Published online Aug 27, 2025. doi: 10.4240/wjgs.v17.i8.108692
Revised: May 30, 2025
Accepted: July 10, 2025
Published online: August 27, 2025
Processing time: 126 Days and 23 Hours
Esophageal bronchogenic cysts (EBCs) are usually discovered incidentally during radiologic or endoscopic examinations. They are rare and prone to misdiagnosis or mistreatment. As a submucosal lesion, the endoscopic ultrasonography (EUS) characteristics of EBCs are unclear.
To analyze the clinicopathological and EUS characteristics of EBCs.
A total of 22 patients with a histological diagnosis of EBCs who underwent EUS examination were retrospectively included. The clinicopathological and EUS fea
Most of the EBCs were asymptomatic, and no malignant transformation or pre
When a submucosal lesion located at the lower esophagus originates from the intrinsic muscle layer, the possibility of EBCs should be noted, the EUS characteristics of which include a hypoecho with a clear boundary and a posterior wall enhancement, a floating echo inside and no blood flow signal, a yellow-green or green pattern on elastography, and no enhancement on contrast EUS.
Core Tip: Esophageal bronchogenic cysts (EBCs) are usually discovered incidentally during radiologic or endoscopic examinations. They are rare and prone to misdiagnosis or mistreatment. We retrospectively analyzed the clinicopathological and endoscopic ultrasonography features of 22 patients with EBCs. Our findings emphasize the critical role of endoscopic ultrasonography for the preoperative diagnosis and treatment of EBCs.
- Citation: Shan GD, Ning LG, Zhang FM, Du HJ, Chen WG, Wang JJ, Li AQ, Xu GQ, Chen HT. Clinicopathological and endoscopic ultrasonography characteristics of esophageal bronchogenic cysts: A single-center study. World J Gastrointest Surg 2025; 17(8): 108692
- URL: https://www.wjgnet.com/1948-9366/full/v17/i8/108692.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i8.108692
Bronchogenic cysts are among the most common bronchopulmonary malformations that arise from abnormal budding of the primitive tracheobronchial tube[1,2]. The location of the cysts depends on the embryological stage of development at which abnormal budding occurs. Approximately 36%-90% of bronchogenic cysts occur in the mediastinum, and most of the remaining cysts involve the lung parenchyma[3]. Bronchogenic cysts have been reported to occur in other unusual locations, such as the pericardium[4], thymus[5], diaphragm[6], retroperitoneum[7], and cervical region[8].
Esophageal bronchogenic cysts (EBCs) are rare, and most of these lesions have been reported as individual case reports[9-12]. There are differences in reports describing their clinical features and uncertainties regarding the optimal treatment strategy[13-15]. While there may be no symptoms when lesion is relatively small, they may occur when the cyst is large, leading to narrowing of the esophagus and obstacles in food passage. Common symptoms include dysphagia, regurgi
Currently, preoperative diagnostic methods include mainly clinical imaging and endoscopy. Clinical imaging, such as computed tomography (CT) and magnetic resonance imaging (MRI), can provide information on the size, quantity, and location of the cyst in the esophageal wall. On preoperative CT, EBCs presented as well-defined, thin-wall cystic lesions with varying densities, and no enhancement of the lesions was found after the administration of contrast agent[1]. However, CT and MRI scans have difficulty detecting and displaying lesions smaller 1 cm. In previous studies, most intramural EBCs presented as subepithelial lesions under endoscopy[9,18]. Conventional endoscopic examination can provide only superficial information about protruding lesions, and endoscopic biopsy is of little significance for most cases, as the lesions present as submucosal lesions. By scanning with an ultrasound probe, endoscopic ultrasonography (EUS) can obtain information about the origin, internal echoes, blood supply, and relationship with the surrounding organs of the cysts. Nevertheless, the role of EUS in the diagnosis of EBCs has rarely been explored[19]. In the present study, we aimed to analyze the clinicopathological and EUS characteristics of EBCs.
Inpatients with a histological diagnosis of EBCs postoperatively were screened through the pathology reports from the Zhejiang University School of Medicine First Affiliated Hospital between 2015 and 2024. Among them, patients without EUS examination or absent of EUS images were excluded. Twenty-two patients with a histological diagnosis of EBCs were included finally. The study was approved by the Ethics Committee of the Zhejiang University School of Medicine First Affiliated Hospital. Baseline demographic and clinicopathological characteristics, including clinical symptoms, sex, age at diagnosis, histopathology, and EUS and CT/MRI images, were collected. All of the included patients had complete EUS images for further analysis.
EUS was performed by experienced endoscopists at the Zhejiang University School of Medicine First Affiliated Hospital. Images were captured and stored in the system The EUS system included Olympus EU-ME2 sonogram processing equipment, an Olympus MAJ-1720 drive system, UM-DP1225R miniature ultrasonic probes with a frequency of 12 MHz, and an Olympus GF-UE260 echoendoscope (Olympus Medical System, Tokyo, Japan). Miniature ultrasonic probes were used in eight patients, and radial echoendoscopy was performed in fourteen patients. EUS were performed by endoscopic experts (over 5 years of EUS diagnosis experience). During miniature ultrasonic examination, the water immersion method was employed, allowing water to cover the lesion and serve as a medium for ultrasound. The size, location, origin, echogenic features, boundary, and blood signal were investigated. All of the EUS images were evaluated independently by 2 physicians with extensive EUS experience (over 10 years of EUS diagnosis experience). Discrepancies were resolved and came to an agreement after discussion with another physician with extensive EUS experience (over 10 years of EUS diagnosis experience).
For contrast-enhanced EUS (CE-EUS), sulfur hexafluoride microbubble (SonoVue) was used as an ultrasound contrast agent. After the SonoVue was injected into a peripheral vein, ultrasound waves disrupt it and produce a signal. An enhancement pattern of blood perfusion was observed[20]. Real-time elastography was performed using a region of interest that included at least 50% of the lesion, as well as surrounding structures. In accordance with standards reported in the international literature, red represents soft tissue, green/yellow represents intermediate organization, and blue represents hard organization[21].
Patient demographics, clinical presentations, CT results, endoscopic and EUS results, and histological information were collected and analyzed. SPSS (IBM SPSS Statistics 26; SPSS Inc., Chicago, IL, United States) was used for statistical analysis. The results are expressed as the means ± SD or the numbers (percentages).
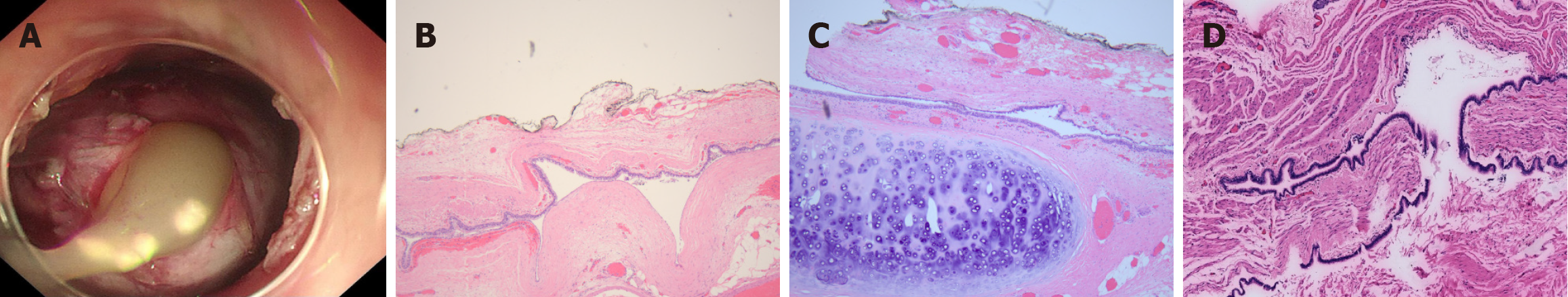
Twenty-two patients with EBCs (mean age 47.0 ± 12.2 years) were included. The male/female ratio was 13/9. Six patients (6/22) presented symptoms, including epigastric distension (2 patients), epigastric discomfort (1 patient), and dysphagia (3 patients). Sixteen patients were asymptomatic, and their cysts were incidentally discovered via abdominal ultrasound, pulmonary CT examination or gastroscopy. The EBCs were predominantly located in the lower esophagus (72.7%, 16/22), with 27.3% (6/22) in the middle esophagus. Postoperative histological diagnoses were derived from 14 patients who underwent endoscopic treatment (2 patients with endoscopic mucosal resection, 12 patients with submucosal tunnel endoscopic resection) and 8 patients with surgery (Table 1). Visible outflow of cystic mucus during operation was identified in 14 patients. The cystic fluid was composed mainly of white (2 patients), yellow (7 patients), and brown (5 patients) cloudy mucus. A typical highly proteinaceous and mucoid yellow content was observed (Figure 1A).
| Characteristics | n (%) |
| Age | 47.0 ± 12.2 |
| Male/female | 13/9 |
| Symptoms | |
| Epigastric distension | 2 (9.1) |
| Epigastric discomfort | 1 (4.5) |
| Dysphagia | 3 (13.7) |
| Asymptomatic | 16 (72.7) |
| Location | |
| Lower esophagus | 16 (72.7) |
| Middle esophagus | 6 (27.3) |
| Treatment | |
| Endoscopic treatment | 14 (63.6) |
| EMR | 2 (14.2) |
| STER | 12 (85.8) |
| Surgery | 8 (36.3) |
| Color of cystic fluid | |
| White | 2 (14.2) |
| Yellow | 7 (50) |
| Brown | 5 (35.8) |
| Pathology | |
| Ciliated columnar epithelium | 22 (100) |
| Smooth muscles | 16 (72.7) |
| Bronchial glands | 8 (36.3) |
| Cartilage | 3 (13.7) |
Histopathology revealed respiratory ciliated columnar epithelium-lined cyst walls with underlying smooth muscles (Figure 1B) and bronchial glands. In three patients, cartilage was identified (Figure 1C). A typical pathological image showed that the lesions was originated from muscularis propria (Figure 1D). No malignant transformation or precancerous changes were discovered via pathological analysis.
Eighteen patients received enhanced CT examination, three received plain CT examination, and one received enhanced MRI examination. None of the patients were preoperatively diagnosed with EBCs by CT or MRI. On CT examination, EBCs presented as suborbicular or oval low-density masses in 16 patients (Supplementary Figure 1A) and slight high-density masses in 2 patients. In one patient, plain CT revealed thickening of the esophageal wall. In two patients, lesions smaller than 1 cm were not detected on CT. The average CT value of the plain scan was 42.4 ± 16.1 HU, whereas the average enhanced CT value was 42.6 ± 13.2 HU. There was slight enhancement in two patients and no enhancement of the lesion in the other sixteen patients after the administration of contrast agent. Two patients with calcifications were identified on CT (Supplementary Figure 1B). Esophageal compression was detected in four patients (Supplementary Figure 1C).
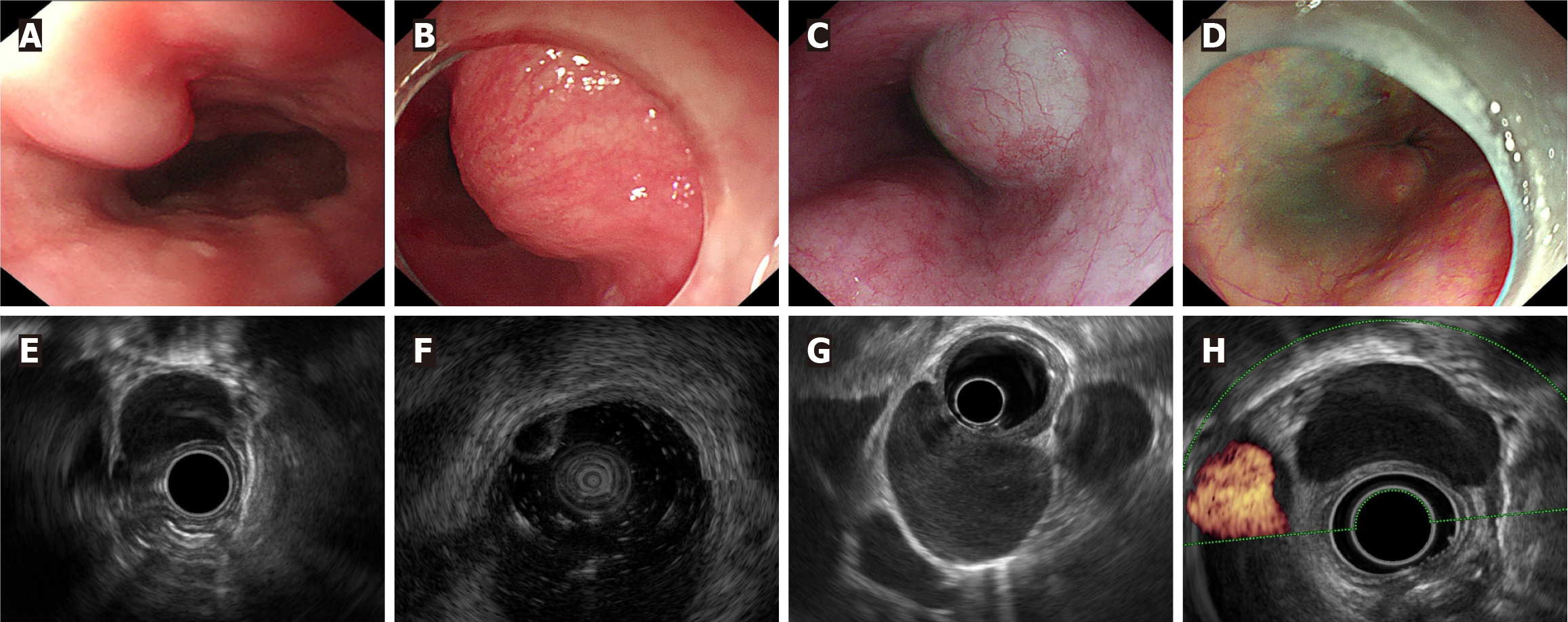
All patients had a single lesion in the esophagus. Endoscopy revealed submucosal lesions in 20 patients, which were covered by normal esophageal mucosa. The endoscopic morphology of the EBCs varied. Six patients had oblong flat bulging lesions (Figure 2A), three had nodular bulging lesions, seven had hemisphere lesions (Figure 2B), and three had dumbbell bulging lesions (Figure 2C). However, in two patients, no lesions were found in the esophageal cavity (Figure 2D).
The average diameter of the lesions on EUS was 3.12 ± 1.25 cm. A total of 81.8% (18/22) of the EBCs presented a predominant extraluminal growth pattern (Table 2). A total of 90.9% (20/22) of the EBCs originated from the muscularis propria (Figure 2E), and 9.1% (2/22) originated from the submucosa (Figure 2F). All of the lesions had clear boundaries. In terms of echo, 77.3% (17/22) were hypoechoic, and 22.7% (5/22) were anechoic. A total of 81.8% (18/22) of the patients had homogeneous echoes (Figure 2G), and 18.2% (4/22) had inhomogeneous echoes. Fourteen patients underwent color Doppler, and no blood flow signal was identified (100%, 14/14) (Figure 2H).
| EUS characteristics | n (%) |
| Average lesion size (cm) | 3.12 ± 1.25 |
| Origin | |
| Muscularis propria | 20 (90.9) |
| Submucosa | 2 (9.1) |
| Growth pattern | |
| Predominant extraluminal growth | 18 (81.8) |
| Intraluminal growth | 4 (18.2) |
| Margin | |
| Smooth and clear | 22 (100) |
| Indistinct | 0 (0) |
| Blood flow signal | 0 (0) |
| Echo | |
| Hypoecho | 17 (77.3) |
| Anecho | 5 (22.7) |
| Punctiform hyperecho | 10 (45.5) |
| Flocculent hypoecho | 8 (36.4) |
| Posterior wall enhancement | 10 (45.5) |
| EUS elastography | 6 |
| Yellow-green or green | 6 (100) |
| Blue | 0 (0) |
| CE-EUS | |
| Enhancement | 0 (0) |
| No enhancement | 5 (100) |
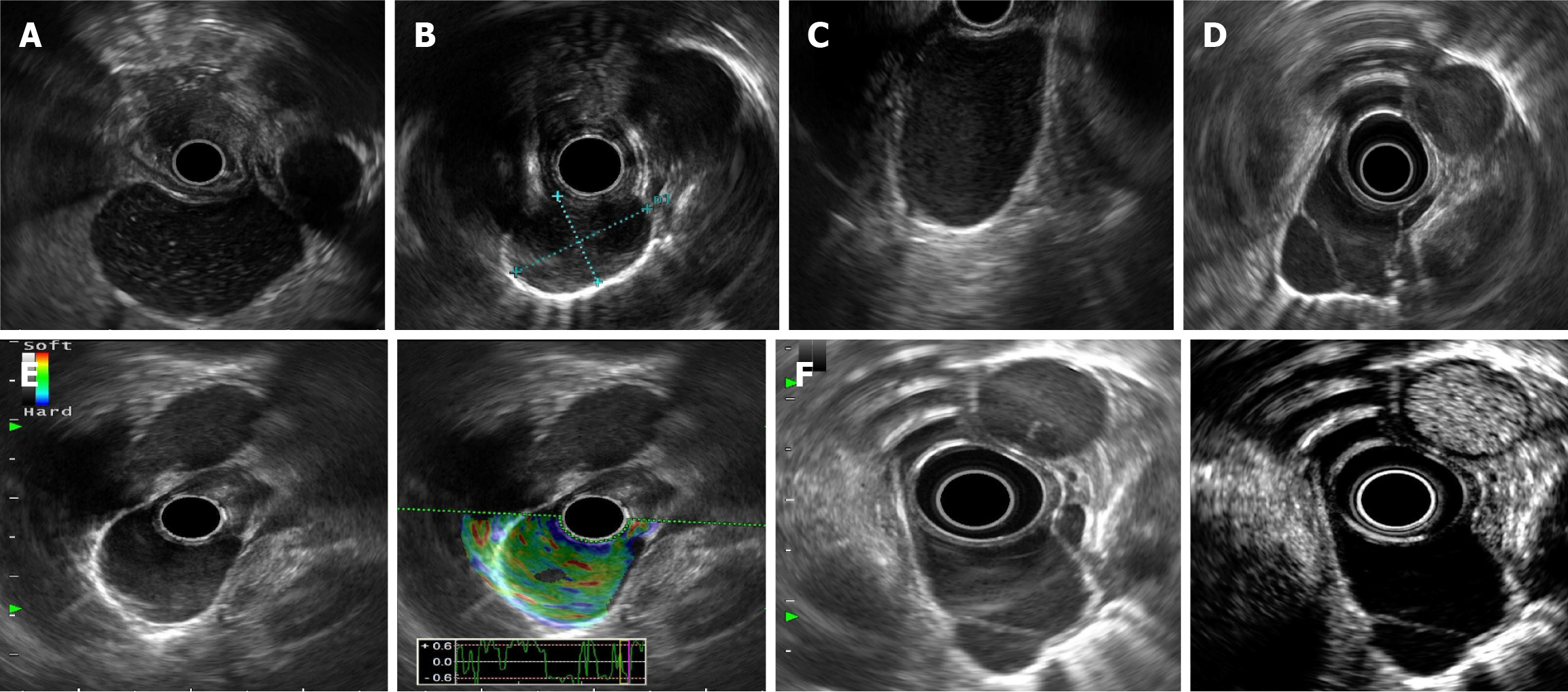
We found floating echoes (presented as a punctiform hyperecho or a flocculent hypoecho) inside the lesion. Among them, 45.5% (10/22) presented with a punctiform hyperecho inside the lesion (Figure 3A), and 36.4% (8/22) of the EBCs presented with a flocculent hypoecho (Figure 3B). A total of 45.5% (10/22) of the patients had a posterior wall enhancement (Figure 3C). In three patients, a separable zone was visible in the capsule cavity (Figure 3D). Six patients with EBCs underwent elasticity imaging, which revealed yellow–green or green patterns (100%, 6/6) (Figure 3E). Five patients with EBCs underwent CE-EUS and showed no enhancement (100%, 5/5) (Figure 3F). Invisible enhancement in the separable zone and capsule wall can be identified.
EBCs are rare, and most of these lesions have been reported as individual case reports[9,15,22]. The patients with EBCs described in the literature were all diagnosed through postoperative pathology. Preoperative diagnosis is complicated because of the low incidence rate and insufficient understanding of EBCs. In the present study, we summarized the EUS characteristics of EBCs and identified several features that are useful for the preoperative diagnosis of EBCs.
We found that the lesions were located in the lower or middle esophagus, which is similar to the findings of previous studies[1,18]. Nevertheless, most of our patients were asymptomatic, even if the lesion was large. One of the reasons may be that the extracellular growth pattern of EBCs and symptoms associated with food passage obstacles are not obvious. Malignant transformation might occur due to prolonged inflammatory responses and metaplastic changes in broncho
None of the patients in our study were diagnosed with EBCs by CT or MRI preoperatively in our study. In two pa
Under EUS, 90.9% (20/22) of EBCs were found to have originated from the muscularis propria. Most of the lesions are hypoechoic and have homogeneous echoes, making it difficult to distinguish them from other hypoechoic solid lesions, such as esophageal leiomyomas. The content of protein and mucus within the cystic fluid determines the appearance of EUS to some extent. If the contents of protein and mucus secreted by the glandular epithelium of an EBC are high, the lesion under EUS is found to be hypoechoic. If the protein and mucus contents are low, it is found to be anechoic. We also detected a posterior wall enhancement in 45.5% (10/22) of the patients. Posterior wall enhancement often occurs on the posterior wall of cysts, abscesses, and other fluid areas but is almost absent from the posterior wall of vascular lumens[23]. Therefore, the presence of posterior wall enhancement indicates fluid content inside the lesion. Interestingly, a punctiform hyperecho and a flocculent low echo were also detected via EUS. We speculate that the punctiform hyperecho may originate from pigments or crystal crystals in the cystic fluid. Furthermore, owing to the uneven distribution of highly proteinaceous and mucoid content, flocculent low echoes can be observed. There is no vascular distribution inside the cyst, resulting in no blood flow signal on color Doppler. Although EUS-guided fine needle aspiration (EUS-FNA) can facilitate diagnosis through the acquisition of ciliated epithelial cells through liquid cytology, the diagnostic yield of EUS-FNA is limited in EBCs[19,24] and is subject to infection. Thus, EUS-FNA is not advocated for EBCs. By identifying these specific features, EUS helps to the diagnosis of EBCs and can distinguish EBCs from other esophageal masses, such as leiomyomas. This capability is something CT/MRI cannot replace. Therefore, EUS should be recommended as a first-line investigation for suspected EBCs.
Elastography is a real-time technique that involves the analysis of tissue stiffness[25]. Changes in tissue stiffness can be associated with various pathologies, including benign and malignant diseases. EUS-guided elastography is primarily used in the evaluation of solid pancreatic lesions[26,27] and enlarged lymph nodes[27,28] and is promising for differentiating cancer lesions from inflammatory lesions[29]. It is also used to distinguish gastrointestinal stromal tumors (GISTs) from other submucosal tumors (SMTs)[30]. Ignee et al[21] reported that 98% of gastric GISTs and 80% of gastric leiomyomas showed a blue pattern on elastography[21]. However, to our knowledge, EUS-guided elastography has not been reported for EBCs. In the present study, six EBCs presented yellow–green or green patterns on elastography. Owing to the proteinaceous and mucoid content inside the lesion, the stiffness of EBCs is usually soft, which may explain the green or yellow pattern on elastography of EBCs. This characteristic may be used to exclude other esophageal SMTs, such as esophageal leiomyomas, esophageal schwannomas, and esophageal stromal tumors.
CE-EUS was specifically developed for contrast-enhanced harmonic imaging and enables the observation of microcirculation and parenchymal perfusion in tumors. A previous systematic review revealed that CE-EUS is useful for the differential diagnosis and risk stratification of SMTs[31]. Kamata et al[32] performed CE-EUS in 58 GISTs and 15 benign SMTs. They revealed that CE-EUS discriminates GISTs from benign lesions with good accuracy. Nevertheless, CE-EUS has not been reported for EBCs. In the present study, we found no enhancement in 100% (5/5) of the EBCs. Owing to the proteinaceous or mucoid content and lack of vascular components inside the cavity, no enhancement can be detected after the injection of SonoVue. We found hypo-enhancement in the separable zone and capsule wall. These characteristics on CE-EUS may be used to differentiate esophageal lesions from solid lesions.
Definitive differentiation of EBCs from other submucosal lesions, especially esophageal leiomyomas, may be difficult. An esophageal leiomyoma is the most common submucosal lesion of the esophagus. Our previous study revealed that the location of leiomyomas in the esophagus is even in the upper, middle or lower esophagus[33]. They mainly originate from the mucosal muscularis or propria muscularis and show homogeneous hypoecho signals on EUS[34]. Although a punctiform hyperecho can be detected on EUS due to calcification inside the leiomyoma, the punctiform hyperecho is fixed and not affected by body position or ultrasound probe movement. Esophageal leiomyomas are hypoenhanced on CE-EUS[35] and blue on EUS elastography[30], which can be used to differentiate them from EBCs.
Both miniature ultrasonic probes and radial echoendoscopy were used in our study. The observation range of mi
EUS-based diagnosis and differentiation can guide the management of EBCs. For lesions suspected to be EBCs on EUS: If the lesion is predominantly within the muscularis propria, endoscopic resection may be recommended. If the lesion extends mostly into the mediastinum, surgical excision is the preferred approach. Additionally, given that EBCs have no reported malignant potential, cystic fluid aspiration or drainage endoscopically may be considered for EUS-suspected EBCs, avoiding unnecessary radical surgical resection. This underscores the critical role of preoperative EUS assessment in optimizing therapeutic strategies. Curative resection of EBCs by endoscopic resection poses significant challenges. In this study, 8 cases achieved an en-bloc and R0 resection, while 6 cases underwent partial resection due to the tendency for intraoperative rupture, which resulted in loss of tension and complicated complete dissection. However, partial resection combined with cauterization remains effective in preventing recurrence, which will be discussed in our next article (unpublished).
As this was a retrospective study, the lesions were dissected from the esophageal wall via endoscopic or surgical procedures rather than complete resection of the entire esophageal wall. Consequently, postoperative pathology be used to fully evaluate the origin of lesions. However, we observed that intraoperative findings could supplement post
We acknowledge that several limitations need to be addressed in our study. First, this was a single-center retrospective study, and only a small number of patients were included. Second, EUS is associated with the risk of interobserver variation depending on observer proficiency and subjective judgment. Miniature ultrasound probes and radial echoendoscopy were used in different patients. However, images from the EBC patients were further read by a skilled EUS expert to avoid interobserver variation. Third, we did not directly compare EBCs with other submucosal lesions of the esophagus. Further studies comparing the EUS features of EBCs with those of other submucosal lesions are needed to aid in the differential diagnosis.
Most EBCs are asymptomatic, and no malignant transformation or precancerous changes are found histologically. When a submucosal lesion located at the lower esophagus originates from the intrinsic muscle layer, the possibility of EBCs should be noted. The EUS characteristics of EBCs include a hypoecho with a clear boundary and posterior wall enhancement, a floating echo inside and no blood flow signal, a yellow-green or green pattern on elastography, and no enhancement on CE-EUS. By effectively understanding the EUS characteristics of EBCs, their preoperative diagnosis becomes possible. Therefore, EUS may become a useful investigational modality for the preoperative diagnosis and treatment of EBCs.
The authors thank all the patients who provided their data at the First Affiliated Hospital of Zhejiang University who contributed to this study.
| 1. | Ko SF, Hsieh MJ, Lin JW, Huang CC, Li CC, Cheung YC, Ng SH. Bronchogenic cyst of the esophagus: clinical and imaging features of seven cases. Clin Imaging. 2006;30:309-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Turkyilmaz A, Eroglu A, Subasi M, Findik G. Intramural esophageal bronchogenic cysts: a review of the literature. Dis Esophagus. 2007;20:461-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 3. | Aktoğu S, Yuncu G, Halilçolar H, Ermete S, Buduneli T. Bronchogenic cysts: clinicopathological presentation and treatment. Eur Respir J. 1996;9:2017-2021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 102] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | Escalante JM, Molina G, Rincón FM, Acosta Buitrago LM, Perez Rivera CJ. Giant Intrapericardial bronchogenic cyst associated with congestive heart failure and atrial fibrillation: a case report. J Cardiothorac Surg. 2021;16:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 5. | Nair V, Moran CA. Unilocular thymic cyst with features of intra-thymic bronchogenic cyst: A clinicopathological correlation of 4 cases. Pathol Res Pract. 2022;238:154121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Tang J, Zeng Z, Deng S, Lin F. Ectopic bronchogenic cyst arising from the diaphragm: a rare case report and literature review. BMC Surg. 2021;21:321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 7. | Hu BY, Yu H, Shen J. A retroperitoneal bronchogenic cyst clinically mimicking an adrenal mass: three case reports and a literature review. J Int Med Res. 2022;50:3000605211072664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 8. | Teissier N, Elmaleh-Bergès M, Ferkdadji L, François M, Van den Abbeele T. Cervical bronchogenic cysts: usual and unusual clinical presentations. Arch Otolaryngol Head Neck Surg. 2008;134:1165-1169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Zhang FM, Chen HT, Ning LG, Xu Y, Xu GQ. Esophageal bronchogenic cyst excised by endoscopic submucosal tunnel dissection: A case report. World J Clin Cases. 2020;8:353-361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Xiang J, Linghu E, Chai N. Case of large intra-esophageal bronchogenic cyst treated with lauromacrogol ablation. Dig Endosc. 2020;32:435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Tang X, Jiang B, Gong W. Endoscopic submucosal tunnel dissection of a bronchogenic esophageal cyst. Endoscopy. 2014;46 Suppl 1 UCTN:E626-E627. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Ceniceros-Cabrales AP, Sánchez-Fernández P. [Esophageal bronchogenic cyst: an uncommon cause of dysphagia in adults. Case report and literature review]. Cir Cir. 2018;86:187-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Xiang J, Linghu E, Li L, Chai N. Endoscopic combined management of a rare esophageal bronchogenic cyst. Dig Liver Dis. 2021;53:125-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Matsumoto R, Sasaki K, Noda M, Tsuruda Y, Kita Y, Uchikado Y, Matsushita D, Arigami T, Mori S, Ohtsuka T. Successful thoracoscopic resection of an esophageal bronchogenic cyst. Gen Thorac Cardiovasc Surg. 2022;70:100-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Chen L, Ye L, Hu B. Gastrointestinal: Endoscopic submucosal tunnel dissection for an esophageal bronchogenic cyst. J Gastroenterol Hepatol. 2023;38:9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Patel SR, Meeker DP, Biscotti CV, Kirby TJ, Rice TW. Presentation and management of bronchogenic cysts in the adult. Chest. 1994;106:79-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 119] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 17. | St-Georges R, Deslauriers J, Duranceau A, Vaillancourt R, Deschamps C, Beauchamp G, Pagé A, Brisson J. Clinical spectrum of bronchogenic cysts of the mediastinum and lung in the adult. Ann Thorac Surg. 1991;52:6-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 208] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 18. | Jiao J, Fan X, Luo L, Zheng Z, Wang B, Liu W. Endoscopic resection in the treatment of intramural esophageal bronchogenic cysts: A retrospective analysis of 17 cases. Dig Liver Dis. 2022;54:1691-1697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 19. | Han C, Lin R, Yu J, Zhang Q, Zhang Y, Liu J, Ding Z, Hou X. A Case Report of Esophageal Bronchogenic Cyst and Review of the Literature With an Emphasis on Endoscopic Ultrasonography Appearance. Medicine (Baltimore). 2016;95:e3111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Lefort C, Gupta V, Lisotti A, Palazzo L, Fusaroli P, Pujol B, Gincul R, Fumex F, Palazzo M, Napoléon B. Diagnosis of gastric submucosal tumors and estimation of malignant risk of GIST by endoscopic ultrasound. Comparison between B mode and contrast-harmonic mode. Dig Liver Dis. 2021;53:1486-1491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 21. | Ignee A, Jenssen C, Hocke M, Dong Y, Wang WP, Cui XW, Woenckhaus M, Iordache S, Saftoiu A, Schuessler G, Dietrich CF. Contrast-enhanced (endoscopic) ultrasound and endoscopic ultrasound elastography in gastrointestinal stromal tumors. Endosc Ultrasound. 2017;6:55-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 22. | Matsuda H, Ishida M, Miyasaka C, Michiura T, Inoue K, Sekimoto M, Tsuta K. Intramural bronchogenic cysts of the esophagus and gastroesophageal junction: A case report. Mol Clin Oncol. 2020;13:162-168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 23. | Quien MM, Saric M. Ultrasound imaging artifacts: How to recognize them and how to avoid them. Echocardiography. 2018;35:1388-1401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 24. | Cassiani J, Crinò SF, Manfrin E, Rivelli M, Gabbrielli A, Guglielmi A, Pedrazzani C. Endoscopic Ultrasound Through-the-Needle Biopsy for the Diagnosis of an Abdominal Bronchogenic Cyst. Clin Endosc. 2021;54:767-770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | Itoh A, Ueno E, Tohno E, Kamma H, Takahashi H, Shiina T, Yamakawa M, Matsumura T. Breast disease: clinical application of US elastography for diagnosis. Radiology. 2006;239:341-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1119] [Cited by in RCA: 1065] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 26. | Iglesias-Garcia J, Larino-Noia J, Abdulkader I, Forteza J, Dominguez-Munoz JE. EUS elastography for the characterization of solid pancreatic masses. Gastrointest Endosc. 2009;70:1101-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 124] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 27. | Giovannini M, Hookey LC, Bories E, Pesenti C, Monges G, Delpero JR. Endoscopic ultrasound elastography: the first step towards virtual biopsy? Preliminary results in 49 patients. Endoscopy. 2006;38:344-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 214] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 28. | Giovannini M, Thomas B, Erwan B, Christian P, Fabrice C, Benjamin E, Geneviève M, Paolo A, Pierre D, Robert Y, Walter S, Hanz S, Carl S, Christoph D, Pierre E, Jean-Luc VL, Jacques D, Peter V, Andrian S. Endoscopic ultrasound elastography for evaluation of lymph nodes and pancreatic masses: a multicenter study. World J Gastroenterol. 2009;15:1587-1593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 204] [Cited by in RCA: 201] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 29. | Iglesias-Garcia J, de la Iglesia-Garcia D, Lariño-Noia J, Dominguez-Muñoz JE. Endoscopic Ultrasound (EUS) Guided Elastography. Diagnostics (Basel). 2023;13:1686. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 30. | Tsuji Y, Kusano C, Gotoda T, Itokawa F, Fukuzawa M, Sofuni A, Matsubayashi J, Nagao T, Itoi T, Moriyasu F. Diagnostic potential of endoscopic ultrasonography-elastography for gastric submucosal tumors: A pilot study. Dig Endosc. 2016;28:173-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 31. | Fusaroli P, Napoleon B, Gincul R, Lefort C, Palazzo L, Palazzo M, Kitano M, Minaga K, Caletti G, Lisotti A. The clinical impact of ultrasound contrast agents in EUS: a systematic review according to the levels of evidence. Gastrointest Endosc. 2016;84:587-596.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 32. | Kamata K, Takenaka M, Kitano M, Omoto S, Miyata T, Minaga K, Yamao K, Imai H, Sakurai T, Watanabe T, Nishida N, Chikugo T, Chiba Y, Imamoto H, Yasuda T, Lisotti A, Fusaroli P, Kudo M. Contrast-enhanced harmonic endoscopic ultrasonography for differential diagnosis of submucosal tumors of the upper gastrointestinal tract. J Gastroenterol Hepatol. 2017;32:1686-1692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 33. | Jiang T, Yu J, Chen L, Chen H, Shan G, Yang M, Xu G. Clinical value of endoscopic ultrasonography for esophageal leiomyoma in elder patients. J Vis Surg. 2017;3:137. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 34. | Sun LJ, Chen X, Dai YN, Xu CF, Ji F, Chen LH, Chen HT, Chen CX. Endoscopic Ultrasonography in the Diagnosis and Treatment Strategy Choice of Esophageal Leiomyoma. Clinics (Sao Paulo). 2017;72:197-201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 35. | Kannengiesser K, Mahlke R, Petersen F, Peters A, Ross M, Kucharzik T, Maaser C. Contrast-enhanced harmonic endoscopic ultrasound is able to discriminate benign submucosal lesions from gastrointestinal stromal tumors. Scand J Gastroenterol. 2012;47:1515-1520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |