Published online Aug 27, 2025. doi: 10.4240/wjgs.v17.i8.107941
Revised: May 8, 2025
Accepted: July 7, 2025
Published online: August 27, 2025
Processing time: 146 Days and 23.6 Hours
Non-tumoral portal vein thrombosis (PVT) is a frequent and challenging com
Core Tip: Non-tumoral portal vein thrombosis (PVT) is a common but serious challenge in liver transplant candidates. It significantly affects postoperative outcomes, especially in advanced cases. A multidisciplinary approach is crucial for the management of PVT. Integrating anticoagulation, interventional radiology, and intraoperative strategies that are tailored to PVT severity is paramount. While early PVT cases can be managed with thrombectomy and portal vein reconstruction, complex cases may require alternative surgical techniques. This manuscript provides a comprehensive analysis of PVT management in patients requiring liver transplant by emphasizing evidence-based strategies and presenting our institutional protocol to optimize outcomes in this high-risk population.
- Citation: Rodrigues PD, Lazzarotto-da-Silva G, Feier FH, Grezzana Filho TJM, Kruel CRP, Leipnitz I, Chedid MF. Non-tumoral portal vein thrombosis in liver transplantation: Surgical perspectives and institutional protocol. World J Gastrointest Surg 2025; 17(8): 107941
- URL: https://www.wjgnet.com/1948-9366/full/v17/i8/107941.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i8.107941
Non-tumoral portal vein thrombosis (PVT) is observed in up to 26% of patients with cirrhosis on the liver transplant waiting list. The prevalence of PVT increases as liver disease progresses[1]. While most cases involve partial thrombosis, complete portal vein occlusion occurs in approximately 44% of cases[2]. Prospective studies evaluating patients for liver transplantation have reported an incidence of PVT as high as 24%[3]. The clinical significance of PVT lies in its substantial impact on patient prognosis, often necessitating specific management strategies. A recent study reported a postoperative mortality rate of 30% in patients with advanced PVT[4], underscoring the importance of optimized treatment approaches.
Effective management of PVT demands a multidisciplinary approach via the integration of preoperative anticoagulation strategies, interventional radiology techniques such as transjugular intrahepatic portosystemic shunts (TIPS), and intraoperative surgical care. Depending on the extent of thrombosis, intraoperative management ranges from simple thrombectomy and primary portal vein anastomosis to complex vascular procedures, including non-physiological reconstructions.
Given the high prevalence and potential impact of PVT on transplant outcomes, it is essential to establish standardized strategies for its management. This manuscript provided an updated overview of PVT in liver transplantation candidates and presented our institutional protocol. We have highlighted key decision-making steps and technical considerations to optimize outcomes in this high-risk population.
Some studies suggest that reduced portal venous flow, particularly values below 15 cm/second (900 mL/minute), constitutes a risk factor for the development of PVT[5]. Additionally, the use of beta-blockers, which decrease portal flow, may contribute to this risk[6]. The concomitant presence of hepatocellular carcinoma (HCC) increases the likelihood of PVT by approximately 25%[7]. Additional clinical factors are also associated with a higher risk of PVT, including a history of previous variceal ligation[2] or upper gastrointestinal bleeding[8]. Similarly, a history of splenectomy or the presence of portosystemic shunts may favor the development of this condition[9]. Finally, evidence indicates that cirrhosis associated with nonalcoholic steatohepatitis (commonly known as NASH) may represent another risk factor for thrombosis[10].
Cirrhosis generally leads to a reduction in both procoagulant and anticoagulant factors. However, some procoagulant factors, such as factor VIII and von Willebrand factor, may be increased and can explain the higher risk of venous thrombosis[7].
Hereditary thrombophili caused by factor V Leiden mutation and the G20210A prothrombin gene mutation increases risk of PVT in cirrhotic patients, despite their low prevalence in this population (6.7% and 5.4%, respectively)[11]. Recently, Fortea et al[12] identified a low incidence (7.8%) of thrombophilia in their population of patients with cirrhosis and PVT, with most presenting antiphospholipid syndrome(66%). Interestingly, patients with thrombophilic disorders tend to have a higher incidence of complete thrombosis and re-thrombosis after anticoagulation discontinuation. In clinical practice, genetic testing for thrombophilia-related mutations is reserved for patients with a family history of thromboembolic events, multiple sites of venous thrombosis, or recurrent venous thrombosis. Patients diagnosed with Budd-Chiari syndrome, which has a high prevalence of patients with a prothrombotic disorder, may also undergo genetic testing.
Doppler ultrasound is generally the first imaging modality to detect PVT in asymptomatic patients, particularly during the follow-up of individuals with cirrhosis. It has a sensitivity of 89% and a specificity of 92% for the diagnosis of complete PVT[13]. For partial thrombosis, sensitivity ranges from 14% to 50%[2]. The limitations of this method include the difficulty of evaluation in obese patients or those with significant intestinal gas distension as well as requiring an experienced professional for accurate assessment[14].
For the diagnosis and staging of PVT, contrast-enhanced imaging modalities such as computed tomography (CT) or magnetic resonance imaging are preferred, particularly in liver transplant candidates. These modalities offer greater sensitivity than ultrasound in detecting hepatic and abdominal neoplasms and are superior in assessing thrombosis of the superior mesenteric and splenic veins[14].
Contrast-enhanced ultrasound (CEUS) can be used as an adjunct in the evaluation of PVT, particularly for differentiating tumor thrombi in patients with hepatic neoplasms, especially HCC. In CEUS, neoplastic thrombi typically exhibit arterial-phase hyperenhancement (wash-in) and portal or late phase hypoenhancement (wash-out). Dissimilarly, non-tumoral thrombi do not change in appearance after intravenous contrast injection. The addition of contrast enhances the sensitivity and specificity of CEUS for distinguishing tumor thrombi, reaching up to 100% and 96%, respectively.
Another method for assessing the likelihood of a neoplastic thrombus of HCC origin is the A-VENA criteria, which include five risk factors: (1) Alpha-fetoprotein > 1000 ng/mL; (2) Venous expansion; (3) Increased vascular enhancement; (4) Neovascularization; and (5) Thrombosis adjacent to a tumor or a previously treated area. When three or more of these factors are present, the A-VENA criteria demonstrate a sensitivity of 100%, specificity of 94%, and negative predictive value of 100% for diagnosing neoplastic etiology[15].
At least 10 classification systems for non-tumoral PVT have been published in the literature[16]. The most widely used for evaluating patients on the liver transplant waiting list is the Yerdel et al[17] classification, which correlates the extent of PVT with surgical management during liver transplantation. For this purpose, the classifications by Charco et al[18] and Jamieson[19] may also be utilized (Table 1).
| Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
| Yerdel et al[17] | Portal thrombosis < 50% with or without minimal extension to the SMV | Portal thrombosis > 50% with or without minimal extension to the SMV | Complete portal and SMV thrombosis in its most proximal extent; preserved flow in the distal SMV | Complete portal and SMV thrombosis |
| Jamieson[19] | Partial or complete portal thrombosis up to the splenomesenteric junction | Portal thrombosis extending to the proximal SMV, with preserved mesenteric flow | Diffuse splanchnic system thrombosis with enlarged collateral vessels | Diffuse splanchnic system thrombosis with thin collaterals |
| Charco et al[18] | Thrombosis restricted to the portal vein, partial or complete | Thrombosis extending to the proximal SMV, with patency of the mesenteric confluence | Diffuse splanchnic system thrombosis with dilated collaterals | Diffuse splanchnic system thrombosis with thin collaterals |
With the aim of providing a more up-to-date and prognostic classification of this pathology, Bhangui et al[20] categorized PVT into non-complex portal thrombosis (Yerdel types I-III, Charco and Jamieson types I and II) and complex portal thrombosis (Yerdel type V, Charco and Jamieson types III and IV).
Another important classification relates to the chronicity of portal thrombosis. It has been conventionally established that portal thrombosis diagnosed for more than 6 months should be considered chronic. In cases where it is not possible to precisely determine when the thrombosis occurred, the following signs are suggestive of chronicity: (1) Calcifications in the vessel wall; (2) Disappearance of the vein and its replacement by a fibrous cord; and (3) Development of collateral circulation with or without portal cavernoma[21].
The impact of non-tumoral PVT on post-liver transplant outcomes is well established. The following systematic literature reviews confirmed that those with PVT experience a significant increase in postoperative mortality compared with patients without this complication. Rodríguez-Castro et al[2] observed that patients with PVT had mortality rates of 10.5% and 18.8% within the first 30 days and after 1 year of liver transplantation, respectively. This was significantly higher than those without PVT (7.7% and 15.3%, respectively). More interestingly, investigations comparing 1-year mortality between patients with partial vs complete PVT found rates of 22% and 42%, respectively. Di Benedetto et al[4] demonstrated in a multicenter retrospective study that included more than 500 cases that patients with Yerdel types III and IV thrombosis had a 90-day mortality rate of 30% compared with 13.7% in patients with Yerdel type II. A recent meta-analysis published by Zanetto et al[22] showed a 5.65-fold increased risk of postoperative mortality in individuals with this complication. In this context, strategies to fully or partially restore portal flow become imperative.
Just as important as the implementation of strategies to restore portal flow is the appropriate timing of clinical management. In some cases, PVT may reflect progressive portal hypertension and contribute to hepatic decompensation, including worsening of ascites and an increased risk of mortality while on the waiting list[23,24]. Therefore, although systematic prioritization of patients with PVT is not recommended currently, careful monitoring and prompt ma
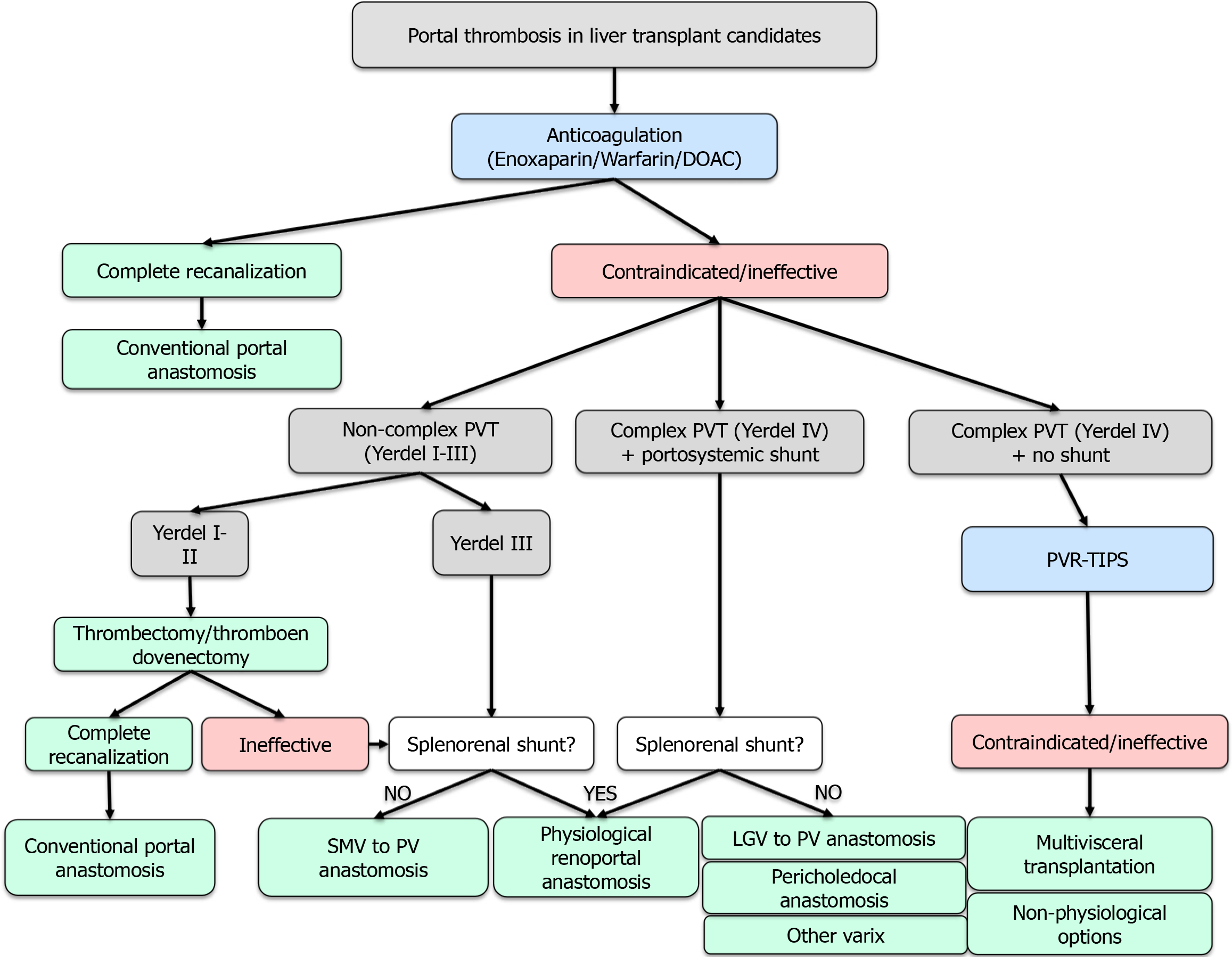
In our unit, the initial assessment of patients includes CT angiography of the abdomen. In patients with large spontaneous portosystemic shunts (> 1 cm), we perform an abdominal Doppler ultrasound with flow quantification. These individuals undergo reassessment with contrast-enhanced abdominal imaging every 3 months after listing to evaluate treatment outcomes (when performed) and/or progression of thrombosis. We use a specific algorithm for the management of these patients (Figure 1).
Anticoagulation can recanalize the portal system in 71% of patients compared with 42% of patients in the absence of this therapy. Additionally, anticoagulation increases the rate of complete recanalization and reduces the progression of PVT. No significant increase in bleeding of any type has been identified. Moreover, when esophageal variceal bleeding was analyzed there was a decrease found in its incidence among treated patients[26].
Several factors are associated with greater anticoagulation efficacy, including initiation of treatment within 6 months of diagnosis, Child A cirrhosis, and partial thrombosis. The average time for portal recanalization ranges from 5-8 months, with most cases occurring within the first year of treatment[27]. When anticoagulation is discontinued, re-thrombosis can occur in up to 38.5% of patients. Therefore, it is important to maintain anticoagulation indefinitely in patients listed for transplantation[28]. Analysis of studies on PVT treatment in patients with cirrhosis suggested that its benefits occur regardless of the specific anticoagulant used[29]. Thus, anticoagulation is recommended for patients with cirrhosis and PVT who are candidates for liver transplantation[30-32].
During the evaluation of patients with cirrhosis and PVT and before anticoagulation initiation, the American College of Gastroenterology[30] and the American Association for the Study of Liver Diseases[31] recommend upper gastroin
Low molecular weight heparin: This drug class is the most extensively studied and is the first-line anticoagulation therapy for patients with cirrhosis[32]. Enoxaparin is the preferred drug in our group. It is administered in two doses of 1 mg/kg[16]. Enoxaparin has a half-life ranging from 3-7 hours when administered subcutaneously. The main contraindications include a history of heparin-induced thrombocytopenia and known hypersensitivity to enoxaparin, benzyl acid derivatives, or porcine-derived products[33]. A dose reduction of 30% is recommended when platelet counts are below 50000[27]. In patients with renal insufficiency [creatinine clearance (CrCl) < 30 mL/minute], a single daily dose is suggested[34].
For anticoagulation reversal before liver transplantation, protamine sulfate can be administered via slow intravenous infusion (not exceeding 5 mg/minute) at a dose of 1 mg of protamine per 1 mg of enoxaparin administered, particularly if enoxaparin was given within 8 hours before the procedure. If the last enoxaparin dose was administered more than 8 hours before transplantation, a reduced dose of 0.5 mg of protamine per 1 mg of enoxaparin may be suggested[35]. A recent consensus recommended anticoagulation reversal if enoxaparin was administered within the prior 12 hours and if intraoperative bleeding due to coagulopathy is present[36].
Fondaparinux: Fondaparinux can be used with efficacy comparable to enoxaparin at a fixed once-daily dose (weight-based dosing)[37], although there are few publications on its use in this context[38]. It is particularly useful in cases of heparin-induced thrombocytopenia. Its use is contraindicated in patients with a CrCl < 30 mL/minute[37].
Vitamin K antagonists (warfarin): Vitamin K antagonists, such as warfarin, are mentioned in the main guidelines as a treatment option, particularly after the initial introduction of low molecular weight heparin. Its oral administration facilitates adherence in patients who have difficulty with other routes. The clinical use is similar to that in patients without cirrhosis but is contraindicated in those with platelet counts below 50000. However, changes in the international normalized ratio (INR), which is common in patients with cirrhosis, can complicate the management of this medication[29]. Warfarin has a half-life range of 36-42 hours[39]. Thus, recent consensus guidelines recommend administering an intravenous dose of vitamin K (10 mg) upon admission for patients with an INR up to 3.5. For patients with an INR > 3.5, it is suggested to administer prothrombin complex concentrate at up to 25 mg/kg before surgery[36,40].
Direct oral anticoagulants: Direct oral anticoagulants (DOACs) such as rivaroxaban, apixaban, dabigatran, and doxaban can be used, particularly in patients with Child-Pugh A. However, specific recommendations for a particular medication are not provided in the latest Baveno VII guidelines[32]. The use of these medications is safe in this population, with portal recanalization outcomes at least comparable to traditional anticoagulants[41,42]. DOACs have a half-life of 9-14 hours[39], and their use is contraindicated in patients with a CrCl < 15 mL/minute, those on dialysis, patients with Child-Pugh C (use of rivaroxaban is contraindicated in patients with Child-Pugh B), and patients with antiphospholipid syndrome[43]. Recent consensus guidelines suggest administering a specific antidote before transplantation, including idarucizumab (praxbind in Brazil, for use with dabigatran) or andexanet alfa (ondexxya in Brazil, for use with apixaban and rivaroxaban)[36].
In liver transplant candidates, the use of TIPS can be an option for portal vein recanalization when anticoagulation has been ineffective or contraindicated[32]. The procedure can be performed via the traditional transjugular route, adding a transhepatic or even a transsplenic route when necessary. This procedure is feasible in 75%-100% of patients, with portal recanalization rates ranging from 57% to 93%[44]. Complete PVT is a significant factor for procedural failure[45], and cavernous transformation is the main factor associated with incomplete recanalization[46]. However, neither condition is considered an absolute contraindication. Reported complications after the procedure include hepatic encephalopathy (ranging from 14% to 60%), bleeding-related complications (such as hemoperitoneum, hematomas at puncture sites, or intra-abdominal organs), sepsis, acute-on-chronic liver failure, and right heart failure[44,46]. Contraindications for TIPS placement include grade 2 or higher hepatic encephalopathy (or recurrent episodes without a precipitating factor), severe systolic or diastolic dysfunction, pulmonary hypertension, sepsis, and a Child-Pugh score greater than 12[47]. In our unit, TIPS is used as a salvage procedure in patients with complex thrombosis associated with the absence of large portosystemic shunts, provided that transsplenic access is feasible.
The prevalence of PVT in patients who underwent liver transplantation are 5%-26%[20]. In some cases, diagnosis is only made intraoperatively, occurring in up to 64% of cases[48]. For patients with Yerdel types I-III PVT (also classified by Bhangui et al[20] as non-complex cases), post-transplant outcomes are comparable to those of patients without PVT if physiological portal reconstruction can be performed. Physiological portal reconstruction is defined as a reconstruction in which splanchnic blood flow is directed to the transplanted liver, even if only partially.
Non-complex PVT: Some authors advocate the effectiveness of simple thrombectomy or thromboendovenectomy (resection of the intimal layer of the vessel) and conventional portal anastomosis in patients with Yerdel types I and II PVT leading to excellent post-transplant results in most patients[48,49]. If a physiological anastomosis can be achieved (e.g., venous conduit between the donor's mesenteric vein and the portal vein of the graft or an extended thrombectomy) in patients with type III PVT, patient and graft survival rates are comparable with those of patients without PVT[50,51]. A crucial factor in decision-making is the presence of spontaneous portosystemic shunts, which occur in 20%-40% of patients with cirrhosis and are considered large when they have a diameter of at least 1 cm[52].
In our group, the decision to ligate these shunts is made intraoperatively. If portal flow increases upon shunt clamping, we proceed with ligation (either directly on the vessel or on the left renal vein in cases of splenorenal shunts). Specifically, in the presence of large splenorenal shunts, when a conventional porto-portal anastomosis is unfeasible or provides inadequate flow (< 1 L/minute), we prioritize renoportal anastomosis, initially avoiding the use of venous grafts from the superior mesenteric vein for portal anastomosis because this would require splenorenal shunt or left renal vein ligation.
Complex PVT: The reported prevalence of complex PVT in studies has ranged from 0%-2.2%[20], and its resolution depends on the presence or absence of portosystemic shunts (either spontaneous or surgical). Among the possible physiological reconstructions for patients with complex PVT and large portosystemic shunts (Charco and Jamieson type III), the preferred approaches, in order of utilization, include renoportal anastomosis, anastomosis using the left gastric vein, and anastomosis using other varices.
Renoportal anastomosis involves an anastomosis between the left renal vein and the graft portal vein. It can be performed in an end-to-end or end-to-side fashion with or without the use of a venous graft. The technique employed by our group has been previously described in another publication[53]. It is considered a physiological approach as long as splenorenal shunts (either spontaneous or surgical) are present. The complication rate is approximately 70%, with the main complications being ascites (27.2%), sepsis (19.6%), and acute kidney injury (18.1%)[54]. One of the primary studies on the outcomes of this technique, conducted in 57 patients, reported a 90-day mortality rate of 8.8%, an anastomosis patency rate of 84% at 5 years, and 5-year graft and patient survival rates of 73% and 76%, respectively[55].
Most publications regarding the use of varices for portal reconstruction in liver transplantation consist of case reports and small case series[20]. The largest study on anastomoses using the left gastric vein, which included 12 patients, reported no 90-day mortality, a 66% postoperative complication rate, and no cases of PVT. The 4-year patient survival rate was 85%[56].
Pericholedochal varices can be used for portal reconstruction, although only a few case series have been published on this technique. This approach requires a biliodigestive anastomosis for biliary reconstruction to minimize the risk of vascular injury during distal bile duct manipulation. Other varices that have been used in portal reconstruction, according to the literature, include the right colic vein, middle colic vein, gastroepiploic vein[20], and the inferior mesenteric vein[57].
A subset of patients presents with diffuse PVT without any shunts suitable for utilization (classified as Jamieson and Charco type IV). In these cases, the following strategies may be considered: (1) Multivisceral transplantation; (2) Cavoportal hemitransposition; and (3) Portal arterialization.
Multivisceral transplantation is considered a physiological reconstruction for these patients, as it replaces the entire portomesenteric system. It may be employed as an upfront therapy or as a backup option if a physiological portal anastomosis is not feasible at the time of transplantation. This is an extremely complex surgical procedure and is associated with significant postoperative morbidity in both the short and long term. The main complications include infections in approximately 90% of patients, graft rejection in 20% of patients, and surgical complications in up to 50% of patients. However, long-term patient survival can reach up to 75% at 10 years[58].
Cavoportal hemitransposition redirects caval blood flow toward the graft portal vein. It can be performed in an end-to-end or end-to-side manner with or without resection of the recipient’s inferior vena cava and with either partial or complete ligation of the vena cava. Postoperative mortality is around 35%. Since this procedure does not address portal hypertension (except in cases with a preexisting mesocaval shunt), the main postoperative complications include refractory ascites, acute kidney injury, upper gastrointestinal bleeding, and lower limb edema[59]. A recent multicenter study found that in patients who underwent cavoportal hemitransposition (with or without portosystemic shunts), 5-year survival was 33%. Additionally, when analyzing only non-physiological anastomoses (in this study, renoportal anastomosis and cavoportal hemitransposition), the 5-year post-transplant survival rate in 13 patients was 6.7%[60].
In cases where none of the previous alternatives are feasible, portal arterialization may be considered as an exceptional procedure. There are only a few case reports and small case series in the literature[20], with a reported mortality rate of 38%[61]. Complications include re-thrombosis, hepatic fibrosis due to excessive portal inflow, right heart failure, and persistent portal hypertension with its associated complications[61].
It is well established that patients with pre-transplant PVT have an increased risk of developing recurrent PVT, regardless of the treatment employed[4,51]. In this context, we recommend performing color Doppler ultrasound of the liver within the first 24 hours after transplantation to assess graft vascularization. Additionally, therapeutic-dose enoxaparin anticoagulation should be resumed as soon as possible. Contraindications to early anticoagulation include evidence of intracavitary bleeding, coagulopathy (INR > 1.5), thrombocytopenia (< 50000), and graft dysfunction[36]. Anticoagulant therapy is maintained for at least 2 months, and the decision regarding its continuation is individualized after this period. Factors that may favor prolonged anticoagulation include other known risk factors such as partial thrombectomy or intimal injury to the portal vein, non-physiological portal vein inflow reconstruction, and the presence of thrombophilic disorders[36]. In such cases, provided the bleeding risk remains acceptable, extended anticoagulation may help reduce the risk of rethrombosis theoretically. Ongoing surveillance with liver imaging is essential to guide therapeutic adjustments during this period.
Non-tumoral PVT remains a complex and high-risk condition in the context of liver transplantation, with significant implications for perioperative management and post-transplant outcomes. Early identification of risk factors and timely diagnosis through appropriate imaging are essential to guide individualized treatment. Anticoagulation remains the cornerstone of initial management, while interventional radiology and surgical approaches must be tailored according to the extent and complexity of thrombosis. Physiological reconstructions are associated with better long-term outcomes, whereas non-physiological alternatives, although sometimes necessary, are linked to increased morbidity and mortality. In some cases, LDLT may represent a theoretical strategy to anticipate transplant timing and avoid complex thrombosis, although further evidence is needed to support its routine use. The implementation of a structured, multidisciplinary protocol, like the one presented in this manuscript, enables consistent decision-making and may improve outcomes for this challenging patient population. Future research should focus on refining patient selection for advanced surgical techniques and optimizing strategies to prevent re-thrombosis after transplantation.
| 1. | Ponziani FR, Zocco MA, Senzolo M, Pompili M, Gasbarrini A, Avolio AW. Portal vein thrombosis and liver transplantation: implications for waiting list period, surgical approach, early and late follow-up. Transplant Rev (Orlando). 2014;28:92-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 2. | Rodríguez-Castro KI, Porte RJ, Nadal E, Germani G, Burra P, Senzolo M. Management of nonneoplastic portal vein thrombosis in the setting of liver transplantation: a systematic review. Transplantation. 2012;94:1145-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 175] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 3. | John BV, Konjeti R, Aggarwal A, Lopez R, Atreja A, Miller C, Zein NN, Carey WD. Impact of untreated portal vein thrombosis on pre and post liver transplant outcomes in cirrhosis. Ann Hepatol. 2013;12:952-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 4. | Di Benedetto F, Magistri P, Di Sandro S, Boetto R, Tandoi F, Camagni S, Lauterio A, Pagano D, Nicolini D, Violi P, Dondossola D, Guglielmo N, Cherchi V, Lai Q, Toti L, Bongini M, Frassoni S, Bagnardi V, Mazzaferro V, Tisone G, Rossi M, Baccarani U, Ettorre GM, Caccamo L, Carraro A, Vivarelli M, Gruttadauria S, De Carlis L, Colledan M, Romagnoli R, Cillo U. Portal vein thrombosis and liver transplantation: management, matching, and outcomes. A retrospective multicenter cohort study. Int J Surg. 2024;110:2874-2882. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Reference Citation Analysis (0)] |
| 5. | Stine JG, Wang J, Shah PM, Argo CK, Intagliata N, Uflacker A, Caldwell SH, Northup PG. Decreased portal vein velocity is predictive of the development of portal vein thrombosis: A matched case-control study. Liver Int. 2018;38:94-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 112] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 6. | Xu X, Guo X, De Stefano V, Silva-Junior G, Goyal H, Bai Z, Zhao Q, Qi X. Nonselective beta-blockers and development of portal vein thrombosis in liver cirrhosis: a systematic review and meta-analysis. Hepatol Int. 2019;13:468-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 7. | Connolly GC, Chen R, Hyrien O, Mantry P, Bozorgzadeh A, Abt P, Khorana AA. Incidence, risk factors and consequences of portal vein and systemic thromboses in hepatocellular carcinoma. Thromb Res. 2008;122:299-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 91] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 8. | Francoz C, Valla D, Durand F. Portal vein thrombosis, cirrhosis, and liver transplantation. J Hepatol. 2012;57:203-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 186] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 9. | Gong C, Qin X, Yang J, Guo T. The Best Anticoagulation Strategy for Cirrhotic Patients who Underwent Splenectomy: A Network Meta-Analysis. Gastroenterol Res Pract. 2017;2017:9216172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 10. | Ghabril M, Agarwal S, Lacerda M, Chalasani N, Kwo P, Tector AJ. Portal Vein Thrombosis Is a Risk Factor for Poor Early Outcomes After Liver Transplantation: Analysis of Risk Factors and Outcomes for Portal Vein Thrombosis in Waitlisted Patients. Transplantation. 2016;100:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 115] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 11. | Ma SD, Wang J, Bezinover D, Kadry Z, Northup PG, Stine JG. Inherited thrombophilia and portal vein thrombosis in cirrhosis: A systematic review and meta-analysis. Res Pract Thromb Haemost. 2019;3:658-667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Fortea JI, Carrera IG, Puente Á, Cuadrado A, Huelin P, Tato CÁ, Fernández PÁ, Montes MDRP, Céspedes JN, López AB, Sanchez FJG, Hoyos ML, Crespo J, Fábrega E. Portal Thrombosis in Cirrhosis: Role of Thrombophilic Disorders. J Clin Med. 2020;9:2822. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Tessler FN, Gehring BJ, Gomes AS, Perrella RR, Ragavendra N, Busuttil RW, Grant EG. Diagnosis of portal vein thrombosis: value of color Doppler imaging. AJR Am J Roentgenol. 1991;157:293-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 123] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 14. | Margini C, Berzigotti A. Portal vein thrombosis: The role of imaging in the clinical setting. Dig Liver Dis. 2017;49:113-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 15. | Sherman CB, Behr S, Dodge JL, Roberts JP, Yao FY, Mehta N. Distinguishing Tumor From Bland Portal Vein Thrombus in Liver Transplant Candidates With Hepatocellular Carcinoma: the A-VENA Criteria. Liver Transpl. 2019;25:207-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 53] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 16. | Senzolo M, Garcia-Tsao G, García-Pagán JC. Current knowledge and management of portal vein thrombosis in cirrhosis. J Hepatol. 2021;75:442-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 129] [Article Influence: 32.3] [Reference Citation Analysis (0)] |
| 17. | Yerdel MA, Gunson B, Mirza D, Karayalçin K, Olliff S, Buckels J, Mayer D, McMaster P, Pirenne J. Portal vein thrombosis in adults undergoing liver transplantation: risk factors, screening, management, and outcome. Transplantation. 2000;69:1873-1881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 527] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 18. | Charco R, Fuster J, Fondevila C, Ferrer J, Mans E, García-Valdecasas JC. Portal vein thrombosis in liver transplantation. Transplant Proc. 2005;37:3904-3905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 19. | Jamieson NV. Changing perspectives in portal vein thrombosis and liver transplantation. Transplantation. 2000;69:1772-1774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 76] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Bhangui P, Lim C, Levesque E, Salloum C, Lahat E, Feray C, Azoulay D. Novel classification of non-malignant portal vein thrombosis: A guide to surgical decision-making during liver transplantation. J Hepatol. 2019;71:1038-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 21. | Turon F, Shalaby S, García-Pagán JC, Hernández-Gea V. Management of portal vein thrombosis in candidates for liver transplant. Liver Transpl. 2024;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 22. | Zanetto A, Rodriguez-Kastro KI, Germani G, Ferrarese A, Cillo U, Burra P, Senzolo M. Mortality in liver transplant recipients with portal vein thrombosis - an updated meta-analysis. Transpl Int. 2018;31:1318-1329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 96] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 23. | Stine JG, Shah PM, Cornella SL, Rudnick SR, Ghabril MS, Stukenborg GJ, Northup PG. Portal vein thrombosis, mortality and hepatic decompensation in patients with cirrhosis: A meta-analysis. World J Hepatol. 2015;7:2774-2780. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 24. | Englesbe MJ, Kubus J, Muhammad W, Sonnenday CJ, Welling T, Punch JD, Lynch RJ, Marrero JA, Pelletier SJ. Portal vein thrombosis and survival in patients with cirrhosis. Liver Transpl. 2010;16:83-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 185] [Article Influence: 12.3] [Reference Citation Analysis (2)] |
| 25. | Kirimker EO, Kabacam G, Keskin O, Goktug UU, Atli M, Bingol-Kologlu M, Karayalcin K, Karademir S, Balci D. Outcomes of Surgical Strategies for Living Donor Liver Transplantation in Patients With Portal Vein Thrombosis: A Cohort Study. Transplant Proc. 2022;54:2217-2223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 26. | Loffredo L, Pastori D, Farcomeni A, Violi F. Effects of Anticoagulants in Patients With Cirrhosis and Portal Vein Thrombosis: A Systematic Review and Meta-analysis. Gastroenterology. 2017;153:480-487.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 298] [Article Influence: 37.3] [Reference Citation Analysis (0)] |
| 27. | Rodriguez-Castro KI, Vitale A, Fadin M, Shalaby S, Zerbinati P, Sartori MT, Landi S, Pettinari I, Piscaglia F, Han G, Burra P, Simioni P, Senzolo M. A prediction model for successful anticoagulation in cirrhotic portal vein thrombosis. Eur J Gastroenterol Hepatol. 2019;31:34-42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 28. | Delgado MG, Seijo S, Yepes I, Achécar L, Catalina MV, García-Criado A, Abraldes JG, de la Peña J, Bañares R, Albillos A, Bosch J, García-Pagán JC. Efficacy and safety of anticoagulation on patients with cirrhosis and portal vein thrombosis. Clin Gastroenterol Hepatol. 2012;10:776-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 288] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 29. | Elkrief L, Hernandez-Gea V, Senzolo M, Albillos A, Baiges A, Berzigotti A, Bureau C, Murad SD, De Gottardi A, Durand F, Garcia-Pagan JC, Lisman T, Mandorfer M, McLin V, Moga L, Nery F, Northup P, Nuzzo A, Paradis V, Patch D, Payancé A, Plaforet V, Plessier A, Poisson J, Roberts L, Salem R, Sarin S, Shukla A, Toso C, Tripathi D, Valla D, Ronot M, Rautou PE; ERN RARE-LIVER and VALDIG, an EASL consortium. Portal vein thrombosis: diagnosis, management, and endpoints for future clinical studies. Lancet Gastroenterol Hepatol. 2024;9:859-883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Reference Citation Analysis (0)] |
| 30. | Simonetto DA, Singal AK, Garcia-Tsao G, Caldwell SH, Ahn J, Kamath PS. ACG Clinical Guideline: Disorders of the Hepatic and Mesenteric Circulation. Am J Gastroenterol. 2020;115:18-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 141] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 31. | Northup PG, Garcia-Pagan JC, Garcia-Tsao G, Intagliata NM, Superina RA, Roberts LN, Lisman T, Valla DC. Vascular Liver Disorders, Portal Vein Thrombosis, and Procedural Bleeding in Patients With Liver Disease: 2020 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2021;73:366-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 372] [Article Influence: 93.0] [Reference Citation Analysis (1)] |
| 32. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1537] [Cited by in RCA: 1499] [Article Influence: 499.7] [Reference Citation Analysis (2)] |
| 33. | United States Food and Drug Administration. Lovenox (enoxaparin sodium injection), for subcutaneous and intravenous use: Prescribing information. Silver Spring (MD): United States Food and Drug Administration. 2021. [Accessed March 20, 2025]. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/020164s129lbl.pdf. |
| 34. | Witt DM, Nieuwlaat R, Clark NP, Ansell J, Holbrook A, Skov J, Shehab N, Mock J, Myers T, Dentali F, Crowther MA, Agarwal A, Bhatt M, Khatib R, Riva JJ, Zhang Y, Guyatt G. American Society of Hematology 2018 guidelines for management of venous thromboembolism: optimal management of anticoagulation therapy. Blood Adv. 2018;2:3257-3291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 361] [Article Influence: 51.6] [Reference Citation Analysis (0)] |
| 35. | Garcia DA, Baglin TP, Weitz JI, Samama MM. Parenteral anticoagulants: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141:e24S-e43S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 775] [Cited by in RCA: 723] [Article Influence: 55.6] [Reference Citation Analysis (0)] |
| 36. | Montalvá E, Rodríguez-Perálvarez M, Blasi A, Bonanad S, Gavín O, Hierro L, Lladó L, Llop E, Pozo-Laderas JC, Colmenero J; on behalf of the Spanish Society of Liver Transplantation and the Spanish Society of Thrombosis and Haemostasis. Consensus Statement on Hemostatic Management, Anticoagulation, and Antiplatelet Therapy in Liver Transplantation. Transplantation. 2022;106:1123-1131. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 37. | United States Food and Drug Administration. ARIXTRA (fondaparinux sodium) Solution for Injection. Silver Spring (MD): United States Food and Drug Administration. 2014. [Accessed March 20, 2025]. Available from: http://www.accessdata.fda.gov/drugsatfda_docs/label/2014/021345s032lbl.pdf. |
| 38. | Zhang ZH, Zhang JW, He P, Zhou Y, Sun CY. Fondaparinux is effective for acute portal vein thrombosis in decompensated cirrhotic patients. Medicine (Baltimore). 2017;96:e8256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 39. | Douketis JD, Spyropoulos AC, Murad MH, Arcelus JI, Dager WE, Dunn AS, Fargo RA, Levy JH, Samama CM, Shah SH, Sherwood MW, Tafur AJ, Tang LV, Moores LK. Perioperative Management of Antithrombotic Therapy: An American College of Chest Physicians Clinical Practice Guideline. Chest. 2022;162:e207-e243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 182] [Article Influence: 60.7] [Reference Citation Analysis (0)] |
| 40. | Bezinover D, Dirkmann D, Findlay J, Guta C, Hartmann M, Nicolau-Raducu R, Mukhtar AM, Moguilevitch M, Pivalizza E, Rosenfeld D, Saner F, Wray C, Wagener G, West J. Perioperative Coagulation Management in Liver Transplant Recipients. Transplantation. 2018;102:578-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 69] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 41. | Nisly SA, Mihm AE, Gillette C, Davis KA, Tillett J. Safety of direct oral anticoagulants in patients with mild to moderate cirrhosis: a systematic review and meta-analysis. J Thromb Thrombolysis. 2021;52:817-827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 42. | Ng CH, Tan DJH, Nistala KRY, Syn N, Xiao J, Tan EXX, Woo FZ, Chew NWS, Huang DQ, Dan YY, Sanyal AJ, Muthiah MD. A network meta-analysis of direct oral anticoagulants for portal vein thrombosis in cirrhosis. Hepatol Int. 2021;15:1196-1206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 43. | De Maria C, Galante A, Fasoli A, De Gottardi A. When and how to use direct oral anticoagulants in patients with advanced chronic liver disease? Curr Opin Pharmacol. 2021;60:111-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 44. | Guo DF, Fan LW, Le Q, Huang CB. Transjugular intrahepatic portosystemic shunt for the prevention of rebleeding in patients with cirrhosis and portal vein thrombosis: Systematic review and meta-analysis. Front Pharmacol. 2022;13:968988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 45. | Lv Y, Bai W, Li K, Wang Z, Guo W, Luo B, Wang J, Wang Q, Wang E, Xia D, Li X, Yuan J, Han N, Niu J, Yin Z, Fan D, Han G. Anticoagulation and Transjugular Intrahepatic Portosystemic Shunt for the Management of Portal Vein Thrombosis in Cirrhosis: A Prospective Observational Study. Am J Gastroenterol. 2021;116:1447-1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 46. | Rodrigues SG, Sixt S, Abraldes JG, De Gottardi A, Klinger C, Bosch J, Baumgartner I, Berzigotti A. Systematic review with meta-analysis: portal vein recanalisation and transjugular intrahepatic portosystemic shunt for portal vein thrombosis. Aliment Pharmacol Ther. 2019;49:20-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 91] [Article Influence: 15.2] [Reference Citation Analysis (0)] |
| 47. | García-Pagán JC, Saffo S, Mandorfer M, Garcia-Tsao G. Where does TIPS fit in the management of patients with cirrhosis? JHEP Rep. 2020;2:100122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 48. | Koh PS, Chan SC, Chok KS, Sharr WW, Wong TC, Sin SL, Lo CM. The friendly incidental portal vein thrombus in liver transplantation. Liver Transpl. 2015;21:944-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 49. | Molmenti EP, Roodhouse TW, Molmenti H, Jaiswal K, Jung G, Marubashi S, Sanchez EQ, Gogel B, Levy MF, Goldstein RM, Fasola CG, Elliott EE, Bursac N, Mulligan D, Gonwa TA, Klintmalm GB. Thrombendvenectomy for organized portal vein thrombosis at the time of liver transplantation. Ann Surg. 2002;235:292-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 112] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 50. | Hibi T, Nishida S, Levi DM, Selvaggi G, Tekin A, Fan J, Ruiz P, Tzakis AG. When and why portal vein thrombosis matters in liver transplantation: a critical audit of 174 cases. Ann Surg. 2014;259:760-766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 123] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 51. | Ravaioli M, Zanello M, Grazi GL, Ercolani G, Cescon M, Del Gaudio M, Cucchetti A, Pinna AD. Portal vein thrombosis and liver transplantation: evolution during 10 years of experience at the University of Bologna. Ann Surg. 2011;253:378-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 92] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 52. | Gomez Gavara C, Bhangui P, Salloum C, Osseis M, Esposito F, Moussallem T, Lahat E, Fuentes L, Compagnon P, Ngongang N, Lim C, Azoulay D. Ligation versus no ligation of spontaneous portosystemic shunts during liver transplantation: Audit of a prospective series of 66 consecutive patients. Liver Transpl. 2018;24:505-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 53. | Grezzana-Filho TJM, Chedid MF, Chedid AD, Bassani-Filho EL, Pereira PF, Leipnitz I, Backes AN, Kruel CRP. How to do liver transplantation using renoportal bypass. ANZ J Surg. 2018;88:236-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 54. | D'Amico G, Hassan A, Diago Uso T, Hashmimoto K, Aucejo FN, Fujiki M, Eghtesad B, Sasaki K, Lindenmeyer CC, Miller CM, Quintini C. Renoportal anastomosis in liver transplantation and its impact on patient outcomes: a systematic literature review. Transpl Int. 2019;32:117-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Azoulay D, Quintini C, Rayar M, Salloum C, Llado L, Diago T, D'Amico G, Ramos E, Fabregat J, Eshkenazy R, Bardou-Jacquet E, Camus C, Compagnon P, Vibert E, Lim C. Renoportal Anastomosis During Liver Transplantation in Patients With Portal Vein Thrombosis: First Long-term Results From a Multicenter Study. Ann Surg. 2022;276:e825-e833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 56. | Ravaioli M, Prosperi E, Pinna A, Siniscalchi A, Fallani G, Frascaroli G, Maroni L, Odaldi F, Serenari M, Cescon M. Restoration of portal flow with varix in liver transplantation for patients with total portal vein thrombosis: An effective strategy in the largest center experience. Clin Transplant. 2021;35:e14303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 57. | Iyer SG, Lau CL, Chang KY, Mak SW, Madhavan KK. Successful living donor liver transplantation in portomesenteric thrombosis. Am J Transplant. 2010;10:1483-1485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 58. | Tekin A, Beduschi T, Vianna R, Mangus RS. Multivisceral transplant as an option to transplant cirrhotic patients with severe portal vein thrombosis. Int J Surg. 2020;82S:115-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 59. | Lerut JP, Lai Q, de Ville de Goyet J. Cavoportal Hemitransposition in Liver Transplantation: Toward a More Safe and Efficient Technique. Liver Transpl. 2020;26:92-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | Fundora Y, Hessheimer AJ, Del Prete L, Maroni L, Lanari J, Barrios O, Clarysse M, Gastaca M, Barrera Gómez M, Bonadona A, Janek J, Boscà A, Álamo Martínez JM, Zozaya G, López Garnica D, Magistri P, León F, Magini G, Patrono D, Ničovský J, Hakeem AR, Nadalin S, McCormack L, Palacios P, Zieniewicz K, Blanco G, Nuño J, Pérez Saborido B, Echeverri J, Bynon JS, Martins PN, López López V, Dayangac M, Lodge JPA, Romagnoli R, Toso C, Santoyo J, Di Benedetto F, Gómez-Gavara C, Rotellar F, Gómez-Bravo MÁ, López Andújar R, Girard E, Valdivieso A, Pirenne J, Lladó L, Germani G, Cescon M, Hashimoto K, Quintini C, Cillo U, Polak WG, Fondevila C. Alternative forms of portal vein revascularization in liver transplant recipients with complex portal vein thrombosis. J Hepatol. 2023;78:794-804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 61. | Bonnet S, Sauvanet A, Bruno O, Sommacale D, Francoz C, Dondero F, Durand F, Belghiti J. Long-term survival after portal vein arterialization for portal vein thrombosis in orthotopic liver transplantation. Gastroenterol Clin Biol. 2010;34:23-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |