Peer-review started: October 20, 2014
First decision: December 12, 2014
Revised: March 6, 2015
Accepted: April 1, 2015
Article in press: April 7, 2015
Published online: May 28, 2015
Medical ultrasound imaging with Doppler plays an essential role in the diagnosis of vascular disease. This study intended to review the clinical use of “to-and-fro” waveform at duplex Doppler ultrasonography (DDU) in the diagnosis of pseudoaneurysms in the arterial vessels of upper and lower extremities, abdominal aorta, carotid and vertebral arteries as well as to review our personal experiences of “to-and-fro” waveform at DDU also. After receiving institutional review board approval, an inclusive literature review was carried out in order to review the scientific foundation of “to-and-fro” waveform at DDU and its clinical use in the diagnosis of pseudoaneurysms in various arterial vessels. Articles published in the English language between 2000 and 2013 were evaluated in this review study. Pseudoaneurysms in arterial vessels of the upper and lower extremities, abdominal aorta, carotid and vertebral arteries characterized by an extraluminal pattern of blood flow, which shows variable echogenicity, interval complexity, and “to-and-fro” flow pattern on color Doppler ultrasonography. In these arterial vessels, Duplex ultrasonography can demonstrate the degree of clotting, pseudoaneurysm communication, the blood flow patterns and velocities. Spectral Doppler applied to pseudoaneurysms lumen revealed systolic and diastolic turbulent blood flow with traditional “to-and-fro” waveform in the communicating channel. Accurate diagnosis of pseudoaneurysm by spectral Doppler is based on the documentation of the “to-and-fro” waveform. The size of pseudoaneurysm determines the appropriate treatment approach as surgical or conservative.
Core tip: A review of the clinical use of “to-and-fro” waveform at duplex Doppler ultrasonography (DDU) in the diagnosis of pseudoaneurysms in the arterial vessels of upper and lower extremities, abdominal aorta, carotid and vertebral arteries as well as to review our personal experiences of “to-and-fro” waveform at DDU also.
- Citation: Mahmoud MZ, Al-Saadi M, Abuderman A, Alzimami KS, Alkhorayef M, Almagli B, Sulieman A. "To-and-fro" waveform in the diagnosis of arterial pseudoaneurysms. World J Radiol 2015; 7(5): 89-99
- URL: https://www.wjgnet.com/1949-8470/full/v7/i5/89.htm
- DOI: https://dx.doi.org/10.4329/wjr.v7.i5.89
A pseudoaneurysm is defined as an arterial wall deficiency, which leads to accumulation of oxygenated blood in the nearby extra-luminal region. Therefore arterial blood spread out of the vessel, forming a sac surrounding by soft tissue and compressed thrombus[1]. Consequently, a pseudoaneurysm is formed as a result of fibrin wall formation nearby the swelling[2]. The basic difference of arterial aneurysm and pseudoaneurysm is that the three-layers of the arterial wall don’t bind the later one[3]. Pseudoaneurysms which are the most common in the femoral and radial arteries, often noticed in the groin and forearm after cardiac catheterization. Furthermore, it may observe also after arterial punctures of blood gas analysis, after the placement of indwelling catheter or after direct arterial trauma[2-5].
Ultrasonography (US) has been widely utilized as a noninvasive imaging modality for the investigation of vascular diseases[6-9]. US which is a valuable tool for diagnosis of pseudoaneurysms has been widely utilized as a noninvasive imaging modality for investigation of vascular disease. The main advantage of US imaging is no use of ionizing radiation, cheap, and availability[10,11]. It has been reported that US has 94% and 97% of sensitivity and specificity, respectively in the diagnosis of postcatheterization pseudoaneurysms, but this sensitivity is not enough to diagnose the pseudoaneurysms of the deep visceral arteries[12,13]. The major limitation of US it is an operator dependent imaging technique, has low sensitivity in the evaluation of deep visceral artery pseudoaneurysm, and evaluation of vessels in trauma patient accompanied with hematoma or fracture[14].
In pseudoaneurysms, Analog US images (grayscale) usually illustrate hypoechoic cystic structures nearby a supplying artery[15,16]. Grayscale can be used to evaluate many pseudoaneurysmal findings such as the size, the number of pseudoaneurysm, and its relation to the artery[17]. However, grayscale is not a conclusive evidence in diagnosis pseudoaneurysm because its findings are accompanied by other clinical conditions such as hematomas and cystic masses either simple or complex[1].
Therefore, Doppler US can be used to confirm the diagnosis. In addition to that, blood flow in a cystic structure distinguished by swirling motion pattern “yin-yang sign”. Also, this type of flow can be detected in saccular aneurysm. Therefore, differential diagnosis is essential for pseudoaneurysm. The cornerstone of pseudoaneurysm diagnosis is dependent upon the appearance of the communicating neck between the arterial vessel and pseudoaneurysmal sac with “to-and-fro” waveform at duplex Doppler ultrasonography (DDU). The “to” represents the arterial blood going into the pseudoaneurysmal sac in systolic cycle, while “fro” illustrate blood exiting the sac in diastolic cycle[18].
In this article authors review the clinical use of “to-and-fro” waveform at DDU in the diagnosis of pseudoaneurysms in various arterial vessels, as well as our personal experiences of “to-and-fro” waveform at DDU in the Radiology Department of King Fahad Medical City (KFMC) at Riyadh, Saudi Arabia.
After receiving institutional review board approval, an inclusive literature review was carried out in order to review the scientific foundation of “to-and-fro” waveform at DDU and its clinical use in the diagnosis of pseudoaneurysms in various arterial vessels of the upper and lower extremities, abdominal Aorta, carotid arteries and vertebral arteries.
The ScienceDirect, PubMed, MEDLINE, NCBI and SAGE database were searched in April 2014 for publications containing information about “to-and-fro” sign in the diagnosis of pseudoaneurysms in various arterial vessels in the title of the report. Abstracts resulting from this search were reviewed for relevance to the clinical outcomes from the procedure. Full manuscripts were retrieved and reviewed if they contained information regarding the evaluation of the evidence on the role of “to-and-fro” sign in the diagnosis of pseudoaneurysms and the published clinical literature in this field.
Only those papers published between 2000 and 2013 were included in the outcomes analysis, and this was due to the tremendous development in this medical diagnostic specialty at the beginning of the new millennium so far. Regarding place of the study or journal type in order to include all available sources of experience.
US is a readily available imaging modality, which does not expose the patient to ionizing radiation. Grayscale and color Doppler techniques are utilized, and standardized protocols in an accredited ultrasound laboratory will increase the likelihood of detection of pseudoaneurysm. Grayscale is often initially performed. Linear (high frequency) US probe has acceptable depth penetration and visualization should be used. After a general overview of the area of concern, attention should be given to any anechoic collections or regions of hematoma[19].
Color Doppler is placed in any anechoic collection to detect flow. The flow can be characterized if the scale is properly set to avoid aliasing due to under sampling. Spectral Doppler is then performed if flow is detected to help characterize arterial vs venous flow. Spectral waveforms may be diagnostic of the pseudoaneurysm and help to exclude arteriovenous fistula (AVF). A low wall filter may be initially used to detect slow flow, and the waveform should fill at least two-thirds of the spectral window. In grayscale, a patent pseudoaneurysm appears as an anechoic rounded or ovoid structure. Because other fluid collections, including cysts, seromas, or hematomas can have this appearance, color Doppler imaging is used to confirm the presence of blood flow within the pseudoaneurysm. When present, thrombus in the pseudoaneurysm appears mildly echogenic or hypoechoic without flow; it may be mural or centrally fill a portion of the pseudoaneurysm lumen[1]. Turbulent blood flow is illustrated by interchangeable coloring appearance, either in red or blue color. If large areas of color aliasing are identified in the adjacent tissues, grayscale may help to differentiate pseudoaneurysm from tissue reverberation associated with AVF[19].
The scientific foundation of “to-and-fro” waveform at DDU and its clinical use in the diagnosis of pseudoaneurysms in various arterial vessels, could be discussed on the basis that, the DDU monitoring will present the conventional “to-and-fro” waveform with blood flow of the bidirectional pattern at the neck of pseudoaneurysm. Occasionally, the neck is the only patent portion of the pseudoaneurysm if partial thrombosis has occurred[19].
The incidence of pseudoaneurysms has increased in hospital based practice, due to the large number of invasive procedures performed[20]. Their incidence varies in the literature due to different definitions, methods of interrogation and presence of certain complications[21]. According to medical literature, the incidence of pseudoaneurysms ranges from 0.1% to 6% and up to 0.5% to 9%, depending on the diagnostic or therapeutic procedure performed[22,23].
The frequency of peripheral arteries pseudoaneurysms is much less in the upper extremities than in the lower extremities (less than 2% of all lesions)[24,25]. Aortic pseudoaneurysms are rare, life-threatening sequelae of cardiac surgery[26]. The incidence, risk factors, and natural history of aortic pseudoaneurysm are unknown, because so few cases have been reported[27]. Pseudoaneurysms of the abdominal aorta are rare, especially those found to be mycotic. Abdominal aorta pseudoaneurysms following trauma have been reported fairly often[28]. Common carotid artery pseudoaneurysms are rare and potentially lethal, and adequate treatment is warranted in order to prevent rupture or neurologic sequelae[29]. Vertebral arteries pseudoaneurysm formation after central line placement has been well documented in the literature, with an incidence rate of 0.05% to 2%[30,31].
Unintentional pseudoaneurysm due to surgical intervention for numerous medical procedure (e.g., pseudoaneurysm can be induced in femoral artery during cardiac catheterization). It accounts up to 70%-80% of the incidence of pseudoaneurysms[32].
It had been estimated that 79% of pseudoaneurysms are traumatic in origin of the internal solid organs such as liver, kidneys, pancreas, and gastrointestinal tract of the digestive system[33].
Pseudoaneurysm can be initiated due to blood vessel erosion by an erosive tumor, either benign or malignant. This is most commonly seen in osteochondroma, neurofibromatosis, leukemia, and lymphoma[34]. Yang et al[35] reported that 25% of pseudoaneurysms are caused by neoplastic aneurysms as choriocarcinoma. Kim et al[36] also reported leukemia and lymphoma as a cause of pseudoaneurysm by damaging the arterial vessel wall.
Pseudoaneurysm can be initiated by primary (wall infection) or secondary (adjacent focus) infection of blood vessels. It has been reported that pseudoaneurysms are more frequent in incidence than true aneurysms, this is because the infection can disturb blood vessels wall more easily[37].
Formation of pseudoaneurysm in blood vessels is caused by destroying the elastic fibers of the media, induced by inflammation. The majority of pseudoaneurysms is caused by Behcet’s syndrome, while pseudoaneurysms caused by primary vasculitis are not common in incidence[38].
Aortic pseudoaneurysms are caused by atherosclerotic ulcer due to disturbance of internal elastic lamina, which can lead to aortic rupture or aortic dissection[39].
Another cause of pseudoaneurysm is infarction of the left ventricle. It occurs due to separation of the left ventricle free wall enclosed by superimposing adherent pericardium, generated what has been named “pseudoaneurysm of the left ventricle”[40].
The gold stander of pseudoaneurysm treatment in general is surgical intervention. The intervention includes arterial ligation, organectomy either partially or totally, and resection using bypass techniques. Surgical treatment is associated with increased morbidity and mortality as compared with minimally invasive treatment options. The complications associated with surgery include bleeding, infection, lymphocele formation, radiculopathy, perioperative myocardial infarction, and death[41].
Endovascular approaches to therapy offers distinct advantage to conventional surgical repair in patients with visceral pseudoaneurysms[42]. Several endovascular techniques have been described to treat pseudoaneurysms. These techniques include catheter-guided embolization with use of coils or detachable balloons[43,44]. Similar management principles are applied to management of aortic pseudoaneurysms[45]. Compared to other techniques, endovascular procedures have lower morbidity and mortality rate in the management of pseudoaneurysm compared to surgical intervention[19].
Percutaneous US-guided thrombin injection is an important treatment option for the treatment of pseudoaneurysms. This approach appears to be a safe and expeditious method for treating postcatheterization femoral pseudoaneurysms. It has significant advantages with respect to ultrasound guided compression repair or surgical repair[19]. Recently, the percutaneous thrombin injection was introduced for the treatment of iatrogenic pseudoaneurysm of femoral artery[46,47].
In addition to that, this procedure can be used to treat arteries above the inguinal ligament and is considered as an alternative technique to US-guided compression in order to avoid arterial rupture[48]. Complications of thrombin injection are uncommon, occurring in 0%-4% of cases[12]. Most reported complications involve the escape of thrombin into the native circulation, causing distal embolization. This occurs in as many as 2% of all patients treated[41].
US-guided compression of pseudoaneurysms is a safe and cost-effective method for achieving pseudoaneurysm thrombosis. However, it has been demonstrated that the success rate is higher and procedure time is much shorter for thrombin injection compared with US compression[48]. Furthermore, compression of pseudoaneurysm is painful to the patient and time-consuming for the practitioner. US-guided compression is more likely to fail in a patient with anticoagulation, large pseudoaneurysm size, chronic pseudoaneurysm, and longer procedure time. The incidence of complications is small but they occasionally do occur[19].
The characteristic appearance of pseudoaneurysm in upper extremities arterial vessels is the extraluminal pattern of blood flow, which shows variable echogenicity, interval complexity, and “to-and-fro” flow pattern on color Doppler ultrasonography (CDUS)[49,50]. It has been estimated that 2% to 3% of pseudoaneurysm in Subclavian artery occur due to blunt trauma, or injuries after clavicle fracture[51].
Pseudoaneurysm of radial artery could be caused also as a result of bacterial infection at cannulation site[8]. It has been considered that radial artery pseudoaneurysm is a rare pathological condition accounting and incidence of 0.048%[52].
Rozen et al[53] reported that pseudoaneurysm of radial artery are a common finding in patients with anticoagulated or patient under antiplatelet treatment. It’s crucial to deliberate pseudoaneurysm diagnosis in any swelling that may presents swelling in order to avoid puncture or incision of the vessel because this swelling could be tender and warm[54].
Several imaging modalities may be used to detect pseudoaneurysms in upper extremity arterial vessels, including conventional arteriography, computed tomography (CT) angiography, radionuclide angiography, and CDUS. US imaging is a diagnostic method of choice required to access pseudoaneurysm before US guided intervention is established for pseudoaneurysm of radial artery[55,56]. CDUS is accurate, noninvasive imaging technique, and widely available. Therefore, it can be used to diagnose pseudoaneurysm of radial artery without even a side effect[51,57,58]. US imaging procedure has the ability to differentiate between solid and cystic lesions adjacent to the radial artery in the wrist area[53]. The sonographic appearance of the radial artery characterized by a feature of sonolucent pulsatile tube[59]. Spectral Doppler of the radial artery pseudoaneurysm is usually shown both “yin-yang” sign and “to-and-fro” waveform[51,57,60].
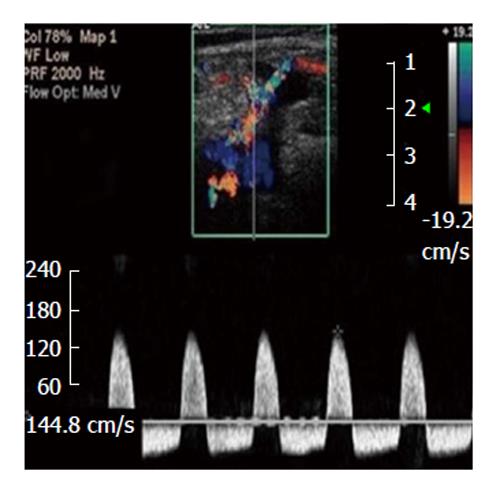
A recent report describes the attempted repair of a brachial artery pseudoaneurysm in an infant that resulted in the thrombosis of the underlying brachial artery and an emergent thrombectomy[61]. The light of the fact that neonates brachial artery injuries are uncommon, but induced by a brachial artery puncture. Therefore, this intervention is not recommended in neonates[62]. In the literature induction of brachial artery pseudoaneurysm due to venipuncture was documented in two instances[63]. Arterial injuries can be diagnosed promptly by using Duplex US imaging technique (Figure 1), without any further need for angiography[64,65]. Also DDU can be up to 95% to 100% sensitive for diagnosing vascular injuries in the hands of highly qualified personnel with a high index of suspicion[5].
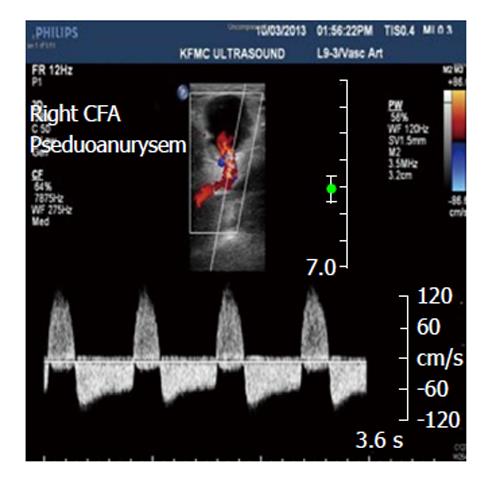
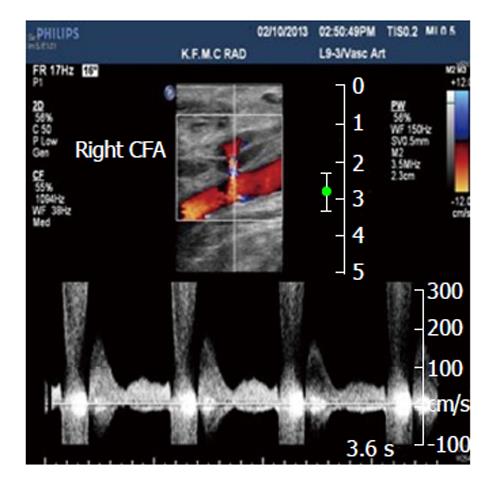
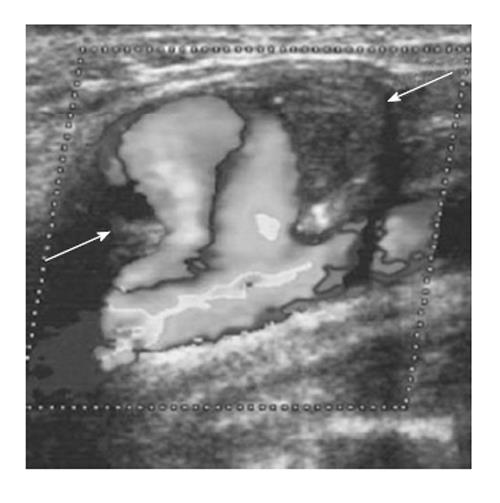
The incidence of pseudoaneurysm in lower extremity arterial vessels was estimated to be ranged from 3.5%-5.5% and 0.1%-0.2% of the Interventional examination and diagnostic radiography, in that order[66,67]. Femoral artery pseudoaneurysms are usually accompanied with a certain features of an audible “to-and-fro” pulsatile mass and touchable thrill (Figure 2). Duplex US of femoral artery (Figure 3) is the diagnostic method of choice for the diagnosis of Pseudoaneurysm[67]. This imaging technique can reveal the blood flow waveform, blood clotting, and the relation with the femoral artery[67].
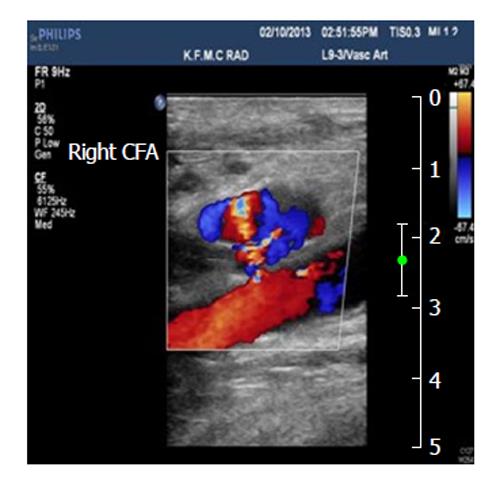
The common femoral artery is the most frequent site of pseudoaneurysm in the lower extremities (Figure 4). This can be attributed to the localization of the common femoral artery inside the neurovascular sheet and it’s supported by the head of the femur. Also the common femoral artery site is the place of choice to introduce cardiac catheterization. The incidence of pseudoaneurysm in the superficial femoral artery is less frequent in occurrence when compare with the common femoral artery because this artery is usually not selected for cardiac catheterization as a result of insufficient supportive tissue around it[68,69].
Also popliteal artery is the most frequent region for pseudoaneurysm incidence because this artery is not supported by muscular tissue to shield it from dilatation and bending, compared to superficial and deep femoral arteries[70]. Enlarging and pulsatile mass located in the popliteal artery are the common features of aneurysmal lesion[70,71]. There are a similarity in diagnostic findings between popliteal artery mycotic pseudoaneurysm and other pseudoaneurysms on the basis of CDUS finding[2,72].
Pseudoaneurysms of the anterior tibial artery and tibioperoneal trunk are exceedingly rare[73,74]. Owen et al[75] reported that pseudoaneurysms of the tibial arteries can be treated using percutaneous injection of thrombin and tissue adhesive. To prevent sudden incidence of a thrombosis in the native vessel, occlusion balloon can be used. An important study reported by Davis et al[76] showed that pseudoaneurysm can be treated with percutaneous infection of thrombin at the posterior tibial and distal superficial femoral arteries. Pseudoaneurysm can be formed during surgical replacement of the knee joint. This can occur either direct (intra-operative) or indirect (intimal plaque disruption)[77].
Some studies reported that the incidence of pseudoaneurysms or aneurysms are rare in the dorsalis pedis artery and usually accompanied with trauma[78-80]. Surgical intervention is preferred to reduce the risk of complication, such as ischemia, arterial rupture, and thrombosis[80,81].
To differentiate between a hematoma and pseudoaneurysm in lower extremities arterial vessels, DDU can be used to establish the accurate diagnosis by demonstrating the relation between the injured artery and aneurysmal neck[82]. In addition, triplex Doppler US can be used for diagnosis of pseudoaneurysm, by presenting “yin-yang” pattern. Bearing in mind that this pattern don’t usually differentiate between pseudoaneurysm and pulsating hematoma[83].
The incidence of abdominal aneurysms has been established by Ertürk et al[84] to be 1% of the overall abdominal aneurysms, concluding that pseudoaneurysms of abdominal aorta has a very low incidence. Pseudoaneurysms of the abdominal aorta are often diagnosed late or after catastrophic complications[85]. Pseudoaneurysms of abdominal aorta caused by medical interventions, these interventions are abdominal surgery, Interventional guided by X-ray imaging of the abdomen, as a complication of abdominal aortic aneurysm, vasculitis, external abdominal trauma, and mycotic aneurysms. Pseudoaneurysms due to external abdominal trauma showed a high incidence in patients treated with anticoagulant or antiplatelet[86].
Shanley et al[87] reported that pseudoaneurysms could be developed in the majority of the visceral artery. A different incidence rate was noted in the splenic artery (46%), renal artery (22%), hepatic artery (16.2%), and pancreaticoduodenal artery (1.3%)[88-90]. Those involving the gastroduodenal artery constitute just 1.5% of all published visceral artery aneurysms[90].
Pseudoaneurysms also took place as a result of a combination of an artery impeded with the wall of pseudocysts[91]. Gastroduodenal and splenic artery pseudoaneurysms are silent in the majority of cases, but in some cases, patients may experience upper abdominal pain or anemia due to bleeding in the gastrointestinal tract or peritoneal cavity[92].
Pseudoaneurysms of splenic artery in different patients are caused by pancreatitis, either chronic or acute pancreatitis. The majority of these patients is characterized by a history of excessive alcohol consumption. The main cause of pseudoaneurysms formation by the aforementioned method is due to the digestion of splenic artery by pancreatic enzymes[93]. Pseudoaneurysm development in the splenic artery due to blunt abdominal trauma had been reported by Sugg et al[94]. Splenic artery slow blood flow is a predisposing factor of pseudoaneurysm as reported by Norotsky et al[95]. In recent year’s noninvasive procedure, therefore the incidence of pseudoaneurysm of splenic artery is increasing in incidence among patients[96]. It has been reported that pseudoaneurysm may develop rarely due to peptic ulcer or as a result of iatrogenic causes. An a tiny number of patients developed pseudoaneurysm in the splenic artery without specific reasons[10].
False aneurysms of the gastroduodenal artery can arise from an impairment in the integrity of the arterial wall, by direct injury via a biopsy needle, enzymatic digestion, as a result of pancreatitis, surgery, or trauma[97]. This defect can lead to the formation of an open communication between the lumen of the artery and its surroundings, which can have two fates. If no soft tissues surround the site of injury, hemorrhage into the peritoneal cavity can occur. The presence of surrounding soft tissue, conversely, can result in containment of the hematoma, which can be followed by fibrosis and enlargement[98]. Pseudoaneurysms have been reported to spontaneous thrombosis, but this is a rare event occurring only under certain conditions[99]. More often, the hematomas become unstable and rupture, being associated with a mortality rate of around 50%[100].
Diagnosis of gastroduodenal and splenic pseudoaneurysm can be made with a number of imaging methods. Contrast-enhanced CT and Doppler sonography are widely used as noninvasive techniques in the diagnosis and monitoring of the lesion[101,102]. On contrast-enhanced CT, a pseudoaneurysm appears as an eccentric mass with a well-defined region of central enhancement in the arterial phase. Doppler sonography shows a mass that generally has a well-defined, solid peripheral component composed of a thrombus and a central anechoic area of varying size. This cavity fills on color Doppler imaging and produces the typical “yin-yang” pattern of pseudoaneurysms anywhere in the body. A “to-and-fro” pattern at the neck of the lesion is confirmatory of a pseudoaneurysm.
Angiography remains the definitive modality used to diagnose, locate, and evaluate the presence of a gastroduodenal and splenic pseudoaneurysm[101,102]. The advantage of this method is that it can be used in the treatment of the lesion as well. Angiography is useful in establishing confirmation of the diagnosis and in cases of an acute rupture or major gastrointestinal bleeding requiring immediate care[93]. The sonographic appearance of abdominal aortic pseudoaneurysm is anechoic blood accumulation in a sac nearby within the artery. This accumulation can be detected by using color Doppler[84]. Sonographic examination of patient using color duplex Doppler revealed a pattern of turbulent flow within pseudoaneurysm illustrated in (Figure 5). Whirlwind flow and “to-and-fro” waveform are seen in the neck of pseudoaneurysm also by using pulsed Doppler[103].
The limitations of color duplex Doppler in the diagnosis of pseudoaneurysms are obese patients and the presence of excessive gasses in the bowel. Neverless ultrasound should be used to establish preliminary diagnosis, especially for patient with pulsatile abdominal masses[84,104].
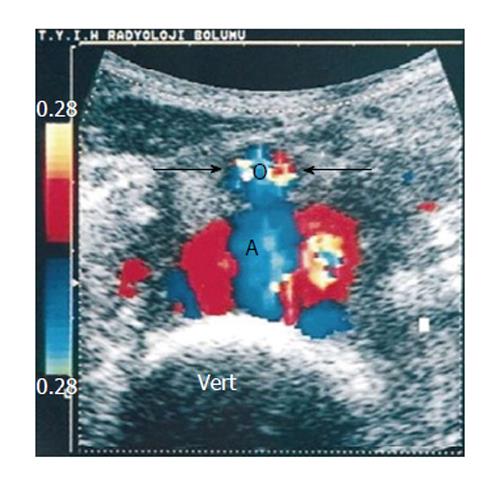
Carotid and vertebral artery pseudoaneurysms are uncommon lesions that may occur as sequelae of blunt trauma, cancer or radiation necrosis, and mycotic infection[104]. Although Doppler ultrasound is a noninvasive imaging procedure, more accurate imaging modalities have been developed such as magnetic resonance angiogram or angiography. However US is the imaging method of choice (Figure 6) to study the midcervical portion of the carotid or vertebral arteries[105-107].
The degree of confidence is high in detection carotid (mid cervical region) and vertebral artery pseudoaneurysms. While the degree of confidence is low in the detection of an intrathoracic segment of the carotid and vertebral arteries[108].
Duplex ultrasound is used on a routine basis to evaluate atherosclerotic lesions. The main findings include dissection, occlusion, pseudoaneurysms, and intimal flaps. Nemours studies used DDU reported that around 92%-100% sensitivity in detection of arterial lesions due to neck trauma[109-111]. The contour of pseudoaneurysms affecting carotid arteries showed variable color flow, depending on the presence of thrombosis[112], while swirling blood flow and “to-and-fro” pattern is shown by spectral Doppler[113]. In common carotid arteries, ultrasound is an effective means of diagnosing a pseudoaneurysm. It may also be used for serial follow the progression of these occurrences once they are diagnosed, as well as to aid in treatment in certain cases[114]. When investigated internal carotid artery pseudoaneurysm by color Doppler it shows swirling of blood flow within the pseudoaneurysm with a communicating channel of the parent artery (yin-yang phenomenon), while pulse Doppler shows “to-and-fro” waveforms[115].
Vertebral artery pseudoaneurysms typically present over the course of several days as a pulsatile mass. Duplex US is used to define the size of the pseudoaneurysms. Adequate visualization of the pseudoaneurysms neck of lesions arising vertebral arteries is limited owing to the overlying clavicle. Angiography is often indicated in order to precisely define the site of injury[30]. However, US examination of vertebral artery pseudoaneurysms is necessary in uncertain or difficult case from the beginning because it is convenient and sensitive in follow-up evaluation[116].
In conclusion, this review study showed that gray scale and Doppler ultrasound play an essential role in the diagnosis of pseudoaneurysms. The use of spectral Doppler in the diagnosis of pseudoaneurysms depends upon the presence of “to-and-fro” waveform. Incidence of arterial pseudoaneurysms are varied in the different body vasculature. Also the choice of pseudoaneurysms treatment is size dependent.
The authors extend their appreciation to the College of Applied Medical Sciences Research Center and the Deanship of Scientific Research at King Saud University for funding this research. In addition, the authors would like to thank the staff of the Radiology Department at KFMC at Riyadh, Saudi Arabia for their cooperation and support during data collection for this article.
P- Reviewer: Aydogdu O, Seicean A S- Editor: Song XX L- Editor: A E- Editor: Liu SQ
| 1. | Saad NE, Saad WE, Davies MG, Waldman DL, Fultz PJ, Rubens DJ. Pseudoaneurysms and the role of minimally invasive techniques in their management. Radiographics. 2005;25 Suppl 1:S173-S189. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 321] [Cited by in F6Publishing: 333] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 2. | Pellerito JS, Taylor KJ. Doppler color imaging. Peripheral arteries. Clin Diagn Ultrasound. 1992;27:97-112. [PubMed] [Cited in This Article: ] |
| 3. | Zierler RE, Zierler BK. Duplex sonography of lower extremity arteries. Semin Ultrasound CT MR. 1997;18:39-56. [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 4. | Pozniak MA, Mitchell C, Ledwidge M. Radial artery pseudoaneurysm: a maneuver to decrease the risk of thrombin therapy. J Ultrasound Med. 2005;24:119-122. [PubMed] [Cited in This Article: ] |
| 5. | Levis JT, Garmel GM. Radial artery pseudoaneurysm formation after cat bite to the wrist. Ann Emerg Med. 2008;51:668-670. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Pellerito JS. Current approach to peripheral arterial sonography. Radiol Clin North Am. 2001;39:553-567. [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Wang HK, Chou YH, Chiou HJ, Chio SY, Chang CY. B-flow ultrasonography of peripheral vascular diseases. J Med Ultrasound. 2005;13:186-195. [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 8. | Gudena R, Khetan N. Swelling of volar aspect of the wrist. Postgrad Med J. 2005;81:e9-e11. [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 9. | Zitsman JL. Pseudoaneurysm after penetrating trauma in children and adolescents. J Pediatr Surg. 1998;33:1574-1577. [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Tessier DJ, Stone WM, Fowl RJ, Abbas MA, Andrews JC, Bower TC, Gloviczki P. Clinical features and management of splenic artery pseudoaneurysm: case series and cumulative review of literature. J Vasc Surg. 2003;38:969-974. [DOI] [Cited in This Article: ] [Cited by in Crossref: 202] [Cited by in F6Publishing: 209] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 11. | Katz DS, Hon M. CT angiography of the lower extremities and aortoiliac system with a multi-detector row helical CT scanner: promise of new opportunities fulfilled. Radiology. 2001;221:7-10. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 45] [Cited by in F6Publishing: 46] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 12. | Morgan R, Belli AM. Current treatment methods for postcatheterization pseudoaneurysms. J Vasc Intervent Radiol. 2003;14:697-710. [DOI] [Cited in This Article: ] [Cited by in Crossref: 197] [Cited by in F6Publishing: 180] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 13. | Katyal S, Oliver JH, Buck DG, Federle MP. Detection of vascular complications after liver transplantation: early experience in multislice CT angiography with volume rendering. AJR Am J Roentgenol. 2000;175:1735-1739. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 78] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 14. | Soto JA, Múnera F, Morales C, Lopera JE, Holguín D, Guarín O, Castrillón G, Sanabria A, García G. Focal arterial injuries of the proximal extremities: helical CT arteriography as the initial method of diagnosis. Radiology. 2001;218:188-194. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 168] [Cited by in F6Publishing: 132] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 15. | Crossin JD, Muradali D, Wilson SR. US of liver transplants: normal and abnormal. Radiographics. 2003;23:1093-1114. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 172] [Cited by in F6Publishing: 135] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 16. | Kwon JH, Kim GS. Obstetric iatrogenic arterial injuries of the uterus: diagnosis with US and treatment with transcatheter arterial embolization. Radiographics. 2002;22:35-46. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 138] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 17. | Krüger K, Zähringer M, Söhngen FD, Gossmann A, Schulte O, Feldmann C, Strohe D, Lackner K. Femoral pseudoaneurysms: management with percutaneous thrombin injections--success rates and effects on systemic coagulation. Radiology. 2003;226:452-458. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 77] [Cited by in F6Publishing: 69] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 18. | Polak JF. The peripheral arteries. Diagnostic ultrasound. St Louis: Mosby 1998; 921-941. [Cited in This Article: ] |
| 19. | Kapoor BS, Haddad HL, Saddekni S, Lockhart ME. Diagnosis and management of pseudoaneurysms: an update. Curr Probl Diagn Radiol. 2009;38:170-188. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 20. | Fransson SG, Nylander E. Vascular injury following cardiac catheterization, coronary angiography, and coronary angioplasty. Eur Heart J. 1994;15:232-235. [PubMed] [Cited in This Article: ] |
| 21. | Kacila M, Vranic H, Hadzimehmedagic A, Sehovic S, Granov N. The frequency of complications of pseudoaneurysms after cardiac interventional diagnostic and therapeutic interventions. Med Arh. 2011;65:78-81. [PubMed] [Cited in This Article: ] |
| 22. | Görge G, Kunz T, Kirstein M. [Non-surgical therapy of iatrogenic false aneurysms]. Dtsch Med Wochenschr. 2003;128:36-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 23. | Righini M, Quéré I, Laroche JP. [Treatment of postcatheterization femoral false aneurysms]. J Mal Vasc. 2004;29:63-72. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Wielenberg A, Borge MA, Demos TC, Lomasney L, Marra G. Traumatic pseudoaneurysm of the brachial artery. Orthopedics. 2000;23:1250, 1322-1324. [PubMed] [Cited in This Article: ] |
| 25. | Szendro G, Golcman L, Klimov A, Yefim C, Johnatan B, Avrahami E, Yechieli B, Yurfest S. Arterial false aneurysms and their modern management. Isr Med Assoc J. 2001;3:5-8. [PubMed] [Cited in This Article: ] |
| 26. | Dhadwal AK, Abrol S, Zisbrod Z, Cunningham JN. Pseudoaneurysms of the ascending aorta following coronary artery bypass surgery. J Card Surg. 2006;21:221-224. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Garisto JD, Medina A, Williams DB, Carrillo RG. Surgical management of a giant ascending aortic pseudoaneurysm. Tex Heart Inst J. 2010;37:710-713. [PubMed] [Cited in This Article: ] |
| 28. | Potts RG, Alguire PC. Pseudoaneurysm of the abdominal aorta: a case report and review of the literature. Am J Med Sci. 1991;301:265-268. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Kim HO, Ji YB, Lee SH, Jung C, Tae K. Cases of common carotid artery pseudoaneurysm treated by stent graft. Case Rep Otolaryngol. 2012;2012:674827. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 30. | Bernik TR, Friedman SG, Scher LA, Safa T. Pseudoaneurysm of the subclavian-vertebral artery junction--case report and review of the literature. Vasc Endovascular Surg. 2002;36:461-464. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 26] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 31. | Cihangiroglu M, Rahman A, Yildirim H, Burma O, Uysal H. Iatrogenic vertebral artery pseudoaneurysm: US, CT and MRI findings. Eur J Radiol. 2002;43:14-18. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 32. | Franklin JA, Brigham D, Bogey WM, Powell CS. Treatment of iatrogenic false aneurysms. J Am Coll Surg. 2003;197:293-301. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 44] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Feliciano DV, Mattox KL. Traumatic aneurysms. Vascular surgery. Philadelphia: Saunders 1989; 996-1003. [Cited in This Article: ] |
| 34. | Smith BL, Munschauer CE, Diamond N, Rivera F. Ruptured internal carotid aneurysm resulting from neurofibromatosis: treatment with intraluminal stent graft. J Vasc Surg. 2000;32:824-828. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 56] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 35. | Yang DM, Yoon MH, Kim HS, Kim HS, Shin DB. Intrarenal pseudoaneurysms complicating renal choriocarcinoma metastases: treatment with coil embolization. Clin Imaging. 2000;24:217-220. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 36. | Kim MD, Kim H, Kang SW, Jeong BG. Nontraumatic hepatic artery pseudoaneurysm associated with acute leukemia: a possible complication of pyogenic liver abscess. Abdom Imaging. 2002;27:458-460. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 37. | Gomes MN, Choyke PL. Infected aortic aneurysms: CT diagnosis. J Cardiovasc Surg (Torino). 1992;33:684-689. [PubMed] [Cited in This Article: ] |
| 38. | Ko GY, Byun JY, Choi BG, Cho SH. The vascular manifestations of Behçet’s disease: angiographic and CT findings. Br J Radiol. 2000;73:1270-1274. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 95] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 39. | Macura KJ, Corl FM, Fishman EK, Bluemke DA. Pathogenesis in acute aortic syndromes: aortic dissection, intramural hematoma, and penetrating atherosclerotic aortic ulcer. AJR Am J Roentgenol. 2003;181:309-316. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 131] [Cited by in F6Publishing: 105] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 40. | Brown SL, Gropler RJ, Harris KM. Distinguishing left ventricular aneurysm from pseudoaneurysm. A review of the literature. Chest. 1997;111:1403-1409. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 118] [Cited by in F6Publishing: 117] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 41. | La Perna L, Olin JW, Goines D, Childs MB, Ouriel K. Ultrasound-guided thrombin injection for the treatment of postcatheterization pseudoaneurysms. Circulation. 2000;102:2391-2395. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 156] [Cited by in F6Publishing: 127] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 42. | Tulsyan N, Kashyap VS, Greenberg RK, Sarac TP, Clair DG, Pierce G, Ouriel K. The endovascular management of visceral artery aneurysms and pseudoaneurysms. J Vasc Surg. 2007;45:276-283; discussion 283. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 399] [Cited by in F6Publishing: 397] [Article Influence: 23.4] [Reference Citation Analysis (1)] |
| 43. | Nicholson AA, Patel J, McPherson S, Shaw DR, Kessel D. Endovascular treatment of visceral aneurysms associated with pancreatitis and a suggested classification with therapeutic implications. J Vasc Interv Radiol. 2006;17:1279-1285. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 44. | Sharma RP, Shetty PC, Burke TH, Shepard AD, Khaja F. Treatment of false aneurysm by using a detachable balloon. AJR Am J Roentgenol. 1987;149:1279-1280. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 12] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 45. | Mitchell JH, Dougherty KG, Strickman NE, Mortazavi A, Krajcer Z. Endovascular repair of paraanastomotic aneurysms after aortic reconstruction. Tex Heart Inst J. 2007;34:148-153. [PubMed] [Cited in This Article: ] |
| 46. | Brophy DP, Sheiman RG, Amatulle P, Akbari CM. Iatrogenic femoral pseudoaneurysms: thrombin injection after failed US-guided compression. Radiology. 2000;214:278-282. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 89] [Cited by in F6Publishing: 92] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 47. | Bydawell G. Percutaneous thrombin injection for pseudoaneurysm treatment. S Afr J Rad. 2013;17:41-42. [DOI] [Cited in This Article: ] |
| 48. | Paulson EK, Sheafor DH, Kliewer MA, Nelson RC, Eisenberg LB, Sebastian MW, Sketch MH. Treatment of iatrogenic femoral arterial pseudoaneurysms: comparison of US-guided thrombin injection with compression repair. Radiology. 2000;215:403-408. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 105] [Cited by in F6Publishing: 106] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 49. | Eisenberg L, Paulson EK, Kliewer MA, Hudson MP, DeLong DM, Carroll BA. Sonographically guided compression repair of pseudoaneurysms: further experience from a single institution. AJR Am J Roentgenol. 1999;173:1567-1573. [PubMed] [Cited in This Article: ] |
| 50. | Chiou HJ, Chou YH, Chiou SY, Wang HK. High-resolution ultrasonography in superficial soft tissue tumors. J Med Ultrasound. 2007;15:152-174. [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 51. | Falk PS, Scuderi PE, Sherertz RJ, Motsinger SM. Infected radial artery pseudoaneurysms occurring after percutaneous cannulation. Chest. 1992;101:490-495. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 46] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 52. | Cozzi DA, Morini F, Casati A, Pacilli M, Salvini V, Cozzi F. Radial artery pseudoaneurysm successfully treated by compression bandage. Arch Dis Child. 2003;88:165-166. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 53. | Rozen G, Samuels DR, Blank A. The to and fro sign: the hallmark of pseudoaneurysm. Isr Med Assoc J. 2001;3:781-782. [PubMed] [Cited in This Article: ] |
| 54. | Barker C, Jefferson P, Ball DR. Portable ultrasound to diagnose true radial artery aneurysm. Anesth Analg. 2007;105:890-891. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Cited by in F6Publishing: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 55. | Gooding GA. Sonography of the radial artery at the wrist. AJR Am J Roentgenol. 1988;150:629-631. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 11] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 56. | Carrafiello G, Laganà D, Mangini M, Recaldini C, Mandas X, Fugazzola C. Post-traumatic pseudoaneurysm of radial artery: percutaneous treatment with thrombin injection. Injury Extra. 2006;37:78-81. [DOI] [Cited in This Article: ] [Cited by in Crossref: 4] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 57. | Lennox A, Griffin M, Nicolaides A, Mansfield A. Regarding “Percutaneous ultrasound guided thrombin injection: a new method for treating postcatheterization femoral pseudoaneurysms”. J Vasc Surg. 1998;28:1120-1121. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 79] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 58. | Davison BD, Polak JF. Arterial injuries: a sonographic approach. Radiol Clin North Am. 2004;42:383-396. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 59. | Truong AT, Thakar DR. Radial artery pseudoaneurysm: a rare complication with serious risk to life and limb. Anesthesiology. 2013;118:188. [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 11] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 60. | Landau D, Schreiber R, Szendro G, Golcman L. Brachial artery pseudoaneurysm in a premature infant. Arch Dis Child Fetal Neonatal Ed. 2003;88:F152-F153. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 61. | Demircin M, Peker O, Tok M, Ozen H. False aneurysm of the brachial artery in an infant following attempted venipuncture. Turk J Pediatr. 1996;38:389-391. [PubMed] [Cited in This Article: ] |
| 62. | Verlato F, Zanon GF, Gamba PG, Verlato G, Rocco S, Orzali A, Camporese G, Signorini GP. Echo Doppler color flow (EDCF) evaluation of vascular pathology in pediatric age groups. Int Angiol. 1996;15:321-327. [PubMed] [Cited in This Article: ] |
| 63. | Dzepina I, Unusic J, Mijatovic D, Bulic K. Pseudoaneurysms of the brachial artery following venipuncture in infants. Pediatr Surg Int. 2004;20:594-597. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 64. | Gullo J, Singletary EM, Larese S. Emergency bedside sonographic diagnosis of subclavian artery pseudoaneurysm with brachial plexopathy after clavicle fracture. Ann Emerg Med. 2013;61:204-206. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 65. | Pero T, Herrick J. Pseudoaneurysm of the radial artery diagnosed by bedside ultrasound. West J Emerg Med. 2009;10:89-91. [PubMed] [Cited in This Article: ] |
| 66. | Goel PK, Modi N, Baijal SS, Kathuria M, Agrawal SK. Sonographically guided thrombin injection for the treatment of femoral artery pseudoaneurysm. Indian Heart J. 2003;55:365-367. [PubMed] [Cited in This Article: ] |
| 67. | Kronzon I. Diagnosis and treatment of iatrogenic femoral artery pseudoaneurysm: a review. J Am Soc Echocardiogr. 1997;10:236-245. [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 62] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 68. | Fellmeth BD, Roberts AC, Bookstein JJ, Freischlag JA, Forsythe JR, Buckner NK, Hye RJ. Postangiographic femoral artery injuries: nonsurgical repair with US-guided compression. Radiology. 1991;178:671-675. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 332] [Cited by in F6Publishing: 279] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 69. | Demirbas O, Batyraliev T, Eksi Z, Pershukov I. Femoral pseudoaneurysm due to diagnostic or interventional angiographic procedures. Angiology. 2005;56:553-556. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 70. | Sadler L, Bolden RO, Lenkey JL. Diagnosis of a ruptured deep femoral artery aneurysm--a case report. Angiology. 1989;40:678-681. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 71. | Harman M, Irmak H, Arslan H, Arslan U, Kayan M. Popliteal artery pseudoaneurysm: a rare complication of brucellosis. J Clin Ultrasound. 2004;32:33-36. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 72. | Murashita T, Yasuda K, Takigami T, Sakuma M, Matsui Y, Sasaki S, Shiiya N. Mycotic aneurysm of the bilateral tibioperoneal trunks associated with bacterial endocarditis: a case report. Int Angiol. 1997;16:176-179. [PubMed] [Cited in This Article: ] |
| 73. | Cappendijk VC, Mouthaan PJ. A true aneurysm of the tibioperoneal trunk. Case report and literature review. Eur J Vasc Endovasc Surg. 1999;18:536-537. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 15] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 74. | McKee MA, Ballard JL. Mycotic aneurysms of the tibioperoneal arteries. Ann Vasc Surg. 1999;13:188-190. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 45] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 75. | Owen RJ, Haslam PJ, Elliott ST, Rose JD, Loose HW. Percutaneous ablation of peripheral pseudoaneurysms using thrombin: a simple and effective solution. Cardiovasc Intervent Radiol. 2000;23:441-446. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 76. | Davis KA, Mansour MA, Kang SS, Labropoulos N, Esposito TJ, Silver GM, Reed RL. Pseudoaneurysms of the extremity without fracture: treatment with percutaneous ultrasound-guided thrombin injection. J Trauma. 2000;49:818-821. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 21] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 77. | Law KY, Cheung KW, Chiu KH, Antonio GE. Pseudoaneurysm of the geniculate artery following total knee arthroplasty: a report of two cases. J Orthop Surg (Hong Kong). 2007;15:386-389. [PubMed] [Cited in This Article: ] |
| 78. | McKee TI, Fisher JB. Dorsalis pedis artery aneurysm: case report and literature review. J Vasc Surg. 2000;31:589-591. [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 79. | Tempest HV, Wilson YG. Acute forefoot ischaemia: an unreported complication of dorsalis pedis artery aneurysm. Eur J Vasc Endovasc Surg. 2001;22:472-473. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 22] [Cited by in F6Publishing: 22] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 80. | Taylor DT, Mansour MA, Bergin JT, Reyes CV, Stuck RM. Aneurysm of the dorsalis pedis artery -- a case report. Vasc Endovascular Surg. 2002;36:241-245. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 16] [Cited by in F6Publishing: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 81. | Ozdemir H, Mahmutyazicioğlu K, Ozkökeli M, Savranlar A, Ozer T, Demirel F. Pseudoaneurysm of the dorsalis pedis artery: color Doppler sonographic and angiographic findings. J Clin Ultrasound. 2003;31:283-287. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 82. | Abu-Yousef MM, Wiese JA, Shamma AR. The „to-and-fro“ sign: duplex Doppler evidence of femoral artery pseudoaneurysm. AJR Am J Roentgenol. 1988;150:632-634. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 96] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 83. | Carroll BA, Graif M, Orron DE. Vascular ultrasound. Peripheral vascular imaging and intervention. St. Louis: Mosby 1992; 211-225. [Cited in This Article: ] |
| 84. | Ertürk H, Erden A, Yurdakul M, Calikoğlu U, Olçer T, Cumhur T. Pseudoaneurysm of the abdominal aorta diagnosed by color duplex Doppler sonography. J Clin Ultrasound. 1999;27:202-205. [DOI] [Cited in This Article: ] |
| 85. | van Herwaarden JA, Waasdorp EJ, Bendermacher BL, van den Berg JC, Teijink JA, Moll FL. Endovascular repair of paraanastomotic aneurysms after previous open aortic prosthetic reconstruction. Ann Vasc Surg. 2004;18:280-286. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 53] [Cited by in F6Publishing: 54] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 86. | Siegel CL, Cohan RH. CT of abdominal aortic aneurysms. AJR Am J Roentgenol. 1994;163:17-29. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 63] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 87. | Shanley CJ, Shah NL, Messina LM. Uncommon splanchnic artery aneurysms: pancreaticoduodenal, gastroduodenal, superior mesenteric, inferior mesenteric, and colic. Ann Vasc Surg. 1996;10:506-515. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 128] [Cited by in F6Publishing: 104] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 88. | Deterling RA Jr. Aneurysm of the visceral arteries. J Cardiovasc Surg (Torino). 1997;12:309-322. [PubMed] [Cited in This Article: ] |
| 89. | White AF, Baum S, Buranasiri S. Aneurysms secondary to pancreatitis. AJR Am J Roentgenol. 1976;127:393-396. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 159] [Cited by in F6Publishing: 167] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 90. | Walter JF, Chuang VP, Bookstein JJ, Reuter SR, Cho KJ, Pulmano CM. Angiography of massive hemorrhage secondary to pancreatic diseases. Radiology. 1977;124:337-342. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 57] [Cited by in F6Publishing: 58] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 91. | Friedman AC. Radiology of the liver, biliary tract, pancreas and spleen. Baltimore: Williams and Wilkins 1987; 674. [Cited in This Article: ] |
| 92. | Stanley JC, Zelenock GB. Splanchnic artery aneurysms. Vascular surgery. Philadelphia: W.B. Saunders Company 1995; 1124-1128. [Cited in This Article: ] |
| 93. | Golzarian J, Nicaise N, Devière J, Ghysels M, Wery D, Dussaussois L, Van Gansbeke D, Struyven J. Transcatheter embolization of pseudoaneurysms complicating pancreatitis. Cardiovasc Intervent Radiol. 1997;20:435-440. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 53] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 94. | Sugg SL, Gerndt SJ, Hamilton BJ, Francis IR, Taheri PA, Rodriguez JL. Pseudoaneurysms of the intraparenchymal splenic artery after blunt abdominal trauma: a complication of nonoperative therapy and its management. J Trauma. 1995;39:593-595. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 34] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 95. | Norotsky MC, Rogers FB, Shackford SR. Delayed presentation of splenic artery pseudoaneurysms following blunt abdominal trauma: case reports. J Trauma. 1995;38:444-447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 96. | Davis KA, Fabian TC, Croce MA, Gavant ML, Flick PA, Minard G, Kudsk KA, Pritchard FE. Improved success in nonoperative management of blunt splenic injuries: embolization of splenic artery pseudoaneurysms. J Trauma. 1998;44:1008-1013; discussion 1013-1015. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 225] [Cited by in F6Publishing: 207] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 97. | Parildar M, Oran I, Memis A. Embolization of visceral pseudoaneurysms with platinum coils and N-butyl cyanoacrylate. Abdom Imaging. 2003;28:36-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 74] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 98. | Bergan J, Yao J. Aneurysms: Diagnosis and Treatment. New York: Grune and Stratton 1982; . [Cited in This Article: ] |
| 99. | Vanlangenhove P, Defreyne L, Kunnen M. Spontaneous thrombosis of a pseudoaneurysm complicating pancreatitis. Abdom Imaging. 1999;24:491-493. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 100. | Stanley JC, Wakefield TW, Graham LM, Whitehouse WM, Zelenock GB, Lindenauer SM. Clinical importance and management of splanchnic artery aneurysms. J Vasc Surg. 1986;3:836-840. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 275] [Cited by in F6Publishing: 236] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 101. | Gabelmann A, Görich J, Merkle EM. Endovascular treatment of visceral artery aneurysms. J Endovasc Ther. 2002;9:38-47. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 102. | Pilleul F, Dugougeat F. Transcatheter embolization of splanchnic aneurysms/pseudoaneurysms: early imaging allows detection of incomplete procedure. J Comput Assist Tomogr. 2002;26:107-112. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 103. | González Llorente J, Gallego Gallego M, Martínez Arnaiz A. Chronic post-traumatic pseudoaneurysm of the abdominal aorta diagnosed by duplex Doppler ultrasonography. A case report. Acta Radiol. 1997;38:121-123. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 104. | Chaikof EL, Shamberger RC, Brewster DC. Traumatic pseudoaneurysms of the abdominal aorta. J Trauma. 1985;25:169-173. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 22] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 105. | Maras D, Lioupis C, Magoufis G, Tsamopoulos N, Moulakakis K, Andrikopoulos V. Covered stent-graft treatment of traumatic internal carotid artery pseudoaneurysms: a review. Cardiovasc Intervent Radiol. 2006;29:958-968. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 110] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 106. | Flor N, Sardanelli F, Ghilardi G, Tentori A, Franceschelli G, Felisati G, Cornalba GP. Common carotid artery pseudoaneurysm after neck dissection: colour Doppler ultrasound and multidetector computed tomography findings. J Laryngol Otol. 2007;121:497-500. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 107. | Nebelsieck J, Sengelhoff C, Nassenstein I, Maintz D, Kuhlenbäumer G, Nabavi DG, Ringelstein EB, Dittrich R. Sensitivity of neurovascular ultrasound for the detection of spontaneous cervical artery dissection. J Clin Neurosci. 2009;16:79-82. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 108. | Kochan JP, Kanamalla US. Imaging in carotid and vertebral artery dissection. Medscape serial online, 2013-07-24, cited 2014-04-10; 1(1): 7 screens. Available from: http://emedicine.medscape.com/article/417341-overview#showall. [Cited in This Article: ] |
| 109. | Montalvo BM, LeBlang SD, Nuñez DB, Ginzburg E, Klose KJ, Becerra JL, Kochan JP. Color Doppler sonography in penetrating injuries of the neck. AJNR Am J Neuroradiol. 1996;17:943-951. [PubMed] [Cited in This Article: ] |
| 110. | Ginzberg E, Montalvo B, LeBlang S, Nunez D, Martin L. The use of duplex ultrasonography in penetrating neck trauma. Arch Surg. 1996;131:691-693. [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 53] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 111. | Demetriades D, Theodorou D, Cornwell E, Berne TV, Asensio J, Belzberg H, Velmahos G, Weaver F, Yellin A. Evaluation of penetrating injuries of the neck: prospective study of 223 patients. World J Surg. 1997;21:41-47; discussion 47-48. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 191] [Cited by in F6Publishing: 204] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 112. | LeBlang SD, Nu-ez DB Jr, Rivas LA, Falcone S, Pogson SE. Helical computed tomographic angiography in penetrating neck trauma. Emerg Radiol. 1997;4:200-206. [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 113. | LeBlang SD, Nunez DB. Noninvasive imaging of cervical vascular injuries. AJR Am J Roentgenol. 2000;174:1269-1278. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 51] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 114. | Scalf TC, Drose JA. Ultrasound diagnosis of a common carotid artery pseudoaneurysm. J Diagn Med Sonogr. 2000;16:202-204. [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 115. | Ramesh A, Muthukumarassamy R, Karthikeyan VS, Rajaraman G, Mishra S. Pseudoaneurysm of internal carotid artery after carotid body tumor excision. Indian J Radiol Imaging. 2013;23:208-211. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 5] [Cited by in F6Publishing: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 116. | Lu CJ, Sun Y, Jeng JS, Huang KM, Hwang BS, Lin WH, Chen RC, Yip PK. Imaging in the diagnosis and follow-up evaluation of vertebral artery dissection. J Ultrasound Med. 2000;19:263-270. [PubMed] [Cited in This Article: ] |