Published online Oct 27, 2023. doi: 10.4240/wjgs.v15.i10.2367
Peer-review started: July 1, 2023
First decision: July 18, 2023
Revised: August 2, 2023
Accepted: August 15, 2023
Article in press: August 15, 2023
Published online: October 27, 2023
Processing time: 118 Days and 5.7 Hours
Hereditary hemorrhagic telangiectasia (HHT) is an autosomal dominant genetic disorder with an incidence of approximately 1 in 5000 in the general population. It is characterized by vasodilation, which affects specific organs, such as the skin, mucous membranes, brain, lungs, gastrointestinal tract, liver, and others. How
A 68-year-old woman was admitted to the emergency department due to four consecutive days of abdominal pain and bloody stool and was subsequently diag
For patients with diffuse superior mesenteric AVFs, selective mesenteric arterial embolization may lead to positive short-term outcomes.
Core Tip: This case report describes a rare case of hereditary hemorrhagic telangiectasia (HHT) involving the portal system and identified a possible gene mutation site. For patients with diffuse superior mesenteric arteriovenous fistulas (AVFs), a selective mesenteric arterial embolization may lead to positive short-term outcomes. In the future, more studies are needed to determine the suitability of this treatment for patients with diffuse superior mesenteric AVFs (SMAVFs) associated with HHT. Additionally, more studies are required to investigate whether new sites of gene mutations can cause or contribute to the development of diffuse SMAVFs in patients with HHT.
- Citation: Wu JL, Zhao ZZ, Chen J, Zhang HW, Luan Z, Li CY, Zhao YM, Jing YJ, Wang SF, Sun G. Hereditary hemorrhagic telangiectasia involving portal venous system: A case report and review of the literature. World J Gastrointest Surg 2023; 15(10): 2367-2375
- URL: https://www.wjgnet.com/1948-9366/full/v15/i10/2367.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i10.2367
Hereditary hemorrhagic telangiectasia (HHT), also known as Osler-Weber-Rendu syndrome, is a common autosomal dominant genetic disease, with an estimated incidence of 1 in 5000[1]. HHT is characterized by vascular malformations that affect specific organs such as the skin, mucosa, brain, lungs, gastrointestinal tract, liver, and others. The most com
A 68-year-old female patient presented to the Emergency Department of our hospital with the chief complaint of abdo
Prior to admission, there was no obvious cause for abdominal stabbing pain, which continued to worsen and changed to dull pain in the left lower abdomen. She had experienced two episodes of dark red bloody stools (approximately 200 mL each time). After defecation, there was no improvement in abdominal pain, and these symptoms were accompanied by abdominal distension, nausea, and low back pain. The patient had no fever, vomiting, or other symptoms over the course of the disease.
The patient had a history of spontaneous epistaxis for 20 years and intermittent rectal bleeding for 10 years, which had been previously neglected in terms of sufficient attention and medical intervention.
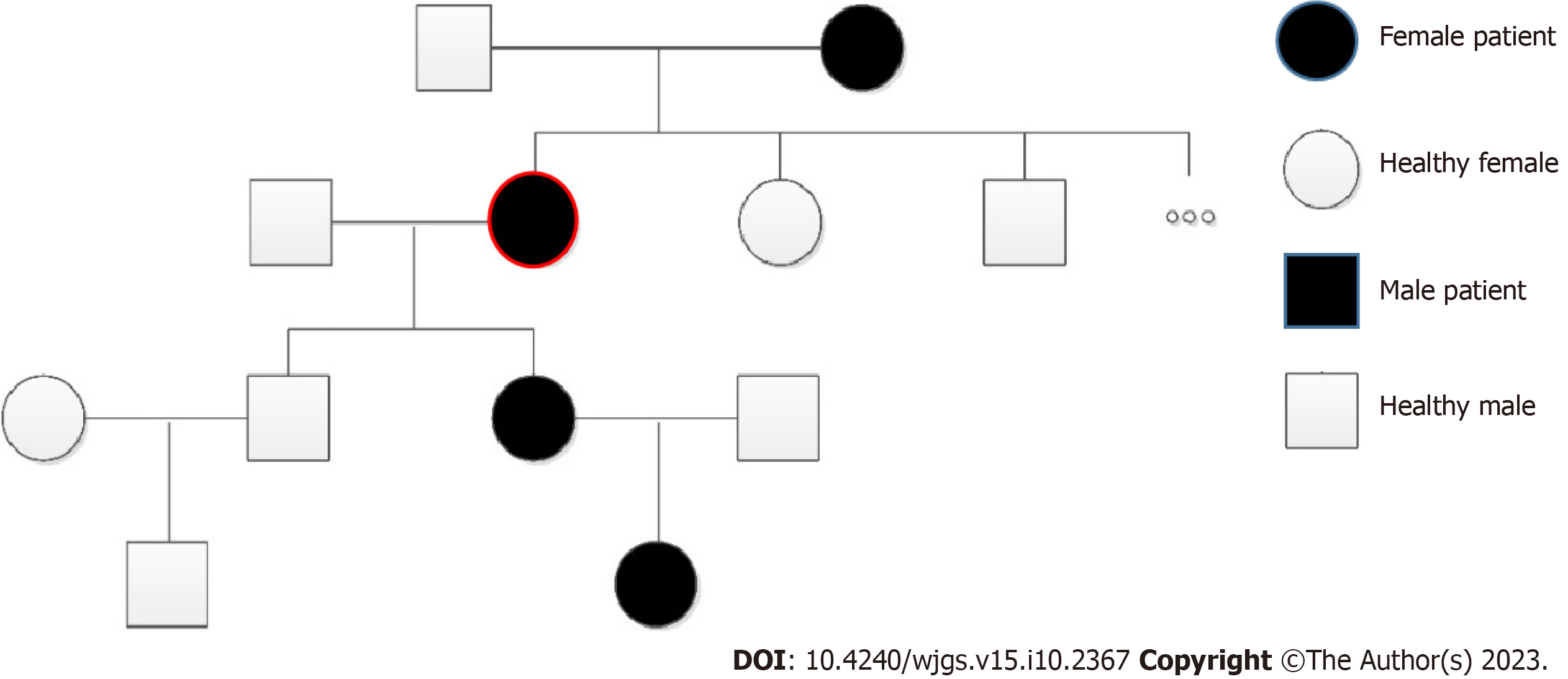
Some of her family members also had a long-term history of spontaneous epistaxis (Figure 1).
During the physical examination, capillary dilatation was identified in the patient’s lips. The patient’s abdomen was distended with tenderness in the lower left quadrant with no signs of rebound tenderness. Bowel sounds were 10 times per minute. No other identifiable abnormalities were found.
The routine blood examination revealed hemoglobin of 115 g/L (normal range: 110-150 g/L), white blood cell count of 8.7 × 109 [normal range: (4-10) × 109], and 81.6% neutrophils (normal range: 50%-70%). A fecal occult blood test was positive. Coagulation function indexes showed varying degrees of decline.
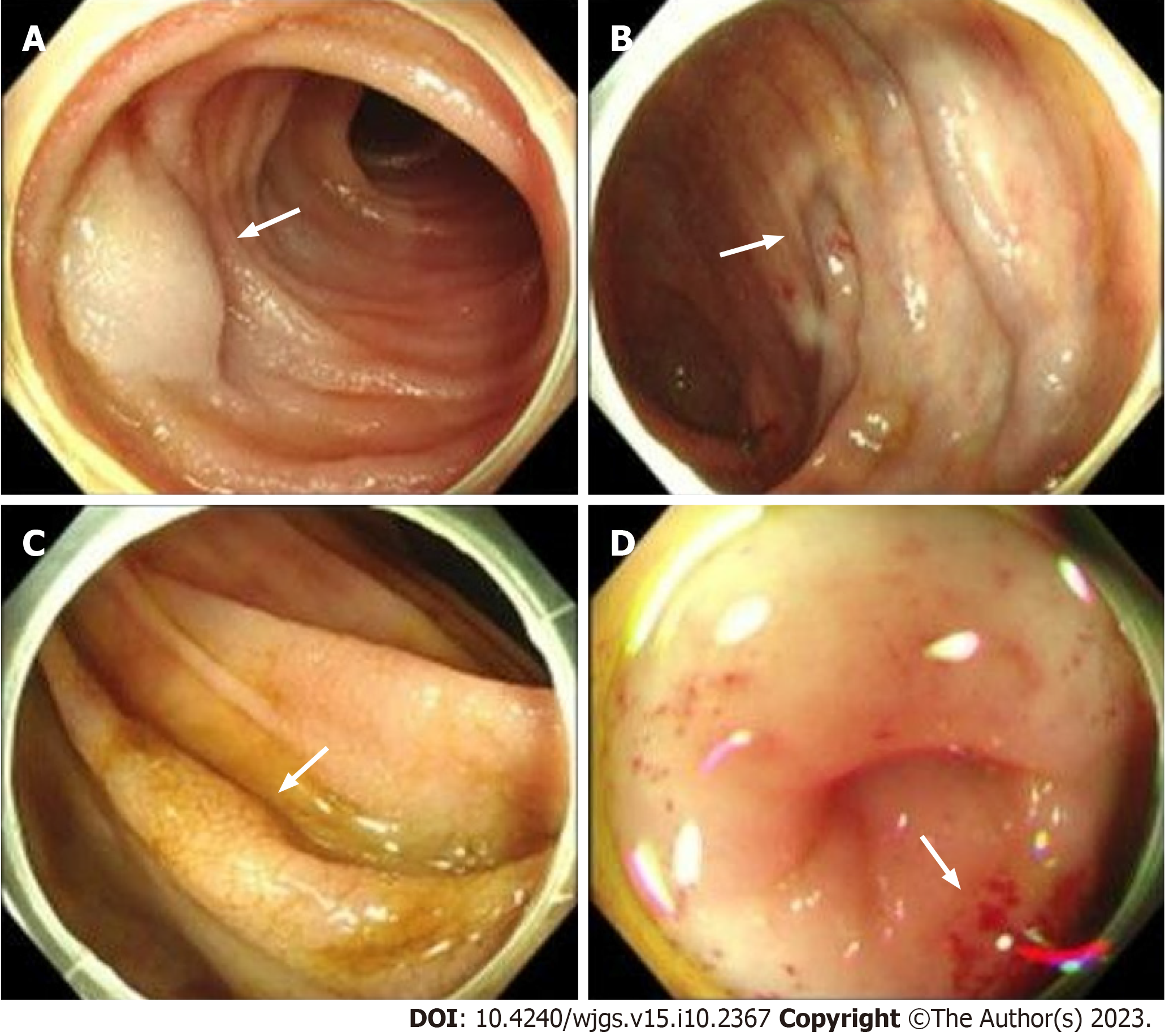
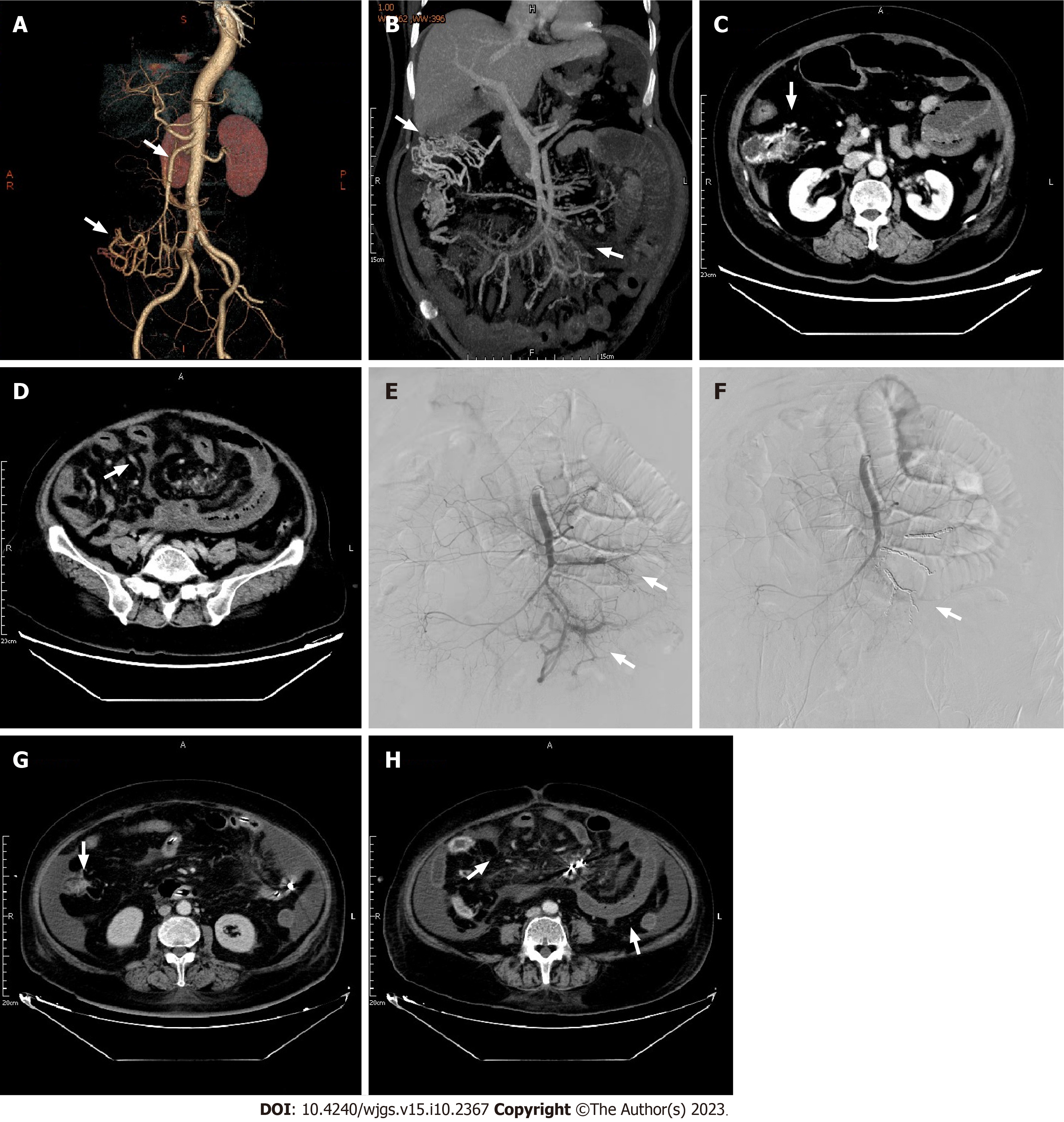
A gastroscopy revealed scattered congestion and edema of the duodenal descending and transverse mucosa, while a colonoscopy showed a blue venous aneurysm or varicose in the ileum at 10 cm from the ileocecal valve. The cecum and ascending colon showed migrated veins and varicose veins (Figures 2A and B). The mucosa was cracked and scale-like (Figures 2C and D). Computed tomography angiography (CTA) of abdominal vessels revealed early mesenteric venous perfusion in the arterial phase that involved the right lower abdomen and left middle abdominal superior mesenteric veins. In the venous phase, there were tortuous vessels around the right upper abdominal intestine, which were con
Based on the above information, the patient was ultimately diagnosed with HHT.
Due to the diffuse congestion and ischemia of the small intestine secondary to the superior mesenteric AVFs (SMAVFs), surgical intervention would involve resection of extensive intestinal segments, leading to adverse effects such as short bowel syndrome, anastomotic stenosis, and others. After being informed of these risks, the patient opted for interventional treatment. Therefore, selective embolization of the SMAVFs was performed. After careful evaluation, it was found that not all sites of the AVFs could be embolized during the selective procedure. If all fistula tracts had been occluded, the patient would have likely developed intestinal ischemic necrosis. Hence, the proportion of embolism had to be app
After the procedure, the patient experienced an improvement in abdominal pain, bloating, and diarrhea. An abdominal CT showed reduced intestinal wall edema with no abnormal perfusion at the embolization site (Figures 3G and H). One week later, the patient had recovered well and was discharged. One month after discharge, the patient was readmitted with abdominal pain and bloody stools. Remaining AVFs were identified and evaluated, and were believed to have caused secondary bowel necrosis. However, due to financial difficulties, the patient declined surgery and conservative treatment had to be adopted. One month later, the patient died from bowel necrosis and abdominal infection.
After searching various databases, we ultimately identified six cases similar to the one reported herein (Table 1). Accor
| Serial number | Publication date | Gender | Age | Clinical manifestation | Treatment | Prognosis | Ref. |
| 1 | 1978 | Male | 63 | Severe epistaxis; gastrointestinal bleeding; ileocolic AVM | NA | NA | [29] |
| 2 | 2003 | Male | 68 | Gastrointestinal bleeding; epistaxis; telangiectasia of skin; pulmonary AVM | Embolization for AVM | No recurrence after 6 mo of follow-up | [30] |
| 3 | 2006 | Female | 73 | Epistaxis; hemolytic anemia; mesenteric AVM | NA | NA | [31] |
| 4 | 2007 | Male | 72 | Epistaxis; gastric telangiectasia; superior mesenteric AVM | Treatment for ascites and varicose veins | NA | [32] |
| 5 | 2016 | Female | 46 | Epistaxis; telangiectasia of skin; AVM of splenic flexure of colon | NA | NA | [33] |
| 6 | 2014 | Female | 68 | Duodenal AVM | NA | NA | [34] |
Internationally, the clinical diagnosis of HHT is mainly based on the Curacao criteria[3]. These criteria have a broad application, especially for the identification of patients and non-patients[1]. The development of genetic technology enables us to diagnose HHT more accurately, particularly for adolescents and children who have no clinical manifestations of epistaxis or visible telangiectasia but do have unknown vascular malformations[4]. HHT can be divided into six common types based on different genotypes (Table 2). Primarily, sequencing methods of the ENG and ACVRL1 genes are currently used[5-8]. Although, a negative result in genetic testing cannot rule out the diagnosis of HHT, since many sporadic cases have unique gene mutations that are not one of the common variations. Many studies have found new variations in the above two gene loci. For example, in a study of a Canadian family, nine new mutation loci were found in the ENG and ACVRL1 genes[5]. In another investigation of a Chinese family, no significant mutation was found in the ENG, ACVRL1, or SMDH genes. After whole exome sequencing, new and unreported variations were found in the N-ethylmaleimide-sensitive factor attachment protein gamma gene[9]. In a genomic investigation conducted in the United Kingdom, comprehensive whole-genome sequencing uncovered a previously unknown heterozygous GDF2 sequence variation in all three affected individuals within an HHT family. Notably, gene screening results for ACVRL1, ENG, and mothers against decapentaplegic homolog 4 (SMAD4) yielded negative findings. In vitro experiments provided compel
| Type | HHT1 | HHT2 | HHT3 | HHT4 | HHT5 | HHT + JPS |
| Mutated gene locus | ENG | ACVRL1 | No specific gene | No specific gene | BMPq/GDF2 | MADH4 |
| Location | 9q34 | 12q3 | 5q31 | 7q14 | NA | NA |
| Affected protein | Endoglin protein | AKL1 protein | NA | NA | NA | SMAD4 protein |
| Proportion | 61% | 37% | NA | NA | NA | 2% |
The different genotypes of HHT can also lead to different clinical manifestations in patients. Studies have found that HHT1 is more prone to cerebral arteriovenous malformations, while HHT2 is more prone to hepatic arteriovenous malformations[11], and the age at diagnosis of HHT1 patients is generally young[12]. A specific type of HHT caused by MADH4 mutations is usually accompanied by juvenile polyposis syndrome[13]. Prior studies have suggested that the ENG, ACVRL1, and MADH4 genes all play a role in regulating cell signaling through the transforming growth factor beta (TGF-β) signaling pathway. The TGF-β family activates the ENG glycoprotein and ALK1 on the cell membrane, leading to changes in gene transcription levels within the cell nucleus that regulate cell proliferation, differentiation, migration, apoptosis, and secretion, ultimately affecting vascular structure and function[14]. Furthermore, studies have found that clinical manifestations differ amongst patients within families, with vascular malformations only appearing in specific organs, suggesting that interpretation solely from a genetic variation perspective cannot fully explain the occurrence of HHT. Therefore, another hypothesis known as the “second strike” theory has been suggested[15]. Firstly, heterozygous variations in the HHT gene lead to the loss of a single allele that encodes for endothelium, which is the first strike. Secondly, environmental or genetic factors provide the second strike. These two factors can also have a synergistic effect. The appearance of HHT clinical symptoms may be the result of multiple factors working together, controlled by complex molecular mechanisms. The cause of the mesenteric AVFs in this patient is currently unknown and requires further study. The “second-strike” hypothesis has been proposed for many years but previously lacked convincing evidence. However, with the advancement of sequencing technologies, Snellings et al[16] have successfully validated three hypo
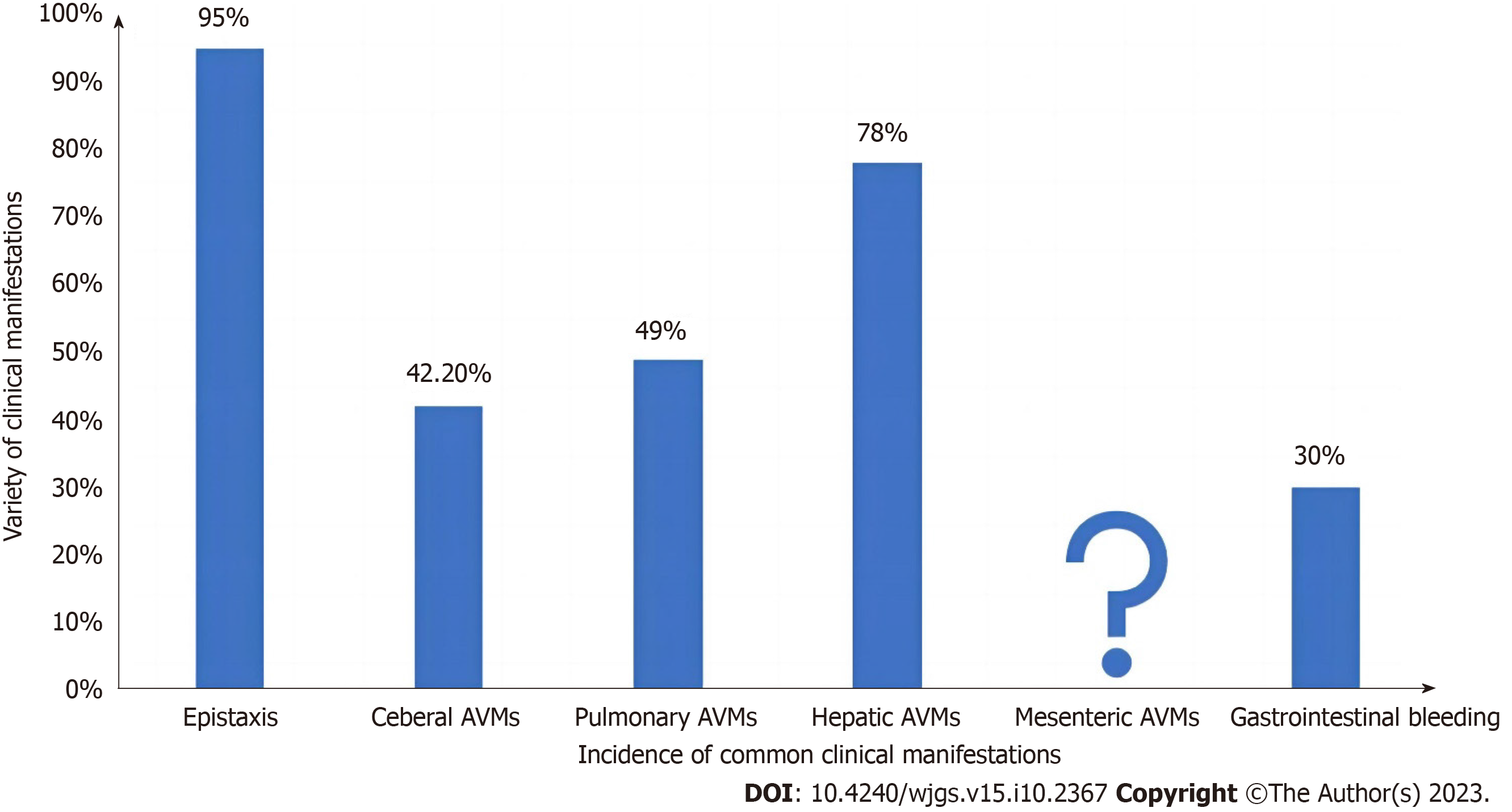
Common clinical manifestations of HHT include epistaxis, cerebral vascular malformations, pulmonary arteriovenous malformations (PAVM), gastrointestinal bleeding, and hepatic arteriovenous malformations (Figure 4). HHT patients typically exhibit lower social index values for EuroQol-visual analogue scale scores, indicating a reduced quality of life compared to the general population. Notably, they attribute a substantial impact on their quality of life to epistaxis (nose
The most common treatment for anemia caused by gastrointestinal bleeding in HHT patients is iron supplementation or blood transfusion, which is not sustainable for long-term treatment. Currently, the treatment options for chronic gastrointestinal bleeding in HHT patients include medication and endoscopic treatment, but they cannot cure AVFs. Al-Samkari and Eng[22] presented several cases of drug therapy for complex HHT-related vascular malformations, all of which achieved favorable treatment outcomes. However, these cases involved more severe manifestations of common symptoms rather than rare clinical presentations similar to the present case. Therefore, the treatment has limited rele
The patient in this case did not exhibit a genetic mutation commonly associated with HHT. Furthermore, for patients with diffuse SMAVF, selective mesenteric arterial embolization may produce some positive outcomes. However, it is still unclear whether selective superior mesenteric arterial embolization is always advantageous in the long term or whether immediate surgery would be more beneficial in cases of intestinal necrosis. Researchers need to continue exploring new pathogenic mutations and the most appropriate treatment methods for rare HHT symptoms, to provide a basis for future prevention and diagnosis, and to deepen our understanding of HHT.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Baysal M, Turkey; Popovic DD, Serbia S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
| 1. | Faughnan ME, Mager JJ, Hetts SW, Palda VA, Lang-Robertson K, Buscarini E, Deslandres E, Kasthuri RS, Lausman A, Poetker D, Ratjen F, Chesnutt MS, Clancy M, Whitehead KJ, Al-Samkari H, Chakinala M, Conrad M, Cortes D, Crocione C, Darling J, de Gussem E, Derksen C, Dupuis-Girod S, Foy P, Geisthoff U, Gossage JR, Hammill A, Heimdal K, Henderson K, Iyer VN, Kjeldsen AD, Komiyama M, Korenblatt K, McDonald J, McMahon J, McWilliams J, Meek ME, Mei-Zahav M, Olitsky S, Palmer S, Pantalone R, Piccirillo JF, Plahn B, Porteous MEM, Post MC, Radovanovic I, Rochon PJ, Rodriguez-Lopez J, Sabba C, Serra M, Shovlin C, Sprecher D, White AJ, Winship I, Zarrabeitia R. Second International Guidelines for the Diagnosis and Management of Hereditary Hemorrhagic Telangiectasia. Ann Intern Med. 2020;173:989-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 279] [Article Influence: 55.8] [Reference Citation Analysis (0)] |
| 2. | Kjeldsen AD, Vase P, Green A. Hereditary haemorrhagic telangiectasia: a population-based study of prevalence and mortality in Danish patients. J Intern Med. 1999;245:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 284] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 3. | Shovlin CL, Guttmacher AE, Buscarini E, Faughnan ME, Hyland RH, Westermann CJ, Kjeldsen AD, Plauchu H. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome). Am J Med Genet. 2000;91:66-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 4. | Faughnan ME, Palda VA, Garcia-Tsao G, Geisthoff UW, McDonald J, Proctor DD, Spears J, Brown DH, Buscarini E, Chesnutt MS, Cottin V, Ganguly A, Gossage JR, Guttmacher AE, Hyland RH, Kennedy SJ, Korzenik J, Mager JJ, Ozanne AP, Piccirillo JF, Picus D, Plauchu H, Porteous ME, Pyeritz RE, Ross DA, Sabba C, Swanson K, Terry P, Wallace MC, Westermann CJ, White RI, Young LH, Zarrabeitia R; HHT Foundation International - Guidelines Working Group. International guidelines for the diagnosis and management of hereditary haemorrhagic telangiectasia. J Med Genet. 2011;48:73-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 698] [Article Influence: 43.6] [Reference Citation Analysis (0)] |
| 5. | Prigoda NL, Savas S, Abdalla SA, Piovesan B, Rushlow D, Vandezande K, Zhang E, Ozcelik H, Gallie BL, Letarte M. Hereditary haemorrhagic telangiectasia: mutation detection, test sensitivity and novel mutations. J Med Genet. 2006;43:722-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 92] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 6. | Cymerman U, Vera S, Karabegovic A, Abdalla S, Letarte M. Characterization of 17 novel endoglin mutations associated with hereditary hemorrhagic telangiectasia. Hum Mutat. 2003;21:482-492. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 7. | Bossler AD, Richards J, George C, Godmilow L, Ganguly A. Novel mutations in ENG and ACVRL1 identified in a series of 200 individuals undergoing clinical genetic testing for hereditary hemorrhagic telangiectasia (HHT): correlation of genotype with phenotype. Hum Mutat. 2006;27:667-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | Kuehl HK, Caselitz M, Hasenkamp S, Wagner S, El-Harith el-HA, Manns MP, Stuhrmann M. Hepatic manifestation is associated with ALK1 in hereditary hemorrhagic telangiectasia: identification of five novel ALK1 and one novel ENG mutations. Hum Mutat. 2005;25:320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Xu Y, Zhang YB, Liang LJ, Tian JL, Lin JM, Wang PP, Li RH, Gu ML, Gao ZC. NAPG mutation in family members with hereditary hemorrhagic telangiectasia in China. BMC Pulm Med. 2021;21:197. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 10. | Balachandar S, Graves TJ, Shimonty A, Kerr K, Kilner J, Xiao S, Slade R, Sroya M, Alikian M, Curetean E, Thomas E, McConnell VPM, McKee S; Boardman-Pretty F; Devereau A, Fowler TA, Caulfield MJ, Alton EW, Ferguson T, Redhead J, McKnight AJ, Thomas GA; Genomics England Research Consortium, Aldred MA, Shovlin CL. Identification and validation of a novel pathogenic variant in GDF2 (BMP9) responsible for hereditary hemorrhagic telangiectasia and pulmonary arteriovenous malformations. Am J Med Genet A. 2022;188:959-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 38] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 11. | Bayrak-Toydemir P, McDonald J, Markewitz B, Lewin S, Miller F, Chou LS, Gedge F, Tang W, Coon H, Mao R. Genotype-phenotype correlation in hereditary hemorrhagic telangiectasia: mutations and manifestations. Am J Med Genet A. 2006;140:463-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 157] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 12. | Sánchez-Martínez R, Iriarte A, Mora-Luján JM, Patier JL, López-Wolf D, Ojeda A, Torralba MA, Juyol MC, Gil R, Añón S, Salazar-Mendiguchía J, Riera-Mestre A; RiHHTa Investigators of the Rare Diseases Working Group from the Spanish Society of Internal Medicine. Current HHT genetic overview in Spain and its phenotypic correlation: data from RiHHTa registry. Orphanet J Rare Dis. 2020;15:138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 13. | Kroon S, Snijder RJ, Faughnan ME, Mager HJ. Systematic screening in hereditary hemorrhagic telangiectasia: a review. Curr Opin Pulm Med. 2018;24:260-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Hata A, Lagna G. Deregulation of Drosha in the pathogenesis of hereditary hemorrhagic telangiectasia. Curr Opin Hematol. 2019;26:161-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Bernabeu C, Bayrak-Toydemir P, McDonald J, Letarte M. Potential Second-Hits in Hereditary Hemorrhagic Telangiectasia. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 16. | Snellings DA, Gallione CJ, Clark DS, Vozoris NT, Faughnan ME, Marchuk DA. Somatic Mutations in Vascular Malformations of Hereditary Hemorrhagic Telangiectasia Result in Bi-allelic Loss of ENG or ACVRL1. Am J Hum Genet. 2019;105:894-906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 94] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 17. | Zarrabeitia R, Fariñas-Álvarez C, Santibáñez M, Señaris B, Fontalba A, Botella LM, Parra JA. Quality of life in patients with hereditary haemorrhagic telangiectasia (HHT). Health Qual Life Outcomes. 2017;15:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 18. | Brinjikji W, Iyer VN, Wood CP, Lanzino G. Prevalence and characteristics of brain arteriovenous malformations in hereditary hemorrhagic telangiectasia: a systematic review and meta-analysis. J Neurosurg. 2017;127:302-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 64] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 19. | Shin SM, Kim HK, Crotty EJ, Hammill AM, Wusik K, Kim DH. CT Angiography Findings of Pulmonary Arteriovenous Malformations in Children and Young Adults With Hereditary Hemorrhagic Telangiectasia. AJR Am J Roentgenol. 2020;214:1369-1376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 20. | Al-Samkari H. Hereditary hemorrhagic telangiectasia: systemic therapies, guidelines, and an evolving standard of care. Blood. 2021;137:888-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 21. | Ginon I, Decullier E, Finet G, Cordier JF, Marion D, Saurin JC, Dupuis-Girod S. Hereditary hemorrhagic telangiectasia, liver vascular malformations and cardiac consequences. Eur J Intern Med. 2013;24:e35-e39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Al-Samkari H, Eng W. A precision medicine approach to hereditary hemorrhagic telangiectasia and complex vascular anomalies. J Thromb Haemost. 2022;20:1077-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 23. | Grujić D, Knežević A, Vojvodić S, Grujić B. Superior Mesenteric Arteriovenous Fistula Presenting with Massive Lethal Upper Gastrointestinal Bleeding 14 Years after Small Bowel Resection. Balkan Med J. 2015;32:214-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Athanasiou A, Michalinos A, Alexandrou A, Georgopoulos S, Felekouras E. Inferior mesenteric arteriovenous fistula: case report and world-literature review. World J Gastroenterol. 2014;20:8298-8303. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 27] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 25. | Shintani T, Mitsuoka H, Masuda M. Transcatheter coil embolization of an iatrogenic superior mesenteric arteriovenous fistula: report of a case. Surg Today. 2011;41:556-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 26. | Wu CG, Li YD, Li MH. Post-traumatic superior mesenteric arteriovenous fistula: endovascular treatment with a covered stent. J Vasc Surg. 2008;47:654-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 27. | Li YL, Zhao CL, He YH, Sun Y, Mao HX, Chang N, Yu Y. [The clinical characteristics and treatment of superior mesenteric arteriovenous fistula]. Henan Med Res. 2019;. |
| 28. | Liu C, Wu H, Fu Z, Tang M, Ruan H, Zhang L. Superior mesenteric arteriovenous fistula presenting as diarrhea: a case report and literature review. Am J Transl Res. 2021;13:8172-8178. [PubMed] |
| 29. | Sörensen R, Holtz U, Banzer D, Khalil M, Hirner A. Differential diagnosis of early opacification of the portal vein and its tributaries during arteriography. Cardiovasc Radiol. 1978;1:179-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 30. | Inouye P, Marcon N, Pugash RA, Hyland RH, Faughnan ME. Embolization of a duodenal arteriovenous malformation in hereditary hemorrhagic telangiectasia: case report and review of the literature. Can J Gastroenterol. 2003;17:661-665. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 31. | van Bon AC, Goey SH, Stuyt PM. A woman with haemolytic anaemia. Neth J Med. 2006;64:259, 261. [PubMed] |
| 32. | Goto T, Dohmen T, Yoneyama K, Monma A, Ohshima S, Shibuya T, Kataoka E, Segawa D, Sato W, Takeuchi S, Anezaki Y, Ishii H, Yamada I, Komatsu M, Watanabe S. Osler-Weber-Rendu disease with esophageal varices and hepatic nodular change. Intern Med. 2007;46:849-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 33. | Ding J, Li X, Song W, Zhao D, Li H, Xia Z. [Analysis of image features of multi-detector helical computed tomography in hereditary hemorrhagic telangiectasia]. Beijing Med J. 2016;. |
| 34. | Zhou S, Song W, He H. [MDCT Findings of Hereditary Hemorrhagic Telangiectasia and Its Clinical Significance]. J Clin Radiol. 2014;. |