Published online Jan 27, 2016. doi: 10.4240/wjgs.v8.i1.41
Peer-review started: June 27, 2015
First decision: September 17, 2015
Revised: October 2, 2015
Accepted: November 10, 2015
Article in press: November 11, 2015
Published online: January 27, 2016
Processing time: 210 Days and 3.8 Hours
This review focuses on the early diagnosis of anal cancer and its precursor lesions through routine screening. A number of risk-stratification strategies as well as screening techniques have been suggested, and currently little consensus exists among national societies. Much of the current clinical rationale for the prevention of anal cancer derives from the similar tumor biology of cervical cancer and the successful use of routine screening to identify cervical cancer and its precursors early in the disease process. It is thought that such a strategy of identifying early anal intraepithelial neoplasia will reduce the incidence of invasive anal cancer. The low prevalence of anal cancer in the general population prevents the use of routine screening. However, routine screening of selected populations has been shown to be a more promising strategy. Potential screening modalities include digital anorectal exam, anal Papanicolaou testing, human papilloma virus co-testing, and high-resolution anoscopy. Additional research associating high-grade dysplasia treatment with anal cancer prevention as well as direct comparisons of screening regimens is necessary to develop further anal cancer screening recommendations.
Core tip: Anal cancer is a low prevalence, highly morbid disease. With the success of secondary prevention practices for other human papilloma virus-associated malignancies, screening strategies may similarly decrease rates of anal cancer. No national guidelines formally support screening. This review summarizes possible screening modalities and what further evidence is needed to support routine screening for anal cancer.
- Citation: Leeds IL, Fang SH. Anal cancer and intraepithelial neoplasia screening: A review. World J Gastrointest Surg 2016; 8(1): 41-51
- URL: https://www.wjgnet.com/1948-9366/full/v8/i1/41.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v8.i1.41
Anal cancer is a rare disease whose outcomes continue to underperform those of other malignancies. There are approximately 7000 cases of anal cancer in the United States annually[1], and the 5-year survival rate is 65.7%[2]. Unlike other common gastrointestinal malignancies, the incidence of anal cancer is increasing at an average rate of 2.2% per year for the last decade. This increasing rate is above a historical baseline in the 1970s and 1980s where anal cancer incidence remained unchanged[2,3]. Much of this increase is due to the rise of new high-risk immunocompromised populations in the last three decades, including chronic human immunodeficiency virus (HIV) infected patients and organ transplant recipients. This review will focus on squamous cell carcinoma of the anus, which is responsible for 80% of all anal cancers[4].
Anal cancer has long been a delayed diagnosis. Historical studies reported a delay of more than two years in diagnosis in more than half of patients[5], and 44% of patients present with disseminated disease at diagnosis[2]. This epidemiologic trend is even more troubling when one considers that early stage anal cancer has been shown to respond exceptionally well to low-morbidity chemoradiation therapy while later stage disease often requires highly morbid and quality of life-changing surgical interventions such as abdominoperineal resection with a permanent colostomy for residual primary tumor and groin dissection for inguinal nodal metastases.
Historically, anal cancer was sufficiently rare that population screenings for the disease were not warranted. After 1997, the American Cancer Society dropped its recommendation for annual digital rectal examinations in favor of colonoscopies and sigmoidoscopies for colorectal cancer screening. This further contributed to a lack of screening for anal cancer, in an era where high-risk anal cancer populations were coming into existence.
It has been recognized that certain groups are at substantially higher risks of anal cancer than the general population. The association of sexually transmitted infections and sexual practices with anal cancer has been recognized since the 1980s[6]. One of the most closely associated sexually transmitted infections has been human papilloma virus (HPV), which was found in 88% of anal cancer patients in a case-controlled cohort[7] as well as in tissue samples containing anal intraepithelial neoplasia, an anal cancer precursor[8]. Other risk factors of anal cancer identified include smoking[9] and organ transplantation[10].
A number of similarities exist between squamous cell anal cancer and cervical cancer. Both occur at squamocolumnar junction epithelium. The transformation zones of the anal canal and the cervix are both characterized by high turnover epithelium that is thought to be particularly vulnerable to malignancy-inducing genetic alterations[11]. Both are HPV-associated which is thought to promulgate changes to cells’ DNA[12]. Immunosuppression is also an increasingly important risk factor for both cancers likely due to the increased activity of HPV seen in immunocompromised tissue substrates[13-15]. Finally, both types of cancer also have widely divergent outcomes for early vs late presenting disease[16].
Unlike anal cancer, diagnoses of cervical cancer have been markedly reduced in the last 40 years. Between 1975 and 2010, the incidence of cervical cancer has decreased by more than 50%[16]. This public health success story is largely attributed to the widespread and routine use of cervical cancer screening, primarily employing the cytology-based cervical Papanicolaou (Pap) test[17]. It is thought that a similar screening effort applied to anal cancer could potentially reverse the disturbing recent trends in disease incidence.
This review focuses on the early diagnosis of anal cancer and its precursor lesions through routine screening. A number of risk-stratification strategies as well as screening techniques have been suggested (Table 1), and currently little consensus exists among national societies (Table 2). No national screening guidelines for anal cancer exist, and the AIDS advocacy groups that note the increased risk of anal cancer in the HIV-positive population differ in their recommended approaches[18-21]. We provide here a balanced examination of the current clinical science to guide both practitioners and policymakers in this rapidly developing field.
| DARE | Anal Pap test | HPV testing | High resolution anoscopy | |
| Sensitivity[56,57,61,62,101] | Not studied | 69%-93% | Alone: 100% | Current diagnostic standard |
| Co-testing with Pap[49]: 72%-96% | ||||
| Specificity[56,57,61,62,101] | Not studied | 32%-59% | Alone: 16% | Current diagnostic standard |
| Resource availability | N/A | Ubiquitous | Ubiquitous | Highly selective centers |
| Provider availability | Universal | Specialty clinics | Specialty clinics | Highly selective centers |
| Learning curve | Part of usual clinical training | Part of usual clinical training | Part of usual clinical training | > 200 cases |
| Current consensus[52] | Annually, all HIV-positive patients | Annually in highest-risk groups | Alone: No recommendation | Second-line screen following positive Pap test |
| Co-testing: No recommendation |
| Routine screening of general population | Routine screening of high-risk individuals | Assesses modalities for diagnosis | Specific modalities assessed | |
| American Society of Colon and Rectal Surgeons[54] | No recommendation | No recommendation | Screening and surveillance | Anal Pap test, high-resolution anoscopy |
| European Society of Medical Oncology | No recommendation | No recommendation | Surveillance only | Digital anorectal exam, standard anoscopy, computed tomography, magnetic resonance imaging |
| European Society of Surgical Oncology | ||||
| European Society for Therapeutic Radiation and Oncology[102] | ||||
| National Comprehensive Cancer Network[103] | No recommendation | No recommendation | Surveillance only | Digital anorectal exam, standard anoscopy |
| Centers for Disease Control and Prevention[104] | No recommendation | No recommendation | Screening and surveillance | Digital anorectal exam, HPV testing |
| New York State Department of Health[52] | No recommendation | All HIV infected adults | Screening only | Digital anorectal exam, anal Pap test, high resolution anoscopy |
| HIV Medicine Association of the Infectious Diseases Society of America[20] | No recommendation | Men who have sex with men, women with a history of abnormal cervical Pap tests, and all HIV-positive persons with genital warts | Screening only | Digital anorectal exam, HPV co-testing, anal Pap test, high resolution anoscopy |
| British HIV Association[21] | No recommendation | No recommendation | Screening and surveillance | Digital anorectal exam, anal Pap test, high resolution anoscopy |
Although this review will not cover the cancer cell biology of anal cancer in detail, a general understanding is helpful because it influences the rationale for routine screening. Much of the current consensus on how anal dysplasia evolves is derived directly from cervical cancer literature. In cervical cancer, it is well recognized that the human papilloma virus infection is a necessary step in the development of cervical dysplasia and ultimately invasive neoplasia[22]. Anal cancer is a more heterogeneous set of malignancies with anal canal tumors that have pathology more similar to squamous cell cervical cancer in addition to perianal squamous cell carcinoma that behaves more like penile or vulvar cancer. Anal canal tumors’ similarity to cervical cancer is also shared by the high rate of HPV co-infection - particularly HPV 16 subtypes - in the latter with studies reporting rates over 90% while perianal tumors’ HPV co-infection rate vary from 30%-80%[23-25]. This ongoing close association between HPV and anal cancer has led to parallel frameworks of oncogenesis for both anal and cervical cancer.
Like cervical cancer, anal cancer is thought to most frequently develop at the transformation zone between squamous and columnar epithelium of the anal canal. HPV infects squamous epithelial cells, and the interaction between virion gene expression and cellular growth regulators leads to loss of differentiation and clonal proliferation[12]. These cells have a predictable pattern of stepwise cellular transformation that leads from normal squamous epithelium to low-grade dysplasia to high-grade dysplasia to invasive cancer[12,26-28].
HPV-associated cellular transformation is characterized by cyclical cellular proliferation and regression. An area of cellular atypia may progress to low-grade dysplasia before then regressing to normal tissue. The occurrence of cancer is when a particular transformed cell line breaks out of this characteristic cycling and linearly progresses to invasive disease[11].
The natural history of disease progression is largely unknown. Historical reports of the incidence of the progression from premalignant to invasive disease range from 2%-9%[29-32]. However, more recent series have reported rates as high as 13%-50% in immunocompromised patients managed expectantly[27,33].
Several case reports point toward anal squamous cell cancer arising in a background of high-grade dysplasia, supporting the dysplasia-to-cancer sequence. Per Scholefield et al[34] the estimated risk of progression from anal intraepithelial neoplasia (AIN) to invasive anal cancer is 10% in 5 years. However, this rate needs to be interpreted with caution as progression rates may vary according to such factors as eradication of disease by surgical therapy and the aforementioned risk factors that increase the risk of progression to invasive disease. Furthermore, Simard et al[35] showed that the incidence of high-grade dysplasia in men in San Francisco has been on the rise - increasing by 11.48% per year between 2000 and 2009.
The basic principle behind screening for anal dysplasia is the early identification of these proliferating cell lines that have established irreversible high-grade dysplasia or local invasive disease. Early stage anal cancer 5-year survival rates exceed 80% while disseminated disease 5-year survival rates are 30%[2]. Clinical studies demonstrating a morbidity or mortality benefit from routine screening for anal dysplasia are currently ongoing (see “Future Directions”), and the success of such efforts for cervical cancer suggests that further consideration is warranted.
Compared to other cancers, anal cancer is rare and no support for general population screening exists. Anal cancer is the 26th most common cancer type in the United States with approximately 7000 cases a year[1]. Although evidence suggests that the majority of anal cancers are initially asymptomatic[36], such a low pre-test probability does not make screening tests feasible for the general population.
However, there are populations with disproportionate prevalence of anal cancer that are more conducive to group-wide screening. Immunosuppressed patients are increasingly recognized as one of the groups at highest risk for anal cancer[13,37]. Much of this recognition has developed over the rise of the HIV/AIDS epidemic in the last three decades. Infection with HIV is associated with a 30-fold increased lifetime risk in anal cancer and a 4-fold increase in 5-year mortality[37,38]. Although sexual practices - particularly anoreceptive intercourse - have been previously associated with anal cancer, recent studies have shown that the risk of anal cancer in HIV-positive individuals exists independently of sexual practices[39,40]. The risk of anal dysplasia progression appears to correlate directly with degree of immunosuppression as measured by T cell CD4+ count with a cell count less than 200 cells/mm3 most closely associated with increased prevalence[41-43]. Surprisingly though increased access to highly active antiretroviral therapies has not eliminated the increased risk of anal cancer in the HIV-infected population. It is thought that immune system restoration does not entirely eliminate the increased risk of dysplastic changes and then antiretroviral treated patients are living longer thereby increasing the lifetime interval risk of disease incidence[44].
Similarly, other immunosuppressed populations share an increased incidence of anal cancer. Increased rates of anal cancer have been identified through controlled studies in kidney[45,46] and liver transplant recipients[47]; anogenital malignancy rates after renal transplant are estimated to be 30- to 100-fold higher than the general population[48].
Currently, no national or international society formally supports routine screening of at-risk populations for anal dysplasia. This lack of recommendation stems from the absence of high-quality studies that demonstrate improved morbidity and mortality for those participating in routine screening. Nevertheless, practice patterns by infectious disease specialists suggest that anal dysplasia screening of high risk individuals is becoming common[49-51], and influential regional societies like the New York State Department of Health AIDS Institute have begun recommending routine annual examination of the anus in all HIV-infected adults and cytologic testing in ultra high-risk HIV-positive patients such as men who have sex with men (MSM), those with a history of condylomata, and women with cervical or vulvar dysplasia[52]. Recent population health studies have even suggested that such selective screening remains inadequate and that anal dysplasia occurs frequently enough in the general HIV-positive population that all should undergo some form of extended screening[53].
The digital anorectal exam (DARE) is widely considered to be an essential but not sufficient component of any anal cancer screening evaluation. Although the American Society of Colon and Rectal Surgeons does not formally recommend routine screening for anal cancer, a visual perianal skin exam, DARE, and anoscopy are the suggested initial workup for any patient with history or symptoms concerning for anal cancer[54]. Any abnormal finding necessitates biopsy. There is no evidence that has demonstrated a screening benefit from physical examination and historic surveillance studies with digital examination suggest low sensitivity for recurrent disease[55]. But the risks to the patient are minimal. The leading guidelines for HIV-infected adults recommend an annual DARE with further screening only if meeting certain high-risk criteria such as MSM, prior history of anogenital condylomas, and women with abnormal cervical or vulvar histology[52]. As the availability of enhanced low cost screening practices such as the anal Pap test and high-resolution anoscopy become more widely available the accepted adequacy of the DARE as a primary screening test will likely diminish.
The cervical Pap test was introduced in the 1960s to help identify premalignant cervical dysplasia that could be intervened upon. Although never demonstrated in a randomized clinical trial, the introduction of the Pap test coincided with a substantially decreased incidence of invasive cervical cancer[17]. The basis of the test is to collect a swabbed cellular sample that is then collected and prepared on a microscope slide for examination by a pathologist. A number of pathology classification systems have been developed with the modified Bethesda System classification in most contemporary use[12].
Anal Pap testing was more recently introduced in the 1990s with a similar methodology and grading scheme as a primary screening tool for a premalignant anal dysplasia[56]. The technique has been well described and is analogous to the cervical Pap test[52]. Sensitivity and specificity have been shown to be similar to cervical cytology[57]. Using large cohort databases for retrospective analysis, Markov modeling of the anal Pap test has demonstrated that its role in anal cancer prevention is likely both cost-effective and efficacious[58].
The test is not without its limitations. Like the cervical Pap test, neither cytologic test has ever been studied in a randomized clinical trial between cytologic screening and expectant management. The anal Pap test also suffers from a similar inter-rater unreliability of cervical cytologic testing which is then further complicated by varied and evolving classification systems[59,60]. Sensitivity and specificity estimates range widely from 69% to 93%, and 32% to 59%, respectively[56,57,61,62]. The anal Pap tests specificity for diagnosing the correct degree of dysplasia is even less accurate with Pap testing routinely reporting low-grade atypia for lesions that ultimately are found to be high-grade dysplasia[63]. Moreover, sensitivity decreases in the highest risk groups. False-negative cytology results in MSM can be as high as 23% for HIV-negative patients, and 45% for HIV-positive patients[64]. Such high rates of missed pathology in high-risk populations most needing effective anal cancer screening have led some to suggest that anal Pap tests are inadequate on their own and should be paired with a direct visual modality such as high-resolution anoscopy in order for them to be considered an appropriate screening test[64]. Even with these limitations, the anal Pap test’s low cost, technical ease and familiarity to many primary care physicians, and acceptable sensitivity have supported its role as the most practical screening option currently available[52,54].
HPV testing is typically performed as part of a Pap test. With modern, liquid-based Pap testing, the same swab sample can be used for both cytology as well as HPV DNA testing[65].
The necessity of HPV for cervical cancer is well established[66] and its relationship as a prerequisite for cervical dysplasia has been used as the rationale for routine HPV testing with abnormal Pap test results. The use of HPV testing as a risk stratification tool for cervical dysplasia has become standard practice[11,67]. Some have even argued that HPV testing as a first-line screening tool for cervical cancer prevention may be sufficient without the need for cytology. Multiple large trials have demonstrated that a single negative HPV test virtually eliminated the risk of death from cervical cancer[68,69]. The United States Food and Drug Administration approved an HPV primary screening test in 2014[65].
The role of HPV in anal cancer is thought to be nearly as important, and many studies have routinely assessed HPV status when screening for anal dysplasia[70-72]. In select high-risk populations, HPV testing has been shown to be an important and clinically useful screening tool in conjunction with anal Pap testing[73]. Descriptive studies also associate more rapid progression of anal dysplasia with high-risk subtypes of HPV[42,74,75]. However, early studies have not shown any benefit to anal cancer prevention with or without HPV testing[76]. More broadly, no guidelines formally recommend HPV testing alone or in combination with Pap testing. Availability of this diagnostic modality is also limited by the lack of coverage by most insurance plans and thereby is a limited offering at most healthcare institutions[77]. The increasing benefit of HPV testing as part of cervical cancer prevention practices suggests that the nature and scope of the use of HPV testing for anal cancer prevention will need reconsideration in the future.
High resolution anoscopy has been proposed as a screening modality that addresses the sensitivity issues of the other methods described above. Modeled off of colposcopy for cervical cancer, high resolution anoscopy uses a high-magnification colposcope with a transparent anoscope to examine the entire anal canal and perianal skin under close visual inspection. Five percent acetic acid is used to identify areas of rapid cell growth; Lugol’s solution is employed to improve biopsy yield and accuracy since higher grade dysplastic lesions initially found with acetic acid will not uptake Lugol’s unlike low-grade dysplasia[78]. Originally reported in Europe in 1989[79], the procedure has been practiced and comprehensively described by its American introducers at the University of California San Francisco since the early 1990s[78].
The benefits of high resolution anoscopy (HRA) remain unchallenged. Decision models have also demonstrated the superiority of HRA-only screening to combined modalities[80]. The leading recommendations from the New York State Department of Health AIDS Institute state that HRA be considered standard of care for any patient with prior abnormal anal Pap test[52]. Its most important contribution being that it effectively addresses some of the limitations of anal cytology-based screening practices[81]. A longitudinal study of 368 asymptomatic MSM undergoing serial HRAs for a mean of 4.2 years found that 11% of high-grade dysplasia identified with HRA coincided with normal results from an anal Pap test[82]. In particular, high-risk groups such as HIV-positive MSM have lower sensitivity results from anal Pap testing[83]. Anal Pap testing may be useful as a way of alerting HRA clinicians to more closely examine suspicious lesions for low- vs high-grade heterogeneity thereby increasing biopsy yields. Some have suggested that HRA and anal Pap test co-testing be performed as a useful quality control measure for HRA[82,83].
Unfortunately, HRA’s usefulness as a screening test is impaired by the logistical needs of its use. Colposcopes are an additional piece of equipment needed for the clinic setting and training for HRA is important[52]. Surprisingly, HRA has been shown to be cost-effective though with the real obstacle being availability[80]. Anecdotally, the original group of researchers who brought HRA to the United States note a high degree of dexterity and technical prowess required to effectively visualize the entire anal canal and obtain reliable biopsy specimens[78]. A new provider logged the first 2 years of cases performed and found that it took approximately 200 cases before results demonstrated no missed high grade lesions found on follow-up[84]. This substantial learning curve and the lack of practitioners trained formally in residency or fellowship have led to a lack of providers able to provide HRA as a screening option. Hence, HRA is typically utilized as a second-line screening tool for abnormal Pap test or HPV results[53,85].
As described above, the anal cancer prevention literature is rife with screening techniques resulting from rational considerations of cancer biology but with minimal clinical evidence demonstrating their efficacy. This situation is further worsened by the lack of clear-cut guidelines from any national or international society of how best to address this rare but devastatingly morbid malignancy. Some of the most important ongoing clinical questions to be answered are noted here.
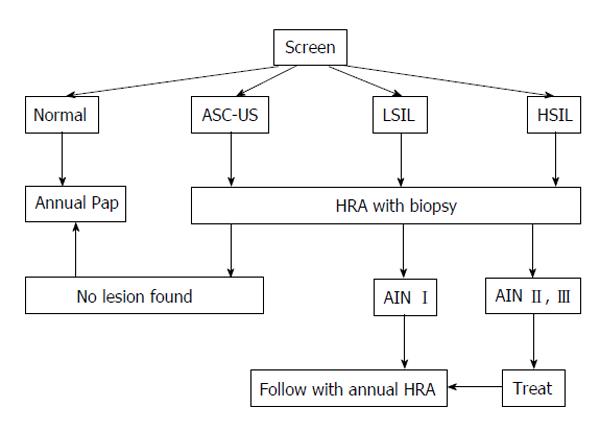
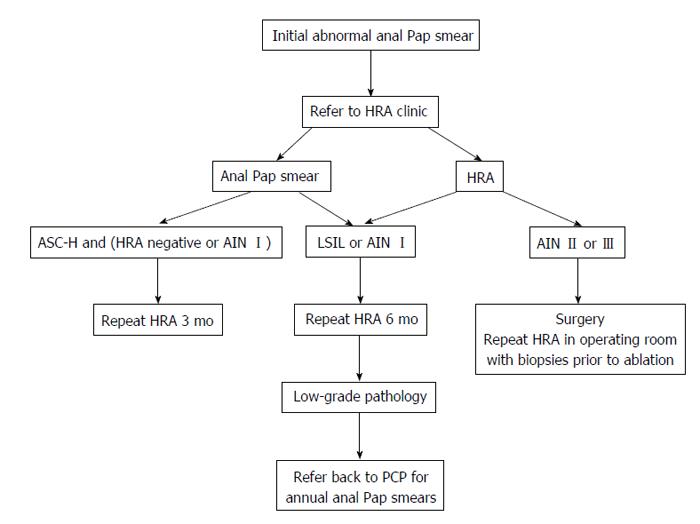
First, one of the most critical areas of further research is optimizing both the screening process and post-screen recommendations for a positive result. Until the data provides further guidance on HPV testing or sufficient HRA-trained providers are available to staff screening clinics, the anal Pap smear will remain the standard of care for anal cancer prevention’s primary screening modality. What to do with a positive screening test is a matter of ongoing debate. Without formal recommendations, individual expert opinion has driven institutions’ screening processes. The most widely disseminated screening algorithm was popularized by researchers at the University of California San Francisco and is reproduced in Figure 1[29]. The authors’ institution uses a modified algorithm that provides HRA screening for all referred patients (Figure 2). The basis of both of these algorithms is that all high-risk patients (e.g., HIV with high-risk sexual history or practices, MSM) get screened annually with an anal Pap test; all atypical cytology results are referred for HRA; and AIN I is followed yearly with HRA while AIN II or III is surgically removed. The diverse modalities for removal of high-grade dysplasia and carcinoma in situ are beyond the scope of this review but little guiding evidence exists and most practice is based off of cervical cancer excisional biopsy techniques.
There is also increasing evidence that the stringent risk stratification currently being employed may be too restrictive. The commonly followed New York State Department of Health’s AIDS Institute guidelines for anal cancer screening stratify HIV-positive patients into intermediate risk vs high risk groups. The latter risk stratified group recommends enhanced screening with annual anal Pap tests for any HIV-positive patient who also endorses MSM behaviors, history of anogenital condylomata, or women with history of abnormal cervical or vulvar pathology[52]. Increasing evidence suggests that HIV-positivity alone affords one a prevalence of approximately 20% for at least some form of anal intraepithelial neoplasia[39,40,64,86,87]. Such high rates of atypia may be unacceptably high for a population that under current recommendations would only receive a symptoms questionnaire, perianal visual inspection, and digital rectal examination. The previously mentioned high rate of cellular turnover and immunosuppression also suggests that less traditionally screened groups such as all anoreceptive sex practitioners and transplants would both warrant from annual screening as well.
Finally, one other line of inquiry that continues to be considered is the perception of patients who have to undergo these anorectal inspections annually awake and often with tissue samples taken. All of the evidence argues against this concern being a real obstacle to routine screening. Self-performed anal visual inspection[88], provider-performed digital rectal exam[89], and anal Pap testing[90,91] have all been explored with high-risk groups with favorable results. A Toronto study repeatedly screened patients for psychological distress at multiple points throughout the patient’s screening algorithm and found less than one-third ever felt negatively distressed throughout the process[92]. Rather than emotional distress, the greatest patient-oriented obstacles to care appear to be lack of knowledge of increase anal cancer risk and economic barriers to screening[90,91,93].
While controversies remain that will continue to shape the management of anal cancer screening today, there are also a number of expected future developments that may drastically change how we approach anal cancer prevention.
An ongoing Australian study may help address the role of HPV testing in anal cancer screening as well as provide more light on an evidence-based screening plan that incorporates one or more of the modalities described in this review. The Study of the Prevention of Anal Cancer (SPANC) is a 3-year prospective cohort that began recruitment in 2010 with follow-up planned through 2018 that will examine the overlapping roles of digital anorectal exam, HPV testing, anal Pap tests, and HRA[94]. Each participant will undergo all of these potential screening studies over multiple time points throughout the study, and it is expected that comparisons of sensitivity and specificity as well as the practical matters of performing each screen will be better understood. The study selected HIV-positive men over 35 years old living in the Sydney area with a total recruitment of over 350 participants. It is likely that the results of this study will provide a major contribution to the ongoing debate surrounding how best to utilize to the various anal cancer screening modalities at clinicians’ disposal.
The ANCHOR Study [anal cancer high-grade squamous intraepithelial neoplasia (HSIL) Outcomes Research] is an ongoing 5-year prospective randomized trial that has the goal of enrolling 5085 patients in the United States. This study aims to follow HIV-positive men over age 35 years with the diagnosis of HSIL over a 5-year period by anal Pap testing and HRA. The two arms of the study include a monitoring arm and a treatment arm for HSIL by ablation through infrared coagulation, with the ultimate goal of determining whether active surveillance with ablative treatment of HSIL will ultimately decrease the incidence of anal cancer[95].
There are also promising early signs to suggest that anal cancer may be an even more rare disease in the future. The HPV vaccine was developed out of the longstanding consensus that HPV infection is a necessary precursor to cervical cancer. Since its 2006 introduction the HPV vaccine has already been shown to reduce the prevalence of HPV infection among vaccinated populations. Estimating the reduction in cervical cancer cases from the already observed reduced in HPV infection suggests that disseminated vaccination will eliminate more than half of cervical cancers each year[96]. The similar tumor biology of cervical cancer and anal cancer suggests that HPV vaccination via herd immunity and more recent recommendations to vaccinate men as well will lead to a similar reduction in HPV-associated anal cancer[97]. There have also been clinical trials to demonstrate the efficacy of the HPV vaccine at reducing anal HPV infection[98,99]. This supportive evidence helped support a change in the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practice’s recommendation to begin routine HPV vaccination of all young males in addition to existing recommendations for female-only vaccination[100]. If general population uptake of these vaccine-based prevention practices is as successful as the early years suggest, it is likely that there will be dramatic reductions in HPV-associated cancer rates. Changes in prevalence will likely influence what kind of secondary prevention measures are appropriate for anal cancer screening later in life.
The low but rising incidence of anal cancer - particularly in vulnerable populations - makes it a concerning and difficult disease to manage with existing evidence-based care. Studies on its diagnosis and management are limited, and nearly all anal cancer guidelines avoid any direct recommendation regarding routine screening. The state of the literature suggests that further descriptive studies will be inadequate to advance consensus. Instead, large randomized clinical trials are necessary to demonstrate the increasing consensus among practitioners that anal cancer screening offers a cost-effective and prevalence lowering intervention in high-risk groups. The SPANC and ANCHOR studies will be helpful in determining whether routine screening through to a cancer diagnosis will ultimately be necessary to build the evidence for a population-wide recommendation.
P- Reviewer: Grundmann RT, Murata A S- Editor: Qi Y L- Editor: A E- Editor: Liu SQ
| 1. | American Cancer Society. Cancer Facts and Figures 2015. Atlanta, GA: American Cancer Society 2015; . |
| 2. | Surveillance Research Program. SEER Stat Fact Sheets: Anal Cancer. Bethesda, MD: National Cancer Institute 2015; . |
| 3. | Nelson RA, Levine AM, Bernstein L, Smith DD, Lai LL. Changing patterns of anal canal carcinoma in the United States. J Clin Oncol. 2013;31:1569-1575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 138] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 4. | Myerson RJ, Karnell LH, Menck HR. The National Cancer Data Base report on carcinoma of the anus. Cancer. 1997;80:805-815. [PubMed] |
| 5. | Möller C, Saksela E. Cancer of the anus and anal canal. Acta Chir Scand. 1970;136:340-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | Daling JR, Weiss NS, Hislop TG, Maden C, Coates RJ, Sherman KJ, Ashley RL, Beagrie M, Ryan JA, Corey L. Sexual practices, sexually transmitted diseases, and the incidence of anal cancer. N Engl J Med. 1987;317:973-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 404] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 7. | Frisch M, Glimelius B, van den Brule AJ, Wohlfahrt J, Meijer CJ, Walboomers JM, Goldman S, Svensson C, Adami HO, Melbye M. Sexually transmitted infection as a cause of anal cancer. N Engl J Med. 1997;337:1350-1358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 475] [Cited by in RCA: 425] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 8. | Scholefield JH, Hickson WG, Smith JH, Rogers K, Sharp F. Anal intraepithelial neoplasia: part of a multifocal disease process. Lancet. 1992;340:1271-1273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 83] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Daling JR, Sherman KJ, Hislop TG, Maden C, Mandelson MT, Beckmann AM, Weiss NS. Cigarette smoking and the risk of anogenital cancer. Am J Epidemiol. 1992;135:180-189. [PubMed] |
| 10. | Adami J, Gäbel H, Lindelöf B, Ekström K, Rydh B, Glimelius B, Ekbom A, Adami HO, Granath F. Cancer risk following organ transplantation: a nationwide cohort study in Sweden. Br J Cancer. 2003;89:1221-1227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 529] [Cited by in RCA: 515] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 11. | Schiffman M, Wentzensen N, Wacholder S, Kinney W, Gage JC, Castle PE. Human papillomavirus testing in the prevention of cervical cancer. J Natl Cancer Inst. 2011;103:368-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 547] [Cited by in RCA: 509] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 12. | Darragh TM, Colgan TJ, Cox JT, Heller DS, Henry MR, Luff RD, McCalmont T, Nayar R, Palefsky JM, Stoler MH. The Lower Anogenital Squamous Terminology Standardization Project for HPV-Associated Lesions: background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. Arch Pathol Lab Med. 2012;136:1266-1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 454] [Cited by in RCA: 428] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 13. | Saslow D, Solomon D, Lawson HW, Killackey M, Kulasingam SL, Cain J, Garcia FA, Moriarty AT, Waxman AG, Wilbur DC. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62:147-172. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 854] [Cited by in RCA: 816] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 14. | Berkhout RJ, Bouwes Bavinck JN, ter Schegget J. Persistence of human papillomavirus DNA in benign and (pre)malignant skin lesions from renal transplant recipients. J Clin Microbiol. 2000;38:2087-2096. [PubMed] |
| 15. | Shamanin V, zur Hausen H, Lavergne D, Proby CM, Leigh IM, Neumann C, Hamm H, Goos M, Haustein UF, Jung EG. Human papillomavirus infections in nonmelanoma skin cancers from renal transplant recipients and nonimmunosuppressed patients. J Natl Cancer Inst. 1996;88:802-811. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 229] [Cited by in RCA: 213] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 16. | Surveillance Research Program. SEER Stat Fact Sheets: Cervix Uteri Cancer. Bethesda, MD: National Cancer Institute 2015; . |
| 17. | van Oortmarssen GJ, Habbema JD. Duration of preclinical cervical cancer and reduction in incidence of invasive cancer following negative pap smears. Int J Epidemiol. 1995;24:300-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 33] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 18. | Darragh TM, Winkler B. Anal cancer and cervical cancer screening: key differences. Cancer Cytopathol. 2011;119:5-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 159] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 19. | Ong JJ, Chen M, Grulich AE, Fairley CK. Regional and national guideline recommendations for digital ano-rectal examination as a means for anal cancer screening in HIV positive men who have sex with men: a systematic review. BMC Cancer. 2014;14:557. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 20. | Aberg JA, Gallant JE, Ghanem KG, Emmanuel P, Zingman BS, Horberg MA. Primary care guidelines for the management of persons infected with HIV: 2013 update by the HIV Medicine Association of the Infectious Diseases Society of America. Clin Infect Dis. 2014;58:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 199] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 21. | Bower M, Palfreeman A, Alfa-Wali M, Bunker C, Burns F, Churchill D, Collins S, Cwynarski K, Edwards S, Fields P. British HIV Association guidelines for HIV-associated malignancies 2014. HIV Med. 2014;15 Suppl 2:1-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 22. | Bosch FX, Muñoz N. The viral etiology of cervical cancer. Virus Res. 2002;89:183-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 125] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 23. | Frisch M, Fenger C, van den Brule AJ, Sørensen P, Meijer CJ, Walboomers JM, Adami HO, Melbye M, Glimelius B. Variants of squamous cell carcinoma of the anal canal and perianal skin and their relation to human papillomaviruses. Cancer Res. 1999;59:753-757. [PubMed] |
| 24. | De Vuyst H, Clifford GM, Nascimento MC, Madeleine MM, Franceschi S. Prevalence and type distribution of human papillomavirus in carcinoma and intraepithelial neoplasia of the vulva, vagina and anus: a meta-analysis. Int J Cancer. 2009;124:1626-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 656] [Cited by in RCA: 665] [Article Influence: 41.6] [Reference Citation Analysis (0)] |
| 25. | Hillman RJ, Garland SM, Gunathilake MP, Stevens M, Kumaradevan N, Lemech C, Ward RL, Meagher A, McHugh L, Jin F. Human papillomavirus (HPV) genotypes in an Australian sample of anal cancers. Int J Cancer. 2014;135:996-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Stoler MH. Human papillomaviruses and cervical neoplasia: a model for carcinogenesis. Int J Gynecol Pathol. 2000;19:16-28. [PubMed] |
| 27. | Watson AJ, Smith BB, Whitehead MR, Sykes PH, Frizelle FA. Malignant progression of anal intra-epithelial neoplasia. ANZ J Surg. 2006;76:715-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 175] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 28. | Kreuter A, Potthoff A, Brockmeyer NH, Gambichler T, Swoboda J, Stücker M, Schmitt M, Pfister H, Wieland U. Anal carcinoma in human immunodeficiency virus-positive men: results of a prospective study from Germany. Br J Dermatol. 2010;162:1269-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 29. | Chin-Hong PV, Palefsky JM. Natural history and clinical management of anal human papillomavirus disease in men and women infected with human immunodeficiency virus. Clin Infect Dis. 2002;35:1127-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 210] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 30. | Cleary RK, Schaldenbrand JD, Fowler JJ, Schuler JM, Lampman RM. Perianal Bowen’s disease and anal intraepithelial neoplasia: review of the literature. Dis Colon Rectum. 1999;42:945-951. [PubMed] |
| 31. | Marfing TE, Abel ME, Gallagher DM. Perianal Bowen‘s disease and associated malignancies. Results of a survey. Dis Colon Rectum. 1987;30:782-785. [PubMed] |
| 32. | Scholefield JH, Castle MT, Watson NF. Malignant transformation of high-grade anal intraepithelial neoplasia. Br J Surg. 2005;92:1133-1136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 159] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 33. | Crum-Cianflone NF, Hullsiek KH, Marconi VC, Ganesan A, Weintrob A, Barthel RV, Agan BK. Anal cancers among HIV-infected persons: HAART is not slowing rising incidence. AIDS. 2010;24:535-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 152] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 34. | Scholefield JH, Harris D, Radcliffe A. Guidelines for management of anal intraepithelial neoplasia. Colorectal Dis. 2011;13 Suppl 1:3-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 35. | Simard EP, Watson M, Saraiya M, Clarke CA, Palefsky JM, Jemal A. Trends in the occurrence of high-grade anal intraepithelial neoplasia in San Francisco: 2000-2009. Cancer. 2013;119:3539-3545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 36. | Hicks CW, Wick EC, Leeds IL, Efron JE, Gearhart SL, Safar B, Fang SH. Patient Symptomatology in Anal Dysplasia. JAMA Surg. 2015;150:563-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 37. | Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet. 2007;370:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1647] [Cited by in RCA: 1612] [Article Influence: 89.6] [Reference Citation Analysis (0)] |
| 38. | Shiels MS, Cole SR, Kirk GD, Poole C. A meta-analysis of the incidence of non-AIDS cancers in HIV-infected individuals. J Acquir Immune Defic Syndr. 2009;52:611-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 396] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 39. | D’Souza G, Wiley DJ, Li X, Chmiel JS, Margolick JB, Cranston RD, Jacobson LP. Incidence and epidemiology of anal cancer in the multicenter AIDS cohort study. J Acquir Immune Defic Syndr. 2008;48:491-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 249] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 40. | Darwich L, Videla S, Cañadas MP, Piñol M, García-Cuyàs F, Vela S, Molina-López RA, Coll J, Sirera G, Clotet B. Distribution of human papillomavirus genotypes in anal cytological and histological specimens from HIV-infected men who have sex with men and men who have sex with women. Dis Colon Rectum. 2013;56:1043-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 41. | Bedimo RJ, McGinnis KA, Dunlap M, Rodriguez-Barradas MC, Justice AC. Incidence of non-AIDS-defining malignancies in HIV-infected versus noninfected patients in the HAART era: impact of immunosuppression. J Acquir Immune Defic Syndr. 2009;52:203-208. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 153] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 42. | de Pokomandy A, Rouleau D, Ghattas G, Trottier H, Vézina S, Coté P, Macleod J, Allaire G, Hadjeres R, Franco EL. HAART and progression to high-grade anal intraepithelial neoplasia in men who have sex with men and are infected with HIV. Clin Infect Dis. 2011;52:1174-1181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 129] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 43. | Guiguet M, Boué F, Cadranel J, Lang JM, Rosenthal E, Costagliola D. Effect of immunodeficiency, HIV viral load, and antiretroviral therapy on the risk of individual malignancies (FHDH-ANRS CO4): a prospective cohort study. Lancet Oncol. 2009;10:1152-1159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 422] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 44. | Piketty C, Selinger-Leneman H, Grabar S, Duvivier C, Bonmarchand M, Abramowitz L, Costagliola D, Mary-Krause M. Marked increase in the incidence of invasive anal cancer among HIV-infected patients despite treatment with combination antiretroviral therapy. AIDS. 2008;22:1203-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 209] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 45. | Ogunbiyi OA, Scholefield JH, Raftery AT, Smith JH, Duffy S, Sharp F, Rogers K. Prevalence of anal human papillomavirus infection and intraepithelial neoplasia in renal allograft recipients. Br J Surg. 1994;81:365-367. [PubMed] |
| 46. | Sillman FH, Sentovich S, Shaffer D. Ano-genital neoplasia in renal transplant patients. Ann Transplant. 1997;2:59-66. [PubMed] |
| 47. | Albright JB, Bonatti H, Stauffer J, Dickson RC, Nguyen J, Harnois D, Jeanpierre C, Hinder R, Steers J, Chua H. Colorectal and anal neoplasms following liver transplantation. Colorectal Dis. 2010;12:657-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 48. | Euvrard S, Kanitakis J, Claudy A. Skin cancers after organ transplantation. N Engl J Med. 2003;348:1681-1691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1186] [Cited by in RCA: 1108] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 49. | Berry JM, Palefsky JM, Jay N, Cheng SC, Darragh TM, Chin-Hong PV. Performance characteristics of anal cytology and human papillomavirus testing in patients with high-resolution anoscopy-guided biopsy of high-grade anal intraepithelial neoplasia. Dis Colon Rectum. 2009;52:239-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 145] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 50. | Swedish KA, Lee EQ, Goldstone SE. The changing picture of high-grade anal intraepithelial neoplasia in men who have sex with men: the effects of 10 years of experience performing high-resolution anoscopy. Dis Colon Rectum. 2011;54:1003-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 51. | Patel J, Salit IE, Berry MJ, de Pokomandy A, Nathan M, Fishman F, Palefsky J, Tinmouth J. Environmental scan of anal cancer screening practices: worldwide survey results. Cancer Med. 2014;3:1052-1061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 52. | New York State Department of Health AIDS Institute. HIV Clinical Resource: anal dysplasia and cancer. New York, NY: New York State Department of Health 2007; . |
| 53. | Weis SE, Vecino I, Pogoda JM, Susa JS, Nevoit J, Radaford D, McNeely P, Colquitt CA, Adams E. Prevalence of anal intraepithelial neoplasia defined by anal cytology screening and high-resolution anoscopy in a primary care population of HIV-infected men and women. Dis Colon Rectum. 2011;54:433-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 54. | Steele SR, Varma MG, Melton GB, Ross HM, Rafferty JF, Buie WD. Practice parameters for anal squamous neoplasms. Dis Colon Rectum. 2012;55:735-749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 55. | Papillon J, Mayer M, Montbarbon JF, Gerard JP, Chassard JL, Bailly C. A new approach to the management of epidermoid carcinoma of the anal canal. Cancer. 1983;51:1830-1837. [PubMed] |
| 56. | Palefsky JM, Holly EA, Hogeboom CJ, Berry JM, Jay N, Darragh TM. Anal cytology as a screening tool for anal squamous intraepithelial lesions. J Acquir Immune Defic Syndr Hum Retrovirol. 1997;14:415-422. [PubMed] |
| 57. | Fox PA, Seet JE, Stebbing J, Francis N, Barton SE, Strauss S, Allen-Mersh TG, Gazzard BG, Bower M. The value of anal cytology and human papillomavirus typing in the detection of anal intraepithelial neoplasia: a review of cases from an anoscopy clinic. Sex Transm Infect. 2005;81:142-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 103] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 58. | Goldie SJ, Kuntz KM, Weinstein MC, Freedberg KA, Palefsky JM. Cost-effectiveness of screening for anal squamous intraepithelial lesions and anal cancer in human immunodeficiency virus-negative homosexual and bisexual men. Am J Med. 2000;108:634-641. [PubMed] |
| 59. | Darragh TM, Winkler B, Souers RJ, Laucirica R, Zhao C, Moriarty AT. Room for improvement: initial experience with anal cytology: observations from the College of American Pathologists interlaboratory comparison program in nongynecologic cytology. Arch Pathol Lab Med. 2013;137:1550-1554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 60. | Darragh TM, Tokugawa D, Castle PE, Follansbee S, Borgonovo S, LaMere BJ, Schwartz L, Gage JC, Fetterman B, Lorey T. Interrater agreement of anal cytology. Cancer Cytopathol. 2013;121:72-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 61. | Arain S, Walts AE, Thomas P, Bose S. The Anal Pap Smear: Cytomorphology of squamous intraepithelial lesions. Cytojournal. 2005;2:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 69] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 62. | Cranston RD, Hart SD, Gornbein JA, Hirschowitz SL, Cortina G, Moe AA. The prevalence, and predictive value, of abnormal anal cytology to diagnose anal dysplasia in a population of HIV-positive men who have sex with men. Int J STD AIDS. 2007;18:77-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 53] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 63. | Betancourt EM, Wahbah MM, Been LC, Chiao EY, Citron DR, Laucirica R. Anal cytology as a predictor of anal intraepithelial neoplasia in HIV-positive men and women. Diagn Cytopathol. 2013;41:697-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 64. | Chin-Hong PV, Berry JM, Cheng SC, Catania JA, Da Costa M, Darragh TM, Fishman F, Jay N, Pollack LM, Palefsky JM. Comparison of patient- and clinician-collected anal cytology samples to screen for human papillomavirus-associated anal intraepithelial neoplasia in men who have sex with men. Ann Intern Med. 2008;149:300-306. [PubMed] |
| 65. | National Cancer Institute. Pap and HPV Testing. Bethesda, MD: National Institutes of Health 2014; . |
| 66. | Katki HA, Kinney WK, Fetterman B, Lorey T, Poitras NE, Cheung L, Demuth F, Schiffman M, Wacholder S, Castle PE. Cervical cancer risk for women undergoing concurrent testing for human papillomavirus and cervical cytology: a population-based study in routine clinical practice. Lancet Oncol. 2011;12:663-672. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 460] [Cited by in RCA: 411] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 67. | Franceschi S, Cuzick J, Herrero R, Dillner J, Wheeler CM. EUROGIN 2008 roadmap on cervical cancer prevention. Int J Cancer. 2009;125:2246-2255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 68. | Wright TC, Stoler MH, Behrens CM, Apple R, Derion T, Wright TL. The ATHENA human papillomavirus study: design, methods, and baseline results. Am J Obstet Gynecol. 2012;206:46.e1-46.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 188] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 69. | Sankaranarayanan R, Nene BM, Shastri SS, Jayant K, Muwonge R, Budukh AM, Hingmire S, Malvi SG, Thorat R, Kothari A. HPV screening for cervical cancer in rural India. N Engl J Med. 2009;360:1385-1394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 812] [Cited by in RCA: 852] [Article Influence: 53.3] [Reference Citation Analysis (0)] |
| 70. | Brickman C, Palefsky JM. Human papillomavirus in the HIV-infected host: epidemiology and pathogenesis in the antiretroviral era. Curr HIV/AIDS Rep. 2015;12:6-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 80] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 71. | Cambou MC, Luz PM, Lake JE, Levi JE, Coutinho JR, de Andrade A, Heinke T, Derrico M, Veloso VG, Friedman RK. Anal human papillomavirus (HPV) prevalences and factors associated with abnormal anal cytology in HIV-infected women in an urban cohort from Rio de Janeiro, Brazil. AIDS Patient Care STDS. 2015;29:4-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 72. | Guimarães MD, Grinsztejn B, Melo VH, Rocha GM, Campos LN, Pilotto JH, Carmo RA, Palefsky JM. Anal HPV prevalence and associated factors among HIV-seropositive men under antiretroviral treatment in Brazil. J Acquir Immune Defic Syndr. 2011;57 Suppl 3:S217-S224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 73. | Heard I, Etienney I, Potard V, Poizot-Martin I, Moore C, Lesage AC, Ressiot E, Crenn-Hebert C, Fléjou JF, Cubie H. High Prevalence of Anal Human Papillomavirus-Associated Cancer Precursors in a Contemporary Cohort of Asymptomatic HIV-Infected Women. Clin Infect Dis. 2015;60:1559-1568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 74. | Kreuter A, Brockmeyer NH, Hochdorfer B, Weissenborn SJ, Stücker M, Swoboda J, Altmeyer P, Pfister H, Wieland U. Clinical spectrum and virologic characteristics of anal intraepithelial neoplasia in HIV infection. J Am Acad Dermatol. 2005;52:603-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 70] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 75. | Latini A, Dona MG, Ronchetti L, Giglio A, Moretto D, Colafigli M, Laquintana V, Frasca M, Zaccarelli M, Antinori A. Prevalence of anal human papillomavirus infection and cytologic abnormalities among HIV-infected and HIV-uninfected men who have sex with men. J Int AIDS Soc. 2014;17:19662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 76. | Cheng SH, Wang CC, Chang SL, Chu FY, Hsueh YM. Oncogenic human papillomavirus is not helpful for cytology screening of the precursor lesions of anal cancers in Taiwanese men who are infected with human immunodeficiency virus. Int J Clin Oncol. 2015;20:943-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 77. | National LGBT Cancer Network. Anal cancer, HIV, and gay/bisexual men. New York, NY: National LGBT Cancer Network 2015; . |
| 78. | Palefsky JM. Practising high-resolution anoscopy. Sex Health. 2012;9:580-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 79] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 79. | Scholefield JH, Sonnex C, Talbot IC, Palmer JG, Whatrup C, Mindel A, Northover JM. Anal and cervical intraepithelial neoplasia: possible parallel. Lancet. 1989;2:765-769. [PubMed] |
| 80. | Lam JM, Hoch JS, Tinmouth J, Sano M, Raboud J, Salit IE. Cost-effectiveness of screening for anal precancers in HIV-positive men. AIDS. 2011;25:635-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 81. | Panther LA, Wagner K, Proper J, Fugelso DK, Chatis PA, Weeden W, Nasser IA, Doweiko JP, Dezube BJ. High resolution anoscopy findings for men who have sex with men: inaccuracy of anal cytology as a predictor of histologic high-grade anal intraepithelial neoplasia and the impact of HIV serostatus. Clin Infect Dis. 2004;38:1490-1492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 111] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 82. | Dalla Pria A, Alfa-Wali M, Fox P, Holmes P, Weir J, Francis N, Bower M. High-resolution anoscopy screening of HIV-positive MSM: longitudinal results from a pilot study. AIDS. 2014;28:861-867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 83. | Botes LP, Pett S, Carr A, Marriott D, Cooper DA, Matthews G, Carbone S, Kumaradevan N, McHugh L, Hillman RJ. Anal cytological abnormalities are poor predictors of high-grade intraepithelial neoplasia amongst HIV-positive men who have sex with men. Sex Health. 2013;10:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 84. | Richel O, Prins JM, de Vries HJ. Screening for anal cancer precursors: what is the learning curve for high-resolution anoscopy? AIDS. 2014;28:1376-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 85. | Chin-Hong PV, Palefsky JM. Human papillomavirus anogenital disease in HIV-infected individuals. Dermatol Ther. 2005;18:67-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 59] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 86. | Darwich L, Cañadas MP, Videla S, Coll J, Piñol M, Cobarsi P, Molina-López RA, Vela S, García-Cuyás F, Llatjos M. Condylomata, cytological abnormalities and human papillomavirus infection in the anal canal in HIV-infected men. HIV Med. 2012;13:549-557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 87. | Piketty C, Darragh TM, Da Costa M, Bruneval P, Heard I, Kazatchkine MD, Palefsky JM. High prevalence of anal human papillomavirus infection and anal cancer precursors among HIV-infected persons in the absence of anal intercourse. Ann Intern Med. 2003;138:453-459. [PubMed] |
| 88. | Ong JJ, Temple-Smith M, Chen M, Walker S, Grulich A, Fairley CK. Exploring anal self-examination as a means of screening for anal cancer in HIV positive men who have sex with men: a qualitative study. BMC Public Health. 2014;14:1257. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 89. | Read TR, Vodstrcil L, Grulich AE, Farmer C, Bradshaw CS, Chen MY, Tabrizi S, Hocking JS, Anderson J, Fairley CK. Acceptability of digital anal cancer screening examinations in HIV-positive homosexual men. HIV Med. 2013;14:491-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 90. | Ferris D, Lambert R, Waller J, Dickens P, Kabaria R, Han CS, Steelman C, Fawole F. Women’s knowledge and attitudes toward anal Pap testing. J Low Genit Tract Dis. 2013;17:463-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 91. | Reed AC, Reiter PL, Smith JS, Palefsky JM, Brewer NT. Gay and bisexual men’s willingness to receive anal Papanicolaou testing. Am J Public Health. 2010;100:1123-1129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 92. | Tinmouth J, Raboud J, Ali M, Malloch L, Su D, Sano M, Lytwyn A, Rourke SB, Rabeneck L, Salit I. The psychological impact of being screened for anal cancer in HIV-infected men who have sex with men. Dis Colon Rectum. 2011;54:352-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 93. | Pitts MK, Fox C, Willis J, Anderson J. What do gay men know about human papillomavirus? Australian gay men’s knowledge and experience of anal cancer screening and human papillomavirus. Sex Transm Dis. 2007;34:170-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 94. | Machalek DA, Grulich AE, Hillman RJ, Jin F, Templeton DJ, Tabrizi SN, Garland SM, Prestage G, McCaffery K, Howard K. The Study of the Prevention of Anal Cancer (SPANC): design and methods of a three-year prospective cohort study. BMC Public Health. 2013;13:946. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 95. | The Anchor Study. 2015. Available from: https://anchorstudy.org/. |
| 96. | Markowitz LE, Hariri S, Lin C, Dunne EF, Steinau M, McQuillan G, Unger ER. Reduction in human papillomavirus (HPV) prevalence among young women following HPV vaccine introduction in the United States, National Health and Nutrition Examination Surveys, 2003-2010. J Infect Dis. 2013;208:385-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 371] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 97. | Crosignani P, De Stefani A, Fara GM, Isidori AM, Lenzi A, Liverani CA, Lombardi A, Mennini FS, Palu’ G, Pecorelli S. Towards the eradication of HPV infection through universal specific vaccination. BMC Public Health. 2013;13:642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 36] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 98. | Kreimer AR, González P, Katki HA, Porras C, Schiffman M, Rodriguez AC, Solomon D, Jiménez S, Schiller JT, Lowy DR. Efficacy of a bivalent HPV 16/18 vaccine against anal HPV 16/18 infection among young women: a nested analysis within the Costa Rica Vaccine Trial. Lancet Oncol. 2011;12:862-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 147] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 99. | Lehtinen M, Dillner J. Clinical trials of human papillomavirus vaccines and beyond. Nat Rev Clin Oncol. 2013;10:400-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 137] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 100. | Centers for Disease Control and Prevention (CDC). Recommendations on the use of quadrivalent human papillomavirus vaccine in males--Advisory Committee on Immunization Practices (ACIP), 2011. MMWR Morb Mortal Wkly Rep. 2011;60:1705-1708. |
| 101. | Salit IE, Lytwyn A, Raboud J, Sano M, Chong S, Diong C, Chapman W, Mahony JB, Tinmouth J. The role of cytology (Pap tests) and human papillomavirus testing in anal cancer screening. AIDS. 2010;24:1307-1313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 128] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 102. | Glynne-Jones R, Northover JM, Cervantes A. Anal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21 Suppl 5:v87-v92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 68] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 103. | National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: anal carcinoma, version 2.2015. Fort Washington, PA: National Comprehensive Cancer Network 2014; . |
| 104. | Workowski KA, Bolan GA. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64:1-137. [PubMed] |