Published online Jan 10, 2012. doi: 10.5306/wjco.v3.i1.7
Revised: October 21, 2011
Accepted: January 7, 2012
Published online: January 10, 2012
The hypercalcemias are a common and heterogeneous group of disorders, ranging from the occasional detection of a high level of serum calcium to a life-treating condition. In a patient presenting with hypercalcemia, a differential diagnosis can be established easily by measuring serum calcium and parathyroid hormone (PTH) concentrations. We describe the case of an 83-year-old man presenting with a severe symptomatic hypercalcemia with high-normal PTH level due to the coexistence of primary hyperparathyroidism and malignancy-associated hypercalcemia. The presence of two conditions producing hypercalcemia was revealed only during in-hospital stay and after the administration of an intravenous bisphosphonate, when the PTH concentration increased rapidly after bisphosphonate treatment with a decrease in serum calcium. The occurrence of two conditions producing hypercalcemia is a rare event in the literature, and should be considered in the presence of an abnormally high serum calcium level associated with normal or high-normal PTH, in order to establish a correct diagnosis and appropriate interventions.
- Citation: Pellicciotti F, Giusti A, Gelli MC, Foderaro S, Ferrari A, Pioli G. Challenges in the differential diagnosis of hypercalcemia: A case of hypercalcemia with normal PTH level. World J Clin Oncol 2012; 3(1): 7-11
- URL: https://www.wjgnet.com/2218-4333/full/v3/i1/7.htm
- DOI: https://dx.doi.org/10.5306/wjco.v3.i1.7
The hypercalcemias (HCa) are a common and heterogeneous group of disorders, ranging from the occasional detection of a high level of serum calcium during routine laboratory assessment to a life-treating condition[1-5]. In general, the signs and symptoms are not specific, and are related to the level of serum calcium, to the rate of calcium increase and to the underlying condition producing HCa. Mild hypercalcemia 11-11.5 mg/dL is usually asymptomatic, while acute onset severe HCa (> 13 mg/dL) may present with lethargy, stupor and coma.
From a pathophysiological point of view, an increase in serum calcium above the reference range is the result of the failure of renal calcium excretion to compensate for an increased influx of calcium into the circulation from the intestine, the kidneys and the skeleton[4,5]. Primary hyperparathyroidism and malignant neoplasms are responsible for more than 90% of all cases of hypercalcemia[5].
In the case of hypercalcemia, a differential diagnosis can be established easily by measuring serum calcium and parathyroid hormone (PTH) concentrations. In clinical practice, serum total calcium concentrations should be adjusted to serum albumin values, while the measurement of ionized serum calcium is rarely needed[4,5].
The finding of an increased serum calcium level in the presence of an inappropriately elevated PTH concentration should suggest a PTH-dependent HCa (primary hyperparathyroidism), while the observation of HCa with suppressed or low-normal PTH values should suggest a PTH-independent hypercalcemia (e.g., granulomatous disorders or malignancy-associated hypercalcemia, MAH)[1,2,4,5].
In the acute clinical setting, the management of severe HCa is independent of the underlying cause, being based on life-treating interventions such as hydration and the prescription of calcium lowering agents (e.g., bisphosphonates). On the other hand, the long-term treatment and prognosis of HCa is highly dependent on the underlying cause[4]. Therefore, a correct differential diagnosis is crucial to maximize the outcome and improve quality of life.
We herein describe the case of an 83-year-old man presenting with hypercalcemia with normal PTH level due to the coexistence of primary hyperparathyroidism and MAH. The occurrence of two conditions producing HCa is a rare event in the literature[6-8], and should be considered in the presence of an abnormally high serum calcium level associated with normal PTH, in order to establish a correct diagnosis and appropriate interventions.
An 83-year-old man was admitted to the Geriatric Acute Care Unit of the Arcispedale Santa Maria Nuova (ASMN, Reggio Emilia) with delirium and other symptoms. The patient was evaluated at the time of admission and followed during hospital stay with serial measurements of laboratory tests.
Serum 25-hydroxy-vitamin D (25-OH-D) was measured by radioimmunoassay using a commercial kit (detection limit 1.5 ng/mL; DiaSorin, Saluggia, Italy). Serum intact PTH (1-84) was assessed using an immunoradiometric method (DiaSorin) with a sensitivity of 0.7 pg/mL (normal range 15 pg/mL-75 pg/mL). The interassay coefficients of variation were between 8.2% and 11% for 25-OH-D and between 3.4% and 4.9% for PTH (depending on the measured concentration). All other parameters were measured using standard automated laboratory methods.
A needle biopsy of axillary lymph nodes and a bone marrow biopsy (obtained from the iliac crest) were performed. Specimens were fixed in 4% buffered formaldehyde (bone marrow biopsy was subsequently decalcified) and processed for routine paraffin embedding. Sections of 5 μm were prepared for routine light microscopy after staining with hematoxylin and eosin. Immunohistochemical staining with the streptavidin-biotin peroxidase detection system was performed using the Ventana automated immunostainer (Ventana Medical System, Tucson, Arizona, United States).
The patient was admitted to the Geriatric Acute Care Unit of the ASMN Hospital at the beginning of August 2010. He was an 83-year-old man, living at home with his wife and walking without aid. His medical history included mild to moderate dementia (started one year before) with minor behavioral symptoms, hypertension, chronic coronary heart disease, carotid atheromatous disease, benign prostatic hyperplasia treated with trans-urethral retrograde prostatectomy, chronic gastritis and duodenal ulcer. Despite having dementia and other comorbidities, he had conserved abilities of daily living. Medications included: trazodone 12.5 mg/bid, bisoprolol 1.25 mg/d, losartan 25 mg/d, acetylsalicylic acid 100 mg/d and rosuvastatin 10 mg/d.
Five days before admission to ASMN Hospital, he was evaluated in the Emergency Department of another hospital for asthenia and worsening of cognitive impairment. Routine blood samples and radiologic evaluations (brain CT and abdominal X-ray) showed severe hyponatremia (121 mmol/L), chronic vascular encephalopathy and coprostasis. Serum calcium was not assessed. He was discharged home without any therapy.
On admission to the Geriatric Unit of the ASMN Hospital, his caregiver referred to persistence of the following symptoms: asthenia, dizziness, recurrent falls, drowsiness, delirium, constipation, polyuria, polydipsia and stupor. The first laboratory assessment confirmed the presence of hyponatremia (129 mmol/L), and demonstrated hypocloremia (87 mmol/L), leukocytosis (10.829/mmc), normocytic anemia (Hb 12.2 mg/dL; MCV 81.4) and renal failure (creatinine 1.6 mg/dL; azotemia 46). Chest and abdominal X-rays were negative. The symptoms were, at this point, correlated to hyponatremia and dehydration-syndrome. Thus, fluid and electrolyte therapy were started.
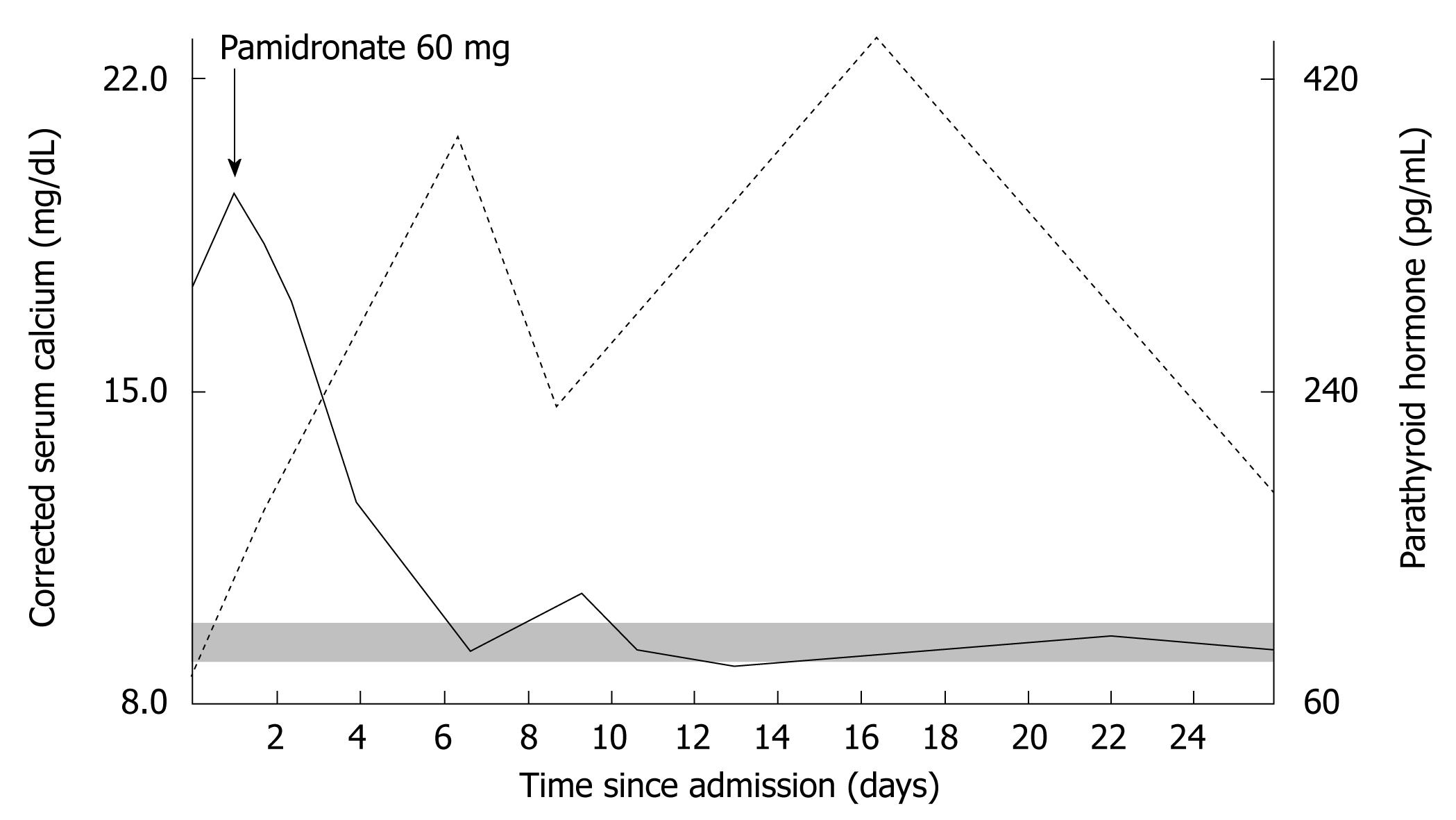
On the second day a routine blood sample showed moderate liver dysfunction, the presence of monoclonal antibodies below the detection limit on serum protein electrophoresis, and severe hypercalcemia (18.2 mg/dL) with PTH (64 pg/mL) within the reference range (Figure 1), associated with hyperphosphatemia (4.8 mg/dL, normal range 2.5 mg/dL-4.5 mg/dL) and hypomagnesemia (1.6 mg/dL, normal range 1.7 mg/dL-2.5 mg/dL).
Given the presence of severe symptomatic hypercalcemia, the patient was treated with re-hydration associated with furosemide, and an intravenous bisphosphonate (pamidronate 60 mg in 500 cc saline in a single administration). As shown in Figure 1, a slight decrease in calcium was observed in the first two days after bisphosphonate treatment, followed by a rapid decrease in serum calcium from day 3. At the same time, PTH demonstrated a rapid and sustained increase in the days following the bisphosphonate infusion (up to 410 pg/mL).
In association with the decrease in serum calcium, a significant improvement in symptoms was observed. In particular, this treatment ameliorated the level of consciousness, resolved dizziness and improved ambulation.
The coexistence of severe symptomatic hypercalcemia with a PTH level within the reference range suggested the presence of two different conditions producing an increase in serum calcium, but opposite effects on PTH (PTH-dependent and PTH-independent hypercalcemia)[1-3].
The sudden increase in PTH (from 64 pg/mL to 410 pg/mL) following the slight decrease in calcium (from 18.2 mg/dL to 15.7 mg/dL; still above the upper limit of the reference range) early after pamidronate infusion, suggested the presence of primary hyperparathyroidism. Further instrumental investigations were undertaken due to a suspected coexisting MAH.
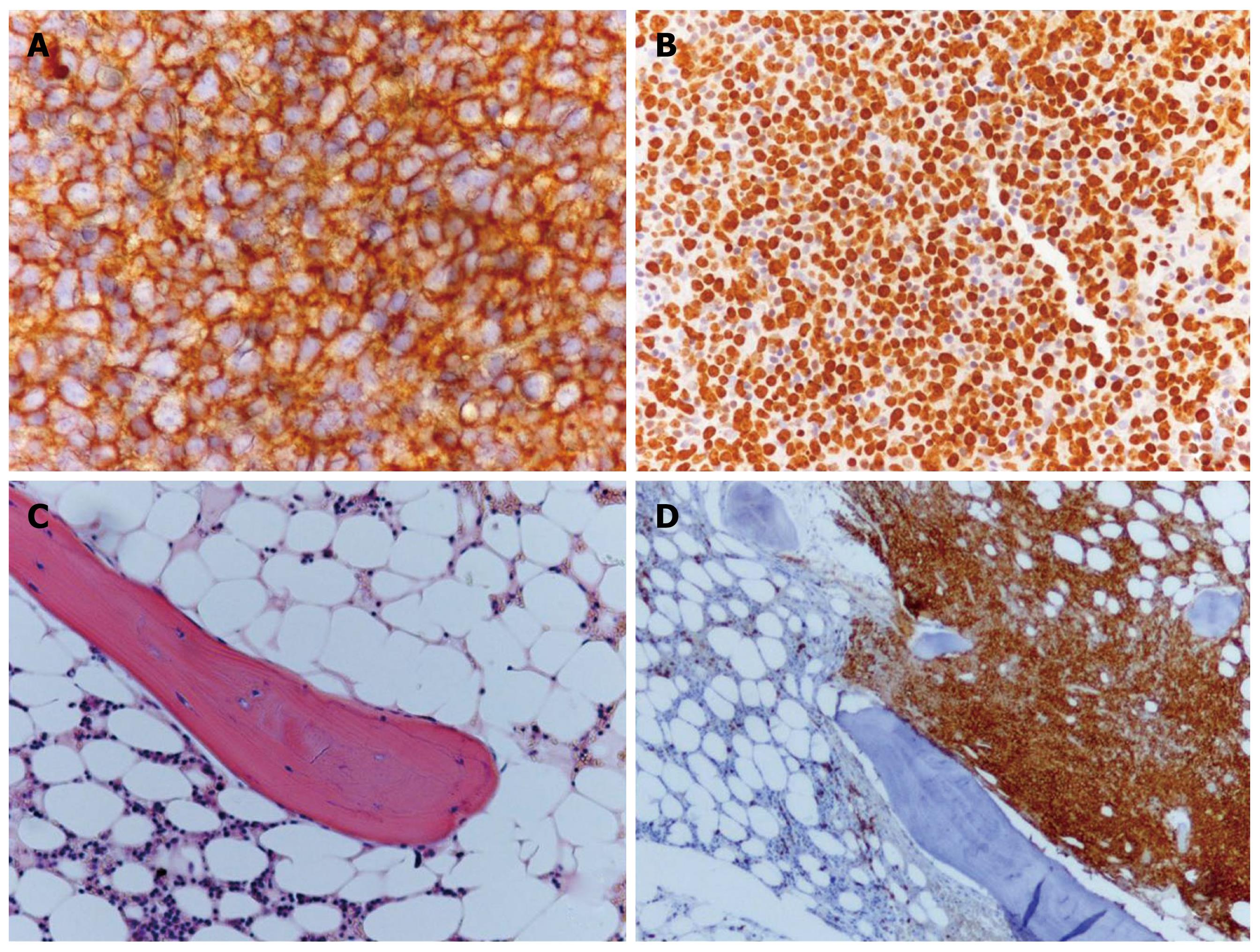
A whole-body bone scan did not detect any areas of increased uptake, and therefore, excluded hypercalcemia due to osteolysis. A whole body CT scan demonstrated the presence of enlarged mediastinal and abdominal lymph nodes suggesting a lymphoproliferative disease. A needle biopsy of axillary lymph nodes was performed. Histological examination revealed a diffuse large B-cell non-Hodgkin lymphoma confirming MAH (Figures 2A and B). The examined tissue was completely replaced by medium to large lymphoid cells with oval to round nuclei. Immunohistochemical investigation revealed a B-cell phenotype (CD20+, Bcl2+, Bcl6+, CD3-, CD10-, CD30-, cyclin D1-). The proliferative fraction detected by Ki67 staining was notably high (80%-90%).
An echographic evaluation of the neck was unable to localize the abnormal parathyroid gland, while a sestamibi scintigraphic evaluation undertaken to localize the hyperfunctioning parathyroid gland demonstrated an area of increased uptake at the base of the right thyroid lobe.
Among the laboratory analyses, a severe vitamin D deficiency was observed. The bone marrow biopsy revealed normal trabecular borders and surface excluding osteomalacia (Figures 2C and D). Cellularity was increased, but hematopoietic marrow lines were reduced. About 70% of bone marrow cellularity was occupied by a lymphoid proliferation, diffusely necrotic with analogous characteristics which were observed in lymph node tissue.
The final diagnosis was non-Hodgkin lymphoma stage 4A associated with primary hyperparathyroidism. When serum calcium was normalized, the patient started chemotherapy. Unfortunately, on the 36th day (six days after the first chemotherapy cycle), the patient’s clinical condition worsened and two days later he died due to irreversible heart failure. Thus, it was not possible to undertake further investigations.
In clinical practice, the differential diagnosis of hypercalcemia is based on the evaluation of serum calcium and PTH levels, with a high PTH concentration suggesting a PTH-dependent hypercalcemia (usually primary hyperparathyroidism), and a suppressed PTH value supporting the diagnosis of a PTH-independent hypercalcemia (most often a MAH or a calcitriol-excess hypercalcemia)[1-4]. In the case described, the coexistence of severe hypercalcemia with a PTH concentration in the normal reference range suggested a more complex diagnosis.
Among the potential causes of hypercalcemia, granulomatous diseases, familial hypocalciuric hypercalcemia and drug-induced HCa (e.g., lithium) were excluded on the basis of the medical and pharmacological history, and first-line investigations[1-3]. The laboratory assessment, undertaken during in-hospital stay, excluded hyperthyroidism, tertiary hyperparathyroidism and other rare disorders such as milk-alkali syndrome[1-3]. Thus, even on the basis of their higher prevalence, primary hyperparathyroidism and MAH were considered the potential cause of HCa in the 83-year-old man described.
The presence of a really high level of calcium with a PTH value within the reference range suggested the possibility of the coexistence of a primary hyperparathyroidism and another not-PTH-mediated hypercalcemic disorder, which, by increasing serum calcium to a level close to the PTH-secretion set-point, was capable of inhibiting secretion of the parathyroids, thus explaining the “atypical” normal PTH concentration. Apart from the diagnosis of lymphoma, which indirectly supported our hypothesis, the dramatic increase in PTH concentration after the infusion of pamidronate further supported our diagnoses. It is probable that pamidronate, by reducing osteoclast-mediated bone resorption and therefore calcium mobilization from the bone tissue, re-established PTH secretion thus reducing serum calcium and its inhibitory action on the parathyroids.
As we also found severe hypovitaminosis D, we also considered the hypothesis that the presence of a secondary hyperparathyroidism could induce an abnormal level of PTH in the presence of MAH. In fact, it is known that the normalization of PTH in secondary hyperparathyroidism, during supplementation with vitamin D, takes a long time, and that even after one year of treatment some patients still have high levels of PTH. This situation is considered to be related to hyperplasia of the parathyroids or to morphological modifications of these glands produced by the long-lasting hypocalcemic stimulus[9]. However, it is commonly believed that the response of the parathyroid glands to a hypercalcemic stimulus is quite fast and relevant, even in the presence of hyperplastic glands as demonstrated by Messa et al[10].
Another potential confounder in the clinical presentation of our case was the low magnesium concentration. Magnesium is essential for PTH secretion, and hypomagnesemia has been shown to blunt PTH increase in conditions such as severe vitamin D deficiency[11,12]. In our case, a rapid and sustained increase in PTH concentration occurred in the absence of magnesium supplementation, suggesting that magnesium deficiency was not the reason for the normal PTH level associated with severe HCa.
Hyponatremia was related to severe dehydration and to loss of urine sodium due to hypercalciuria, and a diagnosis of inappropriate secretion of antidiuretic hormone (trazodone, lymphoma) was excluded. This was also supported by the fact that hyponatremia resolved once the patient was re-hydrated and serum calcium was normalized.
In summary, the case described has some clinical implications: in patients with primary hyperparathyroidism, the coexistence of severe hypercalcemia which is not PTH-mediated, represents a challenge in the differential diagnosis of HCa; therefore, the presence of very high calcium concentrations with normal PTH values should suggest the coexistence of more than one disease producing hypercalcemia; when a patient, specially an older adult, is admitted to an Acute Care Unit or an Emergency Department with worsening cognitive impairment, delirium and asthenia, the calcium concentration should be measured together with routine laboratory and radiologic evaluations.
Peer reviewer: Tim Van den Wyngaert, Dr., Antwerp University Hospital, Wilrijkstraat 10, 2650 Edegem, Belgium
S- Editor Yang XC L- Editor Webster JR E- Editor Zhang DN
| 1. | Silverber SJ, Bilezikian JP. Primary hyperparathyroidism. 7th ed. Primer on the metabolic bone diseases and disorders of mineral metabolism. Washington DC: American Society for Bone and Mineral Research 2008; 301-306. |
| 2. | Horwitz MJ, Hodak SP, Stewart AF. Non-parathyroid hypercalcemia. 7th ed. Primer on the metabolic bone diseases and disorders of mineral metabolism. Washington DC: American Society for Bone and Mineral Research 2008; 307-312. |
| 3. | Moe SM. Disorders involving calcium, phosphorus, and magnesium. Prim Care. 2008;35:215-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 241] [Cited by in RCA: 195] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 4. | Makras P, Papapoulos SE. Medical treatment of hypercalcaemia. Hormones (. Athens). 2009;8:83-95. [PubMed] |
| 5. | Pellitteri PK. Evaluation of hypercalcemia in relation to hyperparathyroidism. Otolaryngol Clin North Am. 2010;43:389-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 6. | Balasanthiran A, Sandler B, Amonoo-Kuofi K, Swamy R, Kaniyur S, Kaplan F. Sarcoid granulomas in the parathyroid gland - a case of dual pathology: hypercalcaemia due to a parathyroid adenoma and coexistent sarcoidosis with granulomas located within the parathyroid adenoma and thyroid gland. Endocr J. 2010;57:603-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Fanari Z, Kadikoy H, Haque W, Pacha O, Abdellatif A. Medical management of primary hyperparathyroidism with concomitant multiple myeloma. Intern Med. 2010;49:581-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 8. | Richey DS, Welch BJ. Concurrent primary hyperparathyroidism and humoral hypercalcemia of malignancy in a patient with clear cell endometrial cancer. South Med J. 2008;101:1266-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Giusti A, Barone A, Pioli G, Girasole G, Razzano M, Pizzonia M, Pedrazzoni M, Palummeri E, Bianchi G. Heterogeneity in serum 25-hydroxy-vitamin D response to cholecalciferol in elderly women with secondary hyperparathyroidism and vitamin D deficiency. J Am Geriatr Soc. 2010;58:1489-1495. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Messa P, Sindici C, Cannella G, Miotti V, Risaliti A, Gropuzzo M, Di Loreto PL, Bresadola F, Mioni G. Persistent secondary hyperparathyroidism after renal transplantation. Kidney Int. 1998;54:1704-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 131] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 11. | Rude RK, Oldham SB, Singer FR. Functional hypoparathyroidism and parathyroid hormone end-organ resistance in human magnesium deficiency. Clin Endocrinol (Oxf). 1976;5:209-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 186] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 12. | Sahota O, Mundey MK, San P, Godber IM, Hosking DJ. Vitamin D insufficiency and the blunted PTH response in established osteoporosis: the role of magnesium deficiency. Osteoporos Int. 2006;17:1013-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 101] [Article Influence: 5.3] [Reference Citation Analysis (0)] |