Published online Aug 26, 2017. doi: 10.4330/wjc.v9.i8.652
Peer-review started: February 22, 2017
First decision: May 8, 2017
Revised: May 13, 2017
Accepted: June 30, 2017
Article in press: July 3, 2017
Published online: August 26, 2017
Processing time: 183 Days and 1.6 Hours
Celiac disease (CD) is an autoimmune response to ingestion of gluten protein, which is found in wheat, rye, and barley grains, and results in both small intestinal manifestations, including villous atrophy, as well as systemic manifestations. The main treatment for the disease is a gluten-free diet (GFD), which typically results in the restoration of the small intestinal villi, and restoration of other affected organ systems, to their normal functioning. In an increasing number of recently published studies, there has been great interest in the occurrence of alterations in the cardiovascular system in untreated CD. Herein, published studies in which CD and cardiovascular terms appear in the title of the study were reviewed. The publications were categorized into one of several types: (1) articles (including cohort and case-control studies); (2) reviews and meta-analyses; (3) case studies (one to three patient reports); (4) letters; (5) editorials; and (6) abstracts (used when no full-length work had been published). The studies were subdivided as either heart or vascular studies, and were further characterized by the particular condition that was evident in conjunction with CD. Publication information was determined using the Google Scholar search tool. For each publication, its type and year of publication were tabulated. Salient information from each article was then compiled. It was determined that there has been a sharp increase in the number of CD - cardiovascular studies since 2000. Most of the publications are either of the type “article” or “case study”. The largest number of documents published concerned CD in conjunction with cardiomyopathy (33 studies), and there have also been substantial numbers of studies published on CD and thrombosis (27), cardiovascular risk (17), atherosclerosis (13), stroke (12), arterial function (11), and ischemic heart disease (11). Based on the published research, it can be concluded that many types of cardiovascular issues can occur in untreated CD patients, but that most tend to resolve on a GFD, often in conjunction with the healing of small intestinal villous atrophy. However, in some cases the alterations are irreversible, underscoring the need for CD screening and treatment when cardiovascular issues arise of unknown etiology.
Core tip: Celiac disease (CD) is a public health concern suffered by about 1% of the population worldwide. It often goes undetected even in developed countries, owing to the varied and occult presentation which can make diagnosis difficult. Untreated, systemic manifestations including cardiovascular ailments can occur. In this review, information concerning the cardiovascular involvement in CD patients is described and discussed. Treatment of CD patients with a gluten free diet can reverse some, but not all of the cardiovascular involvement. Thus the need for prompt diagnosis and treatment.
- Citation: Ciaccio EJ, Lewis SK, Biviano AB, Iyer V, Garan H, Green PH. Cardiovascular involvement in celiac disease. World J Cardiol 2017; 9(8): 652-666
- URL: https://www.wjgnet.com/1949-8462/full/v9/i8/652.htm
- DOI: https://dx.doi.org/10.4330/wjc.v9.i8.652
Celiac disease (CD) is characterized by an immunologic response to gluten, which often results in diffuse inflammatory damage to the small intestinal mucosa, and malabsorption of nutrients[1]. CD is of special interest among chronic diseases due to several factors: (1) it is associated with specific comorbidities; (2) it involves a compromised absorption of nutrients; and (3) a gluten-free diet (GFD) is currently the main long-term treatment[2]. Studies have shown that certain cardiovascular maladies, including cardiomyopathy, myocarditis, arrhythmias, and premature atherosclerosis, are more prevalent in individuals with CD as compared to individuals without the disease[3,4]. In this article, published works concerning the effects of CD on the cardiovascular system, and the risk of cardiovascular disease, are reviewed. The method of some previous analyses is followed to quantitatively characterize the published articles[5-7], and to then compile the most salient information for review.
The Google Scholar search tool was used to find associations between CD and the heart and vascular systems. Keywords pertaining to the heart and vascular system, tabulated in Table 1, were used for search in conjunction with “celiac disease”, “coeliac disease”, or “gluten”. The searches were limited to the co-detection of these terms in the publication title, which is suggestive of the importance of the keywords. The cardiovascular keywords used for search were derived from encyclopedic descriptions of the heart, vascular, and cardiovascular systems. Under these headings, all relevant terms were extracted as keywords. They were categorized as heart terms and vascular terms. The format used for search in Google Scholar was, for example: (1) allintitle: “celiac disease” “myocardial”; (2) allintitle: “coeliac disease” “myocardial”; and (3) allintitle: “gluten” “myocardial”, where the results for the three forms of expression pertaining to CD were then combined. Heart and vascular keywords, tabulated in Table 1, were then combined to form summary topics for analysis. The number of CD/cardiovascular publications per year was then graphed for all of the summary topics.
| Heart terms | Vascular terms |
| Afterload, preload | Angiogenesis |
| Arrhythmia, rhythm | Aorta, aortic |
| Atrial, atrium | Arterial, arteries, artery |
| Atrioventricular | Arteriosclerosis, atherosclerosis |
| Bachmann’s | Atherogenesis, atherogenic |
| Cardiac, cardio | Blood pressure |
| Cardiologist | Cardiovascular |
| Cardiomyopathies, cardiomyopathy | Circulatory |
| Congenital | Circumflex |
| Contractility | Coronary |
| Depolarization | Embolism |
| Effusion | Haemoptysis, hemoptysis |
| Ejection | Hemorrhage |
| Electromechanical | Haemodynamics, Hemodynamics |
| Endocardium, epicardium | Haemosiderosis, hemosiderosis |
| Fibrillation | Stroke |
| Foramen ovale | Thrombosis, thromboembolism |
| Frank-Starling | Vascular |
| Heart | Vein, veins |
| Infarction | Vena cava |
| Ischaemic, ischemic | Venous |
| Mitral | Venule, venules |
| Myocardial, myocardium | |
| Myocarditis | |
| Myocyte | |
| Pericardial, pericardium | |
| Purkinje | |
| QT | |
| Septum | |
| Sinoatrial | |
| Stenosis | |
| Tachycardia | |
| Tricuspid | |
| Valve, valves, valvular |
The type of published document was also recorded for each keyword entry. Each citation used was categorized by type of published documentation as shown in Table 2. There are six possible publication types according to the list. All published documents were categorized as one of the types noted in Table 2. Graphical displays were utilized to separately show the number of CD/cardiovascular documents of each type noted in Table 2 that were published per year. Then for each of the summary topics, the total number of publications of each type in Table 2 were compiled. Also for each of the summary topics, the total number of studies and the mean publication year of the studies for the journals the studies were published in were tabulated. The essential points in each study were then condensed and described in review form, in separate sections, for each of the summary topics.
| Type | Description |
| Articles | Includes research articles, cohort studies and case control studies |
| Reviews | These included reviews of the literature and meta-analyses |
| Case studies | Limited to n = 1-3 patients in the study |
| Letters | These could include comments on other articles as well as case reports in letter form |
| Abstracts | When no full paper had been published, abstracts were included in the references |
| Editorials | These were typically comments on papers published in the same journal issue |
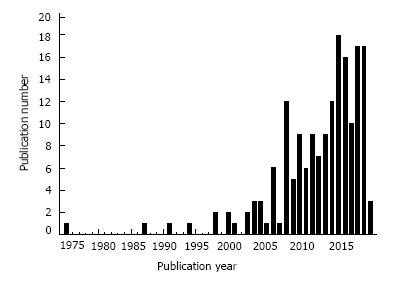
In Figure 1 is presented a graph of the number of publications per year in which CD and cardiovascular terms appeared. The earliest studies in which CD was investigated for cardiovascular function were published in 1970. However until the year 1998, only a handful of such studies were published, after which there began a substantial increase in the publishing of CD/cardiovascular studies. Although there were fluctuations in the number of studies in the 2000s and 2010s, the overall trend was a sharp upward swing in published studies. The data for 2017 only includes the first few weeks of the year.
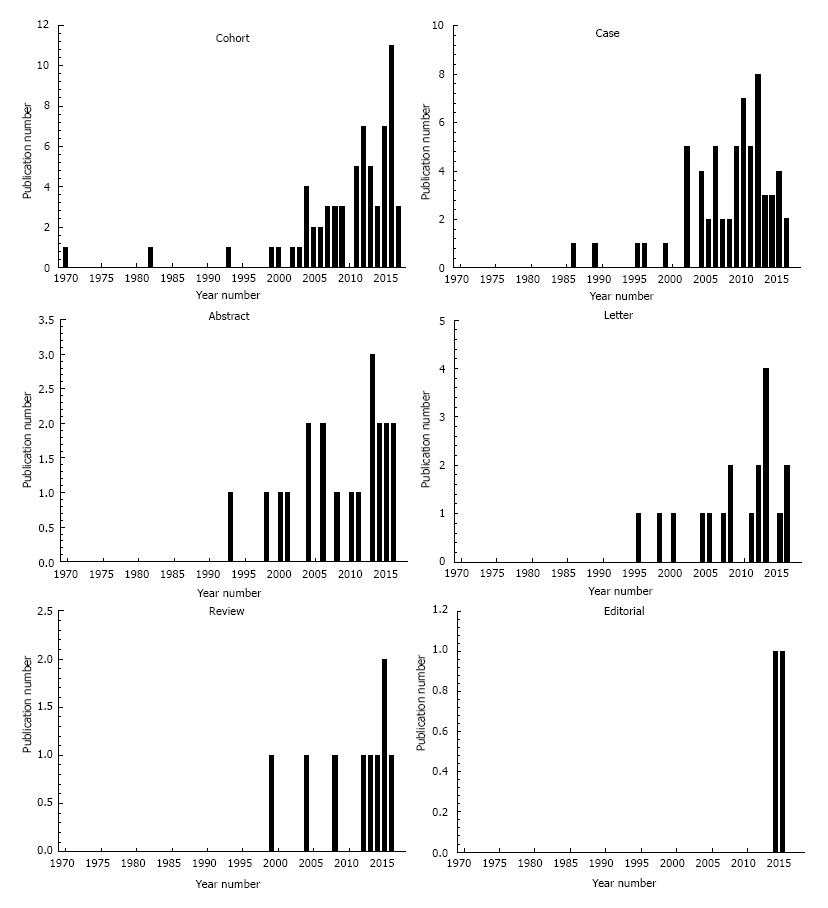
The number of studies for selected types that was published per year is shown in bar graph form in Figure 2. The results are shown for articles (cohort), case studies, abstracts, and letters, and review and editorial publications. Many of the CD/cardiovascular published studies were either articles or case studies. As for graphs of the total studies published that were shown in Figure 1, the graphs of individual published document types in Figure 2 begin to exhibit substantial increases about the year 2000. There were also a number of abstracts and case reports published in the late 1980s and 1990s, as is notable in the case reports graph in Figure 2. There are only a few review and editorial publications to date, but they are recent.
Based on all of this data, in Table 3 are provided, for each document type, the number of published studies for each topic, with the totals for all articles shown in the last row. Most of the published works are either articles (74) or case studies (62). There are also substantial numbers of letters (23) and abstracts (20). The totals for each term are given to the right in the table. The sum total, 190, is greater than the number of cited articles in this review, 180, because a particular citation could be used in more than one review topics section. Furthermore, several citations used in the Introduction were not cardiovascular studies and were not included in Table 3. A number of topics were particularly of interest for CD/cardiovascular publishing. These include papers on CD and cardiomyopathy (33 studies), thrombosis (27), cardiovascular risk (17), atherosclerosis (13), stroke (12), and arterial function and ischemic heart disease (11 each). In Table 4 are shown keyword terms and total number of studies, and median (range) study year of the journal. The median year for all of the studies is 2004 or later, except for the term “haemodynamics” (1998). Thus the possible connection between CD and “haemodynamics” tended to be investigated at earlier dates, as compared with other cardiovascular conditions.
| Keyword | Article | Review | Case | Letter | Abstract | Editorial | Total |
| Arterial function | 6 | 1 | 2 | 2 | 11 | ||
| Atherosclerosis | 4 | 1 | 5 | 3 | 13 | ||
| Angiogenesis | 6 | 1 | 7 | ||||
| Thrombosis | 4 | 2 | 15 | 5 | 1 | 27 | |
| Stroke | 2 | 2 | 7 | 1 | 12 | ||
| Hemorrhage | 7 | 3 | 10 | ||||
| Haemodynamics | 1 | 1 | 2 | 4 | |||
| Vascular - other | 2 | 3 | 5 | ||||
| Pericardial effusion | 2 | 1 | 1 | 4 | |||
| Myocarditis | 3 | 1 | 1 | 1 | 6 | ||
| Cardiomyopathy | 8 | 1 | 21 | 0 | 2 | 1 | 33 |
| Infarction | 4 | 1 | 5 | ||||
| Electromechanical | 6 | 1 | 7 | ||||
| Ischemic heart disease | 6 | 3 | 1 | 1 | 11 | ||
| Rhythm disturbances | 3 | 1 | 1 | 3 | 8 | ||
| Congenital heart defect | 3 | 1 | 4 | ||||
| Heart - other | 3 | 2 | 1 | 6 | |||
| Cardiovascular risk | 11 | 2 | 1 | 3 | 17 | ||
| Total | 74 | 9 | 62 | 23 | 20 | 2 | 190 |
| Category | Topic | Studies1 | Median year | Range |
| Vascular | Arterial function | 11 | 2014 | 2011-2016 |
| Vascular | Atherosclerosis | 13 | 2013 | 2008-2016 |
| Vascular | Angiogenesis | 7 | 2009 | 1970-2013 |
| Vascular | Thrombosis | 27 | 2007 | 1995-2016 |
| Vascular | Stroke | 12 | 2008 | 2001-2017 |
| Vascular | Hemorrhage | 10 | 2007 | 1997-2012 |
| Vascular | Haemodynamics | 4 | 1998 | 1993-2005 |
| Vascular | Other | 5 | 2004 | 1993-2013 |
| Heart | Pericardial effusion | 4 | 2008 | 2000-2014 |
| Heart | Myocarditis | 6 | 2004 | 2002-2012 |
| Heart | Cardiomyopathy | 33 | 2010 | 1986-2016 |
| Heart | Infarction | 5 | 2009 | 2008-2015 |
| Heart | Electromechanical | 7 | 2014 | 2008-2016 |
| Heart | Ischemic heart disease | 11 | 2014 | 2004-2016 |
| Heart | Rhythm disturbances | 8 | 2014 | 1989-2016 |
| Heart | Congenital heart defect | 4 | 2014 | 1982-2016 |
| Heart | Other | 6 | 2012 | 2004-2016 |
| Cardiovas cular disease | Risk factors | 17 | 2013 | 2007-2017 |
In this section, the study results for each keyword of Table 1 are combined into summary topics, to show the general consensus and trends for CD/cardiovascular publications. Thus the most pertinent information from all studies belonging to a particular cardiovascular term was collated by topic. The total number of studies (#) and median and range in years (median year) are shown for published studies concerning each topic. The terms are separated and noted as belonging to vascular or heart categories, followed by cardiovascular risk assessment.
Arterial function is of great concern in CD. Measurements to quantify alterations are made using echocardiography and pulse wave velocity[8,9]. In untreated CD, aortic function can deteriorate, and this deterioration is predictive of subclinical atherosclerosis and future cardiovascular events[8]. Aortic strain and distensibility tend to be significantly lower, and the aortic stiffness index significantly higher, in untreated CD patients vs controls[8,10]. CD patients are at increased risk for coronary artery disease[9], which may have a genetic association[11]. Occlusion of the brachiocephalic trunk and right and left common carotid artery has been noted in CD[12]. In adult CD patients lacking cardiovascular risk factors, abnormal homocysteine, erythrocyte sedimentation rate, C-reactive protein, and insulin levels may, along with inflammation, be contributive to arterial stiffening[9]. Spontaneous coronary artery dissections have been observed as a cause of acute myocardial infarction in CD patients[3]. There is also some support for an association between CD and cerebrovascular disease[13]. Correlation has been shown between restoration of the small intestinal villous atrophy and normalization of vascular parameters in gluten-abstinent CD patients[14]. Yet after onset of the GFD, the lack of a significant reduction in aortic elastic properties suggests that some risk of cardiovascular disease may persist[10]. Type 1 diabetic patients with early presence of micro- or macrovascular complications should always be screened for CD[15]. Type I diabetes mellitus and CD can coexist, and there is evidence that microvascular complications are more severe in patients with both conditions[16,17].
Atherosclerosis has been linked to myocardial infarction and ischemic stroke, with chronic inflammation being a likely pathogenic factor[18]. Untreated adults with CD are at increased risk of early atherosclerosis, as suggested by the presence of chronic inflammation, vascular impairment, unfavorable biochemical patterns[19-21], and relative lack of the classical risk factors. Carotid intima-media thickness values are significantly higher in patients with coexisting diabetes and CD as compared to those patients with diabetes or CD alone[16,22]. CD youth have also been associated with increased risk of developing early atherosclerosis[19]. They are also more likely to have greater mean low density lipoprotein (LDL) cholesterol and thicker carotid intima media as compared with controls, and their endothelium-dependent dilatation is decreased, all of which negatively affect vascular function[23,24]. The GFD enables a reduction in inflammatory parameters, oxidative stress, and insulin resistance, factors that when unchecked can lead to atherosclerosis[10,25]. Gluten avoidance followed by restoration of the intestinal villi to normal function is likely to revert cardiovascular dysfunction in less than 1 years’ time[2,20,21,23,24]. Areas with improved markers on a GFD include the common carotid arteries for intima media thickness, and the humeral artery for endothelium-dependent dilatation[26]. However, patients on the GFD often show weight gain and increase in triglyceride blood levels, which suggests a risk to atherogenicity[27], although alterations of other risk factors do not necessarily support this supposition[28]. CD patients should always be encouraged to choose a healthy GFD[27].
In untreated CD, the overall architecture of the small-bowel mucosal vasculature may be altered, leading to inhibition of angiogenesis[29]. On the GFD, the vasculature normalizes as compared to healthy subjects, in parallel to mucosal recovery, and mucosal autoantibody deposits diminish in the small intestine[29,30]. Autoantibody production in CD mainly targets against transglutaminase 2 (TG2)[29]. These autoantibodies are found in untreated CD patients’ serum[31], but also bound to extracellular TG2 below the epithelial basement membrane and around capillaries in the small intestinal mucosa, as well as in extra-intestinal organs. The autoantibodies can interfere with angiogenesis, including changes in transendothelial migration of lymphocytes[29,32,33], which is probably influenced by common genetic variants in angiogenesis-related genes[34]. In vitro, CD autoantibodies reduce endothelial branching, increase endothelial permeability to macromolecules and lymphocytes, and enhance lymphocyte adhesion to the endothelium[29,35]. Ultrastructural alterations of the small blood vessels embedded in the subepithelial connective tissue of the jejunal mucosa are most severe in CD patients not on the GFD. Similar changes occur when gluten is administered to pediatric CD patients previously on a GFD, and are one of the earliest pathological changes seen in the biopsy material[35].
Thrombotic events are increased in CD[36], can be recurrent[37], and may be present at multiple locations[38] which are observable via Doppler ultrasonographic examination[39]. Thrombophilia may result from hyperhomocysteinemia and deficiencies in protein S, folate, and vitamin B2[40-42]. The thrombotic events in CD may also result from dehydration due to diarrhea[43]. Cerebral venous sinus thrombosis can occur in CD patients[40,44-46], even in absence of gastrointestinal symptoms[45], but can resolve with symptomatic treatment[40]. Venous thrombosis can be a sequela of undetected CD[42,47-54], and may result in thromboembolic events[48,55]. CD may be accompanied by portal vein thrombosis[56,57], and mesenteric[58] or splenic[59] vein thrombosis may present in occult or subclinical celiac disease[58]. There is an increased risk of developing venous thromboembolism from chronic inflammation and vitamin deficiency in CD[44,60,61]. On a GFD, there is favorable evolution of young CD patients with thrombosis[62]. CD should be considered in young patients with thrombosis, especially if the event occurs in an unusual location[62].
Patients with CD have been found to be at an increased risk for stroke, which can persist after onset of the GFD[63,64]. Stroke events can be recurrent[65]. Cerebral infarction and transient hemiplegia may also present in untreated asymptomatic CD patients[66]. CD should be considered as a possible etiology for stroke cases of unknown cause, particularly in youth, whether gastrointestinal manifestations are evident or not[67]. The pathogenesis of stroke in CD youth may involve vitamin B12 deficiency[68] and possibly hyperhomocysteinemia, which may be secondary to folic acid deficiency[69], cerebral arterial vasculopathy, and antiphospholipid syndrome, a secondary autoimmune disorder[70-72]. Children with recurrent acute ischemic stroke should be screened for CD[73]. Because CD is a potentially treatable cause of cerebral vasculopathy and stroke[74], serology-specifically anti TTG antibodies should be included in the evaluation for cryptogenic stroke in childhood, even in the absence of typical gastrointestinal symptoms[72].
When unresponsive CD is treated with corticosteroids and immunosuppression therapy, it can be complicated by the presence of small intestinal lymphoma, and result in hemorrhage[75,76]. During hemorrhage, coagulopathy can occur, which is attributable to vitamin K deficiency associated with malabsorption of multiple fat soluble vitamins in these patients[75]. The immune response to CD, triggered by gluten, can lead to deposition of circulating immune complexes on the membrane of alveolar capillaries, resulting in pulmonary hemosiderosis[77]. Lane-Hamilton syndrome refers to the co-occurrence of idiopathic pulmonary hemosiderosis and CD[78]. Idiopathic pulmonary hemosiderosis is severe and potentially fatal, and is characterized by recurrent episodes of alveolar hemorrhage, hemoptysis, and anemia, and can share a common immune pathway with CD[79]. Left untreated, it can lead to poor prognosis, with progression to pulmonary fibrosis and chronic respiratory limitation[80]. In patients with diffuse alveolar hemorrhage, even in the absence of gastrointestinal symptoms, screening for CD should be done using anti-transglutaminase antibodies[79,81]. If CD is found, the GFD helps control symptoms, enables a reduction of immunosuppressive treatment, and improves clinical course[79]. Improvement of the co-occurrence of CD and pulmonary hemosiderosis over a period of months is found when patients are placed on the GFD[82,83]. Thus patients with pulmonary hemosiderosis should always be screened for CD[80,82]. Patients with hereditary hemorrhagic telangiectasia with unexplained iron malabsorption should also be screened for CD[84].
Alteration of blood flow is an important issue. The pathophysiological changes in the small bowel mucosa during the active phase of CD can induce haemodynamic changes[85] including an abnormal splanchnic circulation[86], and splenic vein obstruction may be present[47]. The postprandial mesenteric blood flow can be significantly increased and delayed in time[86]. A hyperdynamic mesenteric circulation and higher peak systolic velocity of the superior mesenteric artery is often manifested in untreated CD patients as compared with healthy controls and treated celiac patients[85,87]. Treatment with the GFD can improve haemodynamics[85,87].
Several other vascular maladies have been noted to occur in conjunction with CD. The combination of CD, epilepsy, and cerebral calcification is a rare condition known as CEC syndrome[88]. Folate malabsorption is a suggested mechanism, because cerebral calcification can be seen in other conditions related to folate deficiency[88]. Membranous obstruction of the inferior vena cava can also occur[89]. In patients with hyperhomocysteinaemia and sub-clinical CD, endothelial dysfunction is associated with increased systemic vascular resistance that can lead to a reversible form of hypertension[90]. CD adults tend to have a lower prevalence of hypertension and hypercholesterolaemia as compared with the general population[91]. However in patients with hypertension, CD, and hyperhomocysteinaemia (via malabsorption of essential cofactors), CD treatment can improve blood pressure control[90]. Covert hemoptysis may be responsible for disproportionately severe anemia in CD patients[92].
The heart itself can be greatly affected in CD patients. Echocardiography has been used to show that there is a higher incidence of pericardial effusion in CD[93]. In adults, this phenomenon can be asymptomatic[94]. The predisposing factors for pericardial effusion include vessel dysfunction in the presence of high antibody titer, selenium deficiency, and viral infection due to reduced immunological competence, in conjunction with a diminished ability to eliminate toxic free radicals[93,95]. After onset of the GFD and with iron supplement, pericardial effusion, along with peripheral edema and fatigue, decreases[94,96]. Presence of left ventricle dilation, suggesting an initial phase of heart damage, is reversible on the GFD[96]. CD children may also have pericardial fluid, and show no difference compared to those lacking effusion with respect to ECG, chest X-ray, blood cell count, serum enzymes, serum protein, and iron levels[95]. Pericardial effusion is reversible in children when they are treated with a GFD[95,96]. Thus pediatric cardiologists should be alerted to the possibility that mild left ventricular enlargement can be caused by CD[96].
Autoimmune myocarditis may be a complication of CD[97]. Biopsy-proven granulocytic myocarditis of unknown etiology can be found in the setting of silent CD[98]. Progressive heart failure may accompany viral myocarditis in untreated CD; patient condition can improve following standard heart failure treatment and GFD[99]. A strong fluorescence around heart muscle fibers has been noted in untreated CD patients but not in patients on the diet or in controls[100]. This suggests that an autoimmune process toward antigenic components of the myocardium can occur in untreated CD, and lead to cardiac tissue injury, resulting in myocarditis[101]. In these patients, immunosuppression and GFD are often effective therapeutic options[101]. It is thus highly important to screen for CD in patients with these conditions to avoid progression and clinical deterioration[102]. It has been found that the CD prevalence in children with myocarditis is greater than in children without myocarditis[102], who should therefore also receive CD screening.
Cardiomyopathy associated with CD is a serious and potentially lethal condition which requires a multidisciplinary approach involving both a gastroenterologist and a cardiologist[103-106]. There is a higher prevalence of CD in patients with dilated cardiomyopathy[107-109], idiopathic cardiomyopathy[110-113] and ischaemic or valvular cardiomyopathy[110]. There is also a higher prevalence of CD in the relatives of patients with sporadic and inherited dilated cardiomyopathy[114]. Severely dilated left ventricle, concomitant left ventricular dysfunction, very low ejection fraction, pulmonary hemosiderosis, heart block, and/or heart failure have been reported in cardiomyopathy patients with CD[112,115-120]. Severe progressive dilated cardiomyopathy, requiring heart transplantation, can occur[121]. Dilated cardiomyopathy in CD may be accompanied by congestive heart failure and is also becoming increasingly recognized in the pediatric population[122,123]. These children may also present with acute onset congestive heart failure, as well as severe left ventricular systolic dysfunction[123]. Upper limb venous thrombosis and recurrent haemoptysis secondary to pulmonary haemosiderosis may be present[123]. Cirrhotic cardiomyopathy without gastrointestinal symptoms has also been found in pediatric patients[124].
Although a serious condition, the precise cause-and-effect relationship between CD and cardiomyopathy when they occur in tandem is currently unknown[125]. Dilated cardiomyopathy may evolve due to carnitine deficiency[126,127], which is related to CD, but may also develop after onset of the GFD, particularly in patients lacking carnitine supplementation[126]. Idiopathic dilated cardiomyopathy may have an autoimmune mechanism[110,128]. The tTG-positive serology in relatives with echocardiographic abnormalities suggests that immune-mediated mechanisms are at work in subsets of these patients and their families[106]. In individuals with idiopathic congestive cardiomyopathy and CD, ultrastructural and electrophoretic examination of myocardial samples shows a selective loss of actin, and electron microscopy reveals characteristic alterations of enterocyte microvilli[129]. Hence there can be an involvement of the microfilament system in both the myocardial sarcomeres and the enterocytes of these patients[129]. Ischemic cardiomyopathy can occur due to an accelerated atherosclerosis when chronic inflammation is present in CD[130].
Compliance with a GFD is mandatory if patients are to avoid progression of their cardiomyopathy[105,131]. The GFD has been shown to have a beneficial effect on cardiac performance in CD patients with dilated cardiomyopathy[132]. After start of the GFD, abnormal left ventricular dimensions, and diminished cardiac function, including decreased ejection fraction, can improve markedly and may even be completely reversible[103,108,118,128,133]. After two years on a GFD, patients presenting with dilated cardiomyopathy associated with CD show progressive increase in mean serum carnitine levels as compared to values observed prior to the diet[127]. Children with CD and dilated cardiomyopathy also have improved cardiac function once adherent to the GFD[109]. In CD patients on the GFD, there is no association with later onset of myocarditis, cardiomyopathy or pericarditis[134].
Screening for CD in patients with dilated cardiomyopathy, pulmonary haemosiderosis, and iron deficiency anemia in the absence of known etiology is advisable regardless of the intestinal symptoms[105,108,116,123,133]. Serologic tests for IgA-EmA and tissue transglutaminase antibodies should be used to screen for CD[106]. Co-morbidities including iron-deficiency anemia in patients with dilated cardiomyopathy should arouse suspicion of CD[117]. All patients diagnosed with cardiomyopathy and CD should be offered an endomyocardial biopsy for better histological and diagnostic definition[128]. It is beneficial to assess CD in children with dilated cardiomyopathy in the absence of known etiologies[104,135].
Acute myocardial infarction with ST-elevation and spontaneous coronary artery dissection can occur in young patients with CD[3,136]. However, chronic hypocalcemia in untreated CD patients due to poor absorption of minerals can result in electrocardiographic changes that mimic acute myocardial infarction[120]. In CD, there is a higher all-cause mortality one year post-myocardial infarction as compared with the general population[20]. Mesenteric infarction has also occurred as a consequence of CD, and clinicians should be aware of this possibility[137].
Measurement of electromechanical parameters is beneficial to determine the degree of cardiac involvement in CD[138]. Atrial conduction delays are significantly higher in untreated CD as compared with healthy individuals, and may lead to atrial fibrillation[139]. Measurement of atrial electromechanical delay parameters might therefore be useful to predict atrial fibrillation risk[139]. Statistically significant differences in left ventricular function as assessed by echocardiography imaging are found in CD patients vs controls[140]. Patients with CD can have impaired diastolic and systolic function as measured by tissue Doppler echocardiography[141]. Mitral valve prolapse[142] and subclinical myocardial dysfunction of both ventricles[143] can be present in both the pediatric and adult CD population. In children, significantly lower contractility indices and higher left ventricular dimensions are evident as compared with controls[144]. On the GFD, valve regurgitations resolve, and echocardiographic parameters significantly improve[144].
There is an increased risk of ischemic heart disease and higher cardiovascular mortality in CD[145,146] despite the lack of traditional risk factors[26,147] including blood pressure, body mass index, serum cholesterol, lipids, exercise, and smoking[4,148]. First-degree relatives of CD patients are also at an increased risk of ischemic heart disease, but the excess risk is slight[149]. CD and ischemic heart disease may share a common underlying link, rather than a cause-and-effect relationship[18,150]. The underlying association between CD and ischemic heart disease may be chronic inflammation, a major risk factor in the general population; however, potential confounders may also be involved[18,146,148]. After onset of the GFD, persistent villous atrophy detected during follow-up biopsy was not associated with increased risk of ischemic heart disease[151,152].
Compared to controls, untreated CD patients have increased P-wave dispersion and higher interatrial, intra-left atrial, and intra-right atrial conduction delay[153]. Tp-e interval, Tp-e/QT and Tp-e/QTc ratios are also increased in CD[154]. There is a higher prevalence of atrial fibrillation among CD patients[153,155]. Atrial fibrillation, when it occurs, is associated with an increased risk of ischaemic stroke and heart failure[156]. The chronic inflammation that can occur in untreated CD is a recognized risk factor for atrial fibrillation[155,156]. CD patients have slower atrial electrical conduction, which may also increase the risk of atrial fibrillation[153]. However, persistent villous atrophy on follow-up biopsy was not associated with any increased risk of atrial fibrillation[151,152]. It has been reported that CD patients with pulmonary hemosiderosis can develop infranodal heart block, necessitating implantation of a pacemaker[77,157]. These patients may lack digestive manifestations but have iron deficiency and vitamin deficiency anemia[77]. Rhythm alterations in CD can thus result from other pathogenic mechanisms including electrolyte disturbances caused by malabsorption, which can normalize on the GFD[77]. Patients with rhythm disturbances and chronic anemia of unclear origin should therefore be tested for CD[77].
CD patients are likely to more commonly have atrial septal defect as compared to controls[158]. Screening for CD in children with congenital heart defect is recommended, since serum TTG IgA levels are significantly higher in patients with congenital heart defect vs control children[159]. Down syndrome patients with congenital heart defect have higher CD prevalence as compared to patients without congenital heart defect, and CD prevalence in Down syndrome patients is higher than in controls[160]. In children with congenital heart disease and CD, growth improves on a GFD[161].
Chronic hypocalcemia, which may occur in untreated CD due to malabsorption, has been associated with reversible cardiac dysfunction[120]. Untreated CD children tend to show an imbalance of cardiac sympathetic and parasympathetic activity due to enhanced sympathetic tone[162]. This imbalance is still detected after a six months period of GFD[162]. This suggests the presence of a subclinical autonomic nervous system dysfunction[162]. There is a higher prevalence of CD in patients with Postural Orthostatic Tachycardia Syndrome (POTS)[163]; thus these patients should also be screened for CD[164]. Left ventricular hypertrabeculation/noncompaction may be associated with CD[165]. Subclinical systolic dysfunction of the left ventricle may be present in CD children[166].
Cardiovascular disease (CVD) as a whole has many etiologies and is the leading cause of death in developed countries[167,168]. There is a modestly increased risk of CVD in CD patients[63]. Both male and female CD patients may have higher estimated risk for CVD as compared to controls[169,170]. However, CVD risk factors conferred by CD have not been well-defined[171]. This has led to conflicting evidence as to whether CD patients actually have increased baseline risk[172]. CD patients are susceptible to increased platelet activation and increased mean platelet volume and RDW values, factors that contribute to increased risk[173,174]. Using the Framingham Heart Study (FRS) 10-year general CVD risk score, lower values were found among CD patients as compared with controls, which may be due to a lesser body mass index and reduced tobacco use among CD patients[171]. Risk factors other than those measured by the FRS may be observed as increased risk of CVD in CD patients[171]. There can also be a positive association between CD and CVD risk due to ascertainment bias[175]. In CD children, risk factors can be frequently observed as compared with healthy subjects[18]. Overall, certain CVD risk factors have been found to be higher in CD youth as compared with the general population, although neither blood pressure nor overweight and obesity rates were increased[176]. Youth with type 1 diabetes and CD had lower high density lipoprotein (HDL) cholesterol, increasing CVD risk, as compared with non CD patients[177].
The lack of a uniform set of risk factors can influence whether CVD risk is affected by a GFD[172]. In actuality, both risk and protective factors for CVD are likely to be present in CD, at baseline and also on a GFD[172]. Modifiable risk factors for CVD can include body mass index and cholesterol, which have been shown to increase on the GFD[168]. In CD individuals with type 1 diabetes on a GFD, improvement in HDL-cholesterol, and a lower resting heart rate, has been demonstrated as compared with those CD patients without diabetes[178]. On a GFD, the lipid profile of CD patients can also improve[17]. At one year on a GFD, waist circumference may increase, but without significant rise in total or LDL cholesterol[173,174]. The GFD should therefore ideally go beyond gluten exclusion and include body weight control and high quality nutrients[172]. A relatively high proportion of CD children on the GFD had one or more CVD risk factors[179]. The most common CVD risk factors are high fasting triglycerides, elevated blood pressure, and high LDL cholesterol concentrations[179]. Insulin resistance is also found, underscoring the need for CVD screening and dietary counseling targeting the pediatric CD population[179]. Screening for CD and monitoring of HDL cholesterol is recommended in youth with type 1 diabetes[177]. CVD risk factors also include metabolic disorders caused by malabsorption in pediatric CD patients[180]. Hence timely correction of water and electrolyte imbalance, and administration of cardiometabolic therapy, is necessary[180].
Published studies pertaining to the connection between cardiovascular conditions and CD began in the late 1960’s, consisting of a few studies each year, and was followed by a substantial increase beginning about the year 2000. Many of the published studies are either articles (including cohort and case control studies) or case studies consisting of one to three patients. Often, as might be expected, a number of case studies appeared in the literature prior to the cohort studies. Based on the evidence presented in these papers, it is apparent that cardiovascular involvement in CD is a real phenomenon and that there are many manifestations, owing to the multifaceted, systemic physiological changes that can occur in CD. A number of the cardiovascular issues that can occur in untreated CD patients, will resolve on a GFD, often in conjunction with healing of the small intestinal villi. Cardiomyopathy is the most frequently documented cardiovascular condition observed in conjunction with CD, and seems to mostly or completely resolve with appropriate treatment, including a GFD. However, if CD is left unrecognized until a late stage, damage done to the heart may not be entirely reversible. Similarly, to the present time there has been substantial documentation of a number of other cardiovascular conditions found in conjunction with untreated CD including thrombosis and thromboembolism, ischemic heart disease, stroke, and arrhythmia. There has also been significant investigation of CD and risk of cardiovascular disease. On this topic, a current problem is that there is no uniform set of cardiovascular risk factors used for analysis. Future studies should settle the question of how to best treat these co-occurring conditions, and to determine if other cardiovascular manifestations of CD are common phenomena.
Manuscript source: Invited manuscript
Specialty type: Cardiac and cardiovascular systems
Country of origin: United States
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Balaban YH S- Editor: Qi Y L- Editor: A E- Editor: Lu YJ
| 1. | Lodha A, Haran M, Hollander G, Frankel R, Shani J. Celiac disease associated with dilated cardiomyopathy. South Med J. 2009;102:1052-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 2. | Rybak A, Cukrowska B, Socha J, Socha P. Long term follow up of celiac disease-is atherosclerosis a problem? Nutrients. 2014;6:2718-2729. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 3. | Bayar N, Çağırcı G, Üreyen ÇM, Kuş G, Küçükseymen S, Arslan Ş. The relationship between spontaneous multi-vessel coronary artery dissection and celiac disease. Korean Circ J. 2015;45:242-244. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 4. | Wang I, Hopper I. Celiac disease and drug absorption: implications for cardiovascular therapeutics. Cardiovasc Ther. 2014;32:253-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 5. | Ciaccio EJ, Bhagat G, Lewis SK, Green PH. Trends in celiac disease research. Comput Biol Med. 2015;65:369-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 6. | Ciaccio EJ, Bhagat G, Lewis SK, Green PH. Trends in gluten research and its relationship to autoimmune and allergic diseases. Informat Med Unlocked. 2016;3:7-14. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 7. | Ciaccio EJ, Biviano AB, Iyer V, Garan H. Trends in quantitative methods used for atrial fibrillation and ventricular tachycardia analyses. Informat Med Unlocked. 2017;6:12-27. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Sari C, Ertem AG, Sari S, Efe TH, Keles T, Durmaz T, Ertem S, Bozkurt E. Impaired aortic function in patients with coeliac disease. Kardiol Pol. 2015;73:1108-1113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Korkmaz H, Sozen M, Kebapcilar L. Increased arterial stiffness and its relationship with inflammation, insulin, and insulin resistance in celiac disease. Eur J Gastroenterol Hepatol. 2015;27:1193-1199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 10. | Bayar N, Çekin AH, Arslan Ş, Çağırcı G, Küçükseymen S, Çay S, Harmandar FA, Yeşil B. Assessment of Aortic Elasticity in Patients with Celiac Disease. Korean Circ J. 2016;46:239-245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 11. | Jansen H, Willenborg C, Schlesinger S, Ferrario PG, König IR, Erdmann J, Samani NJ, Lieb W, Schunkert H. Genetic variants associated with celiac disease and the risk for coronary artery disease. Mol Genet Genomics. 2015;290:1911-1917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Altabas K, Crne N, Franjić BD, Pavlović D, Josipović J. [Peritoneal dialysis in a patient with right hemiparesis, lupus nephritis, significant insufficiency of arteries of aortic arch and celiac disease: case report]. Acta Med Croatica. 2012;66 Suppl 2:72-75. [PubMed] |
| 13. | Heikkilä K, Koskinen OA, Agarwal A, Tikkinen KA, Mäki M, Kaukinen K. Associations of coeliac disease with coronary heart disease and cerebrovascular disease: A systematic review and meta-analysis. Nutr Metab Cardiovasc Dis. 2015;25:816-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | De Marchi S, Chiarioni G, Prior M, Arosio E. Commentary: coeliac disease and atherosclerosis--hand in hand? Authors’ reply. Aliment Pharmacol Ther. 2013;38:550-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 15. | Pitocco D, Zaccardi F, Martini F, Giubilato S, Liuzzo G, Crea F, Ghirlanda G. The cardiovascular relevance of celiac disease. Diabetes Care. 2012;35:e20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Pitocco D, Giubilato S, Martini F, Zaccardi F, Pazzano V, Manto A, Cammarota G, Di Stasio E, Pedicino D, Liuzzo G. Combined atherogenic effects of celiac disease and type 1 diabetes mellitus. Atherosclerosis. 2011;217:531-535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 17. | Leeds JS, Hopper AD, Hadjivassiliou M, Tesfaye S, Sanders DS. High prevalence of microvascular complications in adults with type 1 diabetes and newly diagnosed celiac disease. Diabetes Care. 2011;34:2158-2163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 18. | Mormile R. Celiac Disease and Ischemic Heart Disease: What is the Link? J Clin Cell Immunol. 2013;4: 2. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 19. | Mormile R. Celiac disease and atherosclerosis: An immunologic puzzle to be solved? Immunol Lett. 2016;180:75-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Rigoni A, De Marchi S, Prior M, Chiarioni G, Arosio E. 100 young adults with celiac disease may be at increased risk of early atherosclerosis. Nutrition, Metabolism Cardiovascular Dis. 2008;18:S58. [DOI] [Full Text] |
| 21. | De Marchi S, Chiarioni G, Prior M, Arosio E. Young adults with coeliac disease may be at increased risk of early atherosclerosis. Aliment Pharmacol Ther. 2013;38:162-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 22. | Martini F, Pitocco D, Zaccardi F, Giubilato S, Pallano V, Manto A, Cammarota G, Liuzzo G, Crea F, Ghirlanda G. Early Detection of cardiovascular disease in patients with associated type 1 diabetes mellitus and celiac disease. 1701 N Beauregard St, Alexandria, VA 22311-1717. USA: Amer Diabetes Assoc 2011; . |
| 23. | Valitutti F, Trovato CM, Barbato M, Cucchiara S. Letter: Atherosclerosis and coeliac disease – Another feature of the changing paradigm? Aliment Pharmacol Therap. 2013;38:559. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 24. | Emilsson L, Carlsson R, James S, Ludvigsson JF. Letter: Coeliac disease and ischaemic heart disease - a true additional risk factor? Authors’ reply. Aliment Pharmacol Therap. 2013;37:1118. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 25. | Aguilar EC, Soares FL, Santos LC, Capettini LS, Teixeira LG, Lemos VS, Alvarez-Leite JI. SUN-LB037: Gluten-free diet reduces cardiovascular risk factors and atherosclerosis development in APOE KO mice. Clin Nutr. 2015;34:S248. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 26. | Demir AM, Kuloğlu Z, Yaman A, Fitöz S, Nergizoğlu G, Kansu A. Carotid intima-media thickness and arterial stiffness as early markers of atherosclerosis in pediatric celiac disease. Turk J Pediatr. 2016;58:172-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Bianchi PI, Biagi F, Borsotti E, Marchese A, Vattiato C, Ferretti F, Balduzzi D, Trotta L, Schiepatti A, Corazza GR. P. 05.13 Peripheral artery disease in coeliac disease. Dig Liver Dis. 2013;45:S123. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 28. | Zanini B, Mazzoncini E, Lanzarotto F, Ricci C, Cesana BM, Villanacci V, Lanzini A. Impact of gluten-free diet on cardiovascular risk factors. A retrospective analysis in a large cohort of coeliac patients. Dig Liver Dis. 2013;45:810-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 29. | Myrsky E, Caja S, Simon-Vecsei Z, Korponay-Szabo IR, Nadalutti C, Collighan R, Mongeot A, Griffin M, Mäki M, Kaukinen K. Celiac disease IgA modulates vascular permeability in vitro through the activity of transglutaminase 2 and RhoA. Cell Mol Life Sci. 2009;66:3375-3385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Myrsky E, Syrjänen M, Korponay-Szabo IR, Mäki M, Kaukinen K, Lindfors K. Altered small-bowel mucosal vascular network in untreated coeliac disease. Scand J Gastroenterol. 2009;44:162-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 31. | Myrsky E, Syrjänen M, Kaukinen K, Korponay-Szabo I, Maki M, Lindfors K. Celiac disease IGA class autoantibodies inhibit the recruitment of vascular smooth muscle cells to capillary walls. J Pediatr Gastroenterol Nutr. 2006;42:E1. |
| 32. | Myrsky E, Kaukinen K, Syrjänen M, Korponay-Szabó IR, Mäki M, Lindfors K. Coeliac disease-specific autoantibodies targeted against transglutaminase 2 disturb angiogenesis. Clin Exp Immunol. 2008;152:111-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 66] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 33. | Kalliokoski S, Sulic AM, Korponay-Szabó IR, Szondy Z, Frias R, Perez MA, Martucciello S, Roivainen A, Pelliniemi LJ, Esposito C. Celiac Disease-Specific TG2-Targeted Autoantibodies Inhibit Angiogenesis Ex Vivo and In Vivo in Mice by Interfering with Endothelial Cell Dynamics. PLoS One. 2013;8:e65887. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 34. | Castellanos-Rubio A, Caja S, Irastorza I, Fernandez-Jimenez N, Plaza-Izurieta L, Vitoria JC, Maki M, Lindfors K, Bilbao JR. Angiogenesis-related gene expression analysis in celiac disease. Autoimmunity. 2012;45:264-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Shiner M. Mucosal vascular changes in coeliac disease and in idiopathic steatorrhoea. Micron. 1970;2:7-18. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 36. | Boucelma M, Saadi M, Boukrara H, Bensalah D, Hakem D, Berrah A. [Association of celiac disease and cerebral venous thrombosis: report of two cases]. J Mal Vasc. 2013;38:47-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 37. | Beyrouti R, Mansour M, Kacem A, Derbali H, Mrissa R. Recurrent cerebral venous thrombosis revealing celiac disease: an exceptional case report. Acta Neurol Belg. 2017;117:341-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 38. | Poulin W, Gaertner S, Cordeanu EM, Mirea C, Andrès E, Stephan D. Stroke revealing celiac disease associated with multiple arterial thrombotic locations. Presse Med. 2015;44:537-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 39. | Çelik G, Çiledağ A, Keskin O, Ensari A, Karabörk A, Kaya A. Sarcoidosis, Celiac disease and deep venous thrombosis: A rare association. Balkan Med J. 2011;28:450-452. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 40. | Bahloul M, Chaari A, Khlaf-Bouaziz N, Kallel H, Chaari L, Ben Hamida C, Chelly H, Rekik N, Bouaziz M. [Celiac disease, cerebral venous thrombosis and protein S deficiency, a fortuitous association?]. J Mal Vasc. 2005;30:228-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 41. | Kallel L, Matri S, Karoui S, Fekih M, Boubaker J, Filali A. Deep venous thrombosis related to protein S deficiency revealing celiac disease. Am J Gastroenterol. 2009;104:256-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 42. | Baryshnikov EN, Krums LM, Vorob’eva NN, Parfenov AI. [Lower extremity deep vein thrombosis associated with gluten-sensitivity celiac disease]. Ter Arkh. 2010;82:52-54. [PubMed] |
| 43. | Lee ES, Pulido JS. Nonischemic central retinal vein occlusion associated with celiac disease. Mayo Clin Proc. 2005;80:157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 44. | Johannesdottir SA, Erichsen R, Horváth-Puhó E, Schmidt M, Sørensen HT. Coeliac disease and risk of venous thromboembolism: a nationwide population-based case-control study. Br J Haematol. 2012;157:499-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 45. | Doğan M, Peker E, Akbayram S, Bektas MS, Basaranoglu M, Cesur Y, Caksen H. Cerebral venous sinus thrombosis in 2 children with celiac disease. Clin Appl Thromb Hemost. 2011;17:466-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 46. | Grover PJ, Jayaram R, Madder H. Management of cerebral venous thrombosis in a patient with Lane-Hamilton syndrome and coeliac disease, epilepsy and cerebral calcification syndrome. Br J Neurosurg. 2010;24:684-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 47. | Ghoshal UC, Saraswat VA, Yachha SK. Association of splenic vein obstruction and coeliac disease in an Indian patient. J Hepatol. 1995;23:358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 48. | Beyan E, Pamukcuoglu M, Beyan C. Deep vein thrombosis associated with celiac disease. Bratisl Lek Listy. 2009;110:263-264. [PubMed] |
| 49. | Grigg AP. Deep venous thrombosis as the presenting feature in a patient with coeliac disease and homocysteinaemia. Aust N Z J Med. 1999;29:566-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 50. | Casella G, Baldini V, Perego D, Brambills M, Fraterrigo RT. A rare association between inflammatory bowel disease (IBD), coeliac disease, membranous glomerulonephritis and leg venous thrombosis associated to heterozygosis for V Leiden factor. Dige Liver Dis. 2000;32:A44. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 51. | Casella G, Perego D, Baldini V, Monti C, Crippa S, Buda CA. A rare association between ulcerative colitis (UC), celiac disease (CD), membranous glomerulonephritis, leg venous thrombosis, and heterozygosity for factor V Leiden. J Gastroenterol. 2002;37:761-762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 52. | Hida M, Erreimi N, Ettair S, Mouane N, Bouchta F. Associated celiac disease and venous thrombosis. Arch Pediatr. 2000;7:215-216. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 53. | Mezalek ZT, Habiba BA, Hicham H, Houda B, Mouna M, Mohamed A, Mohamed A. C0396 Venous thrombosis revealing celiac disease. Three cases. Throm Res. 2012;130:S154-S155. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 54. | Kremer Hovinga JA, Baerlocher G, Wuillemin WA, Solenthaler M. [Deep venous thrombosis of the leg in acquired thrombophilia--hyperhomocysteinemia as a sequela of undetected celiac disease]. Ther Umsch. 1999;56:519-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 55. | Ludvigsson JF, Welander A, Lassila R, Ekbom A, Montgomery SM. Risk of thromboembolism in 14,000 individuals with coeliac disease. Br J Haematol. 2007;139:121-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 56. | Karoui S, Sfar S, Kallel M, Boubaker J, Makni S, Filali A. [Antiphospholipid syndrome revealed by portal vein thrombosis in a patient with celiac disease]. Rev Med Interne. 2004;25:471-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 57. | Zenjari T, Boruchowicz A, Desreumaux P, Laberenne E, Cortot A, Colombel JF. Association of coeliac disease and portal venous thrombosis. Gastroenterol Clin Biol. 1995;19:953-954. [PubMed] |
| 58. | Azzam NA, Al Ashgar H, Dababo M, Al Kahtani N, Shahid M. Mesentric vein thrombosis as a presentation of subclinical celiac disease. Ann Saudi Med. 2006;26:471-473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 59. | Khanna S, Chaudhary D, Kumar P, Mazumdar S. Occult celiac disease presenting as splenic vein thrombosis. Indian J Gastroenterol. 2008;27:38-39. [PubMed] |
| 60. | Ungprasert P, Wijarnpreecha K, Tanratana P. Risk of venous thromboembolism in patients with celiac disease: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2016;31:1240-1245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 61. | Gabrielli M, Santoliquido A, Santarelli L, Fini L, Andrisani MC, Pola P, Gasbarrini G, Gasbarrini A. 6 P Venous thromboembolism, hyperhomocysteinemia and silent coeliac disease: A case report. Digestive and Liver Disease. 2002;34:A27. [DOI] [Full Text] |
| 62. | Zoubeidi H, Ben Salem T, Ben Ghorbel I, Houman MH. Central retinal vein occlusion revealing celiac disease. Eur J Case Reports Int Med. 2016;3. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 63. | Emilsson L, Lebwohl B, Sundström J, Ludvigsson JF. Cardiovascular disease in patients with coeliac disease: A systematic review and meta-analysis. Dig Liver Dis. 2015;47:847-852. [RCA] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 46] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 64. | Ludvigsson JF, West J, Card T, Appelros P. Risk of stroke in 28,000 patients with celiac disease: A nationwide cohort study in Sweden. J Stroke Cerebrovascular Dis. 2012;21:860-867. [RCA] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 65. | Ozge A, Karakelle A, Kaleağasi H. Celiac disease associated with recurrent stroke: a coincidence or cerebral vasculitis? Eur J Neurol. 2001;8:373-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 66. | Millichap JG. Celiac Disease and Stroke. Pediatr Neurol Briefs. 2004;18:64. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 67. | El Moutawakil B, Chourkani N, Sibai M, Moutaouakil F, Rafai M, Bourezgui M, Slassi I. [Celiac disease and ischemic stroke]. Rev Neurol (Paris). 2009;165:962-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 68. | Rachid B, Zouhayr S, Chtaou N, Messouak O, Belahsen F. [Ischemic stroke revealing celiac disease]. Pan Afr Med J. 2010;5:2. [PubMed] |
| 69. | Gefel D, Doncheva M, Ben-Valid E, el Wahab-Daraushe A, Lugassy G, Sela BA. Recurrent stroke in a young patient with celiac disease and hyperhomocysteinemia. Isr Med Assoc J. 2002;4:222-223. [PubMed] |
| 70. | Ajello A, Vitullo G. Stroke and coeliac disease: a new face for an old lady. Italian J Pediatr. 2006;32:259. |
| 71. | Audia S, Duchêne C, Samson M, Muller G, Bielefeld P, Ricolfi F, Giroud M, Besancenot JF. [Stroke in young adults with celiac disease]. Rev Med Interne. 2008;29:228-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 72. | Goodwin FC, Beattie RM, Millar J, Kirkham FJ. Celiac disease and childhood stroke. Pediatr Neurol. 2004;31:139-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 73. | Balci O, Sezer T. The Prevalence of Celiac Disease in Children With Arterial Ischemic Stroke. J Pediatr Hematol Oncol. 2017;39:46-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 74. | Slimani N, Hakem D, Mansouri B, Berrah A. Ischemic stroke revealing a celiac disease: A case report and review of the literature. J Neurol Sci. 2013;333:e248. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 75. | Chen CS, Cumbler EU, Triebling AT. Coagulopathy due to celiac disease presenting as intramuscular hemorrhage. J Gen Intern Med. 2007;22:1608-1612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 76. | Gheorghe L, Popescu I, Gheorghe C, Aposteanu G, Popescu C, Oproiu A. Fatal intestinal hemorrhage complicating ileal lymphoma after cyclosporine for unresponsive celiac disease. Hepato-Gastroenterology. 1996;44:1342-1345. [PubMed] |
| 77. | Callejas Rubio JL, Ortego N, Díez-Ruiz A, Guilarte J, de la Higuera-Torres J. Celiac disease presenting as chronic anemia associated with heart block. Am J Gastroenterol. 1998;93:1391-1392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 78. | Sethi GR, Singhal KK, Puri AS, Mantan M. Benefit of gluten-free diet in idiopathic pulmonary hemosiderosis in association with celiac disease. Pediatr Pulmonol. 2011;46:302-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 79. | Testa ME, Maffey A, Colom A, Agüero L, Rogé I, Andrewartha MS, Teper A. [Pulmonary hemorrhage associated with celiac disease]. Arch Argent Pediatr. 2012;110:e72-e76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 80. | Santos JW, Mello Neto AB, Marchiori RC, Michel GT, Fagundes AL, Tagliari LG, Cancian T. Pulmonary hemosiderosis associated with celiac disease: improvement after a gluten-free diet. J Bras Pneumol. 2012;38:412-414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 81. | Cottin V, Clérici G, Fabien N, Rousset H, Cordier JF. Celiac disease revealed by diffuse alveolar hemorrhage and heart block. Respirat Med Extra. 2006;2:89-91. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 82. | Abuid M, Kopec SE, Fisher K, Fraire A. Alveolar hemorrhage and celiac disease: A case of Lane-Hamilton Syndrome. Am J Respirat Critical Care Med. 2010;181:A4000. [DOI] [Full Text] |
| 83. | Aydogan M, Gundogan A, Ucar E, Tasci C, Gumus S, Polat Z, Deniz O, Tozkoparan E, Ozkan M, Bilgic H. Coexistance of idiopathic pulmonary hemosiderosis and celiac disease: Complete remission with gluten free diet. Eur Respiratory J. 2012;40:P3684. |
| 84. | Lapecorella M, Napolitano M, Mariani G. Hereditary hemorrhagic teleangectasia with celiac disease and atrial fibrillation. Eur J Intern Med. 2007;18:349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 85. | Ertem D, Tüney D, Baloglu H, Pehlivanoglu E. Superior mesenteric artery blood flow in children with celiac disease. J Pediatr Gastroenterol Nutr. 1998;26:140-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 86. | Alvarez D, Vazquez H, Bai JC, Mastai R, Flores D, Boerr L. Superior mesenteric artery blood flow in celiac disease. Dig Dis Sci. 1993;38:1175-1182. [RCA] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 87. | AlAttar WA, Majeed SE, Shubbar AH. Superior mesenteric artery blood flow in celiac disease. Estimation with Doppler ultrasound”. IJGE. 2005;5:38-43. |
| 88. | Bye AM, Andermann F, Robitaille Y, Oliver M, Bohane T, Andermann E. Cortical vascular abnormalities in the syndrome of celiac disease, epilepsy, bilateral occipital calcifications, and folate deficiency. Ann Neurol. 1993;34:399-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 38] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 89. | Martínez F, Berenguer M, Prieto M, Montes H, Rayón M, Berenguer J. Budd-Chiari syndrome caused by membranous obstruction of the inferior vena cava associated with coeliac disease. Dig Liver Dis. 2004;36:157-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 90. | Lim PO, Tzemos N, Farquharson CA, Anderson JE, Deegan P, MacWalter RS, Struthers AD, MacDonald TM. Reversible hypertension following coeliac disease treatment: the role of moderate hyperhomocysteinaemia and vascular endothelial dysfunction. J Hum Hypertens. 2002;16:411-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 91. | West J, Logan RF, Card TR, Smith C, Hubbard R. Risk of vascular disease in adults with diagnosed coeliac disease: a population-based study. Aliment Pharmacol Ther. 2004;20:73-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 118] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 92. | Singhal KK, Janmeja AK, Sodhi R, Punia RS. Hemoptysis in patients of celiac disease with disproportionately severe anemia: tip of the iceberg? Multidiscip Respir Med. 2013;8:25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 93. | Ashrafi F, Darakhshandeh A, Heidarpour M, Tavakoli T, Najafian J. Pericardial effusion in celiac disease. Int J Prev Med. 2014;5:356-359. [PubMed] |
| 94. | Ashrafi F, Darakhshandeh A, Heidarpour M, Tavakoli T, Najafian J. Pericardial effusion in celiac disease: A case report. Int J Prevent Med. 2014;5:356-359. [PubMed] |
| 95. | Riccabona M, Rossipal E. [Pericardial effusion in celiac disease--an incidental finding?]. Wien Klin Wochenschr. 2000;112:27-31. [PubMed] |
| 96. | Nenna R, Ferri M, Thanasi E, Luparia R, Versacci P, Anaclerio S, Bonamico M. P0444 The heart in coeliac disease children and adolescents. An ultrasonographic study. J Pediatr Gastroenterol Nutr. 2004;39:S224-S225. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 97. | Namendys Silva SA, Posadas Calleja JG, Domínguez Cherit G. [Myocardial depression in a patient with celiac disease. A clinical case report and literature review]. Rev Gastroenterol Mex. 2005;70:63-66. [PubMed] |
| 98. | Mazurkiewicz L, Bilinska ZT, Witkowski A, Walczak E, Wagner T, Grzybowski J, Bieganowski A, Michalak E, Wozniewicz B, Ruzyllo W. 351 Unexpected granulocytic myocarditis detected with endomyocardial biopsy manifesting as severe heart failure with conduction disease in a young patient with silent celiac disease. Eur J Heart Failure. 2004;3:89. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 99. | Mazurkiewicz L, Bilińska ZT, Witkowski A, Grzybowski J, Michalak E, Bieganowski A, Walczak E, Wagner T, Woźniewicz B, Ruzyłło W. [Myocardial microabscesses detected by endomyocardial biopsy in a patient with dilated cardiomyopathy and celiac disease: a case report]. Kardiol Pol. 2006;64:733-736. [PubMed] |
| 100. | Sategna-Guidetti C, Franco E, Martini S, Bobbio M. Binding by serum IgA antibodies from patients with coeliac disease to monkey heart tissue. Scand J Gastroenterol. 2004;39:540-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 101. | Frustaci A, Cuoco L, Chimenti C, Pieroni M, Fioravanti G, Gentiloni N, Maseri A, Gasbarrini G. Celiac disease associated with autoimmune myocarditis. Circulation. 2002;105:2611-2618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 115] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 102. | Menezes TM, Motta ME. Celiac disease prevalence in children and adolescents with myocarditis and dilated cardiomiopathy. J Pediatr (Rio J). 2012;88:439-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 103. | Goel NK, McBane RD, Kamath PS. Cardiomyopathy associated with celiac disease. Mayo Clin Proc. 2005;80:674-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 104. | Boskovic A, Kitic I, Prokic D, Stankovic I. Cardiomyopathy associated with celiac disease in childhood. Case Rep Gastrointest Med. 2012;2012:170760. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 105. | Mikolašević I, Zaninović-Jurjević T, Zaputović L, Mavrić Ž, Mulc S, Licul V, Štimac D, Tomulić V, Mijandrušić-Sinčić B. Celiac disease (gluten-sensitive enteropathy) and dilated cardiomyopathy. In Kongres Hrvatskoga Kardiolokog Društva. 2012;7:63-64. |
| 106. | De Bem RS, Da Ro Sa Utiyama SR, Nisihara RM, Fortunato JA, Tondo JA, Carmes ER, Souza RA, Pisani JC, Amarante HM. Celiac disease prevalence in Brazilian dilated cardiomyopathy patients. Dig Dis Sci. 2006;51:1016-1019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 107. | Not T, Faleschini E, Tommasini A, Repetto A, Pasotti M, Baldas V, Spano A, Sblattero D, Marzari R, Campana C. Celiac disease in patients with sporadic and inherited cardiomyopathies and in their relatives. Eur Heart J. 2003;24:1455-1461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 108. | Curione M, Barbato M, Cugini P, Amato S, Da Ros S, Di Bona S. Association of cardiomyopathy and celiac disease: an almost diffuse but still less know entity. A review. Arch Med Sci. 2008;4:103-107. |
| 109. | Gelfond D, Fasano A. Dilated cardiomyopathy and type I diabetes in a patient with celiac disease. J Pediatric Gastroenterol Nutr. 2006;43:E43. [DOI] [Full Text] |
| 110. | Vizzardi E, Lanzarotto F, Carabellese N, Mora A, Bertolazzi S, Benini F, Nodari S, Dei Cas L, Lanzini A. Lack of association of coeliac disease with idiopathic and ischaemic dilated cardiomyopathies. Scand J Clin Lab Invest. 2008;68:692-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 111. | Emilsson L, Andersson B, Elfström P, Green PH, Ludvigsson JF. Risk of idiopathic dilated cardiomyopathy in 29 000 patients with celiac disease. J Am Heart Assoc. 2012;1:e001594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 37] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 112. | Winter Del R JL, Gabrielli N L, Greig D, Inchauste G, Quezada F, Torres M J, Castro G P. [Dilated cardiomyopathy in celiac disease: report of one case]. Rev Med Chil. 2009;137:1469-1473. [PubMed] |
| 113. | Fonager K, Sørensen HT, Nørgård B, Thulstrup AM. Cardiomyopathy in Danish patients with coeliac disease. Lancet. 1999;354:1561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 79] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 114. | Di Lenarda A, Chicco D, Moretti M, Andrea T, Valentina B, Tarcisio N, Sinagra G. Prevalence of organ-specific autoantibodies and coeliac disease in patients with sporadic and inherited dilated cardiomyopathy and their relatives. J Am College Cardiol. 2007;49:60A-60. |
| 115. | Narula N, Rawal P, Kumar RM, Ram Thapa B. Association of celiac disease with cardiomyopathy and pulmonary hemosiderosis. J Trop Pediatr. 2010;56:201-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 116. | Işikay S, Yilmaz K, Kilinç M. Celiac disease with pulmonary haemosiderosis and cardiomyopathy. BMJ Case Rep. 2012;2012. [PubMed] |
| 117. | Khilnani GC, Jain N, Tiwari P, Hadda V, Singh L. A young man with hemoptysis: Rare association of idiopathic pulmonary hemosiderosis, celiac disease and dilated cardiomyopathy. Lung India. 2015;32:70-72. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 118. | McGrath S, Thomas A, Gorard DA. Cardiomyopathy responsive to gluten withdrawal in a patient with coeliac disease. BMJ Case Reports. 2016;2016:bcr2015213301. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 119. | Ouali S, Chabrak S, Larbi N, Kafsi N. [Dilated cardiomyopathy and atrio-ventricular block in coeliac disease. Two case reports]. Arch Mal Coeur Vaiss. 2006;99:1252-1255. [PubMed] |
| 120. | Mavroudis K, Aloumanis K, Stamatis P, Antonakoudis G, Kifnidis K, Antonakoudis C. Irreversible end-stage heart failure in a young patient due to severe chronic hypocalcemia associated with primary hypoparathyroidism and celiac disease. Clin Cardiol. 2010;33:E72-E75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 121. | Barrio JP, Cura G, Ramallo G, Diez M, Vigliano CA, Katus HA, Mereles D. Heart transplantation in rapidly progressive end-stage heart failure associated with celiac disease. BMJ Case Rep. 2011;2011:bcr1220103624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 122. | Sidhu SM, Fishel SC, Dunkerley GR, Lindenberg B. End-stage dilated cardiomyopathy in a 25-year-old presenting with symptoms of gluten enteropathy. Southern Med J. 2004;97:S13. |
| 123. | Poddar B, Shava U, Srivastava A, Kapoor A. Severe heart failure, dilated cardiomyopathy and pulmonary haemosiderosis in coeliac disease: report of two cases. Paediatr Int Child Health. 2014;34:142-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 124. | Roumeliotis N, Hosking M, Guttman O. Celiac disease and cardiomyopathy in an adolescent with occult cirrhosis. Paediatr Child Health. 2012;17:437-439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 125. | Lakhhdar R, Ben Slima H, Drissa M, Drissa H. Familial dilated cardiomyopathy associated with celiac disease. Tunis Med. 2012;90:181-183. [PubMed] |
| 126. | Uslu N, Demir H, Karagöz T, Saltik-Temizel IN. Dilated cardiomyopathy in celiac disease: role of carnitine deficiency. Acta Gastroenterol Belg. 2010;73:530-531. [PubMed] |
| 127. | Curione M, Danese C, Viola F, Di Bona S, Anastasia A, Cugini P, Barbato M. Carnitine deficiency in patients with coeliac disease and idiopathic dilated cardiomyopathy. Nutr Metab Cardiovasc Dis. 2005;15:279-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 128. | Romagnoli E, Boldrini E, Pietrangelo A. Association between celiac disease and idiopathic dilated cardiomyopathy: a case report. Intern Emerg Med. 2011;6:125-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 129. | Chuaqui B, Garrido J, Casanegra P. Actin-deficient cardiomyopathy coexisting with celiac disease: a chance association? Pathol Res Pract. 1986;181:604-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 130. | Ghorbel IB, Hajji R, Feki NB, Salem TB, Lamloum M, Houman MH. Two exceptional complications revealing celiac disease: ischemic cardiomyopathy and pellagra. Int J Celiac Dis. 2015;3:31-32. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 131. | Lerner A, Matthias T. Celiac disease: Intestinal, heart and skin interconnections. Int J Celiac Dis. 2015;3:28-30. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 132. | Curione M, Barbato M, Viola F, Francia P, De Biase L, Cucchiara S. Idiopathic dilated cardiomyopathy associated with coeliac disease: the effect of a gluten-free diet on cardiac performance. Dig Liver Dis. 2002;34:866-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 133. | Glover BM, Treanor NJ, McEneaney DJ. A case of dilated cardiomyopathy associated with coeliac disease. Archives Med Sci. 2007;3:272. |
| 134. | Elfström P, Hamsten A, Montgomery SM, Ekbom A, Ludvigsson JF. Cardiomyopathy, pericarditis and myocarditis in a population-based cohort of inpatients with coeliac disease. J Intern Med. 2007;262:545-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 36] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 135. | Zahmatkeshan M, Fallahpoor M, Amoozgar H. Prevalence of celiac disease in children with idiopathic dilated cardiomyopathy. Iran J Pediatr. 2014;24:587-592. [PubMed] |
| 136. | Picchi A, Pasqualini P, D’Aiello I, Cortese B, Micheli A, Limbruno U. Acute ST-elevation myocardial infarction in a 15-year-old boy with celiac disease and multifactorial thrombotic risk. Thromb Haemost. 2008;99:1116-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 137. | McNeill A, Duthie F, Galloway DJ. Small bowel infarction in a patient with coeliac disease. J Clin Pathol. 2006;59:216-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 138. | Saylan B, Cevik A, Kirsaclioglu CT, Ekici F, Tosun O, Ustundag G. Subclinical cardiac dysfunction in children with coeliac disease: is the gluten-free diet effective? ISRN Gastroenterol. 2012;2012:706937. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 139. | Efe TH, Ertem AG, Coskun Y, Bilgin M, Algul E, Beton O, Asarcikli LD, Erat M, Ayturk M, Yuksel I. Atrial Electromechanical Properties in Coeliac Disease. Heart Lung Circ. 2016;25:160-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 140. | Cenk S, Aylin DB, Fatma Ebru A, Nihal AB, Sevil ÖS, Serdal B, Emine B, Hüseyin A, Telat K, Tahir D, Osman E, Engin B. Assessment of left ventricular function by strain-strain rate echocardiography in patients with celiac disease. Turk J Med Sci. 2014;44:173-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 141. | Akin FE, Sari C, Ozer-Sari S, Demirezer-Bolat A, Durmaz T, Keles T, Ersoy O, Bozkurt E. The evaluation of left ventricular functions with tissue doppler echocardiography in adults with celiac disease. Saudi J Gastroenterol. 2016;22:116-121. [PubMed] |
| 142. | Cuoco L, Muriago M, Castaman R, Di Muzio D, Vescovo G, Salvagnini M. PA. 35 High prevalence of asymptomatic mitral valve prolapse in untreated celiac disease patients. Dig Liver Dis. 2008;40:S88. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 143. | Fathy A, Abo-Haded HM, Al-Ahmadi N, El-Sonbaty MM. Cardiac functions assessment in children with celiac disease and its correlation with the degree of mucosal injury: Doppler tissue imaging study. Saudi J Gastroenterol. 2016;22:441-447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 144. | Lionetti E, Catassi C, Francavilla R, Miraglia Del Giudice M, Sciacca P, Arrigo T, Leonardi S, Salpietro A, Salpietro C, La Rosa M. Subclinic cardiac involvement in paediatric patients with celiac disease: a novel sign for a case finding approach. J Biol Regul Homeost Agents. 2012;26:S63-S68. [PubMed] |
| 145. | Ozturk K, Altun B, Kurt O. Arterial stiffness in patients with celiac disease. Eur J Gastroenterol Hepatol. 2015;27:1479-1480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 146. | Ludvigsson JF, James S, Askling J, Stenestrand U, Ingelsson E. Nationwide cohort study of risk of ischemic heart disease in patients with celiac disease. Circulation. 2011;123:483-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 147. | Emilsson L, Carlsson R, Holmqvist M, James S, Ludvigsson JF. The characterisation and risk factors of ischaemic heart disease in patients with coeliac disease. Aliment Pharmacol Ther. 2013;37:905-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 148. | Lagro J, Duijts L, Jaddoe VW. Letter by Lagro et al regarding article, “Nationwide cohort study of risk of ischemic heart disease in patients with celiac disease”. Circulation. 2011;124:e422; author reply e423. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 149. | Emilsson L, James S, Ludvigsson JF. Ischaemic heart disease in first-degree relatives to coeliac patients. Eur J Clin Invest. 2014;44:359-364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 150. | Bardella MT, Cantù P, Cesana BM, Piodi LP. Severe heart disease in coeliac sprue and inflammatory Bowel disease: a common pathogenesis? Scand J Gastroenterol. 2004;39:1304-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 151. | Lebwohl B, Emilsson L, Fröbert O, Einstein AJ, Green PH, Ludvigsson JF. Mucosal healing and the risk of ischemic heart disease or atrial fibrillation in patients with celiac disease; a population-based study. PLoS One. 2015;10:e0117529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 152. | Lebwohl B, Emilsson L, Fröbert O, Einstein AJ, Green PH, Ludvigsson J. Su1437 Mucosal Healing and Risk of ischemic heart disease in celiac disease: A population-based cohort study. Gastroenterology. 2014;146:S-469. |
| 153. | Bayar N, Çekin AH, Arslan Ş, Çağırcı G, Erkal Z, Çay S, Köklü E, Küçükseymen S. Assessment of Left Atrial Function in Patients with Celiac Disease. Echocardiography. 2015;32:1802-1808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 154. | Yayla Ç, Yüksel M, Yayla KG, Açikgöz SK, Akboga MK, Kaplan M, Coskun O, Akpinar MY, Yalinkiliç ZM, Turak O, Kayaçetin E. PP-138 Assessment of Tp-e interval and Tp-e/QT Ratio in Patients with Celiac Disease. Am J Cardiol. 2016;117:S88. |
| 155. | Gajulapalli RD. Prevalence of atrial fibrillation in patient with celiac disease. Circulation. 2014;130:A12318. |
| 156. | Emilsson L, Smith JG, West J, Melander O, Ludvigsson JF. Increased risk of atrial fibrillation in patients with coeliac disease: a nationwide cohort study. Eur Heart J. 2011;32:2430-2437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 70] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 157. | Mah MW, Priel IE, Humen DP, Brown NE, Sproule BJ. Idiopathic pulmonary hemosiderosis, complete heart block and celiac disease. Can J Cardiol. 1989;5:191-194. [PubMed] |
| 158. | Kumhar M, Kumar C, Kulkarni D. Celiac disease in association with a congenital heart disease. Int J Celiac Dis. 2013;1:14-16. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 159. | Shahramian I, Dehghani SM, Haghighat M, Noori NM, Teimouri A, Sharafi E, Kalili M, Kalvandi G. Serological Evaluation of Celiac Disease in Children with Congenital Heart Defect; A Case Control Study. Middle East J Dig Dis. 2015;7:98-103. [PubMed] |
| 160. | Noori NM, Teimouri A, Nakhaey Moghaddam M, Shahraki T. The prevalence of celiac disease in down syndrome children with and without congenital heart defects. Int J Pediatr. 2016;4:2143-2152. [DOI] [Full Text] |
| 161. | Congdon PJ, Fiddler GI, Littlewood JM, Scott O. Coeliac disease associated with congenital heart disease. Arch Dis Child. 1982;57:78-79. [PubMed] |
| 162. | Barbato M, Viola F, Curione M, Di Bona S, Ambrosini A, Mancini V, Bascietto C, Cucchiara S. P0428 Cardiac sympathetic and parasympathetic balance before and after gluten free diet in children with celiac disease. J. Pediatr Gastroenterol Nutr. 2004;39:S218. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 163. | Penny HA, Ferrar M, Atkinson J, Hoggard N, Hadjivassiliou M, West JN, Sanders DS. PTU-184 Is there a relationship between gluten ingestion and postural tachycardia syndrome? Gut. 2015;64:A144. [DOI] [Full Text] |
| 164. | Penny HA, Aziz I, Ferrar M, Atkinson J, Hoggard N, Hadjivassiliou M, West JN, Sanders DS. PWE-055 Coeliac disease is more common in subjects with postural orthostatic tachycardia syndrome compared to healthy controls. Gut. 2016;65:A165-A166. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 165. | Stöllberger C, Finsterer J. Ischemic stroke in left ventricular noncompaction and celiac disease. Int J Cardiol. 2014;176:534-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 166. | Polat TB, Urganci N, Yalcin Y, Zeybek C, Akdeniz C, Erdem A, Imanov E, Celebi A. Cardiac functions in children with coeliac disease during follow-up: insights from tissue Doppler imaging. Dig Liver Dis. 2008;40:182-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 167. | Norsa L, Shamir R, Zevit N. Gluten-free diet in celiac disease: protective or providing additive risk factors for the development of cardiovascular disease? Nutr Ther Metabol. 2012;30:1-9. |
| 168. | Vanga RR, Kabbani TA, Mukherjee R, Villafuerte-Galvez JA, Pallav K, Dennis M, Kelly CP, Leffler DA. Sa1258 Risk of cardiovascular disease in individuals with celiac disease. Gastroenterology. 2013;144:S-244. [DOI] [Full Text] |
| 169. | Wei L, Spiers E, Reynolds N, Walsh S, Fahey T, MacDonald TM. The association between coeliac disease and cardiovascular disease. Aliment Pharmacol Ther. 2008;27:514-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 170. | Zweig BM, Sheth M, Simpson S, Al-Mallah MH. Association of abdominal aortic calcium with coronary artery calcium and obstructive coronary artery disease: a pilot study. Int J Cardiovasc Imaging. 2012;28:399-404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 171. | Stein AC, Liao C, Paski S, Polonsky T, Semrad CE, Kupfer SS. Obesity and Cardiovascular Risk in Adults With Celiac Disease. J Clin Gastroenterol. 2016;50:545-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 172. | Zanini B, Mazzoncini E, Caselani F, Magni A, Ricci C, Lanzini A. Effect of gluten free diet (GFD) on cardiovascular risk factors: Results in a cohort of 765 celiac disease (CD) patients. Gastroenterology. 2011;140:S-444. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 173. | Cipil H, Baydar I, Altinkaynak K, Ademoglu E, Laloglu E, Filiz M, Sanibas AV, Yildiz F, Sevimli H, Arslan M. Mean platelet volume in a patient with celiac disease: The relationship between metabolic syndrome, impaired fasting glucose and cardiovascular risk. Endoc Abstr. 2016;41:EP788. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 174. | Lewis NR, Logan RF, Sanders DS, Hubbard RB, Holmes GK, West J. Vascular risk profile in incident coeliac disease and change with gluten-free diet. Gut. 2011;60:A87-A88. [DOI] [Full Text] |
| 175. | Ludvigsson JF, de Faire U, Ekbom A, Montgomery SM. Vascular disease in a population-based cohort of individuals hospitalised with coeliac disease. Heart. 2007;93:1111-1115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 176. | Assa A, Frenkel-Nir Y, Tzur D, Katz LH, Shamir R. Cardiovascular Risk Factors in Adolescents With Celiac Disease: A Cross Sectional Population Based Study. J Pediatr Gastroenterol Nutr. 2016; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 177. | Warncke K, Liptay S, Fröhlich-Reiterer E, Scheuing N, Schebek M, Wolf J, Rohrer TR, Meissner T, Holl RW. Vascular risk factors in children, adolescents, and young adults with type 1 diabetes complicated by celiac disease: results from the DPV initiative. Pediatr Diabetes. 2016;17:191-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 178. | Leeds JS, Hopper AD, Hadjivasslliou M, Tesfaye S, Sanders DS. What are the implications of newly-identified coeliac disease in patients with type 1 diabetes mellitus? Effect on glycaemic control, quality of life, cardiac risk factors and peripheral nerve function. Proceedings Nutr Society. 2009;68:E56. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 179. | Norsa L, Shamir R, Zevit N, Verduci E, Hartman C, Ghisleni D, Riva E, Giovannini M. Cardiovascular disease risk factor profiles in children with celiac disease on gluten-free diets. World J Gastroenterol. 2013;19:5658-5664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 180. | Omelchenko OV. Features of functional state of cardiovascular system in children with celiac disease. Childs Health. 2013;45:66-69. |