Published online Feb 10, 2013. doi: 10.5306/wjco.v4.i1.25
Revised: November 26, 2012
Accepted: December 15, 2012
Published online: February 10, 2013
Processing time: 185 Days and 13.7 Hours
Intravenous leiomyomatosis (IVL), showing unusual growth patterns of uterine leiomyoma, is a rare neoplasm characterized by intravascular proliferation of a histologically benign-looking smooth muscle cell tumor mass, but not invading the tissue. To date, less than 300 cases have been reported and fewer than 100 cases with cardiac involvement. Imaging characteristics of IVL are still not clear so it is usually misdiagnosed before surgery. A 36-year-old woman, who had undergone hysterectomy due to hysteromyoma, presented with shortness of breath after activities. Imaging showed IVL with mass involvement of the left ovarian vein, left renal vein, left external and common iliac vein, as well as within the inferior vena cava (IVC), extending into the right atrium. The operation demonstrated that the mass had no stalk and had well-demarcated borders with the wall of the right atrium and IVC. The patient underwent a one-stage combined multidisciplinary thoraco-abdominal operation under general anesthetic. Subsequently, the pathology report confirmed IVL. IVL should be considered in a female patient presenting with an extensive mass in the right side of the heart. Imaging technology, such as echocardiogram, contrast-enhanced computed tomography and magnetic resonance imaging, can provide important information to reveal the mass, the range and path of the lesion, and relates to the surgical plan decision. Consequently, perfect and exact image examination is very necessary pre-operation.
- Citation: Xu ZF, Yong F, Chen YY, Pan AZ. Uterine intravenous leiomyomatosis with cardiac extension: Imaging characteristics and literature review. World J Clin Oncol 2013; 4(1): 25-28
- URL: https://www.wjgnet.com/2218-4333/full/v4/i1/25.htm
- DOI: https://dx.doi.org/10.5306/wjco.v4.i1.25
Intravenous leiomyomatosis (IVL), showing unusual growth patterns of uterine leiomyoma, is a rare neoplasm characterized by intravascular proliferation of a histologically benign-looking smooth muscle cell tumor mass, but not invading the tissue. Birch-Hirschfeld[1] first presented a case of IVL in 1896 and Durck first presented a case of intracardiac extension of IVL in 1907; cases of intracardiac extension account for about 10%[2]. To date, less than 300 cases have been reported in the English literature.
Although histologically benign, IVL might sometimes be malignant in behavior, with not only involvement of pelvic veins, the inferior vena cava (IVC), adrenal and renal veins, but sometimes reaching as far as the right cardiac chambers and the main pulmonary artery, and may result in cardiac symptoms and a cardiac murmur, fainting and even, in some cases, sudden death[3]. Because of its rarity, IVL is usually misdiagnosed pre-operation and diagnosed late, and is subsequently not treated properly. The correct preoperative diagnosis of IVL depends on a huge amount of information, especially a comprehensive imaging examination. Here, we present a case of IVL diagnosed by transthoracic echocardiography, abdominal contrast computed tomography (CT) and CT angiography (CTA) and verified by histopathological evaluation, and we review the literature.
A 36-year-old woman presented with a 1 mo history of shortness of breath after activities with no obvious cause, and not accompanied by palpitation, fever, cough and paroxysmal nocturnal dyspnea, etc. Two years ago, she had undergone hysterectomy due to hysteromyoma and was not found to have any heart disease or relevant history. On examination, there was no abnormality in the physical examination except for cardiac auscultation and mild edema of both lower extremities. II/VI level of systolic blowing murmur and diastolic rumbling murmur were heard in the 4th and 5th intercostals on the left sternum. In addition, incomplete right bundle branch conduction block, increased right heart load and mildly lowered S-T segment were found on the electrocardiogram check. The laboratory examinations revealed normal results, including tumor markers, liver and kidney function, blood and urine examination, etc.
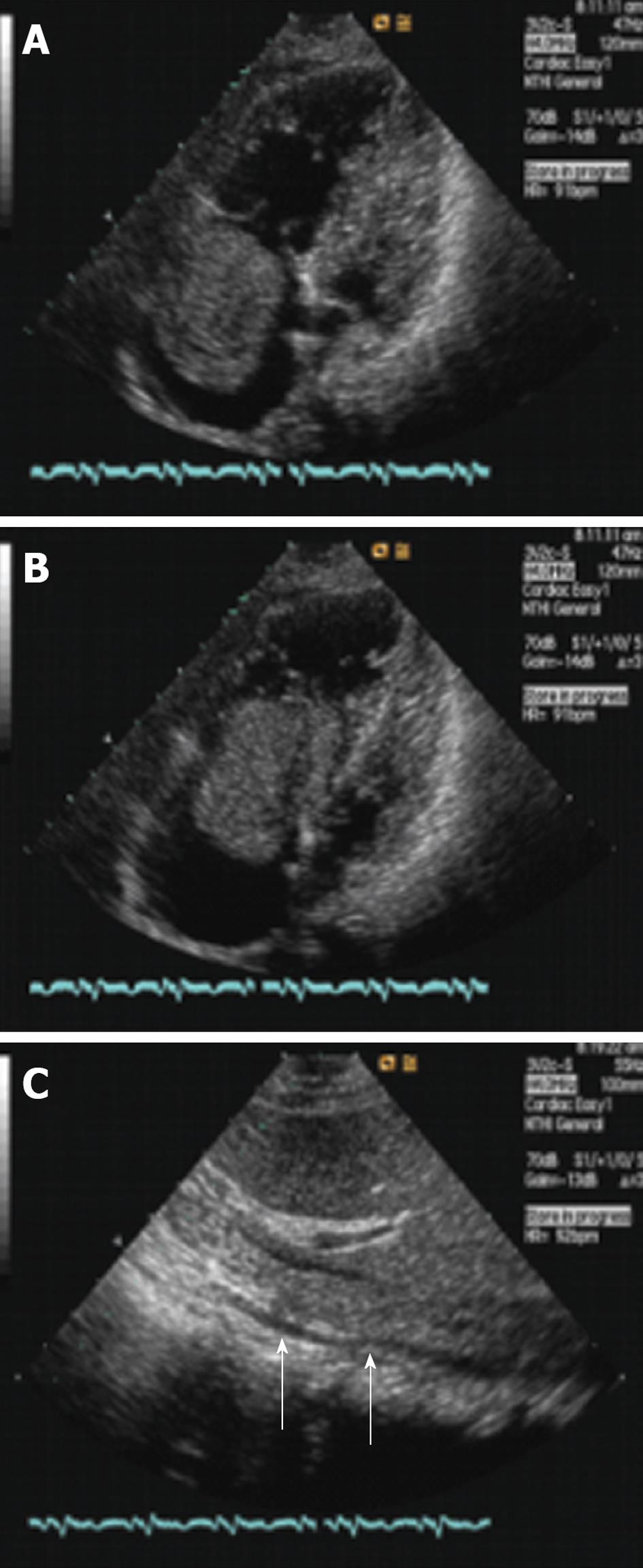
Transthoracic two-dimensional echocardiography showed a mild increase of the right atrium and ventricle and the atrium was filled with an echogenic oval tumor mass which was approximately 6.8 cm × 4.0 cm and moved back and forth through the tricuspid orifice into the right ventricle (Figure 1A and B). The mass size was about 10.7 cm × 1.2 cm and involved the IVC and extended through the lumen (Figure 1C). The mass of the right atrium and IVC was stretched and had well-demarcated borders with the wall of the right atrium and IVC. The presumptive diagnosis of left atrial myxomas was made by an ultrasonic practitioner.
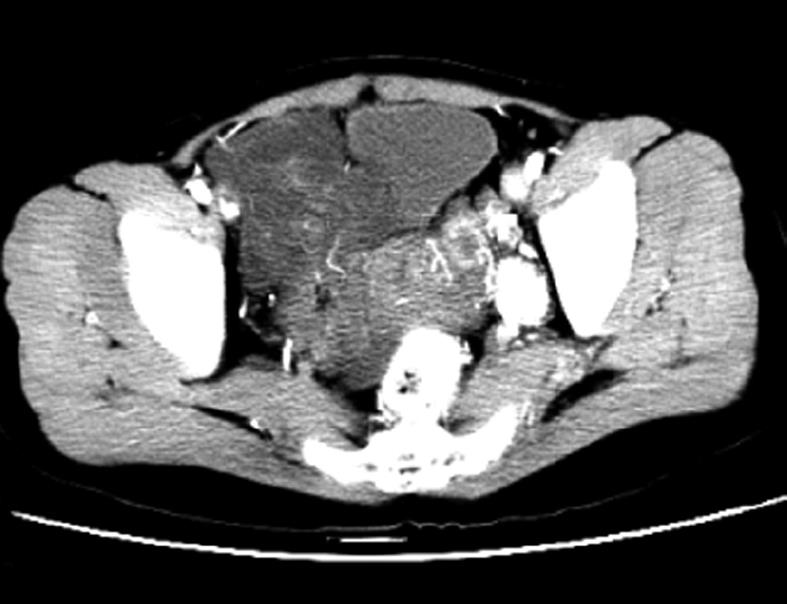
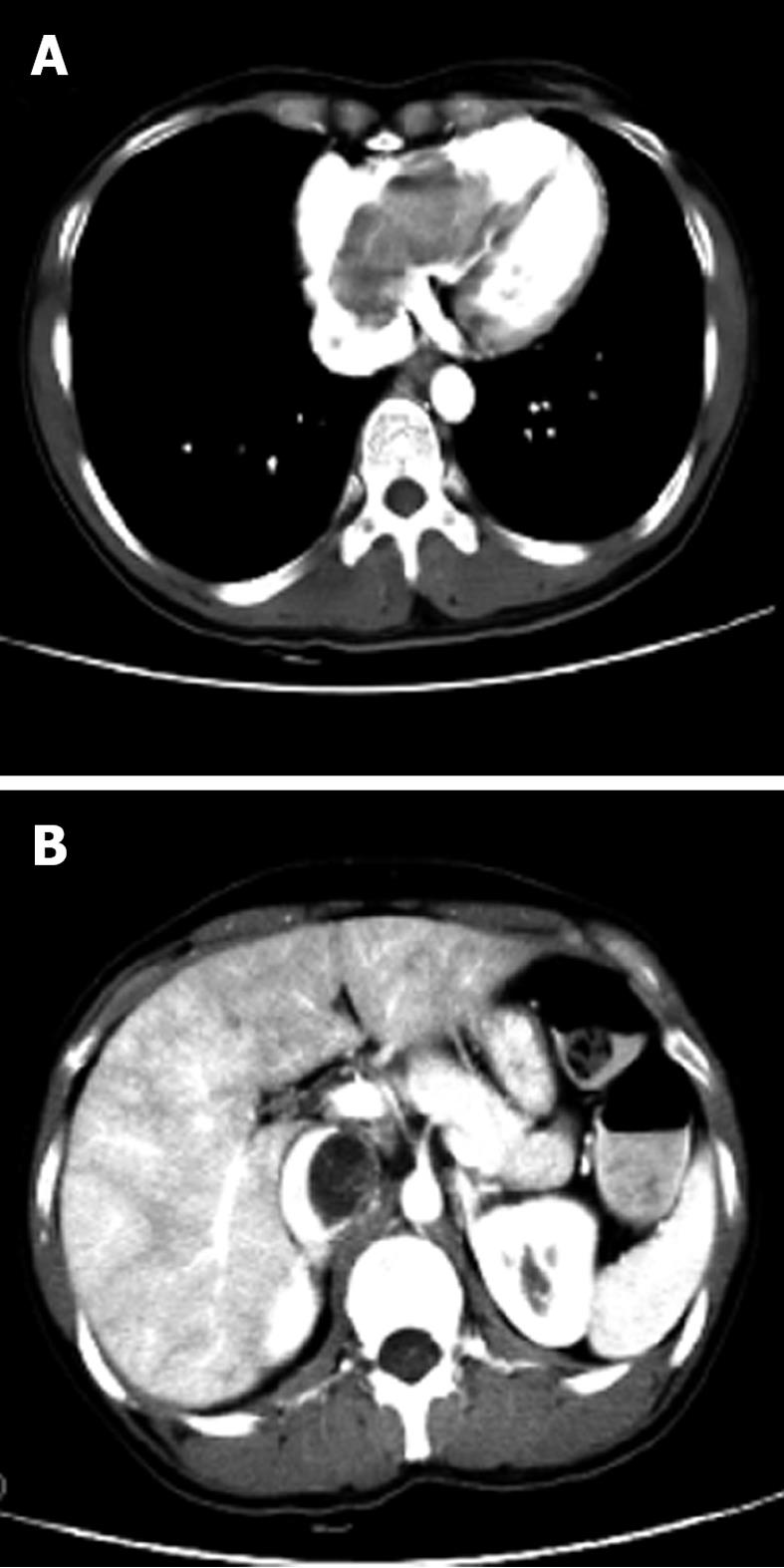
Contrast enhanced CT and CTA of the thoracic, abdominal and pelvic cavity were performed. The pelvic CT revealed a lobulated mass with local heterogeneity contrast enhancement (Figure 2). A low attenuation-filling defect without contrast enhancement was present in the left ovarian vein, left renal vein, left external and common iliac vein, as well as within the IVC, extending into the right atrium (Figure 3). The mass within the vein and right atrium was wide. Hence, the presumptive diagnosis of sarcoma with invasion in to the vein system and right atrium was made; the mass was not in the vein system, except for the thrombus formation.
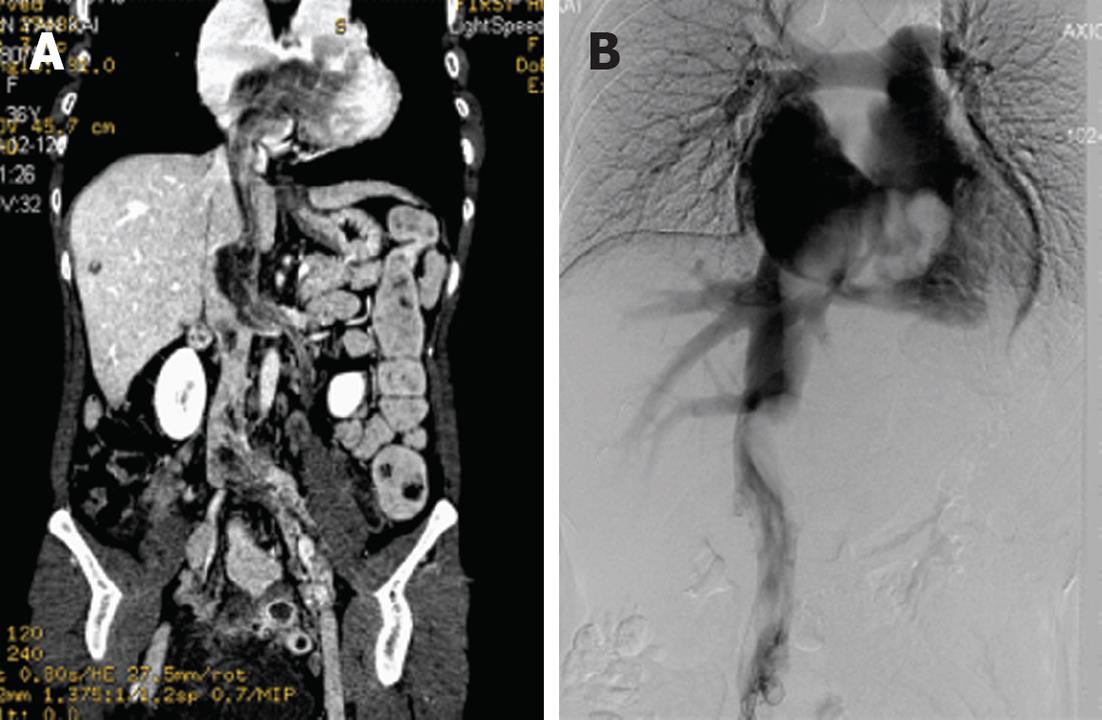
Digital subtraction angiography of the thoracic and abdominal cavity was performed. The imaging demonstrated an irregular mass, like a filling defect from the IVC to the right atrium, and presented as a large tumor within the atrium (Figure 4).
Cardiac and vascular surgery was carried out under general anesthetic and with circulatory arrest. The pelvic masses were partially resected because of extensive lesions, including bilateral accessories. The left ovarian vein was cut off and the IVC and right atrium opened; the size of tumor within the atrium was about 8 cm × 6 cm × 6 cm. The surgeons separated the adhesion between the mass and the wall of the vascular structure and heart and peeled the lesions completely. The length of tumor within the vascular structure and heart was 30 cm. All the masses looked red and had the same pathological features, showing a spindle cell tumor, size consistency, less karyokinesis and with a thick wall vessel in the mesenchyme. Subsequently, the pathological report confirmed IVL.
The neoplastic smooth muscles of IVL are histologically and cytogenetically similar to benign leiomyomata but might behave in a “malignant” fashion, with not only involvement of pelvic veins, IVC, renal veins, pulmonary artery and right cardiac chamber, but also distant metastasis, such as lung, brain, lymph nodes and so on[3-5]. Although cardiac involvement is present in up to 10% of cases[3], in the last 10 years the literature contains more reports of IVL and fewer than 100 cases with cardiac involvement.
So far, the pathogenesis of IVL is still unclear; one possibility is that the tumor originated from smooth muscle in the vessel wall and the other that it is the uterine leiomyoma which invaded the uterine vein and extended extensively. The current patient had a history of hysterectomy 5 years ago. CT imaging revealed an irregular enhancing tumor in the pelvis invading into the left ovarian vein, renal vein and common iliac vein, as well as within the IVC, extending into the right atrium. The mass was not adhesive with the wall of the vascular structure and heart and was resected completely. Therefore, the case was inclined to support the second theory of pathogenesis.
Clinical characteristics of IVL mainly depend on the location and scale of lesion. More distal intravascular extension of the tumor can result in various cardiorespiratory symptoms. Symptoms of abdominal pain, shortness of breath, palpitation and edema of the lower extremities are the most common and occasionally present with Budd-Chiari syndrome, pulmonary embolism and sudden death[3,5]. The prominent clinical manifestation of the present case was shortness of breath and mild edema of the lower extremities. The reason for misdiagnosis pre-operation may be because it is not a typical symptom for IVL diagnosis.
Previously, many case reports described characteristic imaging features, including nodular, enhancing tumors that appear to originate in the uterus and extend into the venous system, causing expansion of the involved vascular structures and heart[6-8]. Echocardiographic features of IVL with cardiac extension include a hyperechoic elongated mobile mass extending from the IVC and an irregular mass in the atrium. The tumor in the atrium usually is misdiagnosed as myxoma, although some key points of a differential diagnosis are advanced[9], as happened in the present case. Usually, more imaging examinations are needed. Contrast-enhanced CT, CTA and post-processing (multi planar reformation, maximum intensity projection ) can directly show the tumor and full-scale path of extension, which is the key for the establishment of an operative plan[6]. A pelvic irregular tumor usually presents with inhomogeneous distinct enhancement and the iliac vein, ovarian veins, IVC and right atrium are usually involved, distended and filled with an enhancing mass. In addition, IVC can be distended with a non-enhancing thrombus. In the present case, CT imaging revealed a lobulated inhomogeneous enhanced mass in the pelvis and a long, serpentine and elongated mobile mass extending from the left ovarian veins, left internal iliac vein, continued to the IVC and extending into the right atrium. However, it is sad that it did not raise the presumptive diagnosis of IVL. In retrospect, once this disease is discovered, the diagnosis of IVL should be considered by combining the CT features with the history of uterine myoma. Pre-surgical CT examination is very important. Magnetic resonance imaging (MRI) features of IVL were also reported, similar to the findings of CT but with advantages of superb soft tissue contrast resolution, direct multi-planar imaging capability and unique ability to assess blood flow.
According to imaging features, the differential diagnosis of IVL mainly includes intravenous thrombus, leiomyosarcoma, right atrial myxoma and tumor thrombosis with malignant carcinoma, for example, renal carcinoma, hepatocellular carcinoma, adrenal cortical carcinoma, etc. The differential diagnosis points of them have been described by previous reports[3,6,8]. Therefore, here, we summarize the diagnosis points of IVL: (1) Middle-aged women, usually with a history of hysterectomy; (2) Pelvic irregular mass with inhomogeneous enhancement, invasion in the pelvic vein and extending to the IVC, sometimes to the right atrium; (3) Lesions widely infringing the venous system is an important feature; (4) The mobile mass within the right atrium is always accompanied with a large mass in the IVC and continued; and (5) The mass in the heart and vein structures have no adhesion with the wall of heart and vein. According to these features, the present case was a typical IVL case but it was misdiagnosed pre-operation; to our knowledge, it was the first case confirmed in our hospital. Although these features strongly suggest the diagnosis of IVL, the final diagnosis depends on histopathology.
Surgical excision is still the best treatment of choice for IVL and complete removal of the tumor is considered essential to prevent a recurrence. In fact, it has a high rate of recurrence because complete resection is a difficult thing to achieve[10]. Once IVL involves the right heart chamber, a combined multidisciplinary thoraco-abdominal operation is required. In the present case, the intra-cardiac and intra-vascular mass was free-floating without involvement of the cardiac structure and vein wall tissue. The left ovarian vein was cut off and then the mass was resected by opening the IVC and right atrium. Only a partial resection of the pelvic tumor was performed because of the widespread tumor and involvement of small vessels in the pelvis. Estrogen would stimulate the tumor to grow[10] and therefore a bilateral oophorectomy was performed.
In conclusion, intracardiac leiomyomatosis should be considered in a female patient presenting with an extensive mass in the right side of the heart. Imaging technology, such as echocardiogram, contrast-enhanced CT and MRI, can provide important information to reveal the mass, the range and path of the lesion, and relates to the surgical plan decision. Consequently, perfect and exact image examination is very necessary pre-operation.
P-Reviewer Nishiyama M S- Editor Huang XZ L- Editor Roemmele A E- Editor Zheng XM
| 1. | Birch-Hirschfeld FV. Lehrbuch der pathologischen Anatomie. 5th ed. Leipzig: FCW Vogel 1896; 226. |
| 2. | Baca López FM, Martínez-Enriquez A, Castrejón-Aivar FJ, Ruanova-León D, Yánez-Gutiérrez L. Echocardiographic study of an intravenous leiomyoma: case report and review of the literature. Echocardiography. 2003;20:723-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Lee S, Kim DK, Narm KS, Cho SH. Pulmonary artery embolization of intravenous leiomyomatosis extending into the right atrium. Korean J Thorac Cardiovasc Surg. 2011;44:243-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 4. | Thian YL, Tan KH, Kwek JW, Wang J, Chern B, Yam KL. Leiomyomatosis peritonealis disseminata and subcutaneous myoma--a rare complication of laparoscopic myomectomy. Abdom Imaging. 2009;34:235-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Lee HJ, Choi J, Kim KR. Pulmonary benign metastasizing leiomyoma associated with intravenous leiomyomatosis of the uterus: clinical behavior and genomic changes supporting a transportation theory. Int J Gynecol Pathol. 2008;27:340-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | Sun C, Wang XM, Liu C, Xv ZD, Wang DP, Sun XL, Deng K. Intravenous leiomyomatosis: diagnosis and follow-up with multislice computed tomography. Am J Surg. 2010;200:e41-e43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Oehler MK, Scopacasa L, Brown M, Kumar G, Edwards J. Intravenous uterine leiomyomatosis extending into the right heart. Aust N Z J Obstet Gynaecol. 2011;51:92-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Li Y, Mei F, Yang C, Lv P, Ouyang C, Jin B. Intravenous leiomyomatosis with right heart involvement-a report of 4 cases and literature review. J Huazhong Univ Sci Technolog Med Sci. 2011;31:586-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Okamura H, Yamaguchi A, Kimura N, Adachi K, Adachi H. Partial resection of intravenous leiomyomatosis with cardiac extension. Gen Thorac Cardiovasc Surg. 2011;59:38-41. [PubMed] |
| 10. | Kokawa K, Yamoto M, Yata C, Mabuchi Y, Umesaki N. Postmenopausal intravenous leiomyomatosis with high levels of estradiol and estrogen receptor. Obstet Gynecol. 2002;100:1124-1126. [PubMed] |