Published online Aug 27, 2021. doi: 10.4240/wjgs.v13.i8.806
Peer-review started: February 12, 2021
First decision: March 30, 2021
Revised: April 9, 2021
Accepted: July 7, 2021
Article in press: July 7, 2021
Published online: August 27, 2021
Processing time: 189 Days and 0.1 Hours
Acute mesenteric ischemia (AMI) is a rare cause of the “acute abdomen”, characterized by impaired blood flow to the intestine. The principle of treatment is restoration of perfusion to ischemic bowel and resection of any necrotic intestine. Surgery and endovascular intervention are two complementary approaches to mesenteric ischemia. Endovascular intervention is not an alternative to the surgical approach, but it has the potential to improve the prognosis of patients with AMI when judiciously combined with a surgical approach. Due to the need for emergent treatment of patients with acute mesenteric ischemia, the treatment strategy needs to be modified for each facility. This review aims to highlight cutting-edge studies and provide reasonable treatment strategies for patients with acute mesenteric ischemia based on available evidence.
Core Tip: The management of acute mesenteric ischemia (AMI) is challenging. An increased number of endovascular interventions were performed in past decades. Endovascular interventions have the potential to improve the prognosis of patients with AMI when combined with a surgical approach. Due to the need for emergent treatment of patients with acute mesenteric ischemia, the treatment strategy needs to be modified for each facility. We discuss the treatment strategy for patients with AMI based on cutting-edge evidence.
- Citation: Sakamoto T, Kubota T, Funakoshi H, Lefor AK. Multidisciplinary management of acute mesenteric ischemia: Surgery and endovascular intervention. World J Gastrointest Surg 2021; 13(8): 806-813
- URL: https://www.wjgnet.com/1948-9366/full/v13/i8/806.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v13.i8.806
Acute mesenteric ischemia (AMI) is a rare cause of the “acute abdomen”, characterized by impaired blood flow to the intestine. The main principle of treatment is restoration of blood flow to the ischemic intestine and resection of any necrotic segments. Early revascularization is important to reduce mortality[1]. Despite advances in technology and appropriate diagnosis and treatment, mortality remains high[2]. The management of patients with AMI is still challenging. Surgery and endovascular interventions are the two major approaches to treat patients with mesenteric ischemia. Surgical embolectomy, reported in 1951, is still a standard procedure for mesenteric embolism[3]. The first successful percutaneous angioplasty for mesenteric ischemia was reported in 1983[4]. Although decades have passed, the basic concepts of treatment have not changed. Endovascular interventions have been developed for a wide range of diseases including those in cardiology, oncology, and trauma, leading to the development of innovative devices and therapeutic approaches. As a result, an increased number of endovascular interventions were performed over the past decades[5]. However, there is no definite consensus regarding the complementary use of a surgical approach and endovascular intervention.
This review aims to highlight the cutting-edge studies highlighting the roles of surgery and endovascular intervention for the treatment of patients with AMI and develop a comprehensive treatment strategy based on available evidence.
AMI is commonly classified as: (1) mesenteric arterial embolization; (2) mesenteric arterial thrombosis; (3) non-occlusive mesenteric ischemia; or (4) mesenteric venous thrombosis. Other less common mechanisms include arterial dissection such as aortic dissection or superior mesenteric artery (SMA) dissection. In this review, we focus on mechanisms (1), (2), (3), and (4) above.
The prevalence of AMI has changed in recent decades. Mesenteric arterial embolization was the most common cause of mesenteric ischemia, which previously accounted for 40%-50%, and recently accounts for 25% of cases[2,6]. The most common sources of emboli include the heart and aortic atheromas. Mesenteric arterial thrombosis was the second most common cause of mesenteric ischemia, which previously accounted for 20%-35% and recently accounts for 40%[2,6]. Thrombotic occlusion occurs in a stenotic mesenteric artery when the flow in the artery decreases. Non-occlusive mesenteric ischemia previously accounted for 5% to 15% and recently accounts for 25% of cases[2,6], which results from generalized impaired perfusion of the mesenteric artery circulation. Although the mechanism is still unknown, heart failure, renal failure, cardiac surgery using cardiopulmonary bypass, and the use of catecholamine are reported as risk factors[7]. Lastly, mesenteric venous thrombosis is a unique type of mesenteric ischemia. The pathophysiology is different from the other types of mesenteric ischemia. Thrombosis in the mesenteric vein impairs blood outflow, resulting in bowel edema, impaired microperfusion, bowel dilation, and ischemia. In many cases, the underlying disease includes malignant neoplasms, inflammatory disease around the mesenteric vein, and coagulation disorders[6].
To establish the diagnosis, details of the clinical presentation such as sudden onset abdominal pain and abdominal pain out of proportion to physical findings are impor
The principles of treatment of mesenteric ischemia include: (1) restoration of perfusion; and (2) excision of necrotic intestine. The surgical approach is the conservative approach to arterial mesenteric ischemia, in which surgeons can restore the circulation to the mesentery and remove necrotic intestine if necessary, during one procedure. However, recent advancements in endovascular intervention for patients with mesenteric ischemia are noteworthy. To determine the optimal treatment strategy, it is critical to decide whether the patient is physiologically stable, which type of mesenteric ischemia the patient has, and whether the patient has peritonitis. In this review, we will discuss the treatment for mesenteric arterial embolism, mesenteric arterial thrombosis, and non-occlusive mesenteric ischemia because the nature and strategy of the treatment of patients with mesenteric venous thrombosis are quite different from other types of mesenteric ischemia.
The usefulness of endovascular intervention in patients with mesenteric ischemia needs to be evaluated for each type of mesenteric ischemia. Theoretically, restoring perfusion is the main goal of treatment. Endovascular intervention contributes to restoration of perfusion, only. In principle, endovascular therapy requires a patient without clinical signs of intestinal necrosis when used as the initial treatment.
Mesenteric arterial embolism and mesenteric arterial thrombosis: There are several options for endovascular intervention to treat patients with occlusive mesenteric ischemia (embolism and thrombosis). Removal of the thrombus or embolus and treatment of underlying stenosis are the primary goals of treatment. For removal of a thrombus or embolus, endovascular embolectomy and endovascular local thrombolysis can be selected. An endovascular embolectomy is an option for mesenteric arterial embolism, and is also useful for thrombosis. It is sometimes difficult to distinguish an embolism from thrombosis because multiple comorbidities are common in elderly patients. Local thrombolysis can be both an initial treatment, or a salvage option if endovascular embolectomy does not revascularize completely. In a retrospective single-center review, among 37 patients with mesenteric arterial embolisms, complete recanalization was achieved in 91.9% of patients with in-hospital mortality of 27.0%[11]. In a study of 50 patients with mesenteric embolism or thrombosis without peritonitis, local thrombolysis was used as the initial treatment and there was 88% technical success with a mortality of 33% including 6 self-limited bleeding episodes[12]. In this study, patients with peritonitis were not candidates for thrombolysis because, unlike embolectomy, thrombolysis can result in bleeding complications. Patients with peritonitis require laparotomy and intestinal resection. The decision to undertake thrombolysis should be made after a multidisciplinary discussion.
After removal of the thrombus or embolus, treatment of the underlying stenosis with angioplasty or/and stenting may be performed during the same procedure, especially in patients with thrombosis. In general, an underlying stenosis can be treated later because this is not necessarily a life-saving procedure.
Non-occlusive mesenteric ischemia: Unlike mesenteric arterial embolism or thrombosis, the major principle of treatment of non-occlusive mesenteric ischemia is management of the underlying disease which led to mesenteric ischemia. A local bolus and continuous infusion of vasodilators such as papaverine and prostaglandin E1 may decrease mortality and the need for bowel resection. In a nationwide observational study, early vasodilator therapy (within 2 d of admission) including papaverine and/or prostaglandin E1 decreased mortality and the need for surgical intervention by 11.6% (34.2% vs 22.6%) and 10.2% (15.3% vs 5.0%), respectively[13]. In patients with non-occlusive mesenteric ischemia, stenotic changes at the origin of the SMA can usually be found. Although stenotic changes are associated with high mortality, no data suggest that intervention at the site of stenosis improves mortality[14]. It is reasonable to perform endovascular angioplasty or stenting in patients whose mesenteric ischemia may be partially caused by mesenteric arterial thrombosis. Routine intervention at the stenotic arterial origin is not suggested.
Surgery is the traditional treatment modality for patients with mesenteric ischemia except for those with non-occlusive mesenteric ischemia. The surgical approach includes procedures for (1) restoring perfusion; and (2) excision of any necrotic intestine. The priority of treatment depends on the patients’ physiologic status. In a patient in shock, removal of the necrotic intestine is the priority because sepsis due to bowel necrosis is strongly suspected. In patients without suspected intestinal necrosis, restoration of perfusion is the priority to avoid intestinal necrosis. With or without bowel resection, a second look laparotomy should be considered to check intestinal viability 24 to 48 h after the first operation.
Mesenteric arterial embolism and mesenteric arterial thrombosis: Surgery as the treatment modality for patients with mesenteric arterial embolism and thrombosis has three objectives including (1) removal of the thrombus or embolus; (2) treatment of the underlying stenosis with angioplasty or bypass; and (3) resection of necrotic intestine. Open embolectomy is still considered to be a conservative procedure for mesenteric arterial embolism[3,15]. In patients with mesenteric arterial embolism alone, open embolectomy can restore perfusion completely without additional procedures such as intestinal resection, angioplasty, or vascular bypass. In patients with mesenteric arterial thrombosis, vascular reconstruction is desirable. There are several procedures for vascular reconstruction with (infrarenal aorta-mesenteric bypass) or without a graft (iliac-ileocolic artery bypass)[16]. There is a report that antegrade bypass is preferred to retrograde bypass due to a lower re-intervention rate[17]. Although theoretically, autografts such as saphenous vein are preferred to synthetic grafts in terms of infection risk, autografts may be more time-consuming to construct than synthetic grafts.
Non-occlusive mesenteric ischemia: The major purpose of surgery in patients with non-occlusive mesenteric ischemia is resection of any necrotic intestine. Patients who are in shock or have peritonitis are candidates for immediate surgical exploration to assess intestinal viability. Diagnostic laparoscopy is reported to be a safe and reliable diagnostic tool in patients with suspected acute mesenteric ischemia[18-20]. A second look laparotomy should be considered 24 to 48 h after the first operation as in patients with mesenteric ischemia of other etiologies.
There are several observational studies and meta-analyses comparing the outcomes of endovascular interventions and surgery. In a single-center study from Spain including 323 patients, the characteristics of patients and their treatment have changed over recent decades. Patients have increased morbidity and the prevalence of arterial embolism has decreased[2]. In light of these changes in the demographics of the patients, care should be taken to interpret the results of studies.
In a nationwide study, using the Swedish Vascular Registry including 42 endovascular and 121 open revascularizations of the SMA from 1999 to 2006, short and long-term mortality was lower in patients who underwent endovascular revascularization; 30 d and 1 year mortality were 28% vs 42% and 39% vs 58%, respectively[21]. One of the largest studies using the National Inpatient Sample database including 5237 patients (35.5% endovascular and 64.5% open revascularization) from 2000 through 2006 revealed that endovascular intervention is associated with lower mortality compared with open surgery (15.6% vs 38.6%). The latest study using the same database included 4665 patients who underwent interventional treatment (24% endovascular and 76% open revascularization) from 2005 through 2009 also showed that endovascular intervention is associated with lower mortality compared with open surgery (24.9% vs 39.3%)[5]. A study using the American College of Surgeons National Surgical Quality Improvement Program database in the United States including 439 patients (6.2% endovascular, 5.2% hybrid, and 88.6% open revascularization) from 2005 to 2010 showed that endovascular intervention was associated with decreased mortality [odds ratio (OR) 0.4, 95%CI 0.2-0.9][22]. A meta-analysis including five non-randomized studies showed that endovascular intervention had a lower prevalence of bowel resection (OR 0.37, 95%CI 0.23-0.59) and 30-d mortality (OR 0.50, 95%CI 0.30-0.83) compared with open surgery[8]. Another meta-analysis including nineteen observational studies also showed that endovascular intervention was associated with a lower prevalence of bowel resection (OR 0.45, 95%CI 0.34-0.59) and 30-d mortality (OR 0.45, 95%CI 0.34-0.59) compared with open surgery[23].
Overall, endovascular interventions were reported to be superior to open surgery. These results are based on retrospective observational studies, although some of them conducted multivariable regression analysis. It is likely that there is selection bias between the populations in using these two approaches. Even with meta-analyses, there is large heterogeneity among the studies[8]. Due to the rarity and underlying nature of the disease, it will be nearly impossible to conduct a randomized controlled trial to compare the two approaches. We need to evaluate the effect of endovascular intervention based on existing studies.
Recently, retrograde open mesenteric stenting (ROMS) has been described[24,25]. This procedure includes a laparotomy and retrograde endovascular revascularization of the superior mesenteric artery[26]. Candidates for this procedure are patients with stenosis at the origin of the SMA. As previously mentioned, vascular reconstruction is desirable for this group of patients but time-consuming to conduct in an emergency setting. One of the advantages of this method over vascular bypass is significantly shorter operative time. A recent study showed that bypass procedures took a longer time than ROMS (302 vs 189 min, P < 0.01)[25]. One of the major concerns after stenting is patency, however, patency rates similar to those after bypass were reported, 76% to 88%[25,27-29].
This approach is a good option for patients requiring laparotomy to both resect necrotic intestine and reconstruct vascular inflow (mesenteric arterial thrombosis).
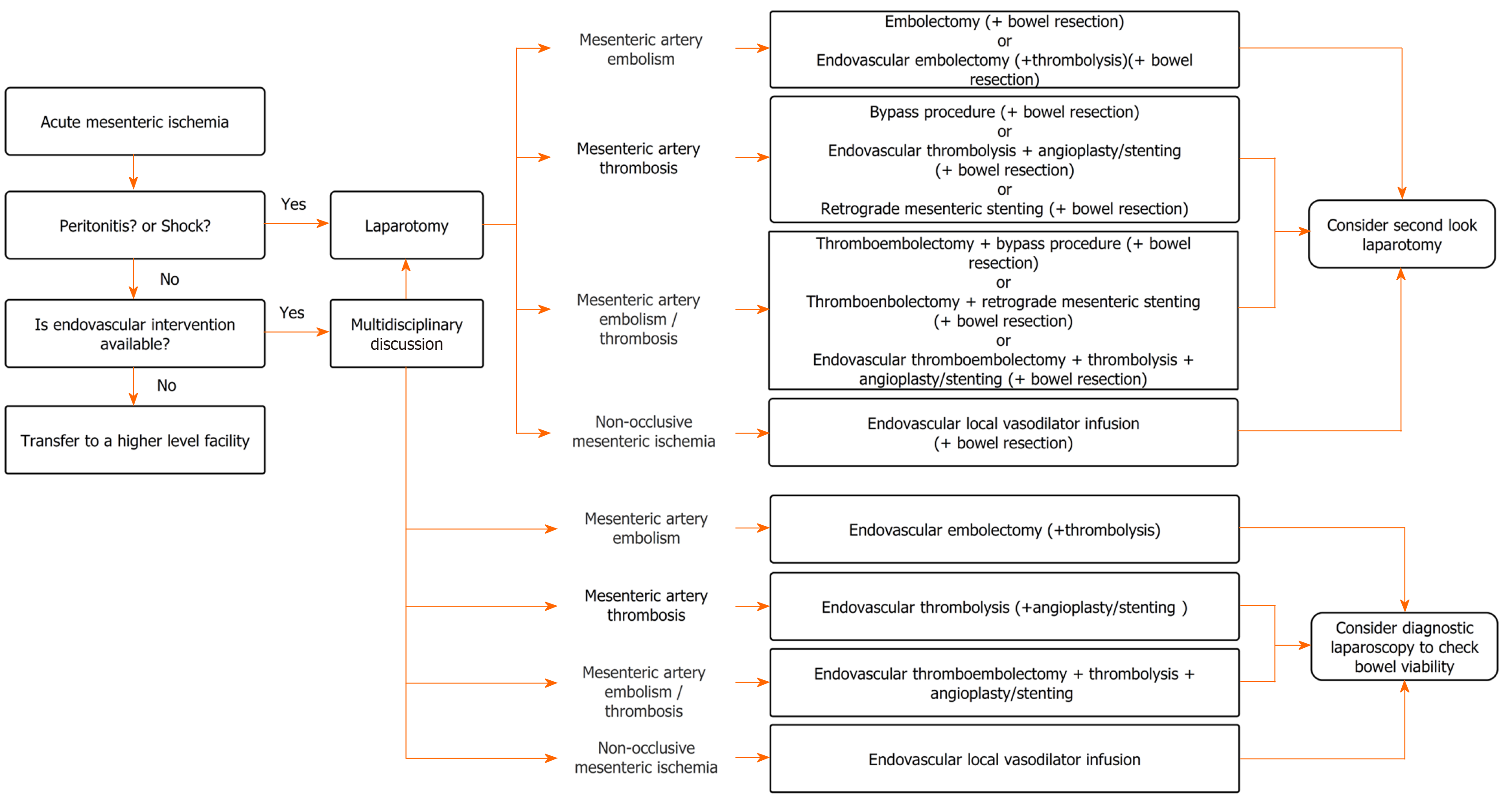
Endovascular intervention is not an alternative to surgery, but has the potential to improve the prognosis of patients with AMI when combined with a surgical approach as a complementary procedure. Since acute mesenteric ischemia requires emergent treatment, the strategy needs to be modified to fit each facility (Figure 1).
Although evidence for the effect of endovascular interventions is limited, there are apparent advantages to endovascular intervention over open surgery in some patients. Recent advancements in endovascular intervention added treatment options such as ROMS. There is an established role for endovascular intervention in the treatment of patients with mesenteric ischemia.
In patients with shock or peritonitis, laparotomy is the primary option due to the high degree of association of these conditions with intestinal necrosis. A subsequent procedure may be needed depending on the etiology. Even after laparotomy, an endovascular intervention can be selected if a hybrid operating room is available. In patients without shock or peritonitis, endovascular intervention is often the preferred option. If the endovascular intervention is unavailable at the facility, transferring the patient to a higher-level facility with these capabilities should be considered. Early revascularization with endovascular treatment followed by close observation or planned laparoscopic exploration is a reasonable strategy.
The major problem when managing patients with mesenteric ischemia is that multi-disciplinary interventions are required. Corcos et al[30] suggested that multidisciplinary treatment has the potential to improve survival of patients with acute mesenteric ischemia. To provide appropriate treatment, vascular surgeons, general surgeons, and interventional radiologists must collaborate. Daily simulation and creating protocols are crucial to successful treatment, and to facilitate the combination of surgery and endovascular interventions, developing a hybrid operating room is important. To rescue non-survivors, factors determining the primary treatment need to be identified in future studies. In addition to endovascular intervention and surgery, perioperative treatment such as oral antibiotics may have the potential to improve survival[31].
In this review, we constructed a decision-making flow chart synthesized from current evidence-based recommendations in recent studies which discuss the appropriate use of interventional radiology and surgery for patients with mesenteric ischemia. Although interventional radiology and surgery for mesenteric ischemia were compared in many studies, these two approaches should be treated as complementary.
The management of patients with acute mesenteric ischemia is a challenge for the vascular surgeon, general surgeon, and interventional radiologist. Although there is not enough data to support a specific approach, an individualized strategy combining surgery and endovascular intervention based on the capabilities at each facility will improve patient outcomes. Given the current technology available, this is not the time to compare surgery and endovascular interventions, but to establish the best treatment strategy for each individual patient considering these as complementary approaches.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: The Japanese Society of Gastroenterology.
Specialty type: Surgery
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Wu W S-Editor: Zhang H L-Editor: Webster JR P-Editor: Zhang YL
| 1. | Plumereau F, Mucci S, Le Naoures P, Finel JB, Hamy A. Acute mesenteric ischemia of arterial origin: importance of early revascularization. J Visc Surg. 2015;152:17-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Acosta-Mérida MA, Marchena-Gómez J, Saavedra-Santana P, Silvestre-Rodríguez J, Artiles-Armas M, Callejón-Cara MM. Surgical Outcomes in Acute Mesenteric Ischemia: Has Anything Changed Over the Years? World J Surg. 2020;44:100-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 3. | KLASS AA. Embolectomy in acute mesenteric occlusion. Ann Surg. 1951;134:913-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 57] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 4. | Roberts L Jr, Wertman DA Jr, Mills SR, Moore AV Jr, Heaston DK. Transluminal angioplasty of the superior mesenteric artery: an alternative to surgical revascularization. AJR Am J Roentgenol. 1983;141:1039-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 5. | Beaulieu RJ, Arnaoutakis KD, Abularrage CJ, Efron DT, Schneider E, Black JH 3rd. Comparison of open and endovascular treatment of acute mesenteric ischemia. J Vasc Surg. 2014;59:159-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 6. | Clair DG, Beach JM. Mesenteric Ischemia. N Engl J Med. 2016;374:959-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 312] [Cited by in RCA: 329] [Article Influence: 36.6] [Reference Citation Analysis (1)] |
| 7. | Sakamoto T, Fujiogi M, Matsui H, Fushimi K, Yasunaga H. Clinical features and outcomes of nonocclusive mesenteric ischemia after cardiac surgery: a retrospective cohort study. Heart Vessels. 2020;35:630-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 8. | Björck M, Koelemay M, Acosta S, Bastos Goncalves F, Kölbel T, Kolkman JJ, Lees T, Lefevre JH, Menyhei G, Oderich G; Esvs Guidelines Committee; Kolh P, de Borst GJ, Chakfe N, Debus S, Hinchliffe R, Kakkos S, Koncar I, Sanddal Lindholt J, Vega de Ceniga M, Vermassen F, Verzini F, Document Reviewers, Geelkerken B, Gloviczki P, Huber T, Naylor R. Editor's Choice - Management of the Diseases of Mesenteric Arteries and Veins: Clinical Practice Guidelines of the European Society of Vascular Surgery (ESVS). Eur J Vasc Endovasc Surg. 2017;53:460-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 431] [Article Influence: 61.6] [Reference Citation Analysis (1)] |
| 9. | Expert Panel on Interventional Radiology, Fidelman N, AbuRahma AF, Cash BD, Kapoor BS, Knuttinen MG, Minocha J, Rochon PJ, Shaw CM, Ray CE Jr, Lorenz JM. ACR Appropriateness Criteria® Radiologic Management of Mesenteric Ischemia. J Am Coll Radiol. 2017;14:S266-S271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Bala M, Kashuk J, Moore EE, Kluger Y, Biffl W, Gomes CA, Ben-Ishay O, Rubinstein C, Balogh ZJ, Civil I, Coccolini F, Leppaniemi A, Peitzman A, Ansaloni L, Sugrue M, Sartelli M, Di Saverio S, Fraga GP, Catena F. Acute mesenteric ischemia: guidelines of the World Society of Emergency Surgery. World J Emerg Surg. 2017;12:38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 307] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 11. | Raupach J, Lojik M, Chovanec V, Renc O, Strýček M, Dvořák P, Hoffmann P, Guňka I, Ferko A, Ryška P, Omran N, Krajina A, Čabelková P, Čermáková E, Malý R. Endovascular Management of Acute Embolic Occlusion of the Superior Mesenteric Artery: A 12-Year Single-Centre Experience. Cardiovasc Intervent Radiol. 2016;39:195-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 12. | Kärkkäinen JM, Lehtimäki TT, Saari P, Hartikainen J, Rantanen T, Paajanen H, Manninen H. Endovascular Therapy as a Primary Revascularization Modality in Acute Mesenteric Ischemia. Cardiovasc Intervent Radiol. 2015;38:1119-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 13. | Takiguchi T, Nakajima M, Ohbe H, Sasabuchi Y, Matsui H, Fushimi K, Kim S, Yokota H, Yasunaga H. Vasodilator Therapy and Mortality in Nonocclusive Mesenteric Ischemia: A Nationwide Observational Study. Crit Care Med. 2020;48:e356-e361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 14. | Minko P, Stroeder J, Groesdonk HV, Graeber S, Klingele M, Buecker A, Schäfers HJ, Katoh M. A scoring-system for angiographic findings in nonocclusive mesenteric ischemia (NOMI): correlation with clinical risk factors and its predictive value. Cardiovasc Intervent Radiol. 2014;37:657-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 15. | Yun WS, Lee KK, Cho J, Kim HK, Huh S. Treatment outcome in patients with acute superior mesenteric artery embolism. Ann Vasc Surg. 2013;27:613-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Swerdlow NJ, Varkevisser RRB, Soden PA, Zettervall SL, McCallum JC, Li C, Wyers MC, Schermerhorn ML. Thirty-Day Outcomes After Open Revascularization for Acute Mesenteric Ischemia From the American College of Surgeons National Surgical Quality Improvement Program. Ann Vasc Surg. 2019;61:148-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 17. | Scali ST, Ayo D, Giles KA, Gray S, Kubilis P, Back M, Fatima J, Arnaoutakis D, Berceli SA, Beck AW, Upchurch GJ, Feezor RJ, Huber TS. Outcomes of antegrade and retrograde open mesenteric bypass for acute mesenteric ischemia. J Vasc Surg. 2019;69:129-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 18. | Chagla L, Kiff R. 'Second look' laparoscopy in the management of acute mesenteric ischemia. Br J Surg. 1994;81:1083. [PubMed] |
| 19. | Zamir G, Reissman P. Diagnostic laparoscopy in mesenteric ischemia. Surg Endosc. 1998;12:390-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 20. | Gonenc M, Dural CA, Kocatas A, Buyukasik S, Karabulut M, Alis H. The impact of early diagnostic laparoscopy on the prognosis of patients with suspected acute mesenteric ischemia. Eur J Trauma Emerg Surg. 2013;39:185-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 21. | Block TA, Acosta S, Björck M. Endovascular and open surgery for acute occlusion of the superior mesenteric artery. J Vasc Surg. 2010;52:959-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 93] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 22. | Branco BC, Montero-Baker MF, Aziz H, Taylor Z, Mills JL. Endovascular Therapy for Acute Mesenteric Ischemia: an NSQIP Analysis. Am Surg. 2015;81:1170-1176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 23. | El Farargy M, Abdel Hadi A, Abou Eisha M, Bashaeb K, Antoniou GA. Systematic review and meta-analysis of endovascular treatment for acute mesenteric ischaemia. Vascular. 2017;25:430-438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Pisimisis GT, Oderich GS. Technique of hybrid retrograde superior mesenteric artery stent placement for acute-on-chronic mesenteric ischemia. Ann Vasc Surg 2011; 25: 132.e7-132. 11;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 25. | Andraska E, Haga L, Li X, Avgerinos E, Singh M, Chaer R, Madigan M, Eslami MH. Retrograde open mesenteric stenting should be considered as the initial approach to acute mesenteric ischemia. J Vasc Surg. 2020;72:1260-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 26. | Rebelo A, Partsakhashvili J, John E, Kleeff J, Ukkat J. Current concepts and evidence on open, endovascular and hybrid treatment of mesenteric ischemia: The retrograde open mesenteric stenting. Surg Pract Sci. 2020;. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 27. | Oderich GS, Macedo R, Stone DH, Woo EY, Panneton JM, Resch T, Dias NV, Sonesson B, Schermerhorn ML, Lee JT, Kalra M, DeMartino RR, Sandri GA, Ramos Tenorio EJ; Low Frequency Vascular Disease Research Consortium Investigators. Multicenter study of retrograde open mesenteric artery stenting through laparotomy for treatment of acute and chronic mesenteric ischemia. J Vasc Surg. 2018;68:470-480.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 28. | Blauw JT, Meerwaldt R, Brusse-Keizer M, Kolkman JJ, Gerrits D, Geelkerken RH; Multidisciplinary Study Group of Mesenteric Ischemia. Retrograde open mesenteric stenting for acute mesenteric ischemia. J Vasc Surg. 2014;60:726-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 29. | Roussel A, Castier Y, Nuzzo A, Pellenc Q, Sibert A, Panis Y, Bouhnik Y, Corcos O. Revascularization of acute mesenteric ischemia after creation of a dedicated multidisciplinary center. J Vasc Surg. 2015;62:1251-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 30. | Corcos O, Castier Y, Sibert A, Gaujoux S, Ronot M, Joly F, Paugam C, Bretagnol F, Abdel-Rehim M, Francis F, Bondjemah V, Ferron M, Zappa M, Amiot A, Stefanescu C, Leseche G, Marmuse JP, Belghiti J, Ruszniewski P, Vilgrain V, Panis Y, Mantz J, Bouhnik Y. Effects of a multimodal management strategy for acute mesenteric ischemia on survival and intestinal failure. Clin Gastroenterol Hepatol 2013; 11: 158-65. e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 93] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 31. | Nuzzo A, Maggiori L, Paugam-Burtz C, Cazals-Hatem D, Ronot M, Huguet A, Becq A, Castier Y, Weiss E, Plessier A, Tran-Dinh A, Joly F, Panis Y, Bouhnik Y, Gault N, Corcos O. Oral Antibiotics Reduce Intestinal Necrosis in Acute Mesenteric Ischemia: A Prospective Cohort Study. Am J Gastroenterol. 2019;114:348-351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 25] [Article Influence: 4.2] [Reference Citation Analysis (0)] |