Published online Dec 18, 2015. doi: 10.5312/wjo.v6.i11.935
Peer-review started: April 2, 2015
First decision: July 10, 2015
Revised: September 2, 2015
Accepted: October 1, 2015
Article in press: October 8, 2015
Published online: December 18, 2015
Processing time: 260 Days and 1.6 Hours
Various early-onset spinal deformities, particularly infantile and juvenile scoliosis (JS), still pose challenges to pediatric orthopedic surgeons. The ideal treatment of these deformities has yet to emerge, as both clinicians and surgeons still face multiple challenges including preservation of thoracic motion, spine and cage, and protection of cardiac and lung growth and function. Elongation-derotation-flexion (EDF) casting is a technique that uses a custom-made thoracolumbar cast based on a three-dimensional correction concept. EDF can control progression of the deformity and - in some cases-coax the initially-curved spine to grow straighter by acting simultaneously in the frontal, sagittal and coronal planes. Here we provide a comprehensive review of how infantile and JS can affect normal spine and thorax and how serial EDF casting can be used to manage these spinal deformities. A fresh review of the literature helps fully understand the principles of the serial EDF casting technique and the effectiveness of conservative treatment in patients with early-onset spinal deformities, particularly infantile and juvenile scolisois.
Core tip: Infantile and juvenile scoliosis still pose challenges to pediatric orthopedic surgeons. Elongation-derotation-flexion (EDF) casting is a technique that uses a custom-made thoracolumbar cast based on a three-dimensional correction concept. EDF can control progression of the deformity and-in some cases-coax the initially-curved spine to grow straighter by acting simultaneously in the frontal, sagittal and coronal planes. A critical literature review is needed in order to understand the principles of the technique and the effectiveness of conservative treatment using the specific EDF casting technique in young and very young patients.
- Citation: Canavese F, Samba A, Dimeglio A, Mansour M, Rousset M. Serial elongation-derotation-flexion casting for children with early-onset scoliosis. World J Orthop 2015; 6(11): 935-943
- URL: https://www.wjgnet.com/2218-5836/full/v6/i11/935.htm
- DOI: https://dx.doi.org/10.5312/wjo.v6.i11.935
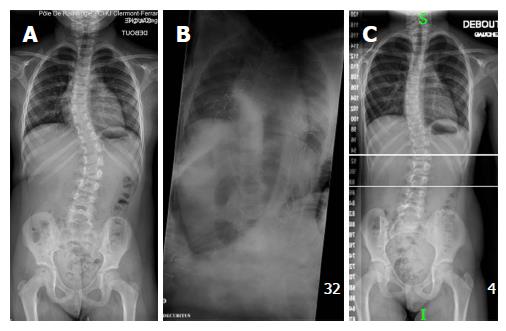
Scoliosis is a three-dimensional deformity of the spine with lateral, anteroposterior and rotational components. In most cases, scoliosis is idiopathic and concerns adolescents (AS), but there are rarer cases of infantile (IS) and juvenile scoliosis (JS) where onset is prior to age 5 and 10 years, respectively. On top of age at onset, IS and JS also present differently to AS in epidemiology, rate of deformity progression, and associated anomalies[1,2].
In young and very young patients, a progressive spinal deformity interferes with the regular and harmonious growth of the vertebral column and thoracic cage. In this subgroup of patients, the distorted spinal growth will lead to an underdeveloped thoracic cage, a short trunk and height, and a disproportionate body habitus[1,2]. IS and JS therefore rank among the most challenging conditions in pediatric orthopedics, as pathologic changes induced on a growing organism by an early-onset spinal deformity can prove dramatic, even leading to death in the most severe cases[2-6].
The goal of any treatment is to correct as soon as possible all distortions secondary to altered spinal growth: underdeveloped thoracic cage, short trunk and height, disproportionate body habitus, low weight, respiratory and cardiac dysfunctions. Tachypnea, dyspnea, chronic obstructive pulmonary disease, tracheomalacia, tachycardia and weight loss often prove more worrisome than the deformity of the spine itself[2,4,5,7,8].
The ideal treatment for early-onset scoliosis (IS and JS in particular) has not yet been identified. In particular, care providers (surgeons, pediatricians, pneumologists, anesthesiologists, physiotherapists) still have to overcome multiple challenges including preservation of thoracic motion, spine and cage, and protection of cardiac and lung growth and function[2,4,6-8].
Arthrodesis carried out in the thoracic spine at an early age is unable to control the negative effects of the deformity on thoracic cage shape, lung parenchyma development or preservation of cardio-pulmonary function. Early spine fusion is not the solution when progressive spinal deformities occurring in young and very young patients have to be managed. Moreover, early arthrodesis of the thoracic spine, is a cause of respiratory insufficiency and adds loss of pulmonary function to the pre-existing spinal deformity[2,6-8].
Elongation-derotation-flexion (EDF) casting is a technique that uses a custom-made thoracolumbar cast based on a three-dimensional correction concept. EDF can control progression of the deformity and-in some cases-coax the initially-curved spine to grow straighter by acting simultaneously in the frontal, sagittal and coronal planes[9,10]. In particular, the EDF cast can be used as a “positive” force to influence spinal growth[10] as it counteracts the negative effects of the spinal deformity on the future spinal development of the growing organism[4,9,10].
Here we provide a comprehensive review of how infantile and JS can affect normal spine and thorax and how serial EDF casting can be used to manage these spinal deformities. A fresh review of the literature helps fully understand the principles of the serial EDF casting technique and the effectiveness of conservative treatment in children with IS and JS.
The first five to ten years of life are a critical period for thoracic cage and spinal column development and growth[2-5,8]. This period is marked by a series of complex phenomena that follow in very rapid succession, and these events need to be well synchronized to preserve harmonious limb and spine relationships, as growth does not progress at the same rate in the different body segments[4,8].
Alterations of the spinal column development and growth can lead to a malformation or deformity. As a result, standing and sitting height, thoracic cage dimensions, and lung development are negatively affected. Patients with progressive early-onset spinal deformity will experience a reduction of longitudinal trunk growth and a loss of normal trunk proportions, leading to disproportionate body habitus. Moreover, in severe scoliosis, growth tends to become asymmetrical due to growth plate disorganization. As a result, this disorganization of the growth plate leads to progressive changes of vertebral bodies’ anatomy. Those changes are responsible for secondary thoracic cage shape modifications, reduction of thoracic motility and altered lung growth[1,2]. This succession of events is similar to a domino. A “domino effect” means that not only is spinal growth affected but size and shape of the thoracic cage are modified as well. This modification of the thoracic cage will subsequently interfere with lung development. Constriction of the thoracic cage as a result of a spinal deformity significantly restricts lung growth and can contribute to serious pulmonary complications such as thoracic insufficiency syndrome, Cor pulmonale and, in the most severe cases, death[2,4-7].
It has been reported that the loss of vital capacity in patients with untreated early onset spinal deformities is about 15% greater compared than adolescent idiopathic scoliosis patients. Pehrsson et al[11] analyzed the mortality and causes of death in 115 untreated scoliosis patients. They compared their findings to the rates expected according to official Swedish statistics, and found that mortality was significantly higher in IS and JS but not AS[11]. In addition, Karol et al[7] reported that a thoracic spine height of at least 18 to 22 cm is necessary to reduce the risk of severe respiratory insufficiency[2,5,7].
The North-American orthopedic surgeon Joseph Risser introduced the cast frame that bears his name to treat scoliosis with casting. The technique is based on the principles of elongation and derotation in order to correct the spinal curvature[12].
In 1964, French surgeons Cotrel and Morel[9] improved the Risser technique by adding the third dimension, called flexion, and subsequently popularized the EDF casting technique.
Between 1975 and 2000, United Kingdom physician Min Mehta modified Cotrel and Morel’s original EDF technique and introduced the concept of serial casting. Under Metha’s protocol, EDF casts are changed every 8 to 12 wk under general anesthesia[9,10].
EDF stands for elongation, derotation, and flexion. It is a method of orthopedic reduction of the scoliotic deformity on a specific reduction frame by traction, postero-lateral compression and rotation, the application of a thoraco-lumbo-sacral plaster cast, and, finally, lateral manual compression[9].
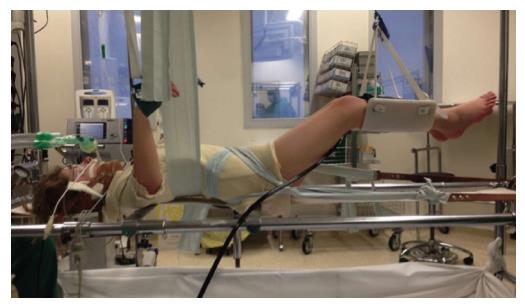
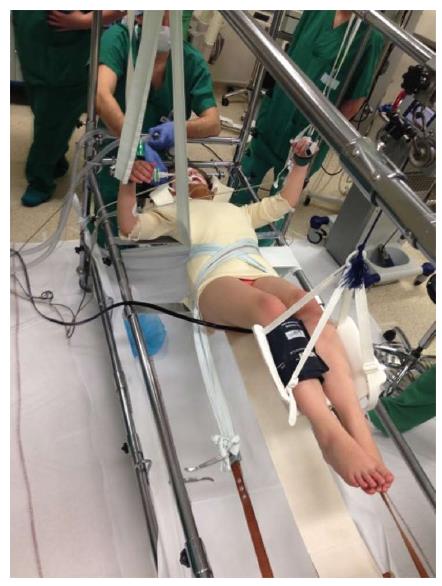
The reduction apparatus is the Cotrel frame that is used to realize an axial correction of the spine, with chin and occiput providing the proximal point of traction and iliac crests providing the distal point of traction. The technique employs harnesses and straps, and the amount of traction is controlled[9] (Figures 1 and 2).
The EDF cast can be performed under general anesthesia[13] or, alternatively, with the patient awake[14]. Both options are effective[13-15]. Canavese et al[13] recently reported that serial EDF casting done under general anesthesia and neuromuscular-blocking drugs improves outcome in patients with JS, specifically by enabling more effective control of curve progression in IS and JS patients compared to EDF casting under general anesthesia alone or no anesthesia. Canavese et al[13] hypothesized that complete muscle relaxation helps the surgeon better derotate the spine while straightening it. Moreover, adding neuromuscular-blocking drugs to the protocol improves immediate outcome and slows curve progression at 24-mo follow-up[13].
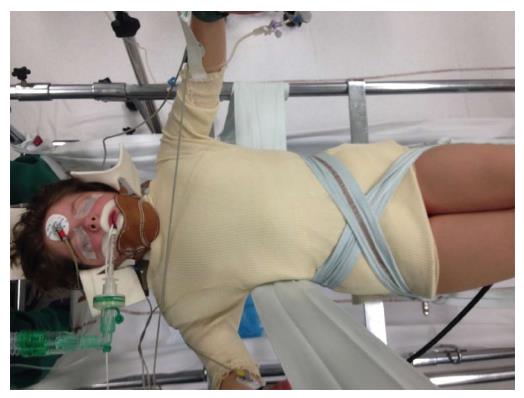
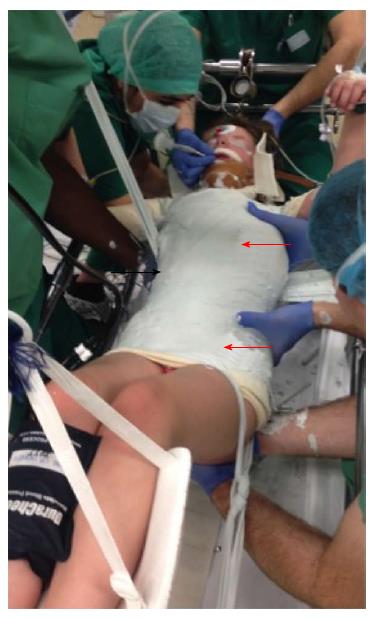
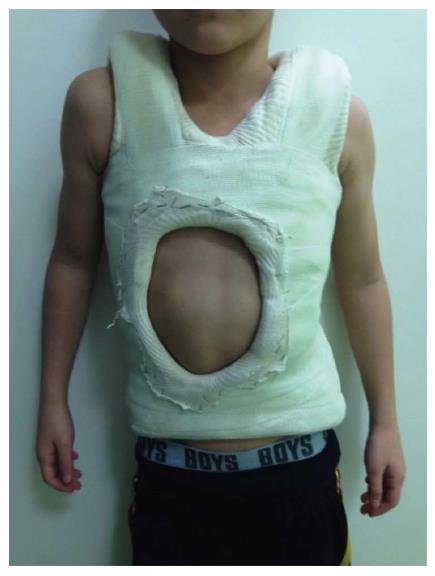
The patient lay on two horizontal metal bars, one supporting the shoulders and the other supporting the pelvis. A strap wrapped around the patient on the convex side of the scoliotic curve is tensioned to reduce it by simultaneously applying a lateral and a posterior reduction force. In cases presenting an “S” curve, a second strap can be applied in the opposite side and direction, level with this second convexity (Figure 3). The two support bars can then be removed to apply the plaster over the straps, making sure it is well-molded, especially at the iliac crests. While the plaster is still malleable, one-hand lateral pressure is applied on the convexity side and two-hand counter-pressure is applied on the concavity side as close as possible to the end vertebrae, and both pressures are maintained until the plaster hardens (Figure 4). Next, a few layers of synthetic fiberglass are added to reinforce the cast, and a thoraco-abdominal window is cut to enable decompression of the anterior abdomen (stomach and bowels) and better expansion of the thoracic cage (Figure 5). The casts are subsequently changed every 8-12 wk.
At first sight, the cast may look like a constrictive force on the thorax, limiting its expansion, but in fact, if well-molded, it does not compress the thoracic cage and allows comfortable respiratory movement (Figure 6). Dhawale et al[16] reviewed data from 37 serial EDF casts to investigate the effects of casting on ventilation in IS, and found that casting led to only transient pulmonary restriction: peak inspiratory pressure increased by 106% at cast application but fell back to near-baseline values once the thoraco-abdominal straps were cut free[13].
The big challenge for the growing spine is to preserve thoracic cage, thoracic spine and lung development without altering spine and thoracic cage motion. Morbidity tied to surgical procedures has sparked a comeback of conservative treatment, i.e., serial EDF casting.
EDF casting is a non-surgical option that can be considered in the management of patients with early-onset spinal deformities, particularly IS and JS. The key to successful treatment is to anticipate curve progression, recognize poor prognosis and apply prophylactic treatment, i.e., serial EDF casting. Factors influencing management and outcome are numerous and heterogeneous in nature: age, curve magnitude, cardiac function, respiratory impairment, nutritional status, neurological examination, presence of pain, loss of function, and patient’s and/or parents’ concerns over self-esteem due to spine and chest deformity.
Serial EDF casting can be used as a delaying tactic, to stop further progression of the deformity for a few years until to definitive fusion, or as a definitive treatment option. Patients aggressively casted before age 20 mo for curves averaging 30 degrees go on to present zero net progression and/or up to a 10-degree reduction in scoliosis at skeletal maturity[9], whereas children undergoing cast treatment after age 30 mo for curves averaging 50 degrees did not go on to gain significant correction, although their spinal curvature did not progress[10]. Fletcher et al[15] confirmed previous findings, especially in children with moderate spinal deformities, and concluded that serial casting can contribute to postpone surgery in selected cases. van Hessem et al[14] recently found that serial casting is also effective for the management of JS patients. They showed that serial casting can stop curve progression and even eliminate the need for surgery[13].
The main advantage of EDF casting is that the spine is left alone. The EDF cast can control the progression of the deformity and-in some cases-coax the initially-curved spine to grow straighter by acting simultaneously in the frontal, sagittal and coronal planes[9,10]. In particular, the EDF cast can be used as a “positive” force to influence spinal growth[10] as it counteracts the negative effects of the spinal deformity on future spinal development of the growing organism[3,8,9]. Serial EDF casting plays a central role in delaying or in some cases even eliminating the need for growth-sparing surgery[10,15], but it is not effective in all patients with early-onset scoliosis. However, unlike surgery, it does not potentially interfere with spine and thoracic cage growth. In particular, it has been shown that fitting growth-sparing devices near and/or on the spine can alter spinal growth and lead to auto-lusion of ribs and vertebral bodies[4,6,7,10]. These changes will contribute to make definitive fusion more challenging with less satisfactory outcomes[17,18].
Serial EDF casting should be considered as a valuable low-risk treatment strategy for patients with IS and JS[15,16,19,20]. The learning curve is rapid, reported rate of complications is low, and most reported complications are relatively minor[9,10,12,16,19]. Potential side-effects of serial EDF casting include dry or itchy skin, skin rash or irritation, blisters, weak muscles, joint pain or stiffness after the cast is removed, and sleep problems (cast intolerance)[20-22].
As reported by Sanders et al[21], the most important issue is temporary procedural chest pressure making ventilation difficult while the cast is setting. However, as soon as the anterior or antero-lateral window is open, respiratory airflow drops back to normal. Sanders et al[21] suggest intubating patients undergoing EDF casting under general anesthesia. Dhawale et al[16] reported that peak inspiratory pressure increased due to transient pulmonary restriction at cast application but fell back to near-baseline values once the window was cut way. They concluded that patients with underlying pulmonary disease are at risk of respiratory complications during the casting process, making it necessary to follow casting with a proper period of observation[16].
Badlani et al[19] reported a subclavian vein thrombosis after EDF cast application for the treatment of progressive infantile kyphoscoliosis. This is the only published case in the English literature. Badlani et al[19] stressed that early and accurate diagnosis of this complication allowed effective treatment and avoided further morbidity for the patient, prompting them to suggest that clinicians performing EDF casting in young patients should be aware of the possibility of this rare complication and know how to quickly diagnose and treat it[19].
The ideal treatment of IS and JS has not been identified. Management strategies must consider the patient’s full lifespan as well as the effects of treatment on spinal and chest growth of a patient with early-onset scoliosis.
Advancements in growth-friendly surgical procedures now offer orthopedic surgeons an array of treatment options for children with IS and JS, including distraction-based techniques (dual-growing rods, magnetic growing rods, vertebral expandable prosthetic titanium ribs), compression-based techniques (vertebral body stapling) and growth-guided techniques (Luque trolley, Shilla technique). However, due to a lack of evidence-based research, the ideal treatment for patients with early-onset scoliosis has not been yet found. Moreover, a significant variation among surgeon’ surgical strategies has been reported[23].
In most cases, growth-sparing surgery is not an isolated intervention and is peppered with numerous complications. Reported rates of surgical complications ranges from 8% to 50%: reported complications include skin problems, wound complications, device migration and/or fractures, anesthetic complications, hardware failure, auto-fusion, infections, and decompensation. Mackenzie et al[24] reported that surgical site infection rate in patients with infantile deformities treated with growth constructs increased from 0% at insertion to up to 29% during lengthening and/or revision procedures[23,24]. Moreover, repeated hospitalizations for lengthening and unplanned surgical procedures increase the child’s time away from school and can have repercussions on the child’s psychological well-being[17,25].
Despite these findings, various articles published over the last few years consider growth-sparing surgery as the gold standard for the management of severe scoliosis in patients younger than 10 years of age[20,23].
However, unlike surgery, it does not potentially interfere with spine and thoracic cage growth. In particular, it has been shown that fitting growth-sparing devices near and/or on the spine can alter spinal growth and lead to auto-lusion of ribs and vertebral bodies[4,6,7,10]. These changes will contribute to make definitive fusion more challenging with less satisfactory outcomes[17,18].
Unlike surgery, serial EDF casting does not potentially interfere with spine and thoracic cage growth. In particular, it can be used as a “positive” corrective force to influence spinal growth[10] as it counteracts the negative effects of the spinal deformity on future spinal development of the growing organism[4,9,10]. However, although serial EDF casting can in some cases delay and/or eliminate the need for surgery, it cannot completely arrest curve progression nor erase the potential need for surgery in all types of curves.
Serial EDF casting should be considered a valuable low-risk treatment strategy[14,16,18] and an alternative to surgery for patients with early-onset scoliosis, in particular for those with IS and JS (Table 1). It has been proven effective in controlling curve progression, but there is still a lack of papers reporting outcomes of serial EDF casting in patients with IS and JS. In particular, there are less than 15 papers reporting on clinical and radiological outcomes in patients with IS and JS treated by serial casting (Table 1) vs over a hundred papers reporting on surgical outcomes in this patient population.
| Ref. | IS or JS | No. of patients | Idiopathic/non-idiopathic | Delay in surgery | % of surgery |
| Scott and Morgan[26] | IS | 28 | - | NA | NA |
| James et al[28] | IS | 212 | - | NA | NA |
| Lloyd-Roberts and Pilcher[29] | IS | 100 | - | NA | NA |
| Conner[27] | IS | 61 | - | NA | NA |
| Mehta[30] | IS | 64 | 64/0 | NA | NA |
| Ceballos et al[31] | IS | 113 | 113/0 | 3.5 yr | NA |
| Mehta[10] | IS | 941/422 (136) | 100/36 | 111 yr | 0%1 |
| 82 yr | 35.7%2 | ||||
| Sanders et al[21] | IS | 55 | 41/14 | 2.1 yr | 17% |
| Smith et al[34] | IS | 17 | NA | NA | 30% |
| Fletcher et al[15] | IS | 29 | 12/17 | 3.3 yr | 28% |
| Baulesh et al[32] | IS | 36 | 19/17 | 2.1 yr | 31% |
| Waldron et al[22] | IS | 20 | 8/12 | 18 mo | 40% |
| Johnston et al[33] | IS/JS | 27 | 11/16 | 20 mo | 55% |
| van Hessem et al[14] | JS | 7 | 7/0 | 4.6 yr | 0% |
| Canavese et al[13] | JS3 | 44 | 36/8 | 2 yr | 15%4 |
| 25%5 | |||||
| 33%6 | |||||
| Demirkiran et al[46] | Congenital scoliosis7 | 11 | NA | 2.1 yr | 0% |
In the early 1950s and late 1960s, Scott and Morgan[26] reported that resolving curves are not uncommon while Conner showed that early-onset deformities associated with developmental anomalies are likely to progress[27]. However, neither James et al[28] nor Lloyd-Roberts and Pilcher[29] were able to identify absolute criteria distinguishing between resolving and progressive curves.
In 1972, Mehta[30] showed that patients aggressively casted before age 20 mo for curves averaging 30 degrees go on to present zero net progression and/or up to a 10-degree reduction in scoliosis at skeletal maturity[10], whereas older children (30 mo of age) with curves averaging 50 degrees had the curve stabilized[10,30]. In a later work, Mehta reviewed 136 children with IS and found that although all children received the same treatment, the results followed different patterns. In the 94 children treated before 24 mo of age, the scoliosis resolved, whereas in the 42 patients treated after age 30 mo, treatment could only reduce but not resolve the deformity. Rate of surgery in this subgroup was 35.7%. Mehta[10,30] highlighted that avoiding delay in treatment was critically important.
Ceballos et al[31] confirmed Mehta’s prognostic criteria. They reviewed 113 patients with IS and concluded that relationship of rib head and corresponding vertebral body at the apex of the curve is the most reliable prognostic sign[31].
In Sanders et al[21]’s review of 55 patients with progressive IS treated with derotational casting, 46 of 55 patients (83%) responded to treatment and did not require surgery at the time of publication[21]. Sanders et al[21] confirmed Mehta’s findings[9,20,30] and reported that cast correction at younger age, moderate curve size, and an idiopathic diagnosis carry a better prognosis than casting at an older age, curve over 60 degrees, and a non-idiopathic diagnosis[21].
Waldron et al[22] reviewed 20 consecutive patients with early-onset scoliosis treated by a Risser cast performed under general anesthesia and found that at the time of publication, 13 of the 20 patients (65%) did not require surgery. They concluded that serial Risser casting under general anesthesia is a safe and effective time-buying strategy for patients with early-onset spinal deformity: relatively large curves can be stabilized and patients are allowed to reach a more suitable age for other forms of treatment[22]. Similarly, Fletcher et al[15] reported a group of 29 patients with IS treated by derotational casting with an average 39-mo delay to surgery, and found that at the time of publication, 21 of the 29 patients (72%) did not require surgery[15]. Although a cure could not be obtained in this cohort of patients, Fletcher et al[15] confirmed that serial casting can postpone surgical intervention and, in some cases, it can be considered as a valid alternative to surgery[15]. Baulesh et al[32] reported that serial casting can preserve normal longitudinal thoracic growth in children with early-onset spinal deformity and can have positive effects on pulmonary function. They reviewed 36 patients with early-onset spinal deformity, and reported a 25-mo delay of surgery in 25 of these 36 patients (69%)[32]. More recently, Morin and Kulkarni[20] reported that serial EDF casting for the treatment of progressive idiopathic IS is an effective tool for Metha’s benign-type curves[10,24], avoiding spinal fusion in about 2/3 of cases[20]. Johnston et al[33] reported that serial casting is a valuable delaying tactic for children with early-onset scoliosis. They reviewed 27 patients with IS and JS treated with serial casting and found that mean Cobb angle remained stable after a mean 2.4 years of treatment. Johnston et al[33] concluded that although serial casting cannot control all spinal deformities, it does not compromise spinal length and avoids surgical complications associated with early growth-sparing surgery[32]. In Smith et al[34]’s review of 31 patients with IS, 17 of the 31 were treated with serial casting and 30% progressed to surgery (5/17), prompting the authors to conclude that serial casting is particularly indicated in patients with smaller and flexible spinal curves[34].
On the other hand, casting and especially bracing has been less successful in JS compared to IS, with reported rates of surgery ranging from 27% to 100% after conservative treatment[13,14,35,36].
JS is a rare disease with diverse clinical presentation, so there are logically scarce few studies reporting clinical and/or radiological outcomes of patients with JS treated by serial casting. Most relevant studies published to date have evaluated the efficacy of rigid brace systems (Table 1). These studies reported a gradually slow loss of correction from the fitting point until the end of the treatment as well as a worsening of the curve after the weaning point[36-38].
Tolo and Gillespie[39] reported that 27% of patients treated with the Milwaukee brace underwent surgery. Similarly, Dabney and Browen[38] reported 33% rate of surgery in patients treated with rigid brace system for scoliosis. Coillard et al[40] using a SpineCor orthosis reported 37% surgeries in patients with JS. Higher percentages of surgery (40% to 100%) have been reported by Kahanovitz et al[41] and Robinson et al[42].
However, recent reports from van Hessem et al[14] and Canavese et al[13] point to more more favorable results in patients with JS treated by serial casting.
van Hessem et al[14] reviewed 7 patients with JS treated by casting and/or bracing and found that casting with patients awake is an effective JS management approach. They reported that Cobb’s angle was reduced 32%, decreasing from 37 degrees to 25 degrees, and none of the patients required surgery at a mean follow-up of 4.6 years[14].
More recently, Canavese et al[13] reviewed a cohort of 44 patients with JS and reported that serial EDF casting under general anesthesia plus neuromuscular-blocking drugs (curare) more effectively controls curve progression in JS patients compared to EDF casting under general anesthesia alone or no anesthesia[13]. They posit that complete muscle relaxation helps the surgeon better derotate the spine while straightening it. Moreover, adding neuromuscular-blocking drugs to the protocol improves immediate outcome and slows curve progression at 24-mo follow-up[13]. Canavese et al[13] found that curve magnitude according to Cobb[43], rib vertebral angle difference according to Mehta[30] and apical vertebral degree according to Nash and Moe[44] were all improved. Rib vertebral angle difference and apical vertebral degree are expressions of vertebral rotation and can be used to better characterize the spinal deformity and/or evaluate effects of brace or surgical treatment[45].
Demirkiran et a[46] have recently applied the principles of serial derotational casting to young and very young patients with congenital spine deformities. Demirkiran et al[46] reviewed 11 children with progressive congenital scoliosis. They reported that none of the patients required surgery for curve progression or developed complications during the treatment period. Demirkiran et al[46] concluded that serial derotational casting is an effective and safe delaying technique to postpone surgery in congenital deformities in the short-term follow-up.
There is a patent lack of papers reporting outcomes of serial EDF casting in patients with IS and JS. At the time of writing, there are less than 15 papers reporting clinical and radiological outcomes in patients with IS and JS treated by serial casting vs over a hundred papers reporting on surgical outcomes in this patient population.
EDF casting is a safe technique that can modify the natural evolution of infantile and juvenile-type scoliosis by reducing and slowing curve progression in both frontal and transverse planes.
P- Reviewer: Metzger PD S- Editor: Ji FF L- Editor: A E- Editor: Jiao XK
| 1. | Koop SE. Infantile and juvenile idiopathic scoliosis. Orthop Clin North Am. 1988;19:331-337. [PubMed] |
| 2. | Nnadi C. Early Onset Scoliosis. Stuttgart, Germany: Thieme 2015; . |
| 3. | Dimeglio A, Bonnel F. Le rachis en croissance. Paris, France: Springer Verlag 1990; . |
| 4. | Canavese F, Dimeglio A, Stebel M, Galeotti M, Canavese B, Cavalli F. Thoracic cage plasticity in prepubertal New Zealand white rabbits submitted to T1-T12 dorsal arthrodesis: computed tomography evaluation, echocardiographic assessment and cardio-pulmonary measurements. Eur Spine J. 2013;22:1101-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Dimeglio A, Canavese F. The growing spine: how spinal deformities influence normal spine and thoracic cage growth. Eur Spine J. 2012;21:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 162] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 6. | Canavese F, Dimeglio A, Volpatti D, Stebel M, Daures JP, Canavese B, Cavalli F. Dorsal arthrodesis of thoracic spine and effects on thorax growth in prepubertal New Zealand white rabbits. Spine (Phila Pa 1976). 2007;32:E443-E450. [PubMed] |
| 7. | Karol LA, Johnston C, Mladenov K, Schochet P, Walters P, Browne RH. Pulmonary function following early thoracic fusion in non-neuromuscular scoliosis. J Bone Joint Surg Am. 2008;90:1272-1281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 318] [Cited by in RCA: 314] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 8. | Dimeglio A. Growth of the spine before age 5 years. J Pediatr Orthop B. 1992;1:102-107. [RCA] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 111] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 9. | Cotrel Y, Morel G. [The elongation-derotation-flexion technic in the correction of scoliosis]. Rev Chir Orthop Reparatrice Appar Mot. 1964;50:59-75. [PubMed] |
| 10. | Mehta MH. Growth as a corrective force in the early treatment of progressive infantile scoliosis. J Bone Joint Surg Br. 2005;87:1237-1247. [PubMed] |
| 11. | Pehrsson K, Larsson S, Oden A, Nachemson A. Long-term follow-up of patients with untreated scoliosis. A study of mortality, causes of death, and symptoms. Spine (Phila Pa 1976). 1992;17:1091-1096. [PubMed] |
| 12. | Risser JC. The application of body casts for the correction of scoliosis. Instr Course Lect. 1955;12:255-259. [PubMed] |
| 13. | Canavese F, Botnari A, Dimeglio A, Samba A, Pereira B, Gerst A, Granier M, Rousset M, Dubousset J. Serial elongation, derotation and flexion (EDF) casting under general anesthesia and neuromuscular blocking drugs improve outcome in patients with juvenile scoliosis: preliminary results. Eur Spine J. 2015;Epub ahead of print. [PubMed] |
| 14. | van Hessem L, Schimmel JJ, Graat HC, de Kleuver M. Effective nonoperative treatment in juvenile idiopathic scoliosis. J Pediatr Orthop B. 2014;23:454-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Fletcher ND, McClung A, Rathjen KE, Denning JR, Browne R, Johnston CE. Serial casting as a delay tactic in the treatment of moderate-to-severe early-onset scoliosis. J Pediatr Orthop. 2012;32:664-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 91] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 16. | Dhawale AA, Shah SA, Reichard S, Holmes L, Brislin R, Rogers K, Mackenzie WG. Casting for infantile scoliosis: the pitfall of increased peak inspiratory pressure. J Pediatr Orthop. 2013;33:63-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 17. | Lattig F, Taurman L, Hell AK. Treatment of Early Onset Spinal Deformity (EOSD) with VEPTR: A Challenge for the Final Correction Spondylodesis: A Case Series. J Spinal Disord Tech. 2012;Epub ahead of print. [PubMed] |
| 18. | Zivkovic V, Büchler P, Ovadia D, Riise R, Stuecker R, Hasler C. Extraspinal ossifications after implantation of vertical expandable prosthetic titanium ribs (VEPTRs). J Child Orthop. 2014;8:237-244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Badlani N, Korenblit A, Hammerberg K. Subclavian vein thrombosis after application of body cast. J Pediatr Orthop. 2013;33:e1-e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 20. | Morin C, Kulkarni S. ED plaster-of-Paris jacket for infantile scoliosis. Eur Spine J. 2014;23 Suppl 4:S412-S418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Sanders JO, D’Astous J, Fitzgerald M, Khoury JG, Kishan S, Sturm PF. Derotational casting for progressive infantile scoliosis. J Pediatr Orthop. 2009;29:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 22. | Waldron SR, Poe-Kochert C, Son-Hing JP, Thompson GH. Early onset scoliosis: the value of serial risser casts. J Pediatr Orthop. 2013;33:775-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 23. | Yang JS, McElroy MJ, Akbarnia BA, Salari P, Oliveira D, Thompson GH, Emans JB, Yazici M, Skaggs DL, Shah SA. Growing rods for spinal deformity: characterizing consensus and variation in current use. J Pediatr Orthop. 2010;30:264-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 24. | Mackenzie WG, Matsumoto H, Williams BA, Corona J, Lee C, Cody SR, Covington L, Saiman L, Flynn JM, Skaggs DL. Surgical site infection following spinal instrumentation for scoliosis: a multicenter analysis of rates, risk factors, and pathogens. J Bone Joint Surg Am. 2013;95:800-806, S1-S2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 123] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 25. | Bess S, Akbarnia BA, Thompson GH, Sponseller PD, Shah SA, El Sebaie H, Boachie-Adjei O, Karlin LI, Canale S, Poe-Kochert C. Complications of growing-rod treatment for early-onset scoliosis: analysis of one hundred and forty patients. J Bone Joint Surg Am. 2010;92:2533-2543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 426] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 26. | Scott JC, Morgan TH. The natural history and prognosis of infantile idiopathic scoliosis. J Bone Joint Surg Br. 1955;37-B:400-413. [PubMed] |
| 27. | Conner AN. Developmental anomalies and prognosis in infantile idiopathic scoliosis. J Bone Joint Surg Br. 1969;51:711-713. [PubMed] |
| 28. | James JI, Lloyd-Roberts GC, Pilcher MF. Infantile structural scoliosis. J Bone Joint Surg Br. 1959;41-B:719-735. [PubMed] |
| 29. | Lloyd-Roberts GC, Pilcher MF. Structural idiopathic scoliosis in infancy: a study of the natural history of 100 patients. J Bone Joint Surg Br. 1965;47:520-523. [PubMed] |
| 30. | Mehta MH. The rib-vertebra angle in the early diagnosis between resolving and progressive infantile scoliosis. J Bone Joint Surg Br. 1972;54:230-243. [PubMed] |
| 31. | Ceballos T, Ferrer-Torrelles M, Castillo F, Fernandez-Paredes E. Prognosis in infantile idiopathic scoliosis. J Bone Joint Surg Am. 1980;62:863-875. [PubMed] |
| 32. | Baulesh DM, Huh J, Judkins T, Garg S, Miller NH, Erickson MA. The role of serial casting in early-onset scoliosis (EOS). J Pediatr Orthop. 2012;32:658-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 33. | Johnston CE, McClung AM, Thompson GH, Poe-Kochert C, Sanders JO. Comparison of growing rod instrumentation versus serial cast treatment of early-onset scoliosis. Spine Deform. 2013;1:339-342. [RCA] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 34. | Smith JR, Samdani AF, Pahys J, Ranade A, Asghar J, Cahill P, Betz RR. The role of bracing, casting, and vertical expandable prosthetic titanium rib for the treatment of infantile idiopathic scoliosis: a single-institution experience with 31 consecutive patients. Clinical article. J Neurosurg Spine. 2009;11:3-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 35. | Grivas TB, Kaspiris A. European braces widely used for conservative scoliosis treatment. Stud Health Technol Inform. 2010;158:157-166. [PubMed] |
| 36. | Lonstein JE, Winter RB. The Milwaukee brace for the treatment of adolescent idiopathic scoliosis. A review of one thousand and twenty patients. J Bone Joint Surg Am. 1994;76:1207-1221. [PubMed] |
| 37. | Gabos PG, Bojescul JA, Bowen JR, Keeler K, Rich L. Long-term follow-up of female patients with idiopathic scoliosis treated with the Wilmington orthosis. J Bone Joint Surg Am. 2004;86-A:1891-1899. [PubMed] |
| 38. | Dabney KW, Browen JR. Juvenile idiopathic scoliosis. Semin Spine Surg. 1991;3:524-530. |
| 39. | Tolo VT, Gillespie R. The characteristics of juvenile idiopathic scoliosis and results of its treatment. J Bone Joint Surg Br. 1978;60-B:181-188. [PubMed] |
| 40. | Coillard C, Leroux MA, Zabjek KF, Rivard CH. SpineCor--a non-rigid brace for the treatment of idiopathic scoliosis: post-treatment results. Eur Spine J. 2003;12:141-148. [PubMed] |
| 41. | Kahanovitz N, Levine DB, Lardone J. The part-time Milwaukee brace treatment of juvenile idiopathic scoliosis. Long-term follow-up. Clin Orthop Relat Res. 1982;145-151. [PubMed] |
| 42. | Robinson CM, McMaster MJ. Juvenile idiopathic scoliosis. Curve patterns and prognosis in one hundred and nine patients. J Bone Joint Surg Am. 1996;78:1140-1148. [PubMed] |
| 43. | Cobb JR. Outline for the study of scoliosis. Instr Course Lect. 1948;5:261-275. |
| 44. | Nash CL, Moe JH. A study of vertebral rotation. J Bone Joint Surg Am. 1969;51:223-229. [PubMed] |
| 45. | Canavese F, Holveck J, De Coulon G, Kaelin A. Analysis of concave and convex rib-vertebral angle, angle difference, and angle ratio in patients with lenke type 1 main thoracic adolescent idiopathic scoliosis treated by observation, bracing or posterior fusion, and instrumentation. J Spinal Disord Tech. 2011;24:506-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Demirkiran HG, Bekmez S, Celilov R, Ayvaz M, Dede O, Yazici M. Serial derotational casting in congenital scoliosis as a time-buying strategy. J Pediatr Orthop. 2015;35:43-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 37] [Article Influence: 3.7] [Reference Citation Analysis (0)] |