Published online Aug 27, 2025. doi: 10.4240/wjgs.v17.i8.109884
Revised: June 17, 2025
Accepted: July 14, 2025
Published online: August 27, 2025
Processing time: 93 Days and 9.3 Hours
The impact of transjugular intrahepatic portosystemic shunt (TIPS) on liver and spleen stiffness remains unclear, as does the association between preoperative liver and spleen stiffness and prognosis following TIPS.
To investigate changes in liver and spleen stiffness after TIPS and examines the relationship between these parameters and the prognosis of post-TIPS patients.
A total of 76 patients with liver cirrhosis and portal hypertension who underwent TIPS were included. Liver and spleen stiffness was assessed using the sound touch quantify (STQ) value, determined via point shear wave elastography in ultrasound imaging. Cox regression analysis was employed to evaluate the relationship between liver and spleen stiffness and cumulative survival in TIPS patients.
The liver STQ value demonstrated a marginally decreasing trend over time (P = 0.052), while the spleen STQ value showed a significantly decreasing trend (P = 0.025). Spleen STQ was positively correlated with portal pressure gradient (PPG) levels (rs = 0.327, P = 0.025). Cox regression analysis indicated that older age [hazard ratio (HR) = 1.063, 95%CI: 0.997-1.133, P = 0.060] and a higher liver STQ value (HR = 1.051, 95%CI: 1.009-1.095, P = 0.018) were associated with an in
Following TIPS, spleen stiffness exhibited a more pronounced change than liver stiffness and was positively associated with PPG. Preoperative liver stiffness serves as a prognostic indicator for survival in patients undergoing TIPS.
Core Tip: This study involved 76 patients with liver cirrhosis who underwent a transjugular intrahepatic portosystemic shunt (TIPS). The stiffness of the liver and spleen was assessed using point shear wave elastography with ultrasonic imaging, which quantifies the sound touch value. The results indicated that spleen stiffness significantly decreased over time following TIPS. Older age and higher liver stiffness were correlated with an elevated risk of mortality following TIPS. In summary, following TIPS, spleen stiffness exhibited a more pronounced change than liver stiffness. Preoperative liver stiffness serves as a prognostic indicator for survival in patients undergoing TIPS.
- Citation: Liu XF, Huang XC, Ye QJ, Yuan LJ, Gao GF, Li JY, Feng DP. Changes in liver and spleen stiffness after transjugular intrahepatic portosystemic shunt and their relationship with prognosis. World J Gastrointest Surg 2025; 17(8): 109884
- URL: https://www.wjgnet.com/1948-9366/full/v17/i8/109884.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i8.109884
Transjugular intrahepatic portosystemic shunt (TIPS) is a critical intervention used to manage complications arising from portal hypertension, such as variceal bleeding and refractory ascites[1]. Understanding the compensatory physiological changes following TIPS and predicting patient prognosis is essential for guiding clinical management and subsequent treatment strategies. This need has driven interest in non-invasive techniques, such as elastography, to evaluate liver and spleen stiffness, which may reflect the severity of portal hypertension and help predict clinical outcomes. Measurements of liver and spleen stiffness obtained through elastography are emerging as valuable tools in this setting. Shear wave elastography (SWE), for example, has been shown to predict reductions in the hepatic vein pressure gradient (HVPG) after TIPS, with spleen stiffness offering greater diagnostic accuracy than liver stiffness and conventional Child-Pugh scores[2].
Liver stiffness measurement has been extensively validated as an independent risk stratification tool across various chronic liver diseases, including primary biliary cholangitis (PBC)[3], nonalcoholic fatty liver disease (NAFLD)[4], and alcohol-related liver disease (ALD)[5]. Numerous studies consistently demonstrate that higher baseline liver stiffness is associated with worse outcomes, such as hepatic decompensation and mortality, across a broad spectrum of liver conditions[6]. In NAFLD, for instance, liver stiffness assessed via magnetic resonance elastography has been linked to the future development of cirrhosis and liver decompensation[7]. Similarly, vibration-controlled transient elastography has demonstrated substantial predictive power for adverse outcomes in PBC, facilitating patient risk stratification based on defined stiffness thresholds[3]. These findings highlight the growing role of liver stiffness measurement in both immediate clinical decision-making and long-term monitoring, thereby enhancing prognostic assessment and overall patient management.
Beyond static assessments, dynamic changes in liver stiffness measurements hold significant prognostic value. The progression or regression of liver stiffness correlates with liver-related events in chronic liver diseases. For example, a 20% increase in liver stiffness is associated with a substantially elevated risk of hepatic decompensation and liver-related mortality[8]. Findings in ALD suggest that both baseline and longitudinal changes in liver stiffness should be considered for accurate prognostic evaluation over time. Post-TIPS changes in liver stiffness, particularly when accompanied by systemic inflammation, further highlight the complex interactions between procedural interventions and underlying liver pathophysiology[9]. The prognostic utility of spleen stiffness is an area of ongoing investigation; existing evidence indicates its superior diagnostic performance, especially in the context of TIPS. In the evaluation of TIPS dysfunction - a challenge historically due to the invasive nature of venography - color Doppler ultrasound combined with spleen stiffness measurement has emerged as a promising non-invasive alternative, showing a strong correlation with HVPG[10]. These developments suggest a simplified and cost-effective approach for monitoring shunt function and potential complications.
The application of ultrasound-based stiffness measurements for prognostic prediction following TIPS, as well as in broader liver disease management, represents a convergence of technological innovation and clinical necessity. Point SWE, measured via sound touch quantify (STQ) value, is a non-invasive and widely utilized imaging technique primarily used to assess liver and spleen stiffness[11]. This study examines changes in liver and spleen stiffness based on STQ value at multiple time points after TIPS and explores the association between preoperative stiffness values and patient prognosis post-TIPS.
Patients with cirrhotic portal hypertension who underwent TIPS at the First Hospital of Shanxi Medical University were enrolled in the Taiyuan Cirrhosis & Portal Hypertension Cohort Study. Eligibility was determined based on specific inclusion and exclusion criteria. The inclusion criteria were: (1) Liver cirrhosis with portal hypertension confirmed by pathology or imaging and treated with TIPS; (2) Age > 18 years; and (3) Provision of signed informed consent. Exclusion criteria were: (1) Presence of malignant tumors; (2) Inability to undergo liver and spleen stiffness assessment via ultrasound; (3) TIPS procedure failure; (4) Prior history of liver or spleen surgery; and (5) Incomplete data. A total of 76 patients met the criteria and were included in the study. All participants provided written informed consent. The study was approved by the Research Ethics Committee of the First Hospital of Shanxi Medical University (Approval No. K-K112).
The data collected in this study included the following categories: (1) Demographic data: Age and gender; (2) Etiology: Viral, alcoholic, immunological, and other causes; (3) Laboratory examinations [alanine aminotransferase, aspartate aminotransferase, albumin, total bilirubin, white blood cell count, red blood cell count, hemoglobin, platelet count, international normalized ratio (INR), and blood ammonia]; (4) Liver function scores (Child-Pugh score, model for end-stage liver disease (MELD) score, and MELD-Na score); (5) Imaging examinations: Liver and spleen stiffness measurements; (6) Hemodynamic examination: Preoperative portal pressure gradient (PPG); and (7) Follow-up in
Indications for TIPS placement included refractory ascites and secondary prevention of variceal bleeding. The procedure was performed using covered stents, following the technique described by Rössle et al[12]. Clinical and ultrasonographic evaluations of the shunt were conducted three days post-procedure. All patients received an 8 mm diameter polytetrafluoroethylene-covered stent (GORE VIATORR).
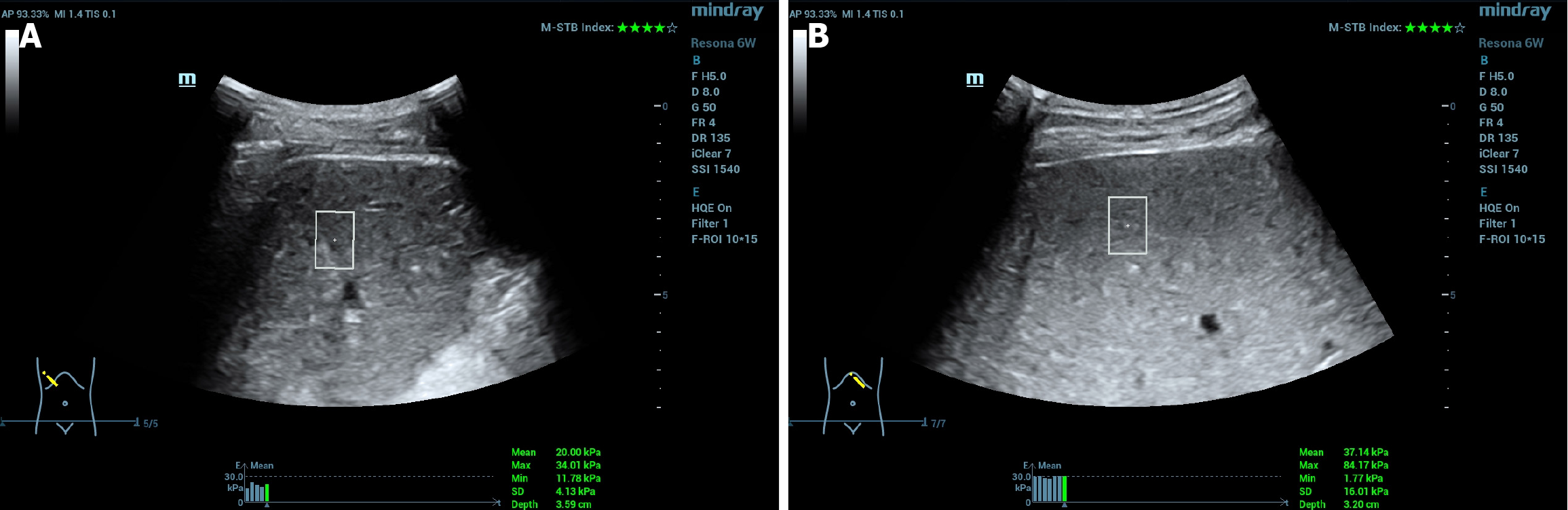
Liver and spleen stiffness was assessed using STQ on a Mindray Resona 6W ultrasound system (Mindray, Shenzhen, China) equipped with SC6-1 convex probes (frequency range: 1.0-6.0 MHz). All examinations were performed by the same ultrasound physician, who has over 10 years of experience. Patients fasted overnight prior to the examination.
Patients were positioned supine, and the probe was placed between the ribs to scan the right lobe of the liver and the spleen, targeting the S5/6 Liver segments and a representative section of the spleen. During the scan, patients were instructed to breathe calmly and to hold their breath for 3-5 s once the sampling frame displayed a uniform and stable image. At this point, the Q-box was activated and centered within the sampling box. Five measurements were taken in the same region, and the median value was recorded. A valid result was defined as having an interquartile range (IQR) to a median ratio (IQR/Med) ≤ 30% and image credibility > 95%. Figure 1 provides a schematic representation of repre
All patients were followed up at 1, 3, 6, 12, and 24 months after undergoing TIPS. During each follow-up visit, par
All statistical analyses were performed using SPSS software (version 24.0). Prior to analysis, normality was assessed using normality tests or P-P plots. Continuous variables are presented as mean ± SD or median with IQR, while categorical variables are expressed as frequency (percentage). Generalized estimating equations were used to evaluate trends in liver and spleen stiffness over time. Spearman correlation analysis was applied to assess relationships between continuous variables, yielding correlation coefficients. Logistic regression analysis was employed to identify risk factors associated with OHE following TIPS. Cox proportional hazard models were used to evaluate the association between liver and spleen stiffness and overall survival in patients with cirrhosis and portal hypertension who underwent TIPS. Kaplan-Meier survival curves were constructed to illustrate survival probabilities and differences between groups were compared using the Log-rank test. Receiver operating characteristic (ROC) analysis was used to assess predictive performance, and comparisons of the area under the ROC curves (AUCs) were conducted using the method of Hanley. In all analyses, two-tailed P values < 0.05 were considered statistically significant.
A total of 76 patients with liver cirrhosis and portal hypertension who underwent TIPS were ultimately included in this prospective cohort study. Among these 76 participants, 35 (46.1%) were male. The ages ranged from 26 to 81 years, with a mean age of 56.8 years. The most common etiology of cirrhosis was viral cirrhosis (32.9%), followed by immunologically associated cirrhosis (14.4%). During the follow-up period, 14 patients (18.4%) experienced an episode of OHE, and 12 patients (15.8%) died. Participants were divided into two groups based on survival status. The age (55.4 years vs 64.1 years), MELD-Na score (11.2 vs 27.0), blood ammonia concentration (18.7 μmol/L vs 24.4 μmol/L), liver STQ value (21.7 kPa vs 34.1 kPa), type IV collagen (148.7 ng/mL vs 197.5 ng/mL) and laminin (101.8 ng/mL vs 158.8 ng/mL) were all significantly higher in patients who died after TIPS, with statistically significant differences. Other variables did not show statistically significant differences between the two groups, as presented in Table 1.
| Parameter | All | Survival | Death | P value |
| Age, years | 56.8 ± 11.8 | 55.4 ± 10.5 | 64.1 ± 15.6 | 0.018 |
| Gender | 0.335 | |||
| Male | 35 (46.1) | 31 (48.4) | 4 (33.3) | |
| Female | 41 (53.9) | 33 (51.6) | 8 (66.7) | |
| Cause of disease | 0.09 | |||
| Viral | 25 (32.9) | 21 (32.8) | 4 (33.3) | |
| Alcoholic | 13 (17.1) | 12 (18.8) | 1 (8.3) | |
| Immunological | 24 (31.6) | 17 (26.6) | 7 (58.3) | |
| Other | 14 (18.4) | 14 (21.9) | 0 (0.0) | |
| Child-Pugh score | 7.7 ± 1.6 | 7.6 ± 1.6 | 8.5 ± 1.5 | 0.069 |
| MELD score | 11.1 ± 7.4 | 10.4 ± 5.9 | 14.6 ± 12.6 | 0.074 |
| MELD-Na | 13.7 ± 16.8 | 11.2 ± 6.4 | 27.0 ± 38.2 | 0.002 |
| ALT, U/L | 25.5 ± 23.9 | 25.2 ± 25.2 | 27.1 ± 16.1 | 0.806 |
| AST, U/L | 37.4 ± 31.8 | 36.6 ± 32.8 | 41.7 ± 26.7 | 0.614 |
| ALB, g/L | 38.6 ± 11.8 | 38.5 ± 11.7 | 39.1 ± 12.5 | 0.887 |
| TBIL, μmol/L | 29.2 ± 18.1 | 28.2 ± 17.9 | 34.9 ± 19.3 | 0.244 |
| WBC, × 109/L | 3.7 ± 2.6 | 3.7 ± 2.8 | 3.6 ± 1.3 | 0.876 |
| RBC, × 109/L | 3.2 ± 0.7 | 3.3 ± 0.7 | 3.0 ± 0.4 | 0.220 |
| HGB, g/L | 93.0 ± 24.4 | 93.4 ± 25.9 | 90.6 ± 14.1 | 0.716 |
| PLT, × 109/L | 89.4 ± 66.6 | 89.2 ± 67.1 | 90.5 ± 66.9 | 0.950 |
| INR | 2.9 ± 10.1 | 2.1 ± 6.8 | 7.2 ± 20.2 | 0.112 |
| Blood ammonia, μmol/L | 19.6 ± 7.4 | 18.7 ± 7.5 | 24.4 ± 5.1 | 0.019 |
| OHE | 0.522 | |||
| Yes | 14 (18.4) | 53 (82.8) | 9 (81.6) | |
| No | 62 (81.6) | 11 (17.2) | 3 (18.4) | |
| PPG, mmHg | 23.9 ± 6.3 | 23.8 ± 6.5 | 24.1 ± 5.4 | 0.913 |
| Liver STQ, kPa | 23.5 ± 11.7 | 21.7 ± 8.8 | 34.1 ± 19.5 | < 0.001 |
| Spleen STQ, kPa | 32.3 ± 10.9 | 32.2 ± 10.7 | 33.1 ± 12.2 | 0.795 |
| Hyaluronidase, ng/mL | 506.3 ± 589.9 | 473.4 ± 566.7 | 696.4 ± 717.5 | 0.299 |
| Type III procollagen, ng/mL | 15.2 ± 7.1 | 15.1 ± 7.2 | 16.3 ± 6.6 | 0.641 |
| Type IV collagen, ng/mL | 155.9 ± 63.9 | 148.7 ± 58.8 | 197.5 ± 79.3 | 0.033 |
| Laminin, ng/mL | 110.2 ± 71.5 | 101.8 ± 60.8 | 158.8 ± 107.9 | 0.026 |
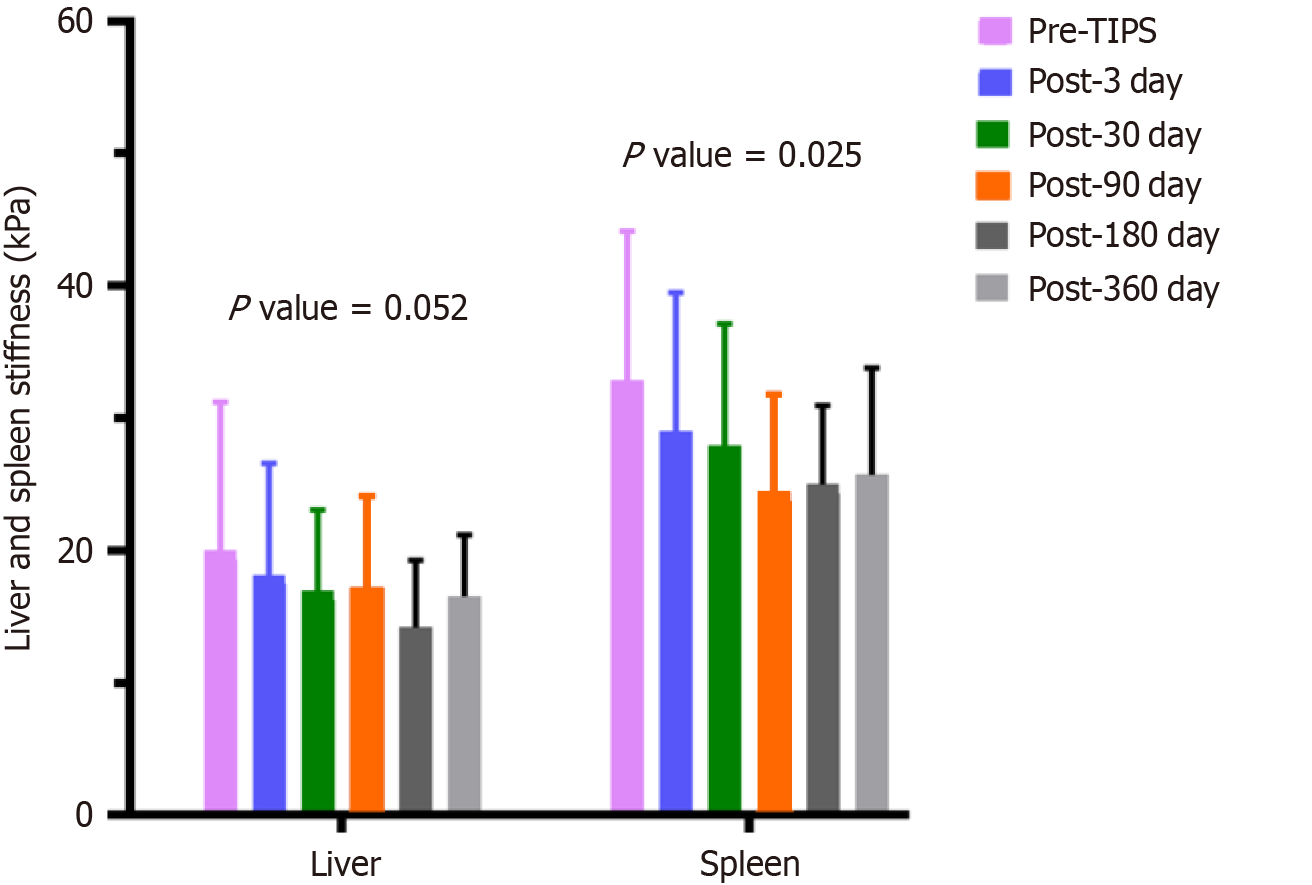
The STQ values were used to evaluate changes in liver and spleen stiffness within one year following TIPS, as illustrated in Figure 2. A generalized estimating equation was applied to assess whether the time after TIPS influenced liver and spleen stiffness based on longitudinal changes in STQ values. The results showed a marginal decreasing trend in liver STQ values, which reflect liver stiffness, over time (P = 0.052). In contrast, spleen STQ values, which reflect spleen stiffness, demonstrated a statistically significant decreasing trend over time (P = 0.025). These findings indicate that the reduction in spleen stiffness after TIPS was more pronounced than the change in liver stiffness.
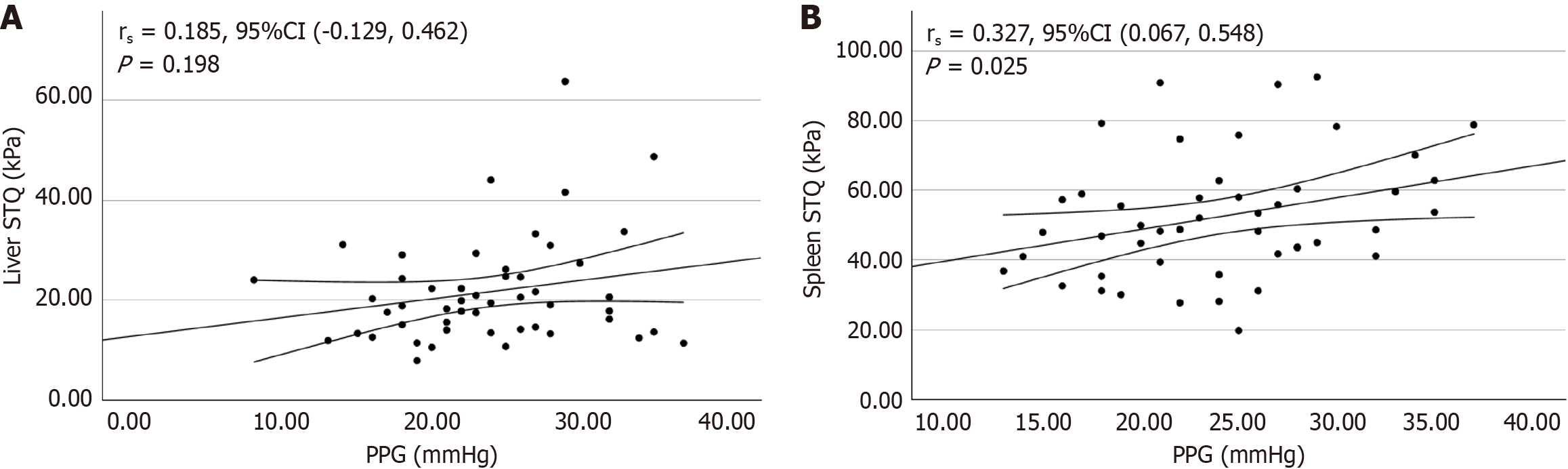
To explore the correlation between ultrasound-based liver and spleen stiffness parameters and portal vein pressure, we analyzed their relationships with PPG. Spearman correlation analysis revealed that spleen STQ values were positively correlated with preoperative PPG levels (rs = 0.327, P = 0.025), showing statistical significance. This suggests that spleen stiffness effectively reflects portal vein pressure. In contrast, liver stiffness did not demonstrate a statistically significant correlation (rs = 0.185, P = 0.198), as shown in Figure 3.
Further analysis was conducted to determine whether liver and spleen stiffness, as measured by ultrasound, were associated with serum fibrosis markers. Spearman correlation analysis indicated that serum type IV collagen and laminin were significantly positively correlated with liver STQ values (rs = 0.35, P = 0.008 for type IV collagen, rs = 0.39, P = 0.002 for laminin). No significant correlations were observed between serological fibrosis indicators and spleen stiffness, as shown in Supplementary Table 1. This finding is consistent with the biological basis that the liver secretes serum fibrosis markers.
We further analyzed the correlation between liver and spleen stiffness and liver function. The results showed that the Child-Pugh score (rs = 0.39, P = 0.014) and blood ammonia (rs = 0.26, P = 0.039) were positively correlated with liver STQ values. This indicates that poorer liver function is associated with increased liver stiffness. In comparison, only the Child-Pugh score demonstrated a positive correlation with spleen STQ values (rs = 0.29, P = 0.020). No statistically significant correlations were observed between other liver function indicators and liver or spleen stiffness, as presented in Supplementary Table 2.
To assess whether liver and spleen stiffness measured by ultrasound imaging is associated with the occurrence of OHE after TIPS, a logistic regression analysis was performed (Supplementary Table 3). The results showed no significant correlation between liver or spleen stiffness and post-TIPS OHE (all P > 0.05).
We analyzed the association between liver and spleen stiffness and mortality following TIPS. Univariable and multivariable Cox regression analyses indicated that older age [hazard ratio (HR) = 1.063, 95%CI: 0.997-1.133, P = 0.060] and a higher liver STQ value (HR = 1.051, 95%CI: 1.009-1.095, P = 0.018) were associated with an increased risk of death after TIPS. Both factors were identified as prognostic risk indicators for post-TIPS survival, as shown in Table 2. Specifically, each 1 kPa increase in liver STQ value corresponded to a higher mortality risk.
| Univariable | Multivariable | |||
| HR (95%CI) | P value | HR (95%CI) | P value | |
| Age, year | 1.070 (1.012, 1.131) | 0.017 | 1.063 (0.997, 1.133) | 0.060 |
| Gender, male | 1.744 (0.525, 5.792) | 0.364 | ||
| Child-Pugh score | 1.378 (0.990, 1.919) | 0.057 | ||
| Blood ammonia, μmol/L | 1.093 (1.013, 1.179) | 0.022 | ||
| OHE | 1.606 (0.435, 5.936) | 0.477 | ||
| Liver STQ, kPa | 1.071 (1.032, 1.111) | < 0.001 | 1.051 (1.009, 1.095) | 0.018 |
| Spleen STQ, kPa | 1.008 (0.954, 1.065) | 0.776 | ||
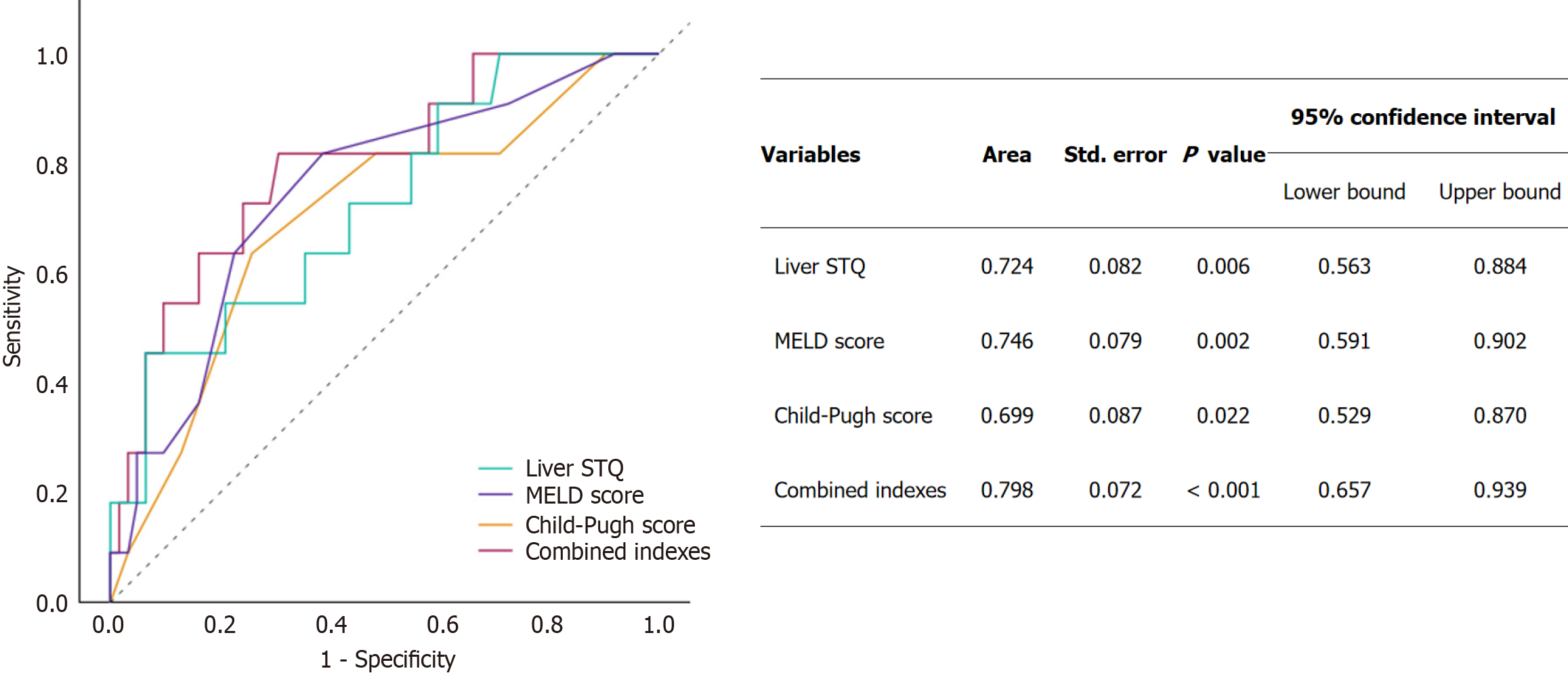
After establishing the relationship between liver STQ value and patient prognosis following TIPS, we further assessed and compared the predictive performance of liver function scores, liver stiffness, and combined indicators for post-TIPS outcomes. The liver STQ value showed an AUC of 0.724, 95%CI: 0.563-0.884, indicating better predictive performance than the Child-Pugh score (AUC = 0.699, 95%CI: 0.529-0.870), and a similar predictive capacity to the MELD score
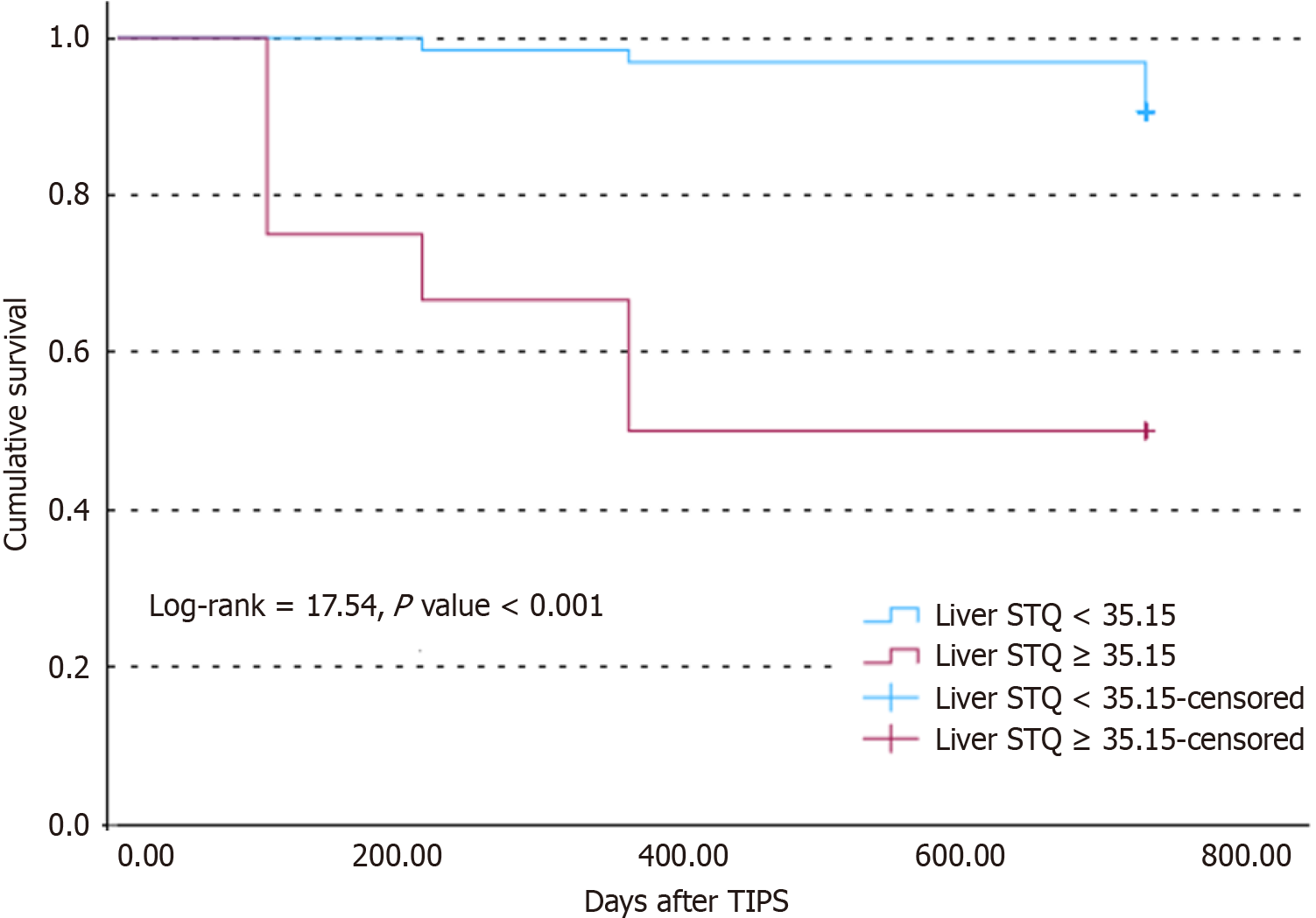
The optimal cut-off value for the liver STQ in predicting prognosis after TIPS was 35.15 kPa. Kaplan–Meier survival analysis demonstrated that patients with liver STQ ≥ 35.15 kPa had significantly shorter overall survival (Log-rank = 17.545, P < 0.001), as shown in Figure 5. The median overall survival was notably longer in the liver STQ < 35.15 kPa group compared to the ≥ 35.15 kPa group (716 days vs 417 days). These findings suggest that liver STQ value measured via ultrasound imaging should be considered an important prognostic indicator following TIPS.
To the best of our knowledge, this is the first report to investigate changes in both liver and spleen stiffness within 12 months after TIPS placement and to demonstrate the prognostic value of liver and spleen stiffness in patients with cirrhosis and portal hypertension following TIPS. The spleen STQ value exhibited a significantly decreasing trend over time post-TIPS. Patients with high liver STQ values prior to TIPS showed poor cumulative survival. Compared with Child-Pugh scores, the liver STQ value emerged as a more powerful prognostic indicator of overall survival in patients with cirrhosis and portal hypertension after TIPS. Liver stiffness based on STQ values should, therefore, be considered when evaluating the post-TIPS survival prognosis.
When comparing changes in liver and spleen stiffness at various intervals following TIPS, statistically significant variations were observed in spleen stiffness measured by STQ values. Over the one-year follow-up period, spleen stiffness initially decreased and subsequently increased. In contrast, liver stiffness exhibited only marginal statistical significance in its change. A small-sample study by Buechter et al[13] (n = 24) similarly reported a statistically significant reduction in spleen stiffness within 28 days after TIPS while noting no statistically significant change in liver stiffness. However, our study identified a marginally significant downward trend in liver stiffness, a discrepancy that may be attributable to sample size limitations. Nevertheless, this finding indirectly emphasizes the clinical value and potential superiority of spleen stiffness assessment in evaluating portal hypertension. This phenomenon can be explained biologically: In the context of portal hypertension, changes in spleen stiffness primarily result from splenic congestion secondary to elevated portal pressure rather than fibrotic degeneration of splenic parenchyma. This reflects hemodynamically induced stiffening. Conversely, liver stiffness arises from both parenchymal fibrosis and hemodynamic factors. Following TIPS-induced reduction in portal pressure and alleviation of hemodynamic stress, spleen stiffness de
To explore whether liver and spleen stiffness measured via ultrasound correlates with liver function and liver fibrosis serological markers, our results indicate a positive correlation between liver stiffness and both liver fibrosis markers and liver function indicators. Both types of indicators directly reflect hepatocellular damage[14], and their association with liver stiffness aligns with established biological principles and previously published reports[15,16]. Furthermore, we assessed the feasibility of using liver and spleen stiffness measurements to reflect portal vein pressure. Correlation analysis showed a positive correlation between spleen stiffness and PPG. However, liver stiffness did not demonstrate a statistically significant correlation with PPG. The absence of a significant relationship between liver stiffness and PPG was unexpected. Upon reviewing the literature, we noted that Giunta et al[17] also reported a significant correlation between spleen stiffness and both PPG and HVPG, while consistent findings regarding liver stiffness were not established - results that align with our observations. Although some studies have reported a correlation between liver stiffness and HVPG, no reports have demonstrated a correlation between liver stiffness and PPG. Based on this, we speculate that discrepancies in these conclusions may be attributable to variations in the degree of liver cirrhosis among different study populations. It is well known that when PPG exceeds 12 mmHg, patients enter the stage of clinically significant portal hypertension. At this stage, changes in portal pressure are largely governed by portal hemodynamics rather than being solely dependent on the extent of hepatic fibrosis. Literature indicates that when the HVPG exceeds 10-12 mmHg, portal pressure becomes relatively independent of liver fibrosis[18]. Liver stiffness primarily reflects the extent of hepatic fibrosis or cirrhosis, whereas portal pressure, measured by the PPG, is influenced by multiple factors. These include increased intrahepatic resistance (associated with fibrosis), as well as prehepatic and posthepatic resistance (e.g., hypersplenism, formation of collateral circulation), and alterations in blood flow dynamics. Therefore, at higher HVPG or PPG levels, the contribution of hepatic fibrosis to overall portal pressure diminishes, which results in a weakened correlation between liver stiffness and PPG. In contrast, the observed correlation between spleen stiffness and PPG suggests that spleen stiffness more accurately reflects changes in portal pressure than liver stiffness. This is likely due to the spleen’s direct involvement in the portal venous system, where the degree of congestion serves as a direct manifestation of portal hypertension. Following TIPS placement, the alleviation of splenic congestion is generally more pronounced than that of hepatic parenchymal stress. As a result, spleen stiffness values demonstrate a stronger association with changes in PPG.
In comparison to the study by Buechter et al[13], which reported a strong correlation between spleen stiffness and portal pressure (r > 0.8 for HVPG), our findings revealed a statistically significant but moderate correlation between spleen stiffness and PPG (r = 0.32). We speculate that this discrepancy may be primarily attributed to the fundamental differences between HVPG and PPG as measures of portal hypertension severity. The correlation between HVPG and PPG was notably weak (r = 0.145), with mean PPG levels approximately 10 mmHg higher than HVPG[19]. Although HVPG is commonly employed as a surrogate marker of portal hypertension, PPG may provide a more accurate reflection of actual portal pressure, particularly in the presence of extensive collateral circulation associated with decompensated cirrhosis, which can compromise the reliability of HVPG. Moreover, our study involved a substantially larger sample size than that of Buechter et al[13]. It uniquely included a longitudinal assessment of liver and spleen stiffness over a full year following TIPS. These long-term observations confer greater clinical relevance for predicting patient prognosis and evaluating the efficacy of the TIPS procedure. Collectively, these findings highlight the superior value of spleen stiffness as an indicator of cirrhosis progression. Although no prior studies have reported a direct relationship between PPG and spleen stiffness, earlier research has confirmed a positive correlation between HVPG and spleen stiffness[13,20], indirectly supporting this conclusion.
Patients with higher preoperative liver stiffness are at greater risk of mortality following TIPS, a finding consistent with previous reports. Jansen et al[9] reported that patients with elevated liver stiffness exhibited poorer prognoses, including shorter survival durations and increased incidence of decompensated events.
Our findings indicate that a high preoperative liver stiffness is positively correlated with increased post-TIPS mortality, a finding consistent with previous reports[9], emphasizing the clinical relevance of preoperative liver stiffness as
Based on the optimal cut-off value for liver STQ measurements, we observed a statistically significant difference in cumulative survival between the two groups, indicating that liver stiffness plays a critical role in predicting mortality among TIPS patients. Compared with previous studies, we found that the cut-off values for liver STQ in predicting mortality are not consistent[23-25]. In our study, the optimal liver stiffness cut-off value using STQ for predicting post-TIPS survival was 35.15 kPa. Compared with related findings, Ishikawa et al[22] reported that in patients with portal hypertension undergoing balloon-retrograde transvenous obliteration, those with liver stiffness < 28.18 kPa exhibited greater improvement in MELD-Na scores. Although this cut-off value is lower than ours, it remains relatively close. We speculate that several factors may account for this difference: (1) The outcomes predicted differed-our study focused on mortality risk, a more severe endpoint than liver function deterioration; (2) The surgical procedures varied between studies; (3) Baseline liver stiffness in that study was lower than in ours (21.7 kPa vs 23.5 kPa); and (4) The methods used for liver stiffness measurement differed, highlighting the need to account for detection technique variability when comparing cut-off values across studies. Additionally, two other studies reported substantially lower liver stiffness thresholds. Braude et al[26] found that liver stiffness > 10 kPa in patients with compensated liver disease was positively associated with mortality. Similarly, Fung et al[27] demonstrated that liver stiffness ≥ 10 kPa in chronic hepatitis B patients significantly correlated with increased mortality risk. The liver stiffness values reported in these two studies are much lower than our cut-off of 35.15 kPa. This discrepancy is primarily due to differences in baseline liver stiffness characteristics among the study populations. It is well established that patients undergoing TIPS are in the decom
This study has several limitations. First, as a single-center investigation with a relatively small sample size, the generalizability of the findings is limited. This limitation may also explain why liver stiffness changes within one year after TIPS were only marginally significant. Nonetheless, our results provide important insights and a foundation for future studies. The role of spleen stiffness should be emphasized in evaluating cirrhosis progression, while preoperative liver stiffness assessment is valuable for guiding TIPS indications and informing prognostic care, thereby potentially improving survival outcomes. Second, patient follow-up in this study was not fully consistent. Due to variations in patient compliance and convenience, some patients were unable to adhere to fixed follow-up intervals at our center. This may have introduced discrepancies in the timing of liver and spleen stiffness assessments. To address this, we grouped patients with similar follow-up intervals wherever possible. The semi-random nature of this grouping also helped mitigate potential bias in our conclusions. This approach may partially explain the marginal statistical significance observed in liver stiffness changes during the first post-TIPS year. To accurately capture the overall trend despite missing time-point data, we applied a generalized estimating equation model to minimize the impact of such missing data on trend analysis. Finally, our study employed a single ultrasound modality to assess liver and spleen stiffness and did not include comparative evaluations of different elastography techniques. Nonetheless, the STQ method used in point SWE is a well-established and clinically validated technique for measuring both liver and spleen stiffness. Compared with traditional transient elastography, point SWE offers several advantages: (1) It allows simultaneous evaluation of liver and spleen stiffness, providing the convenience of integrated measurement and standardized quality control; and (2) It has a high success rate and is less affected by patient factors such as obesity and ascites. In comparison with magnetic resonance elastography, point SWE also offers greater affordability, making it more feasible for widespread clinical screening applications. However, to better understand the accuracy and specificity of these three techniques in evaluating liver and spleen stiffness, comparative studies are indeed warranted.
Spleen stiffness exhibited a significant change following TIPS and demonstrated a positive correlation with PPG, suggesting that spleen stiffness may serve as a more reliable indicator of disease status. Additionally, preoperative liver stiffness was positively correlated with post-TIPS mortality risk, indicating its potential value as a prognostic marker for survival outcomes in patients undergoing TIPS.
| 1. | Rajesh S, George T, Philips CA, Ahamed R, Kumbar S, Mohan N, Mohanan M, Augustine P. Transjugular intrahepatic portosystemic shunt in cirrhosis: An exhaustive critical update. World J Gastroenterol. 2020;26:5561-5596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 86] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 2. | Wang P, Hu X, Xie F. Predictive value of liver and spleen stiffness measurement based on two-dimensional shear wave elastography for the portal vein pressure in patients with compensatory viral cirrhosis. PeerJ. 2023;11:e15956. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 3. | Corpechot C, Carrat F, Gaouar F, Chau F, Hirschfield G, Gulamhusein A, Montano-Loza AJ, Lytvyak E, Schramm C, Pares A, Olivas I, Eaton JE, Osman KT, Dalekos G, Gatselis N, Nevens F, Cazzagon N, Zago A, Russo FP, Abbas N, Trivedi P, Thorburn D, Saffioti F, Barkai L, Roccarina D, Calvaruso V, Fichera A, Delamarre A, Medina-Morales E, Bonder A, Patwardhan V, Rigamonti C, Carbone M, Invernizzi P, Cristoferi L, van der Meer A, de Veer R, Zigmond E, Yehezkel E, Kremer AE, Deibel A, Dumortier J, Bruns T, Große K, Pageaux GP, Wetten A, Dyson J, Jones D, Chazouillères O, Hansen B, de Lédinghen V; Global & ERN Rare-Liver PBC Study Groups. Liver stiffness measurement by vibration-controlled transient elastography improves outcome prediction in primary biliary cholangitis. J Hepatol. 2022;77:1545-1553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 59] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 4. | Gawrieh S, Vilar-Gomez E, Wilson LA, Pike F, Kleiner DE, Neuschwander-Tetri BA, Diehl AM, Dasarathy S, Kowdley KV, Hameed B, Tonascia J, Loomba R, Sanyal AJ, Chalasani N; NASH Clinical Research Network. Increases and decreases in liver stiffness measurement are independently associated with the risk of liver-related events in NAFLD. J Hepatol. 2024;81:600-608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 5. | Thorhauge KH, Semmler G, Johansen S, Lindvig KP, Kjærgaard M, Hansen JK, Torp N, Hansen CD, Andersen P, Hofer BS, Gu W, Israelsen M, Mandorfer M, Reiberger T, Trebicka J, Thiele M, Krag A; Microb-Predict, Galaxy and MicrobLiver consortia. Using liver stiffness to predict and monitor the risk of decompensation and mortality in patients with alcohol-related liver disease. J Hepatol. 2024;81:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Reference Citation Analysis (0)] |
| 6. | Singh S, Fujii LL, Murad MH, Wang Z, Asrani SK, Ehman RL, Kamath PS, Talwalkar JA. Liver stiffness is associated with risk of decompensation, liver cancer, and death in patients with chronic liver diseases: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2013;11:1573-84.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 239] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 7. | Gidener T, Ahmed OT, Larson JJ, Mara KC, Therneau TM, Venkatesh SK, Ehman RL, Yin M, Allen AM. Liver Stiffness by Magnetic Resonance Elastography Predicts Future Cirrhosis, Decompensation, and Death in NAFLD. Clin Gastroenterol Hepatol. 2021;19:1915-1924.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 8. | Semmler G, Yang Z, Fritz L, Köck F, Hofer BS, Balcar L, Hartl L, Jachs M, Stopfer K, Schedlbauer A, Neumayer D, Maurer J, Müllner-Bucsics T, Simbrunner B, Scheiner B, Trauner M, Mandorfer M, Reiberger T, Bauer DJM. Dynamics in Liver Stiffness Measurements Predict Outcomes in Advanced Chronic Liver Disease. Gastroenterology. 2023;165:1041-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 52] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 9. | Jansen C, Möller P, Meyer C, Kolbe CC, Bogs C, Pohlmann A, Schierwagen R, Praktiknjo M, Abdullah Z, Lehmann J, Thomas D, Strassburg CP, Latz E, Mueller S, Rössle M, Trebicka J. Increase in liver stiffness after transjugular intrahepatic portosystemic shunt is associated with inflammation and predicts mortality. Hepatology. 2018;67:1472-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 10. | Attia D, Rodt T, Marquardt S, Hinrichs J, Meyer BC, Gebel M, Wacker F, Manns MP, Potthoff A. Shear wave elastography prior to transjugular intrahepatic portosystemic shunt may predict the decrease in hepatic vein pressure gradient. Abdom Radiol (NY). 2019;44:1127-1134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 11. | Murad Gutiérrez V, Romero Enciso JA. Liver elastography: what it is, how it is done, and how it is interpreted. Radiologia (Engl Ed). 2018;60:183-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Rössle M, Haag K, Ochs A, Sellinger M, Nöldge G, Perarnau JM, Berger E, Blum U, Gabelmann A, Hauenstein K. The transjugular intrahepatic portosystemic stent-shunt procedure for variceal bleeding. N Engl J Med. 1994;330:165-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 522] [Cited by in RCA: 475] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 13. | Buechter M, Manka P, Theysohn JM, Reinboldt M, Canbay A, Kahraman A. Spleen stiffness is positively correlated with HVPG and decreases significantly after TIPS implantation. Dig Liver Dis. 2018;50:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 14. | Lu Y, Tang W, Zhang H, Liu J, Zhong S. Effect of hepatocyte damage in hepatic fibrogenesis of patients infected with Schistosoma japonicum. Infect Immun. 2024;92:e0002624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 15. | Wang J, Wang Q, Yu G, She Q, Zhang W, Zhang J. Correlation Between Liver Stiffness Measured by Shear Wave Elastography and Child-Pugh Classification. J Ultrasound Med. 2018;37:2191-2199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Foschi FG, Domenicali M, Giacomoni P, Dall'Aglio AC, Conti F, Borghi A, Bevilacqua V, Napoli L, Mirici F, Cucchetti A, Ercolani G, Gardini AC, Bellentani S, Gastaldelli A, Giuffrè M, Tiribelli C, Bedogni G; Bagnacavallo Study Group. Is there an association between commonly employed biomarkers of liver fibrosis and liver stiffness in the general population? Ann Hepatol. 2020;19:380-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Giunta M, La Mura V, Conti CB, Casazza G, Tosetti G, Gridavilla D, Segato S, Nicolini A, Primignani M, Lampertico P, Fraquelli M. The Role of Spleen and Liver Elastography and Color-Doppler Ultrasound in the Assessment of Transjugular Intrahepatic Portosystemic Shunt Function. Ultrasound Med Biol. 2020;46:1641-1650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 18. | Jeong SW. Liver stiffness measurement: is it a non-invasive substitution for HVPG? Clin Mol Hepatol. 2013;19:367-369. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 19. | Wang L, Song QK, Yue ZD, Zhao HW, Fan ZH, Wu YF, Liu FQ, Meng K, Zhang L, Jiang HG, Ding YN, Zhang Y. [Study on the correlation between PPG and HVPG in patients with portal hypertension]. Zhonghua Gan Zang Bing Za Zhi. 2022;30:722-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 20. | Song J, Huang J, Huang H, Liu S, Luo Y. Performance of spleen stiffness measurement in prediction of clinical significant portal hypertension: A meta-analysis. Clin Res Hepatol Gastroenterol. 2018;42:216-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 21. | Peng Y, Qi X, Guo X. Child-Pugh Versus MELD Score for the Assessment of Prognosis in Liver Cirrhosis: A Systematic Review and Meta-Analysis of Observational Studies. Medicine (Baltimore). 2016;95:e2877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 344] [Cited by in RCA: 343] [Article Influence: 38.1] [Reference Citation Analysis (0)] |
| 22. | Ishikawa T, Egusa M, Fujioka T, Nishiyama N, Kawamoto D, Sasaki R, Nishimura T, Tanabe N, Oono T, Saeki I, Takami T. A combination of liver stiffness and international normalized ratio is an ideal prognostic predictor of portosystemic shunt occlusion in patients with portal hypertension. J Gastroenterol. 2023;58:246-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 23. | Sporea I, Sirli R, Deleanu A, Tudora A, Curescu M, Cornianu M, Lazar D. Comparison of the liver stiffness measurement by transient elastography with the liver biopsy. World J Gastroenterol. 2008;14:6513-6517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 35] [Cited by in RCA: 39] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 24. | Wong GL. Update of liver fibrosis and steatosis with transient elastography (Fibroscan). Gastroenterol Rep (Oxf). 2013;1:19-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 87] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 25. | Ferraioli G, Tinelli C, Malfitano A, Dal Bello B, Filice G, Filice C; Liver Fibrosis Study Group, Above E, Barbarini G, Brunetti E, Calderon W, Di Gregorio M, Lissandrin R, Ludovisi S, Maiocchi L, Michelone G, Mondelli M, Patruno SF, Perretti A, Poma G, Sacchi P, Zaramella M, Zicchetti M. Performance of real-time strain elastography, transient elastography, and aspartate-to-platelet ratio index in the assessment of fibrosis in chronic hepatitis C. AJR Am J Roentgenol. 2012;199:19-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 47] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 26. | Braude M, Roberts S, Majeed A, Lubel J, Prompen J, Dev A, Sievert W, Bloom S, Gow P, Kemp W. Liver stiffness (Fibroscan®) is a predictor of all-cause mortality in people with non-alcoholic fatty liver disease. Liver Int. 2023;43:90-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 27. | Fung J, Lai CL, Seto WK, Wong DK, Yuen MF. Prognostic significance of liver stiffness for hepatocellular carcinoma and mortality in HBeAg-negative chronic hepatitis B. J Viral Hepat. 2011;18:738-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |