Published online Aug 20, 2013. doi: 10.5321/wjs.v2.i3.40
Revised: April 3, 2013
Accepted: May 7, 2013
Published online: August 20, 2013
Processing time: 157 Days and 10.4 Hours
This paper describes types and characteristics of zirconia materials in relation to their applications in dentistry. The zirconia material typically used today by most manufacturers is a tetragonal polycrystalline zirconia, partially stabilized with yttrium oxide. The mechanical properties of zirconia have been extensively investigated in the scientific literature and zirconia clearly measures up to any other equivalent manufactured material. The biocompatibility of zirconia has also been extensively evaluated and no local or systemic adverse reactions or cytotoxic effects have been found in relation to it. However, ceramic bonding, ageing, light transmission and manufacturing processes are all factors that need to be further evaluated in order to guide the successful use of zirconia as a prosthetic restorative material. Milling zirconia to full-contour might be an alternative to traditionally veneered restorations. A potential adhesion mechanism appears to be the combination of air abrasion with aluminum oxide particles (silanated or not), followed by sintering with materials containing special reactive monomers. Changes in zirconia properties before and after the sintering process have also been investigated. It was found that after sintering, surface roughness was greater, and micro hardness was slightly reduced; however, accurate precision of fit was not affected by the sintering process. Currently, zirconia restorations are manufactured by either soft or hard-milling processes, with the manufacturer of each claiming advantages over the other. Chipping of the veneering porcelain is reported as a common problem and has been labeled as its main clinical setback. As zirconia has demonstrated good mechanical and biological performance, future technology is attempting to improve esthetics and minimize veneer fracture, aiming to create confidence in the dental community towards this all-ceramic system. Milling zirconia to full-contour might be an alternative to traditionally veneered restorations. Finally, implications are drawn for manufacturing, machining, and widespread use of these materials.
Core tip: Although all zirconia is chemically similar, the ultimate product can vary from manufacturer to manufacturer, with materials of varying density, uniformity homogeneity and crystalline transformation. This can be due to varying grain sizes of the powdered material ultimately affecting strength, with variations producing porosity. One type of restoration will not fit every clinical condition but today we have a range of options in zirconia ceramics, including monolithic full-contour type and conventional veneered type zirconia.
- Citation: Saridag S, Tak O, Alniacik G. Basic properties and types of zirconia: An overview. World J Stomatol 2013; 2(3): 40-47
- URL: https://www.wjgnet.com/2218-6263/full/v2/i3/40.htm
- DOI: https://dx.doi.org/10.5321/wjs.v2.i3.40
Zirconium (Zr) is a metal with the atomic number 40. It was first discovered in 1789 by the chemist Martin Klaproth[1-3]. The material has a density of 6.49 g/cm³, a melting point of 1852 °C and a boiling point of 3580 °C. It has a hexagonal crystal structure and is grayish in color. Zr does not occur in nature in a pure state. It can be found in conjunction with silicate oxide with the mineral name Zircon (ZrO2 x SiO2) or as a free oxide (Zirconia, ZrO2) with the mineral name Baddeleyite[4]. These minerals cannot be used as primary materials in dentistry because of impurities of various metal elements that affect color and because of natural radionuclides like urania and thoria, which make them radioactive[5]. Complex and time-consuming processes that result in an effective separation of these elements are necessary in order to produce pure zirconia powders. After purification the material produced can be used as a ceramic biomaterial[4,6,7].
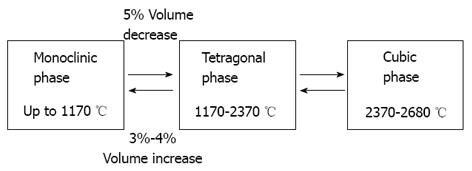
ZrO2 is a polymorphic material and occurs in three forms: monoclinic, tetragonal and cubic. The monoclinic phase is stable at room temperatures up to 1170 °C, the tetragonal at temperatures of 1170-2370 °C and the cubic at over 2370 °C[8,9]. However, noticeable changes in volume are associated with these transformations: during the monoclinic to tetragonal transformation a 5% decrease in volume occurs when zirconium oxide is heated; conversely, a 3%-4% increase in volume is observed during the cooling process[4,10] (Figure 1).
Several different oxides are added to zirconia to stabilize the tetragonal and/or cubic phases. Magnesia (MgO), Yttria (Y2O3), Calcia (CaO), and Ceria (CeO), amongst others, allow the generation of multiphase materials known as Partially Stabilized Zirconia (PSZ), whose microstructure at room temperature generally consists of cubic zirconia as the major phase, with monoclinic and tetragonal zirconia precipitates as the minor phase[4,11,12].
PSZ materials have been tested for their potential use as ceramic biomaterials. Mg-PSZ is one of the most commonly used zirconia-based engineering ceramics[13]. Residual porosity in the mass of the material, a rather coarse grain size (30-40 μm), and difficulties in obtaining Mg-PSZ precursors free of impurities, are all factors that have discouraged the interest of ceramic manufacturers in the development of Mg-PSZ for biomedical applications[4]. It has been reported that reinforcement by phase transformation toughening is less pronounced in Mg-PSZ than in Y-TZP (Yttrium-Tetragonal Zirconia Polycrystals), a finding that is discussed below[13]. Ceria (Ce)-doped zirconia ceramics have been very rarely considered, although they exhibit superior toughness (up to 20 MPa) and show no signs of ageing[14].
In the presence of a small amount of stabilizing oxides, and at room temperature, it is possible to obtain PSZ ceramics in the tetragonal phase only, known as Tetragonal Zirconia Polycrystals (TZP). The finely dispersed tetragonal ZrO2 grains within the cubic matrix, provided that they are small enough, can be maintained in a metastable state that is able to transform into the monoclinic phase[11]. Tetragonal-to-monoclinic phase transformation in zirconia can be induced by stress, temperature and surface treatments[15,16]. Low temperature ageing via phase transformation of zirconia hip joint heads in normal atmospheric conditions has been reported after 10 years of incubation[10]. After the ageing of yttrium-stabilized zirconium dioxide in body fluid or water, some tetragonal-to-monoclinic phase transformation on the surface of zirconium dioxide has also been reported[17,18]. Even though some phase transition does occur, reports indicate that the effect on the material’s mechanical properties is negligible[4,10].
The addition of approximately 2%-3% of mol yttria (Y2O3) as a stabilizing agent in zirconia allows the sintering of fully tetragonal fine-grained zirconia ceramic materials made of 100% small metastable tetragonal grains and known as Y-TZP[11].
Zirconia has mechanical properties similar to those of stainless steel. Its resistance to traction can be as high as 900-1200 MPa and its compression resistance is about 2000 MPa[4]. Cyclical load stresses are also tolerated well by this material. Applying an intermittent force of 28 kN to zirconia substrates, Cales and Stefani found that some 50 billion cycles were necessary to break the samples, but with a force in excess of 90 kN structural failure of the samples occurred after just 15 cycles[19]. Surface treatments can also modify the physical properties of zirconia. One property of zirconia that has not been well studied is the phenomenon of low-temperature degradation or “ageing”. Water and nonaqueous solvents can induce the formation of zirconiahydroxides along a crack. This process accelerates expansion of the fracture and can result in reduced strength, toughness, and density, leading to failure of the restoration[14,20-22]. Surface grinding can also reduce strength[23,24]. Kosmac et al[15] confirmed this observation and reported reduced mean strength and reliability of zirconium oxide after grinding.
Zirconia is characterized by high flexural strength and fracture toughness as a result of a physical property known as transformation toughening[4,25,26]. The incidence of framework fracture was directly related to the design of the FPD, where inlay retained FPDs (IRFPD) showed the highest failure rate[27,28]. The most common complication observed in zirconia-based restorations was fracture of the veneering porcelain, manifesting clinically as chipping fractures of the veneering ceramic with or without exposing the underlying Y-TZP framework[27]. Several factors that may affect the rate of veneering fractures have been investigated. A loss of veneering material may result from an alteration of the crystal structure of the zirconia surface during airborne-particle abrasion of the framework before the veneering process. This may result in a change of the temperature expansion coefficients[15,25]. Other factors include the different surface treatments of the frameworks and the bond strength between the veneering ceramics and zirconia frameworks[29,30].
Sintering a CAD/CAM-milled lithium disilicate layering veneer cap onto the zirconia coping has significantly increased the mechanical strength of crown restorations and represents a cost effective way of fabricating all-ceramic restorations[31]. Milling of new generation full-contour zirconia might be an alternative approach to overcome chipping fractures of veneered zirconia restorations. Fabricating mono-block restorations from pure zirconia could increase the mechanical stability and expand the range of indications[32]. However, no clinical data is available yet.
The biocompatibility of zirconia has been extensively evaluated[4,21,33]. In vitro and in vivo studies have confirmed the high biocompatibility of Y-TZP with the use of very pure zirconia powders that have been purged of their radioactive content[34-39]. No local (cellular) or systemic adverse reactions to the material were reported[4,11,35,40,41]. Recent studies have demonstrated that fewer bacteria accumulate around Y-TZP than titanium[42-44]. This could possibly be explained by different protein adsorption properties[45]. In terms of periodontal health, none of the studies reported any difference or noted any changes in the biological health of the soft and hard tissues around the zirconia-based restorations. Although some data quantified and explored differences in the biocompatibility of zirconia, no instances of gingival inflammation or periodontitis could be shown[46]. These findings have led to the suggestion that zirconium oxide may be a suitable material for manufacturing implant abutments with a low bacterial colonization potential[44].
Zirconia as implant abutment material was first introduced in 1996[47]. A randomized controlled clinical trial comparing zirconia and titanium abutments supported by 40 single implants was published[48]. After being in function for three years, 18 zirconia and 10 titanium abutments were followed-up. Both abutment materials exhibited survival rates of 100%, as well as similar biological and esthetic outcomes. In an animal study, it was shown that the collagen fiber orientation was similar around zirconia and titanium implant necks. For both materials, the fibers run parallel-oblique and parallel to the implant surface[49]. In a clinical study, a similar degree of plaque accumulation was found at zirconia and titanium abutments at three years. In the same study, when zirconia abutments are used as restoration support, there were no significant differences in bone levels between zirconia and titanium abutments after 3-year follow-up[48].
All ceramic materials more satisfactorily address the demand for esthetic restorations than metal ceramic restorations with opaque cores[50,51]. However, the translucency of the most durable zirconia-based ceramic crowns is reported to be less than that of lithium disilicate glass ceramics, for which excellent esthetic results are documented[52-56]. In-Ceram Zirconia (VITA Zahnfabrik, Bad Säckingen, Germany), an aluminum oxide-based ceramic with 35% zirconium dioxide, has a relatively low translucency, equal to that of metal ceramic crowns when evaluated using the contrast ratio method[55]. This could be an obstacle to achieving an esthetically acceptable restoration. Among nonzirconia core materials, an optimal esthetic result has been reported with Procera AllCeram (Nobel Biocare AB, Göteborg, Sweden), which is a 99.9% aluminum oxide densely sintered ceramic[57], and IPS Empress (Ivoclar Vivadent AG, Schaan, Liechtenstein) lithium disilicate glass ceramic[58]. The latter evolved in 2005 to IPS e.max Press (Ivoclar Vivadent AG), with improved translucency and mechanical properties[59,60]. Alumina and glass ceramic have, respectively, fair to high relative translucency; nevertheless, their mechanical properties are lower than ZrO2 ceramics[55,61].
Light transmission through Y-TZP varies as a function of: (1) the composition and thickness of the zirconia framework; and (2) the physical characteristics and degree of glazing of the veneering porcelain[62].
Based on this, the use of zirconia ceramics with different chemical compositions may be significant for clinicians. Additionally, measuring the degree of conversion of different resin luting agents beneath zirconia ceramic materials may produce better clinical outcomes[63]. Future studies should be expanded to include new generation full-contour zirconia[64]. Full-contour zirconia milling blanks are created through a unique patent-pending process. In one process the zirconium oxide powders are milled to further reduce the particle size of zirconium oxide, and mixed with a suitable binder to increase the compaction and density of the green state (compacted powders) and eliminate the closed porosity. The manufacturers claim that, unlike conventional high-pressure milling blank manufacture, this processing gives full-contour zirconia improved light transmission, providing a lower, more natural shade value[65].
Three main types of zirconia are available for use in clinical dentistry[66]. Although they are chemically identical, they have slightly different physical properties (e.g., porosity, density, purity, strength), which may or may not be clinically relevant. Zirconia raw material (as previously mentioned) is not a natural product, but is chemically processed from minerals. With cold isostatic pressing, the powders are shaped into ceramic pre-forms. Cold isostatic pressing is the most accepted procedural technique for shaping Y-TZP and produces stable, chalk-like non-sintered green-stage objects with a very high primary density. The green objects are further stabilized and condensed up to about 95% of the theoretical density by means of sintering without pressure in the oxidized atmosphere of a special furnace, forming pre-sintered-type oxide-ceramic blanks[11,67]. Additional compression can be achieved with Hot Isostatic Postcompaction (HIP) performed at 1000 bar and 50 °C below the sintering temperature[67]. This procedure removes residual porosity and produces dense, fully-sintered-type oxide-ceramic blanks. Carrying out HIP on Y-TZP results in a gray-black material that usually requires subsequent heat treatment to oxidize and restore whiteness[68].
Zirconia ceramics are used in dentistry as materials for frameworks, generally fabricated by means of milling the zirconia block using a CAD/CAM machine system[69-74]. Blocks can be milled either at the green stage, the pre-sintered stage or the completely sintered stage. Green-stage zirconia blocks can be milled using dry carbide burs, pre-sintered zirconia blocks can be milled using carbide burs under cooling liquid, and milling of completely sintered zirconia blocks requires the use of diamonds under cooling liquid[75]. The three available types of zirconia products are shown in Table 1 together with the milling/grinding technology used in each case.
| Milling at green stage (non-sintered) | Cercon base, Cercon (Degudent, Frankfurt, Germany) |
| Lava Frame, Lava (3M ESPE, Seefeld, Germany) | |
| Hint-ELs Zirkon TPZ-G, DigiDent (Girrbach, Pforzheim, Germany) | |
| ZirkonZahn, Steger (Steger, Brunneck, Italy) | |
| Xavex G 100 Zirkon, Etkon (Etkon, Grafelfingen, Germany) | |
| Grinding at pre-sintered stage | In-Ceram YZ Cubes, Cerec InLab (Sirona, Bensheim, Germany) |
| ZS-Blanks, Everest (KaVo, Leutkirch, Germany) | |
| Hint-ELs Zirkon TZP-W, DigiDent (Girrbach, Pforzheim, Germany) | |
| DC-Shrink, Precident DCS (DCS, Allschwil, Switzerland) | |
| LAVA All-Ceramic System (3M ESPE, Seefeld, Germany) | |
| Cercon Smart Ceramics (DeguDent, Hanau, Germany) | |
| Procera Zirconia (Nobel Biocare, Göteborg, Sweden) | |
| Grinding at completely sintered stage | DC-Zirkon, Precident DCS (DCS, Allschwil, Switzerland) |
| Z-Blanks, Everest (KaVo, Leutkirch, Germany) | |
| Zirkon TM, Pro 50, Cynovad (Cynovad, Montreal, Canada) | |
| Hint-ELs Zirkon TZP-HIP, DigiDent (Girrbach, Pforzheim, Germany) | |
| HIP Zirkon, Etkon (Etkon, Grafelfingen, Germany) |
Frameworks made from green and pre-sintered zirconia are milled in an enlarged form to compensate for the shrinkage that occurs during sintering, usually 20%-25% for a partially-sintered framework[76]. The milling process is faster and the wear and tear on hardware is less than when milling from a fully-sintered blank. The framework is subsequently post-sintered in special furnaces (at about 1500 °C) to reach the fully-sintered stage. The color of the zirconia can be individualized with the addition of oxides to the green-stage framework[68].
The question often arises as to which type of zirconia is best to use. It appears that each has advantages and disadvantages. Fully-sintered HIP zirconia has a denser polycrystalline structure with less porosity than non-HIP material, and this should translate clinically into increased resistance to fracture[77]. On the other hand, some investigators have questioned whether the amount of grinding needed during milling of fully sintered zirconia and the heat that is generated, cause surface and structural defects that can have adverse clinical implications[78]. The marginal fit of either type of material, however, is associated with very acceptable clinical results. Margin fitting of milled zirconia is as good as, if not superior to the fit of a restoration fabricated from a high noble alloy. Studies have measured the marginal gap of CAD/CAM-milled zirconia of both varieties and found it to be 40 to 70 μm[79]. However, compared to the alternative method, milling of fully sintered zirconia blocks is a time consuming process that causes greater wear of the diamond burs and is more expensive. Hence, from that point of view, green-stage zirconia could be regarded as more advantageous[67].
The longevity of an indirect restoration is closely related to the integrity of the cement at the margin[80]. Although the use of zirconia ceramics for dental applications is ongoing, the best method to achieve a durable bond between the ceramic and the tooth structure is still unknown[81]. The only consensus found in the literature is that hydrofluoric acid etching and common silane agents are not effective with zirconia ceramics[81-83].
Several studies have investigated the bond strength and the durability of various bonding methods used to form high-strength zirconia ceramics. One technique commonly used to condition the ceramic surface is that of air abrasion[77,84-86]. Air abrasion with aliminium oxide particles is routinely performed to remove layers of contaminants, thus increasing micromechanical retention between the resin cement and the restoration[80,87,88]. These particles may or may not be silica-coated (with tribochemical treatment)[89-91].
Other techniques for the superficial treatment of zirconia ceramics which have been investigated are laser, plasma spraying and fusing glass pearls to the zirconia surface[92,93]. Higher laser power settings (400-600 mJ) cause excessive material deterioration, making them unsuitable as treatments for zirconia surfaces. Irradiation with 200 mJ provides mild surface alterations, with features intermediate between the effects of air abrasion and higher laser intensities[92]. Plasma spraying and glass pearl fusion treatments were found to improve the bond strength of resin cements to the surface. However, they were not compared with conventional methods of surface treatments for Y-TZP ceramics, such as air abrasion and tribochemical coating[93].
In other studies several coating agents were used to enhance the formation of chemical bonding with zirconia but only those agents that contain a phosphate monomer agent were effective in establishing a reliable bond with zirconia materials[84,94].
A recent study focusing on the long-term stability of zirconia resin bonding shows that it is directly related to the chemistry of the materials used, including primers. The authors suggest that a more hydrophobic compound is required to better resist the detrimental effect of hydrolysis in order to gain full benefit from the primers[95-97].
In a novel approach to enhance zirconia resin bond strength, selective infiltration-etching of zirconia-based materials has been tried. This method creates a retentive surface where the adhesive resin can infiltrate and interlock in order to establish a strong and a durable bond with zirconia[98-101].
Several positive characteristics of zirconia, such as biocompatibility, color and mechanical properties, make the material suitable for use in modern dentistry. However, ceramic bonding, ageing, light transmission and manufacturing processes are all factors that need to be further evaluated in order to guide the successful use of zirconia as a prosthetic restorative material. Milling zirconia to full-contour might be an alternative to traditionally veneered restorations.
P- Reviewers Chen S, Hotta M, Payne A S- Editor Song XX L- Editor Hughes D E- Editor Lu YJ
| 1. | Denry I, Kelly JR. State of the art of zirconia for dental applications. Dent Mater. 2008;24:299-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1229] [Cited by in RCA: 993] [Article Influence: 55.2] [Reference Citation Analysis (4)] |
| 2. | Tsuge T. Radiopacity of conventional, resin-modified glass ionomer, and resin-based luting materials. J Oral Sci. 2009;51:223-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 3. | Ban S. Reliability and properties of core materials for all-ceramic dental restorations. Jpn Dent Sci Rev. 2008;44:3-21. [RCA] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 74] [Article Influence: 4.4] [Reference Citation Analysis (4)] |
| 4. | Piconi C, Maccauro G. Zirconia as a ceramic biomaterial. Biomaterials. 1999;20:1-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1872] [Cited by in RCA: 1203] [Article Influence: 46.3] [Reference Citation Analysis (7)] |
| 5. | Porstendörfer J, Reineking A, Willert HC. Radiation risk estimation based on activity measurements of zirconium oxide implants. J Biomed Mater Res. 1996;32:663-667. [PubMed] |
| 6. | Boothe GF, Stewart-Smith D, Wagstaff D, Dibblee M. The radiological aspects of zircon sand use. Health Phys. 1980;38:393-398. [PubMed] [DOI] [Full Text] |
| 7. | Christel P, Meunier A, Dorlot JM, Crolet JM, Witvoet J, Sedel L, Boutin P. Biomechanical compatibility and design of ceramic implants for orthopedic surgery. Ann N Y Acad Sci. 1988;523:234-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 63] [Article Influence: 1.7] [Reference Citation Analysis (3)] |
| 8. | Chevalier J, Gremillard L, Virkar AV, Clarke DR. The tetragonal-monoclinic transformation in zirconia: Lessons learned and future trends. J Am Ceram Soc. 2009;92:1901–1920. [DOI] [Full Text] |
| 9. | Suresh A, Mayo MJ, Porter WD, Rawn CJ. Crystallite and grain-size-dependent phase transformations in yttria-doped zirconia. J Am Ceram. 2003;86:360-362. [DOI] [Full Text] |
| 10. | Hjerppe J, Vallittu PK, Fröberg K, Lassila LV. Effect of sintering time on biaxial strength of zirconium dioxide. Dent Mater. 2009;25:166-171. [PubMed] |
| 11. | Christel P, Meunier A, Heller M, Torre JP, Peille CN. Mechanical properties and short-term in-vivo evaluation of yttrium-oxide-partially-stabilized zirconia. J Biomed Mater Res. 1989;23:45-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 320] [Article Influence: 8.9] [Reference Citation Analysis (1)] |
| 12. | De Aza AH, Chevalier J, Fantozzi G, Schehl M, Torrecillas R. Crack growth resistance of alumina, zirconia and zirconia toughened alumina ceramics for joint prostheses. Biomaterials. 2002;23:937-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 432] [Cited by in RCA: 156] [Article Influence: 6.8] [Reference Citation Analysis (2)] |
| 13. | Sundh A, Sjögren G. Fracture resistance of all-ceramic zirconia bridges with differing phase stabilizers and quality of sintering. Dent Mater. 2006;22:778-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 74] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 14. | Chevalier J. What future for zirconia as a biomaterial? Biomaterials. 2006;27:535-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 797] [Cited by in RCA: 474] [Article Influence: 23.7] [Reference Citation Analysis (1)] |
| 15. | Kosmac T, Oblak C, Jevnikar P, Funduk N, Marion L. The effect of surface grinding and sandblasting on flexural strength and reliability of Y-TZP zirconia ceramic. Dent Mater. 1999;15:426-433. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 453] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 16. | Hannink HJ, Kelly PM, Muddle BC. Transformation Toughening in Zirconia-Containing Ceramics. J Am Ceram Soc. 2000;83:461-487. [DOI] [Full Text] |
| 17. | Sato T, Shimada M. Transformation of yttria-doped tetragonal ZrO, polycrystals by annealing in water. J Am Ceram Soc. 1985;68:356-359. [DOI] [Full Text] |
| 18. | Shimizu K, Oka M, Kumar P, Kotoura Y, Yamamuro T, Makinouchi K, Nakamura T. Time-dependent changes in the mechanical properties of zirconia ceramic. J Biomed Mater Res. 1993;27:729-734. [PubMed] |
| 19. | Cales B, Stefani Y. Mechanical properties and surface analysis of retrieved zirconia femoral hip joint heads after an implantation time of two to three years. J Mater Sci Mater Med. 1994;5:376-380. [DOI] [Full Text] |
| 20. | Swab JJ. Low temperature degradation of Y-TZP materials. J Mater Sci. 1991;26:6706-6714. [DOI] [Full Text] |
| 21. | Manicone PF, Rossi Iommetti P, Raffaelli L. An overview of zirconia ceramics: basic properties and clinical applications. J Dent. 2007;35:819-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 795] [Cited by in RCA: 585] [Article Influence: 32.5] [Reference Citation Analysis (1)] |
| 22. | Lange FF, Dunlop GL, Davis BI. Degradation during aging of transformation-toughned ZrO2-Y2O3 materials at 250 °C. J Amer Ceram Soc. 1986;69:237-240. [DOI] [Full Text] |
| 23. | Işeri U, Ozkurt Z, Yalnız A, Kazazoğlu E. Comparison of different grinding procedures on the flexural strength of zirconia. J Prosthet Dent. 2012;107:309-315. [PubMed] |
| 24. | Luthardt RG, Holzhüter M, Sandkuhl O, Herold V, Schnapp JD, Kuhlisch E, Walter M. Reliability and properties of ground Y-TZP-zirconia ceramics. J Dent Res. 2002;81:487-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 176] [Article Influence: 7.7] [Reference Citation Analysis (1)] |
| 25. | Peláez J, Cogolludo PG, Serrano B, Lozano JF, Suárez MJ. A prospective evaluation of zirconia posterior fixed dental prostheses: three-year clinical results. J Prosthet Dent. 2012;107:373-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (3)] |
| 26. | Saridag S, Sevimay M, Pekkan G. Fracture Resistance of Teeth Restored With All-ceramic Inlays and Onlays: An In Vitro Study. Oper Dent. 2013;Feb 7; [Epub ahead of print]. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 27. | Al-Amleh B, Lyons K, Swain M. Clinical trials in zirconia: a systematic review. J Oral Rehabil. 2010;37:641-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 28. | Saridag S, Ozyesil AG, Pekkan G. Fracture strength and bending of all-ceramic and fiber-reinforced composites in inlay-retained fixed partial dentures. J Dent Sci. 2012;7:159-164. [RCA] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (1)] |
| 29. | Fischer J, Stawarczyk B, Hämmerle CH. Flexural strength of veneering ceramics for zirconia. J Dent. 2008;36:316-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 87] [Article Influence: 5.1] [Reference Citation Analysis (3)] |
| 30. | Hisbergues M, Vendeville S, Vendeville P. Zirconia: Established facts and perspectives for a biomaterial in dental implantology. J Biomed Mater Res B Appl Biomater. 2009;88:519-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 180] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 31. | Beuer F, Schweiger J, Eichberger M, Kappert HF, Gernet W, Edelhoff D. High-strength CAD/CAM-fabricated veneering material sintered to zirconia copings--a new fabrication mode for all-ceramic restorations. Dent Mater. 2009;25:121-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 170] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 32. | Guess PC, Att W, Strub JR. Zirconia in fixed implant prosthodontics. Clin Implant Dent Relat Res. 2012;14:633-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 96] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 33. | Stanford C, Oates T, Beirne R. Zirconia as an implant and restorative biomaterial. Int J Oral Maxillofac Implants. 2006;21:841-844 Available from: http://www.docin.com/p-399839391.html. |
| 34. | Lohmann CH, Dean DD, Köster G, Casasola D, Buchhorn GH, Fink U, Schwartz Z, Boyan BD. Ceramic and PMMA particles differentially affect osteoblast phenotype. Biomaterials. 2002;23:1855-1863. [PubMed] |
| 35. | Ichikawa Y, Akagawa Y, Nikai H, Tsuru H. Tissue compatibility and stability of a new zirconia ceramic in vivo. J Prosthet Dent. 1992;68:322-326. [PubMed] |
| 36. | Torricelli P, Verné E, Brovarone CV, Appendino P, Rustichelli F, Krajewski A, Ravaglioli A, Pierini G, Fini M, Giavaresi G. Biological glass coating on ceramic materials: in vitro evaluation using primary osteoblast cultures from healthy and osteopenic rat bone. Biomaterials. 2001;22:2535-2543. [PubMed] |
| 37. | Covacci V, Bruzzese N, Maccauro G, Andreassi C, Ricci GA, Piconi C, Marmo E, Burger W, Cittadini A. In vitro evaluation of the mutagenic and carcinogenic power of high purity zirconia ceramic. Biomaterials. 1999;20:371-376. [PubMed] |
| 38. | Burger W, Richter HG, Piconi C, Vatteroni R, Cittadini A, Boccalari M. New Y-TZP powders for medical grade zirconia. J Mater Sci Mater Med. 1997;8:113-118. [PubMed] |
| 39. | Takamura K, Hayashi K, Ishinishi N, Yamada T, Sugioka Y. Evaluation of carcinogenicity and chronic toxicity associated with orthopedic implants in mice. J Biomed Mater Res. 1994;28:583-589. [PubMed] |
| 40. | Al-Dohan HM, Yaman P, Dennison JB, Razzoog ME, Lang BR. Shear strength of core-veneer interface in bi-layered ceramics. J Prosthet Dent. 2004;91:349-355. [PubMed] |
| 41. | Josset Y, Oum’Hamed Z, Zarrinpour A, Lorenzato M, Adnet JJ, Laurent-Maquin D. In vitro reactions of human osteoblasts in culture with zirconia and alumina ceramics. J Biomed Mater Res. 1999;47:481-493. [PubMed] |
| 42. | Rimondini L, Cerroni L, Carrassi A, Torricelli P. Bacterial colonization of zirconia ceramic surfaces: an in vitro and in vivo study. Int J Oral Maxillofac Implants. 2002;17:793-798. [PubMed] |
| 43. | Welander M, Abrahamsson I, Berglundh T. The mucosal barrier at implant abutments of different materials. Clin Oral Implants Res. 2008;19:635-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 44. | Scarano A, Piattelli M, Caputi S, Favero GA, Piattelli A. Bacterial adhesion on commercially pure titanium and zirconium oxide disks: an in vivo human study. J Periodontol. 2004;75:292-296. [PubMed] |
| 45. | Milleding P, Carlén A, Wennerberg A, Karlsson S. Protein characterisation of salivary and plasma biofilms formed in vitro on non-corroded and corroded dental ceramic materials. Biomaterials. 2001;22:2545-2555. [PubMed] |
| 46. | Raigrodski AJ, Hillstead MB, Meng GK, Chung KH. Survival and complications of zirconia-based fixed dental prostheses: a systematic review. J Prosthet Dent. 2012;107:170-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 211] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 47. | Wohlwend A, Studer S, Scharer P. Das zirkonoxidabutment ein neues vollkeramisches konzept zur ästhetischen verbesserung der suprastruktur in der implantologie. Quint Zahnt. 1996;22:364-381. |
| 48. | Zembic A, Sailer I, Jung RE, Hämmerle CH. Randomized-controlled clinical trial of customized zirconia and titanium implant abutments for single-tooth implants in canine and posterior regions: 3-year results. Clin Oral Implants Res. 2009;20:802-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 152] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 49. | Tetè S, Mastrangelo F, Bianchi A, Zizzari V, Scarano A. Collagen fiber orientation around machined titanium and zirconia dental implant necks: an animal study. Int J Oral Maxillofac Implants. 2009;24:52-58. [PubMed] |
| 50. | Anusavice KJ. Phillips’ science of dental materials. 11th ed. St. Louis: Elsevier Health Sciences 2003; 655-719. |
| 51. | McLaren EA. All-ceramic alternatives to conventional metal-ceramic restorations. Compend Contin Educ Dent. 1998;19:307-308, 310, 312 passim; quiz 326. [PubMed] |
| 52. | Culp L, McLaren EA. Lithium disilicate: the restorative material of multiple options. Compend Contin Educ Dent. 2010;31:716-720, 722, 724-725. [PubMed] |
| 53. | Koutayas SO, Vagkopoulou T, Pelekanos S, Koidis P, Strub JR. Zirconia in dentistry: part 2. Evidence-based clinical breakthrough. Eur J Esthet Dent. 2009;4:348-380. [PubMed] |
| 54. | Cardoso JA, Almeida PJ, Fernandes S, Silva CL, Pinho A, Fischer A, Simões L. Co-existence of crowns and veneers in the anterior dentition: case report. Eur J Esthet Dent. 2009;4:12-26. [PubMed] |
| 55. | Heffernan MJ, Aquilino SA, Diaz-Arnold AM, Haselton DR, Stanford CM, Vargas MA. Relative translucency of six all-ceramic systems. Part I: core materials. J Prosthet Dent. 2002;88:4-9. [PubMed] |
| 56. | Heffernan MJ, Aquilino SA, Diaz-Arnold AM, Haselton DR, Stanford CM, Vargas MA. Relative translucency of six all-ceramic systems. Part I: core materials. J Prosthet Dent. 2002;88:4-9. [PubMed] |
| 57. | Odman P, Andersson B. Procera AllCeram crowns followed for 5 to 10.5 years: a prospective clinical study. Int J Prosthodont. 2001;14:504-509. [PubMed] |
| 58. | Narcisi EM. Narcisi EM. Three-unit bridge construction in anterior single-pontic areas using a metal-free restorative. Compend Contin Educ Dent. 1999;20:109-112, 114, 116-119, quiz 120. [PubMed] |
| 59. | Conrad HJ, Seong WJ, Pesun IJ. Current ceramic materials and systems with clinical recommendations: a systematic review. J Prosthet Dent. 2007;98:389-404. [PubMed] |
| 60. | Stappert CF, Guess PC, Chitmongkolsuk S, Gerds T, Strub JR. All-ceramic partial coverage restorations on natural molars. Masticatory fatigue loading and fracture resistance. Am J Dent. 2007;20:21-26. [PubMed] |
| 61. | Baldissara P, Llukacej A, Ciocca L, Valandro FL, Scotti R. Translucency of zirconia copings made with different CAD/CAM systems. J Prosthet Dent. 2010;104:6-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 154] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 62. | Hauptmann H, Suttor D, Frank S, Hoescheler H. Material properties of all ceramic zirconia prosthesis. Abstract 2910. J Dent Res. 2000;79:507. |
| 63. | Cekic-Nagas I, Egilmez F, Ergun G. Comparison of light transmittance in different thicknesses of zirconia under various light curing units. J Adv Prosthodont. 2012;4:93-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 64. | Beuer F, Stimmelmayr M, Gueth JF, Edelhoff D, Naumann M. In vitro performance of full-contour zirconia single crowns. Dent Mater. 2012;28:449-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 167] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 65. | Available from: http://www.bruxzir.com/downloads-bruxzir-zirconia-dental-crown/bruxzir-solid-zirconia-business-integration-program.pdf. |
| 66. | Angela C, Volpato M, Gustavo DL, Garbelotto A, Fredel MC, Bondiol F. Application of zirconia in dentistry: biological, mechanical and optical considerations. In: Sikalidis C. Advances in Ceramics - Electric and Magnetic Ceramics, Bioceramics, Ceramics and Environment. InTech 2011; 399-420. [DOI] [Full Text] |
| 67. | Tinschert J, Natt G, Mautsch W, Augthun M, Spiekermann H. Fracture resistance of lithium disilicate-, alumina-, and zirconia-based three-unit fixed partial dentures: a laboratory study. Int J Prosthodont. 2001;14:231-238. [PubMed] |
| 68. | Sundh A, Molin M, Sjögren G. Fracture resistance of yttrium oxide partially-stabilized zirconia all-ceramic bridges after veneering and mechanical fatigue testing. Dent Mater. 2005;21:476-482. [PubMed] |
| 69. | Gehrke P, Alius J, Fischer C, Erdelt KJ, Beuer F. Retentive Strength of Two-Piece CAD/CAM Zirconia Implant Abutments. Clin Implant Dent Relat Res. 2013;Mar 25; [Epub ahead of print]. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 56] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 70. | Hamza TA, Ezzat HA, El-Hossary MM, Katamish HA, Shokry TE, Rosenstiel SF. Accuracy of ceramic restorations made with two CAD/CAM systems. J Prosthet Dent. 2013;109:83-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 71. | Alghazzawi TF, Lemons J, Liu PR, Essig ME, Janowski GM. The failure load of CAD/CAM generated zirconia and glass-ceramic laminate veneers with different preparation designs. J Prosthet Dent. 2012;108:386-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 72. | Kypraiou V, Pelekanos S, Eliades G. Identification of monoclinic phase in CAD/CAM zirconia FPD frameworks. Eur J Esthet Dent. 2012;7:418-429. [PubMed] |
| 73. | Biscaro L, Bonfiglioli R, Soattin M, Vigolo P. An in vivo evaluation of fit of zirconium-oxide based ceramic single crowns, generated with two CAD/CAM systems, in comparison to metal ceramic single crowns. J Prosthodont. 2013;22:36-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 74. | Jalalian E, Atashkar B, Rostami R. The effect of preparation design on the fracture resistance of zirconia crown copings (computer associated design/computer associated machine, CAD/CAM system). J Dent (Tehran). 2011;8:123-129. [PubMed] |
| 75. | Witkowski S. CAD-CAM in dental technology. Quintessence Dent Technol. 2005;28:169. |
| 76. | Raigrodski AJ. Contemporary materials and technologies for all-ceramic fixed partial dentures: a review of the literature. J Prosthet Dent. 2004;92:557-562. [PubMed] |
| 77. | Keough BE, Kay HB, Sager RD. A ten-unit all-ceramic anterior fixed partial denture using Y-TZP zirconia. Pract Proced Aesthet Dent. 2006;18:37-43; quiz 44. [PubMed] |
| 78. | Luthardt RG, Holzhüter MS, Rudolph H, Herold V, Walter MH. CAD/CAM-machining effects on Y-TZP zirconia. Dent Mater. 2004;20:655-662. [PubMed] |
| 79. | Hertlein G, Hoscheler S, Frank S, Suttor D. Marginal fit of CAD/CAM manufactured all ceramic zirconia prostheses. J Dent Res. 2001;80:42-44. |
| 80. | Valandro LF, Ozcan M, Bottino MC, Bottino MA, Scotti R, Bona AD. Bond strength of a resin cement to high-alumina and zirconia-reinforced ceramics: the effect of surface conditioning. J Adhes Dent. 2006;8:175-181. [PubMed] |
| 81. | Kern M, Wegner SM. Bonding to zirconia ceramic: adhesion methods and their durability. Dent Mater. 1998;14:64-71. [PubMed] |
| 82. | Atsu SS, Kilicarslan MA, Kucukesmen HC, Aka PS. Effect of zirconium-oxide ceramic surface treatments on the bond strength to adhesive resin. J Prosthet Dent. 2006;95:430-436. [PubMed] |
| 83. | Yoshida K, Tsuo Y, Atsuta M. Bonding of dual-cured resin cement to zirconia ceramic using phosphate acid ester monomer and zirconate coupler. J Biomed Mater Res. 2006;77:28-33. [PubMed] |
| 84. | Blatz MB, Sadan A, Martin J, Lang B. In vitro evaluation of shear bond strengths of resin to densely-sintered high-purity zirconium-oxide ceramic after long-term storage and thermal cycling. J Prosthet Dent. 2004;91:356-362. [PubMed] |
| 85. | Yang B, Barloi A, Kern M. Influence of air-abrasion on zirconia ceramic bonding using an adhesive composite resin. Dent Mater. 2010;26:44-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 154] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 86. | Xie ZG, Meng XF, Xu LN, Yoshida K, Luo XP, Gu N. Effect of air abrasion and dye on the surface element ratio and resin bond of zirconia ceramic. Biomed Mater. 2011;6:065004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 87. | Wolfart M, Lehmann F, Wolfart S, Kern M. Durability of the resin bond strength to zirconia ceramic after using different surface conditioning methods. Dent Mater. 2007;23:45-50. [PubMed] |
| 88. | Foxton RM, Cavalcanti AN, Nakajima M, Pilecki P, Sherriff M, Melo L, Watson TF. Durability of resin cement bond to aluminium oxide and zirconia ceramics after air abrasion and laser treatment. J Prosthodont. 2011;20:84-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 89. | Curtis AR, Wright AJ, Fleming GJ. The influence of surface modification techniques on the performance of a Y-TZP dental ceramic. J Dent. 2006;34:195-206. [PubMed] |
| 90. | Chen L, Suh BI, Kim J, Tay FR. Evaluation of silica-coating techniques for zirconia bonding. Am J Dent. 2011;24:79-84. [PubMed] |
| 91. | Thompson JY, Stoner BR, Piascik JR, Smith R. Adhesion/cementation to zirconia and other non-silicate ceramics: where are we now? Dent Mater. 2011;27:71-82. [PubMed] |
| 92. | Cavalcanti AN, Pilecki P, Foxton RM, Watson TF, Oliveira MT, Gianinni M, Marchi GM. Evaluation of the surface roughness and morphologic features of Y-TZP ceramics after different surface treatments. Photomed Laser Surg. 2009;27:473-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 4.9] [Reference Citation Analysis (2)] |
| 93. | Derand T, Molin M, Kvam K. Bond strength of composite luting cement to zirconia ceramic surfaces. Dent Mater. 2005;21:1158-1162. [PubMed] |
| 94. | Wegner SM, Kern M. Long-term resin bond strength to zirconia ceramic. J Adhes Dent. 2000;2:139-147. [PubMed] |
| 95. | Mirmohammadi H, Aboushelib MN, Salameh Z, Feilzer AJ, Kleverlaan CJ. Innovations in bonding to zirconia based ceramics: Part III. Phosphate monomer resin cements. Dent Mater. 2010;26:786-792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 96. | Aboushelib MN, Matinlinna JP, Salameh Z, Ounsi H. Innovations in bonding to zirconia-based materials: Part I. Dent Mater. 2008;24:1268-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 97. | Tanaka R, Fujishima A, Shibata Y, Manabe A, Miyazaki T. Cooperation of phosphate monomer and silica modification on zirconia. J Dent Res. 2008;87:666-670. [PubMed] |
| 98. | Aboushelib MN, Feilzer AJ, Kleverlaan CJ. Bonding to zirconia using a new surface treatment. J Prosthodont. 2010;19:340-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 69] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 99. | Komine F, Blatz MB, Matsumura H. Current status of zirconia-based fixed restorations. J Oral Sci. 2010;52:531-539. [PubMed] |
| 100. | Aboushelib MN. Evaluation of zirconia/resin bond strength and interface quality using a new technique. J Adhes Dent. 2011;13:255-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 101. | Aboushelib MN, Kleverlaan CJ, Feilzer AJ. Selective infiltration-etching technique for a strong and durable bond of resin cements to zirconia-based materials. J Prosthet Dent. 2007;98:379-388. [PubMed] |