. [PubMed] |
| 30. | Piehl MA, Brown RS. Use of extreme position changes in acute respiratory failure. Crit Care Med. 1976;4:13-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 261] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 31. | Douglas WW, Rehder K, Beynen FM, Sessler AD, Marsh HM. Improved oxygenation in patients with acute respiratory failure: the prone position. Am Rev Respir Dis. 1977;115:559-566. [PubMed] |
| 32. | Maunder RJ, Shuman WP, McHugh JW, Marglin SI, Butler J. Preservation of normal lung regions in the adult respiratory distress syndrome. Analysis by computed tomography. JAMA. 1986;255:2463-2465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 170] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 33. | Gattinoni L, Presenti A, Torresin A, Baglioni S, Rivolta M, Rossi F, Scarani F, Marcolin R, Cappelletti G. Adult respiratory distress syndrome profiles by computed tomography. J Thorac Imaging. 1986;1:25-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 164] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 34. | Gattinoni L, Pesenti A, Avalli L, Rossi F, Bombino M. Pressure-volume curve of total respiratory system in acute respiratory failure. Computed tomographic scan study. Am Rev Respir Dis. 1987;136:730-736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 526] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 35. | Gattinoni L, Pesenti A. The concept of “baby lung”. Intensive Care Med. 2005;31:776-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 627] [Cited by in RCA: 492] [Article Influence: 24.6] [Reference Citation Analysis (1)] |
| 36. | Gattinoni L, Pesenti A, Carlesso E. Body position changes redistribute lung computed-tomographic density in patients with acute respiratory failure: impact and clinical fallout through the following 20 years. Intensive Care Med. 2013;39:1909-1915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 37. | Langer M, Mascheroni D, Marcolin R, Gattinoni L. The prone position in ARDS patients. A clinical study. Chest. 1988;94:103-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 216] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 38. | Gattinoni L, Pelosi P, Vitale G, Pesenti A, D’Andrea L, Mascheroni D. Body position changes redistribute lung computed-tomographic density in patients with acute respiratory failure. Anesthesiology. 1991;74:15-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 329] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 39. | Bone RC. The ARDS lung. New insights from computed tomography. JAMA. 1993;269:2134-2135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 40. | Pelosi P, D’Andrea L, Vitale G, Pesenti A, Gattinoni L. Vertical gradient of regional lung inflation in adult respiratory distress syndrome. Am J Respir Crit Care Med. 1994;149:8-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 296] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 41. | Gattinoni L, D’Andrea L, Pelosi P, Vitale G, Pesenti A, Fumagalli R. Regional effects and mechanism of positive end-expiratory pressure in early adult respiratory distress syndrome. JAMA. 1993;269:2122-2127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 233] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 42. | Glenny RW, Lamm WJ, Albert RK, Robertson HT. Gravity is a minor determinant of pulmonary blood flow distribution. J Appl Physiol (1985). 1991;71:620-629. [PubMed] |
| 43. | Pelosi P, Crotti S, Brazzi L, Gattinoni L. Computed tomography in adult respiratory distress syndrome: what has it taught us? Eur Respir J. 1996;9:1055-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 95] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 44. | Hubmayr RD. Perspective on lung injury and recruitment: a skeptical look at the opening and collapse story. Am J Respir Crit Care Med. 2002;165:1647-1653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 211] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 45. | Rouby JJ, Puybasset L, Nieszkowska A, Lu Q. Acute respiratory distress syndrome: lessons from computed tomography of the whole lung. Crit Care Med. 2003;31:S285-S295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 168] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 46. | Gattinoni L, Taccone P, Carlesso E, Marini JJ. Prone position in acute respiratory distress syndrome. Rationale, indications, and limits. Am J Respir Crit Care Med. 2013;188:1286-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 288] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 47. | Albert RK, Hubmayr RD. The prone position eliminates compression of the lungs by the heart. Am J Respir Crit Care Med. 2000;161:1660-1665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 234] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 48. | Malbouisson LM, Busch CJ, Puybasset L, Lu Q, Cluzel P, Rouby JJ. Role of the heart in the loss of aeration characterizing lower lobes in acute respiratory distress syndrome. CT Scan ARDS Study Group. Am J Respir Crit Care Med. 2000;161:2005-2012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 102] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 49. | Mure M, Glenny RW, Domino KB, Hlastala MP. Pulmonary gas exchange improves in the prone position with abdominal distension. Am J Respir Crit Care Med. 1998;157:1785-1790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 46] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 50. | Galiatsou E, Kostanti E, Svarna E, Kitsakos A, Koulouras V, Efremidis SC, Nakos G. Prone position augments recruitment and prevents alveolar overinflation in acute lung injury. Am J Respir Crit Care Med. 2006;174:187-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 158] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 51. | Guerin C, Baboi L, Richard JC. Mechanisms of the effects of prone positioning in acute respiratory distress syndrome. Intensive Care Med. 2014;40:1634-1642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 52. | Tawhai MH, Nash MP, Lin CL, Hoffman EA. Supine and prone differences in regional lung density and pleural pressure gradients in the human lung with constant shape. J Appl Physiol (1985). 2009;107:912-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 116] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 53. | Guérin C. Prone ventilation in acute respiratory distress syndrome. Eur Respir Rev. 2014;23:249-257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 54. | Pelosi P, Brazzi L, Gattinoni L. Prone position in acute respiratory distress syndrome. Eur Respir J. 2002;20:1017-1028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 176] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 55. | Arborelius M, Lundin G, Svanberg L, Defares JG. Influence of unilateral hypoxia on blood flow through the lungs in man in lateral position. J Appl Physiol. 1960;15:595-597. [PubMed] |
| 56. | Greene R, Zapol WM, Snider MT, Reid L, Snow R, O’Connell RS, Novelline RA. Early bedside detection of pulmonary vascular occlusion during acute respiratory failure. Am Rev Respir Dis. 1981;124:593-601. [PubMed] |
| 57. | Gattinoni L, Pelosi P, Pesenti A, Brazzi L, Vitale G, Moretto A, Crespi A, Tagliabue M. CT scan in ARDS: clinical and physiopathological insights. Acta Anaesthesiol Scand Suppl. 1991;95:87-94; discussion 94-6. [PubMed] |
| 58. | Richard JC, Janier M, Lavenne F, Berthier V, Lebars D, Annat G, Decailliot F, Guerin C. Effect of position, nitric oxide, and almitrine on lung perfusion in a porcine model of acute lung injury. J Appl Physiol (1985). 2002;93:2181-2191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 59. | Nyrén S, Mure M, Jacobsson H, Larsson SA, Lindahl SG. Pulmonary perfusion is more uniform in the prone than in the supine position: scintigraphy in healthy humans. J Appl Physiol (1985). 1999;86:1135-1141. [PubMed] |
| 60. | Guérin C. Prone position. Curr Opin Crit Care. 2014;20:92-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 61. | Wiener CM, Kirk W, Albert RK. Prone position reverses gravitational distribution of perfusion in dog lungs with oleic acid-induced injury. J Appl Physiol (1985). 1990;68:1386-1392. [PubMed] |
| 62. | Lamm WJ, Graham MM, Albert RK. Mechanism by which the prone position improves oxygenation in acute lung injury. Am J Respir Crit Care Med. 1994;150:184-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 298] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 63. | Guérin C. Ventilation in the prone position in patients with acute lung injury/acute respiratory distress syndrome. Curr Opin Crit Care. 2006;12:50-54. [PubMed] |
| 64. | Petersson J, Rohdin M, Sánchez-Crespo A, Nyrén S, Jacobsson H, Larsson SA, Lindahl SG, Linnarsson D, Glenny RW, Mure M. Paradoxical redistribution of pulmonary blood flow in prone and supine humans exposed to hypergravity. J Appl Physiol (1985). 2006;100:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 65. | Mure M, Domino KB, Lindahl SG, Hlastala MP, Altemeier WA, Glenny RW. Regional ventilation-perfusion distribution is more uniform in the prone position. J Appl Physiol (1985). 2000;88:1076-1083. [PubMed] |
| 66. | Cakar N, der Kloot TV, Youngblood M, Adams A, Nahum A. Oxygenation response to a recruitment maneuver during supine and prone positions in an oleic acid-induced lung injury model. Am J Respir Crit Care Med. 2000;161:1949-1956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 71] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 67. | Rival G, Patry C, Floret N, Navellou JC, Belle E, Capellier G. Prone position and recruitment manoeuvre: the combined effect improves oxygenation. Crit Care. 2011;15:R125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 68. | Oczenski W, Hörmann C, Keller C, Lorenzl N, Kepka A, Schwarz S, Fitzgerald RD. Recruitment maneuvers during prone positioning in patients with acute respiratory distress syndrome. Crit Care Med. 2005;33:54-61; quiz 62. [PubMed] |
| 69. | Nakos G, Tsangaris I, Kostanti E, Nathanail C, Lachana A, Koulouras V, Kastani D. Effect of the prone position on patients with hydrostatic pulmonary edema compared with patients with acute respiratory distress syndrome and pulmonary fibrosis. Am J Respir Crit Care Med. 2000;161:360-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 83] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Wiener CM, McKenna WJ, Myers MJ, Lavender JP, Hughes JM. Left lower lobe ventilation is reduced in patients with cardiomegaly in the supine but not the prone position. Am Rev Respir Dis. 1990;141:150-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 71. | Petersson J, Ax M, Frey J, Sánchez-Crespo A, Lindahl SG, Mure M. Positive end-expiratory pressure redistributes regional blood flow and ventilation differently in supine and prone humans. Anesthesiology. 2010;113:1361-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 72. | Gainnier M, Michelet P, Thirion X, Arnal JM, Sainty JM, Papazian L. Prone position and positive end-expiratory pressure in acute respiratory distress syndrome. Crit Care Med. 2003;31:2719-2726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 57] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 73. | Taccone P, Pesenti A, Latini R, Polli F, Vagginelli F, Mietto C, Caspani L, Raimondi F, Bordone G, Iapichino G. Prone positioning in patients with moderate and severe acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2009;302:1977-1984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 364] [Cited by in RCA: 348] [Article Influence: 21.8] [Reference Citation Analysis (1)] |
| 74. | Gattinoni L, Caironi P. Prone positioning: beyond physiology. Anesthesiology. 2010;113:1262-1264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 75. | Gattinoni L, Tognoni G, Pesenti A, Taccone P, Mascheroni D, Labarta V, Malacrida R, Di Giulio P, Fumagalli R, Pelosi P. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med. 2001;345:568-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 861] [Cited by in RCA: 743] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 76. | Guerin C, Gaillard S, Lemasson S, Ayzac L, Girard R, Beuret P, Palmier B, Le QV, Sirodot M, Rosselli S. Effects of systematic prone positioning in hypoxemic acute respiratory failure: a randomized controlled trial. JAMA. 2004;292:2379-2387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 376] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 77. | Gattinoni L, Bombino M, Pelosi P, Lissoni A, Pesenti A, Fumagalli R, Tagliabue M. Lung structure and function in different stages of severe adult respiratory distress syndrome. JAMA. 1994;271:1772-1779. [PubMed] [DOI] [Full Text] |
| 78. | Kallet RH, Alonso JA, Pittet JF, Matthay MA. Prognostic value of the pulmonary dead-space fraction during the first 6 days of acute respiratory distress syndrome. Respir Care. 2004;49:1008-1014. [PubMed] |
| 79. | Gattinoni L, Vagginelli F, Carlesso E, Taccone P, Conte V, Chiumello D, Valenza F, Caironi P, Pesenti A. Decrease in PaCO2 with prone position is predictive of improved outcome in acute respiratory distress syndrome. Crit Care Med. 2003;31:2727-2733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 189] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 80. | Protti A, Chiumello D, Cressoni M, Carlesso E, Mietto C, Berto V, Lazzerini M, Quintel M, Gattinoni L. Relationship between gas exchange response to prone position and lung recruitability during acute respiratory failure. Intensive Care Med. 2009;35:1011-1017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 81. | Dirkes S, Dickinson S, Havey R, O’brien D. Prone positioning: is it safe and effective? Crit Care Nurs Q. 2012;35:64-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 82. | Servillo G, Roupie E, De Robertis E, Rossano F, Brochard L, Lemaire F, Tufano R. Effects of ventilation in ventral decubitus position on respiratory mechanics in adult respiratory distress syndrome. Intensive Care Med. 1997;23:1219-1224. [PubMed] |
| 83. | Guerin C, Badet M, Rosselli S, Heyer L, Sab JM, Langevin B, Philit F, Fournier G, Robert D. Effects of prone position on alveolar recruitment and oxygenation in acute lung injury. Intensive Care Med. 1999;25:1222-1230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 81] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 84. | Slutsky AS. Lung injury caused by mechanical ventilation. Chest. 1999;116:9S-15S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 412] [Cited by in RCA: 385] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 85. | Bilek AM, Dee KC, Gaver DP. Mechanisms of surface-tension-induced epithelial cell damage in a model of pulmonary airway reopening. J Appl Physiol (1985). 2003;94:770-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 210] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 86. | Albert RK. The role of ventilation-induced surfactant dysfunction and atelectasis in causing acute respiratory distress syndrome. Am J Respir Crit Care Med. 2012;185:702-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 98] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 87. | Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126-2136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1169] [Cited by in RCA: 1346] [Article Influence: 112.2] [Reference Citation Analysis (0)] |
| 88. | Gattinoni L, Protti A. Ventilation in the prone position: for some but not for all? CMAJ. 2008;178:1174-1176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 89. | Chiumello D, Carlesso E, Cadringher P, Caironi P, Valenza F, Polli F, Tallarini F, Cozzi P, Cressoni M, Colombo A. Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am J Respir Crit Care Med. 2008;178:346-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 453] [Cited by in RCA: 495] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 90. | Gattinoni L, Protti A, Caironi P, Carlesso E. Ventilator-induced lung injury: the anatomical and physiological framework. Crit Care Med. 2010;38:S539-S548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 150] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 91. | Gattinoni L, Carlesso E, Caironi P. Stress and strain within the lung. Curr Opin Crit Care. 2012;18:42-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 92] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 92. | Valenza F, Guglielmi M, Maffioletti M, Tedesco C, Maccagni P, Fossali T, Aletti G, Porro GA, Irace M, Carlesso E. Prone position delays the progression of ventilator-induced lung injury in rats: does lung strain distribution play a role? Crit Care Med. 2005;33:361-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 96] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 93. | Broccard A, Shapiro RS, Schmitz LL, Adams AB, Nahum A, Marini JJ. Prone positioning attenuates and redistributes ventilator-induced lung injury in dogs. Crit Care Med. 2000;28:295-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 176] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 94. | Du HL, Yamada Y, Orii R, Suzuki S, Sawamura S, Suwa K, Hanaoka K. Beneficial effects of the prone position on the incidence of barotrauma in oleic acid-induced lung injury under continuous positive pressure ventilation. Acta Anaesthesiol Scand. 1997;41:701-707. [PubMed] |
| 95. | Broccard AF, Shapiro RS, Schmitz LL, Ravenscraft SA, Marini JJ. Influence of prone position on the extent and distribution of lung injury in a high tidal volume oleic acid model of acute respiratory distress syndrome. Crit Care Med. 1997;25:16-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 132] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 96. | Cornejo RA, Díaz JC, Tobar EA, Bruhn AR, Ramos CA, González RA, Repetto CA, Romero CM, Gálvez LR, Llanos O. Effects of prone positioning on lung protection in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2013;188:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 163] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 97. | Roche-Campo F, Aguirre-Bermeo H, Mancebo J. Prone positioning in acute respiratory distress syndrome (ARDS): when and how? Presse Med. 2011;40:e585-e594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 98. | Papazian L, Gainnier M, Marin V, Donati S, Arnal JM, Demory D, Roch A, Forel JM, Bongrand P, Brégeon F. Comparison of prone positioning and high-frequency oscillatory ventilation in patients with acute respiratory distress syndrome. Crit Care Med. 2005;33:2162-2171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 93] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 99. | Nakos G, Batistatou A, Galiatsou E, Konstanti E, Koulouras V, Kanavaros P, Doulis A, Kitsakos A, Karachaliou A, Lekka ME. Lung and ‘end organ’ injury due to mechanical ventilation in animals: comparison between the prone and supine positions. Crit Care. 2006;10:R38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 100. | Park MS, He Q, Edwards MG, Sergew A, Riches DW, Albert RK, Douglas IS. Mitogen-activated protein kinase phosphatase-1 modulates regional effects of injurious mechanical ventilation in rodent lungs. Am J Respir Crit Care Med. 2012;186:72-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 101. | Mackenzie CF. Anatomy, physiology, and pathology of the prone position and postural drainage. Crit Care Med. 2001;29:1084-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 102. | Chatte G, Sab JM, Dubois JM, Sirodot M, Gaussorgues P, Robert D. Prone position in mechanically ventilated patients with severe acute respiratory failure. Am J Respir Crit Care Med. 1997;155:473-478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 215] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 103. | Gillart T, Bazin JE, Guelon D, Constantin JM, Mansoor O, Conio N, Schoeffler P. [Effect of bronchial drainage on the improvement in gas exchange observed in ventral decubitus in ARDS]. Ann Fr Anesth Reanim. 2000;19:156-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 104. | Voggenreiter G, Aufmkolk M, Stiletto RJ, Baacke MG, Waydhas C, Ose C, Bock E, Gotzen L, Obertacke U, Nast-Kolb D. Prone positioning improves oxygenation in post-traumatic lung injury--a prospective randomized trial. J Trauma. 2005;59:333-341; discussion 341-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 77] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 105. | Drakulovic MB, Torres A, Bauer TT, Nicolas JM, Nogué S, Ferrer M. Supine body position as a risk factor for nosocomial pneumonia in mechanically ventilated patients: a randomised trial. Lancet. 1999;354:1851-1858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 840] [Cited by in RCA: 717] [Article Influence: 27.6] [Reference Citation Analysis (0)] |
| 106. | Li Bassi G, Zanella A, Cressoni M, Stylianou M, Kolobow T. Following tracheal intubation, mucus flow is reversed in the semirecumbent position: possible role in the pathogenesis of ventilator-associated pneumonia. Crit Care Med. 2008;36:518-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 65] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 107. | Li Bassi G, Torres A. Ventilator-associated pneumonia: role of positioning. Curr Opin Crit Care. 2011;17:57-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 108. | Pappert D, Rossaint R, Slama K, Grüning T, Falke KJ. Influence of positioning on ventilation-perfusion relationships in severe adult respiratory distress syndrome. Chest. 1994;106:1511-1516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 189] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 109. | Sinclair SE, Altemeier WA, Matute-Bello G, Chi EY. Augmented lung injury due to interaction between hyperoxia and mechanical ventilation. Crit Care Med. 2004;32:2496-2501. [PubMed] |
| 110. | Vieillard-Baron A, Charron C, Caille V, Belliard G, Page B, Jardin F. Prone positioning unloads the right ventricle in severe ARDS. Chest. 2007;132:1440-1446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 172] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 111. | Jozwiak M, Teboul JL, Anguel N, Persichini R, Silva S, Chemla D, Richard C, Monnet X. Beneficial hemodynamic effects of prone positioning in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2013;188:1428-1433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 150] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 112. | Repessé X, Charron C, Vieillard-Baron A. Acute cor pulmonale in ARDS: rationale for protecting the right ventricle. Chest. 2015;147:259-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 109] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 113. | Bull TM, Clark B, McFann K, Moss M. Pulmonary vascular dysfunction is associated with poor outcomes in patients with acute lung injury. Am J Respir Crit Care Med. 2010;182:1123-1128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 162] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 114. | Brücken U, Grensemann J, Wappler F, Sakka SG. Influence of prone positioning on the measurement of transpulmonary thermodilution-derived variables in critically ill patients. Acta Anaesthesiol Scand. 2011;55:1061-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 115. | McAuley DF, Giles S, Fichter H, Perkins GD, Gao F. What is the optimal duration of ventilation in the prone position in acute lung injury and acute respiratory distress syndrome? Intensive Care Med. 2002;28:414-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 62] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 116. | Grensemann J, Bruecken U, Treszl A, Wappler F, Sakka SG. The influence of prone positioning on the accuracy of calibrated and uncalibrated pulse contour-derived cardiac index measurements. Anesth Analg. 2013;116:820-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 117. | De Jong A, Molinari N, Sebbane M, Prades A, Futier E, Jung B, Chanques G, Jaber S. Feasibility and effectiveness of prone position in morbidly obese patients with ARDS: a case-control clinical study. Chest. 2013;143:1554-1561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 118. | Albert RK, Keniston A, Baboi L, Ayzac L, Guérin C. Prone position-induced improvement in gas exchange does not predict improved survival in the acute respiratory distress syndrome. Am J Respir Crit Care Med. 2014;189:494-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 137] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 119. | Papazian L, Paladini MH, Bregeon F, Huiart L, Thirion X, Saux P, Jammes Y, Auffray JP. Is a short trial of prone positioning sufficient to predict the improvement in oxygenation in patients with acute respiratory distress syndrome? Intensive Care Med. 2001;27:1044-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 120. | Mebazaa MS, Abid N, Frikha N, Mestiri T, Ben Ammar MS. [The prone position in acute respiratory distress syndrome: a critical systematic review]. Ann Fr Anesth Reanim. 2007;26:307-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 121. | Kopterides P, Siempos II, Armaganidis A. Prone positioning in hypoxemic respiratory failure: meta-analysis of randomized controlled trials. J Crit Care. 2009;24:89-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 52] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 122. | Marik PE, Iglesias J. A “prone dependent” patient with severe adult respiratory distress syndrome. Crit Care Med. 1997;25:1085-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 123. | Fessler HE, Talmor DS. Should prone positioning be routinely used for lung protection during mechanical ventilation? Respir Care. 2010;55:88-99. [PubMed] |
| 124. | Collins SR, Blank RS. Approaches to refractory hypoxemia in acute respiratory distress syndrome: current understanding, evidence, and debate. Respir Care. 2011;56:1573-1582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 125. | Lim CM, Kim EK, Lee JS, Shim TS, Lee SD, Koh Y, Kim WS, Kim DS, Kim WD. Comparison of the response to the prone position between pulmonary and extrapulmonary acute respiratory distress syndrome. Intensive Care Med. 2001;27:477-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 126. | Lee DL, Chiang HT, Lin SL, Ger LP, Kun MH, Huang YC. Prone-position ventilation induces sustained improvement in oxygenation in patients with acute respiratory distress syndrome who have a large shunt. Crit Care Med. 2002;30:1446-1452. [PubMed] |
| 127. | Reutershan J, Schmitt A, Dietz K, Unertl K, Fretschner R. Alveolar recruitment during prone position: time matters. Clin Sci (Lond). 2006;110:655-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 128. | Jolliet P, Bulpa P, Chevrolet JC. Effects of the prone position on gas exchange and hemodynamics in severe acute respiratory distress syndrome. Crit Care Med. 1998;26:1977-1985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 93] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 129. | Rialp G, Betbesé AJ, Pérez-Márquez M, Mancebo J. Short-term effects of inhaled nitric oxide and prone position in pulmonary and extrapulmonary acute respiratory distress syndrome. Am J Respir Crit Care Med. 2001;164:243-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 48] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 130. | Papazian L, Paladini MH, Bregeon F, Thirion X, Durieux O, Gainnier M, Huiart L, Agostini S, Auffray JP. Can the tomographic aspect characteristics of patients presenting with acute respiratory distress syndrome predict improvement in oxygenation-related response to the prone position? Anesthesiology. 2002;97:599-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 31] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 131. | Girard R, Baboi L, Ayzac L, Richard JC, Guérin C. The impact of patient positioning on pressure ulcers in patients with severe ARDS: results from a multicentre randomised controlled trial on prone positioning. Intensive Care Med. 2014;40:397-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 132. | Mancebo J, Fernández R, Blanch L, Rialp G, Gordo F, Ferrer M, Rodríguez F, Garro P, Ricart P, Vallverdú I. A multicenter trial of prolonged prone ventilation in severe acute respiratory distress syndrome. Am J Respir Crit Care Med. 2006;173:1233-1239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 325] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 133. | Tonelli AR, Zein J, Adams J, Ioannidis JP. Effects of interventions on survival in acute respiratory distress syndrome: an umbrella review of 159 published randomized trials and 29 meta-analyses. Intensive Care Med. 2014;40:769-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 103] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 134. | Beitler JR, Shaefi S, Montesi SB, Devlin A, Loring SH, Talmor D, Malhotra A. Prone positioning reduces mortality from acute respiratory distress syndrome in the low tidal volume era: a meta-analysis. Intensive Care Med. 2014;40:332-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 131] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 135. | Alsaghir AH, Martin CM. Effect of prone positioning in patients with acute respiratory distress syndrome: a meta-analysis. Crit Care Med. 2008;36:603-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 79] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 136. | Gattinoni L, Carlesso E, Taccone P, Polli F, Guérin C, Mancebo J. Prone positioning improves survival in severe ARDS: a pathophysiologic review and individual patient meta-analysis. Minerva Anestesiol. 2010;76:448-454. [PubMed] |
| 137. | Lee JM, Bae W, Lee YJ, Cho YJ. The efficacy and safety of prone positional ventilation in acute respiratory distress syndrome: updated study-level meta-analysis of 11 randomized controlled trials. Crit Care Med. 2014;42:1252-1262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 118] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 138. | Sud S, Friedrich JO, Taccone P, Polli F, Adhikari NK, Latini R, Pesenti A, Guérin C, Mancebo J, Curley MA. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med. 2010;36:585-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 353] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 139. | Park SY, Kim HJ, Yoo KH, Park YB, Kim SW, Lee SJ, Kim EK, Kim JH, Kim YH, Moon JY. The efficacy and safety of prone positioning in adults patients with acute respiratory distress syndrome: a meta-analysis of randomized controlled trials. J Thorac Dis. 2015;7:356-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 140. | Bloomfield R, Noble DW, Sudlow A. Prone position for acute respiratory failure in adults. Cochrane Database Syst Rev. 2015;11:CD008095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 83] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 141. | Modrykamien AM, Gupta P. The acute respiratory distress syndrome. Proc (Bayl Univ Med Cent). 2015;28:163-171. [PubMed] |
| 142. | Vollman KM. Prone positioning for the ARDS patient. Dimens Crit Care Nurs. 1997;16:184-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 143. | Benson AB, Albert RK. Prone positioning for acute respiratory distress syndrome. Clin Chest Med. 2014;35:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 144. | Henderson WR, Griesdale DE, Dominelli P, Ronco JJ. Does prone positioning improve oxygenation and reduce mortality in patients with acute respiratory distress syndrome? Can Respir J. 2014;21:213-215. [PubMed] |
| 145. | Athota KP, Millar D, Branson RD, Tsuei BJ. A practical approach to the use of prone therapy in acute respiratory distress syndrome. Expert Rev Respir Med. 2014;8:453-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (1)] |
| 146. | Samanta S, Samanta S, Wig J, Baronia AK. How safe is the prone position in acute respiratory distress syndrome at late pregnancy? Am J Emerg Med. 2014;32:687.e1-687.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 147. | Guérin C, Mancebo J. Prone positioning and neuromuscular blocking agents are part of standard care in severe ARDS patients: yes. Intensive Care Med. 2015;41:2195-2197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 148. | Sud S, Friedrich JO, Adhikari NK, Taccone P, Mancebo J, Polli F, Latini R, Pesenti A, Curley MA, Fernandez R. Effect of prone positioning during mechanical ventilation on mortality among patients with acute respiratory distress syndrome: a systematic review and meta-analysis. CMAJ. 2014;186:E381-E390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 182] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 149. | Tekwani SS, Murugan R. ‘To prone or not to prone’ in severe ARDS: questions answered, but others remain. Crit Care. 2014;18:305. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 150. | Soo Hoo GW. In prone ventilation, one good turn deserves another. N Engl J Med. 2013;368:2227-2228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 151. | Prone positioning can save lives. BMJ. 2013;346:f3302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 152. | Hale DF, Cannon JW, Batchinsky AI, Cancio LC, Aden JK, White CE, Renz EM, Blackbourne LH, Chung KK. Prone positioning improves oxygenation in adult burn patients with severe acute respiratory distress syndrome. J Trauma Acute Care Surg. 2012;72:1634-1639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 153. | Abroug F, Ouanes-Besbes L, Dachraoui F, Ouanes I, Brochard L. An updated study-level meta-analysis of randomised controlled trials on proning in ARDS and acute lung injury. Crit Care. 2011;15:R6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 154. | Wright AD, Flynn M. Using the prone position for ventilated patients with respiratory failure: a review. Nurs Crit Care. 2011;16:19-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 155. | Sud S, Sud M, Friedrich JO, Adhikari NK. Effect of mechanical ventilation in the prone position on clinical outcomes in patients with acute hypoxemic respiratory failure: a systematic review and meta-analysis. CMAJ. 2008;178:1153-1161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 156. | Fernandez R, Trenchs X, Klamburg J, Castedo J, Serrano JM, Besso G, Tirapu JP, Santos A, Mas A, Parraga M, Jubert P, Frutos F, Añon JM, Garcia M, Rodriguez F, Yebenes JC, Lopez MJ. Prone positioning in acute respiratory distress syndrome: a multicenter randomized clinical trial. Intensive Care Med. 2008;34:1487-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 107] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 157. | Abroug F, Ouanes-Besbes L, Elatrous S, Brochard L. The effect of prone positioning in acute respiratory distress syndrome or acute lung injury: a meta-analysis. Areas of uncertainty and recommendations for research. Intensive Care Med. 2008;34:1002-1011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 158. | Davis JW, Lemaster DM, Moore EC, Eghbalieh B, Bilello JF, Townsend RN, Parks SN, Veneman WL. Prone ventilation in trauma or surgical patients with acute lung injury and adult respiratory distress syndrome: is it beneficial? J Trauma. 2007;62:1201-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 159. | Agrawal SP, Goel AD. Prone position ventilation in Acute Respiratory Distress Syndrome: An overview of the evidences. Indian J Anaesth. 2015;59:246-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |