Published online Jul 24, 2022. doi: 10.5306/wjco.v13.i7.553
Peer-review started: March 9, 2022
First decision: April 17, 2022
Revised: May 10, 2022
Accepted: June 27, 2022
Article in press: June 27, 2022
Published online: July 24, 2022
Processing time: 134 Days and 22.6 Hours
Cancer is a leading cause of death worldwide. Nowadays, the therapies are inadequate and spur demand for improved technologies. Rapid growth in nanotechnology and novel nanomedicine products represents an opportunity to achieve sophisticated targeting strategies and multi-functionality. Nanomedicine is increasingly used to develop new cancer diagnosis and treatment methods since this technology can modulate the biodistribution and the target site accumulation of chemotherapeutic drugs, thereby reducing their toxicity. Cancer nanotechnology and cancer immunotherapy are two parallel themes that have emerged over the last few decades while searching for a cure for cancer. Immunotherapy is revolutionizing cancer treatment, as it can achieve unprecedented responses in advanced-stage patients, including complete cures and long-term survival. A deeper understanding of the human immune system allows the establishment of combination regimens in which immunotherapy is combined with other treatment modalities (as in the case of the nanodrug Ferumoxytol). Furthermore, the combination of gene therapy approaches with nanotechnology that aims to silence or express cancer-relevant genes via one-time treatment is gradually progressing from bench to bedside. The most common example includes lipid-based nanoparticles that target VEGF-Α and KRAS pathways. This review focuses on nanoparticle-based platforms utilized in recent advances aiming to increase the efficacy of currently available cancer therapies. The insights provided and the evidence obtained in this paper indicate a bright future ahead for immuno-oncology applications of engineering nanomedicines.
Core Tip: Despite many years of fundamental and clinical examination and preliminaries of promising new treatments, cancer stays a significant reason for dreariness and mortality. Ongoing investigations propose that nanomedicine gives benefits over conventional treatments for cancer therapy. Immunotherapeutic strategies, such as cancer vaccines, immunomodulatory agents, immune checkpoint inhibitors, natural killer cells, peptides, nucleic acids, and chimeric antigen receptor T-cells, have augmented the development of this treatment either by stimulating cells or blocking the so-called immune checkpoint pathways. The efficacy of nanomedicine treatments and the examination of the advancement in the synergistic plan of immune-targeting combination therapies reviewed in this manuscript have been validated in clinical trials. The field of nanomedicine, therefore, generates new approaches regarding oncologic malignancies.
- Citation: Nteli P, Bajwa DE, Politakis D, Michalopoulos C, Kefala-Narin A, Efstathopoulos EP, Gazouli M. Nanomedicine approaches for treatment of hematologic and oncologic malignancies. World J Clin Oncol 2022; 13(7): 553-566
- URL: https://www.wjgnet.com/2218-4333/full/v13/i7/553.htm
- DOI: https://dx.doi.org/10.5306/wjco.v13.i7.553
Cancer refers to a diverse group of more than 100 different diseases that exhibit a highly complex and multifactorial profile and together serve as one of the leading causes of death worldwide, accounting for nearly 10 million deaths in 2020 alone[1]. The global prevalence of cancer was estimated to rise from 17 million cases annually in 2018 to 27.5 million in 2040[2]. Although the etiology of each type is hugely varying and the clinical manifestations quite heterogeneous, aberrated cellular and tissue regulation is among the trademarks of cancer[3]. Fundamentally, each type results in the accumulation of genetic and epigenetic alterations that dysregulate the cell cycle and promote abnormal cell growth[4].
Collectively, these alterations impair cellular control mechanisms and the responsible regulatory signaling pathways and drive the transformation of normal cells into malignant cells. The malignant cells acquire new biological abilities, referred to as the hallmarks of cancer[5], a set of distinct features including sustained proliferation, evasion of growth suppression and cell death, altered response to metabolic and stress cues, vascularization, invasion and metastasis, and immune modulation. Various genes have been implicated in carcinogenesis, from activated oncogenes and anti-apoptotic genes to inactivated tumor suppressor genes[6,7]. Large-scale genomic analysis and functional studies have facilitated the identification of distinct mutations in different tumor types, allowing the development of valuable diagnostic biomarkers[8] and the stratification of patients towards more personalized therapeutic approaches[9].
The field of oncology focuses on the prevention, diagnosis, and treatment of cancer while implementing various strategies and tools for clinical application. Conventional cancer therapeutics, including chemotherapy, radiation therapy, and surgery, are widely accepted and used; however, they suffer from various drawbacks due to the lack of tumor-specific targeting, dosing, pharmacokinetic limitations, and severe complications[10]. Concurrently, a tremendous body of research work has been generated over the last decades to advance on the one hand, the understanding of the complicated events governing tumorigenesis[11] and, on the other hand, to develop early diagnostic and efficient therapeutic approaches[12]. Towards that goal, a promising branch of biomedicine, nanomedicine, aims to boost these current cancer management strategies. Nanomedicine can be defined as the use of nanomaterials (materials with at least one dimension ranging from 1-100 nm) for the prevention, diagnosis, and ultimately, treatment of diseases[13].
Nanomedicine, the application of nanotechnology in medical practice, aims to bridge the gap between different scientific principles such as physics, chemistry, pharmaceutics, and biology to harness nanotechnology's knowledge and tools with the goal of serving medicine. The unique phenomena that govern the nanoscale enable novel medical applications and are responsible for the exceptional properties that make nanomaterials excellent candidates for therapeutic applications[14]. Despite their minuscule size, nanoparticles hold great potential as drug delivery systems for cancer treatment, and tremendous research has taken place in the last decades to bring this technology from bench to bedside[15].
Nanoparticles as drug carriers have proven to be an effective tool in the fight against cancer[16]. The improved selectivity afforded by these nanocarriers resulted in a significant increase in the efficacy of the carried medicine, while side effects in the host were minimized. It is also feasible to include targeting moieties specific for cell organelles, which boosts the efficacy of the transported medicines even more[17]. Nanoscale platforms come in various sizes, geometries, materials, and targeting moieties, allowing them to target organs, tissues, and individual cells[18]. Because of their distinct benefits, nanomedicines have emerged as a viable alternative to viral vectors, including low toxicity and immunogenicity, sustained and controlled release features, scale-up capacity, and low-cost manufacturing[17].
This review thus focuses on nanoparticle-based platforms utilized in recent advances aimed to increase the efficacy of currently available cancer therapies.
Recently, some of the alternate approaches to treat cancer are based on immunomodulation which employs the host’s own natural defense mechanisms to recognize and selectively eliminate the cancer cells by inducing the immune system[19]. Nanomaterial-mediated immunomodulation can be achieved either directly or indirectly[20]. To the first group belong nanomaterials that act as vaccine adjuvants, as several systems have been reported to improve antigenicity of conjugated weak antigens, while engineered nanosystems have also shown inherent antigenic properties[21]. Recent studies have highlighted the inherent tendency of liposomes to interact extensively with the immune system leading to several immunomodulatory effects, concerning tumor growth[22]. More specifically, circulating proteins are rapidly integrated to the surface of liposomes, forming a protein corona which can function as the interface for biological interactions and contributes to the formation of immune complexes and immunogenic epitope generation from self-antigens, ultimately resulting in the activation or suppression of immune responses[22]. Moreover, increasing evidence is emerging that indicates the functional ability of nanoparticles to polarize macrophages[23]. On the other hand, the multicomponent cargo capacity of delivering immunomodulatory agents in a targeted manner enables their function as delivery platforms that bolster the immune response.
Νanomaterials used in the combating of the immune evasion strategies of cancer operate in three different approaches that include the immunogenic targeting of cancer cells, the reshaping of the tumor’s immune microenvironment, and the stimulation of the peripheral immune network[24].
When targeting cancer cells, nanomedicines typically aim to induce immunogenic cell death (ICD), thereby triggering an immunogenic cascade that leads to an antigen-specific immune response against a broad spectrum of solid tumors. It is now established that ablative cancer treatments, such as radiotherapy, photodynamic therapy, hyperthermia, and photothermal therapy, as well as certain chemotherapeutics can cause tumor cell death[11].
In the context of tumor immune microenvironment (TIME), nanomaterials can be used to modulate the immunosuppressive tumor microenvironment by targeting tumor-associated macrophages (TAMs), regulatory T cells (Treg cells), regulatory B cells, myeloid-derived suppressor cells (MDSCs), as well as cancer-associated fibroblasts. Several nanoparticle-based strategies that target TAMs for suppressing tumor progression include TAM depletion, inhibiting monocyte recruitment, and TAM reprogramming[25]. Recent studies showed that the utilization of dendrimer nanoparticles carrying the chemotherapeutic methotrexate that specifically recognize the folate receptor-2, which is overexpressed in TAMs, increases therapeutic efficacy by depleting TAMs[25]. Considering monocyte recruitment, it is reported that silver nanoparticles have an adjuvant effect inducing recruitment and activation of local macrophages[26]. As for the reprogramming of macrophages, there have been attempts for creating an albumin-derived nanoplatform that delivers both the disulfiram/copper complex and macrophage modulator regorafenib for reprogramming macrophage[27]. In the context of down-regulating Treg cells, a common strategy is the use of checkpoint blockade antibodies (anticytotoxic T lymphocyte-associated protein 4)[25]. A modulating strategy for abnormal MDSC differentiation has been introduced, using lipid-coated biodegradable hollow mesoporous silica nanoparticles[28] in order to induce differentiation of MDSCs to mature DCs, macrophages, and granulocytes.
Nanomedicines can furthermore be applied in cancer vaccination to target the peripheral immune system[29]. This application’s grounds are based on the notions that intradermally or subcutaneously injected nanoparticles drain to LNs and that antigens bound to a nanoparticle are more efficiently processed by APCs. Instead of triggering APCs to present antigens to naive T cells, nanomedicines have also been designed to replace APCs by directly generating cytotoxic T cells[29].
During the last decade, nanoparticle-based immunotherapy formulations have passed from the pre-clinical stage in the clinical trials and several new treatments have been approved. Ferumoxytol is a nanoparticle formulation that contains iron oxide cores that are coated with carboxymethyl dextran. It enhances the production of reactive oxygen species by macrophages via the Fenton reaction, as M1 macrophages release hydrogen peroxides[30]. As an outcome, cancer cell cytotoxicity is enhanced while continued M1 polarization triggered by apoptotic cancer cells creates an autocrine feedback loop that maintains the production of tumor necrosis factor. Because ferumoxytol is FDA-approved, the drug is accessible for cancer patients through ‘off-label’ use[30]. The only cancer vaccines currently in routine clinical use are the Sipuleucel-T and the Talimogene laherparepvec (T-VEC). The FDA-approved nanomedicine for the treatment of prostate cancer is sipuleucel-T which is a personalized vaccine encompassing patients’ ex vivo processed dendritic cells that express a key tumor antigen, prostatic acid phosphatase (PAP)[31,32]. T-VEC is an engineered oncolytic herpes simplex virus type 1 in which the neurovirulence factor ICP34.5 is replaced by the coding sequence for GM-CSF and acts as a single agent in patients with skin and soft tissue metastases[32]. GM-CSF functions to recruit antigen presenting cells to the tumor microenvironment and promote cytotoxic T-cell responses to tumor associated antigens.
The last decade cancer immunotherapies have changed the perspective of cancer treatment (Table 1). The basic immunotherapy options approved are immune checkpoint inhibitors (ICIs).
| Anti-CTLA-4 antibodies |
| Ipilimumab; Colorectal cancer; Melanoma; Renal cell carcinoma |
| Anti-PD-1 antibodies |
| Nivolumab; Bladder cancer; Colorectal cancer; Head and neck cancer; Hepatocellular carcinoma; Hodgkin lymphoma; Melanoma; Non-small-cell lung cancer; Renal cell carcinoma; Cemiplimab; Cutaneous squamous cell carcinoma; Pembrolizumab; Bladder cancer; Cervical cancer; Gastro-oesophageal junction cancers; Head and neck cancer; Hepatocellular carcinoma; Hodgkin lymphoma; Merkel cell carcinoma; Metastatic solid tumours classified as microsatellite instability high or deficient mismatch repair; Non-small-cell lung cancer; Primary mediastinal large B cell lymphoma; Stomach cancer |
| Anti-PD-L1 antibodies |
| Atezolizumab; Bladder cancer; Breast cancer; Non-small-cell lung cancer; Avelumab; Bladder cancer; Merkel cell carcinoma; Durvalumab; Bladder cancer; Non-small-cell lung cancer |
ICIs have stirred up the field of tumour therapy and are now considered first-line therapies for various solid and liquid tumours. The approval of anticytotoxic T lymphocyte-associated protein 4 for advanced stage melanoma in 2011, opened up a new field of exploration that led to the 2018 Nobel Prize in Medicine to James P. Allison and Tasuku Honjo for inhibiting negative immune regulation in cancer[33].
Cancer immunotherapies are defined as therapies that directly or indirectly target any component of the immune system that is involved in the anti-cancer immune response, including the stimulation, enhancement, suppression, or desensitization of the immune system. These therapies are composed of monoclonal antibodies targeting the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and PD-1 receptors and the PD-1 ligand PD-L1, which are involved in the regulation of T cells. As shown in Table 1, there is a plethora of ICIs approved for the treatment of various cancer.
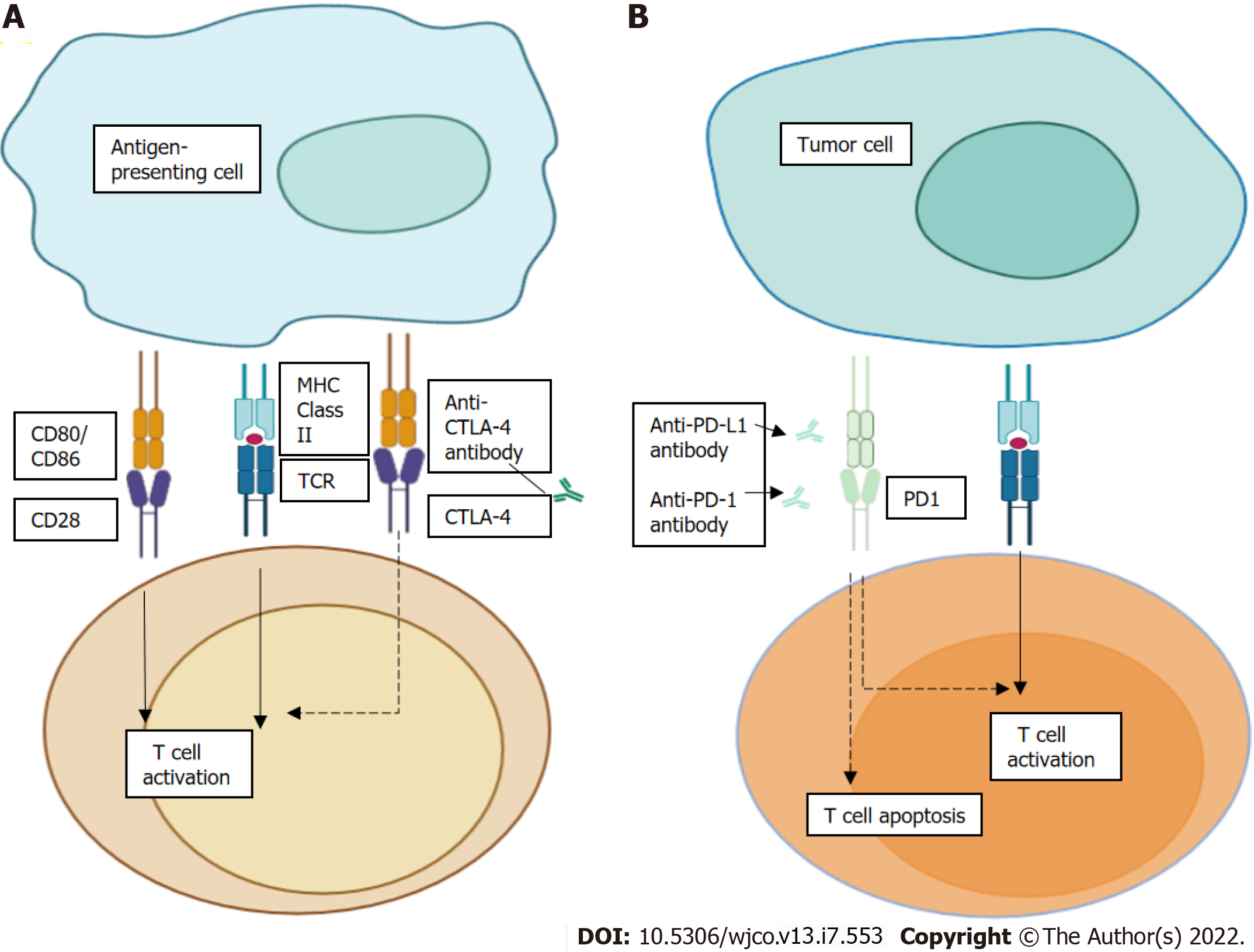
As a general concept, T cell activation needs two signals, first theantigen recognition by the T cell receptor (TCR), and then the antigen presentation by major histocompatibility complex class II molecules on the surface of antigen-presenting cell that leads to signal modulation by CD80 or CD86 binding to the CD28 receptor[34].
CTLA-4 is found on the T cell surface competing with the CD28 receptor to bind CD80 or CD86, thereby blocking T cell activation. Furthermore, CTLA-4 inhibitors block CTLA-4-CD80 or CTLA-4-CD86 binding to facilitate T cell activation (Figure 1A). In Figure 1B, we see PD-1 as a surface receptor that is expressed by T cells and promotes apoptosis of antigen-specific T cells and reduces apoptosis of regulatory T cells through its interaction with its ligand, PD-L1, which is expressed by tumour cells and myeloid cells[35,36]. This interaction is useful in preventing autoimmunity in physiological conditions, but cancer cells exploit this process to escape from immune system activity, upregulating PD-L1 expression[37,38]. PD-1 and PD-L1 inhibitors disrupt the PD-1-PD-L1 interaction, facilitating T cell activation and survival (dashed lines).
The use of ICIs for cancer therapy is increasing; however, only a minority of patients treated with ICIs achieve a durable response. A portion of patients that receive ICIs do not respond to treatment, while others respond initially but ultimately acquire resistance. Primary and acquired resistance are the effect of constantly changing interactions among cancer cells and the immune system. Even in patients with melanoma, which has one of the highest rates of response to ICI, 60%-70% of patients do not experience an objective response to anti-PD-1 therapy[39,40]. Moreover, 20%-30% of patients demonstrate eventual tumour relapse and progression. A key challenge that has emerged with the progressive implementation of ICIs in clinical practice is their uncontrolled collateral effects on the immune system that can lead to so-called immune-related adverse events (irAEs).
ICIs have a different spectrum of toxicities[41] from standard chemotherapy or other biological agents, and most toxicities result from excessive immunity against normal organs. All the primary and secondary (acquired) resistance are a result of complex and constantly evolving interactions between cancer cells and the immune system. The most frequently noted irAEs involve inflammation of gastrointestinal, dermatologic, endocrine, or pulmonary organs. Several clinical trials for ICIs including adjuvant and neo-adjuvant therapies are still in progress.
The role of nanomedicine in ICIs is to ensure an increased therapeutic outcome by using specific nanocarriers. Several formulations are currently investigated in both pre-clinical and clinical studies[42]. Starting from the preclinical studies, at least 12 different nanocarriers are being investigated. These include gold nanostars that are being tested for the PD-L1 blockage, PLGA combined with anti-CTLA4, and incorporation of anti-PD-1 and anti-TIM-3 with liposomes[43-46]. All these preclinical studies are held in mouse models for bladder, breast (4T1 cells), and colon cancer.
An important number of clinical trials are also being performed. These mainly include Nanoparticle Albumin Bound (Nab) formulations that combine ICIs with standard chemotherapeutics (as in the case of paclitaxel and carboplatin in Pembrolizumab, Atezolizumab, and Nivolumab formulations). Non-Nab strategies in nanoparticle-based immunotherapy include the radiosensitizer molecule NBTXR3[42].
Most of these studies will require a considerable amount of time to be completed and for the efficacy of these nanoformulation to be investigated. The poor lymphatic drainage of tumours (part of a phenomenon known as the EPR effect) could theoretically offer an advantage of nanoformulations over conventional ones although due to its complicated nature of EPR, this needs to be validated in these studies[47,48]. At the same time, further considerations are constantly being applied for future applications as the concept of smart nanoplatforms that will be triggered only upon an external stimulus[49-51].
Cancer immunotherapy is considered to eliminate primary as well as metastatic tumors and it is shown to develop immunological memory. It is important to say that nanomedicine can deliver a vast number of immunological agents to the targeted site (i.e., tumor)[52]. Nanomedicines have been explored thoroughly for tumor-targeted drug delivery and reducing the side effects of chemotherapeutic drugs. Tumor targeting is mainly mediated by passive targeting and/or active targeting and has been evaluated based on the average targeting efficiency and clinical impact (Table 2). There are three targeting strategies to boost cancer immunotherapy, including targeting and killing cancer cells to induce specific forms of ICD, TIME, and targeting the peripheral immune system[53].
| Condition | Interventions | Phase | Ref. | Status |
| Solid tumor | ROBO1 CAR-NK cells | I/II | NCT03940820 | Recruiting |
| Ewing sarcoma; Neuroblastoma; Rhabdomyosarcoma; Osteosarcoma; CNS tumors | Allogeneic HCT; Donor NK cell infusion | II | NCT02100891 | Active, not recruiting |
| Brain and CNS tumors; leukemia; lymphoma; chronic myeloproliferative disorders; lymphoproliferative disorder multiple myeloma and plasma cell neoplasm; myelodysplastic syndrome; myelodysplastic/ myeloproliferative neoplasm; unspecified adult solid tumor, protocol specific | Donor NK cell infusion | I/II | NCT00823524 | Completed |
| Malignant solid tumors | NK Immunotherapy | II | NCT02853903 | Completed |
| Malignant solid tumors | NK Immunotherapy | I/II | NCT02857920 | Completed |
| Multiple myeloma | CIML NK cells plus KP1237 and low dose IL-2 | I/II | NCT04634435 | Recruiting |
| Hematological malignancy; | NK cell infusion | I | NCT01853358 | Completed |
| leukemia; lymphoma; myeloma; Hodgkin's disease | NK-92 cells | I | NCT00990717 | Completed |
| Acute lymphoblastic leukemia; chronic lymphoblastic leukemia; B-cell lymphoma | Fludarabine + Cyclophosphamide + CAR-NK-CD19 Cells | I | NCT04796688 | Recruiting |
| Leukemia; lymphoma | NK cell infusion | I | NCT01287104 | Completed |
Natural killer (NK) cells, part of the innate immune system, have been identified as the next-generation therapy for cancer. These cells are lymphocytes with antitumor and antiviral abilities that have several applications. NK cells have memory-like and memory responses after cytokine preactivation, viral infections, and hapten exposure, in addition to being classified as innate lymphoid cells[54]. They have various mechanisms for directly killing cancer cells and enhancing the immune system's ability to fight cancer. Over the last 40 years, NK cell immunotherapy has demonstrated encouraging effects in both preclinical and clinical studies. These cells have been used for years and have been approved by the FDA. The NK-92 cell line (CD56+/CD3-), isolated from a patient with lymphoma, has expected high cytotoxic movement and can be extended under acceptable assembling practice conditions in recombinant interleukin-2[55].
Many nanoparticles have been discovered to be immunotherapy carriers, delivering antitumor immunotherapeutics specifically to tumor cells. These nanoparticles could provide stability, increase solubility, and cause less toxicity to healthy cells. Nanoparticles have the potential to deliver immunotherapeutics directly to cancer sites, which can be explained by their increased duration in the bloodstream without altering the body's physiochemical properties. The lymphoid node secures the nanoparticles prior to their drug conveyance priority and the elimination of toxic waste products. When nanoparticle immunotherapy is used passively to target cancer, there is a significant reduction in cellular cytotoxicity and a favorable outcome. Thus, to achieve an effective outcome, the delivery system must be modified so that the immunotherapeutic carrier enters the intracellular space before accomplishing the immunotherapy. There are several types of nanoparticles, which are classified based on their size, morphology, and physical and chemical properties[56]. Nanotechnology, specifically nanoparticles as drug delivery systems (DDSs), eases targeted medicines and theragnostics. Most nanomedicines include a targeting element, but some do not, yet[57].
Magnetic nanoparticles, Fe3O4, were modified with meso-2,3-dimercaptosuccinic acid, as the affinity of the electron-rich carboxyl group was higher and the orbital in the Fe atom was empty. After obtaining CD56 antibody-modified Fe3O4 nanoparticles aided by a shorter co-culture period, NK-92 cell recruitment and infiltration into solid tumors were improved in the presence of a magnetic field. Biohybrid treatment with NK-CD56 nanoparticles effectively suppressed tumor growth and significantly prolonged the survival of cancer-bearing mice. Finally, by synergizing immune cells with a directional magnetic field that promotes infiltration into solid tumor tissue under magnetic resonance imaging control, antitumor efficiency is significantly improved. Magnetic nanoparticles and NK cells can be utilized for various biomedical applications, as they have proved to possess flexible characteristics to operate in biomedicine[58].
Gold nanoparticles (AuNPs) were coated with PEG and D-(β)-glucosamine, as glucose coating increases the cellular uptake of the nanoparticles. The K562 human erythroleukemia cell line (positive target) and the 888 human melanoma cell line (negative control) were co-cultured with AuNP-labeled NK-92 cells. The results indicate that AuNP-labeled NK-92 cells can specifically identify target cells and keep their cytokine secretion and antitumor function. In addition, gold nanoparticles do not undermine the therapeutic effect of NK-92 cells in vivo. AuNPs could assist NK cells to achieve their aim, the regression of the tumor; therefore, this combinatorial therapy for cancer will reduce or even end the dosage of radiation[59].
Worldwide, approximately 97% of active clinical trials are chimeric antigen receptor (CAR) T-cell-based therapies. Nanoparticles can engineer NK cells to produce CAR-NK therapy by targeting several ligands, such as antibodies to nanoparticles, and accomplish successful targeted delivery[60]. Axicabtagene ciloleucel (refractory diffuse large B-cell lymphoma) and tisagenlecleucel (B-cell precursor acute lymphoblastic leukemia) were two CAR T-cell therapies that cure blood cancer approved by the FDA[61]. Functional antitumor immune response has been shown by adoptive cell transfer studies, such as CAR T-cell therapy [62].
Peptides are a powerful tool in cancer diagnosis and treatment with many advantages and numerous ways to alter their function and use them in oncology. They present with excellent biocompatibility (degradation products are amino acids, which are a natural source of cells). They can be formulated and introduced with all kinds of modifications. By using the process of self-assembly, we can improve the stability of a peptide sequence and create the conditions for better targeting of the diseased organ. Their big advantage depends on their small size and better tissue/cell penetration[63,64]. To minimize the nonselective side effects of chemotherapy, a specific peptide sequence or motif can be used. Nanoparticles based on peptides, can be used to target cancer cells, to minimize systemic drug exposure and increase efficiency of the drug that is to be delivered[63].
Some examples of therapeutic peptides in clinical use nowadays are GnRH agonists for the treatment of prostate and breast cancer (e.g., Buserelin and Nafarelin), GnRH antagonists for the treatment of prostate and breast cancer (e.g., Cetrorelix and Abarelix), and somatostatin agonists for the treatment and diagnosis of GH-producing tumors (e.g., Ocreotide and Lanreotide). In the future, many more peptides will take part in the treatment process against oncology, such as Chlorotoxin and its analogue TM601 (phases I, II, and III clinical trials for diagnosis of glioma), BT1718 (phases I and II for treatment of solid tumors), and P28 (phase I for treatment of various solid tumors)[64].
Drug conjugates is a modern method of using peptides as a tool for drug delivery. They are chemotherapeutic or cytotoxic agents linked to an antibody or a peptide via a linker. They provide enhanced function, higher circulation time, and lower off-target toxicity (to healthy tissues)[65]. An example is the conjugation of paclitaxel to a peptide (Angiopep-2) via an ester/amide bond. Angiopep-2 goes into the cell via transcytosis and crosses the blood-brain barrier, thus facilitating the uptake of the conjugate into the brain for the treatment of patients with solid tumors and brain metastases. The esterase enzyme, which is present in lysosomes, breaks down the ester bond, thus releasing paclitaxel in the brain. In this way, ANG 1005 overcomes the main disadvantage of paclitaxel and gains access into the blood-brain barrier. ANG1005 has been studied in several clinical trials (phase I and phase II) in patients with metastatic brain cancers and the results have shown that it works well against CNS tumors, improves symptoms, and increases survival[66].
Peptide self-assembly is a process in which peptides spontaneously or by a trigger form aggregates. In that form, the transport mechanism provides a higher efficiency of drug loading with better molecule stability and a simultaneous lower ratio of drug loss[67]. The method uses monomers of short amino acid sequences or repeated amino acid sequences that assemble together to form nanostructures. The nanostructure can be made by various building blocks such as dipeptides (the simplest form), surfactant-like peptides, and cyclic peptides[68]. The resultant nanostructure can take the form of nanofibers, nanotubes, micelles, and hydrogels[69-71].
Self-assembly of peptides is divided into spontaneous and trigger types. If the assembly happens in an aqueous solution, it is spontaneous. The peptide molecules that are dissolved in the aqueous solution form non-covalent interactions, such as hydrogen bonding bonds, van der Waals forces, electrostatic, and π-π stacking interactions[69]. If the process of assembly is driven by external factors and does not happen spontaneously, such as temperature, ion concentration, and pH changes, it is called trigger aggregation. The above-mentioned nanostructure can be used for drug delivery, drug stabilization, crossing the blood-brain barrier, neuronal or liver cell regeneration, fibroblast migration, etc[69].
Albeit their central role in governing cell physiology, nucleic acids had not been considered as possible drug candidates until relatively recently when successful protein production was demonstrated upon in vivo administration[70]. Since then, a novel class of drugs, referred to as nucleic acid therapeutics, have emerged[71]. Conventional therapeutics generally exhibit a transient effect and exert their action via protein targeting. This action mode poses significant disadvantages as only a fraction of human proteins can be targeted by pharmaceutical compounds[72]. Combined with the limitations of conventional therapeutics in oncology discussed above, gene therapy offers a promising approach as a one-time treatment targeting the route of the disease - genetics - while contributing to long-standing therapeutic outcomes with high specificity[73]. Developments in nucleic acid design and chemical modifications have assisted in overcoming stability, toxicity, and immunogenicity issues[74-76], and by further harnessing the power of nanomaterials, nucleic acid therapeutics can be loaded into nanocarriers to formulate DDSs with enhanced pharmacokinetic properties[77].
Due to the arduous nature of the causation and phenotype of cancer, it is evident that nucleic acid-based therapeutics must implement a plethora of strategies via different modes of action to target relevant genes and their products in cancer cells or stimulate an immune response against them[78]. Table 3 summarizes the current status of nucleic acid nanomedicines available for cancer treatment. The first strategy implemented in oncology utilizes antisense oligonucleotides (ASOs) and small interfering RNAs (siRNAs) to target disease-relevant mRNAs and inhibit their translation. ASOs are synthetic oligonucleotides complementary to a gene of interest that bind on the pre-mRNA or mRNA of the target gene, hindering cellular post-transcriptional and translational machinery and eventually leading to altered splicing patterns or gene silencing, respectively[79,80]. Lipid nanoparticle (LNP)-based ASOs are under clinical evaluation to treat leukemia[78,81] and solid tumors[81] via targeting Grb2. Furthermore, targeting the anti-apoptotic gene Bcl-2 is also being examined as a possible target in patients with advanced lymphoid malignancies[82].
| Name | Category | Structure | Mode of action | Status |
| ASO | Inhibition of translation of cancer or angiogenesis associated proteins | Synthetic ssDNA or ssRNA oligos complementary to mRNA of interest | Rnase H mediated mRNA degradation | In clinical trials; LNP-based anti-Grb2 ASOs for leukemia[70] and solid tumors[71]; LNP-based anti-Bcl-2 ASOs for advanced lymphoid malignancies[72] |
| siRNA | Inhibition of translation of cancer or angiogenesis associated proteins | Synthetic dsRNA oligos complementary to mRNA of interest | Dicer induces cleavage of dsRNA and RNA-induced silencing complex mRNA degradation | In clinical trials; Polymeric anti-KRAS siRNAs for pancreatic ductal adenocarcinoma[78]; LNP based anti-PKN3 siRNAs in patients with advanced solid tumors[79]; LNP based anti-KSP and anti-VEGF-A siRNAs in patients with solid tumors[80,81]; LNP based anti-PLK1 siRNAs in patients with solid tumors[82] |
| saRNA | Forced exogenous gene expression | Synthetic dsRNA oligos complementary to mRNA of interest | Target gene promoters to induce transcriptional gene activation | In clinical trials; LNP based formulations for treatment of hepatocellular carcinoma[84] and advanced solid tumors[85] |
| miRNA mimics | Regulation of post- transcriptional mRNA expression | Chemically modified dsRNA molecules designed to mimic endogenous microRNAs | Translational repression and gene silencing | Currently only in basic research[87] |
| mRNA vaccines | Forced exogenous antigen expression | Synthetic mRNA | Induction of immune response against cancer cells | In clinical trials; LNP-based mRNA vaccines encoding known tumor-specific antigens are being investigated in early phase clinical trials in patients with HPV-driven squamous cell carcinoma[90], melanoma[90], ovarian[92], pancreatic, lung, and colorectal cancer[93]; Personalized vaccines based on patient specific neo-antigens are being assessed clinically for the treatment of melanoma[95] and breast cancer[96] |
siRNAs are a class of double-stranded RNA molecules involved in the biological process of RNA interference that regulates gene expression[83]. By administering a siRNA complementary to its target mRNA, this natural process is harnessed to selectively silence genes via multiprotein complexes[84]. Activation of the oncogene KRAS is a hallmark of pancreatic ductal adenocarcinoma, the most common type of pancreatic cancer[85]. siG12D-LODER is a biodegradable polymer-based system loaded with siRNA against KRAS, which has completed phase I and is currently being tested in phase II in combination with chemotherapy to determine treatment efficacy[86-88]. Other delivery systems based on LNPs have also completed or are currently being tested in phase I trials against advanced solid cancers in various modalities targeting tumor proliferation or microenvironment[89-92].
Additional strategies based on small activating RNA molecules aim to upregulate the expression of physiologic master gene regulators[93] and are undergoing pre-clinical development for the treatment of hepatocellular carcinoma[94] and advanced solid tumors[95]. Moreover, since misregulated miRNA expression is another feature of cancer, miRNA mimics are also being developed to mimic endogenous miRNAs and restore physiological expression levels[96,97].
The rise of mRNA vaccines has led to a new era of cancer immunotherapy offering considerable benefits, including increased safety and efficacy with expeditious cost-effective manufacturing pipelines, aiming to elicit an immune response upon exposure to a tumor antigen[98,99]. Several LNP-based mRNA vaccines encoding known tumor-specific antigens are being investigated in early phase clinical trials in patients with HPV-driven squamous cell carcinoma, melanoma, ovarian, pancreatic, lung, and colorectal cancer[100-103]. Technological progress in next-generation sequencing has enormously facilitated the discovery of patient-specific neoantigens, novel epitopes arising from tumor-specific mutations that can be used as a template to generate personalized neoantigen vaccines[104]. Such vaccines are being assessed clinically for the treatment of melanoma and breast cancer[105,106].
The field of cancer nanomedicines is rapidly expanding and is expected to revolutionize available treatment options. Nanomaterial-mediated immunomodulation offers a dual aspect of immunomodulation therapies, as they can themselves act as immunomodulatory agents, or they can function as delivery platforms for targeted delivery of other immunomodulating agents[20]. Their unique and tunable properties can be utilized to target the cancer-immunity flow in multiple steps, offering advanced systems that pave a way to reshaping the landscape of clinical cancer treatment. ICIs have launched a new field far beyond CTLA-4 and PD-1. First of all, co-inhibitory signaling pathways, such as HVEM-BTLA and Galectin-9-TIM3, are being studied in cancer and other diseases[107]. Once we learn more about them, we may design rational combinational strategies to concurrently target two or more inhibitory pathways to gain better therapeutic efficacy. Moreover, good results are shown with the combination of immune checkpoint blockade with other immunotherapy regimens to eliminate primary cancer and metastases more effectively. One such strategy has been to combine anti-PD-1/PD-L1 or anti-CTL4 with oncolytic viruses[108,109]. Meanwhile, other types of cancer immunotherapies, including adoptive transfer of CAR T cells, TCR-modified T cells, and cancer vaccines using neo-antigens, have made significant progress in recent years and have shown promise in clinics[110-112]. Future NK cell products will be able to suppress inhibitory signals and tumor proliferation but enhance the activation of the immune system. Evidence of increased NK cell-mediated tumor cell killing has emerged in targeted therapies. To enhance that, nanomedicine approaches immunity with T-cell activation, specific antigen delivery, and the appropriate nanoparticle for the targeting[113]. Nanoparticles will tackle all the obstacles to delivery and engage multiple aspects of the immune system by producing therapeutics to target current and forthcoming diseases[114]. These are only a small portion of the application of nanoparticles with NK cells and their clinical activity because of the heterogeneity of human diseases[115]. These findings, combined with the ability of NK cells to detect immune responses, suggest that NK cells are the keys to the next-generation onco-immunotherapy. In the future days, peptides will play a significant role in the continuous research of cancer therapy and human well-being. Cell-penetrating peptides have the ability to deliver molecules such as drugs, oligonucleotides, and nanoparticles inside cells, without any size restriction[116].
Future research will set the basis for the ideal drug-delivery system, where peptides would reach their target site efficiently without any degradation before and the cargo would be rapidly released and act on the site. Also, the problem of non-selective cellular uptake will be eliminated and thus modern therapy tools for anti-cancer treatment will be created[117,118]. The nanoparticle-mediated delivery of guide RNAs and programmable nucleases such as Cas9 and Cas13 has expanded the portfolio of in vivo tissue-specific genome editing tools available for cancer research in pre-clinical models[119-122]. Alongside advancements in nucleic acid drugs, innovative nanoparticle delivery systems will vastly benefit the field by implementing novel delivery systems, such as nanoclews and surface modifications, allowing the manufacturing of sophisticated nanoparticles[123-127]. Despite the numerous nanotherapeutics being clinically scrutinized, cancer nanomedicines often fail to reach their primary endpoint, and the correlation of the drug behavior between animal models and patient cohorts is often inconsistent[128,129]. Therefore, in silico models should also be implemented to aid in understanding and predicting biological interactions[130]. Finally, multi-omics data, including but not limited to genomics, epigenomics, transcriptomics, and radiomics, can comprehensively be evaluated and reform the field of personalized nanomedicine by allowing the design of customizable medicines based on the patients' profile[131].
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: Greece
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Nath LR, India; Zhu CM, China S-Editor: Wang LL L-Editor: Wang TQ P-Editor: Wang LL
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75126] [Cited by in RCA: 64681] [Article Influence: 16170.3] [Reference Citation Analysis (177)] |
| 2. | Rahib L, Wehner MR, Matrisian LM, Nead KT. Estimated Projection of US Cancer Incidence and Death to 2040. JAMA Netw Open. 2021;4:e214708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 277] [Cited by in RCA: 790] [Article Influence: 197.5] [Reference Citation Analysis (0)] |
| 3. | Fouad YA, Aanei C. Revisiting the hallmarks of cancer. Am J Cancer Res. 2017;7:1016-1036. [PubMed] |
| 4. | Vogelstein B, Kinzler KW. The multistep nature of cancer. Trends Genet. 1993;9:138-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1217] [Cited by in RCA: 1129] [Article Influence: 35.3] [Reference Citation Analysis (0)] |
| 5. | Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51728] [Cited by in RCA: 47160] [Article Influence: 3368.6] [Reference Citation Analysis (5)] |
| 6. | Futreal PA, Coin L, Marshall M, Down T, Hubbard T, Wooster R, Rahman N, Stratton MR. A census of human cancer genes. Nat Rev Cancer. 2004;4:177-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2427] [Cited by in RCA: 2294] [Article Influence: 109.2] [Reference Citation Analysis (0)] |
| 7. | Vogelstein B, Kinzler KW. Cancer genes and the pathways they control. Nat Med. 2004;10:789-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2905] [Cited by in RCA: 2774] [Article Influence: 132.1] [Reference Citation Analysis (0)] |
| 8. | Poste G. Bring on the biomarkers. Nature. 2011;469:156-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 391] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 9. | Meric-Bernstam F, Brusco L, Shaw K, Horombe C, Kopetz S, Davies MA, Routbort M, Piha-Paul SA, Janku F, Ueno N, Hong D, De Groot J, Ravi V, Li Y, Luthra R, Patel K, Broaddus R, Mendelsohn J, Mills GB. Feasibility of Large-Scale Genomic Testing to Facilitate Enrollment Onto Genomically Matched Clinical Trials. J Clin Oncol. 2015;33:2753-2762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 338] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 10. | Damyanov CA. Conventional Treatment of Cancer Realities and Problems. Ann Complement Altern Med 2018; 1: 1002 . |
| 11. | Balani S, Nguyen LV, Eaves CJ. Modeling the process of human tumorigenesis. Nat Commun. 2017;8:15422. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 12. | Atlihan-Gundogdu E, Ilem-Ozdemir D, Ekinci M, Ozgenc E, Demir ES, Sánchez-Dengra B, González-Alvárez I. Recent developments in cancer therapy and diagnosis. J Pharm Investig. 2020;50:349-361. [RCA] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 13. | Soares S, Sousa J, Pais A, Vitorino C. Nanomedicine: Principles, Properties, and Regulatory Issues. Front Chem. 2018;6:360. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 394] [Cited by in RCA: 404] [Article Influence: 57.7] [Reference Citation Analysis (0)] |
| 14. | Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MDP, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S, Habtemariam S, Shin HS. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018;16:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3635] [Cited by in RCA: 3248] [Article Influence: 464.0] [Reference Citation Analysis (0)] |
| 15. | Aghebati-Maleki A, Dolati S, Ahmadi M, Baghbanzhadeh A, Asadi M, Fotouhi A, Yousefi M, Aghebati-Maleki L. Nanoparticles and cancer therapy: Perspectives for application of nanoparticles in the treatment of cancers. J Cell Physiol. 2020;235:1962-1972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 218] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 16. | Datta P, Ray S. Nanoparticulate formulations of radiopharmaceuticals: Strategy to improve targeting and biodistribution properties. J Labelled Comp Radiopharm. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 17. | Guo P, Huang J, Moses MA. Cancer Nanomedicines in an Evolving Oncology Landscape. Trends Pharmacol Sci. 2020;41:730-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 18. | Decuzzi P, Peer D, Mascolo DD, Palange AL, Manghnani PN, Moghimi SM, Farhangrazi ZS, Howard KA, Rosenblum D, Liang T, Chen Z, Wang Z, Zhu JJ, Gu Z, Korin N, Letourneur D, Chauvierre C, van der Meel R, Kiessling F, Lammers T. Roadmap on nanomedicine. Nanotechnology. 2021;32:012001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 19. | Jindal A, Sarkar S, Alam A. Nanomaterials-Mediated Immunomodulation for Cancer Therapeutics. Front Chem. 2021;9:629635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 20. | Kubackova J, Zbytovska J, Holas O. Nanomaterials for direct and indirect immunomodulation: A review of applications. Eur J Pharm Sci. 2020;142:105139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 21. | Debele TA, Yeh CF, Su WP. Cancer Immunotherapy and Application of Nanoparticles in Cancers Immunotherapy as the Delivery of Immunotherapeutic Agents and as the Immunomodulators. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 22. | La-Beck NM, Liu X, Wood LM. Harnessing Liposome Interactions With the Immune System for the Next Breakthrough in Cancer Drug Delivery. Front Pharmacol. 2019;10:220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 23. | Miao X, Leng X, Zhang Q. The Current State of Nanoparticle-Induced Macrophage Polarization and Reprogramming Research. Int J Mol Sci. 2017;18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 156] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 24. | Shi Y, Lammers T. Combining Nanomedicine and Immunotherapy. Acc Chem Res. 2019;52:1543-1554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 315] [Article Influence: 52.5] [Reference Citation Analysis (0)] |
| 25. | Jang H, Kim EH, Chi SG, Kim SH, Yang Y. Nanoparticles Targeting Innate Immune Cells in Tumor Microenvironment. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 26. | Xu Y, Tang H, Liu JH, Wang H, Liu Y. Evaluation of the adjuvant effect of silver nanoparticles both in vitro and in vivo. Toxicol Lett. 2013;219:42-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 27. | Zhao P, Yin W, Wu A, Tang Y, Wang J, Pan Z, Lin T, Zhang M, Chen B, Duan Y, Huang Y. Dual‐Targeting to Cancer Cells and M2 Macrophages via Biomimetic Delivery of Mannosylated Albumin Nanoparticles for Drug‐Resistant Cancer Therapy. Adv Funct Mater. 2017;27:1700403. [DOI] [Full Text] |
| 28. | Kong M, Tang J, Qiao Q, Wu T, Qi Y, Tan S, Gao X, Zhang Z. Biodegradable Hollow Mesoporous Silica Nanoparticles for Regulating Tumor Microenvironment and Enhancing Antitumor Efficiency. Theranostics. 2017;7:3276-3292. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 129] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 29. | Yang J, Wang C, Shi S, Dong C. Nanotechnologies for enhancing cancer immunotherapy. Nano Res. 2020;13:2595-2616. [DOI] [Full Text] |
| 30. | Zanganeh S, Hutter G, Spitler R, Lenkov O, Mahmoudi M, Shaw A, Pajarinen JS, Nejadnik H, Goodman S, Moseley M, Coussens LM, Daldrup-Link HE. Iron oxide nanoparticles inhibit tumour growth by inducing pro-inflammatory macrophage polarization in tumour tissues. Nat Nanotechnol. 2016;11:986-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 999] [Cited by in RCA: 1173] [Article Influence: 130.3] [Reference Citation Analysis (0)] |
| 31. | Anassi E, Ndefo UA. Sipuleucel-T (provenge) injection: the first immunotherapy agent (vaccine) for hormone-refractory prostate cancer. P T. 2011;36:197-202. [PubMed] |
| 32. | Johnson DB, Puzanov I, Kelley MC. Talimogene laherparepvec (T-VEC) for the treatment of advanced melanoma. Immunotherapy. 2015;7:611-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 138] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 33. | Guo ZS. The 2018 Nobel Prize in medicine goes to cancer immunotherapy (editorial for BMC cancer). BMC Cancer. 2018;18:1086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 55] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 34. | Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, Lambotte O, Mariette X, Prat A, Suárez-Almazor ME. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020;6:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 399] [Cited by in RCA: 897] [Article Influence: 179.4] [Reference Citation Analysis (0)] |
| 35. | Fife BT, Pauken KE. The role of the PD-1 pathway in autoimmunity and peripheral tolerance. Ann N Y Acad Sci. 2011;1217:45-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 268] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 36. | Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity. Immunol Rev. 2010;236:219-242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1835] [Cited by in RCA: 1775] [Article Influence: 118.3] [Reference Citation Analysis (0)] |
| 37. | Wang X, Teng F, Kong L, Yu J. PD-L1 expression in human cancers and its association with clinical outcomes. Onco Targets Ther. 2016;9:5023-5039. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 371] [Cited by in RCA: 597] [Article Influence: 66.3] [Reference Citation Analysis (0)] |
| 38. | Gandini S, Massi D, Mandalà M. PD-L1 expression in cancer patients receiving anti PD-1/PD-L1 antibodies: A systematic review and meta-analysis. Crit Rev Oncol Hematol. 2016;100:88-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 303] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 39. | Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L, Carcereny E, Ahn MJ, Felip E, Lee JS, Hellmann MD, Hamid O, Goldman JW, Soria JC, Dolled-Filhart M, Rutledge RZ, Zhang J, Lunceford JK, Rangwala R, Lubiniecki GM, Roach C, Emancipator K, Gandhi L; KEYNOTE-001 Investigators. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med. 2015;372:2018-2028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4095] [Cited by in RCA: 4862] [Article Influence: 486.2] [Reference Citation Analysis (0)] |
| 40. | Ott PA, Bang YJ, Piha-Paul SA, Razak ARA, Bennouna J, Soria JC, Rugo HS, Cohen RB, O'Neil BH, Mehnert JM, Lopez J, Doi T, van Brummelen EMJ, Cristescu R, Yang P, Emancipator K, Stein K, Ayers M, Joe AK, Lunceford JK. T-Cell-Inflamed Gene-Expression Profile, Programmed Death Ligand 1 Expression, and Tumor Mutational Burden Predict Efficacy in Patients Treated With Pembrolizumab Across 20 Cancers: KEYNOTE-028. J Clin Oncol. 2019;37:318-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 669] [Article Influence: 111.5] [Reference Citation Analysis (0)] |
| 41. | Yao L, Jia G, Lu L, Bao Y, Ma W. Factors affecting tumor responders and predictive biomarkers of toxicities in cancer patients treated with immune checkpoint inhibitors. Int Immunopharmacol. 2020;85:106628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 42. | Cremolini C, Vitale E, Rastaldo R, Giachino C. Advanced Nanotechnology for Enhancing Immune Checkpoint Blockade Therapy. Nanomaterials (Basel). 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 43. | Liu Y, Maccarini P, Palmer GM, Etienne W, Zhao Y, Lee CT, Ma X, Inman BA, Vo-Dinh T. Synergistic Immuno Photothermal Nanotherapy (SYMPHONY) for the Treatment of Unresectable and Metastatic Cancers. Sci Rep. 2017;7:8606. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 105] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 44. | Liu Y, Chongsathidkiet P, Crawford BM, Odion R, Dechant CA, Kemeny HR, Cui X, Maccarini PF, Lascola CD, Fecci PE, Vo-Dinh T. Plasmonic gold nanostar-mediated photothermal immunotherapy for brain tumor ablation and immunologic memory. Immunotherapy. 2019;11:1293-1302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 45. | Chen Q, Xu L, Liang C, Wang C, Peng R, Liu Z. Photothermal therapy with immune-adjuvant nanoparticles together with checkpoint blockade for effective cancer immunotherapy. Nat Commun. 2016;7:13193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 969] [Cited by in RCA: 1140] [Article Influence: 126.7] [Reference Citation Analysis (0)] |
| 46. | Huang TY, Huang GL, Zhang CY, Zhuang BW, Liu BX, Su LY, Ye JY, Xu M, Kuang M, Xie XY. Supramolecular Photothermal Nanomedicine Mediated Distant Tumor Inhibition via PD-1 and TIM-3 Blockage. Front Chem. 2020;8:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 181] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 47. | Shi Y, van der Meel R, Chen X, Lammers T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics. 2020;10:7921-7924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 234] [Cited by in RCA: 503] [Article Influence: 100.6] [Reference Citation Analysis (0)] |
| 48. | Subhan MA, Yalamarty SSK, Filipczak N, Parveen F, Torchilin VP. Recent Advances in Tumor Targeting via EPR Effect for Cancer Treatment. J Pers Med. 2021;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 279] [Article Influence: 69.8] [Reference Citation Analysis (0)] |
| 49. | Thakur N, Thakur S, Chatterjee S, Das J, Sil PC. Nanoparticles as Smart Carriers for Enhanced Cancer Immunotherapy. Front Chem. 2020;8:597806. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 50. | Shrestha B, Wang L, Brey EM, Uribe GR, Tang L. Smart Nanoparticles for Chemo-Based Combinational Therapy. Pharmaceutics. 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 51. | Kaushik N, Borkar SB, Nandanwar SK, Panda PK, Choi EH, Kaushik NK. Nanocarrier cancer therapeutics with functional stimuli-responsive mechanisms. J Nanobiotechnology. 2022;20:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 52. | Yu H, de Geest BG. Nanomedicine and cancer immunotherapy. Acta Pharmacol Sin. 2020;41:879-880. [DOI] [Full Text] |
| 53. | Sun Q, Bai X, Sofias AM, van der Meel R, Ruiz-Hernandez E, Storm G, Hennink WE, De Geest B, Kiessling F, Yu HJ, Lammers T, Shi Y. Cancer nanomedicine meets immunotherapy: opportunities and challenges. Acta Pharmacol Sin. 2020;41:954-958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 54. | Mikelez-Alonso I, Magadán S, González-Fernández Á, Borrego F. Natural killer (NK) cell-based immunotherapies and the many faces of NK cell memory: A look into how nanoparticles enhance NK cell activity. Adv Drug Deliv Rev. 2021;176:113860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 55. | Tonn T, Schwabe D, Klingemann HG, Becker S, Esser R, Koehl U, Suttorp M, Seifried E, Ottmann OG, Bug G. Treatment of patients with advanced cancer with the natural killer cell line NK-92. Cytotherapy. 2013;15:1563-1570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 294] [Cited by in RCA: 366] [Article Influence: 30.5] [Reference Citation Analysis (0)] |
| 56. | Muluh TA, Chen Z, Li Y, Xiong K, Jin J, Fu S, Wu J. Enhancing Cancer Immunotherapy Treatment Goals by Using Nanoparticle Delivery System. Int J Nanomedicine. 2021;16:2389-2404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 57. | McGoron AJ. Perspectives on the Future of Nanomedicine to Impact Patients: An Analysis of US Federal Funding and Interventional Clinical Trials. Bioconjug Chem. 2020;31:436-447. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 58. | Zhao S, Duan J, Lou Y, Gao R, Yang S, Wang P, Wang C, Han L, Li M, Ma C, Liang X, Liu H, Sang Y, Gao L. Surface specifically modified NK-92 cells with CD56 antibody conjugated superparamagnetic Fe3O4 nanoparticles for magnetic targeting immunotherapy of solid tumors. Nanoscale. 2021;13:19109-19122. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 59. | Shamalov K, Meir R, Motiei M, Popovtzer R, Cohen CJ. Noninvasive Tracking of Natural Killer Cells Using Gold Nanoparticles. ACS Omega. 2021;6:28507-28514. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 60. | El-Mayta R, Zhang Z, Hamilton AG, Mitchell MJ. Delivery technologies to engineer natural killer cells for cancer immunotherapy. Cancer Gene Ther. 2021;28:947-959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 61. | Abdou P, Wang Z, Chen Q, Chan A, Zhou DR, Gunadhi V, Gu Z. Advances in engineering local drug delivery systems for cancer immunotherapy. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2020;12:e1632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 62. | Acebes-Fernández V, Landeria-Viñuela A, Juanes-Velasco P, Hernández AP, Otazo-Perez A, Manzano-Román R, Gongora R, Fuentes M. Nanomedicine and Onco-Immunotherapy: From the Bench to Bedside to Biomarkers. Nanomaterials (Basel). 2020;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 63. | Li L, Ma B, Wang W. Peptide-Based Nanomaterials for Tumor Immunotherapy. Molecules. 2020;26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 64. | Kurrikoff K, Aphkhazava D, Langel Ü. The future of peptides in cancer treatment. Curr Opin Pharmacol. 2019;47:27-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 65. | Sorolla A, Sorolla MA, Wang E, Ceña V. Peptides, proteins and nanotechnology: a promising synergy for breast cancer targeting and treatment. Expert Opin Drug Deliv. 2020;17:1597-1613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 66. | Alas M, Saghaeidehkordi A, Kaur K. Peptide-Drug Conjugates with Different Linkers for Cancer Therapy. J Med Chem. 2021;64:216-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 151] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 67. | Wang Y, Cheetham AG, Angacian G, Su H, Xie L, Cui H. Peptide-drug conjugates as effective prodrug strategies for targeted delivery. Adv Drug Deliv Rev. 2017;110-111:112-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 377] [Article Influence: 47.1] [Reference Citation Analysis (0)] |
| 68. | Lee S, Trinh THT, Yoo M, Shin J, Lee H, Kim J, Hwang E, Lim YB, Ryou C. Self-Assembling Peptides and Their Application in the Treatment of Diseases. Int J Mol Sci. 2019;20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 112] [Cited by in RCA: 136] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 69. | Conibear AC, Schmid A, Kamalov M, Becker CFW, Bello C. Recent Advances in Peptide-Based Approaches for Cancer Treatment. Curr Med Chem. 2020;27:1174-1205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 70. | Wolff JA, Malone RW, Williams P, Chong W, Acsadi G, Jani A, Felgner PL. Direct gene transfer into mouse muscle in vivo. Science. 1990;247:1465-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2667] [Cited by in RCA: 2758] [Article Influence: 78.8] [Reference Citation Analysis (0)] |
| 71. | Kulkarni JA, Witzigmann D, Thomson SB, Chen S, Leavitt BR, Cullis PR, van der Meel R. The current landscape of nucleic acid therapeutics. Nat Nanotechnol. 2021;16:630-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 795] [Article Influence: 198.8] [Reference Citation Analysis (0)] |
| 72. | Hopkins AL, Groom CR. The druggable genome. Nat Rev Drug Discov. 2002;1:727-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2416] [Cited by in RCA: 2325] [Article Influence: 101.1] [Reference Citation Analysis (0)] |
| 73. | Bulaklak K, Gersbach CA. The once and future gene therapy. Nat Commun. 2020;11:5820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 194] [Article Influence: 38.8] [Reference Citation Analysis (0)] |
| 74. | Ochoa S, Milam VT. Modified Nucleic Acids: Expanding the Capabilities of Functional Oligonucleotides. Molecules. 2020;25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 75. | Sago CD, Kalathoor S, Fitzgerald JP, Lando GN, Djeddar N, Bryksin AV, Dahlman JE. Barcoding chemical modifications into nucleic acids improves drug stability in vivo. J Mater Chem B. 2018;6:7197-7203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 76. | Chen C, Yang Z, Tang X. Chemical modifications of nucleic acid drugs and their delivery systems for gene-based therapy. Med Res Rev. 2018;38:829-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 119] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 77. | Safari J, Zarnegar Z. Advanced drug delivery systems: Nanotechnology of health design A review. J Saudi Chem Soc. 2014;18:85-99. [RCA] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 180] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 78. | Yamada Y. Nucleic Acid Drugs-Current Status, Issues, and Expectations for Exosomes. Cancers (Basel). 2021;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 79. | Dias N, Stein CA. Antisense oligonucleotides: basic concepts and mechanisms. Mol Cancer Ther. 2002;1:347-355. [PubMed] |
| 80. | ClinicalTrials.gov. Clinical Trial of BP1001 in Combination With With Venetoclax Plus Decitabine in AML. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT02781883. |
| 81. | ClinicalTrials.gov. BP1001-A in Patients With Advanced or Recurrent Solid Tumors. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT04196257. |
| 82. | ClinicalTrials.gov. A Clinical Trial of BP1002 in Patients With Advanced Lymphoid Malignancies. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT04072458. |
| 84. | Kim DH, Rossi JJ. Strategies for silencing human disease using RNA interference. Nat Rev Genet. 2007;8:173-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 788] [Cited by in RCA: 783] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 85. | Buscail L, Bournet B, Cordelier P. Role of oncogenic KRAS in the diagnosis, prognosis and treatment of pancreatic cancer. Nat Rev Gastroenterol Hepatol. 2020;17:153-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 468] [Article Influence: 93.6] [Reference Citation Analysis (0)] |
| 86. | Zorde Khvalevsky E, Gabai R, Rachmut IH, Horwitz E, Brunschwig Z, Orbach A, Shemi A, Golan T, Domb AJ, Yavin E, Giladi H, Rivkin L, Simerzin A, Eliakim R, Khalaileh A, Hubert A, Lahav M, Kopelman Y, Goldin E, Dancour A, Hants Y, Arbel-Alon S, Abramovitch R, Galun E. Mutant KRAS is a druggable target for pancreatic cancer. Proc Natl Acad Sci U S A. 2013;110:20723-20728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 227] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 87. | ClinicalTrials.gov. Phase I - Escalating Dose Study of siG12D LODER (Local Drug EluteR) in Patients With Locally Advanced Adenocarcinoma of the Pancreas, and a Single Dose Study of siG12D LODER (Local Drug EluteR) in Patients With Non-operable Adenocarcinoma of the Pancreas. |
| 88. | ClinicalTrials.gov. A Phase 2 Study of siG12D LODER in Combination With Chemotherapy in Patients With Locally Advanced Pancreatic Cancer (PROTACT). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT01676259. |
| 89. | ClinicalTrials.gov. Study With Atu027 in Patients With Advanced Solid Cancer. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT00938574. |
| 90. | ClinicalTrials.gov. Multi-center, Open Label, Extension Study of ALN-VSP02 in Cancer Patients Who Have Responded to ALN-VSP02 Treatment. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT02166255. |
| 91. | ClinicalTrials.gov. Dose Escalation Trial to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Intravenous ALN-VSP02 In Patients With Advanced Solid Tumors With Liver Involvement. [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT00882180. |
| 92. | El Dika I, Lim HY, Yong WP, Lin CC, Yoon JH, Modiano M, Freilich B, Choi HJ, Chao TY, Kelley RK, Brown J, Knox J, Ryoo BY, Yau T, Abou-Alfa GK. An Open‐Label, Multicenter, Phase I, Dose Escalation Study with Phase II Expansion Cohort to Determine the Safety, Pharmacokinetics, and Preliminary Antitumor Activity of Intravenous TKM‐080301 in Subjects with Advanced Hepatocellular Carcinoma. Oncologist. 2019;24:747. |
| 93. | Kwok A, Raulf N, Habib N. Developing small activating RNA as a therapeutic: current challenges and promises. Ther Deliv. 2019;10:151-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 94. | ClinicalTrials.gov. Safety, Tolerability and Anti-tumour Activity Study of MTL-CEBPA Plus Sorafenib (OUTREACH2). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT04710641. |
| 95. | ClinicalTrials.gov. A Study of MTL-CEBPA in Combination With a PD-1 Inhibitor in Patients With Advanced Solid Tumours (TIMEPOINT). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT04105335. |
| 96. | Peng Y, Croce CM. The role of MicroRNAs in human cancer. Signal Transduct Target Ther. 2016;1:15004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1765] [Cited by in RCA: 1702] [Article Influence: 189.1] [Reference Citation Analysis (0)] |
| 97. | O'Neill CP, Dwyer RM. Nanoparticle-Based Delivery of Tumor Suppressor microRNA for Cancer Therapy. Cells. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 98. | Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines - a new era in vaccinology. Nat Rev Drug Discov. 2018;17:261-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1620] [Cited by in RCA: 2806] [Article Influence: 400.9] [Reference Citation Analysis (0)] |
| 99. | Miao L, Zhang Y, Huang L. mRNA vaccine for cancer immunotherapy. Mol Cancer. 2021;20:41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 692] [Cited by in RCA: 579] [Article Influence: 144.8] [Reference Citation Analysis (0)] |
| 100. | ClinicalTrials.gov. HPV Anti-CD40 RNA Vaccine (HARE-40). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT03418480. |
| 101. | ClinicalTrials.gov. Evaluation of the Safety and Tolerability of i.v. Administration of a Cancer Vaccine in Patients With Advanced Melanoma (Lipo-MERIT). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT02410733. |
| 102. | ClinicalTrials.gov. Ovarian Cancer Treatment With a Liposome Formulated mRNA Vaccine in Combination With (Neo-)Adjuvant Chemotherapy (OLIVIA). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT04163094. |
| 103. | ClinicalTrials.gov. A Study of mRNA-5671/V941 as Monotherapy and in Combination With Pembrolizumab (V941-001). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT03948763. |
| 104. | Blass E, Ott PA. Advances in the development of personalized neoantigen-based therapeutic cancer vaccines. Nat Rev Clin Oncol. 2021;18:215-229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 566] [Cited by in RCA: 612] [Article Influence: 153.0] [Reference Citation Analysis (1)] |
| 105. | ClinicalTrials.gov. An Efficacy Study of Adjuvant Treatment With the Personalized Cancer Vaccine mRNA-4157 and Pembrolizumab in Participants With High-Risk Melanoma (KEYNOTE-942). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT03897881. |
| 106. | ClinicalTrials.gov. RNA-Immunotherapy of IVAC_W_bre1_uID and IVAC_M_uID (TNBC-MERIT). [cited 20 April 2022]. Available from: https://clinicaltrials.gov/ct2/show/NCT02316457. |
| 107. | Baumeister SH, Freeman GJ, Dranoff G, Sharpe AH. Coinhibitory Pathways in Immunotherapy for Cancer. Annu Rev Immunol. 2016;34:539-573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 108. | Liu Z, Ravindranathan R, Kalinski P, Guo ZS, Bartlett DL. Rational combination of oncolytic vaccinia virus and PD-L1 blockade works synergistically to enhance therapeutic efficacy. Nat Commun. 2017;8:14754. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 276] [Article Influence: 34.5] [Reference Citation Analysis (1)] |
| 109. | Haanen JBAG. Converting Cold into Hot Tumors by Combining Immunotherapies. Cell. 2017;170:1055-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 198] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 110. | June CH, O'Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science. 2018;359:1361-1365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1289] [Cited by in RCA: 2066] [Article Influence: 295.1] [Reference Citation Analysis (0)] |
| 111. | Rosenberg SA, Restifo NP. Adoptive cell transfer as personalized immunotherapy for human cancer. Science. 2015;348:62-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1877] [Cited by in RCA: 1847] [Article Influence: 184.7] [Reference Citation Analysis (0)] |
| 112. | Ott PA, Hu Z, Keskin DB, Shukla SA, Sun J, Bozym DJ, Zhang W, Luoma A, Giobbie-Hurder A, Peter L, Chen C, Olive O, Carter TA, Li S, Lieb DJ, Eisenhaure T, Gjini E, Stevens J, Lane WJ, Javeri I, Nellaiappan K, Salazar AM, Daley H, Seaman M, Buchbinder EI, Yoon CH, Harden M, Lennon N, Gabriel S, Rodig SJ, Barouch DH, Aster JC, Getz G, Wucherpfennig K, Neuberg D, Ritz J, Lander ES, Fritsch EF, Hacohen N, Wu CJ. An immunogenic personal neoantigen vaccine for patients with melanoma. Nature. 2017;547:217-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2010] [Cited by in RCA: 2054] [Article Influence: 256.8] [Reference Citation Analysis (0)] |
| 113. | Zhuang J, Holay M, Park JH, Fang RH, Zhang J, Zhang L. Nanoparticle Delivery of Immunostimulatory Agents for Cancer Immunotherapy. Theranostics. 2019;9:7826-7848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 114. | Perciani CT, Liu LY, Wood L, MacParland SA. Enhancing Immunity with Nanomedicine: Employing Nanoparticles to Harness the Immune System. ACS Nano. 2021;15:7-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 115. | Youn YS, Bae YH. Perspectives on the past, present, and future of cancer nanomedicine. Adv Drug Deliv Rev. 2018;130:3-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 196] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 116. | Habault J, Poyet JL. Recent Advances in Cell Penetrating Peptide-Based Anticancer Therapies. Molecules. 2019;24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 219] [Article Influence: 36.5] [Reference Citation Analysis (0)] |
| 117. | Liu X, Wu F, Ji Y, Yin L. Recent Advances in Anti-cancer Protein/Peptide Delivery. Bioconjug Chem. 2019;30:305-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 115] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 118. | Klimpel A, Lützenburg T, Neundorf I. Recent advances of anti-cancer therapies including the use of cell-penetrating peptides. Curr Opin Pharmacol. 2019;47:8-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 119. | Anzalone AV, Koblan LW, Liu DR. Genome editing with CRISPR-Cas nucleases, base editors, transposases and prime editors. Nat Biotechnol. 2020;38:824-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 643] [Cited by in RCA: 1419] [Article Influence: 283.8] [Reference Citation Analysis (0)] |
| 120. | Wei T, Cheng Q, Min YL, Olson EN, Siegwart DJ. Systemic nanoparticle delivery of CRISPR-Cas9 ribonucleoproteins for effective tissue specific genome editing. Nat Commun. 2020;11:3232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 160] [Cited by in RCA: 401] [Article Influence: 80.2] [Reference Citation Analysis (0)] |
| 121. | Rosenblum D, Gutkin A, Kedmi R, Ramishetti S, Veiga N, Jacobi AM, Schubert MS, Friedmann-Morvinski D, Cohen ZR, Behlke MA, Lieberman J, Peer D. CRISPR-Cas9 genome editing using targeted lipid nanoparticles for cancer therapy. Sci Adv. 2020;6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 169] [Cited by in RCA: 339] [Article Influence: 67.8] [Reference Citation Analysis (0)] |
| 122. | Zhang L, Wang P, Feng Q, Wang N, Chen Z, Huang Y, Zheng W, Jiang X. Lipid nanoparticle-mediated efficient delivery of CRISPR/Cas9 for tumor therapy. NPG Asia Mater. 2017;9:e441. [DOI] [Full Text] |
| 123. | Sun W, Ji W, Hall JM, Hu Q, Wang C, Beisel CL, Gu Z. Self-assembled DNA nanoclews for the efficient delivery of CRISPR-Cas9 for genome editing. Angew Chem Int Ed Engl. 2015;54:12029-12033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 490] [Cited by in RCA: 473] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 124. | Naeem M, Hoque MZ, Ovais M, Basheer C, Ahmad I. Stimulus-Responsive Smart Nanoparticles-Based CRISPR-Cas Delivery for Therapeutic Genome Editing. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 125. | Liu Q, Zhao K, Wang C, Zhang Z, Zheng C, Zhao Y, Zheng Y, Liu C, An Y, Shi L, Kang C, Liu Y. Multistage Delivery Nanoparticle Facilitates Efficient CRISPR/dCas9 Activation and Tumor Growth Suppression In Vivo. Adv Sci (Weinh). 2019;6:1801423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 127] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 126. | Tang Q, Liu J, Jiang Y, Zhang M, Mao L, Wang M. Cell-Selective Messenger RNA Delivery and CRISPR/Cas9 Genome Editing by Modulating the Interface of Phenylboronic Acid-Derived Lipid Nanoparticles and Cellular Surface Sialic Acid. ACS Appl Mater Interfaces. 2019;11:46585-46590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 127. | Lyu Y, He S, Li J, Jiang Y, Sun H, Miao Y, Pu K. A Photolabile Semiconducting Polymer Nanotransducer for Near-Infrared Regulation of CRISPR/Cas9 Gene Editing. Angew Chem Int Ed Engl. 2019;58:18197-18201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 128. | Hua S, de Matos MBC, Metselaar JM, Storm G. Current Trends and Challenges in the Clinical Translation of Nanoparticulate Nanomedicines: Pathways for Translational Development and Commercialization. Front Pharmacol. 2018;9:790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 408] [Cited by in RCA: 569] [Article Influence: 81.3] [Reference Citation Analysis (0)] |
| 129. | Sun D, Zhou S, Gao W. What Went Wrong with Anticancer Nanomedicine Design and How to Make It Right. ACS Nano. 2020;14:12281-12290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 171] [Article Influence: 34.2] [Reference Citation Analysis (0)] |
| 130. | Brown MR, Hondow N, Brydson R, Rees P, Brown AP, Summers HD. Statistical prediction of nanoparticle delivery: from culture media to cell. Nanotechnology. 2015;26:155101. [PubMed] [DOI] [Full Text] |
| 131. | Rosenblum D, Peer D. Omics-based nanomedicine: the future of personalized oncology. Cancer Lett. 2014;352:126-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 56] [Article Influence: 5.1] [Reference Citation Analysis (0)] |