Revised: March 21, 2013
Accepted: April 10, 2013
Published online: May 6, 2013
Processing time: 131 Days and 19.3 Hours
The graft vs tumor effect produced by the infusion of allo-reactive lymphocytes is considered to be the main mechanism of action in the eradication of tumor cells only reported in allogeneic stem cell transplantation. We present a case of a lymphoma patient infused with his collected bystander lymphocytes from is stem cell autograft after failing to collect enough stem cells to proceed with autologous stem cells transplantation, resulting in tumor response with no treatment related toxicity. This case illustrates the concept of autologous lymphocyte infusion, suggesting the possibility of an autograft vs tumor effect, as an effort to parallel donor lymphocyte infusion in allogeneic stem cell transplantation to create a graft vs tumor effect by increasing donor lymphocytes in the patient.
Core tip: This case report illustrated the possibility of an autograft vs tumor effect by infusion autologous lymphocytes collected during stem cell collection without the side effect graft vs host disease. The infusion of autologous lymphocytes produced tumor regression in our case, suggesting that this treatment modality might benefit lymphoma patients.
- Citation: Porrata LF, Markovic SN. Autologous lymphocytes infusion. World J Hematol 2013; 2(2): 59-61
- URL: https://www.wjgnet.com/2218-6204/full/v2/i2/59.htm
- DOI: https://dx.doi.org/10.5315/wjh.v2.i2.59
The clinical evidence of graft vs tumor effect reported in the allogeneic stem cell transplantation setting has been recognized based on indirect observations: (1) anecdotal reports showing the sudden withdrawal of immuno-suppression in relapsed patients post-allogeneic stem cell transplantation can re-established complete remission; (2) increased relapse rates in patients receiving syngeneic marrow graft compared with recipients of allogeneic grafts; (3) decreased relapse rates post-allogeneic stem cell transplantation in association with graft-vs-host disease; (4) T-cell depletion of an allogeneic graft increases the risk of relapse; and (5) donor lymphocyte infusion induces complete remission after relapse following allogeneic stem cell transplantation[1]. In contrast to allogeneic stem cell transplantation, the high-dose chemotherapy used in autologous stem cell transplantation is considered to be the sole mechanism of action to eradicate resistant tumor clones that escaped standard chemotherapy because of the presumed lack of an autologous graft (autograft) vs tumor effect that parallels the graft vs tumor effect reported in allogeneic stem cell transplantation[1]. However, our published studies reporting that the recovery of absolute lymphocyte count post-autologous stem cell transplantation is associated with superior clinical outcomes without the detrimental effects of graft vs host disease suggests the possibility of an autograft vs tumor effect[2-4]. Furthermore, the discovery that early absolute lymphocyte recovery post-autologous stem cell transplantation, as surrogate marker of host immunity in autologous stem cell transplantation, is directly dependent on the absolute numbers of infused bystander lymphocytes (immune effector cells) harvested during CD34+ stem cells collection argues that the manipulation of the immunocompetent effector cells in the autograft can affect not only immune recovery but also clinical outcomes in the post-autologous stem cell transplantation[3]. Thus, the infusion of collected “bystander” lymphocytes from a stem cell autograft correlates with lymphocyte recovery and clinical outcomes in lymphoma patients undergoing autologous stem cell transplantation. Herein, we report a case of a lymphoma patient that failed to collect enough stem cell to proceed to autologous stem cell transplantation, but received his collected bystander autologous lymphoma resulting in tumor regression.
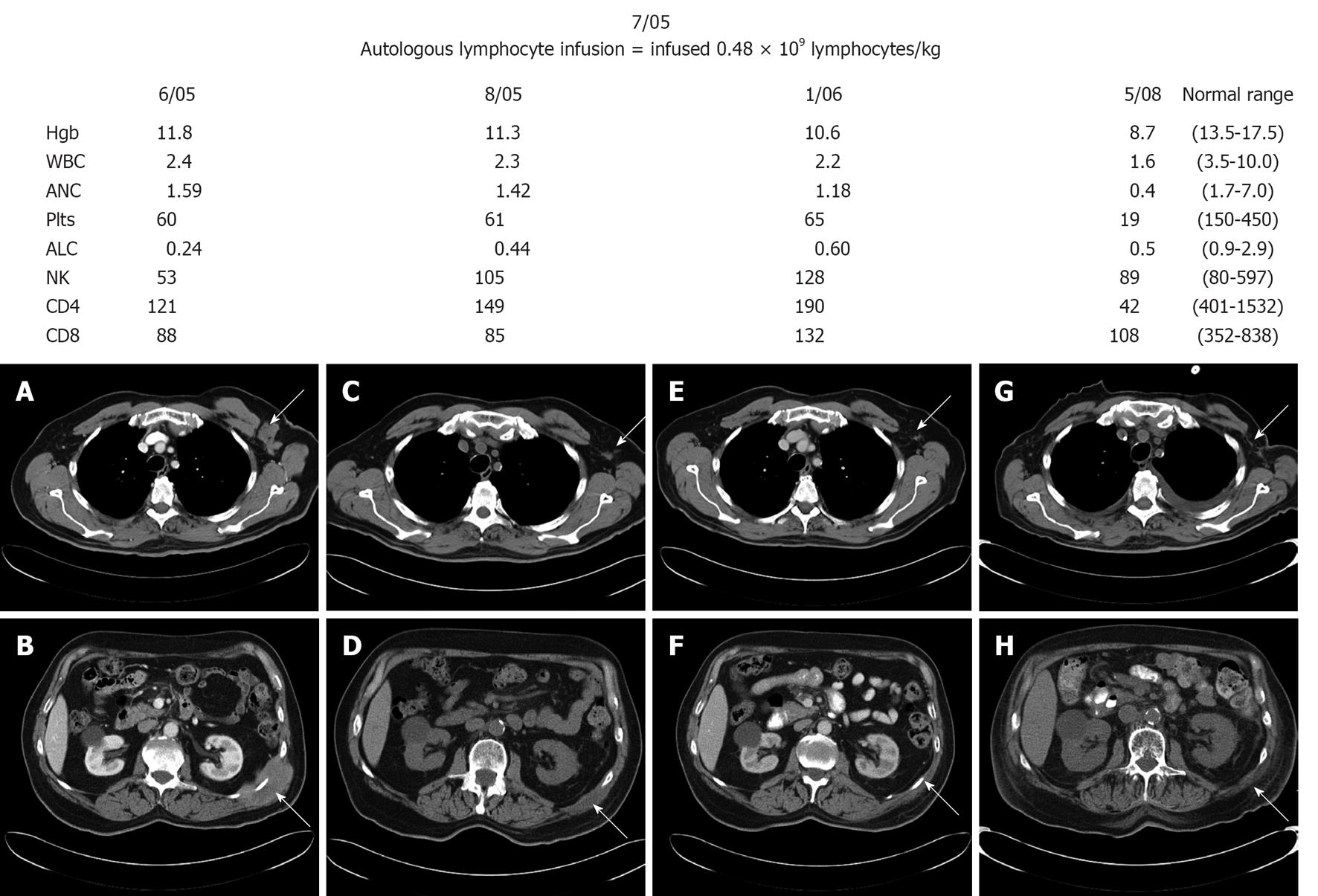
In November 2000, a 70-year-old man presented with worsening abdominal pain, distension, heartburn and early satiety. Patient was evaluated by local physician who identified an abdominal mass measuring 6.0 cm by 5.0 cm. Computer tomography scan demonstrated an abdominal mass measuring 11.0 cm by 7.0 cm arising from the pancreas into the mesenteric. Needle biopsy of the mesenteric mass revealed diffuse large B-cell lymphoma (DLBCL) is Eastern Cooperative Oncology Group performance status was 1, bone marrow biopsy was negative, lactate dehydrogenase of 297 U/L, and his stage was IIE. He achieved a complete remission after six cycles of cyclophosphamide, adriamycin, vincristine, and prednisone chemotherapy in February 2001. In August 2001, the patient presented with hypercalcemia (12.8 mg/dL), and left axillary adenopathy. A bone marrow biopsy demonstrated 80% of DLBCL lymphoma involvement. Salvage chemotherapy with ifosfamide, etoposide and carboplatin was initiated resolving the lymphadenopathy, hypercalcemia and repeated bone marrow biopsy demonstrated 5% lymphoma involvement. In February 2002, patient preceded to peripheral blood stem cell collection. The patient only collected 1.03 × 106/kg stem cells through is peripheral blood; 2.0 × 106 cells/kg is the minimum required for autologous stem cell transplantation. The patient did not proceed to autologous stem cell transplantation due to his low stem cell numbers. The decision was to observe the patient and he did well. In June 2005 he developed new flank pain and a computer tomography scan (Figure 1A and B) demonstrated new masses: left axillary mass of 3.5 cm × 2.6 cm and left flank mass of 8.4 cm × 5.4 cm. Biopsy of the left flank mass revealed DLBCL. The patient refused any further cytotoxic chemotherapy but was willing to consider non-cytotoxic approaches. We discussed with him the option of infusing his autograft lymphocytes that were collected at the same time of his stem cell collection, based on our previous publications in autologous stem cell transplantation that recovery of lymphocytes improves clinical outcomes in lymphomas[2-4]. In July 2005 patient received 0.48 × 109 lymphocytes/kg. The infusion of autologous lymphocytes was approved by our Institutional Review Board and patient consented also for the procedure. The patient did not experience any side effects or toxicities from the autologous lymphocytes infusion. His absolute lymphocyte count prior to the lymphocyte infusion was 0.24 × 109/L. After the infusion, the absolute lymphocyte count improved to 0.44 × 109/L (Figure 1).
His peripheral blood lymphocyte subsets showed normalization of natural killer (NK) cells (Figure 1). Patient received interleukin-2 million units three times a week for 6 wk. Follow-up computer tomography scan in August 2005 showed improvement of the left axillary adenopathy (1.4 cm × 0.5 cm) and the left flank mass (4.3 cm × 1.8 cm) (Figure 1C and D) and subsequent scan on January 2006 (Figure 1E and F) and May 2008 (Figure 1G and H) continued to showed resolution of the masses. In addition, patient’s NK cells count remained in the normal range after the autologous lymphocyte infusion. In May 2008, patient showed pancytopenia and bone marrow biopsy revealed therapy-related myelodysplastic syndrome with refractory cytopenia with multilineage dysplasia and documented monosomy 7 secondary to prior chemotherapy treatment with cyclophosphamide, carboplatin, and etoposide. The bone marrow biopsy was negative for involvement for lymphoma. In September 5, 2008 the patient succumbed to complications from myelodysplastic syndrome with no recurrence of his DLBCL.
To our knowledge; the infusion of autologous bystander lymphocytes from the stem cell autograft in a non-transplant setting has never been reported. We present a case of a lymphoma patient who failed to collect sufficient stem cells for autologous stem cell transplantation. The stored autograft was infused in an attempt to halt lymphoma progression by increasing lymphocyte counts, as an effort to parallel a “donor lymphocyte infusion” of allograft recipients to create “graft-vs-tumor effect” by increasing donor lymphocyte counts in the patient. After the autologous lymphocyte infusion, our patient showed increment of his absolute lymphocyte count with normalization of his NK cells. NK cells have been associated as the key lymphocytes affecting clinical outcomes in lymphoma patients undergoing autologous stem cell transplantation[5]. Patient remained in remission in concordance with normal peripheral blood NK cells counts (Figure 1). A limitation of this case report was the lack of reporting the absolute lymphocyte subsets infused to the patient. From our previous publication the infusion of 0.5 × 106 lymphocytes/kg was associated with faster lymphocyte recovery post-autologous stem cell transplantation. Our patient received 0.48 × 106 lymphocyte/kg resulting in the normalization of the peripheral blood NK cells observed just 1 mo after her infusion of his autologous lymphocytes. Patient did not develop any side effects from the autologous lymphocyte infusion and he did not develop any signs or symptoms of autologous graft verus host disease. Due to the immunosuppressive effects of the tumor, higher number of non-cytotoxic NK cells and overexpression of inhibitory NK cells receptors have been observed in conjunction low numbers of cytotoxic NK cells and reduced expression of activating receptors ailing tumor progression. In our case, the NK cells collected in the autograft where collected when the patient was in complete remission. Thus, it is reasonable to assume that infused NK cells have not been tampered by the immunosuppressive effects of the lymphoma leading to the recognition and eradication of the lymphoma in our case[6]. Our intervention resulted in a dramatic and sustained treatment response with no treatment related toxicity, suggesting the concept of autograft vs tumor effect.
P- Reviewers Shah JJ, Yamakawa M S- Editor Gou SX L- Editor A E- Editor Zheng XM
| 1. | Porrata LF, Litzow MR, Inwards DJ, Gastineau DA, Moore SB, Pineda AA, Bundy KL, Padley DJ, Persky D, Ansell SM. Infused peripheral blood autograft absolute lymphocyte count correlates with day 15 absolute lymphocyte count and clinical outcome after autologous peripheral hematopoietic stem cell transplantation in non-Hodgkin’s lymphoma. Bone Marrow Transplant. 2004;33:291-298. [PubMed] |
| 2. | Porrata LF, Markovic SN. Autograft mediated adoptive immunotherapy of cancer in the context of autologous stem cell transplantation. World J Clin Oncol. 2010;1:29-34. [PubMed] |
| 3. | Porrata LF, Markovic SN. Timely reconstitution of immune competence affects clinical outcome following autologous stem cell transplantation. Clin Exp Med. 2004;4:78-85. [PubMed] |
| 4. | Porrata LF, Gertz MA, Inwards DJ, Litzow MR, Lacy MQ, Tefferi A, Gastineau DA, Dispenzieri A, Ansell SM, Micallef IN. Early lymphocyte recovery predicts superior survival after autologous hematopoietic stem cell transplantation in multiple myeloma or non-Hodgkin lymphoma. Blood. 2001;98:579-585. [PubMed] |
| 5. | Porrata LF, Inwards DJ, Ansell SM, Micallef IN, Johnston PB, Gastineau DA, Litzow MR, Winters JL, Markovic SN. Early lymphocyte recovery predicts superior survival after autologous stem cell transplantation in non-Hodgkin lymphoma: a prospective study. Biol Blood Marrow Transplant. 2008;14:807-816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 125] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 6. | Levy EM, Roberti MP, Mordoh J. Natural killer cells in human cancer: from biological functions to clinical applications. J Biomed Biotechnol. 2011;2011:676198. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 135] [Article Influence: 9.6] [Reference Citation Analysis (0)] |