Published online Jun 9, 2024. doi: 10.5409/wjcp.v13.i2.91478
Revised: April 22, 2024
Accepted: May 6, 2024
Published online: June 9, 2024
Processing time: 161 Days and 6.3 Hours
High-saturated fat (HF) or high-fructose (HFr) consumption in children pre
Core Tip: High-fructose (HFr) or high-fat (HF) intake favors metabolic syndrome (MetS) development by different mechanisms. When combined, HFr exacerbates the effects of HF, leading to a faster and more severe MetS development. Combined HF + HFr is usually present in ultra-processed foods. However, there is a lack of studies in the pediatric population evaluating the impact of restricting the combined intake of carbohydrates and fat in MetS. We reviewed the mechanisms by which HF + HFr produces more severe MetS to support the need for studies targeting the combined intake of HF + HFr in pediatric population to improve the outcomes of different interventions against MetS.
- Citation: Vargas-Vargas MA, González-Montoya M, Torres-Isidro O, García-Berumen CI, Ortiz-Avila O, Calderón-Cortés E, Cortés-Rojo C. Assessing the impact of concurrent high-fructose and high-saturated fat diets on pediatric metabolic syndrome: A review. World J Clin Pediatr 2024; 13(2): 91478
- URL: https://www.wjgnet.com/2219-2808/full/v13/i2/91478.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v13.i2.91478
Industrialization of occidental society has led to profound changes in several aspects of human life, including the introduction of foods that are accessible to the majority of the population, have a long shelf life, and are highly palatable. This has opened the world market for ultra-processed (UPC) foods, a term that refers to industrial modification of food such that the final product is different from the original and generally contains little or no whole food[1]. High amounts of saturated fat or carbohydrates are added to these foods to achieve high palatability and making them energy-dense, the chronic consumption of which is believed to be the main driver of the current obesity pandemic.
In contrast to the current panorama, in both the Middle Ages and Renaissance, obesity was considered a status symbol of affluence and health, triggering the onset of metabolic disorders in adults. Childhood obesity gained attention approximately 40 years ago when the stable prevalence curve of obesity in the pediatric population was found to have become parabolic[2]. Over the last three decades, the global prevalence of obesity has increased by 27.5% in adults and 47.1% in children[3], highlighting the urgent need to understand the main causes of this phenomenon.
Undoubtedly, the increasing accessibility of high-calorie UPC foods in developed and developing countries has played an important role in the early onset of obesity[4,5], which is a key risk factor for the development of metabolic syndrome (MetS)[6]. In adults, MetS is defined as a set of diagnostic symptoms including central obesity with a waist circumference ≥ 102 cm in men or ≥ 88 cm in women, blood pressure (BP) ≥ 130/85 mmHg, triglycerides (TG) ≥ 150 mg/dL, high-density lipoprotein cholesterol (HDL-C) < 40 mg/dL (men) or < 50 mg/dL (women) and fasting glucose ≥ 100 mg/dL[7,8]. In contrast with the well-established criteria for defining MetS in adults, the diagnosis of MetS in children lacks universally accepted criteria[9].
In addition to the excessive consumption of UPC foods, other factors that may contribute to early onset MetS are maternal obesity and the consequent development of gestational diabetes and type 2 diabetes mellitus (T2DM). This metabolic environment enveloping the fetus is related to an alteration in the degree of fetal adiposity and triggers early-onset obesity[10]. An association between the visceral adiposity index, daily energy intake, and MetS has been reported in children and adolescents with obesity aged 8–15 years[11]. Thus, fetal metabolic alterations and early simultaneous consumption of high-fructose (HFr) and high-saturated fat (HF) diets may enhance the pathophysiological mechanisms that contribute to the development of MetS.
Fructose, in HFr, is a glucose isomer widely used as a glucose substitute owing to its high sweetening power. It is a carbohydrate with the highest lipogenic power because its glycolytic metabolism is not subjected to negative feedback regulation. Thus, the hepatic metabolism of fructose yields large amounts of substrate for lipogenesis[12-14]. Moreover, HFr consumption increases hepatic uric acid production, which directly contributes to insulin resistance[15]. Accordingly, a consumption of > 100 g/d HFr may have serious health repercussions[16].
Trans-saturated fats, in HF, are added to UPC products whose fats have been hydrogenated to increase their shelf life[17] and are considered a risk factor for the development of coronary heart diseases, insulin resistance, and obesity accompanied by systemic inflammation[18,19].
Common pathological consequences such as insulin resistance, dyslipidemia, non-alcoholic fatty liver disease (NAFLD), hyperglycemia, and increased BP have been observed when HF or HFr diets were administered separately in animal models of MetS[20], probably because both HF and HFr diets share lipotoxicity as a common injury mechanism[20,21]. Nevertheless, at the molecular and cellular levels, HF and HFr exhibit different injury features. Moreover, when combined, HF and HFr reinforce their deleterious effects and produce more severe MetS phenotypes than when supplemented separately[22-24].
Therefore, this narrative review aimed to highlight the need for increased attention to the consequences of simult
The International Diabetes Federation and the American Diabetes Association suggest that only children older than 10 years should be examined for MetS, whereas for children younger than 10 years, only high waist circumference measurements should be used for screening[26]. According to the International Diabetes Federation, children aged 10-16 years with central adiposity (≥ 90th) and presenting with two of the following criteria: TG ≥ 150 mg/dL; HDL-C < 40 mg/dL; systolic BP ≥ 130 mmHg or diastolic BP ≥ 85 mmHg; and fasting plasma glucose ≥ 100 mg/dL[27] may be diagnosed with MetS. At the beginning of 21st century, only a few studies investigated MetS frequency in childhood and adolescence because of the various criteria used to define MetS[28]. Nevertheless, a systematic review including 169 studies was conducted in 2020 with 550405 children and adolescents from 44 countries across 13 regions worldwide[29]. The results showed that approximately 3% of the children (6-12 years) and 5% of adolescents (13-18 years) had MetS, with some variation across countries and regions. Although these numbers may appear modest, they are the result of a noticeable increase in the past four decades in the global prevalence of pediatric obesity, T2DM, and overall MetS[30].
In the pediatric population, the onset of MetS is driven by an array of risk factors. A prominent factor is the presence of a family history of MetS, particularly when one or both parents have MetS or related disorders[31]. Moreover, lifestyle choices exert a prominent influence, with suboptimal dietary patterns and lack of physical activity significantly contributing to increased risk[32]. In this context, childhood obesity and excessive adiposity particularly concentrated around the abdominal region, have emerged as critical factors that increase the risk of MetS in children and adolescents[33]. This phenomenon is closely linked to a noticeable increase in the availability of high-fat foods and sugar-sweetened drinks, the consumption of which has markedly increased during the past decades, coinciding with an exponential increase in MetS incidence[34-39]. Furthermore, a high-fat diet exacerbates vascular oxidative stress and endothelial dysfunction before the onset of insulin resistance and systemic oxidative stress, thereby contributing to the onset of MetS[40].
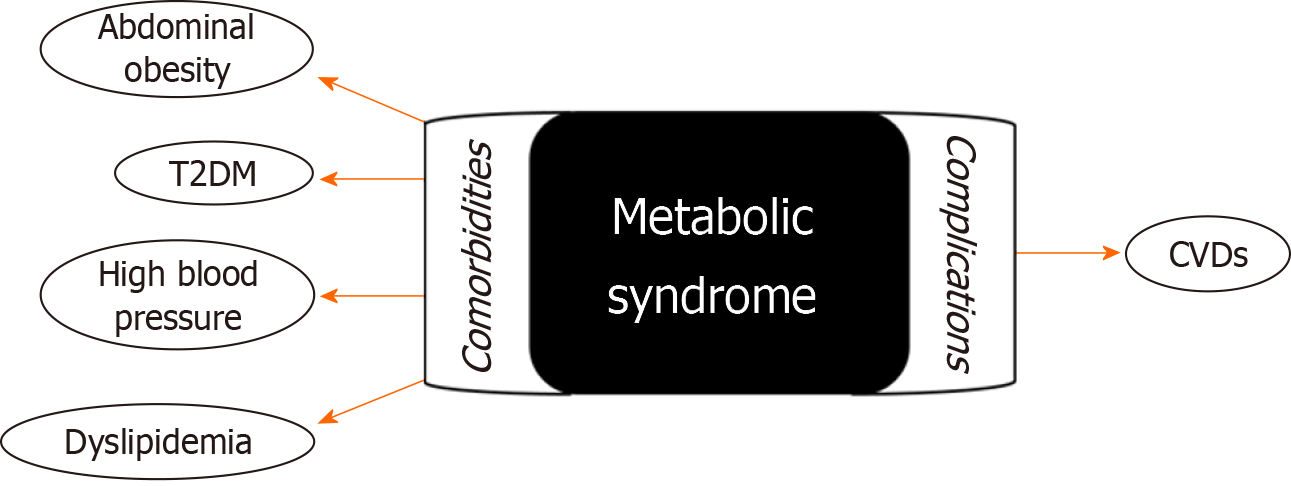
Children and adolescents with obesity frequently display endocrine comorbidities including T2DM, dyslipidemia, polycystic ovary syndrome, central precocious puberty, and MetS[41]. MetS is not an isolated condition, but rather a precursor to a multitude of comorbidities and potential complications.
Among the most common comorbidities associated with MetS in children and adolescents are abdominal obesity, high BP, dyslipidemia, and T2DM[42-44]. In addition, MetS may lead to complications affecting various organ systems, including the liver and kidney[45]. Furthermore, long-term complications of MetS increase the risk of developing cardiovascular diseases (CVDs), such as atherosclerosis and coronary artery disease (Figure 1)[46,47].
The intermediate metabolism of carbohydrates and lipids converges during acetyl-CoA formation for ATP generation or carbon molecule buildup to cope with the biosynthetic needs of the cells. The fate of these fuels is determined by an intricate network of signals involving the endocrine system, and cellular energy needs manifest as variations in the ATP/ADP and nicotinamide adenine dinucleotide [NAD(P)]/NAD(P)H ratios, which control metabolic fluxes via allosteric and post-translational modifications of enzymes. Here, we briefly summarize the fundamental aspects of fructose and fatty acid metabolism and their influence on the development of insulin resistance, emphasizing the disturbances in lipid metabolism. The effects of excessive consumption of carbohydrates and lipids on the overall alterations that influence MetS have been described elsewhere[20,24].
Fructose consumption has not been associated with health issues until recently. Indeed, in the middle of the 20th century, fructose was used as a sweetener by individuals with diabetes because its metabolism is not insulin-dependent. Moreover, it increases acetone synthesis, restores nitrogen balance, and decreases water loss[48]. The following paragraphs summarize the biochemical basis of this notion has changed completely.
Fructose in sugar-supplemented beverages is usually added as HFr corn syrup-55, which is composed of a mixture of 55% fructose and 45% glucose in the form of monosaccharides[49], allowing rapid fructose absorption. Fructose does not undergo enzymatic hydrolysis in the intestine. Fructose absorption by the brush border and basolateral membranes of the small intestine is carried out by the carbohydrate transporter GLUT-5, although the GLUT-8 transporter may also be involved in this process[50-52].
Once in portal blood, fructose is transported into hepatic cells by GLUT2[53]. Fructose is metabolized in hepatocytes via fructolysis, which involves three enzymatic steps that yield triose-phosphate molecules that are available for lipogenesis. Fructose is first phosphorylated by fructokinase to fructose-1-phosphate (F1P), which is cleaved by aldolase B into dihydroxyacetone phosphate (DHAP) and glyceraldehyde; glyceraldehyde is subsequently phosphorylated to glyceraldehyde-3-phosphate (G3P) by triokinases[54]. The unregulated and rapid phosphorylation of fructose by fructokinase and the bypass of fructose metabolism from the major regulatory steps of glycolysis explain the negligible concentration of fructose in the systemic circulation and increased production of triose phosphate available for glycolytic and gluconeogenic pathways[20].
The metabolic fates of G3P and DHAP are dependent on nutritional status, as triose phosphates are involved in gluconeogenesis and de novo lipogenesis (DNL)[55] in starved and fed animals, respectively. In fed animals, triose phosphates are converted to pyruvate, causing enhanced flux into the tricarboxylic acid cycle and increased citrate production, which is exported to the cytosol and transformed by ATP citrate lyase into cytosolic acetyl-CoA, the starting point for DNL[56]. DNL is stimulated by at least two factors derived from fructokinase-mediated fructose phosphorylation, namely, elevated uric acid synthesis supported by phosphate depletion[57], and F1P as a signaling molecule[58]. Moreover, excessive uric acid impairs insulin signaling, thus contributing to insulin resistance[59].
DNL is mainly involved in the development of certain features of MetS, including elevated blood levels of total cholesterol, TG, and free fatty acids (FFA)[60]. At the intracellular level, DNL increases the concentrations of diacylglycerols and ceramides in the liver and skeletal muscle, driving local and systemic insulin resistance[61,62]. Oxidative stress and chronic inflammation are also involved in insulin resistance and may be enhanced by fructose-induced DNL[63].
The hepatic accumulation of fatty acids, a characteristic of MetS, can be due to excessive fat ingestion. TG in HF diets are emulsified by bile acids within the intestinal lumen and then hydrolyzed primarily by pancreatic lipase to sn-2-monoacylglycerols and FFA[64]. In enterocytes, FFA are re-esterified, and the resultant TG are incorporated into chylomicrons for entry into circulation. Chylomicrons are taken up by muscle and adipose tissue owing to the activity of lipoprotein lipase, whereas chylomicron remnants are taken up by parenchymal liver cells[65,66]. An HF diet increases the blood levels of chylomicrons and their remnants. Abnormal chylomicron metabolism in diabetes is associated with atherosclerosis development[67].
Conversely, although the role of passive diffusion in fatty acid transport[68] is small, the active uptake of circulating fatty acids by the liver is dependent on fatty acid transporter proteins (FATP), the expression of which is regulated by peroxisome proliferator-activated receptor gamma. The FATP2 and FATP5 isoforms are found primarily in the liver. In hepatocytes, FFA may be metabolized in the mitochondria via fatty acid β-oxidation to produce ATP and ketone bodies. FFA can also be esterified to G3P and cholesterol for the synthesis of TG and cholesteryl esters, respectively, which accumulate in hepatocytes as small droplets or are secreted into the bloodstream as very low-density lipoprotein particles[69]. An imbalance between fatty acid utilization for energy generation, very low-density lipoprotein secretion, and TG storage causes lipotoxicity in hepatocytes, leading to the development of NAFLD and impaired insulin signaling.
Mitochondrial fatty acid β-oxidation produces large amounts of flavin adenine dinucleotide (FADH2), NADH, and acetyl-CoA. NADH and FADH2 are reoxidized to NAD+ and FAD, respectively, by the mitochondrial electron transport chain (ETC). Excessive TG accumulation in hepatocytes causes mitochondrial dysfunction, inefficient electron transport through the ETC, electron leakage from the redox sites of ETC complexes, and generation of reactive oxygen species (ROS)[70]. Therefore, the overconsumption of fat impairs mitochondrial fatty acid β-oxidation by impairing NADH and FADH2 oxidation. This produces reductive stress in the form of high NADH/NAD+ ratios, which together with excess acetyl-CoA lead to the reversible acetylation and inhibition of several enzymes participating in catabolic pathways, including acyl-CoA dehydrogenases involved in β-fatty acid oxidation and ETC complexes, among others[71]. Overall, in addition to causing the accumulation of lipids such as ceramides and diacylglycerol, which interfere with the insulin receptor[61], enzyme inhibition enhances the impairment of ETC function and causes overproduction of ROS. In turn, ROS oxidize unsaturated lipids in fat deposits, causing lipid peroxidation. Moreover, HF diet-induced ROS triggers proinflammatory signaling, inducing nuclear factor kappa B -dependent production of proinflammatory cytokines, such as tumor necrosis factor (TNF)-α, TNF- β, interleukin-6, interleukin-1β, and inducible nitric oxide synthase. Overall, this enhances insulin resistance and contributes to dyslipidemia[72,73], whereas, at the systemic level, dyslipidemia, ROS, and inflammation favor the development of atherosclerosis and CVD.
As mentioned above, there is a growing interest in exploring the combined effects of HF and HFr diets to mimic the composition of Western diets comprising high-fat foods accompanied by sugar-sweetened beverages typically served in fast food establishments. At least three recent studies in rodents have shown that fructose addition to an HF diet worsens the deleterious effects of HF diet by increasing DNL and enhancing damaging effects of HF on mitochondrial function, oxidative stress, hepatic lipid metabolism, and insulin resistance[22,23,62]. Rats fed the HF + HFr diet displayed higher levels of blood TG, FFA, and insulin than those fed an HF diet alone. The increase in both FFA and TG was believed to be the result of DNL stimulated by HFr consumption, as an HF diet inhibits DNL in the liver and whole body, whereas the opposite occurs with an HF + HFr diet[74]. Hepatic insulin resistance was also higher with the HF + HFr diet than that with the HF diet, which was in concordance with higher ceramides levels, TG levels, mitochondrial dysfunction, and steatosis in rats fed an HF + HFr diet than that in rats fed only an HF diet[62].
The worsening of mitochondrial dysfunction, i.e., impaired mitochondrial fatty acid oxidation, by the HF + HFr diet may be central to the more deleterious effects of this diet than of the HF or HFr diet alone[62], as impaired oxidative phosphorylation and increased severity of steatosis was observed in rats consuming HF + HFr than those consuming only an HF diet or HFr diet[23]. Moreover, mitochondrial dysfunction coincided with higher ROS production and oxidative stress with this diet than that with an HF diet or an HFr diet alone[23,62].
Fructose addition to an HF diet increased the acetylation and inhibition of carnitine-palmitoyl transferase 1a (CPT1a), which mediates fatty acid translocation of fatty acids into the mitochondrial matrix and is the rate-limiting step in fatty acid β-oxidation, and long-chain acyl-CoA dehydrogenase (ACADL), thus contributing to increased liver steatosis[22].
Thus, compared to an HF or HFr diet alone, fructose addition not only worsens but also accelerates the effects of an HF diet by impairing ETC function to a higher degree, probably eliciting higher NADH/NAD+ ratios that induces a greater inhibition of fatty acid β-oxidation via increased acetylation of ACADL and CPT1a. This, in turn, may increase the concentrations of incomplete products of fatty acid oxidation, such as ceramides and TG, causing impaired insulin signaling, which may be reinforced by the exacerbated ROS production induced by HF + HFr diets in the liver mitochondria. Therefore, this raises the question of whether, in childhood, the frequent consumption of UPC fast foods containing HF + HFr triggers early onset of MetS than the consumption of foods containing only HF or HFr, and whether this worsens the outcomes of MetS, namely diabetes and its complications, including early onset of CVD.
A review of the methodology of clinical trials and their limitations, as well as cohort, cross-sectional, and other observational and associative studies related to both HF and HFr consumption patterns in children of different ages, ethnicities, and metabolic conditions, will help to understand the role of HF and HFr diets in metabolic disorders and MetS development. The prevalence of disorders related to lipid metabolism, such as obesity, diabetes, hyperlipidemia, dyslipidemia, glucose and insulin intolerance, hepatic steatosis, and heart risk, increases with age; however, these metabolic risk factors in childhood could predispose to the development of chronic degenerative diseases in adulthood[75-77]. Therefore, it is important to explore how HF and HFr individually, or the simultaneous intake of HF + HFr differentially alters metabolic parameters from childhood, becoming imminent risk factors in the subsequent development of metabolic diseases.
The Dietary Guidelines for Americans 2020-2025 and the joint World Health Organization (WHO)/Food and Agriculture Organization expert consultation recommend limiting saturated fat intake to < 10% of the total energy intake[78]. This is mainly because high saturated fat intake is associated with an increase in metabolic risks, such as high body mass index (BMI), hepatic steatosis, insulin resistance, and high levels of TG, total cholesterol, low-density lipoprotein cholesterol (LDL-C)[79-81]. A high-fat diet and insulin resistance also promote FFA accumulation in the liver, leading to a loss of regulation of normal lipid liver metabolism and the development of NAFLD, which frequently occurs in children and has been reported in cases of severe hepatitis and hepatic fibrosis in children aged 10 years[82].
The focus on trans fatty acids (tFA) as a causal factor of MetS has increased in recent years. Although saturated fatty acids can be derived from natural sources, tFA have an industrial origin in the production of UPC foods. Industrial tFA are produced by the partial hydrogenation of unsaturated vegetable oils, which occurs when vegetable oils are heated. The main sources of industrial tFA are margarine, commercially baked products, deep-fried fast foods, packaged snack foods, and other UPC foods[76,83]. tFA have been identified as an important cause of CVDs and their resulting clinical endpoints, such as stroke and heart attack. Evidence indicates that a high tFA intake is associated with an increased probability of coronary heart disease. Although CVDs and coronary heart diseases mainly present in adults, atherosclerotic lesions in the aorta and coronary arteries can begin to appear in childhood and are positively associated with dyslipidemia and other CVD risk factors, such as elevated total and LDL-C levels. The WHO has prioritized elimination of industrial tFA in processed foods and recommends limiting tFA intake to < 1 energy percentage. Moreover, the United States Food and Drug Administration (FDA) has determined that partially hydrogenated oils are no longer safe for use in human food. However, evidence regarding an association between HF intake and either MetS development or lipid and glucose profile modifications is lacking[84].
Fructose is one of the sweetest naturally occurring carbohydrates, either in the form of sucrose or high-fructose corn syrup (HFCS). It is an important lipogenic substrate that stimulates DNL. Fructose consumption has gradually increased worldwide over the last four decades as the UPC food industry has dramatically expanded, as has sugar-sweetened beverage consumption in children[85].
The main food types with a considerable fructose contribution consumed by children are industrialized and homemade sweetened milk-based drinks and other beverages; whole fruits contribute little to fructose intake[86]. Even moderate consumption of fruits (0.8-1.5 servings per day, 6-7 d a week) can improve the lipid profile by decreasing the total cholesterol, serum TG, and LDL-C levels[87]. Although the WHO discourages feeding fruit juices or other sweetened beverages to children aged under 2 years, babies from 6 months of age drink fructose beverages or derivatives, thus modifying their water consumption patterns and increasing health risks[88]. Additionally, the WHO recommends limiting the consumption of sugar-sweetened foods (including fructose, sucrose, and HFCS) to 10% of the total energy intake and suggests that a reduction of 5% has beneficial health effects[89]. Fructose restriction from 28% to 10% in children aged 3–18 years improved glucose metabolism and lipid profile and decreased levels of blood aspartate aminotransferase, a liver and heart injury marker[90]. In the United States, 13% of the population consumes more than 25% of their total energy intake from added sugars. Alarmingly, the prevalence of added sugar is estimated to be higher in Mexico, ranging from 58% in school-aged boys to 85% in adolescent girls; total sugar intake in the Mexican diet was 365 kcal/d, of which 238 kcal/d was from added sugars and only 127 kcal/d from intrinsic sugars[91,92]. Huerta-Ávila et al[86] showed an association between fructose intake and high adiposity and cardiometabolic risk factors (i.e., high levels of blood glucose, insulin, TG, total cholesterol, LDL-C, and HDL-C) in children from Mexico City (Table 1).
| MetS inductors and interventions | Population (age, n) | Measured parameters | Outcomes | Type of study | Ref. |
| Saturated fatty acids | 6-16 (n = 108); OB children | lipid profiles | High saturated fat intake was associated with higher BMI, NAFLD positivity. TG, total cholesterol, LDL-C and HOMA-IR were significantly higher in SFA consumption group | Cross-sectional study | Maffeis et al[81] |
| Fructose restriction | 8-18 (n = 20) Children with obesity and MetS | Lactate in serum (related with liver fat fraction and visceral adipose tissue) | Fructose restriction produced a 50% decrease in lactate, which was related with decreased de novo lipogenesis and insulin sensitivity | Clinical trial | Erkin-Cakmak et al[85] |
| Sugar sweetened beverages (Fructose) | 6-12 (n = 1,087); OB children | Adiposity (BMI, WC), cardiovascular risk markers (glucose, insulin, HOMA-IR, TG, LDL-C, HDL) | TG, insulin, TC and LDL concentrations and HOMA-IR were significantly higher in OB children | Association study | Huerta-Ávila et al[86] |
| Moderate fruit consumption | 5-19 (n = 14, 755) | Lipid Profile (TC, TG), fasting serum insulin (HOMA-IR) | Moderate fruit consumption (1.5 serving per day 6-7 d a week) was associated with lower odds of lipid disorders, improving the childhood lipid profiles | Cluster-controlled trial | Liu et al[87] |
| Fructose restriction | 3-18 (Latino children n = 2; African-American children n = 16) | Anthropometric parameters (BW, WC, BMI), BP, biochemical measurements (serum lactate, TG, cholesterol and others), glucose and HOMA-IR | Fructose restriction (reduction from 28% to 10%) in children showed reductions in diastolic BP, serum lactate, TG, LDL-C. Glucose tolerance and hyperinsulinemia | Clinical trial | Lustig et al[90] |
| Fructose | 12-16 (n = 1454) | HOMA-IR, uric acid, anthropometric parameters (BW, WC, BMI, body fat percentage) | High consumption of sugar sweetened beverages (> 350 mL/d) were more likely to have elevated fasting serum insulin and HOMA-IR, WC and serum uric acid compared to those not having fructose consumption | Cross-sectional study | Lin et al[96] |
| UPC foods (high fructose-high fat) | 4-8 (n = 307) | Anthropometric parameters (BW, WC, BMI, WHR) and glucose profile | An increase in WC was observed in children with a higher UPC consumption. Not a direct association with altered glucose metabolism was observed | Longitudinal study | Costa et al[97] |
| UPC foods (high fructose-high fat) | 3-4 (n = 346); 7-8 (n = 307) | Lipid profile (TC, TG, HDL, LDL-C) | The higher consumption of UPC, the higher increase in total cholesterol and LDL-C and altered lipoprotein profiles in children | Longitudinal study | Rauber et al[99] |
| UPC foods (high fructose-high fat) | 12-19 (n = 210) Children from Brazilian family program | Urinary fructose excretion Anthropometric parameters and biochemical parameters | Significant association between MetS and the consumption of UPC foods | Cross- sectional study | Tavares et al[100] |
| Fructose | 7-12 (n = 27); 13-15 (n = 25); 16-16 (n = 32); OB children | Anthropometric parameters (BW, Hgt, WC, BMI and WHR), serum lipid profiles | Higher fructose intake from beverages correlate positively with the percentage of body fat, WC, WHR, TC, TG and increased atherogenic indices | Observational study | Czerwonogrodzka-Senczyna et al[101] |
| Fructose | 9-16 (n = 246) | Urinary fructose excretion | Metabolic dysfunctions and urinary excretion only at very high fructose intake levels (> 25% of total energy intake) or hypercaloric diets | Cohort study (DONALD) | Perrar et al[102] |
| Trans fatty acids | 6-13 (n = 54); OB children | Anthropometric parameters, glucose and insulin. HOMA-IR, total lipids, postprandial levels of trans fatty acids | Obese children showed hyperinsulinemia and increased insulin resistance compared with controls. No differences for fasting plasma tFA or dietary tFA intake were observed | Clinical trial | Larqu et al[103] |
| High dairy fat products (total and saturated fat intake) | 4-13 (n = 174) Intervention with low dairy fat products | Anthropometric parameters Pentadecanoic acid and lipid profile | Total fat and saturated fat intakes from dairy foods were lower in the intervention group (low fat dairy products consumption) but did not alter energy intakes or measures of adiposity | Randomized controlled trial | Hendrie and Golley[104] |
Health risks associated with HFr consumption in pediatric populations include dental decay, overweight, obesity, insulin resistance, diabetes, NAFLD, hypertension, and cardiovascular risks[93-95]. Studies have shown that those who consume a greater amount of sugar-sweetened beverages are more likely to have elevated fasting serum insulin levels, insulin resistance, waist circumference, and serum uric acid levels[96].
UPC foods significantly contribute to HF and HFr intake in children. Approximately 41.8% of preschoolers and 47.8% of school-age children have high intakes of these products[97]. The consumption of UPC products has undergone a huge increase worldwide, with high consumption among children up to 12 months of age, with an increase observed in 16-month-old children[98]. Thus, consumption of UPC products early in life in children aged 3-8 years plays a role in altering lipoprotein profiles and could be a key determinant of CVDs. Moreover, high consumption of UPC foods in 12-18-year-old adolescents was associated with the prevalence of MetS and its associated features. Likewise, epidemiological studies (Table 1) have correlated the consumption of UPC foods with metabolic alterations and chronic diseases, such as obesity, T2DM, NAFLD, and CVDs[99-105].
Children and teenagers who consume HFr/HF diets usually develop metabolic disorders such as obesity, NAFLD, insulin resistance, T2DM, and other MetS components[106]. Therefore, it is important to educate the public regarding the risk of excessive consumption of these products and unhealthy lifestyles among children who develop several MetS components. Nevertheless, accomplishing this goal has been challenging, as initiatives aimed at decreasing the consumption of products containing HFr/HF in children and interventions designed for the treatment of associated diseases have not succeeded[107], as evidenced by the current childhood obesity pandemic[108]. However, many research groups, institutions, and organizations worldwide continue to develop strategies to reduce the consumption of products containing HF/HFr and treat disorders caused by excessive intake. An essential aspect of these prevention and treatment programs is the need for multidisciplinary teams to address the problems and improve the probability of success. These programs should prioritize dietary guidance while emphasizing physical exercise and, if needed, pharmacological, psychological, and surgical interventions[109].
Owing to the limited efficacy of therapeutic interventions for the treatment of MetS associated with HFr/HF diets, there has been growing interest in preventive strategies. This may be feasible because many risk factors, excluding genetic influences, can be modified to serve as targets for preventive initiatives[110]. Interventions for both the prevention and treatment of the negative consequences of HF/HFr diets in children focus on reducing the intake of products containing HF or HFr, high-glycemic, and calorie-dense foods[111,112]. Moreover, exclusive breastfeeding should be promoted in infants up to 6 months of age[113]. In addition, children should be encouraged to consume vegetables, fruits, low-fat dairy products, moderate quantities of whole grains, fish, poultry, and nuts, and advised against excessive sodium intake[114].
Currently, there is no effective general dietary intervention that can be applied in all cases or all pediatric populations; thus, a personalized approach is recommended. However, for the management of children who are at risk or have already developed metabolic alterations owing to the consumption of HFr/HF diets, several dietary alternatives have been proposed. One key approach is to guide children on appropriate food portions. A 12-month trial revealed that nutrient-balanced portion-controlled diets using meal plans (55%-60% carbohydrates, 10%-15% protein, and 30% fat) were effective in decreasing BMI among children with obesity. Furthermore, these diets resulted in reduced hunger compared to low-carbohydrate and reduced glycemic load diets[115]. The traffic light diet is another, simple alternative that reduces energy intake and encourages overweight and obese children to consume healthy diets[116]. It involves restricting foods based on their caloric content, and categorizing them by color: Green foods – can be eaten without restriction; yellow foods – should be consumed cautiously; red foods - stop and think before eating these foods[117].
Furthermore, nutritional labels, including nutritional facts tables, guide daily amounts, and front-of-package labels, have been implemented in numerous countries worldwide. These labels are particularly relevant to foods with high unhealthy nutrient content, such as HF/HFr products, which require restricted consumption. Front-of-package labels are the most comprehensible to the general public, particularly to children, resulting in the highest rates of self-reported awareness[118].
In addition, the short-term efficacy of low-carbohydrate and low-fat diets has been demonstrated. Within 6 months of implementation, these regimens resulted in a significant reduction in the BMI of children with obesity and considerably ameliorated fatty liver disease[119]. The fructose and glycemic index/glycemic load diet (FRAGILE) dietary intervention showed that a reduction in fructose intake, particularly HFCS, resulted in a significant reduction in metabolic risk factors and hepatic injury in children with NAFLD[120].
Established diet plans, such as the Mediterranean diet and the Dietary Approaches to Stop Hypertension (DASH) diet, have been applied in patients with metabolic disorders, such as obesity, hypertension, cardiovascular risk, and MetS, which are usually the result of unhealthy dietary patterns, including HF/HFr diets. These diets contain a substantial portion of plant-based foods, including fruits and vegetables, whole grain products, legumes, nuts, and seeds; moderate daily consumption of dairy products, and weekly consumption of poultry and fish[114]. Six weeks of adherence to the DASH diet improved MetS markers and high BP in a group of adolescent women, according to the findings of a randomized controlled trial designed to examine the effect of the DASH diet on metabolic outcomes[121]. In their evaluation of the effectiveness of a Mediterranean-style diet in children and adolescents with obesity, Velázquez-López et al[122] found that a 16-week intervention decreased BMI, glucose levels, and lipid profiles.
Other therapeutic options, such as probiotics or prebiotics, have been investigated to modulate the intestinal microbiota composition and improve alterations caused by the HFr/HF diet, such as obesity, MetS, and NAFLD. Various hypotheses have been proposed to explain the potential therapeutic benefits of probiotics and prebiotics in patients on HFr/HF diets[123]. Prebiotic supplementation inhibited fatty acid synthesis in animal models, which possibly reduced fructose-induced hepatic TG accumulation. This may be due to decreased expression of fatty acid synthase and acetyl CoA carboxylase, which are enzymes that regulate hepatic lipogenesis[124]. Probiotics improved inflammatory liver damage through the modulation of TNF-α and insulin resistance[125]. It has been demonstrated that the consumption of an HF/HFr diet over 8 wk resulted in both gut dysbiosis and a change in the permeability of the gut barrier. As an effective probiotic, Limosilactobacillus reuteri DSM 17938 may be used to prevent the detrimental effects of an HF/HFr diet on the gut[126].
Compelling evidence indicates that modern lifestyle, including a diet composed primarily of HF/HFr-containing UPC foods and a sedentary lifestyle, has socially affected early childhood, as evidenced by an increase in the prevalence of childhood obesity[127]. Consequently, interventions promoting physical activity are frequently employed to counteract the negative effects of HF/HFr consumption in children. A Mayo Clinic review on childhood obesity outlined the following characteristics of physical activity: Age, individual preferences regarding the type of physical activity, and exercise tolerance, which should be considered when establishing physical activity objectives for children. Children aged ≥ 6 years are recommended to participate in at least 60 min of daily physical activity. Toddlers should have a designated time of 60–90 min in an 8-h day for moderate to vigorous physical activity. Preschoolers, however, are recommended to have a longer duration (90–120 min) of physical activity[109]. Parents and schools should encourage children and adolescents to participate in outdoor play and recess activities and walk or bike to school. Positive factors affecting physical activity levels include family, peer, and communal encouragement; proactive and athletic self-perception; desire to prevent weight gain; and personal achievement[128].
Children who are obese face challenges in initiating and sustaining physical activity programs. These issues arise from limited motor abilities resulting from excessive body weight and an inclination toward a sedentary lifestyle. Consequently, such children often develop unpleasant emotions associated with physical activity, leading to a higher possibility of ceding physical activity and establishing a vicious cycle wherein sedentary habits become more prevalent[129]. Stabelini Neto et al[130] suggested that to promote a healthy metabolic profile, adolescents should perform at least 88 min/d of moderate-to-vigorous physical activity. These activities should include games, sports, recreation, planned exercises, and transportation, both in schools and communities. In children, low levels of physical activity are associated with an increased risk of developing MetS[131]. Lifestyle intervention in elementary school students involving a physical activity promotion program to raise awareness among parents and students about MetS-associated risks associated with MetS significantly reduced some major MetS components, including insulin resistance, high BP, and obesity[132].
Physical activity can also improve the composition and functionality of the gut microbiota, representing another benefit that lifestyle changes can bring to children affected by HF/HFr diets who have developed complications such as obesity, NAFLD, or MetS[133].
Pharmacological treatment may be initiated when children fail to lose weight within 6–12 months of lifestyle modification or when they exhibit significant cardiometabolic risks associated with the consumption of HFr/HF diets, although few approved drugs are available for use in children[132]. The FDA has approved the following five drugs for chronic weight management in pediatric patients aged ≥ 12 years who are obese, with a BMI of 30 kg/m2 and weight > 60 kg[134]: Orlistat, phentermine/topiramate (PHEN/TPM), liraglutide, semaglutide, and setmelanotide; these drugs employ various mechanisms of action to combat childhood obesity. Orlistat is a widely recognized medication for weight management in overweight children and adolescents and functions as a selective inhibitor of gastrointestinal lipases, thus inhibiting dietary fat absorption[135]. Orlistat, in conjunction with lifestyle modifications, has been shown in clinical studies to substantially decrease BMI and improve metabolic parameters in children with obesity[136]. As orlistat inhibits the absorption of fat-soluble vitamins, multivitamin supplementation is recommended. Adverse effects of orlistat include diarrhea, abdominal pain, flatulence, and greasy feces[109].
PHEN/TPM is frequently prescribed for short-term weight loss interventions owing to its appetite-suppressing properties. A reduction in caloric intake is induced via appetite suppression when topiramate, which acts on GABA A receptors, is combined with the noradrenergic agonist, phentermine[58]. The use of PHEN/TPM and lifestyle interventions in a 56-wk trial including adolescents with obesity showed a statistically significant reduction in BMI and favorably affected TG and HDL-C levels. Common adverse events include paresthesia, dizziness, dysgeusia, insomnia, constipation, palpitations, tachycardia, and dry mouth[137].
Liraglutide and semaglutide are agonists of the glucagon-like peptide-1 (GLP-1) receptor and affect glucose metabolism and appetite regulation by binding to GLP-1 receptors located in both the brain and peripheral tissues[138,139]. Kelly et al[140] showed that in obese adolescents, the use of liraglutide plus lifestyle therapy led to a significantly greater reduction in BMI. Common side effects of GLP-1 receptor agonists include gastrointestinal symptoms such as nausea, vomiting, diarrhea, and constipation, which typically diminish over time[141].
Setmelanotide, a synthetic analog of melanocyte-stimulating hormone, is a promising treatment for some types of pediatric obesity. It functions as an effective melanocortin-4-receptor (MC4R) agonist in children and adolescents with obesity resulting from genetic mutations that impact the MC4R pathway[142].
Although the FDA has not yet approved metformin for the treatment of obesity in children, there is evidence of its use in studies on pediatric patients with other metabolic disorders, such as insulin resistance, polycystic ovary syndrome, MetS, and childhood T2DM, where metformin has been associated with weight loss, improved insulin sensitivity and glucose levels, and reduced inflammation[131]. Because of the limited pharmacological options for treating metabolic disorders related to HFr/HF diets, such as obesity, MetS, and NAFLD, there is an extensive field of research wherein new therapeutic alternatives are being analyzed to address these issues that affect both pediatric and adult populations. Table 2 shows a summary of recent studies focused on the treatment of these metabolic disorders, which are not limited to the pediatric population. These studies include compounds present in olive oil such as oleanolic acid alone[143] or in combination with metformin[144], as well as some polyphenols[145] and triterpenic acids present in this oil[146]. Other compounds of natural origin under study are γ-oryzanol with ferulic acid[147], quercetin[148], ursolic acid[149], and the non-protein amino acid citrulline[150]. Microencapsulation of herbal extracts with beneficial properties against metabolic syndrome has been studied to improve their pharmacokinetic properties[151]. On the other hand, the use of agents that improve intestinal microbiota and glucose and lipid levels by influencing host metabolism has been explored[152,153]. Likewise, the beneficial effect on lipid metabolism of such divergent drugs as the antihypertensive drugs amlodipine and valsartan[154], GLP-1 receptor agonists[155] and acetyl CoA carboxylase inhibitors of natural or synthetic origin[156] have been explored.
| Model/population | Treatment | Outcomes | Ref. |
| Fructose-induced NAFLD; Neonatal rats | Oleanolic acid | Attenuates the subsequent development of HFr diet-induced NAFLD | Nyakudya et al[143] |
| HFr induced metabolic dysfunction; Rats (Rattus norvegicus) | Oleanolic acid (60 mg/kg); metformin (500 mg/kg) | Both treatments increased mono- and polyunsaturated FFAs, associated with increased glut-4, glut-5 and nrf-1 and decreased acc-1 and fas | Molepo et al[144] |
| MetS induced by HFr diet; female mice | Tyrosol, hydroxytyrosol and salidroside | Improved glucose metabolism and lipid metabolism, including reduced levels of total cholesterol insulin, uric acid, LDL-C, and aspartate aminotransferase | Zhan et al[145] |
| Obesity and related diseases in humans | Olive oil triterpenic acids | Improves glucose and insulin homeostasis, lipid metabolism, adiposity and cardiovascular dysfunction in obesity | Claro-Cala et al [146] |
| HF and HFr diets induced metabolic dysfunction; Sprague dawley rat | Wheat flour, enriched with γ-oryzanol, phytosterol, and ferulic acid | Alleviates hepatic lipid accumulation and insulin resistance through their elevation in the phosphorylation of AMPK and Akt | Guo et al[147] |
| HF diet induced NAFLD; C57BL/6J mice | Quercetin | Decreased insulin resistance and NAFLD activity score, by reducing the intrahepatic lipid accumulation through its ability to modulate lipid metabolism gene expression, cytochrome P450 2E1 (CYP2E1)-dependent lipoperoxidation and related lipotoxicity | Porras et al[148] |
| HFr induced NAFLD; Sprague dawley rats | Ursolic acid | Ursolic acid administration against dietary fructose-induced NAFLD in newborn rats by reducing fructose-induced hepatic lipid accumulation | Mukonowenzou et al[149] |
| Mouse 3T3-L1 fructose-induced NAFLD | Citrulline | Prevented hypertriglyceridemia and attenuated liver fat accumulation | Jegatheesan et al[150] |
| Sprague-Dawley rats MetS; in vitro and in vivo studies | Nanoformulations of herbal extracts | Decrease the lipid profile, inflammation, oxidative damage, and insulin resistance in in vitro and in vivo models of MetS-related complications | Nouri et al[151] |
| MetS humans | Pharmabiotics | Improves gut microbiota profile wich influence serum lipid levels, BP, neuroendocrine cells and immune functions via regulating the metabolism of the host | Nguyen et al[152] |
| MetS; in vivo studies | Seaweed-derived bioactive components | Modulate the gut microbiota by reversing the Firmicutes/Bacteroidetes ratio, increasing the relative abundance of beneficial bacteria and decreasing the abundance of harmful bacteria; these compounds increase the production of short-chain fatty acids and influence glucose and lipid metabolism | Zang et al[153] |
| HFr induced adiposity | Valsartan; Amlodipine | Both treatment reduced triacylglycerol storage in adipocytes by inhibiting PU.1 | Chou et al[154] |
| Hepatic steatosis induced by HFr diet; Wistar rats | β-catenin | Mediates the effect of GLP-1 receptor agonist on ameliorating hepatic steatosis | Gao et al[155] |
| NAFLD and nonalcoholic steatohepatitis Humans | Acetyl-CoA Carboxylase inhibitors | Therapeutic target for MetS as a key regulatory role in fatty acid synthesis and oxidation pathways | Chen et al[156] |
Despite the substantial amount of evidence about the negative effect on metabolic health of HF + HFr diets, there is still controversy about whether fructose consumption is harmful to health in humans. One of the main arguments against a specific effect of fructose on the increase of intrahepatic fat, serum triglyceride levels and insulin tolerance has been that its effects are related to the supply of excess energy and that other equally energy-dense nutrients can cause the same metabolic alterations as fructose[157].
Two meta-analyses showed that isocaloric replacement of other carbohydrates with fructose produced better glycemic control in people with diabetes and had no effect on markers of NAFLD in healthy subjects. However, as stated by the authors of these studies, the data should be taken with caution because the studies were conducted in small populations and over relatively short periods of time[158,159]. Likewise, in another study where the effects of isocaloric intake of glucose and fructose were analyzed in healthy, centrally overweight men, no change in hepatic TG or markers of liver damage were observed. However, it should be noted that the carbohydrates were consumed for a period of two weeks[160].
In contrast to the above studies, in a study conducted with 37 overweight adult individuals, a small decrease in intrahepatic fat was observed with a fructose-restricted diet compared to an isocaloric diet without fructose restriction, although no difference was observed between serum lipid content and glucose tolerance. As in the aforementioned studies, the major limitation of this study was its short duration of six weeks[161]. Consistent with the above study, in another study in 94 lean healthy men where daily carbohydrate intake was increased by daily intake of fructose or sucrose-sweetened beverages for 7 wk, an increase in hepatic lipid synthesis was observed, whereas this was not observed in individuals who ingested glucose-sweetened beverages[162]. A recent meta-analysis has concluded that the effects of fructose on adiposity are rather related to the source of fructose and the energy level at which it is consumed. Intake of sugar-sweetened beverages in amounts above 100 g/d or providing more than 20% of energy was found to result in moderate increases in adiposity. On the contrary, the consumption of fructose sources such as honey, dried fruits, fruits and 100% fruit juice in amounts above 100 g/d or providing more than 20% of the energy do not show harmful effects, and even have moderate beneficial effects[163].
On the other hand, an umbrella review of existing meta-analyses of the effects of sugar consumption on health outcomes showed an association between consumption of sugar-sweetened beverages at hypercaloric levels and an increased risk of MetS[164]. This has led to the recommendation to minimize consumption of sugar-sweetened beverages[165]. This is consistent with the conclusions of some reviews evaluating the effects of fructose consumption in children. The idea that fructose consumption has a negative effect on the development of NAFLD is not without controversy also in children and adolescents[166], which is due in part to the fact that there were only four studies evaluating the effect of fructose or sucrose restriction on liver fat in young people as of 2023[167]. For example, a recent study where biopsies from obese and lean adolescents were analyzed showed that patients with non-alcoholic steatohepatitis have higher sugar intake and that the severity of liver disease correlates with the amount of total carbohydrate and added sugars ingested[168]. In the light of this and other observations, restriction of added sugar intake in the management of NAFLD in at-risk children and adolescents has been suggested like in adult populations[168,169].
The fast-food industry offers highly palatable products that are attractive to children. In general, these products consist of a meal containing HF and a sugar-containing beverage, with fructose as the main component of such beverages. Studies in animals administered HF + HFr diets mimic the consumption of UPC products offered by fast-food establishments better than studies using HF-only or HFr-only diets. Future studies should address the effects of dietary, physical, and pharmacological interventions on the metabolic health of the pediatric population, as cumulative evidence in animal studies shows that HF + HFr diets produce more deleterious effects on health than HF-only or HFr-only diets. This is because of the differential effects of fructose and saturated fats, as, in contrast to HF diets, fructose has a powerful ability to promote both hepatic DNL and uric acid synthesis, both leading to altered blood lipid levels and promoting insulin resistance. Moreover, HFr exacerbates the effects of HF alone by enhancing mitochondrial dysfunction, ROS production, and inflammation, which are the main factors that lead to insulin resistance, diabetes, and CVD. This raises several questions about whether the effectiveness of an intervention targeting alterations in MetS is equal when tested only in the context of HF consumption and when it is tested only in the context of HFr intake. For example, are FDA-approved drugs for the treatment of obesity in children equally effective in the alterations elicited by HF + HFr compared to alterations produced by HF-only or HFr-only? When a child frequently eats fast food, is restricting only the calories provided by fat equally effective as restricting the calories provided by sugar only? What are the more effective prebiotics, probiotics, and obesity drugs that target the alterations elicited by the combined intake of HF + HFr? These and other questions deserve further research using animal models to develop interventions that better target the metabolic alterations produced by combined fat and fructose consumption, to decrease early onset MetS-associated diseases and delay as much as possible the development of MetS later in life.
We thank the anonymous reviewer and editors for their valuable suggestions.
| 1. | Monteiro CA, Cannon G, Moubarac JC, Levy RB, Louzada MLC, Jaime PC. The UN Decade of Nutrition, the NOVA food classification and the trouble with ultra-processing. Public Health Nutr. 2018;21:5-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1167] [Cited by in RCA: 1211] [Article Influence: 173.0] [Reference Citation Analysis (0)] |
| 2. | Lustig RH, Fennoy I. The History of Obesity Research. Horm Res Paediatr. 2022;95:638-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 3. | Apovian CM. Obesity: definition, comorbidities, causes, and burden. Am J Manag Care. 2016;22:s176-s185. [PubMed] |
| 4. | Engin A. The Definition and Prevalence of Obesity and Metabolic Syndrome. Adv Exp Med Biol. 2017;960:1-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 434] [Cited by in RCA: 728] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 5. | Filgueiras AR, Pires de Almeida VB, Koch Nogueira PC, Alvares Domene SM, Eduardo da Silva C, Sesso R, Sawaya AL. Exploring the consumption of ultra-processed foods and its association with food addiction in overweight children. Appetite. 2019;135:137-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 6. | Bovolini A, Garcia J, Andrade MA, Duarte JA. Metabolic Syndrome Pathophysiology and Predisposing Factors. Int J Sports Med. 2021;42:199-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 177] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 7. | Huang PL. A comprehensive definition for metabolic syndrome. Dis Model Mech. 2009;2:231-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1429] [Cited by in RCA: 1436] [Article Influence: 89.8] [Reference Citation Analysis (0)] |
| 8. | Saklayen MG. The Global Epidemic of the Metabolic Syndrome. Curr Hypertens Rep. 2018;20:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1964] [Cited by in RCA: 2413] [Article Influence: 344.7] [Reference Citation Analysis (0)] |
| 9. | Vaquero Alvarez M, Aparicio-Martinez P, Fonseca Pozo FJ, Valle Alonso J, Blancas Sánchez IM, Romero-Saldaña M. A Sustainable Approach to the Metabolic Syndrome in Children and Its Economic Burden. Int J Environ Res Public Health. 2020;17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 10. | Catalano PM, Hauguel-De Mouzon S. Is it time to revisit the Pedersen hypothesis in the face of the obesity epidemic? Am J Obstet Gynecol. 2011;204:479-487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 260] [Cited by in RCA: 253] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 11. | Vizzuso S, Del Torto A, Dilillo D, Calcaterra V, Di Profio E, Leone A, Gilardini L, Bertoli S, Battezzati A, Zuccotti GV, Verduci E. Visceral Adiposity Index (VAI) in Children and Adolescents with Obesity: No Association with Daily Energy Intake but Promising Tool to Identify Metabolic Syndrome (MetS). Nutrients. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 12. | Cheng H, Zhou J, Sun Y, Zhan Q, Zhang D. High fructose diet: A risk factor for immune system dysregulation. Hum Immunol. 2022;83:538-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 13. | Coronati M, Baratta F, Pastori D, Ferro D, Angelico F, Del Ben M. Added Fructose in Non-Alcoholic Fatty Liver Disease and in Metabolic Syndrome: A Narrative Review. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 14. | Harrison S, Couture P, Lamarche B. Diet Quality, Saturated Fat and Metabolic Syndrome. Nutrients. 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 15. | Pan Y, Kong LD. High fructose diet-induced metabolic syndrome: Pathophysiological mechanism and treatment by traditional Chinese medicine. Pharmacol Res. 2018;130:438-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 16. | Lubawy M, Formanowicz D. High-Fructose Diet-Induced Hyperuricemia Accompanying Metabolic Syndrome-Mechanisms and Dietary Therapy Proposals. Int J Environ Res Public Health. 2023;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 27] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 17. | Islam MA, Amin MN, Siddiqui SA, Hossain MP, Sultana F, Kabir MR. Trans fatty acids and lipid profile: A serious risk factor to cardiovascular disease, cancer and diabetes. Diabetes Metab Syndr. 2019;13:1643-1647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 116] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 18. | Nagpal T, Sahu JK, Khare SK, Bashir K, Jan K. Trans fatty acids in food: A review on dietary intake, health impact, regulations and alternatives. J Food Sci. 2021;86:5159-5174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 19. | Dhibi M, Brahmi F, Mnari A, Houas Z, Chargui I, Bchir L, Gazzah N, Alsaif MA, Hammami M. The intake of high fat diet with different trans fatty acid levels differentially induces oxidative stress and non alcoholic fatty liver disease (NAFLD) in rats. Nutr Metab (Lond). 2011;8:65. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 120] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 20. | Herman MA, Birnbaum MJ. Molecular aspects of fructose metabolism and metabolic disease. Cell Metab. 2021;33:2329-2354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 167] [Article Influence: 41.8] [Reference Citation Analysis (0)] |
| 21. | Badin PM, Vila IK, Louche K, Mairal A, Marques MA, Bourlier V, Tavernier G, Langin D, Moro C. High-fat diet-mediated lipotoxicity and insulin resistance is related to impaired lipase expression in mouse skeletal muscle. Endocrinology. 2013;154:1444-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 22. | Softic S, Meyer JG, Wang GX, Gupta MK, Batista TM, Lauritzen HPMM, Fujisaka S, Serra D, Herrero L, Willoughby J, Fitzgerald K, Ilkayeva O, Newgard CB, Gibson BW, Schilling B, Cohen DE, Kahn CR. Dietary Sugars Alter Hepatic Fatty Acid Oxidation via Transcriptional and Post-translational Modifications of Mitochondrial Proteins. Cell Metab. 2019;30:735-753.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 155] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 23. | García-Berumen CI, Ortiz-Avila O, Vargas-Vargas MA, Del Rosario-Tamayo BA, Guajardo-López C, Saavedra-Molina A, Rodríguez-Orozco AR, Cortés-Rojo C. The severity of rat liver injury by fructose and high fat depends on the degree of respiratory dysfunction and oxidative stress induced in mitochondria. Lipids Health Dis. 2019;18:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 24. | Wali JA, Jarzebska N, Raubenheimer D, Simpson SJ, Rodionov RN, O'Sullivan JF. Cardio-Metabolic Effects of High-Fat Diets and Their Underlying Mechanisms-A Narrative Review. Nutrients. 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 25. | World Health Organization. Consideration of the evidence on childhood obesity for the Commission on Ending Childhood Obesity: report of the ad hoc working group on science and evidence for ending childhood obesity. 2016. [cited 22 April 2024]. Available from: https://cesni-biblioteca.org/wp-content/uploads/2020/04/9789241565332_eng_optimize.pdf. |
| 26. | Lee L, Sanders RA. Metabolic syndrome. Pediatr Rev. 2012;33:459-66; quiz 467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Bitew ZW, Alemu A, Ayele EG, Tenaw Z, Alebel A, Worku T. Metabolic syndrome among children and adolescents in low and middle income countries: a systematic review and meta-analysis. Diabetol Metab Syndr. 2020;12:93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 28. | Molnár D. The prevalence of the metabolic syndrome and type 2 diabetes mellitus in children and adolescents. Int J Obes Relat Metab Disord. 2004;28 Suppl 3:S70-S74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 73] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 29. | Noubiap JJ, Nansseu JR, Lontchi-Yimagou E, Nkeck JR, Nyaga UF, Ngouo AT, Tounouga DN, Tianyi FL, Foka AJ, Ndoadoumgue AL, Bigna JJ. Global, regional, and country estimates of metabolic syndrome burden in children and adolescents in 2020: a systematic review and modelling analysis. Lancet Child Adolesc Health. 2022;6:158-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 150] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 30. | Fahed G, Aoun L, Bou Zerdan M, Allam S, Bouferraa Y, Assi HI. Metabolic Syndrome: Updates on Pathophysiology and Management in 2021. Int J Mol Sci. 2022;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 689] [Article Influence: 229.7] [Reference Citation Analysis (0)] |
| 31. | Lipińska A, Koczaj-Bremer M, Jankowski K, Kaźmierczak A, Ciurzyński M, Ou-Pokrzewińska A, Mikocka E, Lewandowski Z, Demkow U, Pruszczyk P. Does family history of metabolic syndrome affect the metabolic profile phenotype in young healthy individuals? Diabetol Metab Syndr. 2014;6:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 32. | Lorzadeh E, Sangsefidi ZS, Mirzaei M, Hosseinzadeh M. Dietary Habits and their Association with Metabolic Syndrome in a sample of Iranian adults: A population-based study. Food Sci Nutr. 2020;8:6217-6225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 33. | Gepstein V, Weiss R. Obesity as the Main Risk Factor for Metabolic Syndrome in Children. Front Endocrinol (Lausanne). 2019;10:568. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 34. | Reisinger C, Nkeh-Chungag BN, Fredriksen PM, Goswami N. The prevalence of pediatric metabolic syndrome-a critical look on the discrepancies between definitions and its clinical importance. Int J Obes (Lond). 2021;45:12-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 35. | Abbasalizad Farhangi M, Mohammadi Tofigh A, Jahangiri L, Nikniaz Z, Nikniaz L. Sugar-sweetened beverages intake and the risk of obesity in children: An updated systematic review and dose-response meta-analysis. Pediatr Obes. 2022;17:e12914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 36. | Akoto S, Tandoh MA, Nsiah K, Asamoah-Boakye O, Annaful VT. Lifestyle habits, macronutrient intake, and obesity prevalence among adolescents in rural-periurban community senior high schools in the Ho municipality of Ghana. Front Nutr. 2022;9:955898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Stoianov A, Adeli K. Central and Metabolic Effects of High Fructose Consumption: Evidence from Animal and Human Studies. Nutr Food Sci Res. 2014;1:3-9. |
| 38. | Jung S, Bae H, Song WS, Jang C. Dietary Fructose and Fructose-Induced Pathologies. Annu Rev Nutr. 2022;42:45-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 39. | Ramírez-López G, Flores-Aldana M, Salmerón J. Associations between dietary patterns and metabolic syndrome in adolescents. Salud Publica Mex. 2019;61:619-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 40. | Steinberger J, Daniels SR, Eckel RH, Hayman L, Lustig RH, McCrindle B, Mietus-Snyder ML; American Heart Association Atherosclerosis, Hypertension, and Obesity in the Young Committee of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular Nursing; and Council on Nutrition, Physical Activity, and Metabolism. Progress and challenges in metabolic syndrome in children and adolescents: a scientific statement from the American Heart Association Atherosclerosis, Hypertension, and Obesity in the Young Committee of the Council on Cardiovascular Disease in the Young; Council on Cardiovascular Nursing; and Council on Nutrition, Physical Activity, and Metabolism. Circulation. 2009;119:628-647. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 504] [Cited by in RCA: 490] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 41. | Lee J, Kim JH. Endocrine comorbidities of pediatric obesity. Clin Exp Pediatr. 2021;64:619-627. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 42. | Abbasi-Ghahramanloo A, Moshiri E, Afrashteh S, Gholami A, Safiri S, Mohammadbeigi A, Ansari H. Comorbidity of metabolic syndrome components in a population-based screening program: A latent class analysis. Med J Islam Repub Iran. 2020;34:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 43. | Lee MK, Lee JH, Sohn SY, Ahn J, Hong OK, Kim MK, Baek KH, Song KH, Han K, Kwon HS. Cumulative exposure to metabolic syndrome in a national population-based cohort of young adults and sex-specific risk for type 2 diabetes. Diabetol Metab Syndr. 2023;15:78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 44. | Al-Hamad D, Raman V. Metabolic syndrome in children and adolescents. Transl Pediatr. 2017;6:397-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 158] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 45. | Prasad GV. Metabolic syndrome and chronic kidney disease: Current status and future directions. World J Nephrol. 2014;3:210-219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 93] [Cited by in RCA: 109] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 46. | Tune JD, Goodwill AG, Sassoon DJ, Mather KJ. Cardiovascular consequences of metabolic syndrome. Transl Res. 2017;183:57-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 356] [Article Influence: 44.5] [Reference Citation Analysis (0)] |
| 47. | Li X, Zhai Y, Zhao J, He H, Li Y, Liu Y, Feng A, Li L, Huang T, Xu A, Lyu J. Impact of Metabolic Syndrome and It's Components on Prognosis in Patients With Cardiovascular Diseases: A Meta-Analysis. Front Cardiovasc Med. 2021;8:704145. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 48. | MOORHOUSE JA, KARK RM. Fructose and diabetes. Am J Med. 1957;23:46-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 49. | Mock K, Lateef S, Benedito VA, Tou JC. High-fructose corn syrup-55 consumption alters hepatic lipid metabolism and promotes triglyceride accumulation. J Nutr Biochem. 2017;39:32-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 50. | Douard V, Ferraris RP. Regulation of the fructose transporter GLUT5 in health and disease. Am J Physiol Endocrinol Metab. 2008;295:E227-E237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 328] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 51. | DeBosch BJ, Chi M, Moley KH. Glucose transporter 8 (GLUT8) regulates enterocyte fructose transport and global mammalian fructose utilization. Endocrinology. 2012;153:4181-4191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 52. | Ferraris RP, Choe JY, Patel CR. Intestinal Absorption of Fructose. Annu Rev Nutr. 2018;38:41-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 120] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 53. | Zhang DM, Jiao RQ, Kong LD. High Dietary Fructose: Direct or Indirect Dangerous Factors Disturbing Tissue and Organ Functions. Nutrients. 2017;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 143] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 54. | Diggle CP, Shires M, Leitch D, Brooke D, Carr IM, Markham AF, Hayward BE, Asipu A, Bonthron DT. Ketohexokinase: expression and localization of the principal fructose-metabolizing enzyme. J Histochem Cytochem. 2009;57:763-774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 146] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 55. | Topping DL, Mayes PA. Comparative effects of fructose and glucose on the lipid and carbohydrate metabolism of perfused rat liver. Br J Nutr. 1976;36:113-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 56. | Sul HS, Wise LS, Brown ML, Rubin CS. Cloning of cDNA sequences for murine ATP-citrate lyase. Construction of recombinant plasmids using an immunopurified mRNA template and evidence for the nutritional regulation of ATP-citrate lyase mRNA content in mouse liver. J Biol Chem. 1984;259:1201-1205. [PubMed] |
| 57. | Yoo TW, Sung KC, Shin HS, Kim BJ, Kim BS, Kang JH, Lee MH, Park JR, Kim H, Rhee EJ, Lee WY, Kim SW, Ryu SH, Keum DG. Relationship between serum uric acid concentration and insulin resistance and metabolic syndrome. Circ J. 2005;69:928-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 264] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 58. | Kim A, Nguyen J, Babaei M, Kim A, Geller DH, Vidmar AP. A Narrative Review: Phentermine and Topiramate for the Treatment of Pediatric Obesity. Adolesc Health Med Ther. 2023;14:125-140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 59. | Zhu Y, Hu Y, Huang T, Zhang Y, Li Z, Luo C, Luo Y, Yuan H, Hisatome I, Yamamoto T, Cheng J. High uric acid directly inhibits insulin signalling and induces insulin resistance. Biochem Biophys Res Commun. 2014;447:707-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 213] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 60. | Huang JP, Cheng ML, Hung CY, Wang CH, Hsieh PS, Shiao MS, Chen JK, Li DE, Hung LM. Docosapentaenoic acid and docosahexaenoic acid are positively associated with insulin sensitivity in rats fed high-fat and high-fructose diets. J Diabetes. 2017;9:936-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 61. | Dekker MJ, Baker C, Naples M, Samsoondar J, Zhang R, Qiu W, Sacco J, Adeli K. Inhibition of sphingolipid synthesis improves dyslipidemia in the diet-induced hamster model of insulin resistance: evidence for the role of sphingosine and sphinganine in hepatic VLDL-apoB100 overproduction. Atherosclerosis. 2013;228:98-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 62. | Crescenzo R, Bianco F, Coppola P, Mazzoli A, Cigliano L, Liverini G, Iossa S. Increased skeletal muscle mitochondrial efficiency in rats with fructose-induced alteration in glucose tolerance. Br J Nutr. 2013;110:1996-2003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 63. | Porto ML, Lírio LM, Dias AT, Batista AT, Campagnaro BP, Mill JG, Meyrelles SS, Baldo MP. Increased oxidative stress and apoptosis in peripheral blood mononuclear cells of fructose-fed rats. Toxicol In Vitro. 2015;29:1977-1981. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 64. | Lowe ME. The triglyceride lipases of the pancreas. J Lipid Res. 2002;43:2007-2016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 207] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 65. | Havel RJ. Postprandial hyperlipidemia and remnant lipoproteins. Curr Opin Lipidol. 1994;5:102-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 140] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 66. | Cohen DE, Fisher EA. Lipoprotein metabolism, dyslipidemia, and nonalcoholic fatty liver disease. Semin Liver Dis. 2013;33:380-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 126] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 67. | Tomkin GH, Owens D. The chylomicron: relationship to atherosclerosis. Int J Vasc Med. 2012;2012:784536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 68. | Mashek DG. Hepatic fatty acid trafficking: multiple forks in the road. Adv Nutr. 2013;4:697-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 112] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 69. | Kawano Y, Cohen DE. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J Gastroenterol. 2013;48:434-441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 545] [Cited by in RCA: 700] [Article Influence: 58.3] [Reference Citation Analysis (0)] |
| 70. | Pessayre D, Berson A, Fromenty B, Mansouri A. Mitochondria in steatohepatitis. Semin Liver Dis. 2001;21:57-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 258] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 71. | Cortés-Rojo C, Vargas-Vargas MA, Olmos-Orizaba BE, Rodríguez-Orozco AR, Calderón-Cortés E. Interplay between NADH oxidation by complex I, glutathione redox state and sirtuin-3, and its role in the development of insulin resistance. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 72. | Dalvi PS, Chalmers JA, Luo V, Han DY, Wellhauser L, Liu Y, Tran DQ, Castel J, Luquet S, Wheeler MB, Belsham DD. High fat induces acute and chronic inflammation in the hypothalamus: effect of high-fat diet, palmitate and TNF-α on appetite-regulating NPY neurons. Int J Obes (Lond). 2017;41:149-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 163] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 73. | García-Berumen CI, Vargas-Vargas MA, Ortiz-Avila O, Piña-Zentella RM, Ramos-Gómez M, Figueroa-García MDC, Mejía-Zepeda R, Rodríguez-Orozco AR, Saavedra-Molina A, Cortés-Rojo C. Avocado oil alleviates non-alcoholic fatty liver disease by improving mitochondrial function, oxidative stress and inflammation in rats fed a high fat-High fructose diet. Front Pharmacol. 2022;13:1089130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 74. | Crescenzo R, Bianco F, Coppola P, Mazzoli A, Tussellino M, Carotenuto R, Liverini G, Iossa S. Fructose supplementation worsens the deleterious effects of short-term high-fat feeding on hepatic steatosis and lipid metabolism in adult rats. Exp Physiol. 2014;99:1203-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 75. | Lloyd LJ, Langley-Evans SC, McMullen S. Childhood obesity and risk of the adult metabolic syndrome: a systematic review. Int J Obes (Lond). 2012;36:1-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 250] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 76. | Zhou LY, Deng MQ, Zhang Q, Xiao XH. Early-life nutrition and metabolic disorders in later life: a new perspective on energy metabolism. Chin Med J (Engl). 2020;133:1961-1970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 77. | He H, Pan L, Du J, Liu F, Jin Y, Ma J, Wang L, Jia P, Hu Z, Shan G. Prevalence of, and biochemical and anthropometric risk factors for, dyslipidemia in children and adolescents aged 7 to 18 years in China: A cross-sectional study. Am J Hum Biol. 2019;31:e23286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 78. | World Health Organization. Saturated fatty acid and trans-fatty acid intake for adults and children: WHO guideline. 17 July 2023. [cited 22 April 2024]. Available from: https://www.who.int/publications/i/item/9789240073630. |
| 79. | Mensink RP; World Health Organization. Effects of saturated fatty acids on serum lipids and lipoproteins: a systematic review and regression analysis. 2016. [cited 22 April 2024]. Available from: https://iris.who.int/handle/10665/246104. |
| 80. | Bowman SA, Clemens JC. Saturated Fat and Food Intakes of Adults: What We Eat in America, NHANES 2017-2018. In: FSRG Dietary Data Briefs [Internet]. Beltsville (MD): United States Department of Agriculture (USDA); 2010–. [PubMed] |
| 81. | Maffeis C, Cendon M, Tomasselli F, Tommasi M, Bresadola I, Fornari E, Morandi A, Olivieri F. Lipid and saturated fatty acids intake and cardiovascular risk factors of obese children and adolescents. Eur J Clin Nutr. 2021;75:1109-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 82. | Sadeghian M, Vafadar M, Torabi A, Torabi S, Fazel M. Prevalence of fatty liver and its related factors in children. J Family Med Prim Care. 2022;11:5604-5608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 83. | Nishida C, Uauy R. WHO Scientific Update on health consequences of trans fatty acids: introduction. Eur J Clin Nutr. 2009;63 Suppl 2:S1-S4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 64] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 84. | Naude CE, Visser ME, Nguyen KA, Durao S, Schoonees A. Effects of total fat intake on bodyweight in children. Cochrane Database Syst Rev. 2018;2:CD012960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 85. | Erkin-Cakmak A, Bains Y, Caccavello R, Noworolski SM, Schwarz JM, Mulligan K, Lustig RH, Gugliucci A. Isocaloric Fructose Restriction Reduces Serum d-Lactate Concentration in Children With Obesity and Metabolic Syndrome. J Clin Endocrinol Metab. 2019;104:3003-3011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 86. | Huerta-Ávila EE, Ramírez-Silva I, Torres-Sánchez LE, Díaz-Benítez CE, Orbe-Orihuela YC, Lagunas-Martínez A, Galván-Portillo M, Flores M, Cruz M, Burguete-García AI. High Relative Abundance of Lactobacillus reuteri and Fructose Intake are Associated with Adiposity and Cardiometabolic Risk Factors in Children from Mexico City. Nutrients. 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 87. | Liu J, Li Y, Wang X, Gao D, Chen L, Chen M, Ma T, Ma Q, Ma Y, Zhang Y, Jiang J, Zou Z, Dong Y, Ma J. Association between Fruit Consumption and Lipid Profile among Children and Adolescents: A National Cross-Sectional Study in China. Nutrients. 2021;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 88. | Hardwick J, Sidnell A. Infant nutrition – diet between 6 and 24 months, implications for paediatric growth, overweight and obesity. Nutrition Bulletin. 2014;39:354-363. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 89. | World Health Organization. Guideline: sugars intake for adults and children. 4 March 2015. [cited 22 April 2024]. Available from: https://www.who.int/news/item/04-03-2015-who-calls-on-countries-to-reduce-sugars-intake-among-adults-and-children. |
| 90. | Lustig RH, Mulligan K, Noworolski SM, Tai VW, Wen MJ, Erkin-Cakmak A, Gugliucci A, Schwarz JM. Isocaloric fructose restriction and metabolic improvement in children with obesity and metabolic syndrome. Obesity (Silver Spring). 2016;24:453-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 131] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 91. | Sánchez-Pimienta TG, Batis C, Lutter CK, Rivera JA. Sugar-Sweetened Beverages Are the Main Sources of Added Sugar Intake in the Mexican Population. J Nutr. 2016;146:1888S-1896S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 130] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 92. | López-Olmedo N, Carriquiry AL, Rodríguez-Ramírez S, Ramírez-Silva I, Espinosa-Montero J, Hernández-Barrera L, Campirano F, Martínez-Tapia B, Rivera JA. Usual Intake of Added Sugars and Saturated Fats Is High while Dietary Fiber Is Low in the Mexican Population. J Nutr. 2016;146:1856S-1865S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 95] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 93. | Dereń K, Weghuber D, Caroli M, Koletzko B, Thivel D, Frelut ML, Socha P, Grossman Z, Hadjipanayis A, Wyszyńska J, Mazur A. Consumption of Sugar-Sweetened Beverages in Paediatric Age: A Position Paper of the European Academy of Paediatrics and the European Childhood Obesity Group. Ann Nutr Metab. 2019;74:296-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 94. | Russo E, Leoncini G, Esposito P, Garibotto G, Pontremoli R, Viazzi F. Fructose and Uric Acid: Major Mediators of Cardiovascular Disease Risk Starting at Pediatric Age. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 95. | Giussani M, Lieti G, Orlando A, Parati G, Genovesi S. Fructose Intake, Hypertension and Cardiometabolic Risk Factors in Children and Adolescents: From Pathophysiology to Clinical Aspects. A Narrative Review. Front Med (Lausanne). 2022;9:792949. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 96. | Lin WT, Chan TF, Huang HL, Lee CY, Tsai S, Wu PW, Yang YC, Wang TN, Lee CH. Fructose-Rich Beverage Intake and Central Adiposity, Uric Acid, and Pediatric Insulin Resistance. J Pediatr. 2016;171:90-6.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 97. | Costa CS, Rauber F, Leffa PS, Sangalli CN, Campagnolo PDB, Vitolo MR. Ultra-processed food consumption and its effects on anthropometric and glucose profile: A longitudinal study during childhood. Nutr Metab Cardiovasc Dis. 2019;29:177-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 150] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 98. | Batalha MA, França AKTDC, Conceição SIOD, Santos AMD, Silva FS, Padilha LL, Silva AAMD. Processed and ultra-processed food consumption among children aged 13 to 35 months and associated factors. Cad Saude Publica. 2017;33:e00152016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 99. | Rauber F, Campagnolo PD, Hoffman DJ, Vitolo MR. Consumption of ultra-processed food products and its effects on children's lipid profiles: a longitudinal study. Nutr Metab Cardiovasc Dis. 2015;25:116-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 290] [Cited by in RCA: 318] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 100. | Tavares LF, Fonseca SC, Garcia Rosa ML, Yokoo EM. Relationship between ultra-processed foods and metabolic syndrome in adolescents from a Brazilian Family Doctor Program. Public Health Nutr. 2012;15:82-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 224] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 101. | Czerwonogrodzka-Senczyna A, Rumińska M, Majcher A, Credo D, Jeznach-Steinhagen A, Pyrżak B. Fructose Consumption and Lipid Metabolism in Obese Children and Adolescents. Adv Exp Med Biol. 2019;1153:91-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 102. | Perrar I, Buyken AE, Penczynski KJ, Remer T, Kuhnle GG, Herder C, Roden M, Della Corte K, Nöthlings U, Alexy U. Relevance of fructose intake in adolescence for fatty liver indices in young adulthood. Eur J Nutr. 2021;60:3029-3041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 103. | Larqué E, Gil-Campos M, Ramírez-Tortosa MC, Linde J, Cañete R, Gil A. Postprandial response of trans fatty acids in prepubertal obese children. Int J Obes (Lond). 2006;30:1488-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 104. | Hendrie GA, Golley RK. Changing from regular-fat to low-fat dairy foods reduces saturated fat intake but not energy intake in 4-13-y-old children. Am J Clin Nutr. 2011;93:1117-1127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 105. | Ivancovsky-Wajcman D, Fliss-Isakov N, Webb M, Bentov I, Shibolet O, Kariv R, Zelber-Sagi S. Ultra-processed food is associated with features of metabolic syndrome and non-alcoholic fatty liver disease. Liver Int. 2021;41:2635-2645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 106. | Magge SN, Goodman E, Armstrong SC; COMMITTEE ON NUTRITION; SECTION ON ENDOCRINOLOGY; SECTION ON OBESITY. The Metabolic Syndrome in Children and Adolescents: Shifting the Focus to Cardiometabolic Risk Factor Clustering. Pediatrics. 2017;140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 259] [Article Influence: 32.4] [Reference Citation Analysis (0)] |
| 107. | The Lancet Diabetes Endocrinology. Childhood obesity: a growing pandemic. Lancet Diabetes Endocrinol. 2022;10:1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 108. | Gelman R, Whelan J, Spiteri S, Duric D, Oakhill W, Cassar S, Love P. Adoption, implementation, and sustainability of early childhood feeding, nutrition and active play interventions in real-world settings: a systematic review. Int J Behav Nutr Phys Act. 2023;20:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Reference Citation Analysis (0)] |
| 109. | Kumar S, Kelly AS. Review of Childhood Obesity: From Epidemiology, Etiology, and Comorbidities to Clinical Assessment and Treatment. Mayo Clin Proc. 2017;92:251-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 678] [Cited by in RCA: 886] [Article Influence: 110.8] [Reference Citation Analysis (0)] |
| 110. | Gortmaker SL, Swinburn BA, Levy D, Carter R, Mabry PL, Finegood DT, Huang T, Marsh T, Moodie ML. Changing the future of obesity: science, policy, and action. Lancet. 2011;378:838-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 678] [Cited by in RCA: 577] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 111. | Kirk S, Brehm B, Saelens BE, Woo JG, Kissel E, D'Alessio D, Bolling C, Daniels SR. Role of carbohydrate modification in weight management among obese children: a randomized clinical trial. J Pediatr. 2012;161:320-7.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 112. | Psaltopoulou T, Tzanninis S, Ntanasis-Stathopoulos I, Panotopoulos G, Kostopoulou M, Tzanninis IG, Tsagianni A, Sergentanis TN. Prevention and treatment of childhood and adolescent obesity: a systematic review of meta-analyses. World J Pediatr. 2019;15:350-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 113. | Wong PD, Anderson LN, Dai DDW, Parkin PC, Maguire JL, Birken CS; TARGet Kids! Collaboration. The Association of Breastfeeding Duration and Early Childhood Cardiometabolic Risk. J Pediatr. 2018;192:80-85.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 114. | Asghari G, Yuzbashian E, Mirmiran P, Hooshmand F, Najafi R, Azizi F. Dietary Approaches to Stop Hypertension (DASH) Dietary Pattern Is Associated with Reduced Incidence of Metabolic Syndrome in Children and Adolescents. J Pediatr. 2016;174:178-184.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 115. | Kirk S, Woo JG, Brehm B, Daniels SR, Saelens BE. Changes in Eating Behaviors of Children with Obesity in Response to Carbohydrate-Modified and Portion-Controlled Diets. Child Obes. 2017;13:377-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 116. | Epstein LH, Paluch RA, Beecher MD, Roemmich JN. Increasing healthy eating vs. reducing high energy-dense foods to treat pediatric obesity. Obesity (Silver Spring). 2008;16:318-326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 145] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 117. | Epstein LH. A Brief History and Future of the Traffic Light Diet. Curr Dev Nutr. 2022;6:nzac120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Reference Citation Analysis (0)] |
| 118. | Hammond D, Acton RB, Rynard VL, White CM, Vanderlee L, Bhawra J, Reyes M, Jáuregui A, Adams J, Roberto CA, Sacks G, Thrasher JF. Awareness, use and understanding of nutrition labels among children and youth from six countries: findings from the 2019 - 2020 International Food Policy Study. Int J Behav Nutr Phys Act. 2023;20:55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 119. | Ramon-Krauel M, Salsberg SL, Ebbeling CB, Voss SD, Mulkern RV, Apura MM, Cooke EA, Sarao K, Jonas MM, Ludwig DS. A low-glycemic-load versus low-fat diet in the treatment of fatty liver in obese children. Child Obes. 2013;9:252-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 120. | Mager DR, Iñiguez IR, Gilmour S, Yap J. The effect of a low fructose and low glycemic index/load (FRAGILE) dietary intervention on indices of liver function, cardiometabolic risk factors, and body composition in children and adolescents with nonalcoholic fatty liver disease (NAFLD). JPEN J Parenter Enteral Nutr. 2015;39:73-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 121. | Saneei P, Hashemipour M, Kelishadi R, Rajaei S, Esmaillzadeh A. Effects of recommendations to follow the Dietary Approaches to Stop Hypertension (DASH) diet v. usual dietary advice on childhood metabolic syndrome: a randomised cross-over clinical trial. Br J Nutr. 2013;110:2250-2259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 122. | Velázquez-López L, Santiago-Díaz G, Nava-Hernández J, Muñoz-Torres AV, Medina-Bravo P, Torres-Tamayo M. Mediterranean-style diet reduces metabolic syndrome components in obese children and adolescents with obesity. BMC Pediatr. 2014;14:175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 125] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 123. | Mijangos-Trejo A, Nuño-Lambarri N, Barbero-Becerra V, Uribe-Esquivel M, Vidal-Cevallos P, Chávez-Tapia N. Prebiotics and Probiotics: Therapeutic Tools for Nonalcoholic Fatty Liver Disease. Int J Mol Sci. 2023;24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 124. | Hu H, Lin A, Kong M, Yao X, Yin M, Xia H, Ma J, Liu H. Intestinal microbiome and NAFLD: molecular insights and therapeutic perspectives. J Gastroenterol. 2020;55:142-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 125] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 125. | Milosevic I, Vujovic A, Barac A, Djelic M, Korac M, Radovanovic Spurnic A, Gmizic I, Stevanovic O, Djordjevic V, Lekic N, Russo E, Amedei A. Gut-Liver Axis, Gut Microbiota, and Its Modulation in the Management of Liver Diseases: A Review of the Literature. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 393] [Cited by in RCA: 355] [Article Influence: 59.2] [Reference Citation Analysis (0)] |
| 126. | Abuqwider J, Di Porzio A, Barrella V, Gatto C, Sequino G, De Filippis F, Crescenzo R, Spagnuolo MS, Cigliano L, Mauriello G, Iossa S, Mazzoli A. Limosilactobacillus reuteri DSM 17938 reverses gut metabolic dysfunction induced by Western diet in adult rats. Front Nutr. 2023;10:1236417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 127. | Almonacid-Fierro A, González-Almonacid J. The pandemic of childhood obesity: Challenges and possibilities from physical activity. Health Promot Perspect. 2022;12:229-230. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 128. | Haverly K, Davison KK. Personal fulfillment motivates adolescents to be physically active. Arch Pediatr Adolesc Med. 2005;159:1115-1120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 129. | Calcaterra V, Vandoni M, Rossi V, Berardo C, Grazi R, Cordaro E, Tranfaglia V, Carnevale Pellino V, Cereda C, Zuccotti G. Use of Physical Activity and Exercise to Reduce Inflammation in Children and Adolescents with Obesity. Int J Environ Res Public Health. 2022;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 39] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 130. | Stabelini Neto A, de Campos W, Dos Santos GC, Mazzardo Junior O. Metabolic syndrome risk score and time expended in moderate to vigorous physical activity in adolescents. BMC Pediatr. 2014;14:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 131. | Herouvi D, Paltoglou G, Soldatou A, Kalpia C, Karanasios S, Karavanaki K. Lifestyle and Pharmacological Interventions and Treatment Indications for the Management of Obesity in Children and Adolescents. Children (Basel). 2023;10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 132. | Weihe P, Weihrauch-Blüher S. Metabolic Syndrome in Children and Adolescents: Diagnostic Criteria, Therapeutic Options and Perspectives. Curr Obes Rep. 2019;8:472-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 142] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 133. | Carbajo-Pescador S, Porras D, García-Mediavilla MV, Martínez-Flórez S, Juarez-Fernández M, Cuevas MJ, Mauriz JL, González-Gallego J, Nistal E, Sánchez-Campos S. Beneficial effects of exercise on gut microbiota functionality and barrier integrity, and gut-liver crosstalk in an in vivo model of early obesity and non-alcoholic fatty liver disease. Dis Model Mech. 2019;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 103] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 134. | US Food and Drug Administration. News & Events for Human Drugs. [cited 16 December 2023]. Available from: https://www.fda.gov/drugs/news-events-human-drugs. |
| 135. | Guerciolini R. Mode of action of orlistat. Int J Obes Relat Metab Disord. 1997;21 Suppl 3:S12-S23. [PubMed] |
| 136. | Cannon CP, Kumar A. Treatment of overweight and obesity: lifestyle, pharmacologic, and surgical options. Clin Cornerstone. 2009;9:55-68; discussion 69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 137. | Kelly AS, Bensignor MO, Hsia DS, Shoemaker AH, Shih W, Peterson C, Varghese ST. Phentermine/Topiramate for the Treatment of Adolescent Obesity. NEJM Evid. 2022;1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 138. | Andreozzi F, Raciti GA, Nigro C, Mannino GC, Procopio T, Davalli AM, Beguinot F, Sesti G, Miele C, Folli F. The GLP-1 receptor agonists exenatide and liraglutide activate Glucose transport by an AMPK-dependent mechanism. J Transl Med. 2016;14:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 71] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 139. | Hjerpsted JB, Flint A, Brooks A, Axelsen MB, Kvist T, Blundell J. Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity. Diabetes Obes Metab. 2018;20:610-619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 155] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 140. | Kelly AS, Auerbach P, Barrientos-Perez M, Gies I, Hale PM, Marcus C, Mastrandrea LD, Prabhu N, Arslanian S; NN8022-4180 Trial Investigators. A Randomized, Controlled Trial of Liraglutide for Adolescents with Obesity. N Engl J Med. 2020;382:2117-2128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 382] [Article Influence: 76.4] [Reference Citation Analysis (1)] |
| 141. | Christou GA, Katsiki N, Kiortsis DN. The Current Role of Liraglutide in the Pharmacotherapy of Obesity. Curr Vasc Pharmacol. 2016;14:201-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 142. | Clément K, van den Akker E, Argente J, Bahm A, Chung WK, Connors H, De Waele K, Farooqi IS, Gonneau-Lejeune J, Gordon G, Kohlsdorf K, Poitou C, Puder L, Swain J, Stewart M, Yuan G, Wabitsch M, Kühnen P; Setmelanotide POMC and LEPR Phase 3 Trial Investigators. Efficacy and safety of setmelanotide, an MC4R agonist, in individuals with severe obesity due to LEPR or POMC deficiency: single-arm, open-label, multicentre, phase 3 trials. Lancet Diabetes Endocrinol. 2020;8:960-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 265] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 143. | Nyakudya TT, Mukwevho E, Nkomozepi P, Erlwanger KH. Neonatal intake of oleanolic acid attenuates the subsequent development of high fructose diet-induced non-alcoholic fatty liver disease in rats. J Dev Orig Health Dis. 2018;9:500-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 144. | Molepo M, Ayeleso A, Nyakudya T, Erlwanger K, Mukwevho E. A Study on Neonatal Intake of Oleanolic Acid and Metformin in Rats (Rattus norvegicus) with Metabolic Dysfunction: Implications on Lipid Metabolism and Glucose Transport. Molecules. 2018;23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 145. | Zhan X, He M, Pei J, Fan W, Mwangi CN, Zhang P, Chai X, Jiang M. Natural Phenylethanoid Supplementation Alleviates Metabolic Syndrome in Female Mice Induced by High-Fructose Diet. Front Pharmacol. 2022;13:850777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 146. | Claro-Cala CM, Jiménez-Altayó F, Zagmutt S, Rodriguez-Rodriguez R. Molecular Mechanisms Underlying the Effects of Olive Oil Triterpenic Acids in Obesity and Related Diseases. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 147. | Guo XX, Zeng Z, Qian YZ, Qiu J, Wang K, Wang Y, Ji BP, Zhou F. Wheat Flour, Enriched with γ-Oryzanol, Phytosterol, and Ferulic Acid, Alleviates Lipid and Glucose Metabolism in High-Fat-Fructose-Fed Rats. Nutrients. 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 148. | Porras D, Nistal E, Martínez-Flórez S, Pisonero-Vaquero S, Olcoz JL, Jover R, González-Gallego J, García-Mediavilla MV, Sánchez-Campos S. Protective effect of quercetin on high-fat diet-induced non-alcoholic fatty liver disease in mice is mediated by modulating intestinal microbiota imbalance and related gut-liver axis activation. Free Radic Biol Med. 2017;102:188-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 374] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 149. | Mukonowenzou NC, Dangarembizi R, Chivandi E, Nkomozepi P, Erlwanger KH. Administration of ursolic acid to new-born pups prevents dietary fructose-induced non-alcoholic fatty liver disease in Sprague Dawley rats. J Dev Orig Health Dis. 2021;12:101-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 150. | Jegatheesan P, Beutheu S, Ventura G, Sarfati G, Nubret E, Kapel N, Waligora-Dupriet AJ, Bergheim I, Cynober L, De-Bandt JP. Effect of specific amino acids on hepatic lipid metabolism in fructose-induced non-alcoholic fatty liver disease. Clin Nutr. 2016;35:175-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 76] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 151. | Nouri Z, Hajialyani M, Izadi Z, Bahramsoltani R, Farzaei MH, Abdollahi M. Nanophytomedicines for the Prevention of Metabolic Syndrome: A Pharmacological and Biopharmaceutical Review. Front Bioeng Biotechnol. 2020;8:425. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 152. | Nguyen TTB, Jin YY, Chung HJ, Hong ST. Pharmabiotics as an Emerging Medication for Metabolic Syndrome and Its Related Diseases. Molecules. 2017;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 153. | Zang L, Baharlooeian M, Terasawa M, Shimada Y, Nishimura N. Beneficial effects of seaweed-derived components on metabolic syndrome via gut microbiota modulation. Front Nutr. 2023;10:1173225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 154. | Chou CL, Li CH, Fang TC. Benefits of Valsartan and Amlodipine in Lipolysis through PU.1 Inhibition in Fructose-Induced Adiposity. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Reference Citation Analysis (0)] |
| 155. | Gao Z, Song GY, Ren LP, Ma HJ, Ma BQ, Chen SC. β-catenin mediates the effect of GLP-1 receptor agonist on ameliorating hepatic steatosis induced by high fructose diet. Eur J Histochem. 2020;64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 156. | Chen L, Duan Y, Wei H, Ning H, Bi C, Zhao Y, Qin Y, Li Y. Acetyl-CoA carboxylase (ACC) as a therapeutic target for metabolic syndrome and recent developments in ACC1/2 inhibitors. Expert Opin Investig Drugs. 2019;28:917-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 157. | Ter Horst KW, Serlie MJ. Fructose Consumption, Lipogenesis, and Non-Alcoholic Fatty Liver Disease. Nutrients. 2017;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 215] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 158. | Cozma AI, Sievenpiper JL, de Souza RJ, Chiavaroli L, Ha V, Wang DD, Mirrahimi A, Yu ME, Carleton AJ, Di Buono M, Jenkins AL, Leiter LA, Wolever TM, Beyene J, Kendall CW, Jenkins DJ. Effect of fructose on glycemic control in diabetes: a systematic review and meta-analysis of controlled feeding trials. Diabetes Care. 2012;35:1611-1620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 137] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 159. | Chiu S, Sievenpiper JL, de Souza RJ, Cozma AI, Mirrahimi A, Carleton AJ, Ha V, Di Buono M, Jenkins AL, Leiter LA, Wolever TM, Don-Wauchope AC, Beyene J, Kendall CW, Jenkins DJ. Effect of fructose on markers of non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of controlled feeding trials. Eur J Clin Nutr. 2014;68:416-423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 216] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 160. | Johnston RD, Stephenson MC, Crossland H, Cordon SM, Palcidi E, Cox EF, Taylor MA, Aithal GP, Macdonald IA. No difference between high-fructose and high-glucose diets on liver triacylglycerol or biochemistry in healthy overweight men. Gastroenterology. 2013;145:1016-1025.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 141] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 161. | Simons N, Veeraiah P, Simons PIHG, Schaper NC, Kooi ME, Schrauwen-Hinderling VB, Feskens EJM, van der Ploeg EMCL, Van den Eynde MDG, Schalkwijk CG, Stehouwer CDA, Brouwers MCGJ. Effects of fructose restriction on liver steatosis (FRUITLESS); a double-blind randomized controlled trial. Am J Clin Nutr. 2021;113:391-400. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 162. | Geidl-Flueck B, Hochuli M, Németh Á, Eberl A, Derron N, Köfeler HC, Tappy L, Berneis K, Spinas GA, Gerber PA. Fructose- and sucrose- but not glucose-sweetened beverages promote hepatic de novo lipogenesis: A randomized controlled trial. J Hepatol. 2021;75:46-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 123] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 163. | Chiavaroli L, Cheung A, Ayoub-Charette S, Ahmed A, Lee D, Au-Yeung F, Qi X, Back S, McGlynn N, Ha V, Lai E, Khan TA, Blanco Mejia S, Zurbau A, Choo VL, de Souza RJ, Wolever TM, Leiter LA, Kendall CW, Jenkins DJ, Sievenpiper JL. Important food sources of fructose-containing sugars and adiposity: A systematic review and meta-analysis of controlled feeding trials. Am J Clin Nutr. 2023;117:741-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 164. | Huang Y, Chen Z, Chen B, Li J, Yuan X, Wang W, Dai T, Chen H, Wang Y, Wang R, Wang P, Guo J, Dong Q, Liu C, Wei Q, Cao D, Liu L. Dietary sugar consumption and health: umbrella review. BMJ. 2023;381:e071609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 104] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 165. | Mozaffarian D. Sugar, sugary drinks, and health: has the evidence achieved the sweet spot for policy action? Lancet Diabetes Endocrinol. 2023;11:448-451. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 166. | DiStefano JK, Shaibi GQ. The relationship between excessive dietary fructose consumption and paediatric fatty liver disease. Pediatr Obes. 2021;16:e12759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 167. | Distefano JK, Gerhard GS. Effects of dietary sugar restriction on hepatic fat in youth with obesity. Minerva Pediatr (Torino). 2023;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 168. | Porto A, Pan Z, Zhou W, Sokol RJ, Klaczkiewicz K, Sundaram SS. Macronutrient and Micronutrient Intake in Adolescents With Non-alcoholic Fatty Liver Disease: The Association With Disease Severity. J Pediatr Gastroenterol Nutr. 2022;75:666-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 169. | Guimber D, Debray D, Bocquet A, Briend A, Chouraqui JP, Darmaun D, Feillet F, Frelut ML, Hankard R, Lapillonne A, Peretti N, Rozé JC, Simeoni U, Turck D, Dupont C, Comité de Nutrition de la Société Française de Pédiatrie Cnsfp. The role of nutrition in non-alcoholic fatty liver disease treatment in obese children. Arch Pediatr. 2022;29:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |