Published online Sep 10, 2019. doi: 10.4291/wjgp.v10.i2.29
Peer-review started: July 16, 2019
First decision: August 2, 2019
Revised: August 16, 2019
Accepted: August 21, 2019
Article in press: August 21, 2019
Published online: September 10, 2019
Processing time: 67 Days and 15.1 Hours
Intraabdominal adhesions develop spontaneously or after an inflammatory process or surgical procedure in the abdomen. They are the most common cause of small bowel obstruction (SBO). SBO occasionally leads to intestinal ischemia (InIs) which can be a life-threatening condition that requires management as soon as possible. We herein report a case of SBO with InIs presented in our institution and treated without intestinal resection.
A 34-year-old man presented at the emergency department after a 12-h-onset diffuse abdominal pain, bloating and nausea. He had a history of traumatic right hepatectomy 11 years ago as well as adhesiolysis and resection of a long part of small bowel 2 years ago. An abdominal computed tomography (CT) showed dilated loops that led to the diagnosis of SBO. Due to deteriorating lactic acidosis, the patient was operated. Torsion of the small bowel around an adhesion led to 2.30 m of ischemic ileum. After the application of N/S 40 °C for 20 min, the intestine showed signs of improvement and it was decided to avoid resection and instead temporary close the abdomen with vacuum-pack technique. At the second-look laparotomy 48 h later, the intestine appeared normal. The patient was discharged on the 8th post-op day in excellent condition.
In case of SBO caused by adhesions, extreme caution is needed if InIs is present, as the clinical signs are mild and you should rely for diagnosis in CT findings and lactate levels. Conservative surgical approach could reverse the effects of InIs, if performed quickly, so that intestinal resection is avoided and should be used even when minimum signs of viability are present.
Core tip: Intraabdominal adhesions are the most common cause of small bowel obstruction which occasionally leads to intestinal ischemia (InIs). InIs can be a life-threatening condition that requires high index of suspicion because the clinical signs are mild and you should rely for diagnosis in computed tomography findings and lactate levels. Lactate, if present is a good indicator of the progress or regression of the disease. Diagnosis of InIs leads to immediate operation. Conservative surgical approach avoiding resection of ischemic bowel, could lead to reversal of InIs and should be used even when minimum signs of viability are present.
- Citation: Vassiliu P, Ntella V, Theodoroleas G, Mantanis Z, Pentara I, Papoutsi E, Mastoraki A, Arkadopoulos N. Successful management of adhesion related small bowel ischemia without intestinal resection: A case report and review of literature. World J Gastrointest Pathophysiol 2019; 10(2): 29-35
- URL: https://www.wjgnet.com/2150-5330/full/v10/i2/29.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v10.i2.29
Small bowel obstruction (SBO) is a major cause of hospital admissions (15% in United States) and is associated with significantly high rates of patients’ morbidity and even mortality[1,2]. Post-operative abdominal adhesions play a pivotal role in SBO detection while 10% of affected patients are attributed to peritonitis and only 5% of SBO cases remain of unknown origin[2]. Generally, abdominal adhesions are created either spontaneously, by an inflammatory process or after a surgical procedure in the abdomen and pelvis[3,4]. Moreover, adhesive SBO (aSBO) occurs in 3% of all laparotomies and 1% during the first postoperative year. Recurrence rate after aSBO is 19%-53%[5]. Patients with SBO usually present with colicky abdominal pain, distention, nausea, vomiting, constipation and obstipation[1,6,7]. Laboratory findings include in the majority of the cases mild leukocytosis and possibly electrolyte disturbances[1,8]. Abdominal computed tomography (CT) remains the gold-standard diagnostic procedure for aSBO[9,10].
Initial management of aSBO is conservative including nasogastric tube for intestinal decompression with parallel administration of intravenous fluids and analgesics and electrolyte correction on occasion[5,11,12]. In accordance with recent investigations 70% of aSBO cases resolve without operative intervention[5]. However, one of the possible outcomes of SBO is intestinal strangulation which eventually leads to ischemia[5,13]. Intestinal ischemia (InIs) is a potentially lethal complication as it presents without specific symptoms and it requires high index of suspicion for establishment of the diagnosis[14]. Elevated lactate levels and acidosis are indicative of an upcoming ischemia and should also be taken into account during the patient’s management[13,15-17]. Additional CT findings implying InIs are mesenteric edema, free peritoneal fluid and a transitional point, while parietal pneumatosis is a sign of irreversible bowel wall necrosis[2,13]. Thus, in case of evidence of small bowel strangulation, subsequent laparotomy is mandatory. We here present a case of aSBO with signs of intestinal strangulation and ischemia due to torsion around a dense adhesion at mid-ileum in a young male patient successfully treated without intestinal resection. Relevant literature is briefly reviewed. Clinical features and imaging findings are discussed, while the role of current diagnostic and therapeutic management of this nosologic entity is emphasized.
Α 34-year-old man was admitted due to a 12-h-onset increasing diffuse abdominal pain.
The patient also appeared to our emergency department with general malaise and reported bloating and nausea.
He also reported similar clinical symptoms in the past. He was a 20 pack-year smoker. Moreover, the patient had a history of partial right hepatectomy due to liver trauma in 2007 and 1.5 m of small bowel resection due to aSBO in 2016.
On meticulous physical examination, he presented mild tachycardia, tachypnea as well as distended abdomen with guarding and diminished bowel sounds.
Apart from elevated lactate levels (Lac = 2.2 mmol/L), common laboratory blood and urine examinations were within normal limits.
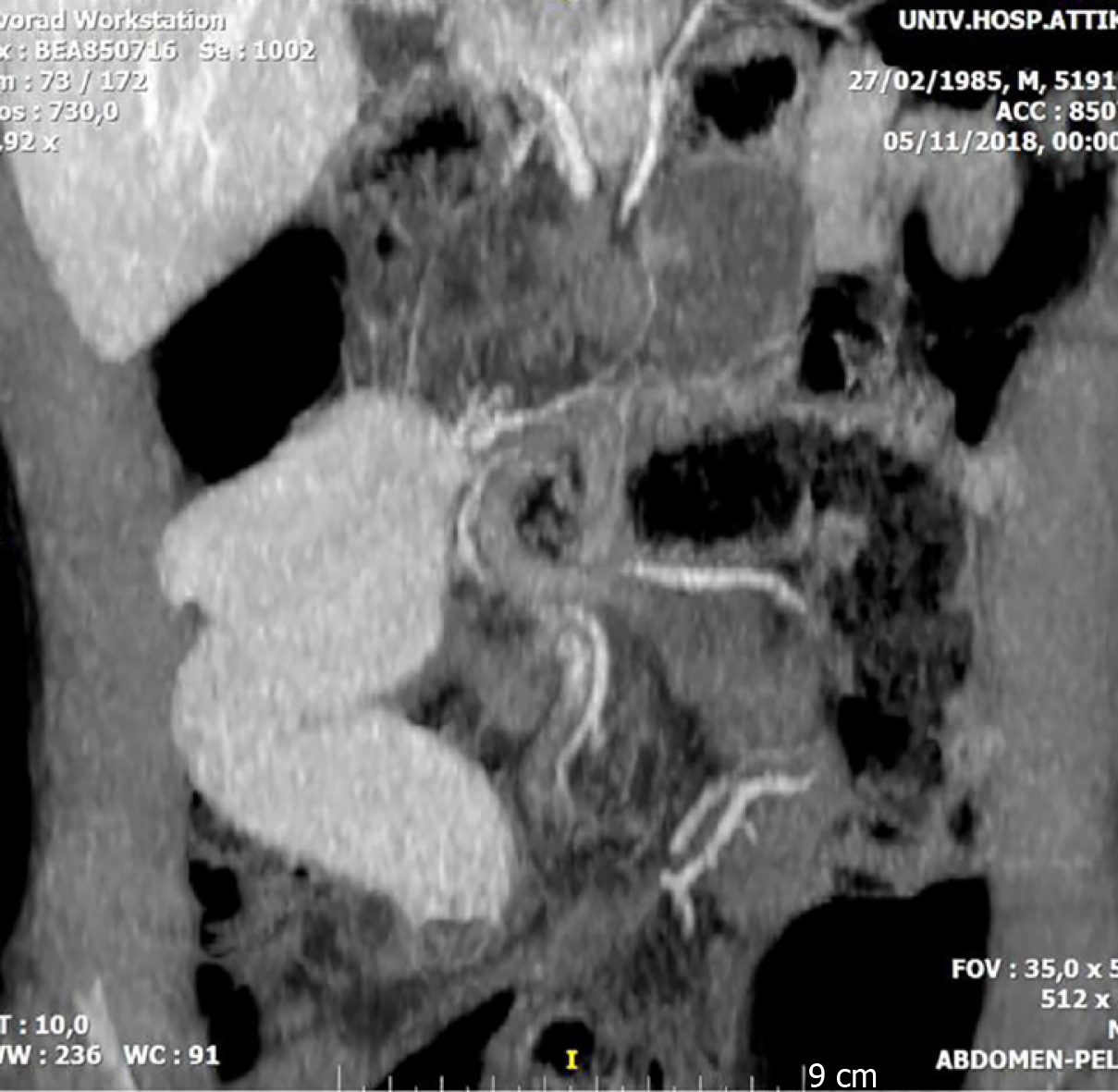
Subsequent ultrasonography was not indicative of pathologic findings. Erect abdominal X-rays depicted limited small bowel fluid levels, indicating intestinal obstruction. Subsequent CT scan identified the presence of bowel dilation and wall edema with parallel winding of the major vessels of gastrointestinal (GI) tract at the transitional zone, assessing InIs without signs of vascular obstruction or thrombosis.
Therefore, the final diagnosis was aSBO (Figure 1). Additional CT angiography as well as low molecular heparin administration was not implemented as signs of thrombosis were absent and intestinal strangulation was verified.
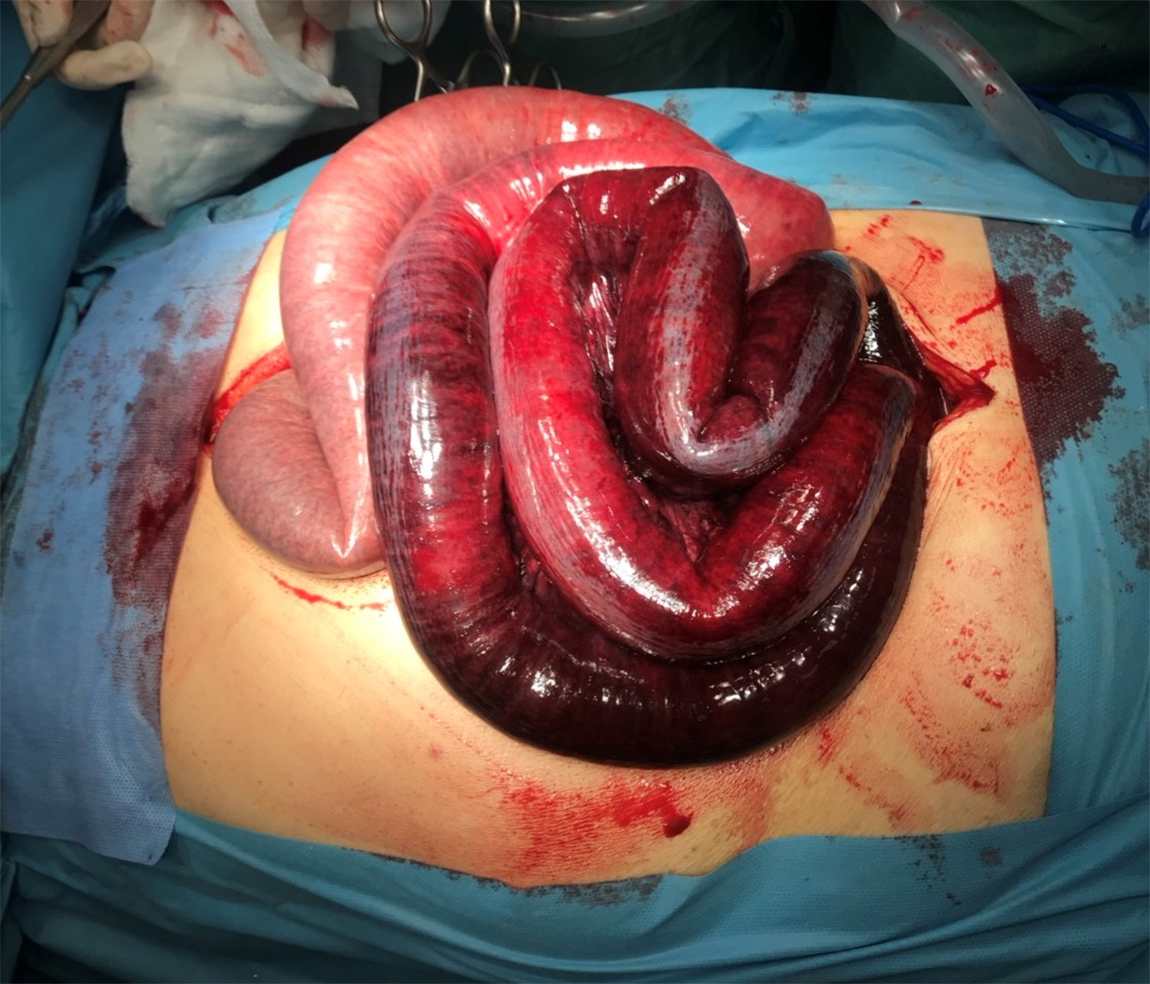
Six hours after admission and fluid resuscitation, the patient was submitted to exploratory laparotomy, due to deteriorating lactic acidosis (Lac = 8 mmol/L) raising strong suspicion of evolving InIs. Surgical operation was performed via a midline incision while the presence of normal bowel from the ligament of Treitz to a length of 1.30 m of jejunum was verified. On the contrary, the rest of the small bowel including ileocolic junction was ischemic and congested due to twirling around an adhesion created at the location of an intestinal anastomosis. A diminutive sign of peristalsis was apparent at the most distal ischemic bowel loops (Figure 2). Resection was reserved as last treatment option due to the short length of the remaining healthy bowel. Rewarming with N/S (40 oC) was applied for 20 min. The intestine, slightly but clearly, improved in color and peristalsis in about half of its ischemic length. Lactic acidosis was also reduced (Lac = 6 mmol/L) during this operation allowing us to postpone resection. The abdomen was temporarily closed with vacuum-pack technique, searching for a second look laparotomy in forty-eight hours. During this period, lactate was normalized (Lac = 0.8 mmol/L).
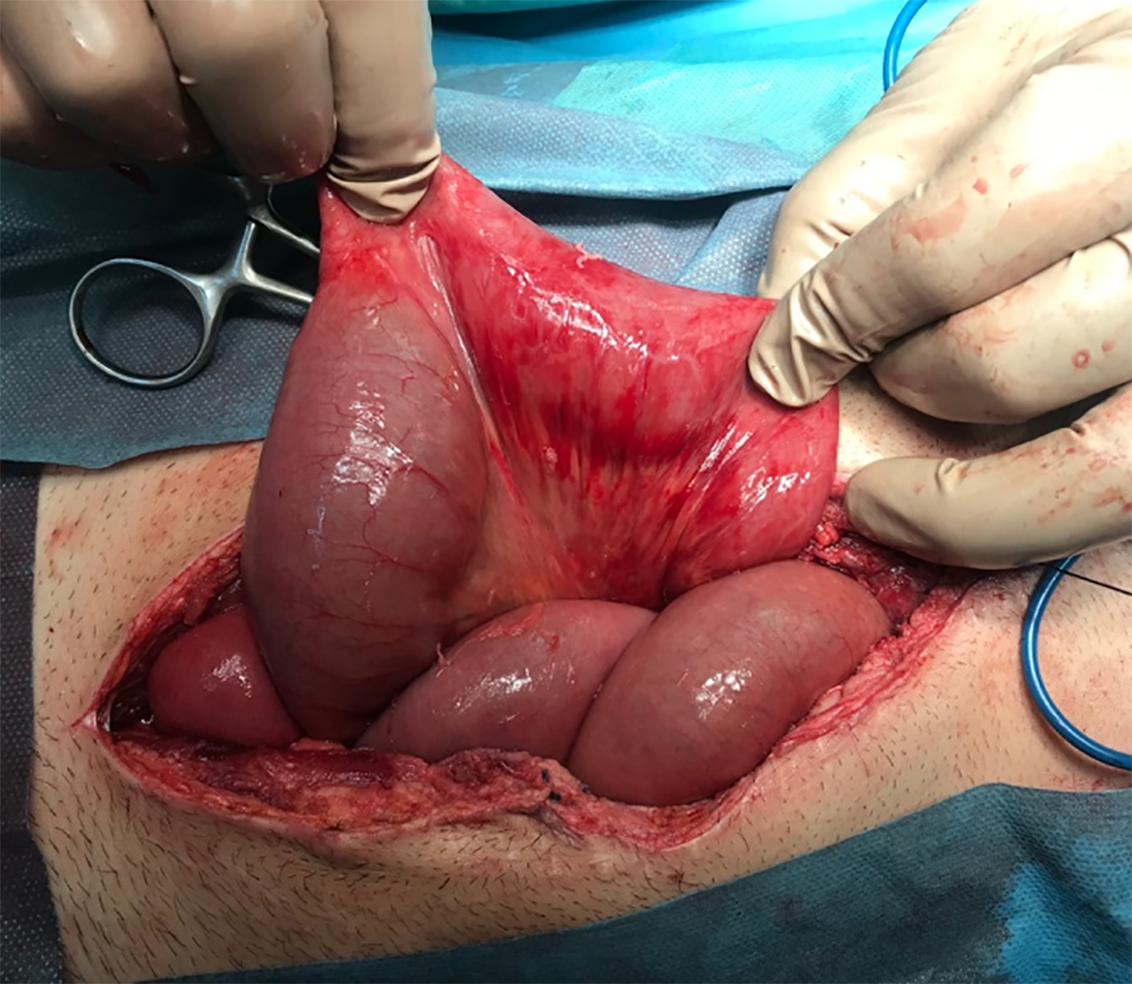
Subsequent second look operation elucidated that the bowel was perfused in all its length and peristalsis was present (Figure 3). The abdomen was closed permanently. Hospital stay was uneventful and he was discharged from the surgical department 8 d postoperatively. Twenty-four hours later, the patient complained for bloody diarrheas, which was expected due to the mucosal apoptosis. Finally, the last 6 mo, the patient remains under intense monitoring without signs of relapse.
Intra-abdominal adhesions are fibrous bands between organs or tissues that are usually formed after an abdominal or pelvic operation, an inflammatory process, or an abdominal trauma[18]. In fact, it has been elucidated, that they may develop after abdominal surgeries in about 67% of the cases and in 97% of patients undergoing gynecological procedures[19,4]. However, the formation of adhesions varies among patients and depends on the severity and type of the initial surgery, potential post-operative complications as well as the presence of intra-peritoneal foreign bodies[3]. In general, according to a recent survey, the overall incidence of aSBO is 4.6%[20]. More specifically, aSBO complicates 19.3% among ileal-anal pouch anastomoses, while affects 11.1% of patients submitted to gynecological surgical operations. Nonetheless, the technique of the procedure affects significantly the development of SBO[20,21]. Consequently, aSBO prevalence was 9.5% in open colectomy versus 4.3% in laparoscopic one and 7.1% in open cholecystectomy vs 0.2% in relevant laparoscopic operation. Contrariwise, there was no difference in the development of aSBO after open or laparoscopic appendectomies (1.4% vs 1.3%)[20]. Finally, laparotomy due to abdominal trauma, was complicated by aSBO in 2.5% of the cases, whereas the incidence of early post-op SBO (within 30 days after surgery) was 24.2%[20,22]. Our case involves a 34-year-old man with a history of partial hepatectomy due to liver trauma 11 years ago, which was followed by a SBO and 1.5 m intestinal resection 2 years ago and now presented with aSBO and subsequent InIs.
Although the clinical presentation of aSBO depends on the location and duration of the obstruction, it usually involves colicky abdominal pain, abdominal distention, nausea and vomiting related to the site of intestinal strangulation[1,23]. Most patients report a history of prior abdominal surgery[24]. In case of ischemia though, intermittent abdominal pain becomes more severe and located while fever should always raise suspicion of InIs and sepsis[1,6]. Meticulous physical examination usually reveals a restless patient with signs of dehydration, poor skin turgor and abdominal distention[1,23]. During inspection visible peristalsis is sometimes observed. In subsequent auscultation, bowel sounds are initially increased and high-pitched, but diminish with advanced SBO[1]. Signs that SBO has resulted in InIs or perforation (peritonitis) include affected vital signs such as tachycardia, tachypnea and pyrexia as well as rebound tenderness, guarding and abdominal rigidity. Our patient presented with a 12-h-onset increasing diffuse abdominal pain, bloating and nausea and on examination mild tachycardia, tachypnea, distended abdomen with guarding and diminished bowel sounds were documented.
Laboratory findings suggestive of SBO more often include mild leukocytosis and possibly electrolyte disturbances due to dehydration and third space volume loss[8]. Elevated lactate levels and acidosis should be also considered through the patient’s management as they strongly indicate intestinal strangulation[13]. In fact, lactate although not universally present at ischemia, when apparent is a good indicator of the progress or regression of the disease[25]. Imaging techniques that are proposed for aSBO vary according to the initial clinical signs, but mostly abdominal CT can contribute to the diagnosis of this condition as it may often accurately identify strangulation in aSBO[26,27]. Imaging findings may also include reduced bowel wall enhancement and mesenteric fluid detection indicative of strangulation as well as small bowel wall thickening associated with aSBO[28,29]. Ideally, only patients who develop strangulation should undergo surgery, and these operations should be performed promptly to avoid transmural necrosis and perforation. Early recognition of SBO strangulation is thus pivotal to help improve the patient outcome by preserving the involved bowel loops and avoiding needless surgical resection[30]. Nevertheless, regardless of the cause, obstructed bowel eventually becomes edemateous, leading to bowel ischemia, inflammation and necrosis. Strangulation is usually verified if at least one of the following criteria is fulfilled including bluish discoloration, loss of arterial pulsation, subserosal and mesenteric hemorrhage, lack of peristalsis and frank infarction. In our patient, abdominal CT scan revealed bowel dilation and wall edema with winding of the major vessels of GI tract predicting InIs.
Our case concerns a young man with aSBO and resultant ischemia of a 2.30 m long intestinal loop which involved the ileocecal valve. Thus, even though initial management of aSBO is usually conservative and the majority of aSBO cases resolve without surgical intervention, given the signs of InIs, laparotomy was mandatory[2,5,11]. The standard procedure for this condition involves resection of the ischemic bowel and primary anastomosis[31]. However, in the present case, resection would lead to short bowel syndrome; a mal-absorptive state caused by massive intestinal resection[32,33]. It usually occurs when the small bowel is less than 2 m long, so that its absorptive function is diminished and the patient cannot maintain its nutritional status and homeostasis by the enteral route alone. These patients are initially dependent on total parenteral nutrition which could progressively lead to liver insufficiency[32,34]. In order to avoid this outcome, taking into consideration that the physiologic consequences of ischemia are still reversible within six hours from onset, and based on diminutive sign of peristalsis at the ischemic ileum, it was decided initially a more conservative approach without intestinal resection[16]. The option of surgical excision was reserved for the second look laparotomy in case that the bowel became necrotic. Fortunately, at the second-look operation the small bowel appeared normal and the resection wasn’t warranted. In this way and even though the intestinal loop at first seemed marginally viable judging from its color and size, the ischemia was reversed, and the patient improved quickly without complications.
The treatment of aSBO should be, at the beginning, conservative excluding cases presented with clinical signs or CT findings predictive of surgical intervention or peritonitis requiring an urgent laparoscopic or laparotomic exploration[2]. On occasion, conservative surgical approach avoiding resection of ischemic bowel, could lead to reversal of InIs and should be initiated even when minimum signs of viability are present and also when less than six hours have passed from the onset of ischemia, time limit allowing reversal of ischemic damage. The second look laparotomy in 24-48 h is an alternative that allows, under the condition of close monitoring (vital signs, clinical evaluation for acute abdomen, lactate levels), the reperfusion and resolution of ischemic impairment of a marginally viable bowel, or deterioration and early resection based on the close monitoring.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Greece
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Huan CM, Lan C, Yuan JY S-Editor: Yan JP L-Editor: A E-Editor: Li X
| 1. | Rami Reddy SR, Cappell MS. A Systematic Review of the Clinical Presentation, Diagnosis, and Treatment of Small Bowel Obstruction. Curr Gastroenterol Rep. 2017;19:28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 142] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 2. | Assenza M, De Gruttola I, Rossi D, Castaldi S, Falaschi F, Giuliano G. Adhesions small bowel obstruction in emergency setting: Conservative or operative treatment? G Chir. 2016;37:145-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 3. | Attard JA, MacLean AR. Adhesive small bowel obstruction: Epidemiology, biology and prevention. Can J Surg. 2007;50:291-300. [PubMed] |
| 4. | Kijima T, Hyakudomi R, Hashimoto T, Kusaka A, Nakatani T, Ishibashi Y. Adhesion-induced chronic abdominal pain: A case report on the diagnostic value of Carnett's test. J Med Case Rep. 2019;13:93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Hajibandeh S, Hajibandeh S, Panda N, Khan RMA, Bandyopadhyay SK, Dalmia S, Malik S, Huq Z, Mansour M. Operative versus non-operative management of adhesive small bowel obstruction: A systematic review and meta-analysis. Int J Surg. 2017;45:58-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Azagury D, Liu RC, Morgan A, Spain DA. Small bowel obstruction: A practical step-by-step evidence-based approach to evaluation, decision making, and management. J Trauma Acute Care Surg. 2015;79:661-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 7. | Maung AA, Johnson DC, Piper GL, Barbosa RR, Rowell SE, Bokhari F, Collins JN, Gordon JR, Ra JH, Kerwin AJ; Eastern Association for the Surgery of Trauma. Evaluation and management of small-bowel obstruction: An Eastern Association for the Surgery of Trauma practice management guideline. J Trauma Acute Care Surg. 2012;73:S362-S369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 177] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 8. | Shi H, Wu B, Wan J, Liu W, Su B. The role of serum intestinal fatty acid binding protein levels and D-lactate levels in the diagnosis of acute intestinal ischemia. Clin Res Hepatol Gastroenterol. 2015;39:373-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 60] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 9. | Chuong AM, Corno L, Beaussier H, Boulay-Coletta I, Millet I, Hodel J, Taourel P, Chatellier G, Zins M. Assessment of Bowel Wall Enhancement for the Diagnosis of Intestinal Ischemia in Patients with Small Bowel Obstruction: Value of Adding Unenhanced CT to Contrast-enhanced CT. Radiology. 2016;280:98-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 10. | Jang KM, Min K, Kim MJ, Koh SH, Jeon EY, Kim IG, Choi D. Diagnostic performance of CT in the detection of intestinal ischemia associated with small-bowel obstruction using maximal attenuation of region of interest. AJR Am J Roentgenol. 2010;194:957-963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 11. | Mosley JG, Shoaib A. Operative versus conservative management of adhesional intestinal obstruction. Br J Surg. 2000;87:362-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 12. | McEntee G, Pender D, Mulvin D, McCullough M, Naeeder S, Farah S, Badurdeen MS, Ferraro V, Cham C, Gillham N. Current spectrum of intestinal obstruction. Br J Surg. 1987;74:976-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 100] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Brillantino A, Iacobellis F, Renzi A, Nasti R, Saldamarco L, Grillo M, Romano L, Castriconi M, Cittadini A, De Palma M, Scaglione M, Di Martino N, Grassi R, Paladino F. Diagnostic value of arterial blood gas lactate concentration in the different forms of mesenteric ischemia. Eur J Trauma Emerg Surg. 2018;44:265-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 14. | Montagnana M, Danese E, Lippi G. Biochemical markers of acute intestinal ischemia: Possibilities and limitations. Ann Transl Med. 2018;6:341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 15. | Ambe PC, Kang K, Papadakis M, Zirngibl H. Can the Preoperative Serum Lactate Level Predict the Extent of Bowel Ischemia in Patients Presenting to the Emergency Department with Acute Mesenteric Ischemia? Biomed Res Int. 2017;2017:8038796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 16. | Clair DG, Beach JM. Mesenteric Ischemia. N Engl J Med. 2016;374:959-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 329] [Article Influence: 36.6] [Reference Citation Analysis (1)] |
| 17. | Nuzzo A, Maggiori L, Ronot M, Becq A, Plessier A, Gault N, Joly F, Castier Y, Vilgrain V, Paugam C, Panis Y, Bouhnik Y, Cazals-Hatem D, Corcos O. Predictive Factors of Intestinal Necrosis in Acute Mesenteric Ischemia: Prospective Study from an Intestinal Stroke Center. Am J Gastroenterol. 2017;112:597-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 140] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 18. | Liakakos T, Thomakos N, Fine PM, Dervenis C, Young RL. Peritoneal adhesions: Etiology, pathophysiology, and clinical significance. Recent advances in prevention and management. Dig Surg. 2001;18:260-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 472] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 19. | Liu Z, Cheng S, Gu C, Pei H, Hong X. Effect of Hydrogen-Rich Saline on Postoperative Intra-Abdominal Adhesion Bands Formation in Mice. Med Sci Monit. 2017;23:5363-5373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Barmparas G, Branco BC, Schnüriger B, Lam L, Inaba K, Demetriades D. The incidence and risk factors of post-laparotomy adhesive small bowel obstruction. J Gastrointest Surg. 2010;14:1619-1628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 103] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 21. | Goussous N, Kemp KM, Bannon MP, Kendrick ML, Srvantstyan B, Khasawneh MA, Zielinski MD. Early postoperative small bowel obstruction: Open vs laparoscopic. Am J Surg. 2015;209:385-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 22. | Kang WS, Park YC, Jo YG, Kim JC. Early postoperative small bowel obstruction after laparotomy for trauma: Incidence and risk factors. Ann Surg Treat Res. 2018;94:94-101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 23. | Kendrick ML. Partial small bowel obstruction: Clinical issues and recent technical advances. Abdom Imaging. 2009;34:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 24. | van Oudheusden TR, Aerts BA, de Hingh IH, Luyer MD. Challenges in diagnosing adhesive small bowel obstruction. World J Gastroenterol. 2013;19:7489-7493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 25. | Leone M, Lefrant JY, Martin C, Constantin JM. Acute mesenteric ischemia, procalcitonin, and intensive care unit. Intensive Care Med. 2015;41:1378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 26. | Lappas JC, Reyes BL, Maglinte DD. Abdominal radiography findings in small-bowel obstruction: Relevance to triage for additional diagnostic imaging. AJR Am J Roentgenol. 2001;176:167-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 44] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 27. | Silva AC, Pimenta M, Guimarães LS. Small bowel obstruction: What to look for. Radiographics. 2009;29:423-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 148] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 28. | Geffroy Y, Boulay-Coletta I, Jullès MC, Nakache S, Taourel P, Zins M. Increased unenhanced bowel-wall attenuation at multidetector CT is highly specific of ischemia complicating small-bowel obstruction. Radiology. 2014;270:159-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 88] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 29. | Millet I, Ruyer A, Alili C, Curros Doyon F, Molinari N, Pages E, Zins M, Taourel P. Adhesive small-bowel obstruction: Value of CT in identifying findings associated with the effectiveness of nonsurgical treatment. Radiology. 2014;273:425-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 30. | Millet I, Boutot D, Faget C, Pages-Bouic E, Molinari N, Zins M, Taourel P. Assessment of Strangulation in Adhesive Small Bowel Obstruction on the Basis of Combined CT Findings: Implications for Clinical Care. Radiology. 2017;285:798-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 31. | Bala M, Kashuk J, Moore EE, Kluger Y, Biffl W, Gomes CA, Ben-Ishay O, Rubinstein C, Balogh ZJ, Civil I, Coccolini F, Leppaniemi A, Peitzman A, Ansaloni L, Sugrue M, Sartelli M, Di Saverio S, Fraga GP, Catena F. Acute mesenteric ischemia: Guidelines of the World Society of Emergency Surgery. World J Emerg Surg. 2017;12:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 306] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 32. | Donohoe CL, Reynolds JV. Short bowel syndrome. Surgeon. 2010;8:270-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 33. | Johnson E, Vu L, Matarese LE. Bacteria, Bones, and Stones: Managing Complications of Short Bowel Syndrome. Nutr Clin Pract. 2018;33:454-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 34. | Lauro A, Lacaille F. Short bowel syndrome in children and adults: From rehabilitation to transplantation. Expert Rev Gastroenterol Hepatol. 2019;13:55-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |