Published online Aug 12, 2014. doi: 10.5318/wjo.v4.i3.56
Revised: May 21, 2014
Accepted: July 12, 2014
Published online: August 12, 2014
Processing time: 177 Days and 19.6 Hours
Macular edema is one of the most common vision-threatening complications of uveitis noted in one third of patients with uveitis. The release of a number of inflammatory mediators induces retinal vascular hyperpermeability leading to uveitic macular edema (UME) which most commonly is of cystoid shape. Fluorescein angiography and non-invasive spectral-domain optical coherence tomography are standard procedures for diagnosis and follow-up of UME with some innovations such as scanning laser ophthalmoscope retro-mode imaging. Effective management of UME requires thorough understanding of the individual case. Proper control of intraocular inflammation is mandatory before targeting macular edema itself. Mainstay of treatment is immunosuppressive therapy with various drug delivery routes including topical, local subconjunctival, peribulbar and sub-Tenon’s, intravitreal and systemic. Clinical trials with biologics are under way to study the efficacy of these agents in suppressing intraocular inflammation and resolution of UME. Visual prognosis in UME depends on numerous factors. Younger age and better visual acuity at baseline are associated with more favorable visual outcome in most studies
Core tip: Cystoid macular edema is among leading causes of visual loss in patients with uveitis. Inflammatory cytokines such as interferon-gamma, interleukin-2, interleukin-10, tumor necrosis factor-alpha and prostaglandins are powerful inflammatory mediators which along with the vascular endothelial growth factor are potent mediators of increased vascular permeability in uveitic macular edema. Scanning laser ophthalmoscope in retro-mode is a novel imaging modality that can show each cystoid space located in any layer of the retina and allows the detection of the extent of cystoid macular edema.
- Citation: Shoughy SS, Kozak I. Updates in uveitic macular edema. World J Ophthalmol 2014; 4(3): 56-62
- URL: https://www.wjgnet.com/2218-6239/full/v4/i3/56.htm
- DOI: https://dx.doi.org/10.5318/wjo.v4.i3.56
Macular edema is one of the most common vision-threatening complications of uveitis. It can affect patients with different types of ocular inflammation[1,2]. Cystoid macular edema (CME), the most common structural type of uveitic macular edema, was found to be the most important cause of both blindness and visual impairment among patients with uveitis, it was noted in 33% of all uveitis patients[3,4]. Visual loss due to cystoid macular edema in patients with uveitis, occurs predominantly in older patients with chronic uveitis[5]. Chronic macular edema has a significant influence on the quality of life of the patients, this is especially important as it tends to affect young people, often between 30 and 50 years of age[6,7].
In adults, cystoid macular edema is the leading cause of visual loss in patients with uveitis. However, the incidence of inflammatory CME in children seems to be lower and it is still the third leading cause of visual loss after macular scars and secondary glaucoma[8]. Macular edema in patients with uveitis was found to account for 41% of visual impairment and 29% of blindness[9]. In this short review we summarize current updates on pathophysiology, diagnosis and treatment of uveitic macular edema.
Effective management of uveitic macular edema requires thorough understanding of the underlying mechanisms of its formation. However, the pathogenesis of uveitic macular edema is not completely understood. Under normal conditions, the fluid volume and content of the macula is controlled by the blood retinal barriers and the pump function of the retinal pigment epithelial cells. The blood retinal barriers are composed of the inner retinal barrier formed by tight junctions of the endothelial cells lining the retinal capillaries and the outer retinal barrier formed by tight junctions between retinal pigment epithelial cells[8]. Most commonly, macular edema results from abnormal hyperpermeability of retinal blood vessels. Among the various tight junction molecules in blood vessel wall, downregulation of occludin has been reported most consistently in the context of blood-retina barrier (BRB) breakdown as well as modulation of aquaporins and dysregulation of caveolar transport[10]. This increase in vascular permeability leads to extravasation of fluid, proteins and other macromolecules into the retinal interstitium[11]. The release of a number of inflammatory mediators induces retinal vascular hyperpermeability. These inflammatory cytokines include interferon-gamma, interleukin-2, interleukin-10 and tumor necrosis factor-alpha[2]. Prostaglandins are powerful lipid derived inflammatory mediators which are generated from the phospholipids in the cell membrane[12,13].
Vascular endothelial growth factor (VEGF) was found to be a potent mediator of increased vascular permeability[14]. Interestingly, it was noted that patients with uveitis and CME have higher concentrations of vascular endothelial growth factor in the aqueous humor as compared with those without CME[15]. Another important factor that contributes to increased vascular leakage is the endothelial damage induced by adherence of leukocyte to the vessel walls, a phenomenon termed leukostasis which is mediated by nitric oxide, adhesion molecules, and other inflammatory mediators[16,17]. The dysfunction of the BRB may not explain the mechanism of macular edema in all cases. Other possible factors that may contribute to the occurrence of maculopathy include the presence of active inflammation, macular or choroidal ischaemia (as a result of active vasculitis), and vitreoretinal traction. Accordingly, treatment of persistent uveitic macular edema will be more successful if the underlying pathogenic mechanisms are properly addressed[18].
The release and diffusion of cytokines may have the predominant role in case of acute inflammation, but the exact factors and events responsible for the development of chronic macular edema in the setting of controlled inflammation have not yet been clearly identified[5]. However, persistence of CME might be secondary to previous inflammatory insults to the retinal pigment epithelium, blood-retina barrier, and persistent cytokines[19]. Leakage from the optic nerve, which is often present in uveitis, may also contribute to the development of persistent macular edema[20,21].
Leakage was found to be amplified by factors that affect the integrity of the retinal blood vessels such as vasodilatation, increased intraluminal pressure, and increased blood flow. Hence, patients with concurrent cardiovascular disease, hypertension, diabetes, or hyperlipidemia have an increased risk of developing macular edema and when present it tends to be more persistent[22]. Smoking was noted to be a risk factor for cystoid macular edema in cases with intermediate uveitis[23,24]. Recently, it was found that two functional genetic variants of interferon regulatory factor 5 (IRF5) may play a role in the development of macular edema in non-anterior uveitis patients through regulation of induction of type I interferon[25].
The presence of macular edema can be detected clinically in cases with clear media. However, biomicroscopic evaluation of macular edema may be difficult when the amount of the fluid and the anatomical changes are minimal. In addition, it is required to have ways to document the extent of the macular edema in order to monitor the progression of macular edema following different treatment modalities.
Fluorescein angiography is a conventional method for the assessment of UME. It is particularly valuable to assess the retinal vascular integrity and to characterize the area of the foveal avascular zone. Fluorescein angiography can also show leakage around optic nerve head which is a common finding in cases with uveitis[26]. The drawbacks of fluorescein angiography include the invasive nature and the need of the contrast with its potential side effects. Furthermore, the interpretation of the fluorescein angiograms might not be easy in the presence of extensive areas of hemorrhage or exudates[27].
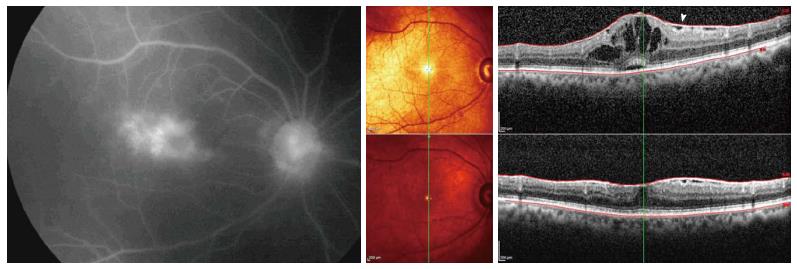
Optical coherence tomography is an effective diagnostic modality for detection of macular edema which produces B-scan cross sectional images of the retinal layers that are comparable to histopathology specimens. It not only allows the determination of the distribution of fluid within the retinal layers but also allows quantification of retinal thickness particularly in patients with CME[28,29]. Three patterns of macular edema were noted in patients with uveitis studied by optical coherence tomography: diffuse macular edema, cystoid macular edema, and serous retinal detachment[29]. In a recent report from the Multicenter Uveitis Steroid Treatment trial, macular edema was associated with impaired visual acuity. Different phenotypes of macular edema were associated with different degrees of visual impairment: cystoid changes without retinal thickening were associated with moderately impaired visual acuity (-5 ETDRS letters), but visual acuity was worse in eyes with retinal thickening (-13 letters) and with both cysts and thickening (-19 letters). Uveitis was also associated with impaired visual field sensitivity, but eyes with macular edema had even worse visual field sensitivity[30].
Epiretinal membrane coexists in a significant percentage of patients with uveitis and may be associated with persistence of macular edema. OCT is helpful in detection and characterization of uveitic ERM[29].
Several studies evaluated the agreement between fluorescein angiography and optical coherence tomography results for the diagnosis of macular edema in patients with uveitis. Optical coherence tomography and fluorescein angiography were found to offer only moderate agreement regarding macular edema status in patients with uveitis, probably because each imaging modality might demonstrate related but nonidentical macular pathologic features. In four hundred seventy-nine eyes with uveitis from 255 patients, macular leakage was present in 40% of cases free of macular thickness with OCT, whereas macular thickness was present in 34% of cases without macular leakage[31]. Because of its lower cost, greater safety, and greater likelihood of obtaining usable information, OCT may be the best initial and follow-up test for evaluation of suspected macular edema. However, obtaining the second test after negative results of the first seems justified when detection of macular leakage or macular thickness would alter management[31]. Both FA and high-resolution OCT are highly sensitive techniques and correlate well in detection of ME. However, there is a small chance that when each test performed alone it might miss existing subtle ME[32]. Therefore, FA and OCT are complementary investigations, each revealing different aspects of the pathophysiology of uveitic ME[33].
The retro-mode of the scanning laser ophthalmoscope is a new method of detecting abnormalities in the retina. It uses an infrared laser and an aperture with a modified central stop that is displaced laterally from the confocal light path. This optical arrangement allows for a clearer and pseudo-3-dimensional image[34]. Scanning laser ophthalmoscope in the retro-mode can show each cystoid space located in any layer of the retina and allows the detection of the extent of cystoid macular oedema[35,36].
Chronic macular edema may lead to permanent loss of vision if not properly treated. It is associated with damage to photoreceptors by ischemia and might lead to retinal thinning and fibrosis[2]. There are no guidelines or consensus on when and how to treat uveitic macular edema and the evidence strength for treatment of macular edema in uveitis is overall low[37]. Macular edema associated with active inflammation requires immediate intervention. Several treatment options exist to address macular edema. The approach used depends on several factors including the laterality of disease, the response to therapy and the side effects of the proposed medication. Management should start with an attempt to treat the underlying cause and control of the ocular inflammation.
Topical therapy for treatment of uveitic macular edema includes corticosteroid and non-steroidal anti-inflammatory drugs (NSAIDs). Treatment with steroids and NSAIDs has been shown to inhibit the release of the inflammatory mediators and was found to decrease vascular permeability[38]. There was no significant difference in the results of treatment in the studies comparing topical NSAIDs with corticosteroids[38].
In the absence of vitreoretinal traction, the administration of indomethacin 0.5% eye drops four times per day in eyes affected with uveitic ME from different etiologies, compared with placebo, was associated with a significant reduction in ME at the 6-mo follow-up visit, as measured by spectral-domain optical coherence tomography[39].
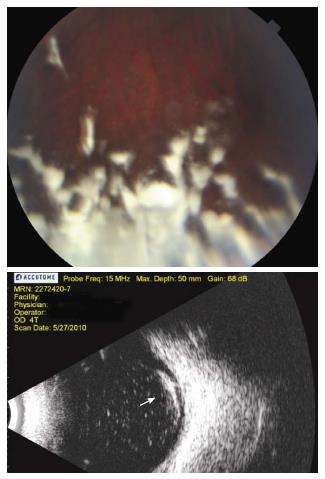
In addition to topical drops and systemic medications, there are various drug delivery routes to treat UME. Local treatment includes injections given subconjuctivally or in the sub-Tenon space, intravitreal injections of drugs and intraocular implantation devices. The advantage of all these is effective delivery of the drug to the proximity of target tissue. Resistant cases of uveitic CME require higher macular concentrations of corticosteroid agents; this can be usually achieved with local therapy such as posterior sub-Tenon injection[10]. Intravitreal triamcinolone acetonide allows high steroid concentration to act locally for maximal effect and duration (Figures 1 and 2). Although intravitreal triamcinolone was found to be often effective in reducing CME, it may not always be effective in improving visual acuity, likely because of pre-existing or long-standing macular damage[40].
Intraocular steroid sustained-delivery device implantation is a relatively new treatment approach for patients requiring frequent intravitreal triamcinolone acetonide injections or chronic treatment with systemic corticosteroids and/or immunosuppressive agents. The Retisert (fluocinolone acetone; Bausch and Lomb Place, Rochester, NY, United States) implant is a non-biodegradable implant, whereas the Ozurdex (dexamethasone; Allergan, Irvine, CA, United States) is biodegradable implant[41,42]. The accumulated effect of repeat dexamethasone pellet implantations was found to improve retinal thickness and resolve ocular inflammation, resulting in restoration of ocular function[43]. Potential complications of all forms of local steroid delivery include increased intraocular pressure and cataract progression[44-46].
Intravitreal injections of anti-VEFG were shown to be useful and therapeutically beneficial in refractory uveitic CME. Intravitreal bevacizumab was found to be associated with anatomic and visual improvement in uveitis patients with CME resistant to medical therapy that persists despite control of the uveitis[19]. Ranibizumab is an antibody fragment which neutralizes all VEGF isoforms and bioactive fragments which also demonstrated a significant improvement in visual acuity and a reduction in macular edema[47,48]. Intravitreal adalimumab was shown in some studies to be of help for refractory uveitis-related macular edema[49]. Local injection therapy can be associated with rare complications. Endophthalmitis and rhegmatogenous retinal detachments have been reported with intravitreal injections of anti-VEFG performed[50,51].
Intravitreal NSAIDs were also evaluated in patients with refractory uveitic cystoid macular edema. Intravitreal injection of diclofenac insignificantly reduced central macular thickness but this was not associated with visual improvement[52].
Several systemic treatment options exist for treating uveitic macular edema including systemic corticosteroids, systemic NSAIDs, systemic immunomodulators, biologic agents and RPE pump inhibitors. Oral steroids are usually reserved to treat patients with significant vision-threatening uveitis as they are associated with systemic side effects[53]. On the other hand, systemic NSAIDs were found to have a limited role, if any, in the treatment of inflammatory cystoid macular edema[54]. Systemic immunomodulator drugs have been found to be effective in the management of uveitic macular edema. Treatment with mycophenolate mofetil may lead to resolution of CME and improve the mean BCVA in patients with uveitis[55,56].
Several biologic agents were evaluated for UME. Intravenous infliximab was found to improve visual acuity and decrease macular thickness in patients with chronic cystoid macular edema associated with uveitis[57]. Efalizumab is an intercellular adhesion molecule inhibitor that was reported as a potential therapy to improve visual acuity and reduce macuar thickness for refractory uveitic macular edema[58]. Acetazolamide, an RPE pump stimulator, may be useful for chronic CME in uveitis. However, the effect is better in cases with quiescent uveitis than in those with chronically active disease[59]. Intravitreal adalimumab showed no efficacy in improving best-corrected visual acuity or reducing central retinal thickness in patients with chronic uveitic macular edema[49].
Pars plana vitrectomy may have a role in the management of selected cases with uveitic macular edema. Clearing vitreous cavity decreases burden of circularing inflammatory cytokines which may contribute to persistence of UME. In eyes with vitreous adhesions and macular traction, vitrectomy surgery with removal of all vitreous adhesions may results in good anatomic and visual outcomes. In a prospective, interventional, randomized, controlled study of 23 eyes of 23 patients, the mean visual acuity in the surgical group improved significantly from logMAR 1.0 (± 0.62) at baseline to 0.55 (± 0.29) at 6 mo (P = 0.011), with 5 (42%) eyes reaching vision of 20/40 or better. CME after vitrectomy improved in the fluorescein angiogram in 4 (33%) eyes, remained unchanged in 7 (58%) eyes and deteriorated in 1 (8%) eye[60]. In addition, vitrectomy has an influence on the efficacy of triamcinolone acetonide injectable solution. In a retrospective review of 20 eyes, it was found that, after intravitreal triamcinolone injection for chronic CME, the mean visual acuity at last follow-up showed statistically significant improvement in non-vitrectomized eyes compared to the almost unaltered mean visual acuity for vitrectomized eyes[61]. A recent study with limited follow-up has shown that treatment with dexamethasone intravitreal implant injection for uveitic macular edema in vitrectomized eyes was associated with favorable visual outcomes and had an acceptable safety profile[62].
Visual prognosis in UME depends on numerous factors. A study reported longitudinal outcomes after 48 mo median follow-up period. Visual acuity at the final follow-up improved in 69%, was deteriorated in 19%, and remained unchanged in 12% of eyes. Younger age and better visual acuity at baseline were associated with more favorable visual outcome. Optical coherence tomography documentation of improvement or total resolution of UME was observed in 77% at the final follow-up[63].
In conclusion, effective management of uveitic macular edema requires thorough understanding of the underlying mechanisms. Proper control of intraocular inflammation is mandatory before targeting macular edema itself. Various diagnostic and therapeutic approaches exist for treatment and monitoring of uveitic macular edema.
P- Reviewer: Arevalo JF, Issa SA, Machida S S- Editor: Song XX L- Editor: A E- Editor: Lu YJ
| 1. | Guex-Crosier Y. The pathogenesis and clinical presentation of macular edema in inflammatory diseases. Doc Ophthalmol. 1999;97:297-309. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 2. | Okhravi N, Lightman S. Cystoid macular edema in uveitis. Ocul Immunol Inflamm. 2003;11:29-38. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 47] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 3. | Rothova A, Suttorp-van Schulten MS, Frits Treffers W, Kijlstra A. Causes and frequency of blindness in patients with intraocular inflammatory disease. Br J Ophthalmol. 1996;80:332-336. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 597] [Cited by in F6Publishing: 630] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 4. | Lardenoye CW, van Kooij B, Rothova A. Impact of macular edema on visual acuity in uveitis. Ophthalmology. 2006;113:1446-1449. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 182] [Cited by in F6Publishing: 195] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 5. | Rothova A. Inflammatory cystoid macular edema. Curr Opin Ophthalmol. 2007;18:487-492. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 62] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 6. | Kiss CG, Barisani-Asenbauer T, Maca S, Richter-Mueksch S, Radner W. Reading performance of patients with uveitis-associated cystoid macular edema. Am J Ophthalmol. 2006;142:620-624. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 7. | Hazel CA, Petre KL, Armstrong RA, Benson MT, Frost NA. Visual function and subjective quality of life compared in subjects with acquired macular disease. Invest Ophthalmol Vis Sci. 2000;41:1309-1315. [PubMed] [Cited in This Article: ] |
| 8. | de Boer J, Steijaert A, van den Bor R, Stellato R, Ossewaarde-van Norel J. Development of Macular Edema and Impact on Visual Acuity in Uveitis Associated with Juvenile Idiopathic Arthritis. Ocul Immunol Inflamm. 2014;Jan 10; Epub ahead of print. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Levin MH, Pistilli M, Daniel E, Gangaputra SS, Nussenblatt RB, Rosenbaum JT, Suhler EB, Thorne JE, Foster CS, Jabs DA. Incidence of visual improvement in uveitis cases with visual impairment caused by macular edema. Ophthalmology. 2014;121:588-595.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 44] [Cited by in F6Publishing: 47] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 10. | Klaassen I, Van Noorden CJ, Schlingemann RO. Molecular basis of the inner blood-retinal barrier and its breakdown in diabetic macular edema and other pathological conditions. Prog Retin Eye Res. 2013;34:19-48. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 402] [Cited by in F6Publishing: 471] [Article Influence: 42.8] [Reference Citation Analysis (1)] |
| 11. | Johnson MW. Etiology and treatment of macular edema. Am J Ophthalmol. 2009;147:11-21.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 120] [Cited by in F6Publishing: 125] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 12. | Ahuja M, Dhake AS, Sharma SK, Majumdar DK. Topical ocular delivery of NSAIDs. AAPS J. 2008;10:229-241. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 147] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 13. | McColgin AZ, Heier JS. Control of intraocular inflammation associated with cataract surgery. Curr Opin Ophthalmol. 2000;11:3-6. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 71] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Senger DR, Connolly DT, Van de Water L, Feder J, Dvorak HF. Purification and NH2-terminal amino acid sequence of guinea pig tumor-secreted vascular permeability factor. Cancer Res. 1990;50:1774-1778. [PubMed] [Cited in This Article: ] |
| 15. | Fine HF, Baffi J, Reed GF, Csaky KG, Nussenblatt RB. Aqueous humor and plasma vascular endothelial growth factor in uveitis-associated cystoid macular edema. Am J Ophthalmol. 2001;132:794-796. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 84] [Cited by in F6Publishing: 89] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 16. | Leal EC, Manivannan A, Hosoya K, Terasaki T, Cunha-Vaz J, Ambrósio AF, Forrester JV. Inducible nitric oxide synthase isoform is a key mediator of leukostasis and blood-retinal barrier breakdown in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2007;48:5257-5265. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 165] [Cited by in F6Publishing: 178] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 17. | Miyamoto K, Khosrof S, Bursell SE, Rohan R, Murata T, Clermont AC, Aiello LP, Ogura Y, Adamis AP. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc Natl Acad Sci USA. 1999;96:10836-10841. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 488] [Cited by in F6Publishing: 539] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 18. | Dick AD. The treatment of chronic uveitic macular oedema. Br J Ophthalmol. 1994;78:1-2. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Cordero Coma M, Sobrin L, Onal S, Christen W, Foster CS. Intravitreal bevacizumab for treatment of uveitic macular edema. Ophthalmology. 2007;114:1574-1579.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Pruett RC, Brockhurst J, Letts NF. Fluorescein angiography of peripheral uveitis. Am J Ophthalmol. 1974;77:448-453. [PubMed] [Cited in This Article: ] |
| 21. | van Kooij B, Probst K, Fijnheer R, Roest M, de Loos W, Rothova A. Risk factors for cystoid macular oedema in patients with uveitis. Eye (Lond). 2008;22:256-260. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 23] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | van Kooij B, Fijnheer R, Roest M, Rothova A. Trace microalbuminuria in inflammatory cystoid macular edema. Am J Ophthalmol. 2004;138:1010-1015. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 23. | Lin P, Loh AR, Margolis TP, Acharya NR. Cigarette smoking as a risk factor for uveitis. Ophthalmology. 2010;117:585-590. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 24. | Thorne JE, Daniel E, Jabs DA, Kedhar SR, Peters GB, Dunn JP. Smoking as a risk factor for cystoid macular edema complicating intermediate uveitis. Am J Ophthalmol. 2008;145:841-846. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Márquez A, Cénit MC, Cordero-Coma M, Ortego-Centeno N, Adán A, Fonollosa A, Díaz Valle D, Pato E, Blanco R, Cañal J. Two functional variants of IRF5 influence the development of macular edema in patients with non-anterior uveitis. PLoS One. 2013;8:e76777. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Gürlü VP, Alimgil ML, Esgin H. Fluorescein angiographic findings in cases with intermediate uveitis in the inactive phase. Can J Ophthalmol. 2007;42:107-109. [PubMed] [DOI] [Cited in This Article: ] |
| 27. | Jittpoonkuson T, Garcia PM, Rosen RB. Correlation between fluorescein angiography and spectral-domain optical coherence tomography in the diagnosis of cystoid macular edema. Br J Ophthalmol. 2010;94:1197-1200. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 33] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 28. | Tran TH, de Smet MD, Bodaghi B, Fardeau C, Cassoux N, Lehoang P. Uveitic macular oedema: correlation between optical coherence tomography patterns with visual acuity and fluorescein angiography. Br J Ophthalmol. 2008;92:922-927. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 72] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 29. | Markomichelakis NN, Halkiadakis I, Pantelia E, Peponis V, Patelis A, Theodossiadis P, Theodossiadis G. Patterns of macular edema in patients with uveitis: qualitative and quantitative assessment using optical coherence tomography. Ophthalmology. 2004;111:946-953. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 158] [Cited by in F6Publishing: 146] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 30. | Taylor SR, Lightman SL, Sugar EA, Jaffe GJ, Freeman WR, Altaweel MM, Kozak I, Holbrook JT, Jabs DA, Kempen JH. The impact of macular edema on visual function in intermediate, posterior, and panuveitis. Ocul Immunol Inflamm. 2012;20:171-181. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 31. | Kempen JH, Sugar EA, Jaffe GJ, Acharya NR, Dunn JP, Elner SG, Lightman SL, Thorne JE, Vitale AT, Altaweel MM. Fluorescein angiography versus optical coherence tomography for diagnosis of uveitic macular edema. Ophthalmology. 2013;120:1852-1859. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 54] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 32. | Kozak I, Morrison VL, Clark TM, Bartsch DU, Lee BR, Falkenstein I, Tammewar AM, Mojana F, Freeman WR. Discrepancy between fluorescein angiography and optical coherence tomography in detection of macular disease. Retina. 2008;28:538-544. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 33. | Ossewaarde-van Norel J, Camfferman LP, Rothova A. Discrepancies between fluorescein angiography and optical coherence tomography in macular edema in uveitis. Am J Ophthalmol. 2012;154:233-239. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 34. | Suzuma K, Tsuiki E, Matsumoto M, Fujikawa A, Kitaoka T. Retro-mode imaging of fibrovascular membrane in proliferative diabetic retinopathy after intravitreal bevacizumab injection. Clin Ophthalmol. 2011;5:897-900. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 35. | Yamamoto M, Mizukami S, Tsujikawa A, Miyoshi N, Yoshimura N. Visualization of cystoid macular oedema using a scanning laser ophthalmoscope in the retro-mode. Clin Experiment Ophthalmol. 2010;38:27-36. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 36. | Lee WJ, Lee BR, Shin YU. Retromode imaging: Review and perspectives. Saudi J Ophthalmol. 2014;28:88-94. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 37. | Davis J. Current concepts in the management of uveitic macular edema. Adv Stud Ophthalmol. 2010;7:60-66. [Cited in This Article: ] |
| 38. | Cho H, Madu A. Etiology and treatment of the inflammatory causes of cystoid macular edema. J Inflamm Res. 2009;2:37-43. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 39. | Allegri P, Murialdo U, Peri S, Carniglia R, Crivelli MG, Compiano S, Autuori S, Mastromarino A, Zurria M, Marrazzo G. Randomized, double-blind, placebo-controlled clinical trial on the efficacy of 0.5% indomethacin eye drops in uveitic macular edema. Invest Ophthalmol Vis Sci. 2014;55:1463-1470. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 40. | Conti SM, Kertes PJ. The use of intravitreal corticosteroids, evidence-based and otherwise. Curr Opin Ophthalmol. 2006;17:235-244. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 41. | Callanan DG, Jaffe GJ, Martin DF, Pearson PA, Comstock TL. Treatment of posterior uveitis with a fluocinolone acetonide implant: three-year clinical trial results. Arch Ophthalmol. 2008;126:1191-1201. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 196] [Cited by in F6Publishing: 209] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 42. | Kuppermann BD, Blumenkranz MS, Haller JA, Williams GA, Weinberg DV, Chou C, Whitcup SM. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol. 2007;125:309-317. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 280] [Cited by in F6Publishing: 300] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 43. | Tomkins-Netzer O, Taylor SR, Bar A, Lula A, Yaganti S, Talat L, Lightman S. Treatment with Repeat Dexamethasone Implants Results in Long-Term Disease Control in Eyes with Noninfectious Uveitis. Ophthalmology. 2014;. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 89] [Cited by in F6Publishing: 96] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 44. | Jea SY, Byon IS, Oum BS. Triamcinolone-induced intraocular pressure elevation: intravitreal injection for macular edema and posterior subtenon injection for uveitis. Korean J Ophthalmol. 2006;20:99-103. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 45. | Friedman DS, Holbrook JT, Ansari H, Alexander J, Burke A, Reed SB, Katz J, Thorne JE, Lightman SL, Kempen JH. Risk of elevated intraocular pressure and glaucoma in patients with uveitis: results of the multicenter uveitis steroid treatment trial. Ophthalmology. 2013;120:1571-1579. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 46. | Lowder C, Belfort R, Lightman S, Foster CS, Robinson MR, Schiffman RM, Li XY, Cui H, Whitcup SM. Dexamethasone intravitreal implant for noninfectious intermediate or posterior uveitis. Arch Ophthalmol. 2011;129:545-553. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 452] [Cited by in F6Publishing: 479] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 47. | Karim R, Tang B. Use of antivascular endothelial growth factor for diabetic macular edema. Clin Ophthalmol. 2010;4:493-517. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 48. | Acharya NR, Hong KC, Lee SM. Ranibizumab for refractory uveitis-related macular edema. Am J Ophthalmol. 2009;148:303-309.e2. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 53] [Cited by in F6Publishing: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 49. | Androudi S, Tsironi E, Kalogeropoulos C, Theodoridou A, Brazitikos P. Intravitreal adalimumab for refractory uveitis-related macular edema. Ophthalmology. 2010;117:1612-1616. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 71] [Cited by in F6Publishing: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 50. | Fintak DR, Shah GK, Blinder KJ, Regillo CD, Pollack J, Heier JS, Hollands H, Sharma S. Incidence of endophthalmitis related to intravitreal injection of bevacizumab and ranibizumab. Retina. 2008;28:1395-1399. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 151] [Cited by in F6Publishing: 175] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 51. | Meyer CH, Michels S, Rodrigues EB, Hager A, Mennel S, Schmidt JC, Helb HM, Farah ME. Incidence of rhegmatogenous retinal detachments after intravitreal antivascular endothelial factor injections. Acta Ophthalmol. 2011;89:70-75. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 76] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 52. | Ramezani A, Fard Esmaeilpour N, Eskandari A, Rabbanikhah Z, Soheilian R, Soheilian M. Intravitreal diclofenac for refractory uveitic cystoid macular edema. J Ophthalmic Vis Res. 2013;8:47-52. [PubMed] [Cited in This Article: ] |
| 53. | Karim R, Sykakis E, Lightman S, Fraser-Bell S. Interventions for the treatment of uveitic macular edema: a systematic review and meta-analysis. Clin Ophthalmol. 2013;7:1109-1144. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 43] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 54. | van Kooij B, De Boer J, Ten Dam N, Fijnheer R, Rothova A. The effect of non-steroidal anti-inflammatory drugs on inflammatory cystoid macular edema. Am J Ophthalmol. 2005;140:563-564. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 55. | Neri P, Mariotti C, Cimino L, Mercanti L, Giovannini A. Long-term control of cystoid macular oedema in noninfectious uveitis with Mycophenolate Mofetil. Int Ophthalmol. 2009;29:127-133. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 39] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 56. | Doycheva D, Zierhut M, Blumenstock G, Stuebiger N, Deuter C. Mycophenolate mofetil in the therapy of uveitic macular edema--long-term results. Ocul Immunol Inflamm. 2012;20:203-211. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 57. | Markomichelakis NN, Theodossiadis PG, Pantelia E, Papaefthimiou S, Theodossiadis GP, Sfikakis PP. Infliximab for chronic cystoid macular edema associated with uveitis. Am J Ophthalmol. 2004;138:648-650. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 127] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 58. | Wang J, Ibrahim M, Turkcuoglu P, Hatef E, Khwaja A, Channa R, Do DV, Nguyen QD. Intercellular adhesion molecule inhibitors as potential therapy for refractory uveitic macular edema. Ocul Immunol Inflamm. 2010;18:395-398. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 59. | Schilling H, Heiligenhaus A, Laube T, Bornfeld N, Jurklies B. Long-term effect of acetazolamide treatment of patients with uveitic chronic cystoid macular edema is limited by persisting inflammation. Retina. 2005;25:182-188. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | Tranos P, Scott R, Zambarakji H, Ayliffe W, Pavesio C, Charteris DG. The effect of pars plana vitrectomy on cystoid macular oedema associated with chronic uveitis: a randomised, controlled pilot study. Br J Ophthalmol. 2006;90:1107-1110. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 48] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 61. | Androudi S, Letko E, Meniconi M, Papadaki T, Ahmed M, Foster CS. Safety and efficacy of intravitreal triamcinolone acetonide for uveitic macular edema. Ocul Immunol Inflamm. 2005;13:205-212. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 56] [Cited by in F6Publishing: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 62. | Adán A, Pelegrín L, Rey A, Llorenç V, Mesquida M, Molins B, Ríos J, Keller J. Dexamethasone intravitreal implant for treatment of uveitic persistent cystoid macular edema in vitrectomized patients. Retina. 2013;33:1435-1440. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 72] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 63. | Tranos PG, Tsaousis KT, Vakalis AN, Asteriades S, Pavesio CE. Long-term follow-up of inflammatory cystoid macular edema. Retina. 2012;32:1624-1628. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |