Published online Jun 24, 2021. doi: 10.5306/wjco.v12.i6.437
Peer-review started: January 19, 2021
First decision: April 6, 2021
Revised: April 7, 2021
Accepted: June 2, 2021
Article in press: June 2, 2021
Published online: June 24, 2021
Processing time: 153 Days and 4 Hours
Patients with bronchogenic carcinoma comprise a high-risk group for coronavirus disease 2019 (COVID-19), pneumonia and related complications. Symptoms of COVID-19 related pulmonary syndrome may be similar to deteriorating symp
Core Tip: Unprecedented times of a pandemic pose a major challenge in maintaining adequate balance between the risk of contracting deadly coronavirus disease 2019 (COVID-19) against the dire consequences of delaying treatment for a life-threatening malignancy. Cancer survivors and patients represent a vulnerable population for COVID-19 related pulmonary syndrome, which can further complicate respiratory and cardiovascular comorbidities they possess. Risk stratification of bronchogenic carcinoma patients and priority imaging may be applied for optimal use of resources during these uncertain times of COVID-19. Utilizing artificial intelligence and deep learning modules based on pattern recognition of image findings during this pandemic has made a lasting impact across future realms.
- Citation: Reddy R. Imaging diagnosis of bronchogenic carcinoma (the forgotten disease) during times of COVID-19 pandemic: Current and future perspectives. World J Clin Oncol 2021; 12(6): 437-457
- URL: https://www.wjgnet.com/2218-4333/full/v12/i6/437.htm
- DOI: https://dx.doi.org/10.5306/wjco.v12.i6.437
In late December 2019, a lower respiratory tract infection was reported in a cluster of patients from Wuhan City, Hubei Province, China. A novel strain of coronavirus was isolated from the bronchoalveolar lavage of infected patients[1]. On January 9, 2020, the World Health Organization named the febrile illness related pulmonary syndrome as coronavirus disease 2019 (COVID-19)[2]. At the time of writing this article on January 19, 2021, the number of confirmed cases stand at 99737233 with 2137990 reported deaths, according to an online virus tracker created by The Lancet and hosted by Johns Hopkins University[3]. Sudden outbreaks of COVID-19 with critically ill patients have put enormous strain on the limited healthcare resources of many countries and caused increased mortality from the pandemic. As of January 2021, the number of confirmed COVID-19 cases globally is over 95 million, vastly affecting every territory other than a few isolated South Atlantic and Pacific Island states and Antarctica[3].
COVID-19 causes mild infection in the majority (80%) of patients showing gradual recovery in about 2 wk[4]. However, COVID-19 has a predilection for middle aged and elderly populations with highest case fatality (8%-15%) seen in the elderly, aged above 80 years. R0 (basic reproduction number) that is each infected individual — on average — transmitting new cases of novel coronavirus was estimated to be 2.2 based on a study that examined the first 425 infected cases in Wuhan[5]. The study also calculated the average incubation period in the group to be 5.2 d[5].
Six subtypes of coronavirus have infected humans in the past, of which four subtypes have caused mild respiratory symptoms, similar to rhinovirus. The other 2 subtypes which are MERS (Middle East respiratory syndrome)-coronavirus and SARS (severe acute respiratory syndrome)-coronavirus have caused severe forms of infection[6]. Relevant epidemiological history and contact history of within 14 d before disease onset have been of tremendous help in contact tracing and cluster tracing of COVID-19 patients[7]. Based on recent epidemiological studies, the incubation period of COVID-19 was calculated to be 1-14 d[8]. Only a minority of COVID-19 patients develop critical illness leading to cardiopulmonary compromise and eventual mortality. The majority of patients have a good prognosis and recovery. COVID-19 related mortality is commonly noted in elderly population with underlying comorbidities.
COVID-19 is a febrile illness associated with dry cough, anosmia and ageusia. Severe cases present with symptoms such as dyspnoea, tachypnoea and severe hypoxemia which can progress to acute respiratory distress syndrome (ARDS) and cardiopulmonary failure. Release of interleukin (IL)-1, IL-6 and tumour necrosis factor-α which are proinflammatory cytokines leads to immune-related pneumonitis of COVID-19 which can progress to profound hypotension, septic shock and acute renal failure[9]. Complications in severe COVID-19 include cardiac failure, metabolic acidosis, rhabdomyolysis, coagulopathy, deep vein thrombosis and pulmonary embolism[10].
Analysis of epidemiological characteristics of 72314 COVID-19 patients in mainland China at “The Chinese Center for Disease Control and Prevention” revealed 107 (0.5%) patients to be affected by cancer[11] and similarly, findings from a study published by Chinese researchers on 1590 COVID-19 patients described 18 (1%) patients with a history of cancer, of which five were diagnosed with bronchogenic carcinoma (5/18, 28%)[12]. Their study also demonstrated a higher risk of accelerated events and deteriorating symptoms more rapidly amongst cancer patients (39%) compared to non-cancerous patients (8%)[12]. Findings from a multicentred study done by Dai et al[13] on confirmed COVID-19 patients which included 105 cancer patients and 536 age-matched non-cancerous patients, revealed that patients with cancer had higher risks in all severe outcomes. Haematological cancer, bronchogenic carcinoma or metastatic cancer (stage IV) had the highest frequency of severe outcomes. Based on the findings of above mentioned two cohorts, it would be fair to infer that patients with carcinoma are a susceptible population for contracting COVID-19 related pulmonary syndrome with a higher risk of related complications and a higher mortality rate. When patients with any form of cancer develop COVID-19 super-infection, the findings are of higher grade/severity with rapid deterioration to end stage events. Further complications include the inability of healthcare stakeholders to adequately streamline ample resources for cancer patients and shortage of services for providing timely antineoplastic treatment which results in poor prognosis.
Unprecedented times of a pandemic pose a major challenge in maintaining adequate balance between the risk of contracting deadly COVID-19 against the dire conse
Transmitted by droplets, novel coronavirus gains entry via angiotensin-converting enzyme 2 receptors located on the epithelial lining of nose, mouth, throat and multiplies within type II epithelial cells within the lung parenchyma[15]. COVID-19 related pulmonary syndrome ranges from mild upper airway symptoms progressing to life-threatening ARDS and related cardiopulmonary failure. Initial COVID-19 pneumonia appearances on histopathology are characterized by alveolar oedema, with patchy and mild inflammatory reaction. Advanced COVID-19 pneumonia can have appearances similar to ARDS characterized by alveolar congestion, oedema, haemo
Symptoms of COVID-19 related pulmonary syndrome may be similar to deteriorating symptoms encountered during bronchogenic carcinoma progression. Similarities between clinical and imaging findings can pose a major challenge to clinicians in distinguishing COVID-19 super-infection from evolving bronchogenic carcinoma. The above-mentioned entities require very different therapeutic approaches. Immune checkpoint inhibitor therapy which is a more widely used treatment for bronchogenic carcinoma can also induce pneumonitis reported in 2% of its patients and may display similar imaging appearances of COVID-19 related pulmonary syndrome[16]. Interstitial pattern of pneumonitis reported in 4% of patients with epidermal growth factor receptor-mutant bronchogenic carcinoma on treatment with tyrosine kinase inhibitor Osimertinib may have similar radiological patterns of COVID-19.
Although consolidation and ground-glass opacification associated with COVID-19 related pulmonary syndrome tends to be more peripheral in location, the radiological changes can mimic treatment induced chemotherapy or radiotherapy and immunotherapy related pneumonitis in bronchogenic carcinoma patients. However, the latter tends to be more confluent and perihilar in location[17]. In situations where clinicians encounter overlap in imaging findings, evaluation of such patients by a multidisciplinary team consisting of oncologists and radiologists should be undertaken with due consideration given for reverse-transcriptase polymerase chain reaction (RT-PCR) testing. Computed tomography (CT) pulmonary angiography represents a valuable imaging investigation for diagnosing pulmonary thromboembolism and for planning further management strategies in patients with COVID-19 and lung cancer related complications[18]. Although the current guidelines do not recommend imaging as the first line investigation for screening of COVID-19 related pneumonia, most countries have adopted diagnostic imaging as an adjunct tool for follow-up of disease progre
Patients with bronchogenic carcinoma have a higher susceptibility to develop severe forms of COVID-19 disease and related complications. Furthermore, patients with associated comorbidities including moderate to severe asthma, chronic obstructive pulmonary disease, cardiovascular disease, diabetes mellitus, chronic kidney disease on dialysis, chronic liver disease, immunosuppression conditions and severe obesity are more at risk of developing severe form of COVID-19 disease. Consequently, patients with bronchogenic carcinoma comprise a high-risk group for COVID-19 pneumonia and related complications. Smoking and immunosuppression induced by steroids are cumulative risk factors for COVID-19 in patients with bronchogenic carcinoma.
A study done at Zhongnan Hospital of Wuhan University, China on 1524 cancer patients admitted to the Department of Radiation and Medical Oncology showed that 0.79% of cancer patients had COVID-19 as against 0.37% of the general population of Wuhan[19]. Findings from a retrospective analysis published in China revealed that patients with cancer, especially advanced bronchogenic carcinoma (28%), are at greater risk of severe disease as compared to non-cancerous patients and the same is confirmed by multiple studies done in this aspect[20].
Smoking increases the risk of complications by up to 1.4-fold for deaths, and by 2.4-fold for mechanical ventilation requirement in patients with COVID-19. At a recent meeting of the “American Association for Cancer Research” annual congress on “COVID-19 and Cancer”, data from a global registry on patients with thoracic cancers infected with COVID-19 revealed a 34.6% mortality rate amongst patients with thoracic cancer[21]. In addition, the most common complications reported were pneumonitis (79.6%), ARDS (26.8%), multiple organ dysfunction (7.6%), and sepsis (5.1%)[21]. The proceedings from the session also attributed the cause of death among patients with bronchogenic carcinoma to COVID-19 and not to carcinoma per se, putting them at greater risk of developing complications, irrespective of the stage of cancer.
Increased inflammation termed as hyperinflammation is common with aging and in cancer patients referred to as “inflammaging”. Poor prognosis in cancer patients and worse outcomes in elderly with severe COVID-19 disease are attributed to this hyperinflammatory disorder characterized by vast immune cell mediated activation of cytokines. Immune checkpoint inhibitors can also cause hyperinflammatory response, leading to enhanced production of cytokines from macrophages and T- lymphocytes.
Immune dysregulation in patients with bronchogenic carcinoma may cause hyperinflammation with increased risk of severe COVID-19 disease. Based on a study by Peng et al[22], which showed an over-representation of cancer patients and bronchogenic carcinoma in particular within their COVID-19 cohort, the authors have proposed three strategies for cancer patients to circumvent the COVID-19 crisis which include; intensifying surveillance of cancer patients infected with COVID-19, increased use of personal protection equipment by cancer patients, and postponing elective surgery for stable cancer patients from endemic areas of COVID-19. Additionally, patients with bronchogenic carcinoma are prone to emergent complications which might require admission to intensive care units or ventilator support which makes them highly vulnerable for contracting COVID-19. Smoking has also been implicated as an independent risk factor for progression to severe COVID-19 disease.
Findings from a study done by Zhou et al[19] at Zhongnan Hospital of Wuhan University, China on 1524 cancer patients admitted to the Department of Radiation and Medical Oncology, revealed that patients with bronchogenic carcinoma harboured a higher risk of COVID-19 compared to the general population regardless of the patient being on anticancer treatment. Also, patients with non-small cell lung cancer (NSCLC) and above the age of sixty have a relatively higher risk of contracting COVID-19[23]. Recommendations were issued by collaboration of the Lung Cancer Study Group, the Chinese Respiratory Oncology Collaboration, Chinese Medical Association and Chinese Thoracic Society regarding management of NSCLC patients during the COVID-19 pandemic. These include intentional postponing of antitumor treatment based on the patient performance status; usage of personal protection equipment for patients with advanced NSCLC; treating advanced NSCLC patients as outpatients whenever possible; promptly identifying symptoms of COVID-19 in patients with NSCLC being masked by antitumor treatment; testing patients for COVID-19 before administering antitumor treatment and tailoring treatment strategies based on subtype of NSCLC and efficacy of treatment[24].
RT-PCR is the gold standard investigation for the diagnosis of COVID-19 although, there can be a delay in results of RT-PCR[25]. Here, the role of imaging investigations has been deciphered for rapid screening of patients with suspected COVID-19. Non-contrast CT scan is particularly recommended as intravenous contrast may mask subtle ground-glass opacification as a typical finding in COVID-19. Additional findings include basal consolidations and reverse-halo sign. However, a typical CT finding such as pleural effusions, pulmonary nodules, mediastinal lymphadenopathy, tree-in-bud opacities and cavitation may be encountered in a small minority of patients and should raise concern for atypical pneumonia caused by Mycoplasma or Chlamydia. Halo sign and pneumothorax should suggest an alternative diagnosis rather than COVID-19[26]. As the imaging findings of COVID-19 are not very specific and may show considerable overlap with other viral/atypical pneumonia, the American College of Radiology (ACR) does not recommend routine use of chest radiography or CT for the screening of COVID-19 in asymptomatic individuals or patients with mild symptoms[27]. However, CT is still recommended for making a diagnosis in symptomatic individuals with a high suspicion of COVID-19 but with a negative RT-PCR result and is being utilized for improving the false negative rate of RT-PCR.
Imaging allows further risk stratification of patients when an initial negative result of RT-PCR requires a risk assessment for high clinical suspicion of COVID-19. The British Society of Thoracic Imaging has provided a template for uniform reporting of chest radiography findings based on classification of patients into four groups which include Non-COVID, COVID normal, COVID indeterminate and COVID cla
There is no role of 18F-fluorodeoxyglucose (18F-FDG) positron emission tomo
The sensitivity of chest radiographs is relatively less ranging between 30%-70% and typical findings of COVID-19 include multifocal translobar consolidation[30]. Pleural effusions are a relatively rare feature. At initial presentation, total positive rate of RT-PCR from nasopharyngeal swabs has been reported to be 59% due to current limitations in kit performance[31]. The European Radiologists Association recommen
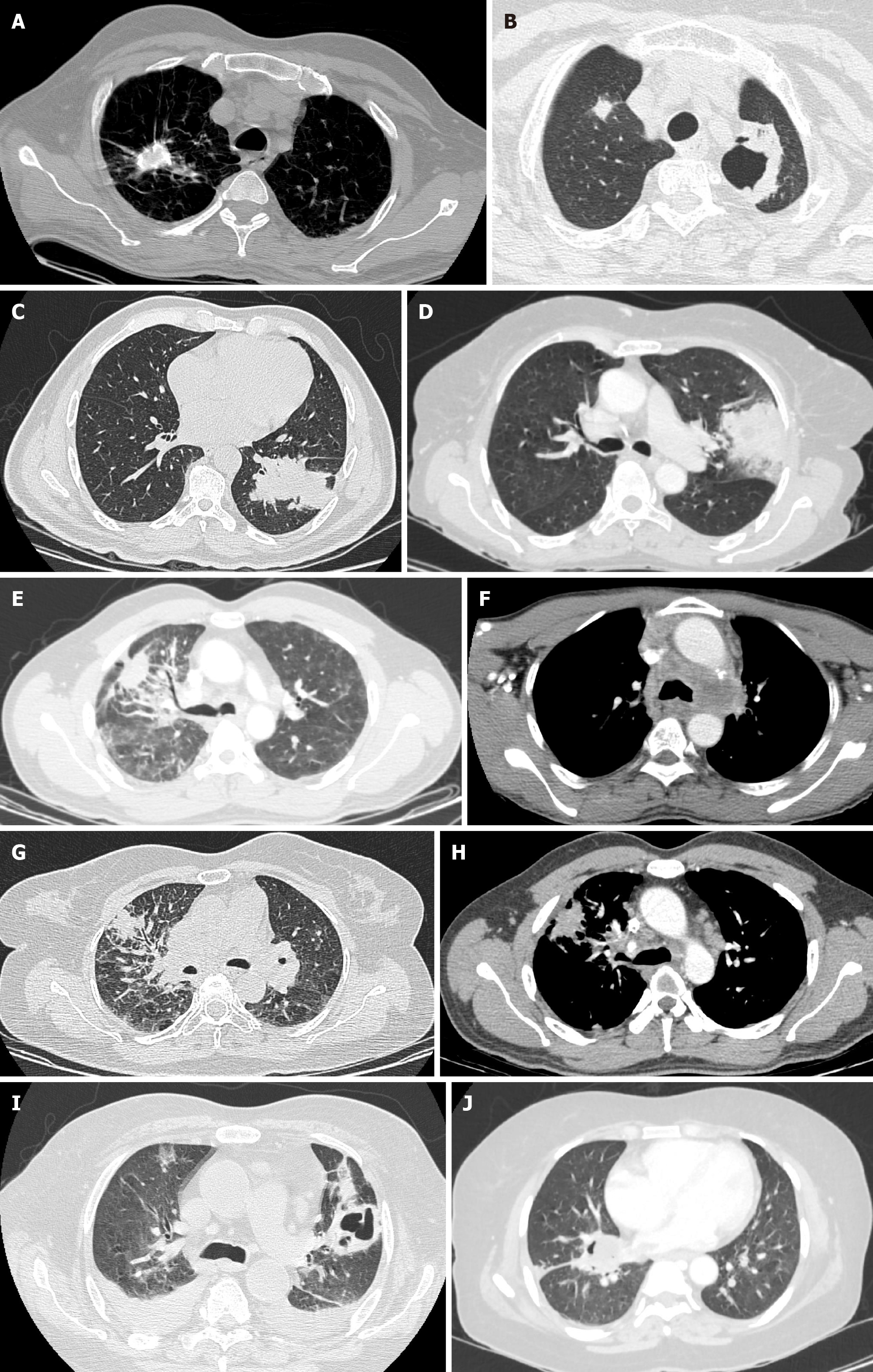
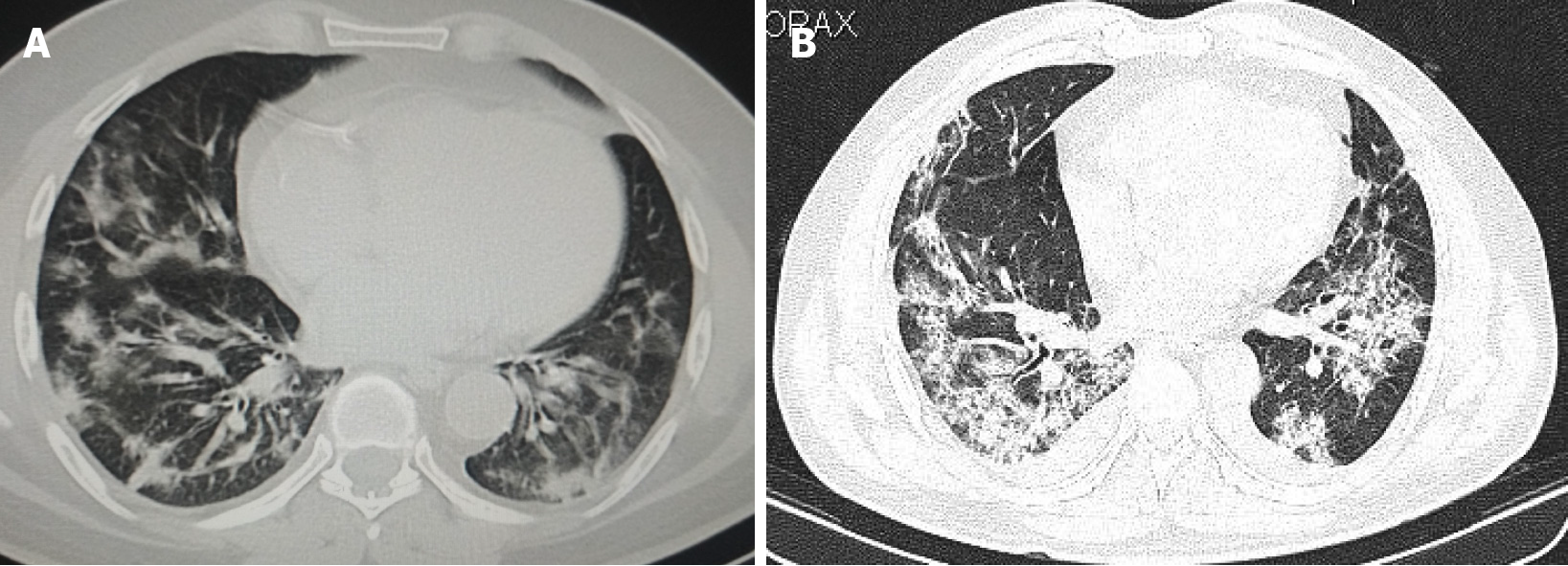
Role of chest CT in the initial diagnosis of COVID-19 related pulmonary syndrome is commendable. Typical imaging findings of COVID-19 pneumonia on chest CT include patchy ground glass opacification in basal and peripheral subpleural distribution with translobar consolidations. Crazy paving pattern is a common finding, whereas reverse-halo sign is rare, but when present is highly specific[33]. CT has a determinant role, not only for early diagnosis of COVID-19, but also for monitoring disease progression and clinical course. Early phase of COVID-19 pneumonia is characterized by ground-glass opacification in peripheral and basal distribution, crazy paving pattern, and traction bronchiectasis. Advanced phase of COVID-19 pneumonia is characterized by progressive transition to consolidation, irregular interlobular septal thickening with extensive lung nodules, and ARDS. Progressive stage of bronchogenic carcinoma or concomitant pneumonia in bronchogenic carcinoma patients may have similar overlapping CT findings of COVID-19 related pulmonary syndrome[34].
An expert consensus statement on ‘Reporting Chest Computed Tomography Findings Related to COVID-19’ has been published by the Radiological Society of North America which categorizes COVID-19 pneumonia based on CT findings into negative for pneumonia, atypical appearances, indeterminate, and typical appearances[35]. CT findings specific for COVID-19 pneumonia include bilateral ground-glass opacification with interlobular septal thickening (crazy paving appearance) predominantly in basal and peripheral distribution with reverse halo sign. CT findings of COVID-19 based on the duration of symptoms include—early stage constituting 0-4 d after onset of respiratory symptoms. Findings during this stage include normal CT scans in 50% of patients with the rest demonstrating focal subpleural ground glass opacification, predominantly in lower lobes. However, typical CT findings are rarely observed during this phase. Progressive stage of COVID-19 constitutes 5-8 d after symptom onset and is characterized by multifocal and bilateral ground glass opaci
General recommendations include minimize aerosol generation and limiting COVID-19 transmission by giving preference to image-guided transthoracic biopsies for diagnosing bronchogenic carcinoma over transbronchial approaches[37]. Giving preference to non-invasive mediastinal staging via imaging on PET/CT and CT and if not feasible preferring mediastinoscopy over bronchoscopy. Endobronchial ultrasound (EBUS) as an investigative tool for nodal staging of stage III disease in absence of radiographically apparent mediastinum may not be required, and for stage III disease with radiographically apparent nodal disease of the mediastinum, no further confirmation with EBUS is required[38].
Cancer survivors and patients represent a vulnerable population for COVID-19 syndrome which can further complicate respiratory and cardiovascular comorbidities they possess. Prevalence of COVID-19 in cancer patients is approx. 1%-6% as compared to their non-cancerous counterparts[39]. And, patients with bronchogenic carcinoma have the highest risk of unfavourable outcomes and complications of COVID-19. Though reasons for this selective affinity of COVID-19 patients is not fully elucidated, lower immune response in cancer patients secondary to treatment regimens may be the cause. Other causes include advanced age of patients and smoking as a risk factor which is prevalent in patients with bronchogenic carcinoma. Additionally, pre-existing cardiovascular and pulmonary comorbidities in patients with bronchogenic carcinoma make them highly vulnerable to COVID-19 and can further complicate the course of the disease. The cumulative risk factors identified in patients with bronchogenic carcinoma complicate the pathway of COVID-19 related pulmonary syndrome in them resulting in an unfavourable outcome.
Streamlining resources by healthcare stakeholders for screening of bronchogenic carcinoma and evaluation of patients with incidentally detected lung nodules are related scenarios which are currently in focus during these uncertain times of a pandemic. National Comprehensive Cancer Network, the United States Preventive Services Taskforce, and CHEST (the American College of Chest Physicians) have issued guidelines for screening of patients to rule out bronchogenic carcinoma and have recommended annual low-dose chest CT to be undertaken in high-risk indivi
Attempts at screening for bronchogenic carcinoma in patients should be delayed until the curve of the pandemic flattens and risks involved with transmission of COVID-19 becomes near minimal. The reasons for the same include increased risk of exposure of patients to COVID-19. Incidental lung findings in asymptomatic individuals exposed to COVID-19 increases the false positive rate and may expose these patients to unnecessary procedures thus increasing the risk further. Thus, by delaying screening for cancer in the current pandemic, resources can be streamlined for managing patients with moderate to severe COVID-19 disease.
The COVID-19 pandemic has strained the healthcare system and drained the resources worldwide due to high transmission rate causing significant mortality and morbidity. Super-infection with COVID-19 in patients with bronchogenic carcinoma increases the risk of complications with further delay in treatment of bronchogenic carcinoma resulting in poor outcomes and mortality. The impact of COVID-19 on patients with bronchogenic carcinoma is multi-faceted and management should be focused in improving negative outcomes. The implications of COVID-19 pandemic in broncho
The current pandemic has forced all healthcare stakeholders to prioritize per value resources and reorganize therapeutic strategies for timely management of patients with COVID-19 related pulmonary syndrome. Since the beginning of the COVID-19 pandemic, the oncology community has been under tremendous strain in managing patients with bronchogenic carcinoma that requires a balance in treating patients, without losing them to COVID-19. Organizations worldwide such as the European Society of Medical Oncology (ESMO), have worked to implement preventative measures and spreading knowledge about the additional risks that cancer patients are prone to during the COVID-19 pandemic[42]. Oncologists have faced the constant dilemma of whether to offer or postpone treatment to patients during this COVID-19 pandemic. Consideration of bronchogenic carcinoma treatment during COVID-19 times requires careful individual assessment as chemotherapy administered may cause transient immune suppression. In addition to this, immunotherapy with immune checkpoint inhibitor therapy may trigger chronic inflammatory changes within the lung parenchyma causing pneumonitis like picture, with resultant poor outcome. However, oncologists currently should carefully weigh the benefits of treating patients with bronchogenic carcinoma vs the risk of getting infected with COVID-19, as delay in treatment may also have a negative effect on the prognosis of bronchogenic carcinoma causing its progression.
Pulmonologists and pulmonary oncologists comprise front-line specialists dealing with the diagnosis and management of patients with bronchogenic carcinoma and COVID-19 related syndrome. It is very crucial for delineating the differential diagnosis of bronchogenic carcinoma and COVID-19, as there is an overlap in symptomatology of the two entities. The delineation and differentiation are based on rapid progression of symptoms, positivity of RT-PCR results to COVID-19, fresh appearance of a patchy infiltrate that is distinct from the baseline pattern of bronchogenic carcinoma initially encountered on imaging.
COVID-19 pulmonary syndrome, bronchogenic carcinoma, chronic obstructive pulmonary disease, pulmonary arterial thromboembolism, lung metastases, radiation pneumonitis, radiation fibrosis and lobar pneumonia are some of the pulmonary conditions for which radiological diagnostic modalities such as chest radiographs, CT and PET/CT form the cornerstone of management and follow up. However, sociocultural approaches and published guidelines for imaging of suspected or confirmed cases of COVID-19 differ for these diagnostic modalities with a temporal and regional variation. Peripheral/subpleural ground-glass opacification with a predilection to bilateral posterior lung segments, multifocal consolidations, crazy paving pattern, vascular dilatation with interlobular septal thickening, traction bronchiectasis and reverse halo sign are some of the typical features of COVID-19 encountered on CT[43]. Whether it is COVID-19 or bronchogenic carcinoma, radio
Choice of chemotherapy, targeted therapy, immunotherapy and supportive care must be tailored based on tumour characteristics and patient comorbidities, considering the risk of COVID-19 super-infection in these patients. Prioritizing the choice of treatment is dependent on active COVID-19 cases in the community and the availability of resources. Prima facie principle in the management of patients with bronchogenic carcinoma is to provide timely enrolment in treatment without unjustified delay for better prognosis. Patients with bronchogenic carcinoma who are asymptomatic or showing features of minimal progression may be monitored at a 4-8 wk interval, and radiotherapy may be offered to patients with local disease progression[44]. However, progressive disease in bronchogenic carcinoma and immune checkpoint inhibitor therapy induced pneumonitis should be differentiated from COVID-19 related pulmo
During these unprecedented times of COVID-19 pandemic, using the least invasive technique holds the key for diagnosing bronchogenic carcinoma in patients. Aerosol-generating procedures such as bronchoscopy should be avoided whenever feasible[46]. A statement issued by the American Association for Bronchology and Interventional Pulmonology regarding the effective use of bronchoscopy in patients with suspected or confirmed cases of COVID-19 also applies to patients with bronchogenic carcinoma[47]. Salient features of the consensus statement include elective bron
Symptoms of patients with bronchogenic carcinoma is due to the tumour itself and may also include cough, dyspnoea and occasional fever. Also, treatment related adverse events caused by immunotherapy, radiotherapy and chemotherapy may also be encountered in these patients. Further, different patterns of bronchogenic carci
Patients with an epidemiological history who fulfil any two of the clinical criteria or patients with no clear epidemiological history but having fulfilled three of the clinical criteria are considered as suspicious patients based on the diagnostic criteria of COVID-19. It is imperative to distinguish patients with clinical characteristics of bronchogenic carcinoma progression from those with COVID-19. Further differential diagnoses to be considered include: (1) Pulmonary infections which may be atypical pneumonia caused by Chlamydia, Mycoplasma, Pneumocystis jirovecii, Legionella or non-COVID-19 viral pneumonia caused by adenovirus, influenza virus, cytomegalovirus and respiratory syncytial virus; (2) Radiation pneumonitis which occurs 1-3 mo after undergoing radiotherapy in patients with bronchogenic carcinoma[49]. Clinical symptoms may include dry cough associated with fever and decreased white blood cell count. Findings on chest CT in radiation pneumonitis are mostly nonspecific and may include ground-glass opacities opacification with patchy infiltrates. Differentiation from COVID-19 related pulmonary syndrome is difficult; (3) Tumour progression in bronchogenic carcinoma may result in lymphangitis carcinomatosa, pneumonic-type lung carcinoma, and obstructive pneumonitis which may present with fever and/or worsening respiratory symptoms; (4) Clinical characteristics of immune checkpoint inhibitor-related pneumonitis include fever, worsening of cough and dyspnoea, ground glass opacification and patchy infiltrates on the chest CT; and (5) Miscellaneous differential diagnoses include cardiac insufficiency and pulmonary thromboembolism.
Imaging plays a crucial role in the diagnosis, management and follow-up of immunotherapy related complications of bronchogenic carcinoma[50]. Centers for Disease Control and Prevention (CDC) has provided recommendations which were endorsed by the ACR, for safeguarding and prioritizing healthcare resources during the COVID-19 pandemic[51]. The recommendations state that imaging for stable patients, inclu
Pulmonary oncologists and radiologists should collaborate for rescheduling and reviewing chest CT examination, based on the symptomology and prognostic value of such investigation during times of COVID-19 pandemic (low priority). A low-dose CT protocol for screening of bronchogenic carcinoma should be temporarily withheld or advertently postponed considering the risk-benefit ratio of such a procedure in the current situation. Follow-up imaging for bronchogenic carcinoma during the course of treatment in the first 6 mo or in case of bronchogenic carcinoma with progression at any point in time should be considered (high priority). Recommendations suggest priority access to diagnostic imaging in patients with clinical picture suspicious for bronchogenic carcinoma (high priority) (Figure 1).
When an individual who satisfies the eligibility criteria is referred for screening of bronchogenic carcinoma, initiation of screening should be delayed during the COVID-19 pandemic based on the consensus statement consistent with CDC guidance[52]. If an individual who satisfies the eligibility criteria is due for a repeat annual chest CT, annual screening examination should be delayed too. Factors that may influence this decision include availability of resources, COVID-19 penetrance in the community, and comorbid conditions.
When an individual who satisfies the eligibility criteria is due now for a surveillance chest CT for an incidentally detected lung nodule, delaying the surveillance CT scan for approximately 3 mo to 6 mo is acceptable based on the consensus statement consistent with CDC guidance[53].
When an individual who satisfies the eligibility criteria is due now for an imaging guided biopsy, delaying the procedure and re-evaluation of the patient with chest CT in approximately 3 mo to 6 mo is acceptable too based on the consensus statement consistent with CDC guidance[54]. However, pre-COVID-19 recommendations suggest further evaluation with PET/CT and/or biopsy for the patient with intermediate and high-risk lung nodules.
When an individual who satisfies the eligibility criteria for surgical treatment of clinical stage I NSCLC, surgery may be deferred based on the consensus statement consistent with CDC guidance after taking into consideration the size, growth rate and 18F-FDG PET avidity of the malignant lesion[55]. Nevertheless, the clinical scenario needs to be discussed at a multidisciplinary tumour board meeting for delaying treatment in an indolent/early-stage bronchogenic carcinoma during times of the COVID-19 pandemic.
Criteria for high priority imaging include: (1) Patients with clinically significant respiratory symptoms with a history of bronchogenic carcinoma; (2) Bronchogenic carcinoma patients with fresh appearance of respiratory symptoms such as dyspnoea, cough and fever; (3) Patients with suspected bronchogenic carcinoma of unknown stage or stage II, III, IV for standard staging work-up; (4) Patients with mass lesion / nodules suspicious for bronchogenic carcinoma stage III/IV for biopsy; and (5) Patients with bronchogenic carcinoma for suspicion of carcinoma progression at any point of time or for evaluation of response during the first 6 mo of treatment.
Criteria for medium priority imaging include: (1) Patients with early bronchogenic carcinoma for determining the stage; (2) Patients with mass lesion / nodules suspi
Criteria for low priority imaging include: (1) Follow-up imaging for patients of bronchogenic carcinoma having underwent radical treatment more than a year ago with high/intermediate risk of relapse; (2) Follow-up imaging for patients of bronchogenic carcinoma having underwent radical treatment with low risk of relapse; (3) Follow-up imaging for solid nodules with benign morphology, nodules with volume of < 50 mm3, partly solid nodule with a non-solid component of size < 8 mm in diameter, or a pleural-based solid nodule of size < 5 mm in diameter; (4) Bronchogenic carcinoma patients with VDT > 600 d; and (5) Low-dose CT screening of patients for bronchogenic carcinoma screening can be deferred until the current COVID-19 pandemic resolves which is unlikely to affect the overall survival rate (Table 1).
| High priority | Medium priority | Low priority |
| Patients with significant respiratory symptoms and/or cancer-related or treatment-related symptoms. In patients with new onset respiratory symptoms such as dyspnoea, cough with or without fever, a CT scan is recommended | Follow-up imaging for high/intermediate risk of relapse in a year after completion of radical treatment | Follow-up imaging for high/intermediate risk of relapse more than 1 yr after completion of radical treatment |
| Standard staging work-up for suspected invasive cancer of unknown stage or stage II/III/IV | Standard staging work-up for early lung cancer (stage I) | Follow-up imaging after radical treatment in low-risk of relapse scenario |
| Biopsies for suspicious nodules or mass for suspected invasive cancer or stage III/IV | Biopsies for suspicious nodules or mass for suspected invasive cancer of unknown stage or stage I/II | |
| Evaluation of active treatment response in the first 6 mo of treatment or for suspicion of tumour progression at any point of time | Evaluation of active treatment response beyond 6 mo of treatment if stable/ controlled situation | |
| Follow-up of nodules of incidental finding with either: (1) Partially solid nodule with a non-solid component of ≥ 8 mm in size; (2) Known VDT 400 d to 600 d; (3) Solid nodule 50 mm3 to 500 mm3; and (4) Pleural-based solid nodule 5 mm to 10 mm in size | Follow-up of nodules of incidental finding with either: (1) Partially solid nodule with a non-solid component of < 8 mm in size; (2) Known VDT > 600 d; (3) Solid nodule < 50 mm3; (4) Pleural-based solid nodule < 5 mm in size; (5) Non-solid nodule < 8 mm in size; and (6) Benign morphology | |
| Pre-planned imaging evaluation per clinical trial protocol | Lung cancer screening can be deferred until the COVID-19 pandemic resolves — it is reasonable for patients in the general population to defer screening low-dose CT, a deferral that is not likely to have an impact on overall survival |
The most significant goal of bronchogenic carcinoma management during these challenging times of the pandemic is to minimize the risk of exposing patients and staff to COVID-19 whilst still managing all life-threatening events related to bronchogenic carcinoma. This priority can be achieved by limiting outpatient visits to the hospital, and extensive testing for COVID-19 in patients undergoing invasive procedure or systemic chemotherapy and immunotherapy.
Ablative radiotherapy and surgical resection are the treatment approaches for patients with stage I/II/resectable stage III NSCLC subtype of bronchogenic carcinoma[56]. However, the principles for surgical resection of bronchogenic carcinoma remain unaffected during this COVID-19 pandemic. Recommendations from CDC and guidelines from professional societies calls for rescheduling of elective surgeries during the current impending pandemic[57]. Recommendations form the American Society of Clinical Oncology state that patients with cancer and treating clinicians should make individual considerations based on risks of delaying surgical resection in these patients[58]. Also, rescheduling of surgical resection for patients with a recent diagnosis of early-stage bronchogenic carcinoma is recommended, as super-infection with COVID-19 may have poor prognostic outcomes.
The European Association of Medical Oncology has recommended considering surgical resection as a priority in the management of early NSCLC and delays in surgery not to exceed 6-8 wk[59]. Recommendations based on the phase of impending COVID-19 pandemic and availability of resources by healthcare institutions, the American College of Surgeons has recently published COVID-19 triage guidelines focusing on surgical care in elective cases[60]. Based on recommendations in semi-urgent settings, surgical intervention is reserved for patients whose survivorship is likely to be compromised when surgical resection is not performed within the next 3 mo. Some of them could be node-positive bronchogenic carcinoma, bronchogenic carcinoma greater than 2 cm in the greatest dimension, and solid or predominantly solid (> 50%) bronchogenic carcinoma[61]. However, surgical resection should be deferred for indolent cases of bronchogenic carcinoma, solid nodules less than 2 cm and ground-glass nodules. Based on recommendations in urgent settings, surgical intervention is reserved for patients likely to have poor prognosis if a surgical intervention is not performed within the next few days, such as surgical complications and tumour associated super-infection[61]. Additionally, stereotactic ablative radiotherapy and neoadjuvant therapy are recommended for these patients.
Surgical resection should be deferred for 2-3 wk in patients with bronchogenic carcinoma who test positive for COVID-19 (Figure 2). When a surgical intervention is deemed inevitable, a specialized negative pressure operating theatre with proper usage of personal protective equipment and a negative-pressure isolation room for postoperative care are recommended[60]. If a surgical procedure is rescheduled, it is advisable for bronchogenic patients to be retested for COVID-19 before the procedure. Reasons for this approach include chemotherapy-induced immunosuppression, which might increase the risk of exposure to COVID-19 and severe pulmonary complications henceforth and to help prevent the risk of contracting COVID-19 during frequent trips to the hospital for chemotherapy cycles. Recommendations suggest using neoadjuvant therapy can achieve better prognosis in patients with bronchogenic carcinoma in whom surgical intervention has been postponed due to the above-mentioned reasons[61]. Home management including telemedicine limiting outpatient visits are recommended for cancer patients during this COVID-19 pandemic[62].
Stereotactic ablative radiotherapy and stereotactic body radiotherapy (SBRT) are non-invasive treatment strategies in the management of early stage (< 5 cm) node-negative NSCLC[61,62]. SBRT is a well-established, highly effective treatment modality with minimal risk and good local growth control of tumour[63]. SBRT involves delivery of single fraction of 24-34 Gy for peripheral tumours of size less than 2 cm or delivery of 5 to 10 fractions of 50-70 Gy for central tumours. Considerations are to be made as to delay treatment for small sized slow-growing tumours or to deliver treatment immediately for larger tumours showing rapid growth in patients with bronchogenic carcinoma in whom SBRT is deemed appropriate. However, during the COVID-19 pandemic maximal use of single fraction treatment with shortening of SBRT fractionation schemes are to be implemented. Another promising treatment modality is brachytherapy which involves insertion of a radioactive source within the tumour causing less radiation dose to surrounding tissues adjacent to the tumour, and is primarily utilized for small endobronchial obstructive lesions, recurrent, or early-stage bronchogenic carcinoma. Since brachytherapy increases the risk of exposure to patients and health care providers, it requires multidisciplinary coordination in an enclosed operating theatre with a brachytherapy suite and related personal protective equipment. Preferentially, during the COVID-19 pandemic, considering external beam radiotherapy for treatment of NSCLC patients and avoiding all brachytherapy procedures is recommended[64].
Local resection, systemic chemotherapy and radiotherapy are treatment modalities currently used in the treatment of locally advanced bronchogenic carcinoma, although, concurrent chemoradiotherapy consisting of platinum-based chemotherapy agents with radiotherapy-based delivery of 30 fractions of 60 Gy can also be administered for patients with stage III NSCLC. For curative treatment of bronchogenic carcinoma, factors to be taken into consideration include the risk of contracting COVID-19, adverse effects related to treatment, and availability of resources. Systemic therapies with shorter treatment course and reduced risk of myelosuppression are recommen
Recommendations for cancer therapy include tailored treatment for patients with bronchogenic carcinoma, including NSCLC after weighing the risk-benefit ratio during times of COVID-19 pandemic. However, the prescribed recommendations are aimed at reducing the risk of COVID-19 transmission. Since NSCLC patients are prone to be reactive during the course of treatment even if drug-induced pneumonitis is suspected, it is necessary to rule out COVID-19 related pulmonary syndrome[66]. In light of the current COVID-19 pandemic, low-dose CT of chest which was traditionally performed for bronchogenic carcinoma screening should be rescheduled especially in patients with underlying cardiopulmonary abnormalities which can put them at increased risks for mortality from COVID-19 pneumonia. Additionally, higher infection rates of COVID-19 in patients with bronchogenic carcinoma is partly attributed to the need to visit medical institutions, especially for performing diagnostic chest imaging, which significantly increases the risk of contact with infected patients and medical personnel alike.
COVID-19 has caused considerable disruption in screening of bronchogenic carcinoma. During the pandemic, there were fewer new patients screened, more patients were apprehensive to visit the healthcare system, and an increased proportion of nodules were suspected to be malignant. However, there were rare instances encountered where incidental detection of bronchogenic carcinoma was made possible while performing routine chest CT scans in patients with COVID-19 related pulmo
Clinical research on bronchogenic carcinoma during the current pandemic has been affected due to implementation of strict measures for limitation of COVID-19 spread, long periods of quarantine, lack of resources and varying work shifts. Major research organizations and committees worldwide have implemented decisions of temporarily halting or even permanently stopping clinical research trials for protocol adherence and for preserving the accuracy of endpoint evaluation. Currently clinical trials focused on the research and development of COVID-19 vaccine are the need of the hour, though there is no real surety that a vaccine might be our only ray of hope in these critical times. It is difficult to justify exposing patients to the risk of COVID-19 in clinical trials with a placebo arm, and hence should be suspended for the time being[67].
During pandemics like the current COVID-19, benefits of artificial intelligence (AI) primarily relate to pattern recognition based on imaging findings and the potential of AI for high throughput analysis[68]. Parallel AI processing of images may facilitate triage of patients in institutions located in regions with limited resources and high disease prevalence. Subtle changes across imaging findings on serial CTs may be overlooked, due to human error. However, recent techniques such as AI and computed aided diagnosis may help quantify these changes with precision, and implementation of trend analysis based on baseline trends may help track disease progression in individuals and across populations[69]. The central theme of AI relies on the plasticity of deep learning networks and in adapting to imaging-based recognition of varying disease patterns. Early AI investigators have laid the foundation for detailed exploration of deep learning algorithms for pattern recognition and quantification of disease severity, and for rapid screening of individuals in situations like the current COVID-19 pandemic.
Literature reviews of published earlier studies have reported feasibility of deve
The COVID-19 pandemic has caused a significant health care crisis and has strained the health care systems of developed and developing nations across the world. Recent ESMO recommendations provide social responsibilities for pulmonary oncologists which include providing timely management for cancer patients, while taking necessary precautions to protect patients and themselves from COVID-19 and providing for fair distribution of resources during the pandemic. Treatment decisions should be aimed at striking a balance between risk vs benefit ratio which require multidisciplinary discussions between patients and caregivers. In resource starved settings such as the current COVID-19 pandemic, cancer patients who commit to treatment also have a duty to follow safe health practices and to undergo self-isolation. Risk stratification of bronchogenic carcinoma patients and priority imaging may be applied for optimal use of resources. For the first time in the era of modern medicine, all of humankind is facing the same threat considering the contagiousness of the coronavirus and the need to reduce nosocomial outbreaks which determine poor prognosis and detrimental outcomes in patients with bronchogenic carcinoma. This also gives us ample opportunity to change our research approach to one that involves better understanding of disease manifestations; and to further explore and promote the applications of chest CT for the safe management of patients and health personnel in the setting of pandemics like the present or any which we might encounter in the near future. It has been over 12 mo since we first heard of the COVID-19 pandemic and while promising developments are being made on the clinical trials front and in the management of patients with bronchogenic carcinoma, there is no certainty of events falling into normalcy in the immediate future. Research findings need to be shared amongst countries to provide best health care to humankind during these uncertain times. Modified and newer therapeutic strategies for patients with bronchogenic carcinoma have been adopted by oncologists around the world for providing uncompromised care within the accepted standards and new guidelines.
I wish to thank Mrs. Sabbavarapu M for her assistance in proofreading and native English editing of the manuscript. Also, I wish to extend my sincere gratitude to Dr. Mathew S for providing the voice-over and technical support required for the preparation of the audio core tip.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Indian Radiological and Imaging Association, No. 945LM/KAR-915A.
Specialty type: Oncology
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Palumbo I, Senchukova M S-Editor: Gao CC L-Editor: Filipodia P-Editor: Yuan YY
| 1. | Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18987] [Cited by in RCA: 17648] [Article Influence: 3529.6] [Reference Citation Analysis (0)] |
| 2. | World Health Organization. "We now have a name for the #2019nCoV disease: COVID 19. I’ll spell it: C O V I D hyphen one nine – COVID 19", Tweet, 11 February. [cited 11 Feb 2020]. In: World Health Organization Tweet [Internet]. Available from: https://twitter.com/WHO/status/1227248333871173632. |
| 3. | Case Dashboard. Wuhan Coronavirus (2019 nCoV) Global Cases (by Johns Hopkins CSSE). [cited 19 Jan 2021]. In: Case Dashboard [Internet]. Available from: https://cn.bing.com/search?q=Coronavirus+statistics&FORM=covwpt&tf=U2VydmljZT1HZW5lcmljQW5zd2VycyBTY2VuYXJpbz1Db3JvbmFWaXJ1c01MIFBvc2l0aW9uPVRPUCBSYW5raW5nRGF0YT1UcnVlIEZvcmNlUGxhY2U9VHJ1ZSBQYWlycz1pbnRlbnQ6Q2hlY2tDb3JvbmFTdGF0cztjb3VudHJ5Y29kZTpVU0E7c3RhdGVDb2RlOk1EO2Fib3ZlbmV3czp0cnVlOyB8&hs=otyxJK7R4M18IDuVesI0heK65VTGkVny0rcFpd8Ejls%3d. |
| 4. | Casas-Rojo JM, Antón-Santos JM, Millán-Núñez-Cortés J, Lumbreras-Bermejo C, Ramos-Rincón JM, Roy-Vallejo E, Artero-Mora A, Arnalich-Fernández F, García-Bruñén JM, Vargas-Núñez JA, Freire-Castro SJ, Manzano-Espinosa L, Perales-Fraile I, Crestelo-Viéitez A, Puchades-Gimeno F, Rodilla-Sala E, Solís-Marquínez MN, Bonet-Tur D, Fidalgo-Moreno MP, Fonseca-Aizpuru EM, Carrasco-Sánchez FJ, Rabadán-Pejenaute E, Rubio-Rivas M, Torres-Peña JD, Gómez-Huelgas R; en nombre del Grupo SEMI-COVID-19 Network. Clinical characteristics of patients hospitalized with COVID-19 in Spain: Results from the SEMI-COVID-19 Registry. Rev Clin Esp (Barc). 2020;220:480-494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 206] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 5. | Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, Ren R, Leung KSM, Lau EHY, Wong JY, Xing X, Xiang N, Wu Y, Li C, Chen Q, Li D, Liu T, Zhao J, Liu M, Tu W, Chen C, Jin L, Yang R, Wang Q, Zhou S, Wang R, Liu H, Luo Y, Liu Y, Shao G, Li H, Tao Z, Yang Y, Deng Z, Liu B, Ma Z, Zhang Y, Shi G, Lam TTY, Wu JT, Gao GF, Cowling BJ, Yang B, Leung GM, Feng Z. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N Engl J Med. 2020;382:1199-1207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11224] [Cited by in RCA: 9319] [Article Influence: 1863.8] [Reference Citation Analysis (0)] |
| 6. | Yang Y, Peng F, Wang R, Yange M, Guan K, Jiang T, Xu G, Sun J, Chang C. The deadly coronaviruses: The 2003 SARS pandemic and the 2020 novel coronavirus epidemic in China. J Autoimmun. 2020;109:102434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 590] [Cited by in RCA: 525] [Article Influence: 105.0] [Reference Citation Analysis (1)] |
| 7. | He F, Deng Y, Li W. Coronavirus disease 2019: What we know? J Med Virol. 2020;92:719-725. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 388] [Cited by in RCA: 436] [Article Influence: 87.2] [Reference Citation Analysis (0)] |
| 8. | McAloon C, Collins Á, Hunt K, Barber A, Byrne AW, Butler F, Casey M, Griffin J, Lane E, McEvoy D, Wall P, Green M, O'Grady L, More SJ. Incubation period of COVID-19: a rapid systematic review and meta-analysis of observational research. BMJ Open. 2020;10:e039652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 383] [Cited by in RCA: 304] [Article Influence: 60.8] [Reference Citation Analysis (0)] |
| 9. | Russano M, Citarella F, Napolitano A, Dell'Aquila E, Cortellini A, Pantano F, Vincenzi B, Tonini G, Santini D. COVID-19 pneumonia and immune-related pneumonitis: critical issues on differential diagnosis, potential interactions, and management. Expert Opin Biol Ther. 2020;20:959-964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 10. | Wang Z, Deng H, Ou C, Liang J, Wang Y, Jiang M, Li S. Clinical symptoms, comorbidities and complications in severe and non-severe patients with COVID-19: A systematic review and meta-analysis without cases duplication. Medicine (Baltimore). 2020;99:e23327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 11. | Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11409] [Cited by in RCA: 11507] [Article Influence: 2301.4] [Reference Citation Analysis (0)] |
| 12. | Guan WJ, Liang WH, Zhao Y, Liang HR, Chen ZS, Li YM, Liu XQ, Chen RC, Tang CL, Wang T, Ou CQ, Li L, Chen PY, Sang L, Wang W, Li JF, Li CC, Ou LM, Cheng B, Xiong S, Ni ZY, Xiang J, Hu Y, Liu L, Shan H, Lei CL, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Cheng LL, Ye F, Li SY, Zheng JP, Zhang NF, Zhong NS, He JX; China Medical Treatment Expert Group for COVID-19. Comorbidity and its impact on 1590 patients with COVID-19 in China: a nationwide analysis. Eur Respir J. 2020;55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1711] [Cited by in RCA: 2189] [Article Influence: 437.8] [Reference Citation Analysis (0)] |
| 13. | Dai M, Liu D, Liu M, Zhou F, Li G, Chen Z, Zhang Z, You H, Wu M, Zheng Q, Xiong Y, Xiong H, Wang C, Chen C, Xiong F, Zhang Y, Peng Y, Ge S, Zhen B, Yu T, Wang L, Wang H, Liu Y, Chen Y, Mei J, Gao X, Li Z, Gan L, He C, Shi Y, Qi Y, Yang J, Tenen DG, Chai L, Mucci LA, Santillana M, Cai H. Patients with Cancer Appear More Vulnerable to SARS-CoV-2: A Multicenter Study during the COVID-19 Outbreak. Cancer Discov. 2020;10:783-791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1142] [Cited by in RCA: 972] [Article Influence: 194.4] [Reference Citation Analysis (0)] |
| 14. | Li L, Yang L, Gui S, Pan F, Ye T, Liang B, Hu Y, Zheng C. Association of clinical and radiographic findings with the outcomes of 93 patients with COVID-19 in Wuhan, China. Theranostics. 2020;10:6113-6121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 53] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 15. | Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y, Ma X, Zhan F, Wang L, Hu T, Zhou H, Hu Z, Zhou W, Zhao L, Chen J, Meng Y, Wang J, Lin Y, Yuan J, Xie Z, Ma J, Liu WJ, Wang D, Xu W, Holmes EC, Gao GF, Wu G, Chen W, Shi W, Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8473] [Cited by in RCA: 7602] [Article Influence: 1520.4] [Reference Citation Analysis (0)] |
| 16. | Rossi E, Schinzari G, Tortora G. Pneumonitis from immune checkpoint inhibitors and COVID-19: current concern in cancer treatment. J Immunother Cancer. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 17. | Rogiers A, Pires da Silva I, Tentori C, Tondini CA, Grimes JM, Trager MH, Nahm S, Zubiri L, Manos M, Bowling P, Elkrief A, Papneja N, Vitale MG, Rose AAN, Borgers JSW, Roy S, Mangana J, Pimentel Muniz T, Cooksley T, Lupu J, Vaisman A, Saibil SD, Butler MO, Menzies AM, Carlino MS, Erdmann M, Berking C, Zimmer L, Schadendorf D, Pala L, Queirolo P, Posch C, Hauschild A, Dummer R, Haanen J, Blank CU, Robert C, Sullivan RJ, Ascierto PA, Miller WH Jr, Stephen Hodi F, Suijkerbuijk KPM, Reynolds KL, Rahma OE, Lorigan PC, Carvajal RD, Lo S, Mandala M, Long GV. Clinical impact of COVID-19 on patients with cancer treated with immune checkpoint inhibition. J Immunother Cancer. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 18. | Lax SF, Skok K, Zechner P, Kessler HH, Kaufmann N, Koelblinger C, Vander K, Bargfrieder U, Trauner M. Pulmonary Arterial Thrombosis in COVID-19 With Fatal Outcome : Results From a Prospective, Single-Center, Clinicopathologic Case Series. Ann Intern Med. 2020;173:350-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 600] [Cited by in RCA: 619] [Article Influence: 123.8] [Reference Citation Analysis (0)] |
| 19. | Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054-1062. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17476] [Cited by in RCA: 18201] [Article Influence: 3640.2] [Reference Citation Analysis (0)] |
| 20. | Liang W, Guan W, Chen R, Wang W, Li J, Xu K, Li C, Ai Q, Lu W, Liang H, Li S, He J. Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China. Lancet Oncol. 2020;21:335-337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3332] [Cited by in RCA: 3125] [Article Influence: 625.0] [Reference Citation Analysis (0)] |
| 21. | Carreira H, Strongman H, Peppa M, McDonald HI, Dos-Santos-Silva I, Stanway S, Smeeth L, Bhaskaran K. Prevalence of COVID-19-related risk factors and risk of severe influenza outcomes in cancer survivors: A matched cohort study using linked English electronic health records data. EClinicalMedicine. 2020;29-30:100656. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 22. | Peng L, Liang F, Xia Y. Risk of COVID-19 in Patients With Cancer. JAMA Oncol. 2020;6:1469-1470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Leclère JB, Fournel L, Etienne H, Al Zreibi C, Onorati I, Roussel A, Castier Y, Martinod E, Le Pimpec-Barthes F, Alifano M, Assouad J, Mordant P; Thoracic Surgery Units of Paris Public Hospitals. Maintaining Surgical Treatment of Non-Small Cell Lung Cancer During the COVID-19 Pandemic in Paris. Ann Thorac Surg. 2021;111:1682-1688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Bertaglia V, Reale ML, Bironzo P, Palesandro E, Mariniello A, Leone G, Tabbò F, Bungaro M, Audisio M, Rapetti S, Di Stefano RF, Carnio S, Artusio E, Capelletto E, Sperone P, Passiglia F, Novello S. Italian survey on the clinical management of non-small cell lung cancer patients during the COVID-19 pandemic: A lesson for the second wave. Crit Rev Oncol Hematol. 2021;157:103189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 25. | Dramé M, Tabue Teguo M, Proye E, Hequet F, Hentzien M, Kanagaratnam L, Godaert L. Should RT-PCR be considered a gold standard in the diagnosis of COVID-19? J Med Virol. 2020;92:2312-2313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 127] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 26. | Kwee TC, Kwee RM. Chest CT in COVID-19: What the Radiologist Needs to Know. Radiographics. 2020;40:1848-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 263] [Cited by in RCA: 250] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 27. | American College of Radiology. ACR Recommendations for the use of chest radiography and computed tomography (CT) for suspected COVID-19 infection. [cited 19 Jan 2021]. In: American College of Radiology [Internet]. Available from: https://www.acr.org/Advocacy-and-Economics/ACR-Position-Statements/Recommendations-for-Chest-Radiography-and-CT-for-Suspected-COVID19-Infection. |
| 28. | McStay R, Johnstone A, Hare SS, Jacob J, Nair A, Rodrigues JCL, Edey A, Robinson G. COVID-19: looking beyond the peak. Challenges and tips for radiologists in follow-up of a novel patient cohort. Clin Radiol. 2021;76:74.e1-74.e14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 29. | Kim H, Hong H, Yoon SH. Diagnostic Performance of CT and Reverse Transcriptase Polymerase Chain Reaction for Coronavirus Disease 2019: A Meta-Analysis. Radiology. 2020;296:E145-E155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 354] [Cited by in RCA: 367] [Article Influence: 73.4] [Reference Citation Analysis (0)] |
| 30. | Cleverley J, Piper J, Jones MM. The role of chest radiography in confirming covid-19 pneumonia. BMJ. 2020;370:m2426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 94] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 31. | Böger B, Fachi MM, Vilhena RO, Cobre AF, Tonin FS, Pontarolo R. Systematic review with meta-analysis of the accuracy of diagnostic tests for COVID-19. Am J Infect Control. 2021;49:21-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 211] [Cited by in RCA: 303] [Article Influence: 75.8] [Reference Citation Analysis (0)] |
| 32. | Fields BKK, Demirjian NL, Gholamrezanezhad A. Coronavirus Disease 2019 (COVID-19) diagnostic technologies: A country-based retrospective analysis of screening and containment procedures during the first wave of the pandemic. Clin Imaging. 2020;67:219-225. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 33. | Besutti G, Giorgi Rossi P, Iotti V, Spaggiari L, Bonacini R, Nitrosi A, Ottone M, Bonelli E, Fasano T, Canovi S, Colla R, Massari M, Lattuada IM, Trabucco L, Pattacini P; Reggio Emilia COVID-19 Working Group. Accuracy of CT in a cohort of symptomatic patients with suspected COVID-19 pneumonia during the outbreak peak in Italy. Eur Radiol. 2020;30:6818-6827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 34. | Robilotti EV, Babady NE, Mead PA, Rolling T, Perez-Johnston R, Bernardes M, Bogler Y, Caldararo M, Figueroa CJ, Glickman MS, Joanow A, Kaltsas A, Lee YJ, Lucca A, Mariano A, Morjaria S, Nawar T, Papanicolaou GA, Predmore J, Redelman-Sidi G, Schmidt E, Seo SK, Sepkowitz K, Shah MK, Wolchok JD, Hohl TM, Taur Y, Kamboj M. Determinants of COVID-19 disease severity in patients with cancer. Nat Med. 2020;26:1218-1223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 446] [Article Influence: 89.2] [Reference Citation Analysis (0)] |
| 35. | Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, Chung M, Henry TS, Kanne JP, Kligerman S, Ko JP, Litt H. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA - Secondary Publication. J Thorac Imaging. 2020;35:219-227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 400] [Cited by in RCA: 564] [Article Influence: 112.8] [Reference Citation Analysis (0)] |
| 36. | Wang A, Gao G, Wang S, Chen M, Qian F, Tang W, Xu Y, Song R, Zhuang L, Ma X, Zhao T, Guo X, Li W, Wang X, Li B, Hu C, Chen Z, Zhang F. Clinical Characteristics and Risk Factors of Acute Respiratory Distress Syndrome (ARDS) in COVID-19 Patients in Beijing, China: A Retrospective Study. Med Sci Monit. 2020;26:e925974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 37. | Donaldsson S, Naver L, Jonsson B, Drevhammar T. COVID-19: minimising contaminated aerosol spreading during CPAP treatment. Arch Dis Child Fetal Neonatal Ed. 2020;105:669-671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 38. | Steinfort DP, Herth FJF, Irving LB, Nguyen PT. Safe performance of diagnostic bronchoscopy/EBUS during the SARS-CoV-2 pandemic. Respirology. 2020;25:703-708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 39. | Fillmore NR, La J, Szalat RE, Tuck DP, Nguyen V, Yildirim C, Do NV, Brophy MT, Munshi NC. Prevalence and outcome of COVID-19 infection in cancer patients: a national Veterans Affairs study. J Natl Cancer Inst. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 69] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 40. | Lang M, Yeung T, Shepard JO, Sharma A, Petranovic M, Flores EJ, McLoud TC, Som A, Saini S, Prabhakar AM, D Succi M, Little BP. Operational Challenges of a Low-Dose CT Lung Cancer Screening Program During the Coronavirus Disease 2019 Pandemic. Chest. 2021;159:1288-1291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 41. | Mazzone PJ, Gould MK, Arenberg DA, Chen AC, Choi HK, Detterbeck FC, Farjah F, Fong KM, Iaccarino JM, Janes SM, Kanne JP, Kazerooni EA, MacMahon H, Naidich DP, Powell CA, Raoof S, Rivera MP, Tanner NT, Tanoue LK, Tremblay A, Vachani A, White CS, Wiener RS, Silvestri GA. Management of Lung Nodules and Lung Cancer Screening During the COVID-19 Pandemic: CHEST Expert Panel Report. J Am Coll Radiol. 2020;17:845-854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 42. | Passaro A, Addeo A, Von Garnier C, Blackhall F, Planchard D, Felip E, Dziadziuszko R, de Marinis F, Reck M, Bouchaab H, Peters S. ESMO Management and treatment adapted recommendations in the COVID-19 era: Lung cancer. ESMO Open. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 43. | Hashemi-Madani N, Emami Z, Janani L, Khamseh ME. Typical chest CT features can determine the severity of COVID-19: A systematic review and meta-analysis of the observational studies. Clin Imaging. 2021;74:67-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 44. | Faivre-Finn C, Fenwick JD, Franks KN, Harrow S, Hatton MQF, Hiley C, McAleese JJ, McDonald F, O'Hare J, Peedell C, Pope T, Powell C, Rulach R, Toy E. Reduced Fractionation in Lung Cancer Patients Treated with Curative-intent Radiotherapy during the COVID-19 Pandemic. Clin Oncol (R Coll Radiol). 2020;32:481-489. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 45. | Sullivan RJ, Johnson DB, Rini BI, Neilan TG, Lovly CM, Moslehi JJ, Reynolds KL. COVID-19 and immune checkpoint inhibitors: initial considerations. J Immunother Cancer. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 46. | Vergnon JM, Trosini-Desert V, Fournier C, Lachkar S, Dutau H, Guibert N, Escarguel B, Froudarakis M; French-Speaking Group on Thoracic Endoscopy (Groupe d’endoscopie de langue française GELF) of the French Language Respiratory Society (Société de pneumologie de langue française; SPLF). Bronchoscopy use in the COVID-19 era. Respir Med Res. 2020;78:100760. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 47. | Wahidi MM, Lamb C, Murgu S, Musani A, Shojaee S, Sachdeva A, Maldonado F, Mahmood K, Kinsey M, Sethi S, Mahajan A, Majid A, Keyes C, Alraiyes AH, Sung A, Hsia D, Eapen G. American Association for Bronchology and Interventional Pulmonology (AABIP) Statement on the Use of Bronchoscopy and Respiratory Specimen Collection in Patients With Suspected or Confirmed COVID-19 Infection. J Bronchology Interv Pulmonol. 2020;27:e52-e54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 48. | Endo F. Re: Holmium laser enucleation of the prostate: a modified enucleation technique and initial results: Y. G. Gong, D. L. He, M. Z. Wang, X. D. Li, G. D. Zhu, Z. H. Zheng, Y. F. Du, L. S. Chang and X. Y. Nan J Urol 2012; 187: 1336-1340. J Urol. 2013;189:395-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 49. | Pascarella G, Strumia A, Piliego C, Bruno F, Del Buono R, Costa F, Scarlata S, Agrò FE. COVID-19 diagnosis and management: a comprehensive review. J Intern Med. 2020;288:192-206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 702] [Cited by in RCA: 733] [Article Influence: 146.6] [Reference Citation Analysis (0)] |
| 50. | Shaverdian N, Shepherd AF, Rimner A, Wu AJ, Simone CB 2nd, Gelblum DY, Gomez DR. Need for Caution in the Diagnosis of Radiation Pneumonitis During the COVID-19 Pandemic. Adv Radiat Oncol. 2020;5:617-620. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 51. | Beer L, Hochmair M, Kifjak D, Haug AR, Prayer F, Mayerhoefer ME, Herold C, Prosch H. Particular findings on lung CT in patients undergoing immunotherapy for bronchogenic carcinoma. Wien Klin Wochenschr. 2020;132:467-474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 52. | Kumar S, Chmura S, Robinson C, Lin SH, Gadgeel SM, Donington J, Feliciano J, Stinchcombe TE, Werner-Wasik M, Edelman MJ, Moghanaki D. Alternative Multidisciplinary Management Options for Locally Advanced NSCLC During the Coronavirus Disease 2019 Global Pandemic. J Thorac Oncol. 2020;15:1137-1146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 53. | Kidane B, Spicer J, Kim JO, Fiset PO, Abdulkarim B, Malthaner R, Palma D. SABR-BRIDGE: Stereotactic ABlative Radiotherapy Before Resection to AvoId Delay for Early-Stage LunG Cancer or OligomEts During the COVID-19 Pandemic. Front Oncol. 2020;10:580189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 54. | Moletta L, Pierobon ES, Capovilla G, Costantini M, Salvador R, Merigliano S, Valmasoni M. International guidelines and recommendations for surgery during Covid-19 pandemic: A Systematic Review. Int J Surg. 2020;79:180-188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 188] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 55. | Pennell NA, Dillmon M, Levit LA, Moushey EA, Alva AS, Blau S, Cannon TL, Dickson NR, Diehn M, Gonen M, Gonzalez MM, Hensold JO, Hinyard LJ, King T, Lindsey SC, Magnuson A, Marron J, McAneny BL, McDonnell TM, Mileham KF, Nasso SF, Nowakowski GS, Oettel KR, Patel MI, Patt DA, Perlmutter J, Pickard TA, Rodriguez G, Rosenberg AR, Russo B, Szczepanek C, Smith CB, Srivastava P, Teplinsky E, Thota R, Traina TA, Zon R, Bourbeau B, Bruinooge SS, Foster S, Grubbs S, Hagerty K, Hurley P, Kamin D, Phillips J, Schenkel C, Schilsky RL, Burris HA 3rd. American Society of Clinical Oncology Road to Recovery Report: Learning From the COVID-19 Experience to Improve Clinical Research and Cancer Care. J Clin Oncol. 2021;39:155-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 71] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 56. | Lambertini M, Toss A, Passaro A, Criscitiello C, Cremolini C, Cardone C, Loupakis F, Viscardi G, Meattini I, Dieci MV, Ferrara R, Giusti R, Maio MD. Cancer care during the spread of coronavirus disease 2019 (COVID-19) in Italy: young oncologists' perspective. ESMO Open. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 124] [Cited by in RCA: 137] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 57. | Howe JR, Bartlett DL, Tyler DS, Wong SL, Hunt KK, DeMatteo RP; Society of Surgical Oncology. COVID-19 Guideline Modifications as CMS Announces "Opening Up America Again": Comments from the Society of Surgical Oncology. Ann Surg Oncol. 2020;27:2111-2113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 58. | Ti LK, Ang LS, Foong TW, Ng BSW. What we do when a COVID-19 patient needs an operation: operating room preparation and guidance. Can J Anaesth. 2020;67:756-758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 265] [Cited by in RCA: 284] [Article Influence: 56.8] [Reference Citation Analysis (0)] |
| 59. | Bartlett DL, Howe JR, Chang G, Crago A, Hogg M, Karakousis G, Levine E, Maker A, Mamounas E, McGuire K, Merchant N, Shibata D, Sohn V, Solorzano C, Turaga K, White R, Yang A, Yoon S; Society of Surgical Oncology. Management of Cancer Surgery Cases During the COVID-19 Pandemic: Considerations. Ann Surg Oncol. 2020;27:1717-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 164] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 60. | Elkaddoum R, Haddad FG, Eid R, Kourie HR. Telemedicine for cancer patients during COVID-19 pandemic: between threats and opportunities. Future Oncol. 2020;16:1225-1227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 61. | Moreno AC, Fellman B, Hobbs BP, Liao Z, Gomez DR, Chen A, Hahn SM, Chang JY, Lin SH. Biologically Effective Dose in Stereotactic Body Radiotherapy and Survival for Patients With Early-Stage NSCLC. J Thorac Oncol. 2020;15:101-109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 62. | Mayne NR, Lin BK, Darling AJ, Raman V, Patel DC, Liou DZ, D'Amico TA, Yang CJ. Stereotactic Body Radiotherapy Versus Delayed Surgery for Early-stage Non-small-cell Lung Cancer. Ann Surg. 2020;272:925-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 63. | Dingemans AC, Soo RA, Jazieh AR, Rice SJ, Kim YT, Teo LLS, Warren GW, Xiao SY, Smit EF, Aerts JG, Yoon SH, Veronesi G, De Cobelli F, Ramalingam SS, Garassino MC, Wynes MW, Behera M, Haanen J, Lu S, Peters S, Ahn MJ, Scagliotti GV, Adjei AA, Belani CP. Treatment Guidance for Patients With Lung Cancer During the Coronavirus 2019 Pandemic. J Thorac Oncol. 2020;15:1119-1136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 64. | Xu Y, Liu H, Hu K, Wang M. Clinical recommendations on lung cancer management during the COVID-19 pandemic. Thorac Cancer. 2020;11:2067-2074. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 65. | Dumoulin DW, Gietema HA, Paats MS, Hendriks LEL, Cornelissen R. Differentiation of COVID-19 Pneumonitis and ICI Induced Pneumonitis. Front Oncol. 2020;10:577696. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 66. | Sathian B, Asim M, Banerjee I, Pizarro AB, Roy B, van Teijlingen ER, do Nascimento IJB, Alhamad HK. Impact of COVID-19 on clinical trials and clinical research: A systematic review. Nepal J Epidemiol. 2020;10:878-887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 157] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 67. | Albadr MAA, Tiun S, Ayob M, Al-Dhief FT, Omar K, Hamzah FA. Optimised genetic algorithm-extreme learning machine approach for automatic COVID-19 detection. PLoS One. 2020;15:e0242899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 68. | Chassagnon G, Vakalopoulou M, Paragios N, Revel MP. Artificial intelligence applications for thoracic imaging. Eur J Radiol. 2020;123:108774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 111] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 69. | Ather S, Kadir T, Gleeson F. Artificial intelligence and radiomics in pulmonary nodule management: current status and future applications. Clin Radiol. 2020;75:13-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 91] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 70. | Gonem S, Janssens W, Das N, Topalovic M. Applications of artificial intelligence and machine learning in respiratory medicine. Thorax. 2020;75:695-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |