Published online Mar 28, 2017. doi: 10.5412/wjsp.v7.i1.1
Peer-review started: February 7, 2017
First decision: March 7, 2017
Revised: March 15, 2017
Accepted: March 20, 2017
Article in press: March 21, 2017
Published online: March 28, 2017
Processing time: 72 Days and 12.8 Hours
The best approach to achieve cure in esophageal cancer is a combination of chemo-radiation and surgery. However, complications occur in half of patients. The current report, reports a rare but severe complication: Complete obstruction of the esophagus, induced by preoperative chemo-radiation therapy. Normally, strictures are treated by repeated dilatations, however, in case of complete obstruction, the perforation rate of standard blind anterograde wire access and dilation is severely increased. In order to minimize the risk of perforations, the rendezvous technique was introduced. This technique involves an anterograde approach in combination with a retrograde approach in order to open and dilatate the esophagus. While technical success rates between 83% and 100% have been reported in literature, data on clinical outcomes are scarcer. The limited amount of studies available claim that success was achieved in almost half of patients. The patient in our case currently has an oral diet without restrictions and rates his quality of life with a VAS-score ten out of ten.
Core tip: A rare but severe complication of chemo-radiation of the esophagus, in case of esophageal cancer, is a complete esophageal obstruction. Blind anterograde wire access and dilatation leads to a higher risk of perforation, due to lack of visualization and a disrupted anatomy. For this reason the rendezvous technique was introduced, which involves an anterograde approach in combination with a retrograde approach in order to open and dilatate the esophagus.
- Citation: Klinkert M, de Jong MC, Sosef MN, van Nunen AB, Belgers HJ. Surgical treatment of a rare complication after endoscopic stent placement for anastomotic leakage after esophageal resection. World J Surg Proced 2017; 7(1): 1-5
- URL: https://www.wjgnet.com/2219-2832/full/v7/i1/1.htm
- DOI: https://dx.doi.org/10.5412/wjsp.v7.i1.1
Esophageal carcinoma is the eighth most common cancer worldwide[1]. Moreover, it is the sixth most common cause of cancer-related death[1,2], with a 5-year-overall survival of only 18.4% for all patients following diagnosis[3]. In case curative intent surgery is performed, 5-year-overall survival increases to 34%; moreover when surgery is performed subsequently to a neoadjuvant chemo-radiation regimen, overall survival rates increase to 47%[4].
The best approach to achieve cure is a combination of chemo-radiation and surgery[5,6], as the CROSS-trial demonstrated that neoadjuvant chemo-radiotherapy (nCRT) improves survival among patients with potentially curable esophageal cancer[4]. Moreover, this increase in survival is achieved without a concomitant increase in postoperative morbidity or mortality[7].
Nevertheless, a high complication rate after esophagectomy is reported, as complications occur after more than half of procedures - open or minimally invasive[8]. While it is generally thought that the complication risk is lower in laparoscopy, due to diminished surgical trauma[5,9], the incidence of postoperative morbidity remains high with incidences ranging from 17% up to 74%[8,10,11]. Most common, severe complications include anastomotic leakage, conduit necrosis, chyle leakage, vocal cord injury or palsy and death[8]. Importantly, these complications are known to directly impact other outcomes and cause prolonged hospital stay and readmission, early cancer recurrence and diminished overall survival and also a lower quality of life[8,12-14]. More rare complications have also been described such as herniation of abdominal structures through the diaphragm and - even more rare - complete obstruction of the esophagus partly due to the preoperative chemo-radiation therapy. Postoperative mortality rates are reported in 7% to 9% of cases[8,9].
A 64-year-old man with a mid-esophageal squamous cell carcinoma (eT3N3M0) underwent a minimal invasive transthoracic esophagectomy, following neoadjuvant treatment with chemo-radiation - CROSS scheme.
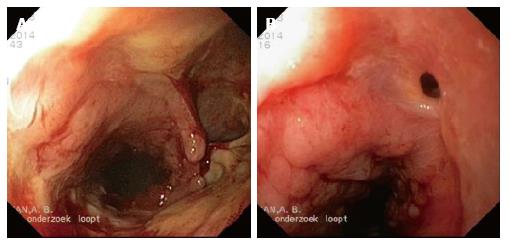
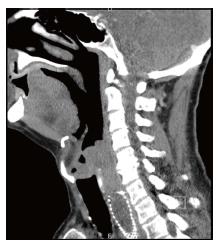
On postoperative day three (POD3), an elevated white blood count was noticed and therefore a chest- and abdominal computed axial tomography (CAT)-scan was performed, which showed suspicion of a pneumonia for which antibiotic treatment was started. However, 5 d later (POD8) a repeated CAT-scan was performed as the patient showed signs of clinical deterioration. This CAT-scan demonstrated free air and fluid around the anastomosis suspicious for anastomotic leakage. Upper endoscopy showed superficial circumferential ischemia of the top of the stomach-tube over a length of 7 cm, while no visible anastomotic leakage was seen. The ischemic area was fully shielded by a partially covered-stent (SEMS) (Ultraflex®). A planned re-endoscopy after 14 d, showed transmural ischemic ulceration over a length of at least 7 cm (Figure 1A). Initially therefore, the decision was taken to disconnect the anastomosis and to thereby create an esophagostomy, however during the re-laparoscopy this was found to be impossible due to the amount of adhesions. Therefore, a new partially covered SEMS (Ultraflex®) was endoscopically placed. The subsequent SEMS replacement endoscopy after 4 wk showed extensive tissue ingrowth into the stent, which was subsequently removed and replaced by a new partially covered stent of the same type. It was noted that the defect due to ischemia was much smaller than previously (Figure 1B). The removal of this SEMS, one month later, was complicated by a prolapse of esophageal mucosal tissue obstructing the upper esophageal entrance completely. We managed to insert a guidewire and place a fully covered SEMS (Hanaro®). Repeat endoscopy the following day, unfortunately showed it was impossible to pass the UES with either the gastroscope or fiber laryngoscope. A subsequently performed CAT-scan confirmed this finding, as it showed a complete obstruction of the upper esophagus over a distance of approximately 4-5 cm (Figure 2). A procedure under general anesthesia was planned. In order to pass this obstruction, the gastroenterologist struggled to identify the anatomical oral route, while the esophageal orifice was not found. Simultaneously, the surgeon performed a laparoscopy and entered the gastric conduit in a retrograde fashion. By moving proximally through the stent, the obstructed proximal esophagus was reached. Step by step, the surgeon, using a biopsy forceps, dissected upwards. At the level of the oral orifice, the oral mucosa could be perforated using a needle knife under light transillumination of the gastroscope. After this, the guide wire could pass through the obstruction (a so-called rendez-vous procedure). Following correct placement of the guidewire, a dilatation was performed and a new fully covered SEMS (Hanaro®) was placed to keep the orifice open. After the covered SEMS (Hanaro®) was removed, no defects in the mucosa underneath it were seen.
Eleven dilatation sessions later, no defects in the mucosa were seen and the stent was removed. After this, our patients’ ability of oral ingestion was fully restored. The patient is currently no longer dependent on any tube-feeding and rates his quality of life with a VAS-score 10 out of 10.
Strictures of the proximal esophagus have been reported in 3% to 4% of patients with head and neck cancer who underwent treatment with radiation therapy[15]. These strictures are usually treated by repeated endoscopic dilatations, however complications of perforation or fistulae are described[15]. Complete obstruction in the proximal esophagus is a rare condition and can occur as a result of radiotherapy, gastro-esophageal reflux disease or caustic injury[16]. In our case, the hypothesis is that the traumatic stent removal, in combination with the neoadjuvant (chemo)radiation therapy, caused the (complete) obstruction of the esophagus.
In case of complete obstruction, the perforation rate of the standard blind anterograde wire access and subsequent dilation is increased, due the suboptimal visualization and distorted anatomy[15]. In order to minimize the incidence of this particular complication, the rendezvous technique was proposed, which was first described in 1998 by van Twisk et al[17]. This technique involves an anterograde approach, in combination with a retrograde approach, in order to open and dilatate the esophagus. This technique is therefore also called combined anterograde-retrograde endoscopic dilatation, abbreviated to CARD[16].
The aim of this procedure is to gain access through the complete obstructed esophagus. Technical success rates between 83% and 100% have been reported in literature[15,16,18-20], exemplifying its technical feasibility. The advantage of this procedure lays in the fact that it is safer than the blind anterograde approach, thereby theoretically reducing the risk of a perforation[15,16]. In addition, in case a mucosal puncture does occur, during the retrograde maneuver this perforation occurs away from the mediastinum, thereby reducing the risk for potential complications of this puncture[15].
However perforations after the rendez-vous procedure have been described, as Maple et al[15] reported two micro-perforations. These perforations, which were probably caused by the guide wire, resolved without (surgical) therapy. Conceivably, these micro-perforations occur more frequently than recognized, as they probably lack clinical relevance in the majority of cases[15].
Apart from the guidewire as the source of perforation, the type of scope can also be of influence of risk of perforation. While Bertolini et al[16] only used a thin, nasal gastroscope and reported one single case of mediastinal emphysema, Goguen et al[18] used an adult gastroscope and reported a pneumomediastinum in 18% of patients, an esophageal perforation in 5% of patients and a gastrostomy tube site problem in 16% of patients.
While the technical feasibility of the rendez-vous approach has been described by different authors, the actual purpose of the rendez-vous approach, i.e., to restore the ability of oral ingestion, is largely overlooked in series published thus far. Very scarce data have been published on the clinical side of this topic. This is a difficult parameter to report on, while there are very little disease-specific measures for this patient-group, as only swallowing, nutritional status and quality of life are acknowledged outcomes. In an attempt to specify nutritional intake following the rendez-vous procedure, Bertolini et al[16] decided to use the Functional Oral Intake Scale (FOIS), a scale specifically designed to document changes in functional oral intake in stroke patients. In their case-series, they reported that half of patients remained PEG-tube independent, while overall there was a relative improvement in swallowing (FOIS level ≥ 3) and consequently an improved quality of life[16]. The largest case-series published thus far is by Grooteman et al[21], they reported that 44% of their 25 patients were able to eat at least soft food. The other 56% of patients remained PEG-tube dependent (FOIS level < 3).
In summary, esophageal stricture is a serious long-term complication of (chemo)radiation therapy of the head and neck area. While currently fairly uncommon, more patients may suffer of strictures in coming years. With the current CROSS-scheme for esophageal carcinoma, which involves chemo-radiation therapy, the incidence of this complication might rise. Moreover, because this particular neoadjuvant regimen has prolonged the overall survival for this group of patients, it does not only enlarge the possible pool of patients at danger, but also prolongs the period during which the risk of developing an esophageal stricture exists. Although, anterograde dilatation is the most common approach, it is accompanied by a relative high risk of complications and cannot always be performed - like in our current case. Therefore, the rendez-vous technique is a good alternative for this group of patients. Data have shown that the rendez-vous procedure is a technically feasible and effective approach for patients with a complete obstruction of their esophagus. Data on clinical outcomes are scarcer; although the limited studies available claim that success was achieved in almost half of patients. Our patient currently has an oral diet without restrictions (FOIS level 7) and rates his quality of life with a VAS-score ten out of ten.
A 64-year-old man with a mid-esophageal carcinoma underwent a minimal invasive transthoracic esophagectomy (following neoadjuvant chemo-radiation) which was complicated by an anastomotic leakage and he developed a total obstruction of his esophagus due traumatic stent replacements.
On postoperative day three, an elevated white blood count was noticed, which led to performance of a subsequent computed axial tomography (CAT)-scan. Antibiotics were started by a suspicion on a pneumonia. Five days later, due clinical deterioration, a new CAT-scan was performed, which showed a suspicion for an anastomotic leakage.
Pneumonia, anastomotic leakage, total obstruction of the esophagus.
Elevated white blood count.
A chest- and abdominal-CAT scan showed signs of an anastomotic leakage, which was confirmed by an upper endoscopy - a stent was placed and after a few (traumatic) stent replacements, a re-placement endoscopy failed to pass the UES, a CAT-scan showed a total obstruction of the esophagus.
A rendez-vous technique was used to dilatate the complete obstructed esophagus.
Complete obstruction in the proximal esophagus is a rare condition and can occur as a result of radiotherapy, gastro-esophageal reflux disease or caustic injury. Strictures are usually treated by repeated endoscopic dilatation, however in case of complete obstruction the perforation rate of standard blind anterograde wire access and subsequent dilation is increased, therefore the rendezvous technique is developed.
Rendez-vous technique is an anterograde- and retrograde approach, in order to open and dilatate the esophagus.
In this case report, the authors showed that chemo-radiation (with or without the combination of a traumatic stent replacements) caused a complete obstruction of the esophagus. The incidence of this complication might rise due the neoadjuvant chemo-radiation treatment and the fact that this regime prolonged the overall survival and the period during the risk of developing structures, of patients suffering of esophageal cancer.
This is a very interesting case report.
Manuscript source: Unsolicited manuscript
Specialty type: Surgery
Country of origin: The Netherlands
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Agresta F, Barreto S, Chello M, Mocellin S S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Herszényi L, Tulassay Z. Epidemiology of gastrointestinal and liver tumors. Eur Rev Med Pharmacol Sci. 2010;14:249-258. [PubMed] |
| 2. | Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23762] [Cited by in RCA: 25543] [Article Influence: 1824.5] [Reference Citation Analysis (7)] |
| 3. | van Hagen P, Hulshof MC, van Lanschot JJ, Steyerberg EW, van Berge Henegouwen MI, Wijnhoven BP, Richel DJ, Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ. Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med. 2012;366:2074-2084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3288] [Cited by in RCA: 4080] [Article Influence: 313.8] [Reference Citation Analysis (0)] |
| 4. | Wan J, Che Y, Kang N, Zhang R. Surgical Method, Postoperative Complications, and Gastrointestinal Motility of Thoraco-Laparoscopy 3-Field Esophagectomy in Treatment of Esophageal Cancer. Med Sci Monit. 2016;22:2056-2065. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Shapiro J, van Lanschot JJ, Hulshof MC, van Hagen P, van Berge Henegouwen MI, Wijnhoven BP, van Laarhoven HW, Nieuwenhuijzen GA, Hospers GA, Bonenkamp JJ. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): long-term results of a randomised controlled trial. Lancet Oncol. 2015;16:1090-1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1292] [Cited by in RCA: 1826] [Article Influence: 182.6] [Reference Citation Analysis (0)] |
| 6. | Nederlof N, Slaman AE, van Hagen P, van der Gaast A, Slankamenac K, Gisbertz SS, van Lanschot JJ, Wijnhoven BP, van Berge Henegouwen MI. Using the Comprehensive Complication Index to Assess the Impact of Neoadjuvant Chemoradiotherapy on Complication Severity After Esophagectomy for Cancer. Ann Surg Oncol. 2016;23:3964-3971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 45] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Low DE, Alderson D, Cecconello I, Chang AC, Darling GE, DJourno XB, Griffin SM, Hölscher AH, Hofstetter WL, Jobe BA. International Consensus on Standardization of Data Collection for Complications Associated With Esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann Surg. 2015;262:286-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 517] [Cited by in RCA: 846] [Article Influence: 84.6] [Reference Citation Analysis (1)] |
| 8. | Finks JF, Osborne NH, Birkmeyer JD. Trends in hospital volume and operative mortality for high-risk surgery. N Engl J Med. 2011;364:2128-2137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1119] [Cited by in RCA: 1066] [Article Influence: 76.1] [Reference Citation Analysis (0)] |
| 9. | Dunst CM, Swanström LL. Minimally invasive esophagectomy. J Gastrointest Surg. 2010;14 Suppl 1:S108-S114. [PubMed] |
| 10. | Courrech Staal EF, Aleman BM, Boot H, van Velthuysen ML, van Tinteren H, van Sandick JW. Systematic review of the benefits and risks of neoadjuvant chemoradiation for oesophageal cancer. Br J Surg. 2010;97:1482-1496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 11. | Derogar M, Orsini N, Sadr-Azodi O, Lagergren P. Influence of major postoperative complications on health-related quality of life among long-term survivors of esophageal cancer surgery. J Clin Oncol. 2012;30:1615-1619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 164] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 12. | Scarpa M, Saadeh LM, Fasolo A, Alfieri R, Cagol M, Cavallin F, Pinto E, Zaninotto G, Ancona E, Castoro C. Health-related quality of life in patients with oesophageal cancer: analysis at different steps of the treatment pathway. J Gastrointest Surg. 2013;17:421-433. [PubMed] |
| 13. | Viklund P, Lindblad M, Lagergren J. Influence of surgery-related factors on quality of life after esophageal or cardia cancer resection. World J Surg. 2005;29:841-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 66] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Maple JT, Petersen BT, Baron TH, Kasperbauer JL, Wong Kee Song LM, Larson MV. Endoscopic management of radiation-induced complete upper esophageal obstruction with an antegrade-retrograde rendezvous technique. Gastrointest Endosc. 2006;64:822-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 48] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 15. | Bertolini R, Meyenberger C, Putora PM, Albrecht F, Broglie MA, Stoeckli SJ, Sulz MC. Endoscopic dilation of complete oesophageal obstructions with a combined antegrade-retrograde rendezvous technique. World J Gastroenterol. 2016;22:2366-2372. [PubMed] |
| 16. | van Twisk JJ, Brummer RJ, Manni JJ. Retrograde approach to pharyngo-esophageal obstruction. Gastrointest Endosc. 1998;48:296-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 49] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 17. | Goguen LA, Norris CM, Jaklitsch MT, Sullivan CA, Posner MR, Haddad RI, Tishler RB, Burke E, Annino DJ. Combined antegrade and retrograde esophageal dilation for head and neck cancer-related complete esophageal stenosis. Laryngoscope. 2010;120:261-266. [PubMed] |
| 18. | Fowlkes J, Zald PB, Andersen P. Management of complete esophageal stricture after treatment of head and neck cancer using combined anterograde retrograde esophageal dilation. Head Neck. 2012;34:821-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Dellon ES, Cullen NR, Madanick RD, Buckmire RA, Grimm IS, Weissler MC, Couch ME, Shaheen NJ. Outcomes of a combined antegrade and retrograde approach for dilatation of radiation-induced esophageal strictures (with video). Gastrointest Endosc. 2010;71:1122-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 20. | Grooteman KV, Wong Kee Song LM, Vleggaar FP, Siersema PD, Baron TH. Functional outcome of patients treated for radiation-induced complete esophageal obstruction after successful endoscopic recanalization (with video). Gastrointest Endosc. 2014;80:175-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |