Published online Mar 9, 2022. doi: 10.5409/wjcp.v11.i2.136
Peer-review started: May 26, 2021
First decision: July 30, 2021
Revised: August 3, 2021
Accepted: January 8, 2022
Article in press: January 8, 2022
Published online: March 9, 2022
Processing time: 286 Days and 19.2 Hours
In the last two decades two new paradigms changed our way of perceiving primary immunodeficiencies: An increasing number of immune defects are more associated with inflammatory or autoimmune features rather than with infections. Some primary immune defects are due to hyperactive pathways that can be targeted by specific inhibitors, providing innovative precision treatments that can change the natural history of diseases. In this article we review some of these “druggable” inborn errors of immunity and describe how they can be suspected and diagnosed in diverse pediatric and adult medicine specialties. Since the availability of precision treatments can dramatically impact the course of these diseases, preventing the development of organ damage, it is crucial to widen the awareness of these conditions and to provide practical hints for a prompt detection and cure.
Core Tip: High-throughput genetic testing have allowed to describe monogenic immune disorders, characterized by combinations of infective, inflammatory, autoimmune, lymphoproliferative, neoplastic features. The term “inborn errors of immunity” (IEIs) is increasingly proposed instead of “primary immunodeficiency” to include defects with a prevalently dysregulatory pathogenesis, resulting in autoimmunity, inflammation, lymphoproliferation, risk of malignancies. It is crucial to widen the awareness of these disorders, as they may mimic multifactorial disorders (rheumatology, gastroenterology, hematology, dermatology, allergology) and some of these are druggable. The awareness of druggable IEIs is the focus of this review, with the aim of favoring a prompter diagnosis and a better cure.
- Citation: Boz V, Zanchi C, Levantino L, Riccio G, Tommasini A. Druggable monogenic immune defects hidden in diverse medical specialties: Focus on overlap syndromes. World J Clin Pediatr 2022; 11(2): 136-150
- URL: https://www.wjgnet.com/2219-2808/full/v11/i2/136.htm
- DOI: https://dx.doi.org/10.5409/wjcp.v11.i2.136
Primary immunodeficiencies are a growing group of monogenic disorders related to dysregulated immune processes, which can result in autoinflammation, autoimmunity, lymphoproliferation and/or risk of malignancy in addition to the paradigmatic recurrent infections: In this sense, the term “inborn errors of immunity” (IEIs) has recently been proposed to underline the heterogeneous phenotype of immune deficiencies[1,2].
Improved diagnostics of monogenic immune disorders, together with the availability of medications acting on disease-related mechanisms recently led to the development of precision therapies which can improve or correct the phenotype of some IEIs[3-6]. The mutations involved in these disorders are usually associated with gain-of-function (GOF) of proteins (often kinases) or hyperactivity of pathways, which can be targeted by specific medications and thus sometimes referred to as “druggable”. For concision, we indicate IEIs with druggable pathogenic mechanisms as druggable IEI (D-IEIs).
Of note, since immunodeficiency may develop significant organ damage due to infection or autoimmunity, early detection of D-IEIs is crucial to benefit from appropriate treatments[7]. Although a deep clinical-laboratory evaluation can help an experienced immunologist to concentrate suspicion on one of these disorders, the use of next generation sequencing (NGS) offers a powerful tool to diagnose D-IEIs, allowing to examine all the candidate genes at once[8-10]. However, due to the wide heterogeneity of IEIs, it may be difficult to select patients for genetic analysis.
In fact, from a phenotypic point of view, due to their origin from general disturbances in immune regulation, D-IEIs tend to affect multiple organs and systems, composing complex clinical pictures that overlap disorders of distinct medical specialties, and tend to fully manifest over the time, with the definition of typical clinical pictures only in adults. Thus, patients with D-IEI can initially be diagnosed – especially in pediatric age - with common multifactorial disorders, pertaining to various medical specialties and displaying atypical clinical presentations such as unusual age of development, multiorgan involvement and response to therapies. These factors are congruent with the immune dysregulation theory.
In light of this, the aim of this review is to widen the awareness of “druggable” IEIs which may be hidden in various medical specialties, in order to promote an earlier diagnosis and a better therapy in this field.
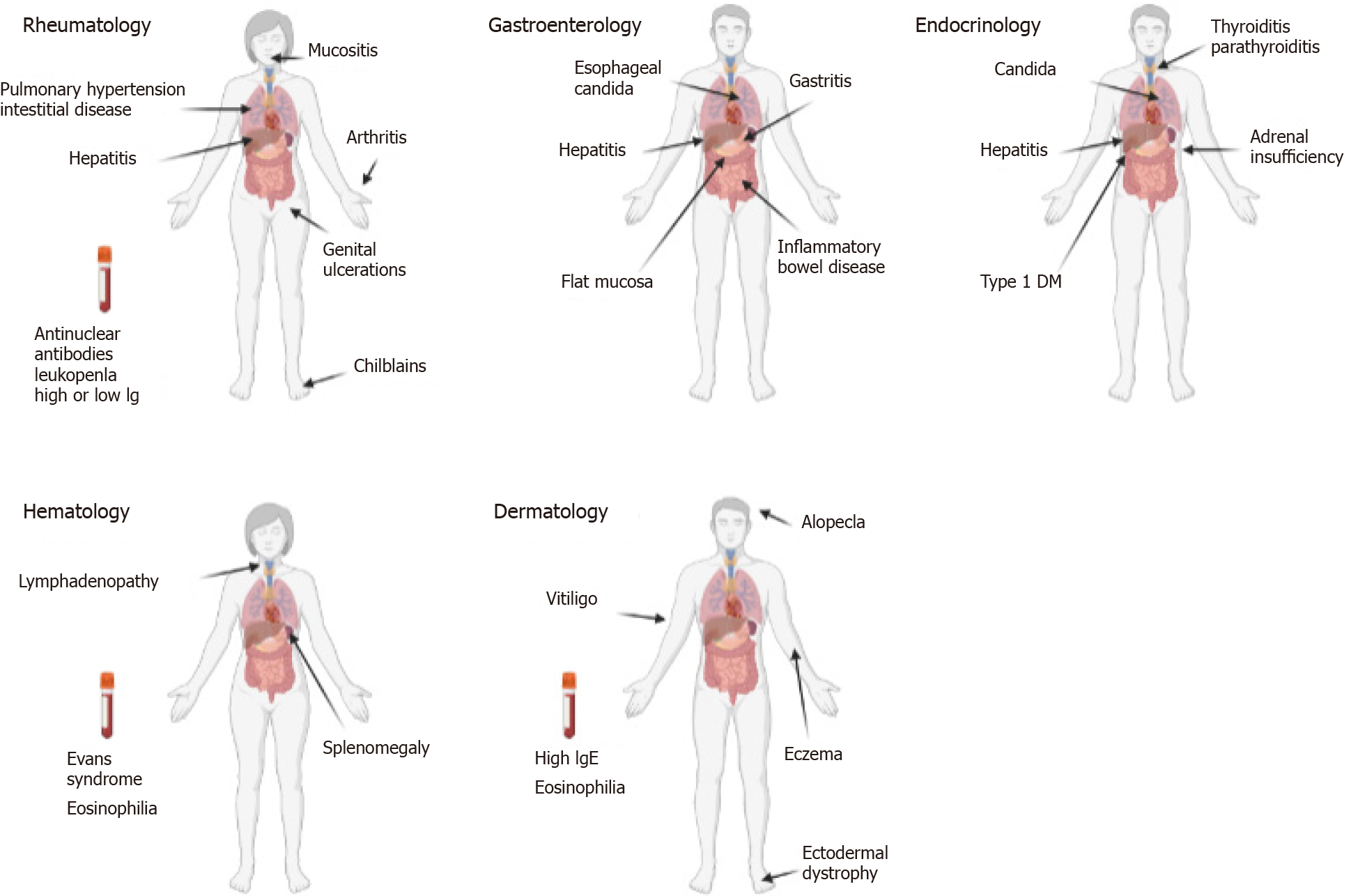
We present a list of druggable IEIs, with prevalent autoimmune, inflammatory and/or lymphoproliferative aspects, which may mimic common multifactorial disorders, and therefore are at risk of being missed, until significant organ damage manifests. Since effective treatments are now available for immune disorders, it is of crucial importance to consider the possibility of a primary immune defect in subjects presenting with clinical pictures suggestive of immune dysregulation, particularly those that overlap distinct rheumatological, gastroenterological, endocrinological and dermatological/allergic disorders (Table 1). As described elsewhere, there is now a trend of anticipating the time of genetic analysis, reserving more in-depth immunological investigations for a later time, with the aim of determining the role of any variants of uncertain significance found in candidate genes[7].
| Gene | Meccanism | Immune assessment | Clinicsautoimmunity | Lymphoproliferation | Infections | Therapy | |
| APDS | PIK3CDPIK3R1 | PI3K delta hyperactivation | Hypogammaglobulinemia IgA and IgG lowSenescent CD8 T cellsDNT | IBD; diabetes; arthritis | Lymphadenopathy, splenomegaly | Recurrent respiratory infections; herpes virus infections | HSCT; antibioticsrituximab and rapamycin; PI3Kδ inhibitors |
| STAT3 GOF | STAT3 | STAT3 hyperactivation | Hypogammaglobulinemia; decrease NK cells; decrease memory B cells; decrease regulatory T cells | Autoimmune cytopenia; diabetes; thyroiditis; arthritis | Adenopathy, hepatosplenomegaly | Herpes virus infections; fungal infections; bacterial infections; respiratory infections | JACK inhibitors |
| APECED | AIRE | Decrease of negative selection of autoreactive T cells in thymus | Autoantibodies;CD8+ effector T cells;FOXP3+ regulatory T cells | Autoimmune hypoparathyroidism;Addison’s disease | Chronic Candida infection | Hormone replacement therapy according to affected organs; immunosuppressive therapies; rituximab | |
| CTLA4 deficiency | CTLA4 | Defective switch off of lymphocyte activation | Hypogammaglobulinemia; DNT;increase of regulatory T cells with reduced expression of FOXP3;CD19+ B cells and switched memory B | Autoimmunecytopenia; hemolytic anemia and thrombocytopenia | Splenomegaly;chronic lymphadenopathy;hepatomegaly | Respiratory tract infections | Sirolimus; abatacept; HSCT |
| LRBA deficiency | LRBA | Defective switch off of lymphocyte activation | Hypogammaglobulinemia; DNT; FOXP3+regulatory T cells;CD19+ B cells;Natural Killer cells; increase of CD4+ and CD8+ memory T cells | Autoimmune gastritis;autoimmunecytopenia; hemolytic anemia; IBD;Autoimmune enteropathy | Splenomegaly;hepatomegaly | Respiratory infections | sirolimus; abatacept; HSCT |
| IPEX | FOXP3 | Failure of immune tolerance | Loss of FOXP3+ T cells;increased of Th2 and Th17 cells;autoantibodiesHypergammaglobulinemia IgA, IgE | Autoimmune enteropathy; autoimmune hemolytic anemia; autoimmune thrombocytopenia; autoimmune neutropenia; autoimmune thyroiditis; nephropathy; hepatitis | Skin infections | Glucocorticoids;Msirolimus;Mtacrolimus; abatacept; HSCT | |
| STAT1 GOF | STAT1 | STAT1 hyperactivation due to increase STAT1 phosphorylation | Low Th17 cells; low switched memory B cells;Hypergammaglobulinemia IgG | Chronic mucocutaneous candidiasis; hypothyroidism; autoimmune cytopenia, hepatopathy; psoriasis | Hepatomegaly; splenomegaly | Fungal, viral and mycobacterial infections; skin infections; Respiratory infections | Antifungal treatment; antibiotic prophylaxis; JACK inhibitors |
| DADA2 | ADA2 | Reduced activity level of the adenosine deaminase 2 | Hypogammaglobulinemia; increases macrophage release of TNF-α; upregulation of neutrophil activity; upregulation of pro-inflammatory cytokines; upregulation of type 1 interferon stimulated genes; aberrant B cell development and differentiation; decrease in NK | Vasculitis, immunodeficiency; autoimmune neutropenia; autoimmune cytopenia | Splenomegaly; lymphadenopathy; hepatomegaly | Verrucosis; herpes virus infections; increased susceptibility to infection with dsDNA viruses | Anti-TNF treatment (etanercept, infliximab,adalimumab); high-dose of glucocorticoids; HSCT; immunosuppressive drugs in isolated cases (mycophenolate, azathioprine, cyclosporine, rituximab, sirolimus, tacrolimus) |
| TNFAIP3 deficiency | TNFAIP3 | Excessive activation of NF-kB signalling | Antinuclear and anti-DNA antibodies; increased production of interferons and proinflammatory cytokines | Autoimmune cytopenias | Anti-TNF treatment; anti-IL1 treatment; glucocorticoid; colchicine |
IPEX is a monogenic immune disorder (due to mutations in FOXP3) characterized by an impaired development of Treg cells, resulting in failure of peripheral immune tolerance, with autoimmunity and allergic manifestations[11-13]. The disease typically presents in infancy with enteropathy, cutaneous disorders with eczema and nail changes, and endocrinopathies [e.g., type 1 diabetes mellitus (T1DM), thyroiditis]. Several other autoimmune manifestations may also be found. The treatment may benefit from sirolimus or tacrolimus, in addition to nutrition and glucocorticoids, but only hematopoietic stem cell transplantation allows a cure, the success of which is related to an early diagnosis[14-16]. Proof of concept for immunoregulation with abatacept was obtained in scurfy mice, which are considered a good animal model for the IPEX[17,18].
APECED is a monogenic immune disorder (due to mutations in AIRE) characterized by an abnormal presentation of self-antigens in the thymus, resulting in the failure of central immune tolerance, with autoimmunity[19-21]. The disease usually presents in infancy with recurrent and severe candidiasis (with susceptibility associated to IL17-neutralizing antibodies) and parathyroid and adrenal autoimmunity, but over time other autoimmune disorders (e.g., hepatitis, thyroiditis, vitiligo, alopecia, gastritis) are also observed[22,23]. Even if there is still no precision therapy for APECED, it is important to make an early diagnosis to establish a proper follow-up with prompt detection of new autoimmune phenomena, infections and malignancies[24,25].
CTLA4 and LRBA deficiencies are monogenic immune disorders associated with an impaired regulation of lymphocyte activation and development, resulting in autoimmunity and lymphoproliferation, but also infections[26-30]. Clinical features include hepatosplenomegaly, enteropathy, eczema, autoimmune cytopenia, arthritis, lupus-like features, hypogammaglobulinemia recurrence of infections and risk of malignancies (particularly due to chronic EBV infection). hematopoietic stem cell transplantation (HSCT) can cure the disease, however the treatment of milder cases may benefit from the use of CTLA4-Ig (abatacept)[28,30,31].
APDS (type I and II) are monogenic immune disorders associated with an impaired regulation of T and B cells maturation and survival, resulting in lymphoproliferation, autoimmunity and infections[32-35]. Clinical features include recurrent infections (especially respiratory, often complicated by the development of bronchiectasis and cutaneous) lymphoproliferative manifestations with risk of lymphoma[36-38], enteropathy and systemic lupus erythematosus (SLE)-like features. The immune defect is complex, with hypogammaglobulinemia with normal or increased IgM, reduced number of recent thymic emigrants and accumulation of senescent CD8 T cells. The pathogenic mechanisms can be partially reversed with drugs inhibiting the PIK3delta kinase, with a great potential in reducing the disease severity[39].
Monoallelic GOF mutations in STAT1 are associated with susceptibility to infections from bacteria and fungi, autoimmune disorders and rheumatologic manifestations, due to increased activation of interferon stimulated genes[40,41]. Since hyperactive STAT1 still depends on the trigger from Janus kinases, the use of JAK inhibitors can partially restore a physiological balance with great clinical benefit both on inflammatory and on infectious symptoms[42,43]. In a recent report, treatment with JAK inhibitors led to the reversal of autoimmune diabetes in a boy with STAT1 GOG[40].
Monoallelic GOF mutations in STAT3 are associated with autoimmune and lymphoproliferative disorders[44]. Patients may present autoimmune enteropathy, celiac disease-like changes in the jejunum, eczema, autoimmune polyendocrinopathy, lymphoproliferation with increased CD4- CD8- double negative T cells and risk of hematologic malignancies and hypogammaglobulinemia with recurrent infection. The use of JAK inhibitors can lead to significant clinical improvement in this case too[45].
DADA2 deficiency is a combined immunodeficiency due to the defective function of the adenosine deaminase-2enzyme. The disease is associated with lymphoproliferation, variable hypogammaglobulinemia and susceptibility to infection, arthritis, livedo reticularis, erythema nodosum, purpura and vasculitis with a picture of polyarteritis nodosa and ischemic strokes[46,47]. The main complaints of the disease are driven by TNF-alpha: Thus, there is a formal recommendation to start anti-TNF treatment as early as possible[48].
A20 haploinsufficiency is a monogenic immune disorder (due to mutations in TNFAIP3) characterized by an abnormal activation of NF-kB signalling, resulting in a phenotype similar to Behcet’s disease (BD)[49]. Indeed, clinical features include uveitis, recurrent oral and genital ulcerations, rash, abscesses and periodic fever. However, some patients may present with ulcerative colitis or with signs of SLE-like autoantibodies, increased production of interferons, autoimmune cytopenias and sometimes nephritis[50-52]. Anti-TNF treatment has been proven of great efficacy in several patients, even if it could be ineffective on lupus-related complaints[53].
There are many other rare IEIs that may present with complex features of immune dysregulation, such as lymphoproliferation, autoimmunity, inflammation, and risk of malignancies. However, the therapeutic implications of diagnosing these IEIs are not as straightforward as those for druggable diseases.
IEIs may present in diverse medical specialties mimicking more common multifactorial disorders. However, there are typical clinical pictures or peculiar sets of features that can raise suspicion of an IEI. After discussing the relevance of such conditions to specific medical specialties, we will propose red flags to help address the suspicion of an IEI in a multidisciplinary setting (Figure 1).
SLE: SLE is quite rare in children before pubertal age[54-56]. Cases with very early onset should always raise suspicion of an underpinning genetic disorder, with particular reference to complement deficiencies and interferonopathies[57,58]. Some cases may be anticipated by blood cells cytopenia or liver involvement. Arthritis may present a devious clinical course with slow development of contractures. A significant history of infections can sometimes be recorded. In many cases, the clinical picture is not the one that is the most typical of SLE, and classification criteria for SLE are not always completely met. NGS gene panels or whole exome sequencing have been proposed to allow an early detection of monogenic mimics of SLE[59,60]. A recent large study demonstrated that a monogenic cause could be found in 23% of patients meeting at least one of the following inclusion criteria: i.e., (1) Age of disease onset under 5 years; (2) Family history of autoimmune disease; (3) Syndromic SLE; and (4) Complicated conditions, such as life-threatening and refractory SLE[61]. Of particular importance is a prompt detection of druggable disorders like interferonopathies or STAT1 GOF immunodeficiency, which can benefit from the use of JAK inhibitors[42,60,62,63], or immune dysregulation deficiencies such as activated PIK3d syndrome that can benefit from PIK3δ inhibitors[61,64,65].
BD: BD is a complex inflammatory and autoimmune disorder with great clinical heterogeneity. BD is rare in pediatrics and often presents for many years in an incomplete form, mainly with recurrent oral and/or genital ulcerations and sometimes periodic fevers. Vasculitis, central nervous system or eye involvement are more typical of older children and adults. BD occurring in early childhood can also be underpinned by monogenic immune defects. Mounting evidence supports the opportunity of searching for monogenic mimics of BD in pediatrics, in particular in subjects with very early disease onset, positive familial history and severe phenotypes[52,66-68]. Some of these monogenic cases may present clinical pictures overlapping with SLE or Inflammatory bowel disease (IBD), as is in the case of STAT1 GOF and A20 haploinsufficiency. The molecular diagnosis in these cases can allow a targeted therapeutic choice and a proper follow-up.
Juvenile idiopathic arthritis is not so rare and is rarely associated with an underlying monogenic disorder. However, there are rare atypical cases, usually with polyarticular involvement refractory to conventional therapies, which may be associated with inflammatory involvement of liver or lungs, uncovering a more complex inflammatory pathogenesis, as in the cases of interferon-related disorders like COPA syndrome (also known as Autoimmune Interstitial Lung, Joint, and Kidney disease, OMIM # 616414), which can benefit from JAK inhibitors[42,69]. Other rare monogenic causes of arthritis in children include immune dysregulation disorders like CTLA4 or LRBA deficiency[30,70], which can be effectively treated by abatacept[30,70], Blau disease and LACC1 deficiency, which present some overlap with sarcoidosis-like granulomatous disorders[71,72].
IBD: IBD can occur at any age, however cases with very early onset are more likely due to monogenic defects[73,74]. Although the majority of monogenic IBD cases occur in children diagnosed before 6 years of age (prevalence of 7%-10%), recent reports suggest the presence of rare variants causing monogenic IBD also in children diagnosed older than 6-years of age. Several genes involved in monogenic IBD were identified, classified in six categories based on action mechanisms, namely defects in the epithelial barrier, T- and B-cell defect, hyperinflammatory and autoinflammatory disorders, phagocytic defects and immunoregulation defects, included IL-10 signaling defects[75].
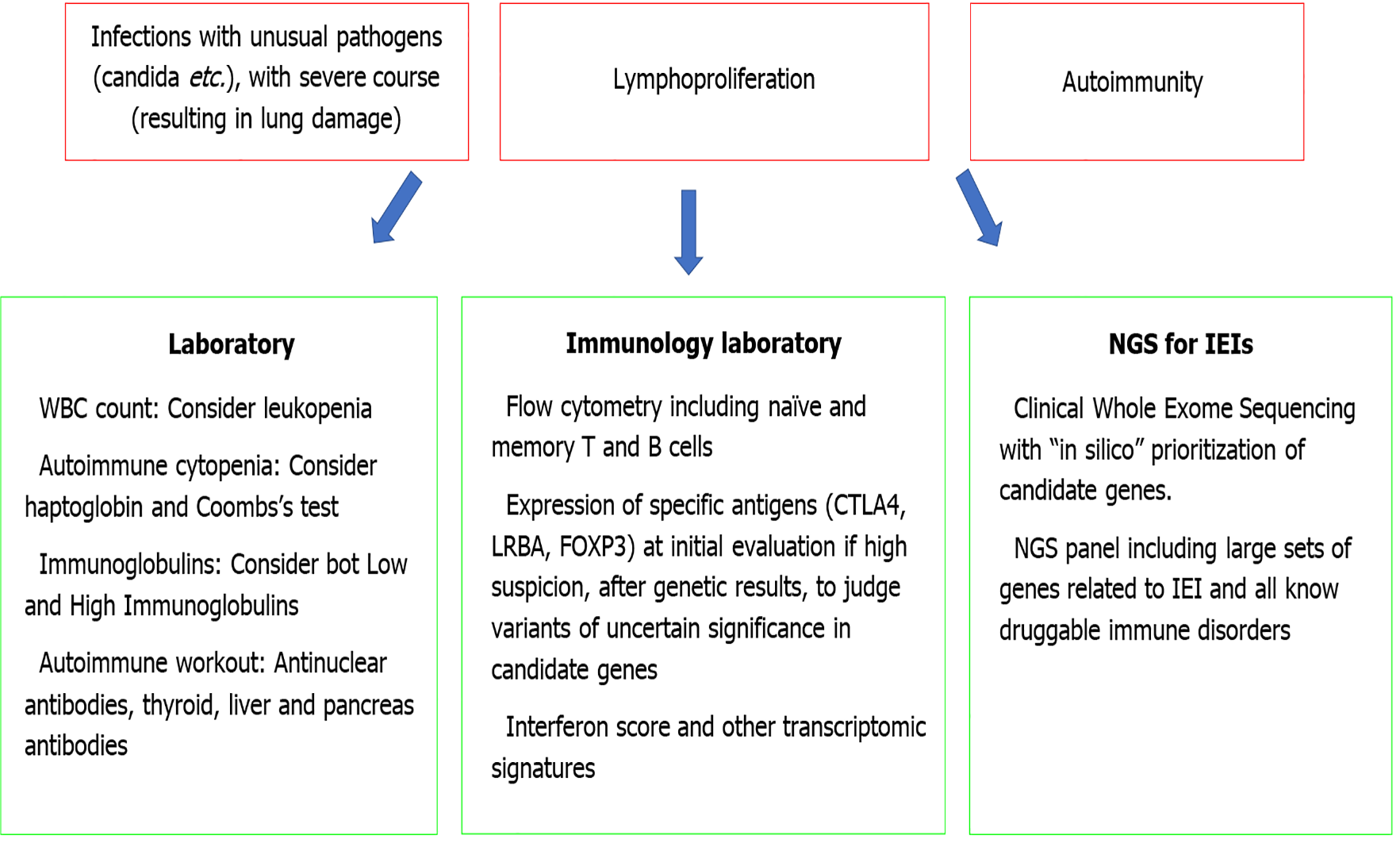
The clinical picture can be indistinguishable from IBD, however the presence of consanguinity, family history of autoimmune diseases and some histological and clinical features associated with extraintestinal manifestations, should raise the suspicion of an IEI. For example, autoimmune enteropathy and eosinophilic infiltrates may support the diagnosis of an underlying inborn immune defect. On a clinical ground, the presence of lymphoproliferative signs, the association with autoimmune phenomena in other organs, the increased burden of infections and the refractoriness to conventional therapies should prompt considering an IEI. The finding of lymphopenia, neutropenia or hypogammaglobulinemia can help address the suspicion of specific conditions. Eosinophilia can also be of great clinical significance. Some typical immunodeficiencies should be considered in cases with very early onset in infancy: Wiskott Aldrich Syndrome may present in the first days of life with inflammatory colitis, thrombocytopenia and infectious or inflammatory complaints; chronic granulomatous disease can mimic an IBD even before the occurrence of serious infections; combined immunodeficiency can present with intestinal inflammation and failure to thrive. Severe perianal disease, folliculitis and arthritis in early infancy suggest the presence of IL-10 signaling defects[76]. It is crucial to consider all these possibilities, as the diagnostic workout can be quite straightforward, if we pay attention to blood cell count, platelet count and volume, lymphocyte subsets and basic functional assays like the study of the oxidative burst in neutrophils or the dihydrorhodamine assay[7]. Various recent experiences proved the utility of performing high throughput genetic testing in children with very early onset IBD or in those with complex clinical pictures supportive of widespread immunodysregulation, with the aim of planning appropriate and targeted treatment[77].
Autoimmune enteropathy: Autoimmune enteropathy is a rare disorder characterized by intractable diarrhea, growth failure, presence of anti-enterocyte autoantibodies and typical mucosal changes with lymphocyte infiltrates and increased apoptotic cells[78,79]. Most cases occur during the first year of life with severe primarily secretory diarrhea[78]. The association with extra-intestinal diseases like insulin-dependent diabetes, thyroiditis, membranous glomerulopathy, interstitial nephritis and the presence of numerous autoantibodies (e.g., antinuclear, anti-smooth-muscle, anti-parietal cells, pancreatic islets...), should raise suspicion of IPEX syndrome. Furthermore, early diarrhea and malabsorption can occur in up to 25% of patients with APECED, due to the destruction of intestinal endocrine cells; in these cases, small bowel biopsies show mild damage, in contrast with the inflammation present in autoimmune enteropathy. An early enteropathy with a relative paucity of inflammatory cells in a patient with a history of recurrent infections should be suspicious for CVID[80,81]. Recent literature reports a series of patients with both LRBA deficiency and CTLA-4 haploinsufficiency with gastrointestinal manifestations, including autoimmune enteropathy, lymphocytic duodenitis resembling celiac disease and autoimmune gastritis[82,83].
Parenteral nutrition, steroids and immunosuppressants like cyclosporin A and tacrolimus are the cornerstones of the therapy. If an IEI can be found in a significant proportion of children with early onset IBD, this is even more true for autoimmune enteropathy[78]. In most cases underpinned by IEIs, HSCT can be the treatment of choice. However, when HSCT is not possible or has to be delayed, a treatment with abatacept or sirolimus may be a valuable option for CTLA4 and LRBA deficiency or for IPEX respectively[30].
Atrophic autoimmune gastritis: Atrophic autoimmune gastritis is an autoimmune disorder associated with chronic gastric autoimmunity, vitamin B12-dependent anemia and increased risk of developing gastric cancer[84]. This condition is often found associated with other immune disorders like common variable immunodeficiency, autoimmune thyroid disease and T1DM[85]. However, some patients may initially present only gastrointestinal complaints with gastritis[83]. Considering that this is a rare disorder in children, the likelihood of finding a monogenic cause is high, and an immunologic and genetic workup should be carried out before the patient develops further autoimmune phenomena. APECED, IPEX and immune dysregulatory disorders are examples of monogenic diseases that can present with autoimmune atrophic gastritis, even if it is rare for autoimmune gastritis to be the sole complaint[83,86,87].
Non celiac flat mucosa: The main cause of flat mucosa in jejunum is active celiac disease (CD), due to gluten-dependent immune activation in the lamina propria of the intestinal mucosa[88,89]. Similar findings can be found in subjects in whom CD has been ruled out, based on negative testing for anti-transglutaminase antibodies and/or absence of the predisposing HLA haplotypes and/or refractoriness to gluten free diet[83,90]. In these cases, intestinal inflammation may be related to an immune defect like common variable immunodeficiency. A flat jejunal mucosa has been described in subjects with immune dysregulatory diseases including IPEX, CTLA4 and LRB immunodeficiency, often in association with other gastrointestinal immune-mediated diseases, like autoimmune gastritis, autoimmune enteropathy or inflammatory bowel disease[83,91].
Esophageal candidiasis: Muco-cutaneous candidiasis is rarely observed in healthy children above the age of 1 year. Seldomly, therapies with oral glucocorticoids may facilitate the development of candidiasis in older children too, however the recurrence of the problem and the extension of the infection to the esophagus should always prompt the suspicion of an underlying immune defect. The underlying causes of chronic muco-cutaneous candidiasis may be monogenic, such as single gene mutations in the autoimmune regulator, signal transducer and activator of transcription-1 (STAT1) and -3 (STAT3), and many others genes (CARD9, TYK2, DOCK8, CD25, IL-1RA, RORC..), or the result of polymorphisms in genes encoding Dectin-1, NACHT LRR and PYD-containing protein 3, protein tyrosine phosphatase non receptor type-22, and Toll-like receptors which contribute to candida infection susceptibility[92-94]. It is worth noting that candida infections can sometimes be misinterpreted as the results of glucocorticoid treatments administered for other immune complaints, as some patients may present SLE-like phenomena (IL12RB1, STAT1 GOF) or autoimmune manifestations (APECED). Indeed, it is uncertain whether severe diffuse mucosal candidiasis reported in a subset of subjects with SLE are the result of immunosuppressive therapy or the marker of a possible underlying immunodeficiency[95].
Autoimmune polyglandular syndromes: Endocrine glands are the most typical targets of organ-specific autoimmunity, probably related to the cell-specific expression of proteins involved in the highly specialized machinery of hormone production. Based on distinct patterns of involvement of diverse endocrine systems, autoimmune polyglandular syndromes (APS) have been classified in three groups (APS1-3). Overall, APS have been associated with a general failure of maintaining immune tolerance to specialized tissue. This can be due to a defective presentation of tissue antigens in the thymus during lymphocyte development (as in APECED), improper control of autoreactive lymphocytes in target organs (as in IPEX and IPEX-like disorders) or to breakdown of tolerance by medications (as with checkpoint inhibitors used to induce anti-cancer immunity).
The combination of hypoparathyroidism and adrenal insufficiency (APS1) with muco-cutaneous candidiasis and ectodermal dystrophy is typical of APECED, however patients may initially present only with a single autoimmune disease. In these cases, the search for autoantibodies can help anticipate further autoimmune disorders, avoiding the risks of a hyperacute onset of disease. APS2 is characterized by T1DM, autoimmune thyroiditis and Addison Disease and is considered a multifactorial disorder associated with the HLA class II locus. APS3 is characterized by T1DM and autoimmune thyroiditis and can be either due to monogenic druggable immune defects (IPEX-like disorders) or to multifactorial causes including HLA class II variants. The presence of dermatitis, autoimmune cytopenia or lymphoproliferation in addition to autoimmune endocrine diseases should always raise suspicion of a monogenic immune dysregulation disorder.
The therapy is mainly based on the replacement of defective hormones. However, in cases associated with significant immune dysregulation, a prompt immune modulation can prevent the development of further autoimmune or infectious diseases, in particular in the cases of druggable IEIs responsive to abatacept and/or sirolimus.
Evans syndrome: Evans syndrome is characterized by the association of autoimmune hemolytic anemia with immune thrombocytopenic purpura. The two autoimmune conditions can occur simultaneously or in sequence. In some cases, autoimmune neutropenia can also be present. The term “Evans syndrome” refers to cases in which another definite diagnosis has not been made. However, a search for underlying immune defects may reveal the presence of a monogenic disease in a significant proportion of cases, in particular among those associated with signs of lymphoproliferation, that may be due to immune dysregulation immunodeficiencies[96]. The diagnosis of autoimmune lymphoproliferative syndromes (ALPS), activated PI3Kδ syndromes, IPEX syndrome, and CTLA4 or LRBA deficiencies can pave the way to the administration of targeted therapies like sirolimus, PI3Kδ inhibitors, and abatacept. Of note, sirolimus has been proven effective also in subjects with idiopathic Evans syndrome, suggesting that the disease may share relevant pathogenic features with ALPS. Since autoimmune cytopenias may be the presenting clinical condition in subjects with a common variable immunodeficiency or in SLE, a study of immunoglobulins, antinuclear antibodies and lymphocyte subpopulations is warranted in all subjects with Evans syndrome. Specific cytometric analyses may also give significant hints for rare IEIs[7].
Refractory eczema: Eczema is a common complaint in young children. If not treated properly, an active eczema can favor the development of allergies and other immune disturbances, fueling a vicious circle of inflammation, scratch injuries and infection. In most cases the disease can be easily treated until it wanes and disappears with age. However, in some rare cases the eczematous dermatitis shows a severe course and a refractoriness to treatments from the first weeks of life. These cases are often associated with poor growth or failure to thrive and sometimes with a history of infections. Blood examinations are usually performed to rule out a severe combined immunodeficiency or a Wiskott Aldrich Syndrome. However, other immune dysregulations are not as easy to diagnose. Peripheral eosinophilia is a clue to diagnosis, but a genetic panel for primary immune defects may be worthwhile in all severe cases. A diagnostic algorithm for IEIs associated with atopic phenotypes has been recently proposed by the Immunology Task Force of the Italian Society of Pediatric Allergy and Immunology[97].
Alopecia, vitiligo: Autoimmune alopecia and vitiligo may present in children at any age. The presence of autoimmune disorders in relatives, like thyroiditis is also common. However, if these complaints present together with other autoimmune or inflammatory disorders or with laboratory abnormalities like eosinophilia or a positive inflammatory index, it may be reasonable to perform an immunological and genetic investigation. For example, when associated with panniculitis, alopecia can rise suspicion of an interferon related disease like CANDLE syndrome, which can benefit from JAK inhibitors; if associated with severe dermatitis it may make you think of an immune dysregulation, like IPEX or CTLA4 or LRBA deficiency. It is worth noting that tofacitinib and other JAK inhibitors, and to a lesser extent abatacept, have been successfully used in subjects with alopecia areata even in the absence of a known underlying immune defect[98-100].
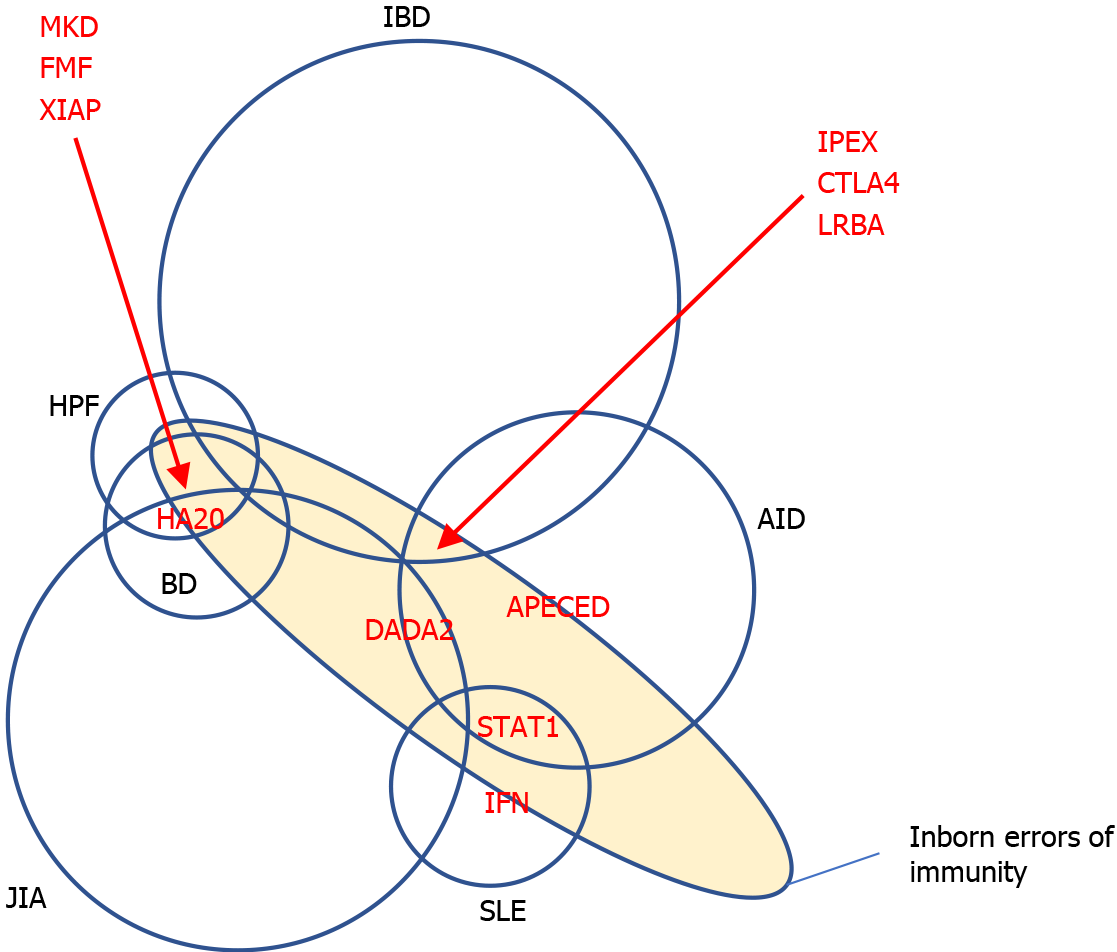
As described in the paragraphs above, IEIs may present with simultaneous or sequential involvements of various organs and systems. Many cases actually present substantial clinical overlap between disorders pertaining to different medical specialties. Whilst overlap syndromes are not rare in adult rheumatology or gastroenterology, this kind of conditions are not frequently encountered in children. Thus, we propose that IEIs should be considered in every child with immunological complaints overlapping diagnoses that are not usually seen in combination at this age. Since not all cases appear severe at beginning, the correct diagnosis may be often delayed to adult age or even missed. However, to recognize the underpinning monogenic disorder, in particular for druggable ones, it is crucial to choose molecularly targeted therapies able to prevent the development of further damages.
In Figure 2, we propose a schematic view of some druggable IEIs in areas of intersection between more common gnoseological entities. In Table 2, we highlight some “red flags” that could help consider a druggable IEI when dealing with complex immune disorders in any medical specialty.
| Clinical red flags |
| Early onset in childhood: The development of complex inflammatory disorders before puberty and particularly before early childhood rises suspicion of a congenital immune dysregulation |
| Overlap of symptoms in distinct specialties: A clinical history of distinct rheumatologic and non-rheumatologic conditions is not common in pediatrics, addressing a monogenic disorder |
| Lymphoproliferative manifestations: The presence of splenomegaly and/or lymphadenopathy in association with inflammatory or autoimmune diseases suggests an underlying inborn error of immunity. |
| Recurrent infections: The recurrence of severe or atypical infections (especially candidiasis) in association with inflammatory or autoimmune diseases is rarely a consequence of immunomodulatory therapies in children, but it does suggest an immunological defect |
| Familiarity with autoimmunity: The clustering of autoimmune disorders in families acknowledges a likely monogenic cause |
| Laboratory red flags |
| Hypogammaglobulinemia |
| Hypergammaglobulinemia |
| Leukopenia |
| Hypereosinophilia |
| Wide positivity of autoantibodies |
| Positive interferon signature |
Monogenic immune disorders are very rare, especially in subjects with adolescent/adult-onset diseases. Monogenic causes are more likely in subjects with a very early onset than in older ages. We thus provide some hints on when to suspect a group of monogenic disorders, the natural history of which can be favorably influenced by the availability of effective treatments (Figure 3).
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: European Society for Immunodeficiencies; Italian Primary Immunodeficiency Network; Italian Society of Pediatrics.
Specialty type: Immunology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Pongcharoen S S-Editor: Liu M L-Editor: A P-Editor: Liu M
| 1. | Azizi G, Tavakol M, Yazdani R, Delavari S, Moeini Shad T, Rasouli SE, Jamee M, Pashangzadeh S, Kalantari A, Shariat M, Shafiei A, Mohammadi J, Hassanpour G, Chavoshzadeh Z, Mahdaviani SA, Momen T, Behniafard N, Nabavi M, Bemanian MH, Arshi S, Molatefi R, Sherkat R, Shirkani A, Alyasin S, Jabbari-Azad F, Ghaffari J, Mesdaghi M, Ahanchian H, Khoshkhui M, Eslamian MH, Cheraghi T, Dabbaghzadeh A, Nasiri Kalmarzi R, Esmaeilzadeh H, Tafaroji J, Khalili A, Sadeghi-Shabestari M, Darougar S, Moghtaderi M, Ahmadiafshar A, Shakerian B, Heidarzadeh M, Ghalebaghi B, Fathi SM, Darabi B, Fallahpour M, Mohsenzadeh A, Ebrahimi S, Sharafian S, Vosughimotlagh A, Tafakoridelbari M, Rahimi Haji-Abadi M, Ashournia P, Razaghian A, Rezaei A, Salami F, Shirmast P, Bazargan N, Mamishi S, Khazaei HA, Negahdari B, Shokri S, Nabavizadeh SH, Bazregari S, Ghasemi R, Bayat S, Eshaghi H, Rezaei N, Abolhassani H, Aghamohammadi A. Autoimmune manifestations among 461 patients with monogenic inborn errors of immunity. Pediatr Allergy Immunol. 2021;32:1335-1348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Bousfiha A, Jeddane L, Picard C, Al-Herz W, Ailal F, Chatila T, Cunningham-Rundles C, Etzioni A, Franco JL, Holland SM, Klein C, Morio T, Ochs HD, Oksenhendler E, Puck J, Torgerson TR, Casanova JL, Sullivan KE, Tangye SG. Human Inborn Errors of Immunity: 2019 Update of the IUIS Phenotypical Classification. J Clin Immunol. 2020;40:66-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 340] [Cited by in RCA: 469] [Article Influence: 93.8] [Reference Citation Analysis (0)] |
| 3. | Lenardo M, Lo B, Lucas CL. Genomics of Immune Diseases and New Therapies. Annu Rev Immunol. 2016;34:121-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 4. | Valencic E, Smid A, Jakopin Z, Tommasini A, Mlinaric-Rascan I. Repositioning Drugs for Rare Immune Diseases: Hopes and Challenges for a Precision Medicine. Curr Med Chem. 2018;25:2764-2782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 5. | Ballow M, Leiding JW. Precision Medicine in the Treatment of Primary Immune Deficiency Patients With Disorders of Immune Dysregulation. Clin Rev Allergy Immunol. 2021 epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Chellapandian D, Chitty-Lopez M, Leiding JW. Precision Therapy for the Treatment of Primary Immunodysregulatory Diseases. Immunol Allergy Clin North Am. 2020;40:511-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 7. | Rispoli F, Valencic E, Girardelli M, Pin A, Tesser A, Piscianz E, Boz V, Faletra F, Severini GM, Taddio A, Tommasini A. Immunity and Genetics at the Revolving Doors of Diagnostics in Primary Immunodeficiencies. Diagnostics (Basel). 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Quinn J, Modell V, Holle J, Truty R, Aradhya S, Johnson B, Orange J, Modell F. Jeffrey's insights: Jeffrey Modell Foundation's global genetic sequencing pilot program to identify specific primary immunodeficiency defects to optimize disease management and treatment. Immunol Res. 2020;68:126-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 9. | Chinn IK, Orange JS. A 2020 update on the use of genetic testing for patients with primary immunodeficiency. Expert Rev Clin Immunol. 2020;16:897-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Bosch B, Itan Y, Meyts I. Whole-exome sequencing for detecting inborn errors of immunity: overview and perspectives. F1000Res. 2017;6:2056. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Gambineri E, Torgerson TR, Ochs HD. Immune dysregulation, polyendocrinopathy, enteropathy, and X-linked inheritance (IPEX), a syndrome of systemic autoimmunity caused by mutations of FOXP3, a critical regulator of T-cell homeostasis. Curr Opin Rheumatol. 2003;15:430-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 425] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 12. | Bennett CL, Christie J, Ramsdell F, Brunkow ME, Ferguson PJ, Whitesell L, Kelly TE, Saulsbury FT, Chance PF, Ochs HD. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2432] [Cited by in RCA: 2489] [Article Influence: 103.7] [Reference Citation Analysis (0)] |
| 13. | Wildin RS, Ramsdell F, Peake J, Faravelli F, Casanova JL, Buist N, Levy-Lahad E, Mazzella M, Goulet O, Perroni L, Bricarelli FD, Byrne G, McEuen M, Proll S, Appleby M, Brunkow ME. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat Genet. 2001;27:18-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1371] [Cited by in RCA: 1380] [Article Influence: 57.5] [Reference Citation Analysis (0)] |
| 14. | Barzaghi F, Amaya Hernandez LC, Neven B, Ricci S, Kucuk ZY, Bleesing JJ, Nademi Z, Slatter MA, Ulloa ER, Shcherbina A, Roppelt A, Worth A, Silva J, Aiuti A, Murguia-Favela L, Speckmann C, Carneiro-Sampaio M, Fernandes JF, Baris S, Ozen A, Karakoc-Aydiner E, Kiykim A, Schulz A, Steinmann S, Notarangelo LD, Gambineri E, Lionetti P, Shearer WT, Forbes LR, Martinez C, Moshous D, Blanche S, Fisher A, Ruemmele FM, Tissandier C, Ouachee-Chardin M, Rieux-Laucat F, Cavazzana M, Qasim W, Lucarelli B, Albert MH, Kobayashi I, Alonso L, Diaz De Heredia C, Kanegane H, Lawitschka A, Seo JJ, Gonzalez-Vicent M, Diaz MA, Goyal RK, Sauer MG, Yesilipek A, Kim M, Yilmaz-Demirdag Y, Bhatia M, Khlevner J, Richmond Padilla EJ, Martino S, Montin D, Neth O, Molinos-Quintana A, Valverde-Fernandez J, Broides A, Pinsk V, Ballauf A, Haerynck F, Bordon V, Dhooge C, Garcia-Lloret ML, Bredius RG, Kałwak K, Haddad E, Seidel MG, Duckers G, Pai SY, Dvorak CC, Ehl S, Locatelli F, Goldman F, Gennery AR, Cowan MJ, Roncarolo MG, Bacchetta R; Primary Immune Deficiency Treatment Consortium (PIDTC) and the Inborn Errors Working Party (IEWP) of the European Society for Blood and Marrow Transplantation (EBMT). Long-term follow-up of IPEX syndrome patients after different therapeutic strategies: An international multicenter retrospective study. J Allergy Clin Immunol. 2018;141:1036-1049.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 207] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 15. | Battaglia M, Stabilini A, Roncarolo MG. Rapamycin selectively expands CD4+CD25+FoxP3+ regulatory T cells. Blood. 2005;105:4743-4748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 961] [Article Influence: 48.1] [Reference Citation Analysis (0)] |
| 16. | Bevacqua M, Baldo F, Pastore S, Valencic E, Tommasini A, Maestro A, Rabusin M, Arbo A, Barbi E. Off-Label Use of Sirolimus and Everolimus in a Pediatric Center: A Case Series and Review of the Literature. Paediatr Drugs. 2019;21:185-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 17. | Singh N, Chandler PR, Seki Y, Baban B, Takezaki M, Kahler DJ, Munn DH, Larsen CP, Mellor AL, Iwashima M. Role of CD28 in fatal autoimmune disorder in scurfy mice. Blood. 2007;110:1199-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 18. | Ochs HD, Gambineri E, Torgerson TR. IPEX, FOXP3 and regulatory T-cells: a model for autoimmunity. Immunol Res. 2007;38:112-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 128] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 19. | Anderson MS, Su MA. AIRE expands: new roles in immune tole rance and beyond. Nat Rev Immunol. 2016;16:247-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 170] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 20. | Malchow S, Leventhal DS, Lee V, Nishi S, Socci ND, Savage PA. Aire Enforces Immune Tolerance by Directing Autoreactive T Cells into the Regulatory T Cell Lineage. Immunity. 2016;44:1102-1113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 158] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 21. | Notarangelo LD, Mazza C, Forino C, Mazzolari E, Buzi F. AIRE and immunological tolerance: insights from the study of autoimmune polyendocrinopathy candidiasis and ectodermal dystrophy. Curr Opin Allergy Clin Immunol. 2004;4:491-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Sarkadi AK, Taskó S, Csorba G, Tóth B, Erdős M, Maródi L. Autoantibodies to IL-17A may be correlated with the severity of mucocutaneous candidiasis in APECED patients. J Clin Immunol. 2014;34:181-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Mazza C, Buzi F, Ortolani F, Vitali A, Notarangelo LD, Weber G, Bacchetta R, Soresina A, Lougaris V, Greggio NA, Taddio A, Pasic S, de Vroede M, Pac M, Kilic SS, Ozden S, Rusconi R, Martino S, Capalbo D, Salerno M, Pignata C, Radetti G, Maggiore G, Plebani A, Badolato R. Clinical heterogeneity and diagnostic delay of autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy syndrome. Clin Immunol. 2011;139:6-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Bruserud Ø, Oftedal BE, Landegren N, Erichsen MM, Bratland E, Lima K, Jørgensen AP, Myhre AG, Svartberg J, Fougner KJ, Bakke Å, Nedrebø BG, Mella B, Breivik L, Viken MK, Knappskog PM, Marthinussen MC, Løvås K, Kämpe O, Wolff AB, Husebye ES. A Longitudinal Follow-up of Autoimmune Polyendocrine Syndrome Type 1. J Clin Endocrinol Metab. 2016;101:2975-2983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 25. | Borchers J, Pukkala E, Mäkitie O, Laakso S. Patients With APECED Have Increased Early Mortality Due to Endocrine Causes, Malignancies and infections. J Clin Endocrinol Metab. 2020;105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 26. | Verma N, Burns SO, Walker LSK, Sansom DM. Immune deficiency and autoimmunity in patients with CTLA-4 (CD152) mutations. Clin Exp Immunol. 2017;190:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 98] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 27. | Schwab C, Gabrysch A, Olbrich P, Patiño V, Warnatz K, Wolff D, Hoshino A, Kobayashi M, Imai K, Takagi M, Dybedal I, Haddock JA, Sansom DM, Lucena JM, Seidl M, Schmitt-Graeff A, Reiser V, Emmerich F, Frede N, Bulashevska A, Salzer U, Schubert D, Hayakawa S, Okada S, Kanariou M, Kucuk ZY, Chapdelaine H, Petruzelkova L, Sumnik Z, Sediva A, Slatter M, Arkwright PD, Cant A, Lorenz HM, Giese T, Lougaris V, Plebani A, Price C, Sullivan KE, Moutschen M, Litzman J, Freiberger T, van de Veerdonk FL, Recher M, Albert MH, Hauck F, Seneviratne S, Pachlopnik Schmid J, Kolios A, Unglik G, Klemann C, Speckmann C, Ehl S, Leichtner A, Blumberg R, Franke A, Snapper S, Zeissig S, Cunningham-Rundles C, Giulino-Roth L, Elemento O, Dückers G, Niehues T, Fronkova E, Kanderová V, Platt CD, Chou J, Chatila TA, Geha R, McDermott E, Bunn S, Kurzai M, Schulz A, Alsina L, Casals F, Deyà-Martinez A, Hambleton S, Kanegane H, Taskén K, Neth O, Grimbacher B. Phenotype, penetrance, and treatment of 133 cytotoxic T-lymphocyte antigen 4-insufficient subjects. J Allergy Clin Immunol. 2018;142:1932-1946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 330] [Article Influence: 47.1] [Reference Citation Analysis (0)] |
| 28. | Jamee M, Hosseinzadeh S, Sharifinejad N, Zaki-Dizaji M, Matloubi M, Hasani M, Baris S, Alsabbagh M, Lo B, Azizi G. Comprehensive comparison between 222 CTLA-4 haploinsufficiency and 212 LRBA deficiency patients: a systematic review. Clin Exp Immunol. 2021;205:28-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 29. | Krone KA, Winant AJ, Vargas SO, Platt CD, Bartnikas LM, Janssen E, Lillehei C, Lee EY, Fishman MP, Casey A. Pulmonary manifestations of immune dysregulation in CTLA-4 haploinsufficiency and LRBA deficiency. Pediatr Pulmonol. 2021;56:2232-2241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Tesch VK, Abolhassani H, Shadur B, Zobel J, Mareika Y, Sharapova S, Karakoc-Aydiner E, Rivière JG, Garcia-Prat M, Moes N, Haerynck F, Gonzales-Granado LI, Santos Pérez JL, Mukhina A, Shcherbina A, Aghamohammadi A, Hammarström L, Dogu F, Haskologlu S, İkincioğulları AI, Köstel Bal S, Baris S, Kilic SS, Karaca NE, Kutukculer N, Girschick H, Kolios A, Keles S, Uygun V, Stepensky P, Worth A, van Montfrans JM, Peters AMJ, Meyts I, Adeli M, Marzollo A, Padem N, Khojah AM, Chavoshzadeh Z, Avbelj Stefanija M, Bakhtiar S, Florkin B, Meeths M, Gamez L, Grimbacher B, Seppänen MRJ, Lankester A, Gennery AR, Seidel MG; Inborn Errors, Clinical, and Registry Working Parties of the European Society for Blood and Marrow Transplantation and the European Society for Immunodeficiencies. Long-term outcome of LRBA deficiency in 76 patients after various treatment modalities as evaluated by the immune deficiency and dysregulation activity (IDDA) score. J Allergy Clin Immunol. 2020;145:1452-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 101] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 31. | Lo B, Zhang K, Lu W, Zheng L, Zhang Q, Kanellopoulou C, Zhang Y, Liu Z, Fritz JM, Marsh R, Husami A, Kissell D, Nortman S, Chaturvedi V, Haines H, Young LR, Mo J, Filipovich AH, Bleesing JJ, Mustillo P, Stephens M, Rueda CM, Chougnet CA, Hoebe K, McElwee J, Hughes JD, Karakoc-Aydiner E, Matthews HF, Price S, Su HC, Rao VK, Lenardo MJ, Jordan MB. AUTOIMMUNE DISEASE. Patients with LRBA deficiency show CTLA4 Loss and immune dysregulation responsive to abatacept therapy. Science. 2015;349:436-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 485] [Article Influence: 48.5] [Reference Citation Analysis (0)] |
| 32. | Tessarin G, Rossi S, Baronio M, Gazzurelli L, Colpani M, Benvenuto A, Zunica F, Cardinale F, Martire B, Brescia L, Costagliola G, Luti L, Casazza G, Menconi MC, Saettini F, Palumbo L, Girelli MF, Badolato R, Lanzi G, Chiarini M, Moratto D, Meini A, Giliani S, Bondioni MP, Plebani A, Lougaris V. Activated Phosphoinositide 3-Kinase Delta Syndrome 1: Clinical and Immunological Data from an Italian Cohort of Patients. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 33. | Ewertowska M, Grześk E, Urbańczyk A, Dąbrowska A, Bąbol-Pokora K, Łęcka M, Kołtan S. Activated phosphoinositide 3-kinase delta syndrome 1 and 2 (APDS 1 and APDS 2): similarities and differences based on clinical presentation in two boys. Allergy Asthma Clin Immunol. 2020;16:22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Maccari ME, Abolhassani H, Aghamohammadi A, Aiuti A, Aleinikova O, Bangs C, Baris S, Barzaghi F, Baxendale H, Buckland M, Burns SO, Cancrini C, Cant A, Cathébras P, Cavazzana M, Chandra A, Conti F, Coulter T, Devlin LA, Edgar JDM, Faust S, Fischer A, Garcia-Prat M, Hammarström L, Heeg M, Jolles S, Karakoc-Aydiner E, Kindle G, Kiykim A, Kumararatne D, Grimbacher B, Longhurst H, Mahlaoui N, Milota T, Moreira F, Moshous D, Mukhina A, Neth O, Neven B, Nieters A, Olbrich P, Ozen A, Pachlopnik Schmid J, Picard C, Prader S, Rae W, Reichenbach J, Rusch S, Savic S, Scarselli A, Scheible R, Sediva A, Sharapova SO, Shcherbina A, Slatter M, Soler-Palacin P, Stanislas A, Suarez F, Tucci F, Uhlmann A, van Montfrans J, Warnatz K, Williams AP, Wood P, Kracker S, Condliffe AM, Ehl S. Disease Evolution and Response to Rapamycin in Activated Phosphoinositide 3-Kinase δ Syndrome: The European Society for Immunodeficiencies-Activated Phosphoinositide 3-Kinase δ Syndrome Registry. Front Immunol. 2018;9:543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 146] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 35. | Lucas CL, Chandra A, Nejentsev S, Condliffe AM, Okkenhaug K. PI3Kδ and primary immunodeficiencies. Nat Rev Immunol. 2016;16:702-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 252] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 36. | Angulo I, Vadas O, Garçon F, Banham-Hall E, Plagnol V, Leahy TR, Baxendale H, Coulter T, Curtis J, Wu C, Blake-Palmer K, Perisic O, Smyth D, Maes M, Fiddler C, Juss J, Cilliers D, Markelj G, Chandra A, Farmer G, Kielkowska A, Clark J, Kracker S, Debré M, Picard C, Pellier I, Jabado N, Morris JA, Barcenas-Morales G, Fischer A, Stephens L, Hawkins P, Barrett JC, Abinun M, Clatworthy M, Durandy A, Doffinger R, Chilvers ER, Cant AJ, Kumararatne D, Okkenhaug K, Williams RL, Condliffe A, Nejentsev S. Phosphoinositide 3-kinase δ gene mutation predisposes to respiratory infection and airway damage. Science. 2013;342:866-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 481] [Article Influence: 40.1] [Reference Citation Analysis (0)] |
| 37. | Carpier JM, Lucas CL. Epstein-Barr Virus Susceptibility in Activated PI3Kδ Syndrome (APDS) Immunodeficiency. Front Immunol. 2017;8:2005. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 38. | Durandy A, Kracker S. Increased activation of PI3 kinase-δ predisposes to B-cell lymphoma. Blood. 2020;135:638-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 39. | Valencic E, Grasso AG, Conversano E, Lucafò M, Piscianz E, Gregori M, Conti F, Cancrini C, Tommasini A. Theophylline as a precision therapy in a young girl with PIK3R1 immunodeficiency. J Allergy Clin Immunol Pract. 2018;6:2165-2167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 40. | Chaimowitz NS, Ebenezer SJ, Hanson IC, Anderson M, Forbes LR. STAT1 Gain of Function, Type 1 Diabetes, and Reversal with JAK Inhibition. N Engl J Med. 2020;383:1494-1496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 41. | van de Veerdonk FL, Plantinga TS, Hoischen A, Smeekens SP, Joosten LA, Gilissen C, Arts P, Rosentul DC, Carmichael AJ, Smits-van der Graaf CA, Kullberg BJ, van der Meer JW, Lilic D, Veltman JA, Netea MG. STAT1 mutations in autosomal dominant chronic mucocutaneous candidiasis. N Engl J Med. 2011;365:54-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 557] [Cited by in RCA: 507] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 42. | Pin A, Tesser A, Pastore S, Moressa V, Valencic E, Arbo A, Maestro A, Tommasini A, Taddio A. Biological and Clinical Changes in a Pediatric Series Treated with Off-Label JAK Inhibitors. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 43. | Weinacht KG, Charbonnier LM, Alroqi F, Plant A, Qiao Q, Wu H, Ma C, Torgerson TR, Rosenzweig SD, Fleisher TA, Notarangelo LD, Hanson IC, Forbes LR, Chatila TA. Ruxolitinib reverses dysregulated T helper cell responses and controls autoimmunity caused by a novel signal transducer and activator of transcription 1 (STAT1) gain-of-function mutation. J Allergy Clin Immunol. 2017;139:1629-1640.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 120] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 44. | Flanagan SE, Haapaniemi E, Russell MA, Caswell R, Allen HL, De Franco E, McDonald TJ, Rajala H, Ramelius A, Barton J, Heiskanen K, Heiskanen-Kosma T, Kajosaari M, Murphy NP, Milenkovic T, Seppänen M, Lernmark Å, Mustjoki S, Otonkoski T, Kere J, Morgan NG, Ellard S, Hattersley AT. Activating germline mutations in STAT3 cause early-onset multi-organ autoimmune disease. Nat Genet. 2014;46:812-814. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 379] [Cited by in RCA: 390] [Article Influence: 35.5] [Reference Citation Analysis (0)] |
| 45. | Forbes LR, Vogel TP, Cooper MA, Castro-Wagner J, Schussler E, Weinacht KG, Plant AS, Su HC, Allenspach EJ, Slatter M, Abinun M, Lilic D, Cunningham-Rundles C, Eckstein O, Olbrich P, Guillerman RP, Patel NC, Demirdag YY, Zerbe C, Freeman AF, Holland SM, Szabolcs P, Gennery A, Torgerson TR, Milner JD, Leiding JW. Jakinibs for the treatment of immune dysregulation in patients with gain-of-function signal transducer and activator of transcription 1 (STAT1) or STAT3 mutations. J Allergy Clin Immunol. 2018;142:1665-1669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 198] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 46. | Caorsi R, Penco F, Grossi A, Insalaco A, Omenetti A, Alessio M, Conti G, Marchetti F, Picco P, Tommasini A, Martino S, Malattia C, Gallizzi R, Podda RA, Salis A, Falcini F, Schena F, Garbarino F, Morreale A, Pardeo M, Ventrici C, Passarelli C, Zhou Q, Severino M, Gandolfo C, Damonte G, Martini A, Ravelli A, Aksentijevich I, Ceccherini I, Gattorno M. ADA2 deficiency (DADA2) as an unrecognised cause of early onset polyarteritis nodosa and stroke: a multicentre national study. Ann Rheum Dis. 2017;76:1648-1656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 169] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 47. | Insalaco A, Moneta GM, Pardeo M, Caiello I, Messia V, Bracaglia C, Passarelli C, De Benedetti F. Variable Clinical Phenotypes and Relation of Interferon Signature with Disease Activity in ADA2 Deficiency. J Rheumatol. 2019;46:523-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 48. | Sahin S, Adrovic A, Kasapcopur O. A monogenic autoinflammatory disease with fatal vasculitis: deficiency of adenosine deaminase 2. Curr Opin Rheumatol. 2020;32:3-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 49. | Yu MP, Xu XS, Zhou Q, Deuitch N, Lu MP. Haploinsufficiency of A20 (HA20): updates on the genetics, phenotype, pathogenesis and treatment. World J Pediatr. 2020;16:575-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 50. | Aeschlimann FA, Batu ED, Canna SW, Go E, Gül A, Hoffmann P, Leavis HL, Ozen S, Schwartz DM, Stone DL, van Royen-Kerkof A, Kastner DL, Aksentijevich I, Laxer RM. A20 haploinsufficiency (HA20): clinical phenotypes and disease course of patients with a newly recognised NF-kB-mediated autoinflammatory disease. Ann Rheum Dis. 2018;77:728-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 164] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 51. | Franco-Jarava C, Wang H, Martin-Nalda A, Alvarez SD, García-Prat M, Bodet D, García-Patos V, Plaja A, Rudilla F, Rodriguez-Sureda V, García-Latorre L, Aksentijevich I, Colobran R, Soler-Palacín P. TNFAIP3 haploinsufficiency is the cause of autoinflammatory manifestations in a patient with a deletion of 13Mb on chromosome 6. Clin Immunol. 2018;191:44-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 52. | Girardelli M, Valencic E, Moressa V, Margagliotta R, Tesser A, Pastore S, Spadola O, Athanasakis E, Severini GM, Taddio A, Tommasini A. Genetic and immunologic findings in children with recurrent aphthous stomatitis with systemic inflammation. Pediatr Rheumatol Online J. 2021;19:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 53. | Aeschlimann FA, Laxer RM. Haploinsufficiency of A20 and other paediatric inflammatory disorders with mucosal involvement. Curr Opin Rheumatol. 2018;30:506-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 54. | Massias JS, Smith EMD, Al-Abadi E, Armon K, Bailey K, Ciurtin C, Davidson J, Gardner-Medwin J, Haslam K, Hawley DP, Leahy A, Leone V, McErlane F, Mewar D, Modgil G, Moots R, Pilkington C, Ramanan AV, Rangaraj S, Riley P, Sridhar A, Wilkinson N, Beresford MW, Hedrich CM. Clinical and laboratory characteristics in juvenile-onset systemic lupus erythematosus across age groups. Lupus. 2020;29:474-481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 73] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 55. | Malattia C, Martini A. Paediatric-onset systemic lupus erythematosus. Best Pract Res Clin Rheumatol. 2013;27:351-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 82] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 56. | Charras A, Smith E, Hedrich CM. Systemic Lupus Erythematosus in Children and Young People. Curr Rheumatol Rep. 2021;23:20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 96] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 57. | Hiraki LT, Silverman ED. Genomics of Systemic Lupus Erythematosus: Insights Gained by Studying Monogenic Young-Onset Systemic Lupus Erythematosus. Rheum Dis Clin North Am. 2017;43:415-434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 58. | Tak AS, Satapathy J, Jana M, Sinha A, Jat KR, Bagri NK. Monogenic lupus with homozygous C4A deficiency presenting as bronchiectasis and immune-mediated thrombocytopenia. Rheumatol Int. 2021 epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 59. | Batu ED, Koşukcu C, Taşkıran E, Sahin S, Akman S, Sözeri B, Ünsal E, Bilginer Y, Kasapcopur O, Alikaşifoğlu M, Ozen S. Whole Exome Sequencing in Early-onset Systemic Lupus Erythematosus. J Rheumatol. 2018;45:1671-1679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 60. | Tirosh I, Spielman S, Barel O, Ram R, Stauber T, Paret G, Rubinsthein M, Pessach IM, Gerstein M, Anikster Y, Shukrun R, Dagan A, Adler K, Pode-Shakked B, Volkov A, Perelman M, Greenberger S, Somech R, Lahav E, Majmundar AJ, Padeh S, Hildebrandt F, Vivante A. Whole exome sequencing in childhood-onset lupus frequently detects single gene etiologies. Pediatr Rheumatol Online J. 2019;17:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 61. | Li G, Liu H, Li Y, Zhang T, Yao W, Guan W, Shi Y, Wu B, Xu H, Sun L. Genetic heterogeneity in Chinese children with systemic lupus erythematosus. Clin Exp Rheumatol. 2021;39:214-222. [PubMed] |
| 62. | Omarjee O, Picard C, Frachette C, Moreews M, Rieux-Laucat F, Soulas-Sprauel P, Viel S, Lega JC, Bader-Meunier B, Walzer T, Mathieu AL, Cimaz R, Belot A. Monogenic lupus: Dissecting heterogeneity. Autoimmun Rev. 2019;18:102361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 85] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 63. | Tesser A, de Carvalho LM, Sandrin-Garcia P, Pin A, Pastore S, Taddio A, Roberti LR, de Paula Queiroz RG, Ferriani VPL, Crovella S, Tommasini A. Higher interferon score and normal complement levels may identify a distinct clinical subset in children with systemic lupus erythematosus. Arthritis Res Ther. 2020;22:91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 64. | Li G, Li Y, Liu H, Shi Y, Guan W, Zhang T, Yao W, Wu B, Xu H, Sun L. Genetic heterogeneity of pediatric systemic lupus erythematosus with lymphoproliferation. Medicine (Baltimore). 2020;99:e20232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 65. | Jamee M, Moniri S, Zaki-Dizaji M, Olbrich P, Yazdani R, Jadidi-Niaragh F, Aghamahdi F, Abolhassani H, Condliffe AM, Aghamohammadi A, Azizi G. Clinical, Immunological, and Genetic Features in Patients with Activated PI3Kδ Syndrome (APDS): a Systematic Review. Clin Rev Allergy Immunol. 2020;59:323-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 66. | Pain CE. Juvenile-onset Behçet's syndrome and mimics. Clin Immunol. 2020;214:108381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 67. | Papadopoulou C, Omoyinmi E, Standing A, Pain CE, Booth C, D'Arco F, Gilmour K, Buckland M, Eleftheriou D, Brogan PA. Monogenic mimics of Behçet's disease in the young. Rheumatology (Oxford). 2019;58:1227-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 68. | Kim HY, Song JY, Kim WI, Ko HC, Park SE, Jang JH, Kim SH. The First Case of an Infant with Familial A20 Haploinsufficiency in Korea. J Korean Med Sci. 2020;35:e252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 69. | Krutzke S, Rietschel C, Horneff G. Baricitinib in therapy of COPA syndrome in a 15-year-old girl. Eur J Rheumatol. 2019;1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 70. | Semo Oz R, S Tesher M. Arthritis in children with LRBA deficiency - case report and literature review. Pediatr Rheumatol Online J. 2019;17:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 71. | Hou X, Qu H, Zhang S, Qi X, Hakonarson H, Xia Q, Li J. The Multi-Omics Architecture of Juvenile Idiopathic Arthritis. Cells. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 72. | Rosé CD, Wouters CH, Meiorin S, Doyle TM, Davey MP, Rosenbaum JT, Martin TM. Pediatric granulomatous arthritis: an international registry. Arthritis Rheum. 2006;54:3337-3344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 134] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 73. | Ouahed J, Spencer E, Kotlarz D, Shouval DS, Kowalik M, Peng K, Field M, Grushkin-Lerner L, Pai SY, Bousvaros A, Cho J, Argmann C, Schadt E, Mcgovern DPB, Mokry M, Nieuwenhuis E, Clevers H, Powrie F, Uhlig H, Klein C, Muise A, Dubinsky M, Snapper SB. Very Early Onset Inflammatory Bowel Disease: A Clinical Approach With a Focus on the Role of Genetics and Underlying Immune Deficiencies. Inflamm Bowel Dis. 2020;26:820-842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 130] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 74. | Nambu R, Warner N, Mulder DJ, Kotlarz D, McGovern DPB, Cho J, Klein C, Snapper SB, Griffiths AM, Iwama I, Muise AM. A Systematic Review of Monogenic Inflammatory Bowel Disease. Clin Gastroenterol Hepatol. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 74] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 75. | Nambu R, Muise AM. Advanced Understanding of Monogenic Inflammatory Bowel Disease. Front Pediatr. 2020;8:618918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 76. | Glocker EO, Kotlarz D, Klein C, Shah N, Grimbacher B. IL-10 and IL-10 receptor defects in humans. Ann N Y Acad Sci. 2011;1246:102-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 197] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 77. | Lega S, Pin A, Arrigo S, Cifaldi C, Girardelli M, Bianco AM, Malamisura M, Angelino G, Faraci S, Rea F, Romeo EF, Aloi M, Romano C, Barabino A, Martelossi S, Tommasini A, Di Matteo G, Cancrini C, De Angelis P, Finocchi A, Bramuzzo M. Diagnostic Approach to Monogenic Inflammatory Bowel Disease in Clinical Practice: A Ten-Year Multicentric Experience. Inflamm Bowel Dis. 2020;26:720-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 78. | Singhi AD, Goyal A, Davison JM, Regueiro MD, Roche RL, Ranganathan S. Pediatric autoimmune enteropathy: an entity frequently associated with immunodeficiency disorders. Mod Pathol. 2014;27:543-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 79. | Uchida T, Suzuki T, Kikuchi A, Kakuta F, Ishige T, Nakayama Y, Kanegane H, Etani Y, Mizuochi T, Fujiwara SI, Nambu R, Suyama K, Tanaka M, Yoden A, Abukawa D, Sasahara Y, Kure S. Comprehensive targeted sequencing identifies monogenic disorders in patients with early-onset refractory diarrhea. J Pediatr Gastroenterol Nutr. 2020;71:333-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 80. | Uzzan M, Ko HM, Mehandru S, Cunningham-Rundles C. Gastrointestinal Disorders Associated with Common Variable Immune Deficiency (CVID) and Chronic Granulomatous Disease (CGD). Curr Gastroenterol Rep. 2016;18:17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 87] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 81. | Fernando SL, Jang HS, Li J. The Immune Dysregulation of Common Variable Immunodeficiency Disorders. Immunol Lett. 2021;230:21-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 82. | Miyazaki H, Hoshi N, Kohashi M, Tokunaga E, Ku Y, Takenaka H, Ooi M, Yamamoto N, Uemura S, Nishimura N, Iijima K, Jimbo K, Okano T, Hoshino A, Imai K, Kanegane H, Kobayashi I, Kodama Y. A case of autoimmune enteropathy with CTLA4 haploinsufficiency. Intest Res. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 83. | Boz V, Valencic E, Girardelli M, Pin A, Gàmez-Diaz L, Tommasini A, Lega S, Bramuzzo M. Case Report: Refractory Autoimmune Gastritis Responsive to Abatacept in LRBA Deficiency. Front Immunol. 2021;12:619246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 84. | Lahner E, Conti L, Annibale B, Corleto VD. Current Perspectives in Atrophic Gastritis. Curr Gastroenterol Rep. 2020;22:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 85. | Lam-Tse WK, Batstra MR, Koeleman BP, Roep BO, Bruining MG, Aanstoot HJ, Drexhage HA. The association between autoimmune thyroiditis, autoimmune gastritis and type 1 diabetes. Pediatr Endocrinol Rev. 2003;1:22-37. [PubMed] |
| 86. | Kutluǧ Ş, Boztuǧ K, Yıldıran A. Development of multiple gallstones in a child with lipopolysaccharide-responsive beige-like anchor protein mutation. Cent Eur J Immunol. 2019;44:332-335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 87. | Kluger N, Jokinen M, Krohn K, Ranki A. Gastrointestinal manifestations in APECED syndrome. J Clin Gastroenterol. 2013;47:112-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 88. | Uche-Anya E, Lebwohl B. Celiac disease: clinical update. Curr Opin Gastroenterol. 2021;37:619-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 89. | Troncone R, Greco L, Auricchio S. Gluten-sensitive enteropathy. Pediatr Clin North Am. 1996;43:355-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 90. | Sari S, Dogu F, Hwa V, Haskologlu S, Dauber A, Rosenfeld R, Polat M, Kuloglu Z, Kansu A, Dalgic B, Ikinciogullari A. A Successful HSCT in a Girl with Novel LRBA Mutation with Refractory Celiac Disease. J Clin Immunol. 2016;36:8-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 91. | Costa-Carvalho BT, de Moraes-Pinto MI, de Almeida LC, de Seixas Alves MT, Maia RP, de Souza RL, Barreto M, Lourenço L, Vicente AM, Coutinho A, Carneiro-Sampaio M. A remarkable depletion of both naïve CD4+ and CD8+ with high proportion of memory T cells in an IPEX infant with a FOXP3 mutation in the forkhead domain. Scand J Immunol. 2008;68:85-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 92. | Glocker E, Grimbacher B. Chronic mucocutaneous candidiasis and congenital susceptibility to Candida. Curr Opin Allergy Clin Immunol. 2010;10:542-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 93. | Okada S, Asano T, Moriya K, Boisson-Dupuis S, Kobayashi M, Casanova JL, Puel A. Human STAT1 Gain-of-Function Heterozygous Mutations: Chronic Mucocutaneous Candidiasis and Type I Interferonopathy. J Clin Immunol. 2020;40:1065-1081. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 109] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 94. | Pichard DC, Freeman AF, Cowen EW. Primary immunodeficiency update: Part II. Syndromes associated with mucocutaneous candidiasis and noninfectious cutaneous manifestations. J Am Acad Dermatol. 2015;73:367-81; quiz 381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 95. | Errante PR, Perazzio SF, Frazão JB, da Silva NP, Andrade LE. Primary immunodeficiency association with systemic lupus erythematosus: review of literature and lessons learned by the Rheumatology Division of a tertiary university hospital at São Paulo, Brazil. Rev Bras Reumatol Engl Ed. 2016;56:58-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 96. | Abraham RS. How to evaluate for immunodeficiency in patients with autoimmune cytopenias: Laboratory evaluation for the diagnosis of inborn errors of immunity associated with immune dysregulation. Hematology Am Soc Hematol Educ Program. 2020;2020:661-672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 97. | Castagnoli R, Lougaris V, Giardino G, Volpi S, Leonardi L, La Torre F, Federici S, Corrente S, Cinicola BL, Soresina A, Cancrini C, Marseglia GL, Cardinale F; Immunology Task Force of the Italian Society of Pediatric Allergy and Immunology (SIAIP). Inborn errors of immunity with atopic phenotypes: A practical guide for allergists. World Allergy Organ J. 2021;14:100513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 98. | Mackay-Wiggan J, Sallee BN, Wang EHC, Sansaricq F, Nguyen N, Kim C, Chen JC, Christiano AM, Clynes R. An open-label study evaluating the efficacy of abatacept in alopecia areata. J Am Acad Dermatol. 2021;84:841-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 99. | Dai Z, Chen J, Chang Y, Christiano AM. Selective inhibition of JAK3 signaling is sufficient to reverse alopecia areata. JCI Insight. 2021;6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 100. | King B, Guttman-Yassky E, Peeva E, Banerjee A, Sinclair R, Pavel AB, Zhu L, Cox LA, Craiglow B, Chen L, Banfield C, Page K, Zhang W, Vincent MS. A phase 2a randomized, placebo-controlled study to evaluate the efficacy and safety of the oral Janus kinase inhibitors ritlecitinib and brepocitinib in alopecia areata: 24-week results. J Am Acad Dermatol. 2021;85:379-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 127] [Article Influence: 31.8] [Reference Citation Analysis (0)] |