Published online Jan 27, 2023. doi: 10.4240/wjgs.v15.i1.114
Peer-review started: September 28, 2022
First decision: November 27, 2022
Revised: November 30, 2022
Accepted: December 23, 2022
Article in press: December 23, 2022
Published online: January 27, 2023
Processing time: 111 Days and 18.7 Hours
A meshoma formation and erosion to the small intestine is rare. Herein, we report one case of a meshoma that was not treated early; causing it to displace and erode the small intestine, with infection, complete control of symptoms was achieved after removal of the infected patch mass, no recurrence of hernia after 2 years of follow-up.
A 62-year-old male patient presented with recurrent abdominal pain repeatedly for 1 wk, which has worsened 2 d before admition, accompanied by fever. Five years before presentation he underwent right inguinal hernia Plug and patch repair approach. Two years ago, a computed tomography scan revealed a right lower abdominal mass with soft tissue density, measuring approximately 30 mm × 17 mm, which was diagnosed as meshoma that was not treated. The patient had poorly controlled diabetes in the past year.
The formation of meshoma is rare, and that if not treated in time it might erode and require resection of the involved organ.
Core Tip: The formation of meshoma increases the risk of infection. According to the literature, keep the surgical field clean when placing the mesh, pay attention to the flatness of the mesh, avoid curling and folding, and avoid any direct contact with the viscera. After the formation of the meshoma, surgery to remove the meshoma as early as possible is recommended.
- Citation: Wu JF, Chen J, Hong F. Intestinal erosion caused by meshoma displacement: A case report. World J Gastrointest Surg 2023; 15(1): 114-120
- URL: https://www.wjgnet.com/1948-9366/full/v15/i1/114.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i1.114
Tension-free repair with mesh has become a standard surgical modality in adult inguinal hernia repair[1,2], significantly reducing the recurrence rate after inguinal hernia repair. As the use of polypropylene mesh in tension-free hernia repair has become more widespread, the clinical problems associated with it have also received increasing attention. At present, it has been proved that the foreign body reaction caused by implanted prosthetic materials can cause a series of complications, such as mesh displacement, adhesion and erosion, meshoma, chronic pain and even mesh infection.
Most mesh infections are acute, which usually occur during postoperative hospitalization or within 1-2 wk of discharge. The infection is mainly superficial and rarely involves the mesh. If not treated promptly, it can develop into a chronic mesh infection, which often ends up involving the mesh, forming a chronic infected sinus tract and causing delayed wound healing. Clinical manifestations of mesh infection include fever, painful local swelling, scleroma, erythema and even purulent discharge and fistula formation with the skin, the rate of mesh infection in open hernia repair is higher than that in laparoscopic hernia repair[3].
Conservative treatment including intravenous antibiotics, percutaneous puncture drainage or negative pressure suction to the wound has a high failure rate as the mesh is already infected and persists as a foreign body deep as a source of infection[4,5]. If the wound is not healed after repeated debridement and dressing change, the infected mesh should be removed as soon as possible[6]. It is still the most commonly used method to treat infection in clinic practice.
A 62-year-old male patient was admitted to the hospital with right lower abdominal pain for 1 wk, which has worsened in the last 2 d.
The patient’s diabetes found in the past year and poor control of blood glucose level.
He had a history of multiple inguinal hernia repairs and had undergone a non-mesh repair of a left inguinal hernia at the age of 40 years. At the age of 57, he was diagnosed with a right inguinal hernia and underwent a plug and patch approach. At the age of 60, he underwent a transabdominal preperitoneal patch procedure for a recurrent left inguinal hernia.
The patient had no family history of inguinal hernia disease.
Temperature 38.2 °C, heart rate 118 bpm, a hard mass was palpated in the right lower abdomen, about 4 cm × 3 cm in extent, poorly defined, with localized skin pressure and rebound pain, no myalgias, bowel sounds 4 bpm.
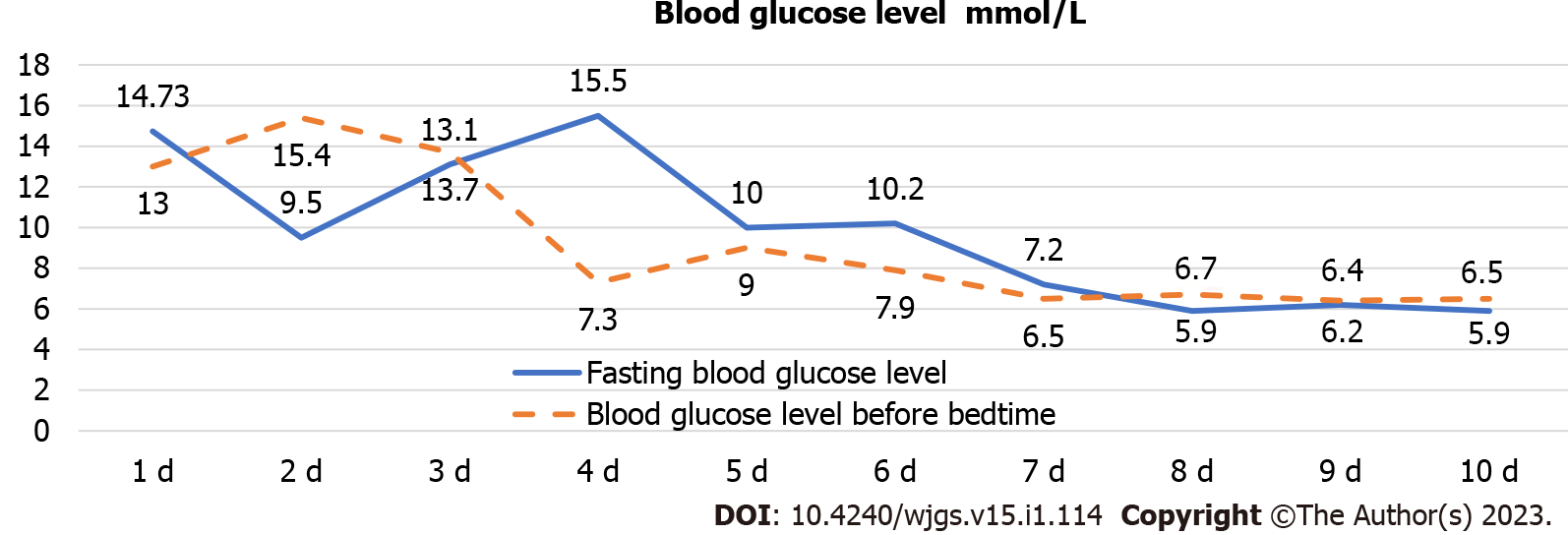
Complete blood count: White blood count (WBC) 12.5, reference 3.5-9.5 with units of 109/L; neutrophils 11.4, reference 1.8-6.3 with units of 109/L; C-reactive protein (CRP) 146.56, reference 0-8 with units of mg/L; fecal occult blood test (+); fasting blood sugar 14.73, reference 3.89-6.11 with units of mmol/L; hemoglobin A1c 12.2%, reference 3.6-6.5.
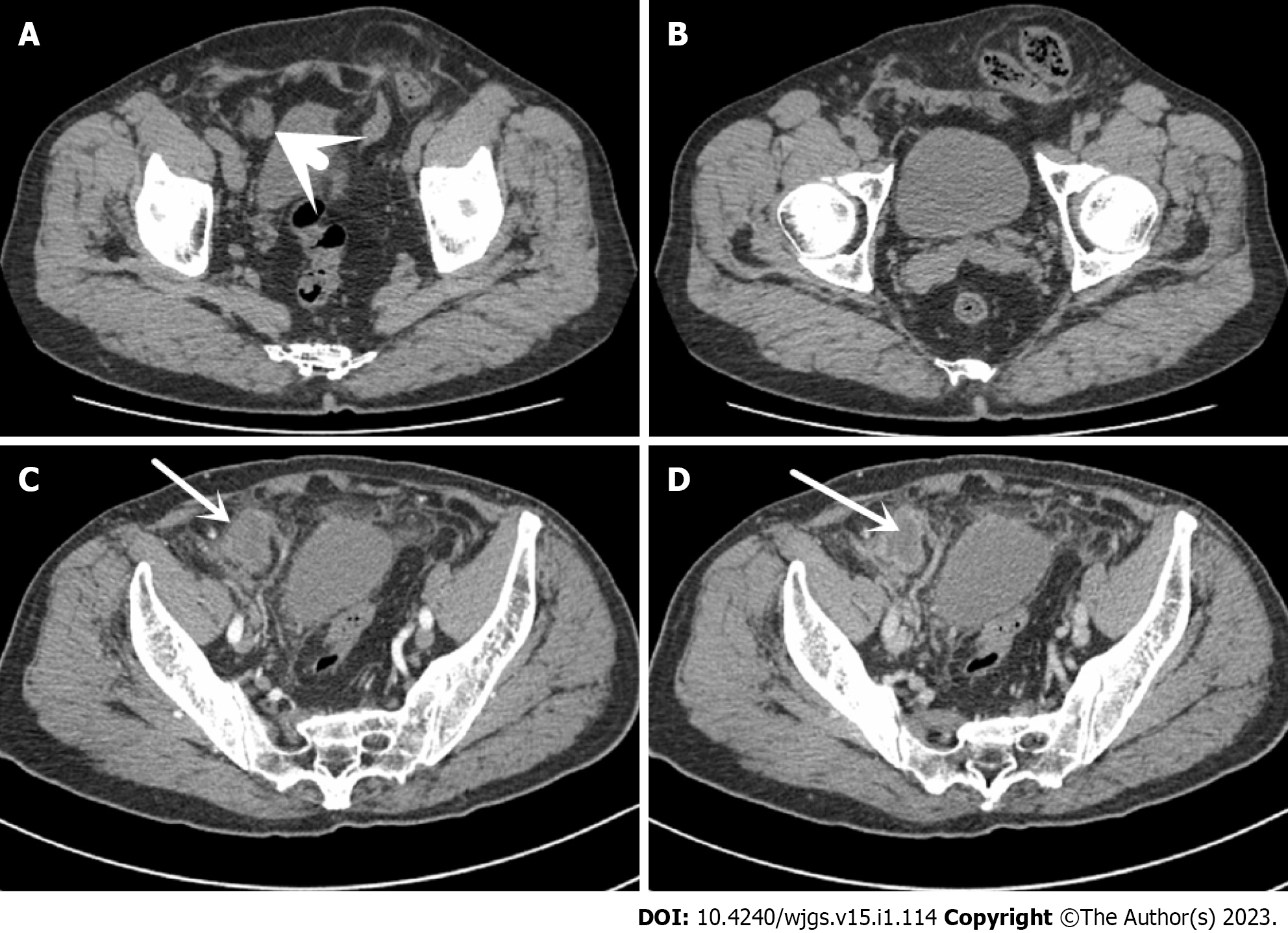
A computed tomography (CT) scan 2 years ago revealed a sigmoid herniation into the left scrotum and a right lower abdominal mass with soft tissue density, measuring approximately 30 mm × 17 mm, with a clear surrounding fatty space. This preoperative contrast-enhanced CT scan of abdomen: a mass soft tissue density shadow with a size of about 32 mm × 26 mm can be seen in the lower right abdomen, with lower density in the center of the lesion, poorly defined borders and blurred surrounding fatty spaces, with enhanced edges and no enhancement in the central region (Figure 1).
Mesh infection, meshoma, gastrointestinal bleeding, and type 2 diabetes.
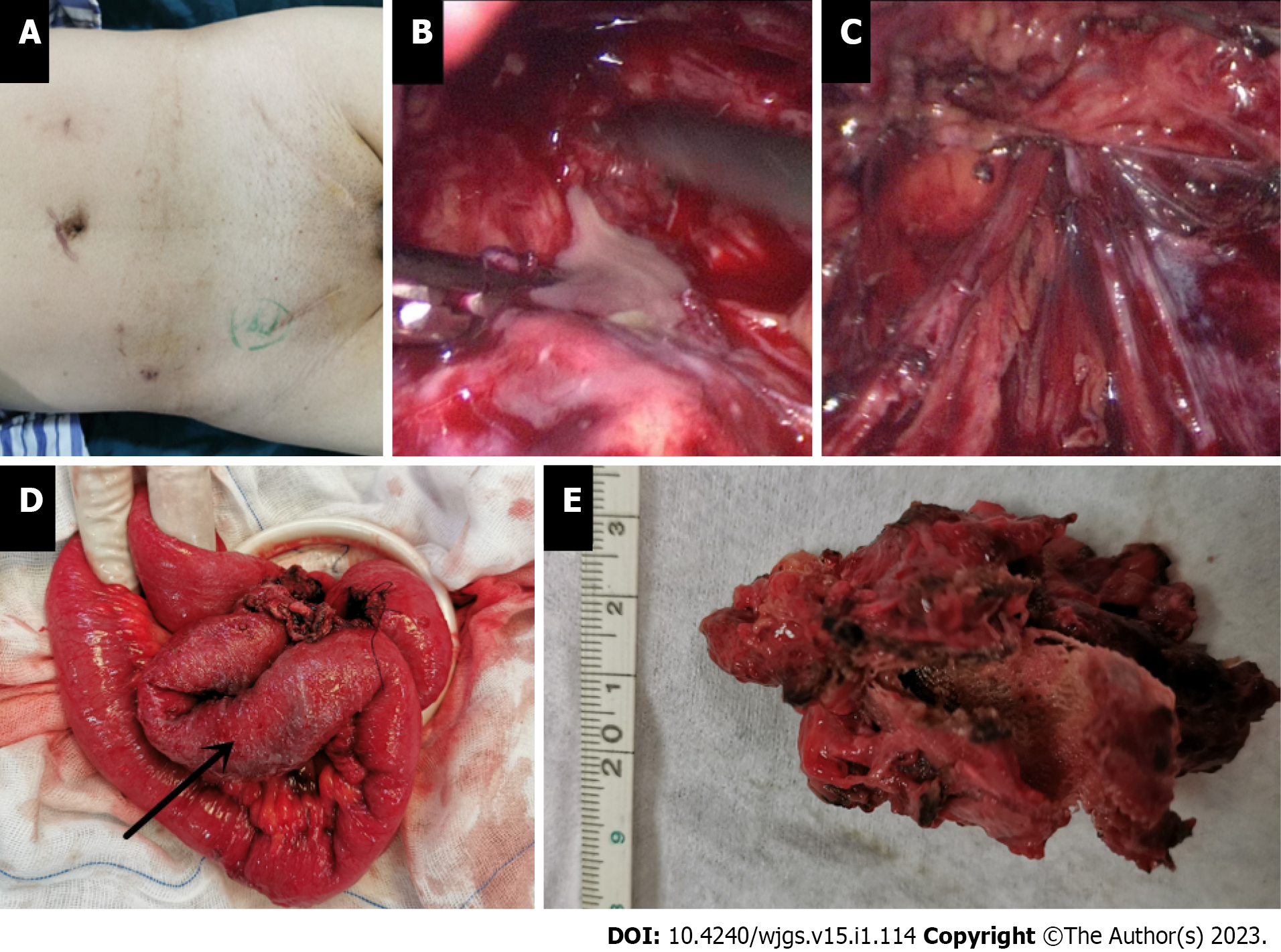
Comparing abdominal CT in 2018 and 2020, it was found that the right lower abdominal mass was not significantly enlarged, the center of the mass was not enhanced, and the surrounding fatty spaces were blurred. Combined with the patient’s diabetes found in the past year and poor control of blood sugar level, the right inguinal hernia was treated with mesh plug plain patch 5 years ago. This right lower abdominal mass was initially considered as meshoma with infection. After intravenous antibiotic treatment for about 10 d before operation, the patient’s body temperature, WBC and CRP were normal, and the blood sugar levels were well controlled after standardized hypoglycemic treatment (Figure 2), but the mass did not shrink. It was discussed and the decision was made to perform a laparoscopic exploration. Intraoperatively, a portion of the right lower abdominal ileal canal was found to be congested and oedematous, with dense adhesions forming to the abdominal wall (Figure 3). The central texture was firm, and sharp dissection with scissors revealed a mesh-like structure with outflow of pus (Figure 3B). The mass was completely separated from the abdominal wall, but not from the small bowel, so it was converted to open surgery. It was found that the mesh had eroded the ileal canal, so part of the ileum was resected along with the mass. Postoperative autopsy revealed a central mesh structure and a cavity in the mass (Figure 3E).
Gastrointestinal endoscopy 1 mo after surgery did not reveal abnormal lesions. There was no recurrence of the inguinal hernia during the 2-year follow-up.
The incidence of mesh infections ranges from 0.11% to 5.00%[7,8] and 0.7%-2.0% after laparoscopic inguinal hernia repair and up to 6%-10% in open-mesh techniques[9]. Risk factors for mesh infection include the patient’s underlying disease such as diabetes, coronary artery disease, chronic obstructive pulmonary disease, smoking, morbid obesity, malnutrition and immune deficiency[7,10]. But also, the type of mesh, the timing of the procedure, the surgical approach, whether the mesh was placed flat, the management of early postoperative complications such as haematoma or seroma requiring surgical management, the surgeon’s experience, and the use of improperly sterilized instruments, etc. The most common pathogens are Staphylococcus aureus and Staphylococcus epidermidis, in addition to several anaerobic bacteria of the genus Streptococcus and Enterobacteriaceae that play a key role in the pathogenesis of hernia repair mesh infections[11,12]. The bacterial culture of this patient yielded Streptococcus agalactiae (Group B Streptococcus, group B strep, GBS), which is a Gram-positive conditional pathogenic bacterium that is commensal in the human intestine and vagina. It causes severe and potentially fatal infections mainly in neonates and the elderly, while carriers have no obvious symptoms[13], the drug sensitivity of this patient was sensitive to penicillin, moxifloxacin, vancomycin and tegacyclin, except for tetracycline and clindamycin.
When the patch becomes infected, bacteria attach to the surface of the mesh and can form a microenvironment called “biofilm”[3,4,14]. Through its three-dimensional structure, the biofilm provides mechanical stability for bacteria and physical protection against external stressors (immune cells and therapeutic compounds, including antibiotics), rendering any conservative therapeutic measures ineffective[4]. As a result, this infection is usually persistent, with poor response to antibiotics and often requiring surgical treatment. It has been suggested that removal of partial mesh is associated with a 50% incidence of persistent prosthetic infection and a significant increase in the incidence of surgical site occurrence and reoperation. In the case of clean contaminated wounds and mesh-associated infection or fistula, the previous mesh should be completely removed whenever safe and feasible[15]. Open debridement can be very extensive and complex, and complete resection is often not achieved, especially when internal organs, especially hollow organs such as the bladder, colon or small bowel, are heavily attached to the abdominal wall or eroded by the mesh[16]. In such cases, the laparoscopic technique demonstrates its advantages. This technique allows a thorough exploration of the abdominal cavity, locating the focus of infection, determining whether there are adhesions and whether internal organs are involved and guiding further treatment, thus allowing unnecessary destruction of the healthy layers of the abdominal wall and nearby organs to be avoided.
Mesh infections can lead to catastrophic consequences, severely affecting the lives of patients and increasing the cost of healthcare to society[17]. This makes preventing patch infections far better than treating them. According to the literature conclusion, the following suggestions are made for the placement of the patch[18,19]. Firstly, choose the right type of mesh. Although patches are widely used in inguinal hernia repair, guidelines do not recommend the use of mesh plugs[1]. Mesh plugs are more likely to enter the abdominal cavity due to their conical shape and heavier weight, but are equally more likely to cause friction and even erosion of the organs. Among the various types of mesh, polypropylene is the preferred material because it is chemically inert, stable, non-immunogenic, non-toxic, flexible, and lightweight, has high tensile strength and is relatively resistant to infection. Secondly, attention needs to be paid to the details of the surgical procedure. It is necessary to keep the surgical field clean when placing the mesh, pay attention to the flatness of the mesh, avoid curling and folding, and avoid any direct contact with the viscera (vital), which can greatly reduce complications. When not fixed, inadequately fixed or inadequately dissected so that there is insufficient space for the mesh, this can lead to folding and curling of the mesh and eventually to the formation of a bulbous mass called a meshoma, a phenomenon first identified and named by Amid[20] in 2004. Thirdly, we must also not neglect the importance of surgical documentation. The size of the original hernia defect, the type of repair, the mesh material, the exact anatomical position of the mesh placement and the fixation technique must be accurately documented to provide as much information as possible for possible reoperation in the future. Finally, improving the patient’s own physical state. For example, quitting smoking, controlling diabetes and reducing the patient’s weight can greatly reduce the risk of wound complications[5].
The incidence of mesh infection is rare and, when it does occur, it has serious consequences for the patient and poses therapeutic difficulties for the surgeon, while treatment outcomes are often unsatisfactory. The formation of meshoma can not only cause mesh infection, but may even erode and require removal of the affected organ if left untreated. Therefore, after the formation of the meshoma, surgery to remove the meshoma as early as possible is recommended.
We thank the patient and all the rest doctors and nurses in our department.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Emergency medicine
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Dayan D, Israel; Ko J, South Korea; Musa Y, United States S-Editor: Chen YL L-Editor: A P-Editor: Chen YL
| 1. | HerniaSurge Group. International guidelines for groin hernia management. Hernia. 2018;22:1-165. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1355] [Cited by in RCA: 1258] [Article Influence: 179.7] [Reference Citation Analysis (1)] |
| 2. | Simons MP, Aufenacker T, Bay-Nielsen M, Bouillot JL, Campanelli G, Conze J, de Lange D, Fortelny R, Heikkinen T, Kingsnorth A, Kukleta J, Morales-Conde S, Nordin P, Schumpelick V, Smedberg S, Smietanski M, Weber G, Miserez M. European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia. 2009;13:343-403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 949] [Cited by in RCA: 898] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 3. | Wilson RB, Farooque Y. Risks and Prevention of Surgical Site Infection After Hernia Mesh Repair and the Predictive Utility of ACS-NSQIP. J Gastrointest Surg. 2022;26:950-964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 4. | Romanò CL, Romanò D, Morelli I, Drago L. The Concept of Biofilm-Related Implant Malfunction and "Low-Grade Infection". Adv Exp Med Biol. 2017;971:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 5. | Timmer AS, Claessen JJM, Brouwer de Koning IM, Haenen SM, Belt EJT, Bastiaansen AJNM, Verdaasdonk EGG, Wolffenbuttel CP, Schreurs WH, Draaisma WA, Boermeester MA. Clinical outcomes of open abdominal wall reconstruction with the use of a polypropylene reinforced tissue matrix: a multicenter retrospective study. Hernia. 2022;26:1241-1250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 6. | Jin C, Shen Y, Chen J. Laparoscopic evaluation and management of 47 patients with late-onset mesh infection after inguinal hernioplasty. Hernia. 2020;24:381-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Warren JA, Love M, Cobb WS, Beffa LR, Couto FJ, Hancock BH, Morrow D, Ewing JA, Carbonell AM. Factors affecting salvage rate of infected prosthetic mesh. Am J Surg. 2020;220:751-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 8. | Neumayer L, Giobbie-Hurder A, Jonasson O, Fitzgibbons R Jr, Dunlop D, Gibbs J, Reda D, Henderson W; Veterans Affairs Cooperative Studies Program 456 Investigators. Open mesh versus laparoscopic mesh repair of inguinal hernia. N Engl J Med. 2004;350:1819-1827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 731] [Cited by in RCA: 653] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 9. | LeBlanc KA, Whitaker JM, Bellanger DE, Rhynes VK. Laparoscopic incisional and ventral hernioplasty: lessons learned from 200 patients. Hernia. 2003;7:118-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 121] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 10. | Rosemar A, Angerås U, Rosengren A, Nordin P. Effect of body mass index on groin hernia surgery. Ann Surg. 2010;252:397-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 11. | Taylor SG, O'Dwyer PJ. Chronic groin sepsis following tension-free inguinal hernioplasty. Br J Surg. 1999;86:562-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 150] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 12. | Xu X, Zhan M, Li X, Chen T, Yang L. In vivo Analysis of the Resistance of the Meshes to Escherichia coli Infection. Front Surg. 2021;8:644227. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Sørensen UBS, Klaas IC, Boes J, Farre M. The distribution of clones of Streptococcus agalactiae (group B streptococci) among herdspersons and dairy cows demonstrates lack of host specificity for some lineages. Vet Microbiol. 2019;235:71-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 14. | Jacombs ASW, Karatassas A, Klosterhalfen B, Richter K, Patiniott P, Hensman C. Biofilms and effective porosity of hernia mesh: are they silent assassins? Hernia. 2020;24:197-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 15. | Kao AM, Arnold MR, Otero J, Huang LC, Prasad T, Lincourt AE, Augenstein VA. Comparison of Outcomes After Partial Versus Complete Mesh Excision. Ann Surg. 2020;272:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 16. | Li J. Total extraperitoneal (TEP) management of mesh erosion into bladder following transabdominal preperitoneal inguinal hernia repair (TAPP). Hernia. 2020;24:205-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 17. | Bueno-Lledó J, Torregrosa-Gallud A, Sala-Hernandez A, Carbonell-Tatay F, Pastor PG, Diana SB, Hernández JI. Predictors of mesh infection and explantation after abdominal wall hernia repair. Am J Surg. 2017;213:50-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 18. | Zhi Z, Cui H, Han W, Deng C, Li X. What is the outcome of late-onset infected mesh removal after open tension-free inguinal hernioplasty: 3-year follow-up. Hernia. 2022;. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 19. | Marmolejo A, Farell J, Ruiz Funes AP, Ayala S, Sánchez A, Navarro CA, Ramírez NA, García L, Daes J. Critical view of the myopectineal orifice: a scoring system to objectively evaluate transabdominal preperitoneal inguinal hernia repair. Surg Endosc. 2022;36:5094-5103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Amid PK. Radiologic images of meshoma: a new phenomenon causing chronic pain after prosthetic repair of abdominal wall hernias. Arch Surg. 2004;139:1297-1298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 3.8] [Reference Citation Analysis (0)] |