Published online May 27, 2023. doi: 10.4240/wjgs.v15.i5.965
Peer-review started: November 25, 2022
First decision: February 15, 2023
Revised: March 7, 2023
Accepted: April 7, 2023
Article in press: April 7, 2023
Published online: May 27, 2023
Processing time: 182 Days and 4.3 Hours
Cholangiocarcinoma (CC) is a very aggressive cancer with a poor prognosis. As surgery is the only curative therapy, preoperative evaluation of the tumor extent is essential for surgical planning. Although high-quality image modalities such as computed tomography and magnetic resonance imaging have been used extensively in preoperative evaluation, the accuracy is low. To obtain precise localization of tumor spread arising from the hilar region preoperatively, the development of an acceptable imaging modality is still an unmet need.
A 52-year-old female presented to our emergency department with jaundice, abdominal pain, and fever. Initially, she was treated for cholangitis. Endoscopic retrograde cholangiopancreatography with the cholangiogram showed long segment filling defect in the common hepatic duct with dilatation of bilateral intrahepatic ducts. Transpapillary biopsy was performed, and the pathology suggested intraductal papillary neoplasm with high-grade dysplasia. After treatment of cholangitis, contrasted-enhanced computed tomography revealed a hilar lesion with undetermined Bismuth-Corlette classification. SpyGlass cholangioscopy showed that the lesion involved the confluence of the common hepatic duct with one skip lesion in the posterior branch of the right intrahepatic duct, which was not detected by previous image modalities. The surgical plan was modified from extended left hepatectomy to extended right hepatectomy. The final diagnosis was hilar CC, pT2aN0M0. The patient has remained disease-free for more than 3 years.
SpyGlass cholangioscopy may have a role in precision localization of hilar CC to provide surgeons with more information before the operation.
Core Tip: The precise localization of hilar cholangiocarcinoma (CC) is important for surgical planning. This case highlights the important role that SpyGlass cholangioscopy can have in precise localization. However, SpyGlass cholangioscopy may be difficult to perform during the first encounter because of obstruction. A two-step approach for obstructive jaundice caused by hilar CC was proposed: (1) Insertion of biliary plastic stents to relieve jaundice and dilate the stricture site; and (2) Removal of biliary plastic stents after relieving jaundice and subsequent examination of hilar CC involvement by SpyGlass cholangioscopy.
- Citation: Chiang CH, Chen KC, Devereaux B, Chung CS, Kuo KC, Lin CC, Lin CK, Wang HP, Chen KH. Precise mapping of hilar cholangiocarcinoma with a skip lesion by SpyGlass cholangioscopy: A case report. World J Gastrointest Surg 2023; 15(5): 965-971
- URL: https://www.wjgnet.com/1948-9366/full/v15/i5/965.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i5.965
Since the first clinical use of SpyGlass cholangioscopy (Microvasive Endoscopy; Boston Scientific Corp, Natick, MA, United States) in 2007, numerous clinical studies of the SpyGlass system have been published[1]. However, no definite guidelines were created for the application of SpyGlass cholangioscopy in cholangiocarcinoma (CC) mapping. One study showed that the overall procedure success rate was 89%, and the yielding rate of device-assisted biopsy for histological examination was 88%[2]. Another study showed that the accuracy of SpyGlass visual impression for differentiating malignant from benign ductal lesions was 89%, and the targeted biopsy accuracy rate was 82%[3].
Many patients diagnosed with early stage hilar CC who accepted radical left or right extended hepatectomy have a high recurrence rate (up to 52% 3 years after R0 resection)[4]. Intraepithelial neoplasia skip lesions and intraductal papillary neoplasm in the bile duct or indeterminate classification of CC could lead to the high recurrence rate[5,6]. The advent of SpyGlass cholangioscopy with high resolution images of the bile duct may provide a role in the precise localization of hilar CC. This will lead to more accurate and precise surgical planning.
Herein, we reported a case of hilar CC that was precisely mapped by SpyGlass cholangioscopy, which detected an intrahepatic duct (IHD) skip lesion. In addition, we demonstrated the potential benefit of preoperative SpyGlass cholangioscopy in the field of precise localization of hilar CC to guide the surgical plan.
A 52-year-old female presented with acute onset right upper quadrant abdominal pain for 1 d.
Three days prior to presentation, the patient experienced progressive tea-colored urine and generalized yellowish skin with an itching sensation. She had a fever of 38 °C.
The patient denied any past illness.
The patient denied any personal and family history.
Physical examination showed right upper quadrant tenderness without rebound pain.
The biochemistry test revealed hyperbilirubinemia (12.5 mg/dL; normal range: 0.2-1.5 mg/dL).
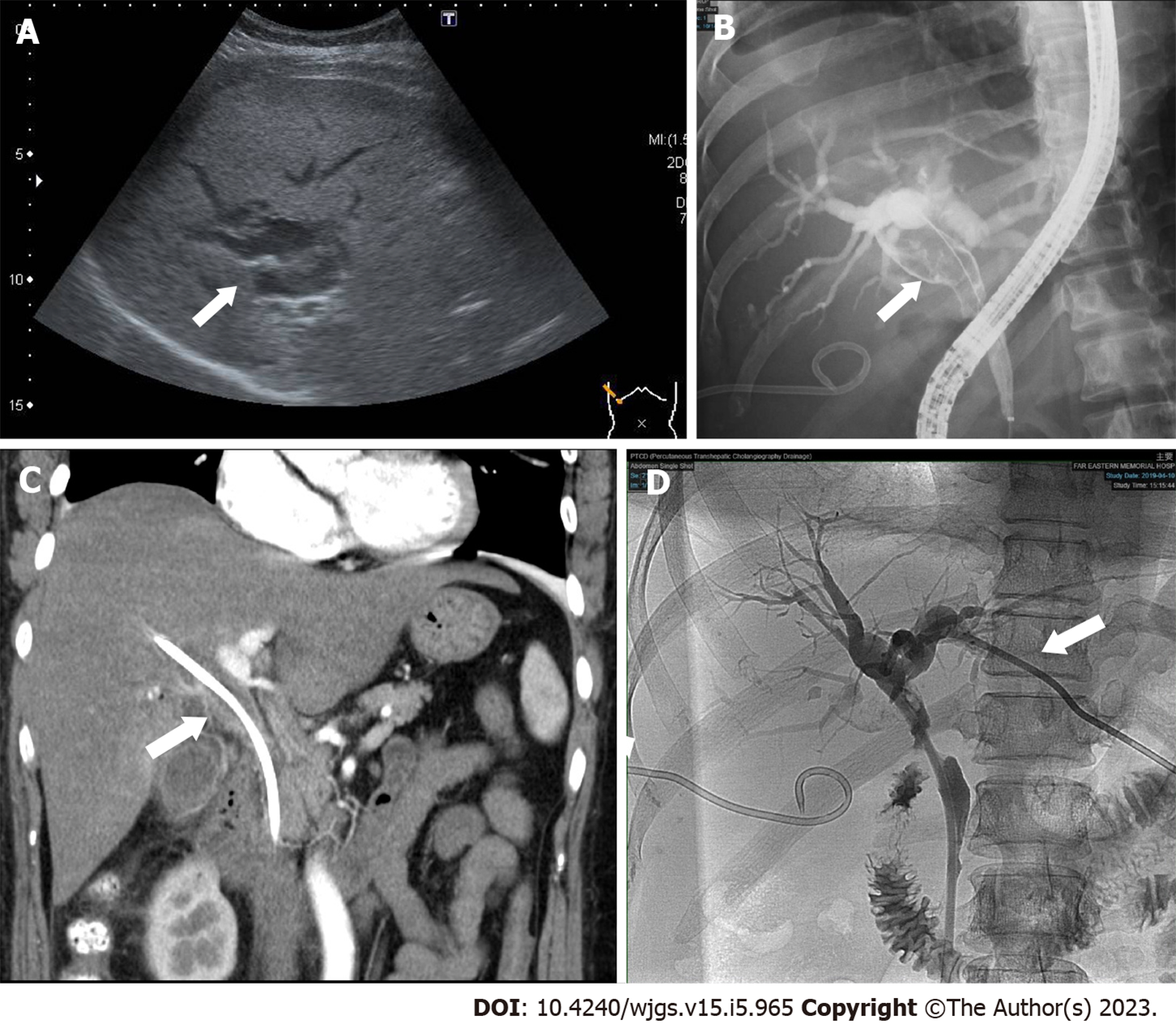
Subsequent abdominal sonography revealed acute calculous cholecystitis, suspected common bile duct (CBD) stone with dilated CBD, and bilateral IHDs (Figure 1A). Ultrasound-guided percutaneous gallbladder drainage and endoscopic retrograde cholangiopancreatography (ERCP) were performed to relieve the patient’s symptoms. The cholangiogram revealed a long filling defect from the proximal CBD to the common hepatic duct (CHD) confluence (Figure 1B). Moreover, some tissue fragments were extracted via balloon lithotripsy during the ERCP exam and sent for pathological examination. In addition, brushing cytology was performed. Endoscopic retrograde biliary drainage was placed into the right IHD to relieve the patient’s jaundice symptoms. Furthermore, subsequent contrast-enhanced computed tomography revealed similar findings as ERCP (Figure 1C). Later, left percutaneous transhepatic cholangiography drainage was performed to relieve unresolved jaundice after the endoscopic retrograde biliary drainage (Figure 1D). The patient’s hyperbilirubinemia was resolved gradually. The final pathology of the CHD lesion showed intraductal papillary neoplasm, with high-grade dysplasia.
CC, pT2aN0.
After serial work-up and management, a general surgeon was consulted for the curative surgery. Initially, extended left hepatectomy was planned for the patient because the patient’s right lobe was larger than the left lobe. This strategy would preserve more liver without preoperative portal vein embolization.
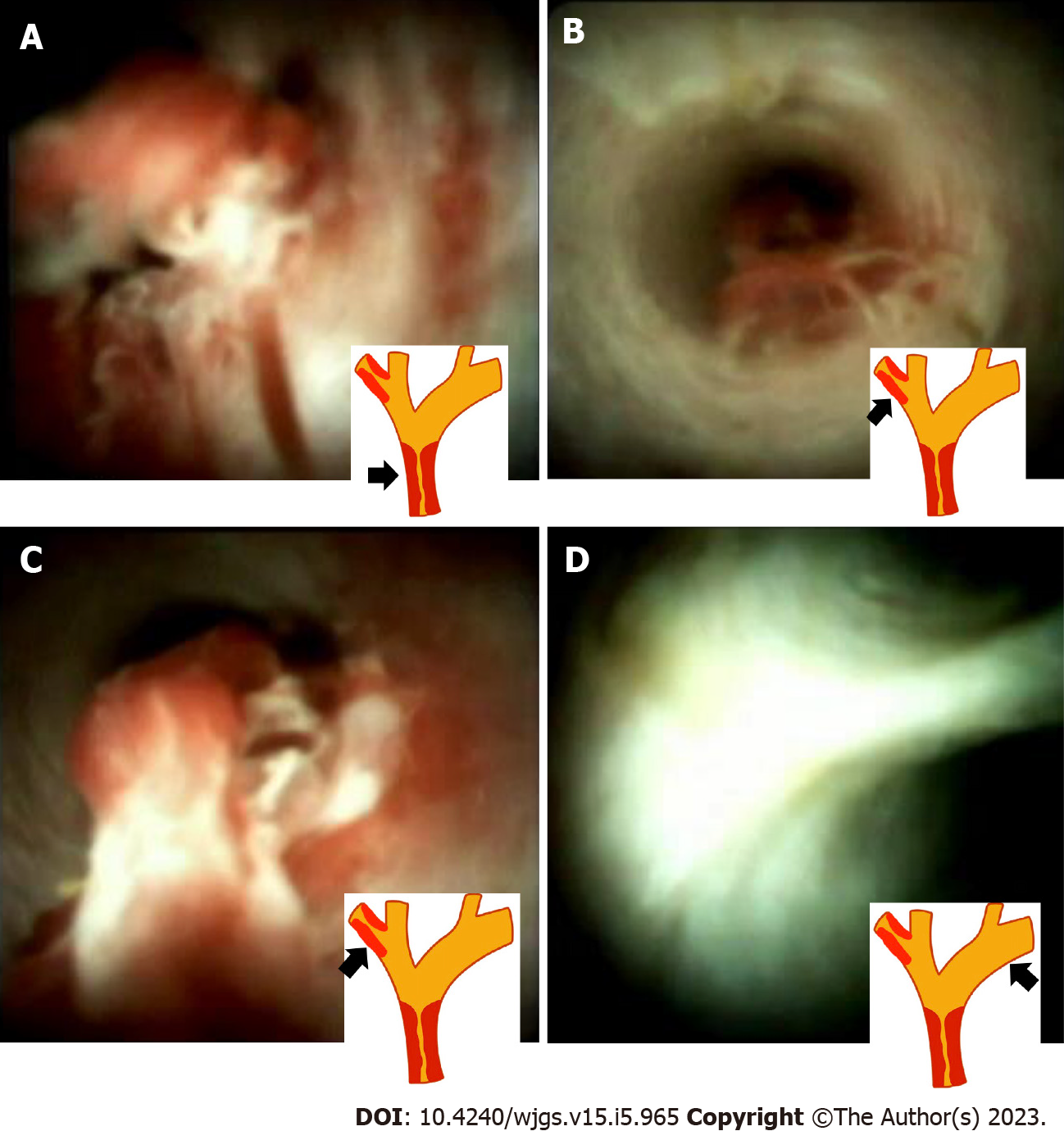
However, the surgical plan was modified after the SpyGlass cholangioscopy, which revealed an intraductal lesion from the CHD to the margin of bifurcation of the IHDs (Figure 2A). A skip lesion in the right posterior branch of the bile duct (2 cm proximally away from bifurcation of the CHD) (Figure 2B and C). The cholangioscopy showed normal appearance of the mucosa in the left proximal IHD (Figure 2D). Based on these findings, the surgeon changed the initial surgical plan of extended left hepatectomy (LH) to extended right hepatectomy (RH).
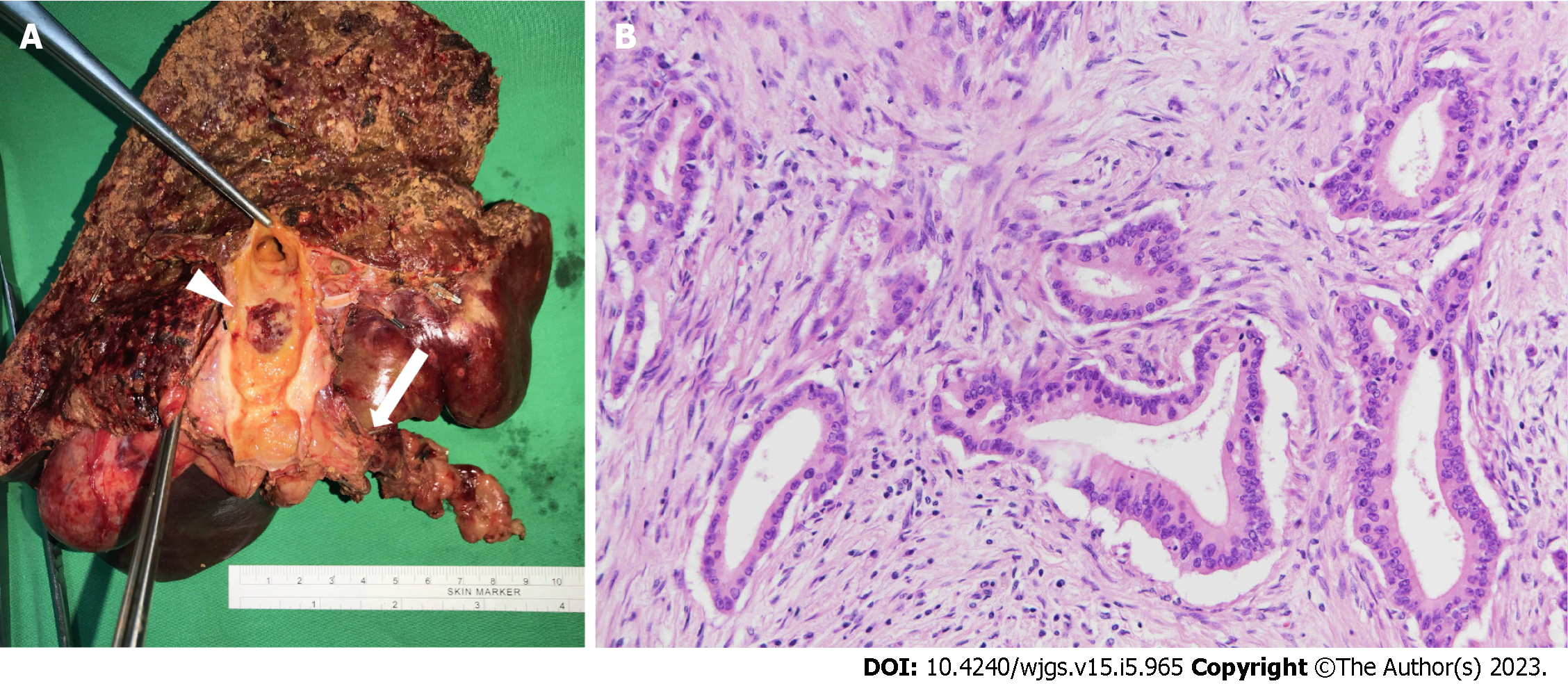
Before the operation, embolization of the right portal vein was performed because the volume of the left lobe of the liver would not be adequate after extended RH. Atrophy of the right lobe of the liver and enlargement of the left lobe of the liver was confirmed 4 wk later by imaging with indocyanine green (retention ratio: 6%; normal range: < 10 %). Laparoscopic extended RH was performed uneventfully 8 wk after portal vein embolization. The surgical specimen showed one skip lesion in the right posterior branch hepatic duct, which was compatible with the SpyGlass cholangioscopy findings (Figure 3A, arrow). The histology showed well differentiated CC that invaded beyond the wall of the bile duct to the surrounding adipose tissue (Figure 3B).
During the follow-up period of more than 3 years, the patient’s symptoms resolved and have not recurred. Liver triphasic computed tomography confirmed that the patient has remained disease free.
CC is the second most common hepatic malignancy, following hepatocellular carcinoma. Hilar CC is the most common type, accounting for 50%-60% of CC cases[6]. The only curative treatment is extended semihepatectomy with hilar resection. Therefore, choosing left or right extended hepatectomy is the most crucial issue for surgeons. The choice depends on the precise localization of the hilar lesion preoperatively. Before 2007, ERCP plus triphasic dynamic computed tomography or magnetic resonance cholangiopancreatography were the only methods to identify the location of the lesion and perform preoperative mapping[7,8].
Since the SpyGlass cholangioscopy system first appeared in 2007, many studies have shown its diagnostic and therapeutic value over indeterminate biliary strictures[9-11]. However, no studies have focused on the preoperative mapping of hilar CC. According to the previous studies and case series, the high recurrence rate of hilar CC may result from skip lesions or residual intraductal papillary neoplasm of the bile duct on the contralateral IHD[4]. For this case, a residual intraductal papillary neoplasm skip lesion in the right posterior branch of the bile duct was detected by SpyGlass cholangioscopy, which was valuable information for surgical planning. Typically for Bismuth type I or type II hilar CC, extended LH is preferred because extended LH and extended RH with en bloc resection have shown similar long-term survival. Extended LH hilar en bloc resections are more feasible and safer perioperatively and postoperatively[12,13]. However, because more precise information of the tumor extent was detected by SpyGlass cholangioscopy, the surgical plan was changed for our patient.
Based on the experience from this case, a two-step approach for obstructive jaundice caused by hilar CC was proposed: (1) Insertion of biliary plastic stents to relieve jaundice and to dilate the stricture site; and (2) Removal of biliary plastic stents after jaundice is resolved and examination of the hilar CC extent by SpyGlass cholangioscopy. Notably, cross-sectional images were still required for evaluation of nodal involvement and distal metastases. In addition, for patients with periductal-infiltrating type CC, SpyGlass cholangioscopy may have a limited role. Thus, combination of SpyGlass cholangioscopy, cross-sectional imaging, and cholangiogram may provide more precise information of hilar CC localization and staging, which will provide the most suitable treatment for patients.
SpyGlass cholangioscopy may have an important role in the precise localization of hilar CC for surgical planning. However, a larger scale study is warranted.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Taiwan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kurita A, Japan; Zharikov YO, Russia S-Editor: Fan JR L-Editor: A P-Editor: Zhang XD
| 1. | Chen YK, Pleskow DK. SpyGlass single-operator peroral cholangiopancreatoscopy system for the diagnosis and therapy of bile-duct disorders: a clinical feasibility study (with video). Gastrointest Endosc. 2007;65:832-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 285] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 2. | Chen YK, Parsi MA, Binmoeller KF, Hawes RH, Pleskow DK, Slivka A, Haluszka O, Petersen BT, Sherman S, Devière J, Meisner S, Stevens PD, Costamagna G, Ponchon T, Peetermans JA, Neuhaus H. Single-operator cholangioscopy in patients requiring evaluation of bile duct disease or therapy of biliary stones (with videos). Gastrointest Endosc. 2011;74:805-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 204] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 3. | Ramchandani M, Reddy DN, Gupta R, Lakhtakia S, Tandan M, Darisetty S, Sekaran A, Rao GV. Role of single-operator peroral cholangioscopy in the diagnosis of indeterminate biliary lesions: a single-center, prospective study. Gastrointest Endosc. 2011;74:511-519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 133] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 4. | Kobayashi A, Miwa S, Nakata T, Miyagawa S. Disease recurrence patterns after R0 resection of hilar cholangiocarcinoma. Br J Surg. 2010;97:56-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 5. | Geramizadeh B. Precursor Lesions of Cholangiocarcinoma: A Clinicopathologic Review. Clin Pathol. 2020;13:2632010X20925045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 6. | Banales JM, Cardinale V, Carpino G, Marzioni M, Andersen JB, Invernizzi P, Lind GE, Folseraas T, Forbes SJ, Fouassier L, Geier A, Calvisi DF, Mertens JC, Trauner M, Benedetti A, Maroni L, Vaquero J, Macias RI, Raggi C, Perugorria MJ, Gaudio E, Boberg KM, Marin JJ, Alvaro D. Expert consensus document: Cholangiocarcinoma: current knowledge and future perspectives consensus statement from the European Network for the Study of Cholangiocarcinoma (ENS-CCA). Nat Rev Gastroenterol Hepatol. 2016;13:261-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 731] [Cited by in RCA: 972] [Article Influence: 108.0] [Reference Citation Analysis (0)] |
| 7. | Hemming AW, Reed AI, Fujita S, Foley DP, Howard RJ. Surgical management of hilar cholangiocarcinoma. Ann Surg. 2005;241:693-9; discussion 699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 250] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 8. | Brandi G, Venturi M, Pantaleo MA, Ercolani G; GICO. Cholangiocarcinoma: Current opinion on clinical practice diagnostic and therapeutic algorithms: A review of the literature and a long-standing experience of a referral center. Dig Liver Dis. 2016;48:231-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 63] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 9. | Tieu AH, Kumbhari V, Jakhete N, Onyimba F, Patel Y, Shin EJ, Li Z. Diagnostic and therapeutic utility of SpyGlass(®) peroral cholangioscopy in intraductal biliary disease: single-center, retrospective, cohort study. Dig Endosc. 2015;27:479-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Lenze F, Bokemeyer A, Gross D, Nowacki T, Bettenworth D, Ullerich H. Safety, diagnostic accuracy and therapeutic efficacy of digital single-operator cholangioscopy. United European Gastroenterol J. 2018;6:902-909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 11. | Pereira P, Vilas-Boas F, Peixoto A, Andrade P, Lopes J, Macedo G. How SpyGlass™ May Impact Endoscopic Retrograde Cholangiopancreatography Practice and Patient Management. GE Port J Gastroenterol. 2018;25:132-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 12. | Kovalenko YA, Zharikov YO, Konchina NA, Gurmikov BN, Marinova LA, Zhao AV. Perihilar cholangiocarcinoma: A different concept for radical resection. Surg Oncol. 2020;33:270-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 13. | Bednarsch J, Czigany Z, Lurje I, Tacke F, Strnad P, Ulmer TF, Gaisa NT, Bruners P, Neumann UP, Lurje G. Left- vs right-sided hepatectomy with hilar en-bloc resection in perihilar cholangiocarcinoma. HPB (Oxford). 2020;22:437-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |