Published online Feb 4, 2014. doi: 10.5492/wjccm.v3.i1.8
Revised: October 30, 2013
Accepted: November 18, 2013
Published online: February 4, 2014
Processing time: 236 Days and 21.2 Hours
Pneumothorax is a potentially lethal complication associated with mechanical ventilation. Most of the patients with pneumothorax from mechanical ventilation have underlying lung diseases; pneumothorax is rare in intubated patients with normal lungs. Tension pneumothorax is more common in ventilated patients with prompt recognition and treatment of pneumothorax being important to minimize morbidity and mortality. Underlying lung diseases are associated with ventilator-related pneumothorax with pneumothoraces occurring most commonly during the early phase of mechanical ventilation. The diagnosis of pneumothorax in critical illness is established from the patients’ history, physical examination and radiological investigation, although the appearances of a pneumothorax on a supine radiograph may be different from the classic appearance on an erect radiograph. For this reason, ultrasonography is beneficial for excluding the diagnosis of pneumothorax. Respiration-dependent movement of the visceral pleura and lung surface with respect to the parietal pleura and chest wall can be easily visualized with transthoracic sonography given that the presence of air in the pleural space prevents sonographic visualization of visceral pleura movements. Mechanically ventilated patients with a pneumothorax require tube thoracostomy placement because of the high risk of tension pneumothorax. Small-bore catheters are now preferred in the majority of ventilated patients. Furthermore, if there are clinical signs of a tension pneumothorax, emergency needle decompression followed by tube thoracostomy is widely advocated. Patients with pneumothorax related to mechanical ventilation who have tension pneumothorax, a higher acute physiology and chronic health evaluation II score or PaO2/FiO2 < 200 mmHg were found to have higher mortality.
Core tip: Patients with pneumothorax related to mechanical ventilation (PRMV) have a high mortality rate. PRMV often occurs in the early stage of mechanical ventilation and it may recur on the other side of lung in a short period of time. Low compliance is associated with a high incidence of PRMV, with PRMV being more related to the underlying process than the ventilatory setting. PRMV patients with tension pneumothorax, higher acute physiology and chronic health evaluation score or PaO2/FiO2 < 200 mmHg have a higher mortality.
- Citation: Hsu CW, Sun SF. Iatrogenic pneumothorax related to mechanical ventilation. World J Crit Care Med 2014; 3(1): 8-14
- URL: https://www.wjgnet.com/2220-3141/full/v3/i1/8.htm
- DOI: https://dx.doi.org/10.5492/wjccm.v3.i1.8
Pneumothorax, defined as the presence of air in the pleural space, is a serious complication of mechanical ventilation and is associated with increased morbidity and mortality[1,2]. Pneumothorax can be categorized as primary, secondary, iatrogenic or traumatic according to etiology. Mechanical ventilation was found to be the common cause of iatrogenic pneumothorax in an intensive care unit (ICU)[3]. Pneumothorax is rare in intubated patients with normal lungs and most patients with pneumothorax related to mechanical ventilation (PRMV) have underlying lung diseases that range from primary obstructive lung disease to secondary pneumonia and acute respiratory distress syndrome (ARDS)[4-6]. In critical illness, pneumothoraces may be difficult to diagnose when they have different clinical presentations and their locations are atypical and complicated by other disease processes in unconscious patients[7,8]. Prompt recognition and treatment of pneumothorax is important to minimize morbidity and mortality[9] because if pneumothorax is not diagnosed quickly, once tension pneumothorax ensues, it usually has a malignant course leading to death if untreated[10]. Sudden fall in oxygen saturation followed by hypotension are often observed clinically in these patients[11,12]. Tension pneumothorax is more common in ventilated patients, occurring in 30%-97% percent of all pneumothoraces[13-17].
Pneumothorax was found to be an independent predictor of mortality during mechanical ventilation[18] and was associated with a significant increase in the ICU length of stay, hospital stay and mortality in all mechanically ventilated patients[3]. The mortality rates are high, ranging from 46% to 77% if barotrauma is a complication of mechanical ventilation[2,3,14,15,19-24].
Patients who received mechanical ventilation have an approximate incidence of barotrauma of 4%-15%[4,13,19,25]. It has been reported that a 14%-87% incidence of pneumothorax occurs depending on severity and duration of ARDS and mode of ventilator for management[1,21,26]. A prior study found that the incidence rate of pneumothorax decreased after the implementation of protective lung strategies in pediatric patients with severe ARDS[27]. The incidence of barotrauma has been reported to be as low as 0.5% in postoperative patients[23].
Development of pneumothorax is most closely correlated with underlying lung disease[1,21]. Pneumonia is an important predisposing factor in the development of pulmonary barotrauma in mechanically ventilated patients[28]. Necrotizing bacterial pneumonia can cause air leaks into the pleura that result in development of pneumothorax[29].
Chronic obstructive pulmonary disease and asthma are also common underlying diseases associated with pneumothorax. High airway pressures are required to overcome severe bronchial obstruction. However, because of a variability of obstruction in the different airways, there is a mal-distribution of mechanical tidal volume, which promotes gas trapping and non-uniform alveolar distension. That is why these patients are at risk of pneumothorax[30].
PRMV is most closely correlated with ARDS[31], a heterogeneous disease in which the lung is physiologically small and with low compliance[32]. The dependent lung regions tend to be collapsed and the lung regions subject to high pressure overinflation and alveolar rupture are nondependent regions when positive end-expiratory pressure (PEEP) inflates and recruits some of collapsed regions during mechanical ventilation[33,34]. Subpleural and intrapulmonary air cysts occur in ARDS patients and the rupture of these air cysts may be a cause of pneumothorax[14]. Whether the pneumothorax in ARDS arises from overinflation of normal lung regions or from cyst rupture has not yet been conclusively established[21].
Other diseases such as lung cancer, tuberculosis, bronchiectasis, cystic fibrosis, idiopathic pulmonary fibrosis, sarcoidosis, histiocytosis X, Marfan’s syndrome, Ehlers-Danlos syndrome, lymphangioleiomyomatosis, rheumatoid arthritis and other connective tissue disease may predispose to the development of a pneumothorax[35].
An early investigation reported that peak airway pressure over 50 cm H2O is associated with increased risk of alveolar rupture during mechanical ventilation[4]. There have also been correlations made between high peak airway pressure and the development of pneumothorax[15,36]. High PEEP had been reported to be associated with pneumothorax[1] but several studies have found no such relationship[15,17,23,28,37]. Increased pressure is not enough by itself to produce alveolar rupture, with some studies demonstrating that pneumothorax is related to high tidal volume[37]. In animal studies, there is evidence to conclude that lung overdistension rather than high airway pressure is the primary cause of alveolar and interstitial injury[38,39].
A clinical study showed that when plateau pressure was maintained lower than 35 cm H2O, pneumothorax was unavoidable[40]. Although one study showed that the incidence of pneumothorax decreased after implementation of protective lung strategies[27], several recent studies comparing low tidal volume with conventional ventilation in ARDS failed to demonstrate any reduction in barotrauma when low tidal volumes were used[41-43]. Previous literature showed the incidence of barotrauma did not relate to the ventilatory settings[22,31,41,44] but a low compliance was associated with a high incidence of barotrauma, which suggested that barotrauma has been more related to the underlying process than the ventilatory setting[40].
Pneumothorax is secondary to ruptured alveoli and dissection of air along the vascular sheaths passing to the mediastinum, subcutaneous tissue and retroperitoneum. Therefore, it may manifest as pulmonary interstitial emphysema, pneumomediastinum, pneumoperitoneum or subcutaneous emphysema[32]. Patients may present with tachycardia, chest pain, tachypnea, agitation, hypotension, cyanosis or consciousness change. Tachycardia is the most common finding.
It has been observed that the median time from mechanical ventilation to the development of pneumothorax was 4 d[3]. Anzueto et al[45] reported that 80% of their patients developed pneumothorax within the first 3 d of mechanical ventilation. Gattinoni et al[21] found that late ARDS (> 2 wk of mechanical ventilation) patients had an increased incidence of pneumothoraces when compared to early ARDS (less than 1 wk of mechanical ventilation) patients[21]. However, Gammon et al[31] reported that pneumothoraces occurred most commonly during the early phase of ARDS, with a declining risk over time.
Approximately 7.4%-10% of patients with first episode pneumothorax will develop second episode pneumothorax on the other lung during mechanical ventilation[3,31]. Clinically, we should take notice of these patients because the pneumothorax might attack the lung on the other side.
Tension pneumothorax is common in mechanically ventilated patients[13-17]. Mechanical ventilation will increase gas flow through pleura defects, allowing more air to pass per unit time, and resulting in a more rapid intrapleural pressure rise with earlier mechanical compressive effects and rapid progress to cardiorespiratory collapse and death[46].
The diagnosis of pneumothorax in critical illness is established from patients’ history, physical examination and radiological investigation. The factors related to the underlying lung disease are important in the history. Examination of the respiratory and cardiovascular systems may help establish the diagnosis of pneumothorax. However, examination findings may vary according to the size of pneumothorax and presence of limited cardiorespiratory reserve[47], with patients with a small pneumothorax (one involving < 15% of the hemithorax) possibly having a normal physical examination. Careful inspection and repeated auscultation of the chest is therefore crucial. Contralateral tracheal deviation, hyperresonant percussion over the chest and decreased breathing sounds might be noted. Reduction in tidal volume during pressure controlled ventilation and increased airway pressure with volume controlled ventilation might be found from ventilators. A pulsus paradoxus on the arterial trace and increased central venous pressure from central venous catheterization may be observed[8,9,46,47].
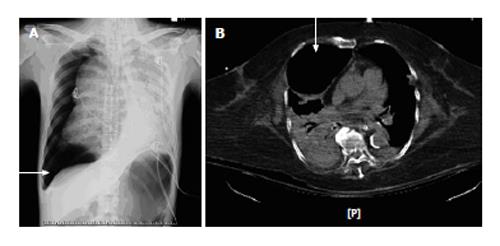
It should be noted that many of the above findings are nonspecific and have not been a reliable indicator of pneumothorax given that dyspnea severity can be out of proportion to the size of the pneumothorax. The radiographic data thus remains the gold standard for the diagnosis of pneumothorax[8]. The chest radiograph may show radiolucent hemithorax with apparent lung edge and absent lung marking. Mediastinal emphysema was the initial manifestation of extra-alveolar air[1]. Although an erect posteroanterior chest radiograph may help to estimate the pneumothorax size[48], it is not practical in critical illness. However, the supine anteroposterior chest radiographs are available in the ICU, although the appearances of a pneumothorax on a supine radiograph may be different from the classic appearances on an erect radiograph. Air may lie in the costophrenic angle extending more inferiorly than usual (Figure 1A), which has been called deep sulcus sign[49]. Concurrent lung disease may lead to different distributions of free air in the pleural space than in relatively normal lungs[50] or it may lead to loculated gas collections if there is associated pleural disease[51]. Some radiographic findings that may be confused with pneumothorax are skin folds, visceral gas within the gas and emphysema bullae. A chest computed tomography (CT) examination, in contrast, can differentiate these diagnoses and is the gold standard test for both the diagnosis and sizing of pneumothorax (Figure 1B)[52]. CT is an excellent tool to differentiate bullous lung disease and prevent unnecessary drainage attempts that may result in the creation of a parenchymal-pleural fistula[53]. Chest radiographs may not distinguish between emphysematous bullae and pneumothorax, thus possibly resulting in potentially catastrophic insertion of intercostal chest drain into emphysematous bullae in mechanically ventilated patients. Unfortunately, it is not always practical or safe to transport critically ill patients for a CT scan to exclude pneumothorax, particularly when the patient is hemodynamically unstable.
Transthoracic sonography is a diagnostic tool for pneumothorax[54-56]. There are several advantages of ultrasonography over standard chest radiography and CT scanning, including the lower radiation, portability, real-time imaging and the ability to easily perform dynamic and repeat evaluation. The main use of ultrasonography for assessment of pneumothorax lies in its capacity to rule out a pneumothorax. Respiration-dependent movement of the visceral pleura and lung surface with respect to the parietal pleura and chest wall can be easily visualized with real-time transthoracic sonography. This characteristic is known as lung sliding or gliding sign[56-58]. At the boundary between the pleura and the ventilated lung, intensive band-like reverberation echoes (comet-tail artifacts) are evoked during breathing movement[54,57]. M-mode images of patients without pneumothorax show breath-dependent movements as a single, thin comet-tail artifact. The presence of air in the pleural space prevents sonographic visualization of visceral pleura movements and the gliding sign and comet-tail artifacts disappear[57,58]. M-mode images of patients with pneumothorax show the immobility of these artifacts known as frozen echoes under breathing movement.
One study showed transthoracic ultrasound was more sensitive than chest radiography in the detection of pneumothorax[59]. Another study comparing ultrasonography to CT scan and chest radiographs for diagnosis of occult pneumothorax revealed that the use of ultrasonography detected 92% of occult pneumothoraces diagnosed with CT scan[60]. In trained hands, an ultrasound examination may obviate the need for empiric tube thoracostomy for suspected tension pneumothorax.
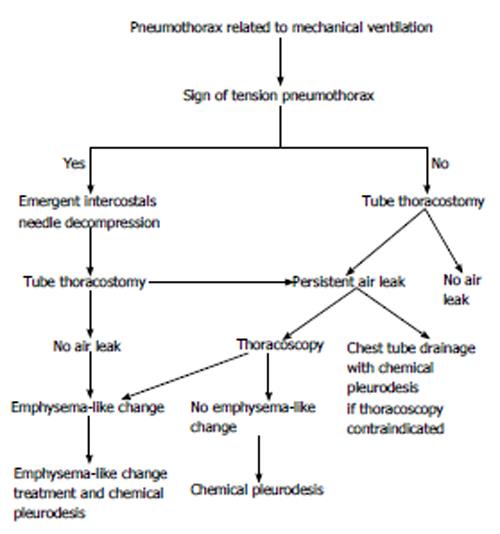
Most mechanically ventilated patients with a pneumothorax require tube thoracostomy placement because of the high risk of tension pneumothorax[61]. The strategy of managing pneumothorax is shown in Figure 2. It is not advisable to wait for a radiograph if there are clinical signs of a tension pneumothorax. Emergency needle decompression followed by tube thoracostomy is widely advocated[46]. In critically ill patients with minimal pulmonary reserve, even a small pneumothorax can have adverse cardiopulmonary effects[62]. Positive pressure ventilation can exacerbate air leaks and prevent pleural healing, potentially causing a rapid increase in the size and severity of existing pneumothorax.
The traditional treatment for pneumothorax in mechanically ventilated patients has been chest tube thoracostomy[61], an image-guided small catheter whose size ranges from 7 to 10 Fr, to become an effective therapeutic option for pneumothorax[63]. A retrospective review of 62 ventilated patients who underwent small-bore chest tube drainage as the primary management of pneumothorax found a 68.6% success rate, defined as no residual air seen in the follow-up chest radiograph, and with no major complications[64]. These results compare favorably with previous data showing a success rate of 55% with the same definition for success as in the previous study with a large bore tube[20].
In patients with a persistent air leak or failure of the lung to expand, early thoracic surgical consultation is generally requested within 3-5 d[65]. Although surgical intervention is considered very effective and safe with a low recurrence rate, the studies reaching this conclusion do not include critically ill patients[66,67].
Muscle relaxants might be effective to decrease the incidence of pneumothorax for patients with ARDS. A recent study by Papazian et al[68] reported a significant reduction in pneumothoraces in patients with severe ARDS who received 48 h of paralysis.
Mechanically ventilated patients with pneumothorax had a significantly higher mortality rate than those without pneumothorax. de Lassence et al[3] reported that iatrogenic pneumothorax was associated with a greater than twofold increase in the risk of death. In terms of iatrogenic pneumothorax, the mortality rate of patients with ventilator-related pneumothorax was significantly higher than that of patients with procedure-related pneumothorax[20]. Ventilator-related pneumothorax patients with tension pneumothorax had a higher risk of death[20].
Pneumothorax is a medical emergency and a disease with a high mortality rate; it requires a careful awareness, prompt recognition and intervention to reduce morbidity and mortality. Most patients with PRMV have underlying pulmonary diseases, the most common of which are pneumonia, ARDS and obstructive lung disease. Pneumothorax presentation and radiographic findings may be subtle or atypical. Whereas a CT scan is the gold standard for diagnosis, the use of ultrasonography has benefits in both diagnosis and management of pneumothorax. Most of the ventilated patients with a pneumothorax require immediate treatment with tube thoracostomy because of the high risk of progression to a tension pneumothorax. Small-bore catheters are now preferred in the majority of ventilated patients.
P- Reviewers: Kastelik JA, Kawai H S- Editor: Zhai HH L- Editor: Roemmele A E- Editor: Liu SQ
| 1. | Gammon RB, Shin MS, Buchalter SE. Pulmonary barotrauma in mechanical ventilation. Patterns and risk factors. Chest. 1992;102:568-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 131] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 2. | Schnapp LM, Chin DP, Szaflarski N, Matthay MA. Frequency and importance of barotrauma in 100 patients with acute lung injury. Crit Care Med. 1995;23:272-278. [PubMed] |
| 3. | de Lassence A, Timsit JF, Tafflet M, Azoulay E, Jamali S, Vincent F, Cohen Y, Garrouste-Orgeas M, Alberti C, Dreyfuss D. Pneumothorax in the intensive care unit: incidence, risk factors, and outcome. Anesthesiology. 2006;104:5-13. [PubMed] |
| 4. | Petersen GW, Baier H. Incidence of pulmonary barotrauma in a medical ICU. Crit Care Med. 1983;11:67-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Pierson DJ. Complications associated with mechanical ventilation. Crit Care Clin. 1990;6:711-724. [PubMed] |
| 6. | Parker JC, Hernandez LA, Peevy KJ. Mechanisms of ventilator-induced lung injury. Crit Care Med. 1993;21:131-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 371] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 7. | Tocino IM, Miller MH, Fairfax WR. Distribution of pneumothorax in the supine and semirecumbent critically ill adult. AJR Am J Roentgenol. 1985;144:901-905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 151] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 8. | Rankine JJ, Thomas AN, Fluechter D. Diagnosis of pneumothorax in critically ill adults. Postgrad Med J. 2000;76:399-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Yarmus L, Feller-Kopman D. Pneumothorax in the critically ill patient. Chest. 2012;141:1098-1105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 10. | Rojas R, Wasserberger J, Balasubramaniam S. Unsuspected tension pneumothorax as a hidden cause of unsuccessful resuscitation. Ann Emerg Med. 1983;12:411-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 11. | Coats TJ, Wilson AW, Xeropotamous N. Pre-hospital management of patients with severe thoracic injury. Injury. 1995;26:581-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 12. | Deakin CD, Davies G, Wilson A. Simple thoracostomy avoids chest drain insertion in prehospital trauma. J Trauma. 1995;39:373-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 68] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 13. | Zwillich CW, Pierson DJ, Creagh CE, Sutton FD, Schatz E, Petty TL. Complications of assisted ventilation. A prospective study of 354 consecutive episodes. Am J Med. 1974;57:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 247] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 14. | Rohlfing BM, Webb WR, Schlobohm RM. Ventilator-related extra-alveolar air in adults. Radiology. 1976;121:25-31. [PubMed] |
| 15. | Woodring JH. Pulmonary interstitial emphysema in the adult respiratory distress syndrome. Crit Care Med. 1985;13:786-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 85] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 16. | Johnson TH, Altman AR. Pulmonary interstitial gas: first sign of barotrauma due to PEEP therapy. Crit Care Med. 1979;7:532-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Zimmerman JE, Dunbar BS, Klingenmaier CH. Management of subcutaneous emphysema, pneumomediastinum, and pneumothorax during respirator therapy. Crit Care Med. 1975;3:69-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Esteban A, Anzueto A, Frutos F, Alía I, Brochard L, Stewart TE, Benito S, Epstein SK, Apezteguía C, Nightingale P. Characteristics and outcomes in adult patients receiving mechanical ventilation: a 28-day international study. JAMA. 2002;287:345-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1117] [Cited by in RCA: 1068] [Article Influence: 46.4] [Reference Citation Analysis (0)] |
| 19. | de Latorre FJ, Tomasa A, Klamburg J, Leon C, Soler M, Rius J. Incidence of pneumothorax and pneumomediastinum in patients with aspiration pneumonia requiring ventilatory support. Chest. 1977;72:141-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 20. | Chen KY, Jerng JS, Liao WY, Ding LW, Kuo LC, Wang JY, Yang PC. Pneumothorax in the ICU: patient outcomes and prognostic factors. Chest. 2002;122:678-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Gattinoni L, Bombino M, Pelosi P, Lissoni A, Pesenti A, Fumagalli R, Tagliabue M. Lung structure and function in different stages of severe adult respiratory distress syndrome. JAMA. 1994;271:1772-1779. [PubMed] [DOI] [Full Text] |
| 22. | Weg JG, Anzueto A, Balk RA, Wiedemann HP, Pattishall EN, Schork MA, Wagner LA. The relation of pneumothorax and other air leaks to mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338:341-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 105] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 23. | Cullen DJ, Caldera DL. The incidence of ventilator-induced pulmonary barotrauma in critically ill patients. Anesthesiology. 1979;50:185-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 67] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 24. | Fleming WH, Bowen JC. Early complications of long-term respiratory support. J Thorac Cardiovasc Surg. 1972;64:729-738. [PubMed] |
| 25. | Strange C. Pleural complications in the intensive care unit. Clin Chest Med. 1999;20:317-327. [PubMed] |
| 26. | Jantz MA, Pierson DJ. Pneumothorax and barotrauma. Clin Chest Med. 1994;15:75-91. [PubMed] |
| 27. | Miller MP, Sagy M. Pressure characteristics of mechanical ventilation and incidence of pneumothorax before and after the implementation of protective lung strategies in the management of pediatric patients with severe ARDS. Chest. 2008;134:969-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 28. | Kumar A, Pontoppidan H, Falke KJ, Wilson RS, Laver MB. Pulmonary barotrauma during mechanical ventilation. Crit Care Med. 1973;1:181-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 119] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 29. | Fukagawa T. Centromere DNA, proteins and kinetochore assembly in vertebrate cells. Chromosome Res. 2004;12:557-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 30. | Marcy TW. Barotrauma: detection, recognition, and management. Chest. 1993;104:578-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 31. | Gammon RB, Shin MS, Groves RH, Hardin JM, Hsu C, Buchalter SE. Clinical risk factors for pulmonary barotrauma: a multivariate analysis. Am J Respir Crit Care Med. 1995;152:1235-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 32. | Gattinoni L, Pesenti A. The concept of “baby lung”. Intensive Care Med. 2005;31:776-784. [PubMed] |
| 33. | Gattinoni L, Mascheroni D, Torresin A, Marcolin R, Fumagalli R, Vesconi S, Rossi GP, Rossi F, Baglioni S, Bassi F. Morphological response to positive end expiratory pressure in acute respiratory failure. Computerized tomography study. Intensive Care Med. 1986;12:137-142. [PubMed] |
| 34. | Gattinoni L, Pesenti A, Bombino M, Baglioni S, Rivolta M, Rossi F, Rossi G, Fumagalli R, Marcolin R, Mascheroni D. Relationships between lung computed tomographic density, gas exchange, and PEEP in acute respiratory failure. Anesthesiology. 1988;69:824-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 363] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 35. | Noppen M, De Keukeleire T. Pneumothorax. Respiration. 2008;76:121-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 177] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 36. | Haake R, Schlichtig R, Ulstad DR, Henschen RR. Barotrauma. Pathophysiology, risk factors, and prevention. Chest. 1987;91:608-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 120] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 37. | Steier M, Ching N, Roberts EB, Nealon TF. Pneumothorax complicating continuous ventilatory support. J Thorac Cardiovasc Surg. 1974;67:17-23. [PubMed] |
| 38. | Dreyfuss D, Saumon G. Ventilator-induced lung injury: lessons from experimental studies. Am J Respir Crit Care Med. 1998;157:294-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1587] [Cited by in RCA: 1434] [Article Influence: 53.1] [Reference Citation Analysis (0)] |
| 39. | Dreyfuss D, Saumon G. Barotrauma is volutrauma, but which volume is the one responsible. Intensive Care Med. 1992;18:139-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 127] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 40. | Boussarsar M, Thierry G, Jaber S, Roudot-Thoraval F, Lemaire F, Brochard L. Relationship between ventilatory settings and barotrauma in the acute respiratory distress syndrome. Intensive Care Med. 2002;28:406-413. [PubMed] |
| 41. | Brochard L, Roudot-Thoraval F, Roupie E, Delclaux C, Chastre J, Fernandez-Mondéjar E, Clémenti E, Mancebo J, Factor P, Matamis D. Tidal volume reduction for prevention of ventilator-induced lung injury in acute respiratory distress syndrome. The Multicenter Trail Group on Tidal Volume reduction in ARDS. Am J Respir Crit Care Med. 1998;158:1831-1838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 544] [Cited by in RCA: 473] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 42. | Stewart TE, Meade MO, Cook DJ, Granton JT, Hodder RV, Lapinsky SE, Mazer CD, McLean RF, Rogovein TS, Schouten BD. Evaluation of a ventilation strategy to prevent barotrauma in patients at high risk for acute respiratory distress syndrome. Pressure- and Volume-Limited Ventilation Strategy Group. N Engl J Med. 1998;338:355-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 626] [Cited by in RCA: 545] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 43. | Brower RG, Shanholtz CB, Fessler HE, Shade DM, White P, Wiener CM, Teeter JG, Dodd-o JM, Almog Y, Piantadosi S. Prospective, randomized, controlled clinical trial comparing traditional versus reduced tidal volume ventilation in acute respiratory distress syndrome patients. Crit Care Med. 1999;27:1492-1498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 44. | Morris AH, Wallace CJ, Menlove RL, Clemmer TP, Orme JF, Weaver LK, Dean NC, Thomas F, East TD, Pace NL. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med. 1994;149:295-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 750] [Cited by in RCA: 577] [Article Influence: 18.6] [Reference Citation Analysis (0)] |
| 45. | Anzueto A, Frutos-Vivar F, Esteban A, Alía I, Brochard L, Stewart T, Benito S, Tobin MJ, Elizalde J, Palizas F. Incidence, risk factors and outcome of barotrauma in mechanically ventilated patients. Intensive Care Med. 2004;30:612-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 155] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 46. | Leigh-Smith S, Harris T. Tension pneumothorax--time for a re-think. Emerg Med J. 2005;22:8-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 178] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 47. | Currie GP, Alluri R, Christie GL, Legge JS. Pneumothorax: an update. Postgrad Med J. 2007;83:461-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 48. | Noppen M, Alexander P, Driesen P, Slabbynck H, Verstraete A. Quantification of the size of primary spontaneous pneumothorax: accuracy of the Light index. Respiration. 2001;68:396-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 38] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 49. | Chiles C, Ravin CE. Radiographic recognition of pneumothorax in the intensive care unit. Crit Care Med. 1986;14:677-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 47] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 50. | Gobien RP, Reines HD, Schabel SI. Localized tension pneumothorax: unrecognized form of barotrauma in adult respiratory distress syndrome. Radiology. 1982;142:15-19. [PubMed] |
| 52. | Kelly AM, Weldon D, Tsang AY, Graham CA. Comparison between two methods for estimating pneumothorax size from chest X-rays. Respir Med. 2006;100:1356-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 53. | Phillips GD, Trotman-Dickenson B, Hodson ME, Geddes DM. Role of CT in the management of pneumothorax in patients with complex cystic lung disease. Chest. 1997;112:275-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 54. | Wernecke K, Galanski M, Peters PE, Hansen J. Pneumothorax: evaluation by ultrasound--preliminary results. J Thorac Imaging. 1987;2:76-78. [PubMed] |
| 55. | Targhetta R, Bourgeois JM, Chavagneux R, Marty-Double C, Balmes P. Ultrasonographic approach to diagnosing hydropneumothorax. Chest. 1992;101:931-934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 54] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 56. | Targhetta R, Bourgeois JM, Chavagneux R, Coste E, Amy D, Balmes P, Pourcelot L. Ultrasonic signs of pneumothorax: preliminary work. J Clin Ultrasound. 1993;21:245-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 33] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 57. | Wernecke K. Ultrasound study of the pleura. Eur Radiol. 2000;10:1515-1523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 30] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 58. | Reissig A, Kroegel C. Accuracy of transthoracic sonography in excluding post-interventional pneumothorax and hydropneumothorax. Comparison to chest radiography. Eur J Radiol. 2005;53:463-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 59. | Jalli R, Sefidbakht S, Jafari SH. Value of ultrasound in diagnosis of pneumothorax: a prospective study. Emerg Radiol. 2013;20:131-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 60. | Soldati G, Testa A, Sher S, Pignataro G, La Sala M, Silveri NG. Occult traumatic pneumothorax: diagnostic accuracy of lung ultrasonography in the emergency department. Chest. 2008;133:204-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 244] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 61. | Gilbert TB, McGrath BJ, Soberman M. Chest tubes: indications, placement, management, and complications. J Intensive Care Med. 1993;8:73-86. [PubMed] |
| 62. | Marcy TW, Marini JJ. Respiratory distress in the ventilated patient. Clin Chest Med. 1994;15:55-73. [PubMed] |
| 63. | Chon KS, vanSonnenberg E, D’Agostino HB, O’Laoide RM, Colt HG, Hart E. CT-guided catheter drainage of loculated thoracic air collections in mechanically ventilated patients with acute respiratory distress syndrome. AJR Am J Roentgenol. 1999;173:1345-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 64. | Lin YC, Tu CY, Liang SJ, Chen HJ, Chen W, Hsia TC, Shih CM, Hsu WH. Pigtail catheter for the management of pneumothorax in mechanically ventilated patients. Am J Emerg Med. 2010;28:466-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 65. | MacDuff A, Arnold A, Harvey J; BTS Pleural Disease Guideline Group. Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010. Thorax. 2010;65 Suppl 2:ii18-ii31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 624] [Cited by in RCA: 815] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 66. | Hatz RA, Kaps MF, Meimarakis G, Loehe F, Müller C, Fürst H. Long-term results after video-assisted thoracoscopic surgery for first-time and recurrent spontaneous pneumothorax. Ann Thorac Surg. 2000;70:253-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 125] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 67. | Thomas P, Le Mee F, Le Hors H, Sielezneff I, Auge A, Giudicelli R, Fuentes P. [Results of surgical treatment of persistent or recurrent pneumothorax]. Ann Chir. 1993;47:136-140. [PubMed] |
| 68. | Papazian L, Forel JM, Gacouin A, Penot-Ragon C, Perrin G, Loundou A, Jaber S, Arnal JM, Perez D, Seghboyan JM. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363:1107-1116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1562] [Cited by in RCA: 1551] [Article Influence: 103.4] [Reference Citation Analysis (0)] |