Published online Sep 10, 2019. doi: 10.4291/wjgp.v10.i2.17
Peer-review started: May 9, 2019
First decision: June 6, 2019
Revised: June 30, 2019
Accepted: August 21, 2019
Article in press: August 21, 2019
Published online: September 10, 2019
Processing time: 134 Days and 21.1 Hours
The indications of immune checkpoint inhibitors (ICPIs) for cancer treatment have rapidly expanded, and their use is increasing in clinical settings worldwide. Despite the considerable clinical benefits of ICPIs, frequent immune-related adverse events (irAEs) have become nonnegligible concerns. Among irAEs, ICPI-induced colitis/diarrhea is frequent and recognized not only by oncologists but also by gastroenterologists or endoscopists. The endoscopic findings show similarity to those of inflammatory bowel disease to a certain extent, particularly ulcerative colitis, but do not seem to be identical. The pathological findings of ICPI-induced colitis may vary among drug classes. They show acute or chronic inflammation, but it may depend on the time of colitis suggested by colonoscopy, including biopsy or treatment intervention. In the case of chronic inflammation determined by biopsy, the endoscopy findings may overlap with those of inflammatory bowel disease. Here, we provide a comprehensive review of ICPI-induced colitis based on clinical, endoscopic and pathologic findings.
Core tip: Immune checkpoint inhibitor (ICPI)-induced colitis/diarrhea is frequent and recognized not only by oncologists but also by gastroenterologists or endoscopists. The endoscopic findings resemble those of inflammatory bowel disease to a certain extent, particularly ulcerative colitis, but are not identical. The pathological findings of ICPI-induced colitis may vary among drug classes. The findings show acute or chronic phases but may depend on the diagnostic timing or treatment intervention. Colonoscopy with biopsy is necessary to confirm ICPI-induced colitis, and early evaluation may avoid exacerbating or prolonging colitis due to treatment resistance.
- Citation: Nishida T, Iijima H, Adachi S. Immune checkpoint inhibitor-induced diarrhea/colitis: Endoscopic and pathologic findings. World J Gastrointest Pathophysiol 2019; 10(2): 17-28
- URL: https://www.wjgnet.com/2150-5330/full/v10/i2/17.htm
- DOI: https://dx.doi.org/10.4291/wjgp.v10.i2.17
In 1992, Ishida et al[1] identified a protein on activated T lymphocytes called programmed cell death protein 1 (PD-1), a key player in tumor immunology. In 1996, Leach et al[2] identified a protein called cytotoxic T-lymphocyte antigen-4 (CTLA-4), another major blocking pathway for the human immune system that was similar to PD-1. Since then, their discoveries have led to the development of immune checkpoint inhibitors (ICPIs) as anticancer drugs and have brought about a major revolution in cancer treatment strategy. Both CTLA-4 and PD-1 deliver negative signals to T-cell-mediated excessive immune activation, known as checkpoints, and ICPIs disrupt the signals mediated by CTLA-4 and PD-1 to prevent T cells from blocking pathways. By inhibiting immune checkpoints, activation of T cells is maintained, thereby helping cancer cells to induce cytotoxic T cell-mediated death. In 2018, Professor Honjo and Professor Allison won the Nobel prize in Physiology or Medicine for their work.
Presently, there are six ICPIs available and approved by the United States Food and Drug Administration for different cancers. Despite the significant clinical benefits of ICPIs, frequent immune-related adverse events (irAEs) in the skin, endocrine organs, gastrointestinal (GI) tract, liver, and lungs and in the musculoskeletal, renal, nervous, hematologic, cardiovascular, and ocular systems have become nonnegligible concerns. Most irAEs have a delayed onset and prolonged duration compared with those from chemotherapy[3]. The incidence of irAEs appears to be similar across tumor types[4]. Among irAEs, ICPI-induced colitis/diarrhea is frequent and recognized not only by oncologists but also by gastroenterologists or endoscopists. In this review, we provide a comprehensive review of ICPI-induced colitis based on clinical, endoscopic and pathologic findings.
ICPI-induced diarrhea occurs after an average of three infusions[5], although it can occur immediately after the first infusion. Recent reports suggest that the onset timing of ICPI-induced diarrhea/colitis may differ by ICPI type. ICPI-induced diarrhea/colitis induced by ipilimumab (anti-CTLA-4) usually occurs 6 to 7 wk after the initiation of ipilimumab[6]. The median time from last the ipilimumab treatment to diarrhea onset is 11-14 d (range 0-59 d)[7,8]. On the other hand, Wang et al[9] reported that 3.2% of patients (30/973) receiving anti-PD-1 developed ICPI-induced colitis at a median of 25.4 wk (range 0.6-120 wk). ICPI-induced diarrhea/colitis induced by anti-PD-1 seems to occur later than that induced by anti-CTLA-4. After the combined use of ipilimumab and nivolumab or pembrolizumab, 24.4% of patients (79/324) developed ICPI-induced diarrhea/colitis significantly earlier, at a median of 7.2 wk (range 0.7-51 wk)[9]. Because the ranges of its onset timing are widely distributed, it is difficult to predict the development of ICPI-induced diarrhea/colitis. In addition, it may be influenced by other drugs, including NSAIDs, antibiotics, or previous anticancer drugs. Moreover, it seems difficult to predict the development of colitis before patients have symptoms[10]. We should keep in mind that ICPI-related colitis can occur at any point, even after discontinuation of ICPIs.
Geukes Foppen et al[11] reported total colonoscopy in 62 of 92 patients (67%) suspected of ICPI-induced colitis. Of these patients, 68% showed pancolitis (> 3 affected segments), and the ascending colon had more severe colitis than the descending colon. In cases where a total colonoscopy was not performed, patients with colitis in the ascending colon can be underestimated by sigmoidoscopy alone. Abdominal computed tomography (CT) findings may be useful not only to evaluate perforation, obstruction, and toxic megacolon but also to evaluate inflamed lesions due to ICPIs. The common CT findings of 16 patients treated with ipilimumab showed that 75% of patients had diffuse colitis patterns, and 25% had segmental colitis[12]. CT was not sufficient to diagnose colitis when using endoscopic evaluation as the gold standard because it has a high false-negative rate and low sensitivity[13]. In contrast, Garcia-Neuer et al[14] reported that CT was useful for predicting ICPI-induced colitis with a positive predictive value of 96% and a negative likelihood ratio of 0.2 in 34 diarrhea patients who underwent both CT and colonoscopy with biopsy. Early sigmoidoscopy without bowel preparation has merit to assess ICPI-induced colitis because it can be performed more easily and earlier than total colonoscopy. Therefore, the combined use of sigmoidoscopy and CT may be useful to evaluate ICPI-induced colitis at an earlier stage.
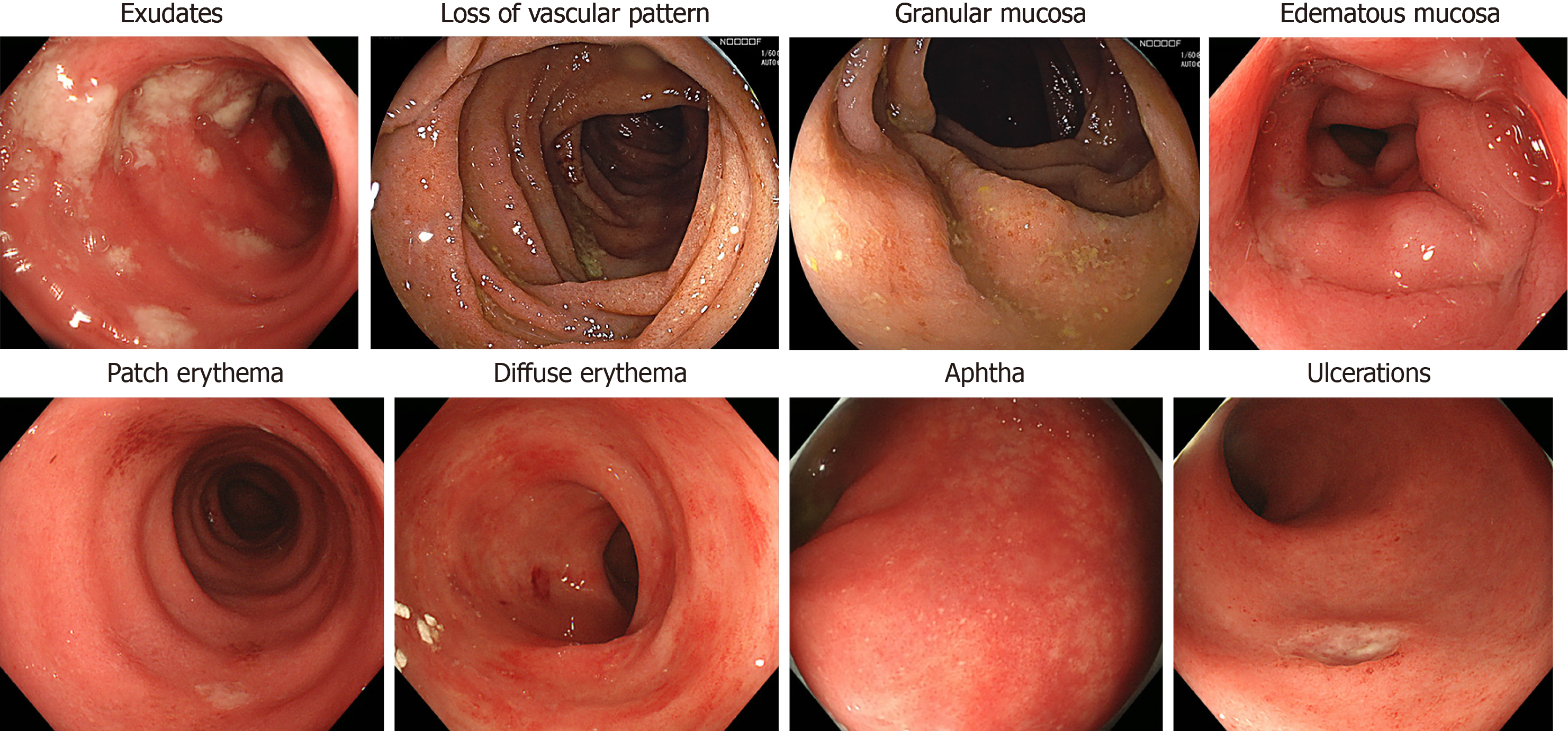
There are several reports about the endoscopic findings of ICPI-induced colitis. Wang et al[13] observed that endoscopic inflammatory findings were found in more than 80% of patients with ICPI-induced diarrhea/colitis. Common endoscopic inflammation findings are reported as exudates, loss of vascular pattern, granular or edematous mucosa, patch or diffuse erythema, aphtha and ulcerations (Figure 1)[15,16]. Most of the inflammatory changes, including pathological changes, are dominantly more diffuse than patchy[10], but patchy distribution was endoscopically observed in half of the patients with diarrhea[17]. These endoscopic findings resemble those of inflammatory bowel disease (IBD) to a certain extent, particularly with ulcerative colitis (UC)[16,18], but sometimes look different from a UC-like pattern (Table 1).
| Endoscopic and pathological findings of immune-related diarrhea and colitis | |
| Endoscopic findings | |
| Endoscopic features | (1) Exudates; (2) loss of vascular pattern; (3) granular or edematous mucosa; (4) patch or diffuse erythema; (5) aphtha; (6) ulceration |
| Inflammatory distribution | (1) Diffuse; (2) patchy (dominantly more diffuse than patchy) |
| Risk factors for steroid-refractory colitis | (1) Extensively inflamed area (e.g., pancolitis); (2) deeper ulceration |
| Pathological findings | |
| Anti-CTLA-4 associated colitis | Like autoimmune colitis: (1) lamina propria expansion due to dense lymphoplasmacytic infiltrate; (2) increased intraepithelial lymphocytosis; (3) apoptosis in the crypts; (4) neutrophilic cryptitis and crypt abscess; (5) occasional prominent eosinophilia in the lamina propria; (6) the lack of findings of basal plasmacytosis, crypt distortion, or granulomas |
| Anti-PD1/anti-PDL1-associated colitis | (1) Expansion of lamina propria by lymphoplasmacytic infiltrate; (2) the increase in intraepithelial neutrophils and neutrophilic crypt abscess; (3) crypt distortion; (4) increased crypt cell apoptosis |
Wang et al[13] reported in 53 patients with diarrhea, clinical symptoms did not always correlate with other endoscopic findings except for the presence of ulceration, which had a strong relationship with higher colitis. Similarly, another retrospective study showed that there was no significant correlation between diarrhea/colitis symptoms and endoscopic findings in 92 patients who developed diarrhea.
They also reported that pancolitis and the presence of ulceration are indicators for steroid-refractory colitis[11]. Geukes Foppen et al[11] reported that the Mayo score was associated with the presence of ulceration. Abu-Sbeih et al[19] categorized endoscopic findings as low-risk and high-risk for steroid-responsiveness. High-risk findings included either ulcers deeper than 2 mm and/or larger than 1 cm in surface area or endoscopically extensive colitis from the proximal colon to the splenic flexure. These patients require frequent use of infliximab or vedolizumab and more frequent and longer hospital stays than non-high-risk patients[19]. They also reported that timely early colonoscopy decreased the duration of steroid treatment[19]. If the colonoscopy shows normal mucosal findings, we are not always able to exclude the presence of ICPI-induced colitis, as cases of isolated ileitis[20] or enteritis without colitis[21] can also occur. We can also rule out microscopic colitis or other infectious diseases such as Clostridioides difficile or cytomegalovirus[7]. Therefore, early colonoscopy with mucosal biopsy from colorectal and ileum-end mucosa is necessary not only to evaluate the severity and distribution of colitis[11] but also to ensure shorter and less intense treatment[19].
The histologic features of ICPI-associated colitis may vary among drug classes, i.e., CTLA-4 inhibitors and PD-1/PDL1 inhibitors. Although they are nonspecific, some findings can be helpful clues to diagnose and speculate about the class of inhibitors. On the other hand, there is significant overlap between ICPI-associated colitis and other types of colitis, making the differential diagnosis difficult.
The histologic findings of CTLA-4-associated colitis are relatively consistent across most studies. The previously reported histologic features of CTLA-4 associated colitis are similar to those of autoimmune colitis[22]. They include lamina propria expansion due to dense lymphoplasmacytic infiltrate, increased intraepithelial lymphocytosis, and apoptosis in the crypts. Neutrophilic cryptitis and crypt abscess are also found. At times, there is prominent eosinophilia in the lamina propria. Although dense lymphoplasmacytic lamina propria expansion is reminiscent of other mimics, the lack of findings of basal plasmacytosis, crypt distortion, or granulomas can help the differentiation.
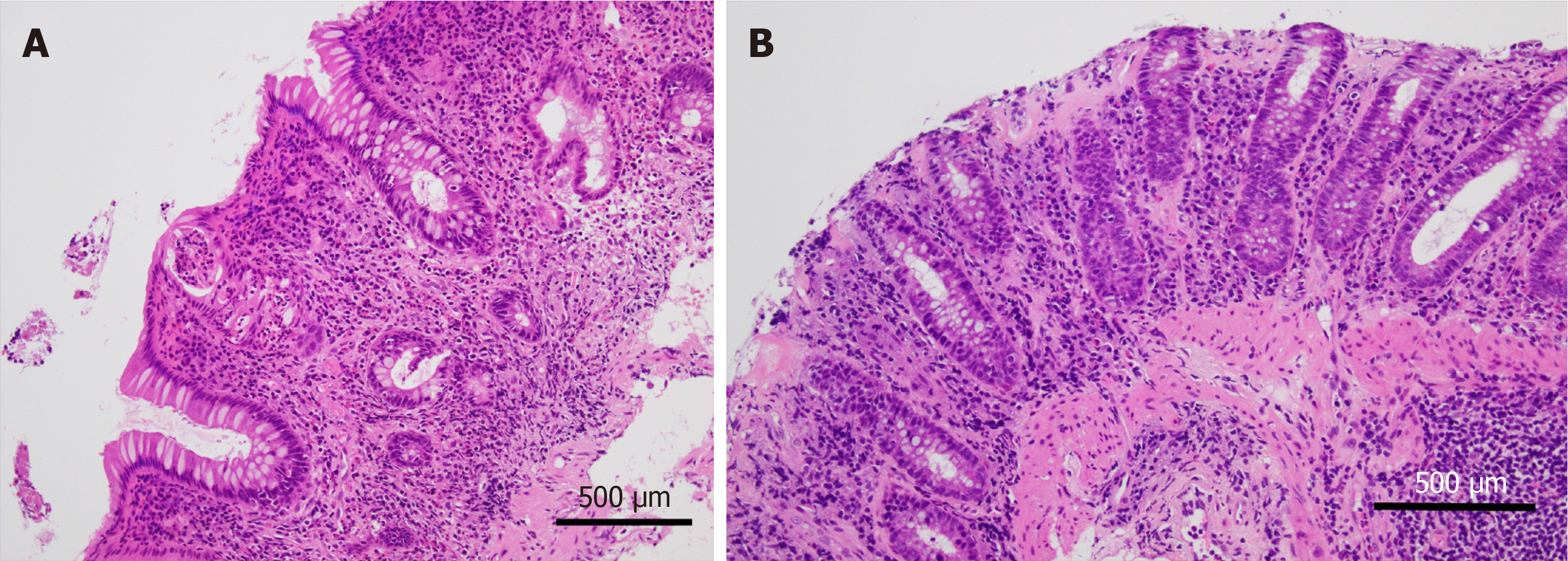
The most common findings of anti-PD1/anti-PDL1-associated colitis are the expansion of the lamina propria by lymphoplasmacytic infiltrate and features of active colitis[23-27]. The latter are characterized by an increase in intraepithelial neutrophils and neutrophilic crypt abscess (Figure 2A). Other findings include crypt distortion, increased crypt cell apoptosis, features of ischemic colitis, and collagenous colitis (Figure 2B). Although, in the study by Gonzalez et al[26], there were no cases with increased intraepithelial lymphocytosis commonly observed in CTLA-4-associated colitis, Chen et al[23] and Bavi et al[27] described features of lymphocytic colitis in a minority of their cases with anti-PD1/anti-PDL1. In the latter studies, a PD-1 inhibitor and CLTA-4 inhibitor were prescribed for their patient population either in combination or sequentially. Therefore, it is unlikely that this finding is related to PD-1 inhibition alone.
As mentioned, the histologic features of ICPI-associated colitis are nonspecific and can mimic other type of colitis, including infectious colitis, IBD, graft versus host disease (GVHD), and other drug-induced colitis. Although infectious colitis typically shows features of active colitis, increased apoptosis and crypt atrophy/dropout are not typical features[28]. ICPI-associated colitis lacks the features of chronicity that characterize IBD[29]. The lamina propria expansion by lymphoplasmacytic infiltrate can discriminate from GVHD although increased crypt apoptosis is the sine qua none of the diagnosis of GVHD[30]. Despite the histopathological differential diagnostic points, clinical correlation and medical history are indispensable for discrimination between ICPI-associated colitis and mimics (Table 1).
IrAEs involving the GI tract range from mild to severe events[31] and are well reported for anti-CTLA4 but less well reported for anti-PD-1 and anti-PD-L1 and for combined anti-CTLA4 plus anti-PD-1. Most clinical trials distinguish diarrhea from colitis even though they overlap in most practical cases. Diarrhea is evaluated based on an increase in stool per day or ostomy output. Colitis is evaluated based on clinical symptoms (abdominal pain, mucus or blood in stool) or diagnostic observations based on radiographic and/or colonoscopy findings. The severity is usually classified based on the Common Terminology Criteria for Adverse Events[32] (Table 2).
| CTCAE Term | Definition | Grade 1 | Grade 2 | Grade 3 | Grade 4 | Grade 5 | CTCAE v5.0 Change |
| Diarrhea | A disorder characterized by an increase in frequency and/or loose or watery bowel movements | Increase of < 4 stools per day over baseline; mild increase in ostomy output compared to baseline | Increase of 4-6 stools per day over baseline; moderate increase in ostomy output compared to baseline; limiting instrumental ADL | Increase of ≥ 7 stools per day over baseline; hospitalization indicated; severe increase in ostomy output compared to baseline; limiting self-care ADL | Life-threatening consequences; urgent intervention indicated | Death | Clarification: Grade 2, 3, Definition |
| Colitis | A disorder characterized by inflammation of the colon | Asymptomatic; clinical or diagnostic observations only; intervention not indicated | Abdominal pain; mucus or blood in stool | Severe abdominal pain; peritoneal signs | Life-threatening consequences; urgent intervention indicated | Death | Addition: Navigational note; Clarification: Grade 3 |
Moderate to severe ICPI-related colitis may lead to severe deterioration in organ function and quality of life and life-threatening events. Diarrhea and colitis occurred in 8% to 22% of patients treated with anti-CTLA4[15]. A recent systemic review reported that 613 fatal ICPI toxic events were fond from 2009 through January 2018 searched by Vigilyze, which included 135 anti–CTLA-4 deaths and 32 combination anti-CTLA-4 plus anti-PD-1 deaths from colitis (27%)[33]. Colonic perforation was reported to occur in 1-5% of melanoma patients treated with ipilimumab (anti-CTLA-4)[7,15,34], and 0.6% of patients treated with ipilimumab died due to ICPI-induced colitis[35].
Anti-CTLA4-related colitis is reportedly associated with mouth ulcers, anal lesions and extraintestinal irAEs[17]. A recent meta-analysis of 34 studies that included 8863 patients in clinical trials revealed that, for anti-CTLA4 alone (ipilimumab), all grades of colitis occurred in 9.1% (95% confidence interval (CI), 6.6%-12.5%) of participants, grade 3/4 colitis occurred in 6.8% (95%CI: 5.3%-8.6%) of participants, and grade 3/4 diarrhea occurred in 7.9% (95%CI: 5.5%-11.4%) of participants. Similarly, for anti-PD-1 alone (nivolumab or pembrolizumab), the rates were 1.4% (95%CI: 1.1%-1.8%), 0.9% (95%CI: 0.7%-1.3%), and 1.3% (95%CI: 1.0%-1.7%), respectively. For anti-PD-L1 alone (atezolizumab), the rates were 1.0% (95%CI: 0.4%-2.2%), 0.6% (95%CI: 0.2%-1.6%), and 0.3% (95%CI: 0.1%-1.1%), respectively[36]. For anti-CTLA4 (Ipilimumab) plus anti-PD-1 (nivolumab), the rates were 13.6% (95%CI: 7.7%-22.9%), 9.4% (95%CI: 4.8%-117.4%), and 9.2% (95%CI: 6.8%-12.3%), respectively. ICPI-induced diarrhea/colitis induced by anti-CTLA-4 can develop more often and more severely than ICPI-induced diarrhea/colitis induced by anti-PD-1. Combined anti-CTLA4 plus anti-PD-1 treatment is also more strongly associated with diarrhea/colitis than single-drug treatment[36]. Ipilimumab is commonly used at either 10 mg/kg or 3 mg/kg. There were similar rates of severe colitis at these doses, but severe diarrhea was more frequent at a dose of 10 mg/kg than at 3 mg/kg[36]. Recently, Marthey et al[17] showed that the use of nonsteroidal anti-inflammatory drugs (NSAIDs) was associated with an increased risk of ICPI-induced colitis induced by CTLA-4 (2/38, 5% vs 11/35, 31%, P = 0.003). Therefore, the use of NSAIDs may affect the incidence of ICPI-induced diarrhea/colitis. Table 3 shows a summary of the incidence of immune-related diarrhea or colitis based on representative clinical trials.
| ICPI | Target | Author | Year | Plus other drugs | n | Cancer type | Any gradediarrhea/colitis, n (%) | Grade 3-5diarrhea/colitis, n (%) |
| Nivolumab | PD-1 | Topalian et al[41] | 2012 | None | 296 | Solid cancer | 33 (11)/ND | 3 (1)/ND |
| Weber et al[42] | 2013 | None ipilimumab-naive | 34 | Melanoma | 13 (38.2)/0 (0) | Not observed1 | ||
| Ipilimumab-refractory | 56 | 11 (19.6)/0 (0) | ||||||
| Weber et al[43] | 2015 | None | 268 | Melanoma2 | 30 (11.2)/ND | 1 (0.4)/ND | ||
| Larkin et al[44] | 2015 | None | 315 | Melanoma | 60 (19.2)/4 (1.3) | 7 (2.2)/2 (0.6) | ||
| Ferris et al[45] | 2016 | None | 236 | SCCHN | 16 (6.8)/0 (0) | 0 (0)/0 (0) | ||
| Kang et al[46] | 2017 | None | 330 | GC/GEJC | 23 (7)/2 (1) | 2 (1)/1 (< 1) | ||
| Pembrolizumab | PD-1 | Hamid et al[47] | 2013 | None | 135 | Melanoma | 27 (20) | 1(1) |
| Garon et al[48] | 2015 | None | 495 | NSCLC | 40 (8.1)/ND | 3 (0.6)/ND | ||
| Ribas et al[49] | 2015 | None | 361 | Melanoma2 | 32 (8.9)/5 (1.4) | 2 (0.6)/2 (0.6) | ||
| Herbst et al[50] | 2016 | None | 690 | NSCLC | 46 (6.7)/6 (0.9) | 2 (0.3)/4 (0.6) | ||
| Ribas et al[51] | 2016 | None | 655 | Melanoma | 115 (18)/11(2) | 6 (1)/7 (1.1) | ||
| Mok et al[52] | 2019 | None | 636 | NSCLC | 34 (5)/7 (1) | 5 (< 1)/4 (< 1) | ||
| Ipilimumab | CTLA-4 | Weber et al[53] | 2008 | None | 88 | Melanoma | ND | 5 (5.6)/4 (4.5) |
| Weber et al[54] | 2009 | None | 57 | Melanoma | 20 (35)/ND | 10 (18)/ND | ||
| budesonide | 58 | 19 (33)/ND | 8 (14)/ND | |||||
| Wolchok et al[55] | 2010 | None | 214 | Melanoma | 58 (27)/ND | 11(5.1)/ND | ||
| Hodi et al[56] | 2010 | None | 131 | Melanoma | 43 (32.8)/10 (7.6) | 7 (5.3)/7 (5.3) | ||
| gp100 | 380 | 146 (38.4)/20 (5.3)3 | 17 (4.5)/12(3.2)3 | |||||
| Robert et al[57] | 2011 | Dacarbazine | 247 | Melanoma | 81 (32.8)/11 (4.5) | 10 (4.0)/5 (2.0) | ||
| Margolin et al[58] | 2012 | None | 72 | Melanoma | 30 (42)/ND | 6 (8.3)/ND | ||
| Kwon et al[59] | 2014 | None | 399 | Prostate cancer | 199 (51)/27 (7) | 64 (16)/18 (5) | ||
| Larkin et al[44] | 2015 | None | 311 | Melanoma | 103 (33.1)/36 (11.6) | 19 (6.1)/27 (8.7) | ||
| Eggermont et al[35] | 2016 | None | 471 | Melanoma | 194 (41.2)/73 (15.5) | 46 (9.8)/39 (8.2) | ||
| Ipilimumab plus nivolumab | CTLA4 and PD1 | Wolchok et al[60] | 2013 | None | 53 | Melanoma | 18 (34.0)/5 (9) | 3 (6)/2 (4) |
| Larkin et al[44] | 2015 | None | 315 | Melanoma | 138 (44.1)/37 (11.8) | 29 (9.3)/24 (7.7) | ||
| Schadendorf et al[61] | 2017 | None | 407 | Melanoma | 30 (7.4)/40 (9.8) | 25 (6.1)/32 (7.9) | ||
| Wolchok et al[62] | 2017 | None | 313 | Melanoma | 142 (45)/40 (13) | 29 (9)/26 (8) | ||
| Hellmann et al[63] | 2017 | None | 77 | NSCLC | 16 (21)/4 (5.2) | 1 (1.3)/3 (3.9) | ||
| Motzer et al[64] | 2018 | None | 547 | Renal cell carcinoma | 145 (27)/ND | 21 (4)/ND | ||
| Durvalumab | PD-L1 | Antonia et al[65] | 2017 | None | 473 | NSCLC | 87 (18.3)/ND | 3 (0.6)/ND |
| Motzer et al[66] | 2018 | None | 475 | NSCLC | 88 (18.5)/ND | 3 (0.6)/ND | ||
| Loibl et al[67] | 2019 | None | 92 | Breast cancer | 26 (28.3)/ND | 3 (3.3)/ND | ||
| Atezolizumab | PD-L1 | Herbst et al[68] | 2014 | None | 277 | Solid tumors or hematological malignancies | 29 (10.5)/ND | 0 (0)/ND |
| Rosenberg et al[69] | 2016 | None | 311 | Urothelial carcinoma | 24 (8)/3 (1) | 1 (0.3)/2 (1) | ||
| Fehrenbacher et al[70] | 2016 | None | 142 | NSCLC | ND | ND/2 (1) | ||
| Socinski et al[71] | 2018 | ABCP | 393 | NSCLC | 70 (17.8) | 11 (2.8) | ||
| Avelumab | PD-L1 | Chung et al[72] | 2019 | None | 150 | GC/GEJC | ND/2 (1.3) | ND/1 (0.7)4 |
| Barlesi et al[73] | 2019 | None | 396 | NSCLC | 24 (6)/ND | 0 (0)/ND |
In the case of grade 1 diarrhea/colitis, antidiarrheal drugs and/or oral hydration with electrolyte substitution can be initiated. In cases of persistent or grade 2 or higher diarrhea or rectal bleeding, it is necessary to confirm colitis or to rule out GI infection by testing for stool leukocytes, stool cultures, IBD, or tumor-related GI symptoms. In particular, Clostridioides difficile toxin and/or antigen test, cytomegalovirus DNA polymerase chain reaction, and tests for stool ova and parasites should be carried out in every patient with diarrhea treated with ICPIs. Sigmoidoscopy or colonoscopy combined with mucosal biopsy needs to be performed to evaluate the presence of colitis and to rule out GI metastasis because it is not uncommon in lung cancer or melanoma. If ICPI-induced colitis is diagnosed, an oral steroid is recommended. In the case of grade 3/4 diarrhea/colitis or persistent symptoms after oral steroids for several days, changing the treatment to intravenous steroids should be considered, and an infusion solution with electrolytes should be given. If patients respond to intravenous steroids within several days, they should be switched to oral steroids and tapered. However, if they fail to respond to steroid infusion, treatment with anti-TNF-α should be considered[15,37]. Recently, a case series reported that vedolizumab was a safer and more theoretic alternative than anti-TNF in patients with steroid-dependent or partially refractory ICPI-induced enterocolitis[38]. In the near future, vedolizumab may be effective and safe because it inhibits the migration of mucosal-associated T lymphocytes without inducing immune suppression and does not show an increased risk of serious infections in patients with UC or Crohn’s disease[39,40].
The combination of endoscopic and pathological findings may help diagnose ICPI-induced colitis as well as exclude infectious colitis, including Clostridioides difficile or cytomegalovirus, ischemic colitis, other drug-induced colitis, or segmental diverticular colitis. However, there are no specific findings because the endoscopic and pathological findings can depend on the time of colitis proven by biopsy or treatment intervention. In cases of persistent or grade 2 or higher diarrhea or rectal bleeding, colonoscopy evaluation is necessary to confirm ICPI-induced colitis and to rule out other diseases. Early evaluation and intervention may avoid exacerbating or prolonging colitis.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Japan
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Abd Elhamid SM, Morini S, Yang ZH S-Editor: Yan JP L-Editor: A E-Editor: Li X
| 1. | Ishida Y, Agata Y, Shibahara K, Honjo T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992;11:3887-3895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1899] [Cited by in RCA: 2201] [Article Influence: 66.7] [Reference Citation Analysis (0)] |
| 2. | Leach DR, Krummel MF, Allison JP. Enhancement of antitumor immunity by CTLA-4 blockade. Science. 1996;271:1734-1736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2582] [Cited by in RCA: 2811] [Article Influence: 96.9] [Reference Citation Analysis (0)] |
| 3. | Matsubara T, Nishida T, Higaki Y, Tomita R, Shimakoshi H, Shimoda A, Osugi N, Sugimoto A, Takahashi K, Nakamatsu D, Mukai K, Yamamoto M, Fukui K, Adachi S, Inada M. Nivolumab Induces Sustained Liver Injury in a Patient with Malignant Melanoma. Intern Med. 2018;57:1789-1792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 4. | Maughan BL, Bailey E, Gill DM, Agarwal N. Incidence of Immune-Related Adverse Events with Program Death Receptor-1- and Program Death Receptor-1 Ligand-Directed Therapies in Genitourinary Cancers. Front Oncol. 2017;7:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 5. | Bertrand A, Kostine M, Barnetche T, Truchetet ME, Schaeverbeke T. Immune related adverse events associated with anti-CTLA-4 antibodies: Systematic review and meta-analysis. BMC Med. 2015;13:211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 511] [Cited by in RCA: 520] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 6. | Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30:2691-2697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1058] [Cited by in RCA: 1107] [Article Influence: 85.2] [Reference Citation Analysis (0)] |
| 7. | Beck KE, Blansfield JA, Tran KQ, Feldman AL, Hughes MS, Royal RE, Kammula US, Topalian SL, Sherry RM, Kleiner D, Quezado M, Lowy I, Yellin M, Rosenberg SA, Yang JC. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J Clin Oncol. 2006;24:2283-2289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 700] [Cited by in RCA: 647] [Article Influence: 34.1] [Reference Citation Analysis (0)] |
| 8. | Lord JD, Hackman RC, Moklebust A, Thompson JA, Higano CS, Chielens D, Steinbach G, McDonald GB. Refractory colitis following anti-CTLA4 antibody therapy: Analysis of mucosal FOXP3+ T cells. Dig Dis Sci. 2010;55:1396-1405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 82] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 9. | Wang DY, Mooradian MJ, Kim D, Shah NJ, Fenton SE, Conry RM, Mehta R, Silk AW, Zhou A, Compton ML, Al-Rohil RN, Lee S, Voorhees AL, Ha L, McKee S, Norrell JT, Mehnert J, Puzanov I, Sosman JA, Chandra S, Gibney GT, Rapisuwon S, Eroglu Z, Sullivan R, Johnson DB. Clinical characterization of colitis arising from anti-PD-1 based therapy. Oncoimmunology. 2018;8:e1524695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 10. | Berman D, Parker SM, Siegel J, Chasalow SD, Weber J, Galbraith S, Targan SR, Wang HL. Blockade of cytotoxic T-lymphocyte antigen-4 by ipilimumab results in dysregulation of gastrointestinal immunity in patients with advanced melanoma. Cancer Immun. 2010;10:11. [PubMed] |
| 11. | Geukes Foppen MH, Rozeman EA, van Wilpe S, Postma C, Snaebjornsson P, van Thienen JV, van Leerdam ME, van den Heuvel M, Blank CU, van Dieren J, Haanen JBAG. Immune checkpoint inhibition-related colitis: Symptoms, endoscopic features, histology and response to management. ESMO Open. 2018;3:e000278. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 207] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 12. | Kim KW, Ramaiya NH, Krajewski KM, Shinagare AB, Howard SA, Jagannathan JP, Ibrahim N. Ipilimumab-associated colitis: CT findings. AJR Am J Roentgenol. 2013;200:W468-W474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 116] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 13. | Wang Y, Abu-Sbeih H, Mao E, Ali N, Qiao W, Trinh VA, Zobniw C, Johnson DH, Samdani R, Lum P, Shuttlesworth G, Blechacz B, Bresalier R, Miller E, Thirumurthi S, Richards D, Raju G, Stroehlein J, Diab A. Endoscopic and Histologic Features of Immune Checkpoint Inhibitor-Related Colitis. Inflamm Bowel Dis. 2018;24:1695-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 180] [Article Influence: 25.7] [Reference Citation Analysis (4)] |
| 14. | Garcia-Neuer M, Marmarelis ME, Jangi SR, Luke JJ, Ibrahim N, Davis M, Weinberg J, Donahue H, Bailey N, Hodi FS, Buchbinder EL, Ott PA. Diagnostic Comparison of CT Scans and Colonoscopy for Immune-Related Colitis in Ipilimumab-Treated Advanced Melanoma Patients. Cancer Immunol Res. 2017;5:286-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 15. | Gupta A, De Felice KM, Loftus EV, Khanna S. Systematic review: Colitis associated with anti-CTLA-4 therapy. Aliment Pharmacol Ther. 2015;42:406-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 202] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 16. | Kubo K, Kato M, Mabe K. Nivolumab-Associated Colitis Mimicking Ulcerative Colitis. Clin Gastroenterol Hepatol. 2017;15:A35-A36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 17. | Marthey L, Mateus C, Mussini C, Nachury M, Nancey S, Grange F, Zallot C, Peyrin-Biroulet L, Rahier JF, Bourdier de Beauregard M, Mortier L, Coutzac C, Soularue E, Lanoy E, Kapel N, Planchard D, Chaput N, Robert C, Carbonnel F. Cancer Immunotherapy with Anti-CTLA-4 Monoclonal Antibodies Induces an Inflammatory Bowel Disease. J Crohns Colitis. 2016;10:395-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 267] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 18. | Yamauchi R, Araki T, Mitsuyama K, Tokito T, Ishii H, Yoshioka S, Kuwaki K, Mori A, Yoshimura T, Tsuruta O, Torimura T. The characteristics of nivolumab-induced colitis: An evaluation of three cases and a literature review. BMC Gastroenterol. 2018;18:135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 44] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 19. | Abu-Sbeih H, Ali FS, Luo W, Qiao W, Raju GS, Wang Y. Importance of endoscopic and histological evaluation in the management of immune checkpoint inhibitor-induced colitis. J Immunother Cancer. 2018;6:95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 155] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 20. | Venditti O, De Lisi D, Caricato M, Caputo D, Capolupo GT, Taffon C, Pagliara E, Battisi S, Frezza AM, Onetti Muda A, Tonini G, Santini D. Ipilimumab and immune-mediated adverse events: A case report of anti-CTLA4 induced ileitis. BMC Cancer. 2015;15:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Messmer M, Upreti S, Tarabishy Y, Mazumder N, Chowdhury R, Yarchoan M, Holdhoff M. Ipilimumab-Induced Enteritis without Colitis: A New Challenge. Case Rep Oncol. 2016;9:705-713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 22. | Oble DA, Mino-Kenudson M, Goldsmith J, Hodi FS, Seliem RM, Dranoff G, Mihm M, Hasserjian R, Lauwers GY. Alpha-CTLA-4 mAb-associated panenteritis: A histologic and immunohistochemical analysis. Am J Surg Pathol. 2008;32:1130-1137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 90] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 23. | Chen JH, Pezhouh MK, Lauwers GY, Masia R. Histopathologic Features of Colitis Due to Immunotherapy With Anti-PD-1 Antibodies. Am J Surg Pathol. 2017;41:643-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 138] [Article Influence: 17.3] [Reference Citation Analysis (1)] |
| 24. | Baroudjian B, Lourenco N, Pagès C, Chami I, Maillet M, Bertheau P, Bagot M, Gornet JM, Lebbé C, Allez M. Anti-PD1-induced collagenous colitis in a melanoma patient. Melanoma Res. 2016;26:308-311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 25. | Marginean EC. The Ever-Changing Landscape of Drug-Induced Injury of the Lower Gastrointestinal Tract. Arch Pathol Lab Med. 2016;140:748-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 26. | Gonzalez RS, Salaria SN, Bohannon CD, Huber AR, Feely MM, Shi C. PD-1 inhibitor gastroenterocolitis: Case series and appraisal of 'immunomodulatory gastroenterocolitis'. Histopathology. 2017;70:558-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 152] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 27. | Bavi P, Butler M, Serra S, Chetty R. Immune modulator-induced changes in the gastrointestinal tract. Histopathology. 2017;71:494-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Turner K. Fenoglio-Preiser’s Gastrointest Pathology. Infectious Colitis. 4th ed. Philadelphia: Wolters Kluwer 2017; 754-796. |
| 29. | Gordon I. Fenoglio-Preiser’s Gastrointest Pathology. Philadelphia: Wolters Kluwer 2017; 814-816. |
| 30. | Turner K. Fenoglio-Preiser’s Gastrointest Pathology. 4th ed. Philadelphia: Wolters Kluwer 2017; 814-816. |
| 31. | Michot JM, Bigenwald C, Champiat S, Collins M, Carbonnel F, Postel-Vinay S, Berdelou A, Varga A, Bahleda R, Hollebecque A, Massard C, Fuerea A, Ribrag V, Gazzah A, Armand JP, Amellal N, Angevin E, Noel N, Boutros C, Mateus C, Robert C, Soria JC, Marabelle A, Lambotte O. Immune-related adverse events with immune checkpoint blockade: A comprehensive review. Eur J Cancer. 2016;54:139-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1248] [Cited by in RCA: 1571] [Article Influence: 174.6] [Reference Citation Analysis (0)] |
| 32. | NCI Common Terminology Criteria for Adverse Events (CTCAE) data files [cited 3 April 2019]. Available from: https://evs.nci.nih.gov/ftp1/CTCAE/About.html. |
| 33. | Wang DY, Salem JE, Cohen JV, Chandra S, Menzer C, Ye F, Zhao S, Das S, Beckermann KE, Ha L, Rathmell WK, Ancell KK, Balko JM, Bowman C, Davis EJ, Chism DD, Horn L, Long GV, Carlino MS, Lebrun-Vignes B, Eroglu Z, Hassel JC, Menzies AM, Sosman JA, Sullivan RJ, Moslehi JJ, Johnson DB. Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol. 2018;4:1721-1728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1108] [Cited by in RCA: 1849] [Article Influence: 264.1] [Reference Citation Analysis (0)] |
| 34. | Yervoy: Highlights of Presecribing Information [cited 27 March 2019]. 2015. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2015/125377s073lbl.pdf. |
| 35. | Eggermont AM, Chiarion-Sileni V, Grob JJ, Dummer R, Wolchok JD, Schmidt H, Hamid O, Robert C, Ascierto PA, Richards JM, Lebbé C, Ferraresi V, Smylie M, Weber JS, Maio M, Bastholt L, Mortier L, Thomas L, Tahir S, Hauschild A, Hassel JC, Hodi FS, Taitt C, de Pril V, de Schaetzen G, Suciu S, Testori A. Prolonged Survival in Stage III Melanoma with Ipilimumab Adjuvant Therapy. N Engl J Med. 2016;375:1845-1855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 964] [Cited by in RCA: 1024] [Article Influence: 113.8] [Reference Citation Analysis (0)] |
| 36. | Wang DY, Ye F, Zhao S, Johnson DB. Incidence of immune checkpoint inhibitor-related colitis in solid tumor patients: A systematic review and meta-analysis. Oncoimmunology. 2017;6:e1344805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 155] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 37. | Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, Jordan K; ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv119-iv142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1607] [Cited by in RCA: 1503] [Article Influence: 187.9] [Reference Citation Analysis (1)] |
| 38. | Bergqvist V, Hertervig E, Gedeon P, Kopljar M, Griph H, Kinhult S, Carneiro A, Marsal J. Vedolizumab treatment for immune checkpoint inhibitor-induced enterocolitis. Cancer Immunol Immunother. 2017;66:581-592. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 149] [Cited by in RCA: 179] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 39. | Sandborn WJ, Feagan BG, Rutgeerts P, Hanauer S, Colombel JF, Sands BE, Lukas M, Fedorak RN, Lee S, Bressler B, Fox I, Rosario M, Sankoh S, Xu J, Stephens K, Milch C, Parikh A; GEMINI 2 Study Group. Vedolizumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2013;369:711-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1416] [Cited by in RCA: 1568] [Article Influence: 130.7] [Reference Citation Analysis (1)] |
| 40. | Feagan BG, Rutgeerts P, Sands BE, Hanauer S, Colombel JF, Sandborn WJ, Van Assche G, Axler J, Kim HJ, Danese S, Fox I, Milch C, Sankoh S, Wyant T, Xu J, Parikh A; GEMINI 1 Study Group. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369:699-710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1576] [Cited by in RCA: 1865] [Article Influence: 155.4] [Reference Citation Analysis (1)] |
| 41. | Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD, Sosman JA, Atkins MB, Leming PD, Spigel DR, Antonia SJ, Horn L, Drake CG, Pardoll DM, Chen L, Sharfman WH, Anders RA, Taube JM, McMiller TL, Xu H, Korman AJ, Jure-Kunkel M, Agrawal S, McDonald D, Kollia GD, Gupta A, Wigginton JM, Sznol M. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443-2454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8900] [Cited by in RCA: 9908] [Article Influence: 762.2] [Reference Citation Analysis (0)] |
| 42. | Weber JS, Kudchadkar RR, Yu B, Gallenstein D, Horak CE, Inzunza HD, Zhao X, Martinez AJ, Wang W, Gibney G, Kroeger J, Eysmans C, Sarnaik AA, Chen YA. Safety, efficacy, and biomarkers of nivolumab with vaccine in ipilimumab-refractory or -naive melanoma. J Clin Oncol. 2013;31:4311-4318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 446] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 43. | Weber JS, D'Angelo SP, Minor D, Hodi FS, Gutzmer R, Neyns B, Hoeller C, Khushalani NI, Miller WH, Lao CD, Linette GP, Thomas L, Lorigan P, Grossmann KF, Hassel JC, Maio M, Sznol M, Ascierto PA, Mohr P, Chmielowski B, Bryce A, Svane IM, Grob JJ, Krackhardt AM, Horak C, Lambert A, Yang AS, Larkin J. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti-CTLA-4 treatment (CheckMate 037): A randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16:375-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1853] [Cited by in RCA: 2106] [Article Influence: 210.6] [Reference Citation Analysis (0)] |
| 44. | Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M, Rutkowski P, Ferrucci PF, Hill A, Wagstaff J, Carlino MS, Haanen JB, Maio M, Marquez-Rodas I, McArthur GA, Ascierto PA, Long GV, Callahan MK, Postow MA, Grossmann K, Sznol M, Dreno B, Bastholt L, Yang A, Rollin LM, Horak C, Hodi FS, Wolchok JD. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N Engl J Med. 2015;373:23-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6070] [Cited by in RCA: 6207] [Article Influence: 620.7] [Reference Citation Analysis (0)] |
| 45. | Ferris RL, Blumenschein G, Fayette J, Guigay J, Colevas AD, Licitra L, Harrington K, Kasper S, Vokes EE, Even C, Worden F, Saba NF, Iglesias Docampo LC, Haddad R, Rordorf T, Kiyota N, Tahara M, Monga M, Lynch M, Geese WJ, Kopit J, Shaw JW, Gillison ML. Nivolumab for Recurrent Squamous-Cell Carcinoma of the Head and Neck. N Engl J Med. 2016;375:1856-1867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3044] [Cited by in RCA: 3695] [Article Influence: 410.6] [Reference Citation Analysis (0)] |
| 46. | Kang YK, Boku N, Satoh T, Ryu MH, Chao Y, Kato K, Chung HC, Chen JS, Muro K, Kang WK, Yeh KH, Yoshikawa T, Oh SC, Bai LY, Tamura T, Lee KW, Hamamoto Y, Kim JG, Chin K, Oh DY, Minashi K, Cho JY, Tsuda M, Chen LT. Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390:2461-2471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1283] [Cited by in RCA: 1714] [Article Influence: 214.3] [Reference Citation Analysis (0)] |
| 47. | Hamid O, Robert C, Daud A, Hodi FS, Hwu WJ, Kefford R, Wolchok JD, Hersey P, Joseph RW, Weber JS, Dronca R, Gangadhar TC, Patnaik A, Zarour H, Joshua AM, Gergich K, Elassaiss-Schaap J, Algazi A, Mateus C, Boasberg P, Tumeh PC, Chmielowski B, Ebbinghaus SW, Li XN, Kang SP, Ribas A. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2712] [Cited by in RCA: 2725] [Article Influence: 227.1] [Reference Citation Analysis (0)] |
| 48. | Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L, Carcereny E, Ahn MJ, Felip E, Lee JS, Hellmann MD, Hamid O, Goldman JW, Soria JC, Dolled-Filhart M, Rutledge RZ, Zhang J, Lunceford JK, Rangwala R, Lubiniecki GM, Roach C, Emancipator K, Gandhi L; KEYNOTE-001 Investigators. Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med. 2015;372:2018-2028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4095] [Cited by in RCA: 4860] [Article Influence: 486.0] [Reference Citation Analysis (0)] |
| 49. | Ribas A, Puzanov I, Dummer R, Schadendorf D, Hamid O, Robert C, Hodi FS, Schachter J, Pavlick AC, Lewis KD, Cranmer LD, Blank CU, O'Day SJ, Ascierto PA, Salama AK, Margolin KA, Loquai C, Eigentler TK, Gangadhar TC, Carlino MS, Agarwala SS, Moschos SJ, Sosman JA, Goldinger SM, Shapira-Frommer R, Gonzalez R, Kirkwood JM, Wolchok JD, Eggermont A, Li XN, Zhou W, Zernhelt AM, Lis J, Ebbinghaus S, Kang SP, Daud A. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): A randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16:908-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1137] [Cited by in RCA: 1268] [Article Influence: 126.8] [Reference Citation Analysis (0)] |
| 50. | Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, Majem M, Fidler MJ, de Castro G, Garrido M, Lubiniecki GM, Shentu Y, Im E, Dolled-Filhart M, Garon EB. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet. 2016;387:1540-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4116] [Cited by in RCA: 5052] [Article Influence: 561.3] [Reference Citation Analysis (0)] |
| 51. | Ribas A, Hamid O, Daud A, Hodi FS, Wolchok JD, Kefford R, Joshua AM, Patnaik A, Hwu WJ, Weber JS, Gangadhar TC, Hersey P, Dronca R, Joseph RW, Zarour H, Chmielowski B, Lawrence DP, Algazi A, Rizvi NA, Hoffner B, Mateus C, Gergich K, Lindia JA, Giannotti M, Li XN, Ebbinghaus S, Kang SP, Robert C. Association of Pembrolizumab With Tumor Response and Survival Among Patients With Advanced Melanoma. JAMA. 2016;315:1600-1609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 722] [Cited by in RCA: 829] [Article Influence: 92.1] [Reference Citation Analysis (0)] |
| 52. | Mok TSK, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, Castro G, Srimuninnimit V, Laktionov KK, Bondarenko I, Kubota K, Lubiniecki GM, Zhang J, Kush D, Lopes G; KEYNOTE-042 Investigators. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised, open-label, controlled, phase 3 trial. Lancet. 2019;393:1819-1830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1597] [Cited by in RCA: 2410] [Article Influence: 401.7] [Reference Citation Analysis (0)] |
| 53. | Weber JS, O'Day S, Urba W, Powderly J, Nichol G, Yellin M, Snively J, Hersh E. Phase I/II study of ipilimumab for patients with metastatic melanoma. J Clin Oncol. 2008;26:5950-5956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 362] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 54. | Weber J, Thompson JA, Hamid O, Minor D, Amin A, Ron I, Ridolfi R, Assi H, Maraveyas A, Berman D, Siegel J, O'Day SJ. A randomized, double-blind, placebo-controlled, phase II study comparing the tolerability and efficacy of ipilimumab administered with or without prophylactic budesonide in patients with unresectable stage III or IV melanoma. Clin Cancer Res. 2009;15:5591-5598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 444] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 55. | Wolchok JD, Neyns B, Linette G, Negrier S, Lutzky J, Thomas L, Waterfield W, Schadendorf D, Smylie M, Guthrie T, Grob JJ, Chesney J, Chin K, Chen K, Hoos A, O'Day SJ, Lebbé C. Ipilimumab monotherapy in patients with pretreated advanced melanoma: A randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 2010;11:155-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 862] [Cited by in RCA: 913] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 56. | Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbé C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10799] [Cited by in RCA: 11784] [Article Influence: 785.6] [Reference Citation Analysis (0)] |
| 57. | Robert C, Thomas L, Bondarenko I, O'Day S, Weber J, Garbe C, Lebbe C, Baurain JF, Testori A, Grob JJ, Davidson N, Richards J, Maio M, Hauschild A, Miller WH, Gascon P, Lotem M, Harmankaya K, Ibrahim R, Francis S, Chen TT, Humphrey R, Hoos A, Wolchok JD. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011;364:2517-2526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3331] [Cited by in RCA: 3409] [Article Influence: 243.5] [Reference Citation Analysis (0)] |
| 58. | Margolin K, Ernstoff MS, Hamid O, Lawrence D, McDermott D, Puzanov I, Wolchok JD, Clark JI, Sznol M, Logan TF, Richards J, Michener T, Balogh A, Heller KN, Hodi FS. Ipilimumab in patients with melanoma and brain metastases: An open-label, phase 2 trial. Lancet Oncol. 2012;13:459-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 797] [Cited by in RCA: 851] [Article Influence: 65.5] [Reference Citation Analysis (0)] |
| 59. | Kwon ED, Drake CG, Scher HI, Fizazi K, Bossi A, van den Eertwegh AJ, Krainer M, Houede N, Santos R, Mahammedi H, Ng S, Maio M, Franke FA, Sundar S, Agarwal N, Bergman AM, Ciuleanu TE, Korbenfeld E, Sengeløv L, Hansen S, Logothetis C, Beer TM, McHenry MB, Gagnier P, Liu D, Gerritsen WR; CA184-043 Investigators. Ipilimumab versus placebo after radiotherapy in patients with metastatic castration-resistant prostate cancer that had progressed after docetaxel chemotherapy (CA184-043): A multicentre, randomised, double-blind, phase 3 trial. Lancet Oncol. 2014;15:700-712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1157] [Cited by in RCA: 1188] [Article Influence: 108.0] [Reference Citation Analysis (0)] |
| 60. | Wolchok JD, Kluger H, Callahan MK, Postow MA, Rizvi NA, Lesokhin AM, Segal NH, Ariyan CE, Gordon RA, Reed K, Burke MM, Caldwell A, Kronenberg SA, Agunwamba BU, Zhang X, Lowy I, Inzunza HD, Feely W, Horak CE, Hong Q, Korman AJ, Wigginton JM, Gupta A, Sznol M. Nivolumab plus ipilimumab in advanced melanoma. N Engl J Med. 2013;369:122-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3192] [Cited by in RCA: 3321] [Article Influence: 276.8] [Reference Citation Analysis (0)] |
| 61. | Schadendorf D, Wolchok JD, Hodi FS, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob JJ, Cowey CL, Lao CD, Chesney J, Robert C, Grossmann K, McDermott D, Walker D, Bhore R, Larkin J, Postow MA. Efficacy and Safety Outcomes in Patients With Advanced Melanoma Who Discontinued Treatment With Nivolumab and Ipilimumab Because of Adverse Events: A Pooled Analysis of Randomized Phase II and III Trials. J Clin Oncol. 2017;35:3807-3814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 349] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 62. | Wolchok JD, Chiarion-Sileni V, Gonzalez R, Rutkowski P, Grob JJ, Cowey CL, Lao CD, Wagstaff J, Schadendorf D, Ferrucci PF, Smylie M, Dummer R, Hill A, Hogg D, Haanen J, Carlino MS, Bechter O, Maio M, Marquez-Rodas I, Guidoboni M, McArthur G, Lebbé C, Ascierto PA, Long GV, Cebon J, Sosman J, Postow MA, Callahan MK, Walker D, Rollin L, Bhore R, Hodi FS, Larkin J. Overall Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N Engl J Med. 2017;377:1345-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2362] [Cited by in RCA: 2779] [Article Influence: 347.4] [Reference Citation Analysis (0)] |
| 63. | Hellmann MD, Rizvi NA, Goldman JW, Gettinger SN, Borghaei H, Brahmer JR, Ready NE, Gerber DE, Chow LQ, Juergens RA, Shepherd FA, Laurie SA, Geese WJ, Agrawal S, Young TC, Li X, Antonia SJ. Nivolumab plus ipilimumab as first-line treatment for advanced non-small-cell lung cancer (CheckMate 012): Results of an open-label, phase 1, multicohort study. Lancet Oncol. 2017;18:31-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 701] [Cited by in RCA: 775] [Article Influence: 96.9] [Reference Citation Analysis (0)] |
| 64. | Motzer RJ, Tannir NM, McDermott DF, Arén Frontera O, Melichar B, Choueiri TK, Plimack ER, Barthélémy P, Porta C, George S, Powles T, Donskov F, Neiman V, Kollmannsberger CK, Salman P, Gurney H, Hawkins R, Ravaud A, Grimm MO, Bracarda S, Barrios CH, Tomita Y, Castellano D, Rini BI, Chen AC, Mekan S, McHenry MB, Wind-Rotolo M, Doan J, Sharma P, Hammers HJ, Escudier B; CheckMate 214 Investigators. Nivolumab plus Ipilimumab versus Sunitinib in Advanced Renal-Cell Carcinoma. N Engl J Med. 2018;378:1277-1290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2567] [Cited by in RCA: 3401] [Article Influence: 485.9] [Reference Citation Analysis (0)] |
| 65. | Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, Yokoi T, Chiappori A, Lee KH, de Wit M, Cho BC, Bourhaba M, Quantin X, Tokito T, Mekhail T, Planchard D, Kim YC, Karapetis CS, Hiret S, Ostoros G, Kubota K, Gray JE, Paz-Ares L, de Castro Carpeño J, Wadsworth C, Melillo G, Jiang H, Huang Y, Dennis PA, Özgüroğlu M; PACIFIC Investigators. Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N Engl J Med. 2017;377:1919-1929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2750] [Cited by in RCA: 3241] [Article Influence: 405.1] [Reference Citation Analysis (0)] |
| 66. | Motzer RJ, Penkov K, Haanen J, Rini B, Albiges L, Campbell MT, Venugopal B, Kollmannsberger C, Negrier S, Uemura M, Lee JL, Vasiliev A, Miller WH, Gurney H, Schmidinger M, Larkin J, Atkins MB, Bedke J, Alekseev B, Wang J, Mariani M, Robbins PB, Chudnovsky A, Fowst C, Hariharan S, Huang B, di Pietro A, Choueiri TK. Avelumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med. 2019;380:1103-1115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1857] [Article Influence: 309.5] [Reference Citation Analysis (0)] |
| 67. | Loibl S, Untch M, Burchardi N, Huober J, Sinn BV, Blohmer JU, Grischke EM, Furlanetto J, Tesch H, Hanusch C, Engels K, Rezai M, Jackisch C, Schmitt WD, von Minckwitz G, Thomalla J, Kümmel S, Rautenberg B, Fasching PA, Weber K, Rhiem K, Denkert C, Schneeweiss A. A randomised phase II study investigating durvalumab in addition to an anthracycline taxane-based neoadjuvant therapy in early triple negative breast cancer - clinical results and biomarker analysis of GeparNuevo study. Ann Oncol. 2019;pii:mdz158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 477] [Article Influence: 95.4] [Reference Citation Analysis (0)] |
| 68. | Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN, Kohrt HE, Horn L, Lawrence DP, Rost S, Leabman M, Xiao Y, Mokatrin A, Koeppen H, Hegde PS, Mellman I, Chen DS, Hodi FS. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature. 2014;515:563-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3965] [Cited by in RCA: 4183] [Article Influence: 380.3] [Reference Citation Analysis (0)] |
| 69. | Rosenberg JE, Hoffman-Censits J, Powles T, van der Heijden MS, Balar AV, Necchi A, Dawson N, O'Donnell PH, Balmanoukian A, Loriot Y, Srinivas S, Retz MM, Grivas P, Joseph RW, Galsky MD, Fleming MT, Petrylak DP, Perez-Gracia JL, Burris HA, Castellano D, Canil C, Bellmunt J, Bajorin D, Nickles D, Bourgon R, Frampton GM, Cui N, Mariathasan S, Abidoye O, Fine GD, Dreicer R. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: A single-arm, multicentre, phase 2 trial. Lancet. 2016;387:1909-1920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2517] [Cited by in RCA: 2894] [Article Influence: 321.6] [Reference Citation Analysis (0)] |
| 70. | Fehrenbacher L, Spira A, Ballinger M, Kowanetz M, Vansteenkiste J, Mazieres J, Park K, Smith D, Artal-Cortes A, Lewanski C, Braiteh F, Waterkamp D, He P, Zou W, Chen DS, Yi J, Sandler A, Rittmeyer A; POPLAR Study Group. Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): A multicentre, open-label, phase 2 randomised controlled trial. Lancet. 2016;387:1837-1846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1779] [Cited by in RCA: 2203] [Article Influence: 244.8] [Reference Citation Analysis (0)] |
| 71. | Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, Rodríguez-Abreu D, Moro-Sibilot D, Thomas CA, Barlesi F, Finley G, Kelsch C, Lee A, Coleman S, Deng Y, Shen Y, Kowanetz M, Lopez-Chavez A, Sandler A, Reck M; IMpower150 Study Group. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N Engl J Med. 2018;378:2288-2301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2076] [Cited by in RCA: 2770] [Article Influence: 395.7] [Reference Citation Analysis (0)] |
| 72. | Chung HC, Arkenau HT, Lee J, Rha SY, Oh DY, Wyrwicz L, Kang YK, Lee KW, Infante JR, Lee SS, Kemeny M, Keilholz U, Melichar B, Mita A, Plummer R, Smith D, Gelb AB, Xiong H, Hong J, Chand V, Safran H. Avelumab (anti-PD-L1) as first-line switch-maintenance or second-line therapy in patients with advanced gastric or gastroesophageal junction cancer: Phase 1b results from the JAVELIN Solid Tumor trial. J Immunother Cancer. 2019;7:30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 88] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 73. | Barlesi F, Vansteenkiste J, Spigel D, Ishii H, Garassino M, de Marinis F, Özgüroğlu M, Szczesna A, Polychronis A, Uslu R, Krzakowski M, Lee JS, Calabrò L, Arén Frontera O, Ellers-Lenz B, Bajars M, Ruisi M, Park K. Avelumab versus docetaxel in patients with platinum-treated advanced non-small-cell lung cancer (JAVELIN Lung 200): An open-label, randomised, phase 3 study. Lancet Oncol. 2018;19:1468-1479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 342] [Article Influence: 48.9] [Reference Citation Analysis (0)] |