Published online Jul 26, 2014. doi: 10.4330/wjc.v6.i7.602
Revised: February 22, 2014
Accepted: May 31, 2014
Published online: July 26, 2014
Processing time: 236 Days and 6.5 Hours
In 1990, takotsubo cardiomyopathy (TCM) was first discovered and reported by a Japanese cardiovascular specialist. Since then, this heart disease has gained worldwide acceptance as an independent disease entity. TCM is an important entity that differs from acute myocardial infarction. It occurs more often in postmenopausal elderly women, is characterized by a transient hypokinesis of the left ventricular (LV) apex, and is associated with emotional or physical stress. Wall motion abnormality of the LV apex is generally transient and resolves within a few days to several weeks. Its prognosis is generally good. However, there are some reports of serious TCM complications, including hypotension, heart failure, ventricular rupture, thrombosis involving the LV apex, and torsade de pointes. It has been suggested that coronary spasm, coronary microvascular dysfunction, catecholamine toxicity and myocarditis might contribute to the pathogenesis of TCM. However, its pathophysiology is not clearly understood.
Core tip: Takotsubo cardiomyopathy (TCM) is an important disease entity that differs from acute myocardial infarction. It occurs more often in postmenopausal elderly women, is characterized by a transient hypokinesis of the left ventricular (LV) apex, and is associated with emotional or physical stress. Wall motion abnormality of the LV apex is generally transient and resolves within a few days to several weeks. The prognosis of TCM is generally good. It has been suggested that coronary spasm, coronary microvascular dysfunction, catecholamine toxicity and myocarditis might contribute to the pathogenesis of TCM. However, its pathophysiology is not clearly understood.
- Citation: Komamura K, Fukui M, Iwasaku T, Hirotani S, Masuyama T. Takotsubo cardiomyopathy: Pathophysiology, diagnosis and treatment. World J Cardiol 2014; 6(7): 602-609
- URL: https://www.wjgnet.com/1949-8462/full/v6/i7/602.htm
- DOI: https://dx.doi.org/10.4330/wjc.v6.i7.602
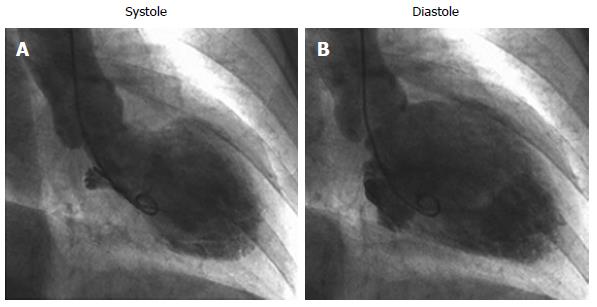
Takotsubo cardiomyopathy (TCM) is a transient wall motion abnormality of the left ventricular (LV) apex accompanied with emotional or physical stress that usually resolves completely. Takotsubo is a Japanese word meaning a pot with a narrow neck and a round bottom used to catch octopuses. Left ventriculography during systole of patients with TCM demonstrates such a shape. Although TCM is a novel concept, the number of cases reported is increasing rapidly. Other words have been used to refer this cardiomyopathy, including stress-related cardiomyopathy[1], transient LV apical ballooning syndrome[2,3], broken heart (heartbreak) syndrome, and ampulla cardiomyopathy[4]. In 2006, the American Heart Association incorporated this disease under the class of acquired cardiomyopathies[5]. This article aimed to review this newly recognized cardiomyopathy, paying particular attention to clinical characteristics, pathophysiology, diagnosis, and treatment.
TCM symptoms were considered extremely rare until the past 20 years. The increasing number of medical reports on these symptoms has highlighted the higher incidence of TCM than that previously reported. Currently, 1000 or more studies reporting cases of TCM have been published. According to a retrospective review, patients with TCM accounted for approximately 2% of all the patients with suspected acute coronary syndrome[6,7]. Further, 90% of these patients were postmenopausal women[8,9]. A few reports indicated that the average age of TCM patients was 68 years, although children or young adults may also be affected[10,11]. Another report indicated that most men with TCM were inpatients, which suggests that physical stresses might play a role for the progress of the disease[12]. In a recent study, demographic and clinical course data in patients with TCM were compared between the United States and Japan. Few Japanese patients with TCM had a history of overt coronary disease (CAD) and family history of early-onset CAD. However, there was no significant difference in long-term prognosis and the recurrence rate between the United States and Japanese patients with TCM[13].
The diagnosis of TCM remains controversial. The diagnostic criteria most widely accepted were published by the Mayo Clinic[14] in 2004. In 2008, a new criterion was added to them: a normal epicardial coronary artery (Table 1)[15]. Kawai et al[16] classified this disease as a syndrome of unknown etiology that was characterized by acute balloon-like dilation in the LV apex (Table 2). As shown by these two diagnostic criteria, the patients with TCM have nonspecific or normal findings on physical examination; however, the clinical course resembles that of acute coronary syndrome or acute decompensated heart failure[14-16]. The most common presenting symptoms listed in the diagnostic criteria are chest pain and dyspnea. In rare cases, patients developed palpitations, nausea, vomiting, syncope, or cardiogenic shock[14-16].
| Suspicion of AMI based on precordial pain and ST elevation observed on the acute-phase ECG |
| Transient hypokinesia or akinesia of the middle and apical regions of the LV and functional hyperkinesia of the basal region, observed on ventriculography or echocardiography |
| Normal coronary arteries confirmed by arteriography (luminal narrowing of less than 50% in all the coronary arteries) in the first 24 h after the onset of symptoms |
| Absence of recent significant head injury, intracranial hemorrhage, suspicion of pheochromocytoma, myocarditis, or hypertrophic cardio myopathy |
| Exclusion criteria |
| Significant organic stenosis or spasm of a coronary artery. In particular, AMI due to a lesion of the anterior descending artery of the left coronary artery, which irrigates a large territory including the apex of the LV (urgent coronary angiography is desirable in order to view the image in the acute phase; during the chronic phase, coronary angiography is necessary to confirm the presence or absence of significant stenotic lesions or abnormal lesions that could explain the ventricular contraction) |
| Cerebrovascular disturbances |
| Pheochromocytoma |
| Viral or idiopathic myocarditis |
| (Note: Coronary angiography is required for the exclusion of coronary artery lesions. Takotsubo-like myocardial dysfunction can occur in conditions such as cerebrovascular disorders or pheochromocytoma) |
| Diagnostic references |
| Symptoms: Precordial pain and dyspnea similar to the findings in the acute coronary syndrome. TCM can also occur without symptoms |
| Triggers: Emotional or physical stress, although it can also occur without any obvious trigger |
| Age and gender: There is a recognized tendency to a higher frequency in elderly individuals, principally women |
| Ventricular morphology: Apical ballooning with rapid recovery on ventriculography and echocardiography |
| ECG: ST elevation may be observed immediately after the event. T waves progressively become negative in various leads and the QT interval progressively lengthens. These changes gradually improve, but the T waves may remain negative for months. Pathological Q waves and alterations of the QRS voltage may be observed in the acute phase |
| Cardiac biomarkers: There is only a slight rise in the cardiac enzymes and troponin |
| Nuclear medicine scan of the heart: Abnormalities may be detected on myocardial gamma scan in some cases |
| Prognosis: Recovery is rapid in most cases, but some patients develop acute pulmonary edema and other sequel, even death |
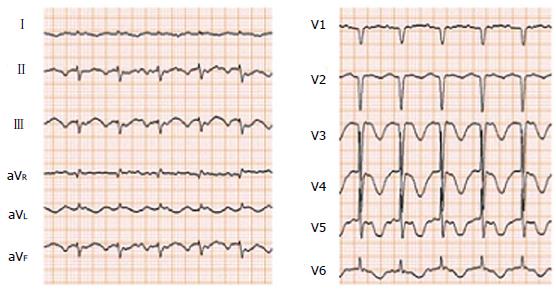
The following six symptoms are especially indicative of TCM: (1) acute onset and stressful inducement: One of the unique features of TCM is its relation with stressful emotional or physical events. This characteristic was described in nearly two-thirds of the patients who developed TCM[17]. Unlike acute coronary syndrome, with an onset peak early in the morning, TCM presents in the afternoon in most cases when stressful inducible events are likely to occur; (2) electrocardiographic characteristics: Although the initial electrocardiogram (ECG) of patients with TCM is nonspecific, an ST segment elevation can be found mainly in the precordial leads in 50% of patients at onset[18,19]. In addition, reciprocal ST-segment depression in the inferior wall leads is unlikely[20]. In comparison with patients with base deformity, inverted T waves are more frequently observed in patients with apex balloon-like dilation[21] and they resolve spontaneously within a few weeks to several months (Figure 1). Furthermore, patients with TCM usually present abnormal Q waves in precordial leads. These Q waves are transient in most patients and generally resolve within a few days to several weeks[22]; (3) cardiac enzymes: In most patients with TCM, there is slight elevation in the cardiac enzyme level on admission[6,20]. The enzyme levels decrease rapidly and do not seem to have prognostic significance[22]; (4) absence of coronary lesion: It is characteristic that no specific coronary lesions are detected in TCM[23,24]. Generally, patients with TCM have chest pain, changes in ECG, elevation of cardiac enzyme levels, and wall motion abnormalities. Therefore, coronary angiography has to be conducted to rule out acute coronary syndrome; (5) balloon-like dilation of the ventricle: In contrast with acute myocardial infarction, LV wall motion abnormalities are found beyond a single coronary artery perfusion area in patients with TCM. Most patients with TCM show loss of motion or hypokinesia at the apex and an apical balloon-like dilation pattern associated with preservation of the base (Figure 2). However, cases of a TCM subtype without abnormalities of the apex were reported recently[25,26]. TCM is essentially characterized by LV failure, although, approximately, one-third of patients also have abnormalities in the right ventricle[27]. Cardiac magnetic resonance imaging (MRI) is a suitable method to establish the diagnosis of TCM because this modality allows the accurate identification of reversible myocardium damage by visualization of wall motion abnormalities in each area, quantification of ventricular function, and assessment of inflammation and fibrosis. This modality brings new insight into the pathophysiology of TCM. It could enable early treatment of acute symptoms, raise awareness, and improve clinical outcomes. Cardiac MRI is appropriate to evaluate wall motion abnormalities and LV ejection fraction, and to confirm the absence of delayed gadolinium enhancement in patients with TCM. This allows differentiation of TCM from myocardial infarction and myocarditis, both pathologies associated with delayed gadolinium enhancement[17]. Although coronary computed tomography angiography is not applicable to the first diagnosis of patients with TCM, there are many reports on its use for clinical course evaluation after TCM onset; (6) recovery of cardiac function: One of the characteristics of TCM is that thorough recovery of cardiac function is achieved. In contrast to other serious wall motion abnormalities at onset, recovery of ventricular function is proven in follow-up evaluations. Most patients with TCM show significant improvement of systolic function within a week and achieve complete recovery by the end of third or fourth week after onset. Generally, another diagnosis should be considered in patients with suspected TCM whose systolic function is not normalized within 12 wk after onset.
The differential diagnosis of TCM includes the following: esophageal spasm, gastroesophageal reflux disease, myocardial infarction, myocardial ischemia, unstable angina, acute coronary syndrome, angina, aortic dissection, myocarditis, acute pericarditis, pneumothorax, cardiogenic pulmonary edema, pulmonary embolism, Boerhaave syndrome (spontaneous esophageal rupture), cardiac tamponade, cardiogenic shock, cocaine-induced cardiomyopathy, dilated cardiomyopathy, hypertrophic cardiomyopathy, and coronary artery spasm.
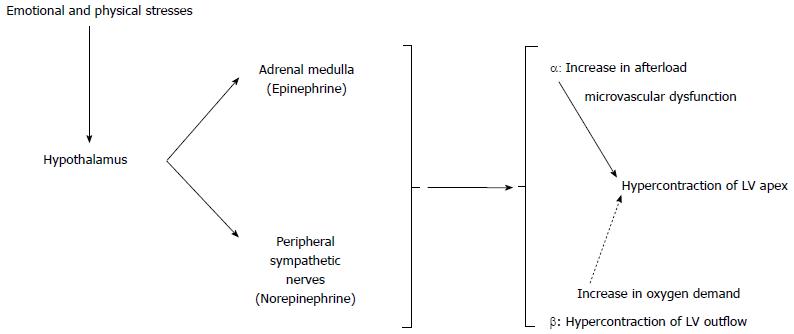
The exact pathogenesis of TCM is unknown, but various hypotheses have been suggested and discussed, including coronary microvascular dysfunction, coronary artery spasm, catecholamine-induced myocardial stunning, reperfusion injury following acute coronary syndrome, myocardial microinfarction and abnormalities in cardiac fatty acid metabolism. Currently, catecholamine-induced cardiotoxicity and microvasculature dysfunction are the most supported theories.
Wittstein et al[22] found that the serum catecholamine concentration was two to three times greater in patients with TCM than that in patients with myocardial infarction, and described that serious emotional stress is a precipitating factor. It has been reported that exogenously administered catecholamines and pheochromocytoma cause typical characteristics of TCM, which supports this theory further[28,29].
Lyon et al[30] advocated a theory called “stimulus trafficking” that could explain the decline of myocyte contractile function in patients with TCM. Supraphysiological levels of catecholamines induce β2-coupling from Gs to Gi. Therefore, the decline of myocyte contractile function is evidenced by hypokinesia in ECG. Involvement of the apex can be attributed to higher adrenoceptor density in the apex than in the base[31]. The rationale of stimulus trafficking is that a switch to Gi occurs to protect the myocytes from the strong stimulation of Gs, which causes apoptosis. Slow increases in serum troponin level explain early minimal necrosis of the myocardial tissue. Nef et al[32] showed increased activity of the phosphatidyl inositol 3-kinase-protein kinase B (PI3K/AKT) signaling pathway, which has important anti-apoptosis functions and plays a role in the rapid recovery of myocytes. Thus, the transient LV dysfunction can be attributed to the PI3K/AKT pathway and inversely switching from Gi to Gs, associated with the homogeneous, prompt and clinically thorough recovery of systolic function observed in TCM.
Patients with TCM consistently present microvasculature dysfunction findings[33]. The characteristics of microvasculature dysfunction after acute psychological stress in patients with TCM include abnormality of endothelium-dependent vasodilation, excessive vasoconstriction, and impairment of myocardial perfusion[34]. Uchida et al[35] reported that extensive endothelial cell apoptosis was observed by myocardial biopsy. According to another report, increased susceptibility to ergonovine or acetylcholine followed by large vessel spasm, similar to vasospastic angina, may contribute to transient LV dysfunction[36]. However, because only 30% of patients showed the characteristics of vasospasm in a challenge test, this theory was ruled out[37,38]. Afonso et al[39] demonstrated that circulatory disturbance, indicating coronary microvascular dysfunction was found on a myocardial contrast echocardiography and the epicardial coronary arteries were normal.
Myocardial biopsy of patients with TCM showed regions with contraction band necrosis, inflammatory cell infiltration, and localized fibrosis[40]. These changes were caused by direct catecholamine toxicity on cardiac muscle cells[41]. Morel et al[42] found that C-reactive protein levels and white blood cell counts increased with the increase in norepinephrine levels in patients with TCM and inferred that catecholamines produced more systemic inflammation via the induction of proinflammatory cytokines such as tumor necrosis factor-α and interleukin-6. Several studies have pointed out that the remarkable myocardial edema, observed on cardiac MRI, occurs despite normal perfusion, which provides further evidence to support the inflammation theory[43,44]. Ueyama et al[45] examined restraint stress in rats with TCM and reported that heme oxygenase 1 (HO-1) levels, a marker of oxidative stress that has cardioprotective properties, was increased significantly. Macrophages play an important main role in oxidative stress induction and expression of β- and α-adrenergic receptors. As a result of pretreatment with β- and α-antagonists, HO-1 expression and its altering gene expression, decreased.
More than 90% of patients with TCM are postmenopausal women. In fact, in a study to investigate if hormone replacement therapy had an effect on TCM, the authors concluded that none of the 31 patients with TCM received estrogen replacement therapy[46]. Moreover, Ueyama et al[47] demonstrated that the decrease in LV function was greater in ovariectomized rats subjected to restraint stress than in rats receiving estradiol supplementation. The myocytes are known to express estrogen receptor-α and estrogen receptor-β. According to Ueyama et al[47], estrogen enhanced transcription of cardioprotective factors such as heat shock protein and atrial natriuretic peptide, and in turn, protected against the toxic effects of catecholamines, calcium overload and reduced oxidative stress[48].
A study reported on the prevalence of mood disorders and use of antidepressants in patients with TCM[28]. When patients with depressive disorders experienced a stressful event, vagus nerve tension was decreased and response to adrenal medullary hormone was increased, which may be relevant to the cause of the disease[49]. Further, some patients with depression showed very high noradrenaline extravasation[50].
Certain polymorphisms of α- and β-adrenergic receptors are associated with neurogenic stunned myocardium that occurs as symptom of subarachnoid hemorrhage and has overlapping pathophysiology with TCM[51]. Although adrenoceptor polymorphisms have not yet been identified in patients with TCM, patients with this disease showed L41Q polymorphism of G protein coupled receptor kinase (GRK5) more frequently compared with the control group[52]. L41Q polymorphism of GRK5 responds to catecholamine stimulation and attenuates the response of β-adrenergic receptors. Under catecholamine stimulation, balloon dilation of the ventricle may occur either by negative inotropic effect by β-receptor decoupling or ischemia because of an imbalance between α1-adrenergic coronary artery vasoconstriction and β-adrenergic vasodilation. These reports suggest the very interesting possibility that the susceptibility to TCM in individuals may be partially related to genetic factors.
Treatment of TCM during the acute phase is mainly symptomatic treatment. Intra-aortic balloon pump equipment is required for hemodynamically unstable patients in addition to cardiopulmonary circulatory support and continuous veno-venous hemofiltration[53-55]. There is controversy on the use of cardiac stimulants because of increased circulating catecholamines[56]. However, cardiac stimulants are used in 20%-40% of patients with TCM[2,57]. Levosimendan may be beneficial because of its inotropic action and vasodilator effect[30,58]. Usage of anticoagulants may be considered at least until systolic function is recovered.
For patients with severe LV outflow tract obstruction with hemodynamic compromise, treatment with a β-blocker or α-adrenoceptor agonist such as phenylephrine and volume expansion should be considered. Calcium channel blockers can be used to decrease LV outflow tract pressure gradient. It is of utmost importance to avoid treatment with nitrites or inotropic drugs in these cases[59-63]. For patients with suspected vasospasm, the use of calcium channel blockers such as verapamil or diltiazem is suggested[64].
Hemodynamically stable patients are often treated with diuretics, angiotensin-converting enzyme (ACE) inhibitors and β-blockers. To reduce the risk of thromboembolism, patients with loss of motion of the LV apex should be treated with anticoagulant therapy until the contractility of the apex is improved unless there is a definite contraindication.
There is no consensus regarding long-term management of TCM, although it is reasonable to treat patients with β-blockers and ACE inhibitors during the ventricular recovery period. However, no data support the continuous use of these drugs for the prevention of TCM recurrence or improvement of survival rate. After LV function normalizes, physicians may consider discontinuation of these drugs.
Patients with TCM usually have a good prognosis, and almost perfect recovery is observed in 96% of the cases[65]. Mortality rate in hospital vary at one to two percent[18,66]. TCM was formerly thought to follow a relatively benign course. However, Sharkey et al[18] described that approximately 5% of TCM patients experienced cardiac arrest. While their long-term survival rate is the same as that in healthy subjects, patients with TCM have a greater risk of death at the time of initial onset[65]. Elesber et al[65] reported that the most frequent chief complaint was chest pain (30%) and that recurrence of the symptom occurred in 11% of patients with TCM after a 4-year follow-up. Some studies have been conducted to assess prognostic indicators such as ECG findings, signs of thrombolysis in myocardial infarction, grade of myocardial perfusion, and N-terminal pro-brain natriuretic peptide level. However, a definite outcome marker has not been established[66-68].
A lot of attention has been focused on TCM recently and this entity has been characterized as a transient LV dysfunction with rapid recovery generally induced by a stressful emotional or physical event. The number of TCM cases continues to increase. Because of close resemblance of its presentation and clinical course to acute myocardial infarction, we believe that TCM should be included in one of the differential diagnosis for acute myocardial infarction. Although the cause of this disease has not been completely understood to date, some promising hypotheses have been suggested. The occurrence of this disease is attributed to the large-scale production of catecholamines that causes myocardial hypokinesia via directcardiomyocyte toxicity and induction of coronary microvascular dysfunction. Further, the high prevalence of TCM in postmenopausal women suggests an important role of estrogen for myocardial protection. Another hypothesis includes oxidative/inflammatory stress-induced myocardial dysfunction. Although the treatment of TCM remains controversial, adrenergic blockade is suggested as a reasonable therapy based on the presumptive pathophysiology of TCM.
P- Reviewer: Al-Biltagi M, Celikyurt YU S- Editor: Ji FF L- Editor: A E- Editor: Wu HL
| 1. | Pavin D, Le Breton H, Daubert C. Human stress cardiomyopathy mimicking acute myocardial syndrome. Heart. 1997;78:509-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 130] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 2. | Tsuchihashi K, Ueshima K, Uchida T, Oh-mura N, Kimura K, Owa M, Yoshiyama M, Miyazaki S, Haze K, Ogawa H. Transient left ventricular apical ballooning without coronary artery stenosis: a novel heart syndrome mimicking acute myocardial infarction. Angina Pectoris-Myocardial Infarction Investigations in Japan. J Am Coll Cardiol. 2001;38:11-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1181] [Cited by in RCA: 1142] [Article Influence: 47.6] [Reference Citation Analysis (0)] |
| 3. | Desmet WJ, Adriaenssens BF, Dens JA. Apical ballooning of the left ventricle: first series in white patients. Heart. 2003;89:1027-1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 353] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 4. | Sharkey SW, Lesser JR, Maron MS, Maron BJ. Why not just call it tako-tsubo cardiomyopathy: a discussion of nomenclature. J Am Coll Cardiol. 2011;57:1496-1497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 5. | Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB. Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation. 2006;113:1807-1816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2284] [Cited by in RCA: 2243] [Article Influence: 118.1] [Reference Citation Analysis (0)] |
| 6. | Parodi G, Del Pace S, Carrabba N, Salvadori C, Memisha G, Simonetti I, Antoniucci D, Gensini GF. Incidence, clinical findings, and outcome of women with left ventricular apical ballooning syndrome. Am J Cardiol. 2007;99:182-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 121] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 7. | Eshtehardi P, Koestner SC, Adorjan P, Windecker S, Meier B, Hess OM, Wahl A, Cook S. Transient apical ballooning syndrome--clinical characteristics, ballooning pattern, and long-term follow-up in a Swiss population. Int J Cardiol. 2009;135:370-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 86] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 8. | Strunk B, Shaw RE, Bull S, Adams J, Baer M, Gershengorn K, Kao A, Keeffe B, Sklar J, Sperling D. High incidence of focal left ventricular wall motion abnormalities and normal coronary arteries in patients with myocardial infarctions presenting to a community hospital. J Invasive Cardiol. 2006;18:376-381. [PubMed] |
| 9. | Wedekind H, Möller K, Scholz KH. Tako-tsubo cardiomyopathy. Incidence in patients with acute coronary syndrome. Herz. 2006;31:339-346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 41] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 10. | Bajolle F, Basquin A, Lucron H, Bonnet D. Acute ischemic cardiomyopathy after extreme emotional stress in a child. Congenit Heart Dis. 2009;4:387-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 11. | Maruyama S, Nomura Y, Fukushige T, Eguchi T, Nishi J, Yoshinaga M, Kawano Y. Suspected takotsubo cardiomyopathy caused by withdrawal of bupirenorphine in a child. Circ J. 2006;70:509-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Kurisu S, Inoue I, Kawagoe T, Ishihara M, Shimatani Y, Nakama Y, Kagawa E, Dai K, Ikenaga H. Presentation of Tako-tsubo cardiomyopathy in men and women. Clin Cardiol. 2010;33:42-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 42] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Maekawa Y, Kawamura A, Yuasa S, Nesto RW, Fukuda K. Direct comparison of Takotsubo cardiomyopathy between Japan and USA: 3-year follow-up study. Intern Med. 2012;51:257-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 14. | Bybee KA, Kara T, Prasad A, Lerman A, Barsness GW, Wright RS, Rihal CS. Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med. 2004;141:858-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1048] [Cited by in RCA: 1020] [Article Influence: 48.6] [Reference Citation Analysis (0)] |
| 15. | Prasad A, Lerman A, Rihal CS. Apical ballooning syndrome (Tako-Tsubo or stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J. 2008;155:408-417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1193] [Cited by in RCA: 1297] [Article Influence: 76.3] [Reference Citation Analysis (0)] |
| 16. | Kawai S, Kitabatake A, Tomoike H. Guidelines for diagnosis of takotsubo (ampulla) cardiomyopathy. Circ J. 2007;71:990-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 236] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 17. | Eitel I, von Knobelsdorff-Brenkenhoff F, Bernhardt P, Carbone I, Muellerleile K, Aldrovandi A, Francone M, Desch S, Gutberlet M, Strohm O. Clinical characteristics and cardiovascular magnetic resonance findings in stress (takotsubo) cardiomyopathy. JAMA. 2011;306:277-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 390] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 18. | Sharkey SW, Windenburg DC, Lesser JR, Maron MS, Hauser RG, Lesser JN, Haas TS, Hodges JS, Maron BJ. Natural history and expansive clinical profile of stress (tako-tsubo) cardiomyopathy. J Am Coll Cardiol. 2010;55:333-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 610] [Cited by in RCA: 705] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 19. | Sanchez-Jimenez EF. Initial clinical presentation of Takotsubo cardiomyopathy with-a focus on electrocardiographic changes: A literature review of cases. World J Cardiol. 2013;5:228-241. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 20. | Ogura R, Hiasa Y, Takahashi T, Yamaguchi K, Fujiwara K, Ohara Y, Nada T, Ogata T, Kusunoki K, Yuba K. Specific findings of the standard 12-lead ECG in patients with ‘Takotsubo’ cardiomyopathy: comparison with the findings of acute anterior myocardial infarction. Circ J. 2003;67:687-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 183] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 21. | Hahn JY, Gwon HC, Park SW, Choi SH, Choi JH, Choi JO, Lee SC, On YK, Kim JS, Kim DK, Jeon ES, Lee SH, Hong KP, Park JE. The clinical features of transient left ventricular nonapical ballooning syndrome: comparison with apical ballooning syndrome. Am Heart J. 2007;154:1166-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 22. | Wittstein IS, Thiemann DR, Lima JA, Baughman KL, Schulman SP, Gerstenblith G, Wu KC, Rade JJ, Bivalacqua TJ, Champion HC. Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med. 2005;352:539-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2159] [Cited by in RCA: 2156] [Article Influence: 107.8] [Reference Citation Analysis (0)] |
| 23. | Pilgrim TM, Wyss TR. Takotsubo cardiomyopathy or transient left ventricular apical ballooning syndrome: A systematic review. Int J Cardiol. 2008;124:283-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 363] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 24. | Hoyt J, Lerman A, Lennon RJ, Rihal CS, Prasad A. Left anterior descending artery length and coronary atherosclerosis in apical ballooning syndrome (Takotsubo/stress induced cardiomyopathy). Int J Cardiol. 2010;145:112-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 25. | Hurst RT, Askew JW, Reuss CS, Lee RW, Sweeney JP, Fortuin FD, Oh JK, Tajik AJ. Transient midventricular ballooning syndrome: a new variant. J Am Coll Cardiol. 2006;48:579-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 205] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 26. | Reuss CS, Lester SJ, Hurst RT, Askew JW, Nager P, Lusk J, Altemose GT, Tajik AJ. Isolated left ventricular basal ballooning phenotype of transient cardiomyopathy in young women. Am J Cardiol. 2007;99:1451-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 27. | Elesber AA, Prasad A, Bybee KA, Valeti U, Motiei A, Lerman A, Chandrasekaran K, Rihal CS. Transient cardiac apical ballooning syndrome: prevalence and clinical implications of right ventricular involvement. J Am Coll Cardiol. 2006;47:1082-1083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 168] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 28. | Abraham J, Mudd JO, Kapur NK, Klein K, Champion HC, Wittstein IS. Stress cardiomyopathy after intravenous administration of catecholamines and beta-receptor agonists. J Am Coll Cardiol. 2009;53:1320-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 324] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 29. | Marcovitz PA, Czako P, Rosenblatt S, Billecke SS. Pheochromocytoma presenting with Takotsubo syndrome. J Interv Cardiol. 2010;23:437-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 30. | Lyon AR, Rees PS, Prasad S, Poole-Wilson PA, Harding SE. Stress (Takotsubo) cardiomyopathy--a novel pathophysiological hypothesis to explain catecholamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med. 2008;5:22-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 700] [Cited by in RCA: 643] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 31. | Mori H, Ishikawa S, Kojima S, Hayashi J, Watanabe Y, Hoffman JI, Okino H. Increased responsiveness of left ventricular apical myocardium to adrenergic stimuli. Cardiovasc Res. 1993;27:192-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 276] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 32. | Nef HM, Möllmann H, Hilpert P, Troidl C, Voss S, Rolf A, Behrens CB, Weber M, Hamm CW, Elsässer A. Activated cell survival cascade protects cardiomyocytes from cell death in Tako-Tsubo cardiomyopathy. Eur J Heart Fail. 2009;11:758-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 69] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 33. | Galiuto L, De Caterina AR, Porfidia A, Paraggio L, Barchetta S, Locorotondo G, Rebuzzi AG, Crea F. Reversible coronary microvascular dysfunction: a common pathogenetic mechanism in Apical Ballooning or Tako-Tsubo Syndrome. Eur Heart J. 2010;31:1319-1327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 209] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 34. | Martin EA, Prasad A, Rihal CS, Lerman LO, Lerman A. Endothelial function and vascular response to mental stress are impaired in patients with apical ballooning syndrome. J Am Coll Cardiol. 2010;56:1840-1846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 125] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 35. | Uchida Y, Egami H, Uchida Y, Sakurai T, Kanai M, Shirai S, Nakagawa O, Oshima T. Possible participation of endothelial cell apoptosis of coronary microvessels in the genesis of Takotsubo cardiomyopathy. Clin Cardiol. 2010;33:371-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 36. | Angelini P. Transient left ventricular apical ballooning: A unifying pathophysiologic theory at the edge of Prinzmetal angina. Catheter Cardiovasc Interv. 2008;71:342-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 79] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 37. | Gianni M, Dentali F, Grandi AM, Sumner G, Hiralal R, Lonn E. Apical ballooning syndrome or takotsubo cardiomyopathy: a systematic review. Eur Heart J. 2006;27:1523-1529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 973] [Cited by in RCA: 990] [Article Influence: 52.1] [Reference Citation Analysis (0)] |
| 38. | Kurisu S, Sato H, Kawagoe T, Ishihara M, Shimatani Y, Nishioka K, Kono Y, Umemura T, Nakamura S. Tako-tsubo-like left ventricular dysfunction with ST-segment elevation: a novel cardiac syndrome mimicking acute myocardial infarction. Am Heart J. 2002;143:448-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 609] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 39. | Afonso L, Bachour K, Awad K, Sandidge G. Takotsubo cardiomyopathy: pathogenetic insights and myocardial perfusion kinetics using myocardial contrast echocardiography. Eur J Echocardiogr. 2008;9:849-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 40. | Nef HM, Möllmann H, Kostin S, Troidl C, Voss S, Weber M, Dill T, Rolf A, Brandt R, Hamm CW. Tako-Tsubo cardiomyopathy: intraindividual structural analysis in the acute phase and after functional recovery. Eur Heart J. 2007;28:2456-2464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 288] [Cited by in RCA: 324] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 41. | Khullar M, Datta BN, Wahi PL, Chakravarti RN. Catecholamine-induced experimental cardiomyopathy--a histopathological, histochemical and ultrastructural study. Indian Heart J. 1989;41:307-313. [PubMed] |
| 42. | Morel O, Sauer F, Imperiale A, Cimarelli S, Blondet C, Jesel L, Trinh A, De Poli F, Ohlmann P, Constantinesco A. Importance of inflammation and neurohumoral activation in Takotsubo cardiomyopathy. J Card Fail. 2009;15:206-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 79] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 43. | Eitel I, Lücke C, Grothoff M, Sareban M, Schuler G, Thiele H, Gutberlet M. Inflammation in takotsubo cardiomyopathy: insights from cardiovascular magnetic resonance imaging. Eur Radiol. 2010;20:422-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 115] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 44. | Avegliano G, Huguet M, Costabel JP, Ronderos R, Bijnens B, Kuschnir P, Thierer J, Tobón-Gomez C, Martinez GO, Frangi A. Morphologic pattern of late gadolinium enhancement in Takotsubo cardiomyopathy detected by early cardiovascular magnetic resonance. Clin Cardiol. 2011;34:178-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 45. | Ueyama T, Kawabe T, Hano T, Tsuruo Y, Ueda K, Ichinose M, Kimura H, Yoshida K. Upregulation of heme oxygenase-1 in an animal model of Takotsubo cardiomyopathy. Circ J. 2009;73:1141-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 46. | Kuo BT, Choubey R, Novaro GM. Reduced estrogen in menopause may predispose women to takotsubo cardiomyopathy. Gend Med. 2010;7:71-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 58] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 47. | Ueyama T, Hano T, Kasamatsu K, Yamamoto K, Tsuruo Y, Nishio I. Estrogen attenuates the emotional stress-induced cardiac responses in the animal model of Tako-tsubo (Ampulla) cardiomyopathy. J Cardiovasc Pharmacol. 2003;42 Suppl 1:S117-S119. [PubMed] |
| 48. | Migliore F, Bilato C, Isabella G, Iliceto S, Tarantini G. Haemodynamic effects of acute intravenous metoprolol in apical ballooning syndrome with dynamic left ventricular outflow tract obstruction. Eur J Heart Fail. 2010;12:305-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 49. | Cevik C, Nugent K. The role of cardiac autonomic control in the pathogenesis of tako-tsubo cardiomyopathy. Am Heart J. 2008;156:e31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 50. | Barton DA, Dawood T, Lambert EA, Esler MD, Haikerwal D, Brenchley C, Socratous F, Kaye DM, Schlaich MP, Hickie I. Sympathetic activity in major depressive disorder: identifying those at increased cardiac risk? J Hypertens. 2007;25:2117-2124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 204] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 51. | Zaroff JG, Pawlikowska L, Miss JC, Yarlagadda S, Ha C, Achrol A, Kwok PY, McCulloch CE, Lawton MT, Ko N. Adrenoceptor polymorphisms and the risk of cardiac injury and dysfunction after subarachnoid hemorrhage. Stroke. 2006;37:1680-1685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 52. | Spinelli L, Trimarco V, Di Marino S, Marino M, Iaccarino G, Trimarco B. L41Q polymorphism of the G protein coupled receptor kinase 5 is associated with left ventricular apical ballooning syndrome. Eur J Heart Fail. 2010;12:13-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 53. | Patel HM, Kantharia BK, Morris DL, Yazdanfar S. Takotsubo syndrome in African-American women with atypical presentations: a single-center experience. Clin Cardiol. 2007;30:14-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 54. | Cangella F, Medolla A, De Fazio G, Iuliano C, Curcio N, Salemme L, Mottola G, Agrusta M. Stress induced cardiomyopathy presenting as acute coronary syndrome: Tako-Tsubo in Mercogliano, Southern Italy. Cardiovasc Ultrasound. 2007;5:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 55. | Bybee KA, Murphy J, Prasad A, Wright RS, Lerman A, Rihal CS, Chareonthaitawee P. Acute impairment of regional myocardial glucose uptake in the apical ballooning (takotsubo) syndrome. J Nucl Cardiol. 2006;13:244-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 56. | Sharkey SW, Lesser JR, Zenovich AG, Maron MS, Lindberg J, Longe TF, Maron BJ. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation. 2005;111:472-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 732] [Cited by in RCA: 696] [Article Influence: 34.8] [Reference Citation Analysis (0)] |
| 57. | Dote K, Sato H, Tateishi H, Uchida T, Ishihara M. Myocardial stunning due to simultaneous multivessel coronary spasms: a review of 5 cases. J Cardiol. 1991;21:203-214. [PubMed] |
| 58. | Padayachee L. Levosimendan: the inotrope of choice in cardiogenic shock secondary to takotsubo cardiomyopathy? Heart Lung Circ. 2007;16 Suppl 3:S65-S70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 59. | Bielecka-Dabrowa A, Mikhailidis DP, Hannam S, Rysz J, Michalska M, Akashi YJ, Banach M. Takotsubo cardiomyopathy--the current state of knowledge. Int J Cardiol. 2010;142:120-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 57] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 60. | Page SP, Pantazis A, Elliott PM. Acute myocardial ischemia associated with latent left ventricular outflow tract obstruction in the absence of left ventricular hypertrophy. J Am Soc Echocardiogr. 2007;20:772.e1-772.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 61. | Thorne KD, Kerut EK, Moore CK. Apical ballooning “tako-tsubo” syndrome associated with transient left ventricular outflow tract obstruction. Echocardiography. 2007;24:770-772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 62. | Penas-Lado M, Barriales-Villa R, Goicolea J. Transient left ventricular apical ballooning and outflow tract obstruction. J Am Coll Cardiol. 2003;42:1143-114; author reply 1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 63. | Yoshioka T, Hashimoto A, Tsuchihashi K, Nagao K, Kyuma M, Ooiwa H, Nozawa A, Shimoshige S, Eguchi M, Wakabayashi T. Clinical implications of midventricular obstruction and intravenous propranolol use in transient left ventricular apical ballooning (Tako-tsubo cardiomyopathy). Am Heart J. 2008;155:526.e1-526.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 76] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 64. | Ibanez B, Navarro F, Cordoba M, M-Alberca P, Farre J. Tako-tsubo transient left ventricular apical ballooning: is intravascular ultrasound the key to resolve the enigma? Heart. 2005;91:102-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 141] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 65. | Elesber AA, Prasad A, Lennon RJ, Wright RS, Lerman A, Rihal CS. Four-year recurrence rate and prognosis of the apical ballooning syndrome. J Am Coll Cardiol. 2007;50:448-452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 393] [Cited by in RCA: 404] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 66. | Dib C, Prasad A, Friedman PA, Ahmad E, Rihal CS, Hammill SC, Asirvatham SJ. Malignant arrhythmia in apical ballooning syndrome: risk factors and outcomes. Indian Pacing Electrophysiol J. 2008;8:182-192. [PubMed] |
| 67. | Ionescu CN, Aguilar-Lopez CA, Sakr AE, Ghantous AE, Donohue TJ. Long-term outcome of Tako-tsubo cardiomyopathy. Heart Lung Circ. 2010;19:601-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 68. | Nef HM, Möllmann H, Weber M, Deetjen A, Brandt R, Hamm CW, Elsässer A. Release pattern of cardiac biomarkers in left ventricular apical ballooning. Int J Cardiol. 2007;115:128-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 1.0] [Reference Citation Analysis (0)] |