Published online Dec 15, 2015. doi: 10.4251/wjgo.v7.i12.445
Peer-review started: May 7, 2015
First decision: June 6, 2015
Revised: June 20, 2015
Accepted: September 30, 2015
Article in press: October 9, 2015
Published online: December 15, 2015
Processing time: 229 Days and 20 Hours
Cytoreductive surgery combined with intraperitoneal chemotherapy can improve survival in appropriately selected patients with colorectal peritoneal metastases. Outcomes are best in those patients in whom a complete cytoreduction can be achieved. Unresectable disease is however encountered in approximately one-quarter of patients at laparotomy. The merits, or otherwise, of proceeding with an incomplete cytoreduction in this setting are unclear. We performed a review of published outcomes following incomplete cytoreduction for colorectal peritoneal metastases. Using the electronic databases, PubMed and MEDLINE, a systematic search of available literature published during the period January 1997 to September 2014 was conducted. Following application of exclusion criteria, 19 papers were identified and included in this review. These comprised fifteen case series, 3 case control studies and one randomised control trial. In the nineteen studies included in this review, 2790 patients underwent cytoreductive surgery with or without intraperitoneal chemotherapy for peritoneal metastases of colorectal origin. Of these, 1732 (62%) underwent a complete cytoreduction while 986 (35%) patients underwent an incomplete cytoreduction. Median survival in the complete cytoreduction group ranged from 11 to 62 mo while survival in the latter group ranged from 2.4 to 32 mo. Of the 986 patients with an incomplete cytoreduction, 331 patients received intraperitoneal chemotherapy and survival in this cohort ranged from 4.5 to 32 mo. An incomplete cytoreduction, with or without intraperitoneal chemotherapy, does not appear to confer a survival benefit. The limited available data points to a palliative benefit in a subset of patients. In the absence of high quality data, the decision as to whether or not to proceed with surgery should be made on an individual patient basis.
Core tip: Cytoreductive surgery combined with intraperitoneal chemotherapy for colorectal peritoneal metastases improves survival in appropriately selected patients following complete cytoreduction. The merits of an incomplete cytoreduction, with or without intraperitoneal chemotherapy, are unclear. The available evidence is heterogenous and of poor quality. The current review has not shown a benefit to surgery in the setting of unresectable disease. Certain patients, particularly those with ascites may however gain from a quality of life point of view.
- Citation: Heaney RM, Shields C, Mulsow J. Outcome following incomplete surgical cytoreduction combined with intraperitoneal chemotherapy for colorectal peritoneal metastases. World J Gastrointest Oncol 2015; 7(12): 445-454
- URL: https://www.wjgnet.com/1948-5204/full/v7/i12/445.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v7.i12.445
Peritoneal metastases (PM) are found in approximately 10% of individuals undergoing resection for colorectal cancer[1] and ultimately occur in up to 35% patients[2-4]. After hepatic metastases, the most common site of cancer recurrence after curative primary resection is the peritoneum[5,6]. Peritoneal metastases have traditionally been associated with a poor prognosis, with patients frequently referred for palliative care. In this setting, median survival in the order of five to seven months was typical[1,7,8]. Recent advances have allowed the introduction of new, more targeted approaches combining systemic chemotherapy with biological agents such as bevacizumab. However, best survival rates achieved with these combinations rarely exceed twenty months[9-11].
Over the past 20 years, a number of studies have shown a survival benefit following combined cytoreductive surgery (CRS) and intraperitoneal chemotherapy for patients with colorectal peritoneal metastases. The primary aim of cytoreductive surgery is to eliminate all macroscopic disease through peritonectomy procedures, and multi-visceral resections if necessary[12,13]. Cytoreductive surgery is combined with intraperitoneal chemotherapy with the aim of irradiating residual microscopic disease[14,15]. A 2003 randomized control trial by Verwaal et al[16], showed that patients treated with cytoreduction and heated intraperitoneal chemotherapy (HIPEC) had a median survival of 22.4 mo compared with 12.6 mo for those assigned to systemic chemotherapy alone. Favourable outcomes using this approach have since been demonstrated in a number of case series, including a 2010 multicentre study of 523 patients in which patients undergoing cytoreduction and HIPEC had a median survival of 30.1 mo[17]. The results of a recent meta-analysis by Mirnezami et al[18] further supported these encouraging outcomes, with patients undergoing CRS and HIPEC having superior two and five year survival rates when compared to those receiving systemic chemotherapy alone.
In patients undergoing surgery for colorectal peritoneal metastases, a correlation between the completeness of cytoreduction and survival has been shown in a number of studies and confirmed in a recent meta-analysis[18]. Verwaal et al[19] found that patients with a complete cytoreduction had a median survival of fifty-two months compared with an eight month median survival in patients with an incomplete cytoreduction. A complete cytoreduction is more likely to be possible and beneficial in the absence of biliary, ureteric, or multilevel bowel obstruction and in patients with lower volume disease [peritoneal carcinomatosis index (PCI) less than twenty][20].
Unfortunately, the pre-operative prediction of those patients in whom a complete cytoreduction will be achievable is difficult. It is well accepted that CT scanning and conventional imaging techniques have a poor sensitivity for identifying peritoneal metastases[21,22]. This can lead to underdiagnosis and understaging, with the result that unresectable disease is first discovered at laparotomy. In an attempt to overcome these limitations, many centres now utilise staging laparoscopy to pre-operatively assess operability and calculate the PCI. A 2012 cohort study by Iversen et al[23] found that while pre-operative laparoscopy reduced the rates of open and closed laparotomy, it understated peritoneal tumour in 56% of patients. Furthermore, in patients with metachronous disease, post-operative adhesions may reduce the reliability of the approach in accurately determining the extent and site of recurrent tumour[23]. Ultimately, the tumour burden, PCI, and resectability can only be reliably calculated at laparotomy[24].
While it is evident that a complete cytoreduction combined with intraperitoneal chemotherapy confers a survival benefit, it is not clear what impact, if any, an incomplete cytoreduction has on overall survival and quality of life. In this paper we aim to review the current literature to address the question of whether surgery should be abandoned if a complete cytoreduction cannot be achieved or, would the patient benefit in terms of symptomatic relief or prolongation of life, from an incomplete cytoreduction combined with intraperitoneal chemotherapy.
A systematic search of available literature using the PubMed and MEDLINE databases provided by the National Library of Medicine was conducted. Search terms included the following keywords, or combinations thereof: “colorectal”, “peritoneal”, “metastases”, “cytoreductive surgery”, “intraperitoneal”, “HIPEC” and “chemotherapy”. The “related citations” function was utilised to broaden the search. Additional relevant publications were obtained by reviewing the reference sections of all selected articles.
Only papers published in the English language during the period January 1997 to September 2014 were included. Only original research articles that studied peritoneal metastases of colorectal origin were included. Papers that omitted the breakdown of survival data according to the primary cancer and a completeness of cytoreduction score (CC-score) were excluded. Studies reporting iterative cytoreductive procedures were also eliminated.
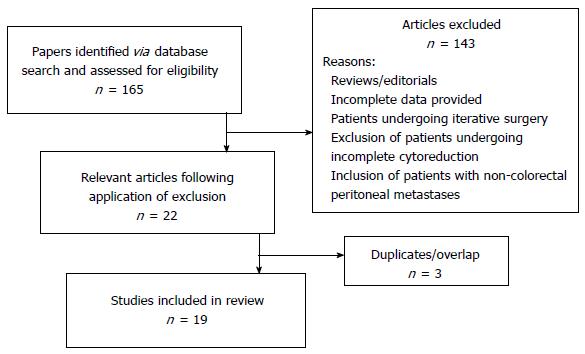
The full text articles of 165 publications were obtained and their relevance assessed. 143 papers were excluded based on the aforementioned criteria. A total of 22 eligible publications were identified. Further review of these papers identified likely overlap between study centres and patient cohorts. Best efforts were made to eliminate any duplication however this was not possible with regard to one paper, in which one-quarter of patients were included in a separate retrospective multicentre study[6,17]. Following this further analysis, 19 papers were ultimately deemed appropriate for inclusion in the current study. This comprised 15 case series[6,17,25-37], 3 case control studies[38-40] and one randomised control trial (RCT)[16]. Figure 1 summarises the selection process.
Eleven of the nineteen studies utilised the completeness of cytoreduction score (CC-Score) as described by Jacquet et al[41] in which a CC-0 score indicates that no macroscopic peritoneal tumour remains after cytoreduction, a CC-1 score indicates that persisting tumour nodules are < 2.5 mm, a CC-2 score indicates residual tumour nodules between 2.5 mm and 2.5 cm and finally a CC-3 score indicates tumour nodules > 2.5 cm or a confluence of unresected tumour. The remaining eight studies used four different scoring systems, which for the purpose of this review, were analysed and assigned the most appropriate CC-Score based on the size of the remaining tumour nodules[6,29,30,33,35-37,40]. Three of the papers utilised the R-score, where R0 indicates complete cytoreduction, R1 indicates the persistence of microscopic disease, and R2a, R2b and R2c indicate residual tumour nodules measuring < 5 mm, 5 mm-2 cm and > 2 cm respectively[16,29,37]. Five studies failed to identify specific tumour measurements. Two of these classified resections as: no evidence of macroscopic disease, persisting microscopic disease and persisting macroscopic disease and for comparison purposes, these were assigned scores of CC-0, CC-1 and CC-2/3 respectively[30,33]. The other three studies used the categories; macroscopically complete or macroscopically incomplete, and were assigned appropriate CC scores[35,36,40]. Complete cytoreduction refers to a CC-0 or 1 score whereas a CC-2 or 3 score is classified as incomplete.
In the nineteen studies analysed, a total of 2790 patients underwent cytoreductive surgery and intraperitoneal chemotherapy for peritoneal metastases of colorectal origin during the period 1997 to 2014. Median age provided in seventeen of the studies ranged from 47-67 years. Of the 2790 patients, a CC-0/1 cytoreduction was achieved in 1732 (62%) while a CC-2/3 resection was achieved in 986 (35%). The 986 patients who had final resection scores of CC-2/3 form the basis of our review. The remaining 72 patients are not included in our analysis as a result of unknown or unassigned CC-scores and patients who were lost to follow up or not included in the original analyses.
Five studies included patients with synchronous peritoneal metastases only[27,30,35-37]. The study by Pestieau et al[26] included patients undergoing resection for both synchronous and metachronous disease, with all but one of the patients in the incomplete cytoreduction group having metachronous peritoneal metastases. The remaining studies included patients with both synchronous and metachronous disease.
No data was provided regarding the delivery of neo-adjuvant therapy in eighteen of the included studies. The remaining study only included patients who had not received systemic treatment prior to surgery[31].
Twelve studies included patients with intra-abdominal disease only[6,16,17,25,26,28,29,32,37-40] and five of these included patients with peritoneal metastases alone[16,28,32,38,40]. In the seven studies that included patients with non-peritoneal intra-abdominal metastases (hepatic metastases), only one paper included the resection of hepatic metastases in the overall CC-score[29]. In three of these seven studies[6,17,37], the CC-score referred to resection of peritoneal disease only and the remaining three studies did not specify whether the resection of hepatic metastases affected the final CC-score[25,26,39]. Cavaliere et al[25] found that the presence of extensive disease at the porta hepatis increased the likelihood of an incomplete cytoreduction. Four of the other studies included patients with extra-abdominal distant metastases (lung and supraclavicular nodes[27], lung only[31], and the other two studies did not specify the site of distant disease[30,36]). The remaining three studies did not specify whether patients also had non peritoneal distant metastases[33-35].
Only two of the studies provided information regarding the actual PCI, or equivalent, in patients in whom a complete cytoreduction was not possible[26,34]. The 55 patients in the study by Pestieau et al[26] in whom a complete cytoreduction was not possible, had a median PCI of 20.7 ± 7.6 compared to a PCI of 15.4 ± 7.6 in patients in whom a CC-0/1 resection was possible.
Chua et al[34] reported a series of three patients with colorectal peritoneal metastases who underwent incomplete cytoreduction. Two of the patients had a PCI of 11 while the third had a score of 39.The reasons for failure to clear all macroscopic disease in this study were extensive small bowel involvement, liver metastases, and extensive involvement of the lesser sac and diaphragm[34]. Similarly, Winer et al[32] found that extensive small bowel involvement or small bowel mesenteric deposits resulted in an aborted or incomplete resection in their 4 patients. Finally, Park et al[31] identified the presence of metastases covering a substantial amount of the peritoneal surface as the reason for failure of cytoreduction in the 5 patients in the incomplete cytoreduction group.
One study specifically reported outcomes in patients with signet ring histology only[32]. Winer et al[32] found that tumours with signet ring histology responded poorly to CRS and HIPEC and their five CC-2/3 patients had a median survival of 2.4 mo. In the remaining studies no correlation between completeness of cytoreduction and tumour histological subtype or differentiation was reported.
In the nineteen studies analysed, 986 patients underwent a CC-2/3 or incomplete resection. No study reported the specific resections that were undertaken in the incomplete cytoreduction group. Two studies reported that in the presence of distant metastases or extensive disease not amenable to a complete cytoreduction, a radical resection of peritoneal disease was not pursued and palliative surgery was performed[30,31]. This, in one study involved the removal of gross tumour deposits or disease that was likely to cause gastrointestinal obstruction, without the administration of intraperitoneal chemotherapy[30] and in the other entailed omentectomy with EPIC[31]. Four studies included patients with extra-abdominal distant metastases. In one study, 10/27 patients had extra-abdominal distant metastases, of whom 5 patients underwent resection of extra-abdominal disease[27]. In another study no patient had resection of their distant disease[30] and the other two studies did not document whether distant metastases were resected[31,36].
In three studies[28,38,39], all patients received some form of systemic therapy (neoadjuvant, adjuvant or both). Three series did not document whether their patient cohort received chemotherapy or not and in the remaining thirteen studies the number of the CC-2/3 patients who received systemic therapy was not documented.
In twelve studies[6,16,17,26,28,29,31,33,34,37,39,40] all patients received intraperitoneal chemotherapy (EPIC, IPHC or both) following incomplete cytoreduction (331). In one study the number of patients receiving intraperitoneal chemotherapy was not specified[27]. In the series reported by Huang et al[38], a comparison of intraperitoneal chemotherapy vs no intraperitoneal therapy following incomplete cytoreduction was performed. Five studies did not utilise this treatment modality. Mitomycin C was the most frequently used chemotherapeutic agent[16,26,27,31,34,37,38] followed by fluorouracil[6,28,40].
A breakdown of survival by completeness of cytroreduction is outlined in Table 1. All studies reported survival using the Kaplan Meier method. Patients in whom a CC-0 resection was achieved had a median survival of 25 to 62 mo, whereas following CC-1 median survival ranged from 11 to 35 mo. In studies where the CC-0 and CC-1 groups were analysed together, a median survival of 15.8 to 42 mo was reported. The 986 patients in whom a CC-2/3 cytoreduction was achieved had a median survival ranging from 2.4 to 32 mo. From the data reported, it is possible to identify only 63 patients in the CC-2/3 group who definitively had peritoneal metastases only and median survival in this cohort ranged from 2.4 to 11 mo. Further analysis of survival data for the CC-2/3 group is outlined in Table 2. Ten of the nineteen studies calculated 5 year survival rates. Two studies documented 5 year survival rates of 3%[30] and 4.7%[36] while the others reported no 5 year survival following incomplete cytoreduction.
| Ref. | Year | Study size,n | CC-0,n | Median survival(mo) | CC-1,n | Median survival,(mo) | CC-0/1,n | Median survival(mo) | CC-2/3,n | Median survival(mo) |
| Pestieau et al[26] | 2000 | 104 | - | - | - | - | 44 | 24 | 55 | 12 |
| Verwaal et al[16] | 2003 | 49 | 18 | - | 21 | 20 | - | - | 10 | 5 |
| Glehen et al[6] | 2004 | 506 | 271 | 32.4 | 106 | 24 | - | - | 129 | 8.4 |
| Carmignani et al[27] | 2004 | 27 | - | - | - | - | 15 | 20.6 | 12 | 9 |
| Mahteme et al[40] | 2004 | 18 | - | - | - | - | 11 | 32 | 7 | 10 |
| Füzün et al[28] | 2006 | 29 | 8 | 62 | 18 | 21 | 26 | 37 | 3 | 7 |
| Shen et al[29] | 2008 | 77 | 13 | NR | 35 | 15.2 | - | - | 29 | 4.5 |
| Varban et al[37] | 2009 | 14 | - | - | - | - | 9 | 23 | 5 | 15.4 |
| Chua et al[34] | 2010 | 58 | - | - | - | - | 3 | 19 | ||
| Elias et al[17] | 2010 | 523 | 439 | 33 | 53 | 20 | - | - | 22 | 7 |
| Cavaliere et al[25] | 2011 | 146 | 124 | 25 | 11 | 11 | - | - | 11 | 8 |
| Mulsow et al[30] | 2011 | 125 | - | - | - | - | 31 | 25 | 94 | 8 |
| Chua et al[39] | 2011 | 110 | 72 | 46 | 27 | 35 | - | - | 11 | 32 |
| Matsuda et al[35] | 2012 | 153 | - | - | - | - | 31 | 42 | 122 | 10 |
| Park et al[31] | 2013 | 29 | 24 | - | 0 | - | - | - | 5 | 12 |
| Winer et al[32] | 2013 | 30 | 14 | - | 9 | - | 23 | 15.8 | 4 | 2.4 |
| Huang et al[38] with HIPEC | 2013 | 33 | - | - | - | - | 14 | 21.7 | 19 | 11 |
| Huang et al[38] without HIPEC | 2013 | 29 | - | - | - | - | 9 | 18.3 | 20 | 5 |
| Kobayashi et al[36] | 2014 | 564 | - | - | - | - | 160 | 30.5 | 404 | 12 |
| Ceelen et al[33] | 2014 | 166 | 79 | 49 | 66 | 22 | - | - | 21 | 12 |
| Ref. | Year | Study size,n | Control1median survival(mo) | CC-2/3,n | Median survival (mo) | 1 yr | 2 yr | 3 yr | 4 yr | 5 yr |
| Pestieau et al[26] | 2000 | 104 | - | 55 | 12 | - | - | - | - | 0.0% |
| Verwaal et al[16] | 2003 | 49 | 12.6 | 10 | 5 | - | - | - | - | - |
| Glehen et al[6] | 2004 | 506 | - | 129 | 8.4 | 38.0% | - | 6.0% | - | 0.0% |
| Carmignani et al[27] | 2004 | 27 | - | 12 | 9 | - | - | - | - | - |
| Mahteme et al[40] | 2004 | 18 | 14 | 7 | 10 | - | - | - | - | - |
| Füzün et al[28] | 2006 | 29 | - | 3 | 7 | - | - | - | - | - |
| Shen et al[29] | 2008 | 77 | - | 29 | 4.5 | - | - | 6.0% | - | 0.0% |
| Varban et al[37] | 2009 | 14 | - | 5 | 15.4 | - | 40.0% | - | 20.8% | - |
| Chua et al[34] | 2010 | 58 | - | 3 | 19 | - | - | - | - | 0.0% |
| Elias et al[17] | 2010 | 523 | - | 22 | 7 | - | - | 8.5% | - | 0.0% |
| Cavaliere et al[25] | 2011 | 146 | - | 11 | 8 | - | 0.0% | - | - | 0.0% |
| Mulsow et al[30] | 2011 | 125 | - | 94 | 8 | 39.0% | 17.0% | - | - | 3.0% |
| Chua et al[39] | 2011 | 110 | - | 11 | 32 | - | - | - | - | - |
| Matsuda et al[35] | 2012 | 153 | - | 122 | 10 | - | - | - | - | 0.0% |
| Park et al[31] | 2013 | 29 | - | 5 | 12 | - | - | 0.0% | - | 0.0% |
| Winer et al[32] | 2013 | 30 | - | 4 | 2.4 | - | - | - | - | - |
| Huang et al[38] with HIPEC | 2013 | 33 | - | 19 | 11 | - | - | - | - | - |
| Huang et al[38] without HIPEC | 2013 | 29 | - | 20 | 5 | - | - | - | - | - |
| Kobayashi et al[36] | 2014 | 564 | - | 404 | 12 | - | - | - | - | 4.7% |
| Ceelen et al[33] | 2014 | 166 | - | 21 | 12 | - | - | - | - | - |
The randomised control trial by Verwaal et al[16] was the only study to report perioperative mortality for the incomplete cytoreduction group. Seven out of ten patients in the CC-2/3 group died in comparison to 1/18 (5.5%) patients in the CC-0 group. Furthermore, 80% of the grade 4 toxicities and complications occurred in the CC-2/3 group. The treatment related mortality for the experimental arm (complete and incomplete cytoreduction) was 8%.
It is now broadly accepted that the treatment modality of complete cytoreductive surgery with intraperitoneal chemotherapy confers a survival benefit for appropriately selected patients with peritoneal metastases of colorectal origin. It is also accepted that a complete cytoreduction is associated with better outcomes than an incomplete resection. It is less clear however, whether an incomplete cytoreduction is of benefit with respect to survival or quality of life when compared to non-operative approaches. This question is particularly relevant in the setting of unresectable disease first encountered at laparotomy for planned CRS and HIPEC. Despite advances in staging, this situation arises in up to one quarter of patients[23,42,43] and poses a dilemma for the surgeon; should they proceed and remove resectable disease, combining it with HIPEC, or should the procedure be abandoned?
From the current literature review it is not possible to make firm conclusions as to the merits or otherwise of persisting with CRS and HIPEC when it is apparent that it will not be possible to resect all disease. This difficulty arises mainly due to the heterogeneity of patients and reported approaches, most of which has been taken from uncontrolled studies. In the only RCT in the series, the 10 patients in the incomplete group had a median survival of 5 mo compared with 12.6 mo for those undergoing systemic chemotherapy[16]. Perioperative mortality in the incomplete group was high however, impacting significantly on overall survival for this group. Similar results were obtained in the case control series by Mahteme et al[40] who reported median survival of 10 mo in those undergoing CRS vs 14 mo for those in the standard chemotherapy group. It is however noteworthy that sixteen of the nineteen studies in the current review reported median survival of 12 mo or less following incomplete cytoreduction. While most of the studies were non-comparative, it is clear that these outcomes are no better than historical controls, or those reported with best systemic treatment. These outcomes suggest no survival advantage to an incomplete cytoreduction. There was however great heterogeneity of patients (many of whom had non-peritoneal distant disease), the extent of surgery, use of HIPEC, and the delivery of systemic treatment. From the literature it is noted that peritoneal metastases of appendiceal adenocarcinoma origin tend to have a better overall survival when compared to true colorectal peritoneal metastases[13,44].
The value of intraperitoneal chemotherapy after CC-2/3 is also unknown. For the twelve studies in the current review in which all patients received intraperitoneal chemotherapy, median survival for the patients with incomplete cytoreduction ranged from 4.1 to 32 mo and, for the five without intraperitoneal chemotherapy, from 2.4 to 15 mo[25,30,32,35,36] (Table 3). In a case-control study, Huang et al[38] compared outcomes in CRS/HIPEC with CRS alone. Those in the HIPEC group had a median survival of 11 mo vs 5 mo in those who underwent CRS alone. It is important to note that the cut off point, with respect to the size of residual tumour, for use of intraperitoneal chemotherapy varied across the studies in this review, varying from > 1 mm[17] to > 5 mm[6].
| Ref. | CC-2/3, n | Median survival (mo) |
| With intraperitoneal chemotherapy | ||
| Shen et al[29] | 29 | 4.5 |
| Verwaal et al[16] | 10 | 5 |
| Elias et al[17] | 22 | 7 |
| Füzün et al[28] | 3 | 7 |
| Glehen et al[6] | 129 | 8.4 |
| Carmignani et al[27] | 12 | 9 |
| Mahteme et al[40] | 7 | 10 |
| Huang et al[38] with HIPEC | 19 | 11 |
| Park et al[31] | 5 | 12 |
| Ceelen et al[33] | 21 | 12 |
| Pestieau et al[26] | 55 | 12 |
| Varban et al[37] | 5 | 15.4 |
| Chua et al[34] | 3 | 19 |
| Chua et al[39] | 11 | 32 |
| Without intraperitoneal chemotherapy | ||
| Winer et al[32] | 4 | 2.4 |
| Huang et al[38] without HIPEC | 20 | 5 |
| Cavaliere et al[25] | 11 | 8 |
| Mulsow et al[30] | 94 | 8 |
| Matsuda et al[35] | 122 | 10 |
| Kobayashi et al[36] | 404 | 12 |
While its impact on survival remains debatable, an incomplete cytoreduction with HIPEC may provide symptom relief. Malignant ascites results in abdominal distension and dyspnoea and symptomatic relief with paracentesis is transient at best as failure to treat the cause of the ascites results in rapid reaccumulation of the fluid[45]. Chua et al[34] specifically assessed a small number of patients who underwent CC-2/3 resection for colorectal PM. Two out of the three patients reported resolution of their symptoms (abdominal pain, anorexia, distension) postoperatively. Randle et al[46] found that partial cytoreduction and HIPEC was successful in treating malignant ascites (no radiological evidence of ascites three months post-operatively) in 84% (243/288) of patients with peritoneal metastases from a variety of primary tumours. This suggests a potential role for intraperitoneal chemotherapy in controlling ascites and improving symptoms. Garofalo et al[47] found that laparoscopic HIPEC successfully treated debilitating ascites in 3 patients with PM of colorectal origin. Valle et al[48] showed a benefit (at least in the short term) to laparoscopic HIPEC with mitomycin C in patients with PM of colorectal origin. At one month post-operatively, forty-nine out of fifty-two patients (94%) were free of ascites. Eleven of these patients had PM of colorectal origin. These findings suggest a potential benefit, in the presence of unresectable disease at laparotomy, from intraperitoneal chemotherapy. Patients may also benefit, albeit temporarily, from formation of an ostomy, intestinal bypass or adhesiolysis[49].
Factors associated with unresectability have been documented in the literature. The two main causative factors are 5-7 abdominal regions affected by PM and extensive small bowel or mesenteric involvement[50,51]. Only three of the studies in the current review documented the reasons for unresectability. These included extensive small bowel involvement, hepatic metastases and extensive disease involving the diaphragm and lesser sac[31,32,34]. Similar causative factors were identified in a series reported by van Oudheusden and colleagues[43], however, the main factor was found to be a high PCI with 50% (41/82) of patients undergoing an open and closed laparotomy upon the discovery of a PCI exceeding 20. The literature suggests that a complete cytoreduction confers little survival benefit in patients with a PCI > 17[52] and the association between a high PCI and poor outcomes is documented in eight of the studies[6,16,17,25-28,38] in this review. However, while there are reports showing favourable outcomes in a subset of these patients[53,54], selecting which patients with a high PCI to operate on remains a challenge.
It appears that tumour biology can impact on resectability and outcomes in peritoneal carcinomatosis. Winer et al[32] reported outcomes in patients with poorly differentiated tumours. They found that PM was a common finding in patients with signet ring cell subtype in the primary tumour and accounted for 14% of the cytoreductive surgeries performed in their institute. Tumours with signet ring histology are more likely to have metastasised at initial presentation and have an extremely poor prognosis despite advances in systemic chemotherapy[55,56] .The median survival for patients with an incomplete cytoreduction in Winer’s series was 2.4 mo. Winer et al[32] and Ceelen et al[33] concluded that aggressive histological subtypes, including signet ring, are particularly resistant to cytoreductive surgery and this finding is also reported by Chua et al[57]. PM with signet ring histology is also associated with an increased risk of death in those undergoing open and closed laparotomy[43] as well as an increased risk of recurrence[58]. These results suggest that patients with signet ring subtype and peritoneal metastases should be approached with caution, particularly when there is a question regarding resectability.
From the published literature it would appear that there is a survival benefit to the delivery of adjuvant systemic treatment following incomplete cytoreduction. Chua et al[39] compared outcomes of patients with peritoneal metastases who received systemic chemotherapy vs patients who underwent an incomplete cytoreduction, intraperitoneal chemotherapy and systemic chemotherapy. Those in the systemic chemotherapy group alone had median survivals of 11 to 23 mo (depending on the chemotherapy regimen delivered - standard, modern and modern chemotherapy with biological agents) while the median survival of patients who underwent an incomplete cytoreduction was 32 mo. Overall, they found that the administration of a modern chemotherapy regimen improved survival in patients who underwent an incomplete CRS/HIPEC when compared with standard chemotherapy and that an incomplete cytoreduction conferred a survival benefit over systemic chemotherapy alone. Similarly, Glehen et al[6] found that adjuvant chemotherapy (irinotecan or oxaliplatin) following incomplete cytoreduction significantly improved survival, when compared with no adjuvant chemotherapy.
Klaver et al[59] and Hompes et al[50] specifically assessed the impact of palliative chemotherapy (without cytoreductive surgery or HIPEC) on survival in patients with colorectal PM. In a population based study, Klaver et al[59] found that patients who received systemic chemotherapy survived for up to 66 wk vs 11 wk for those who didn’t. They also found that peritoneal metastases are somewhat resistant to fluorouracil monotherapy but may be sensitive to modern, multi-agent chemotherapy regimens[59]. Hompes et al[50] reported a series of 43 patients with unresectable PM in whom no resection was performed at laparotomy. The overall median survival for these patients was 6.3 mo with those who received palliative chemotherapy having a slightly improved survival of 9.3 mo vs 3.1 for those without.
Verwaal et al[16] showed particularly adverse peri-operative outcomes associated with an incomplete cytoreduction. More recent studies do not provide sufficient data to support or refute this finding. It is however clear that the morbidity associated with CRS and HIPEC has improved significantly and is now comparable with other major gastrointestinal procedures[60]. This factor must be taken into account if a decision is to be made to proceed on an individual patient basis, even when a complete cytoreduction will not be possible.
It is generally accepted that the preoperative diagnosis of unresectable peritoneal carcinomatosis precludes an attempt at cytoreductive surgery and patients should not routinely be exposed to an unnecessary laparotomy. In the setting of unresectable peritoneal disease discovered at laparotomy, there is currently no evidence that an incomplete cytoreduction, with or without HIPEC, will improve survival. However, the available data is of poor quality and the decision to proceed must be made on an individual patient basis, taking into account the site and extent of disease, tumour biology and any palliative benefit that may result, and balancing this with the risk of morbidity. Certain patients, particularly those with ascites may receive a quality of life benefit following incomplete cytoreduction and intraperitoneal chemotherapy.
P- Reviewer: van Oudheusden TR S- Editor: Yu J L- Editor: A E- Editor: Wu HL
| 1. | Chu DZ, Lang NP, Thompson C, Osteen PK, Westbrook KC. Peritoneal carcinomatosis in nongynecologic malignancy. A prospective study of prognostic factors. Cancer. 1989;63:364-367. [PubMed] |
| 2. | Dawson LE, Russell AH, Tong D, Wisbeck WM. Adenocarcinoma of the sigmoid colon: sites of initial dissemination and clinical patterns of recurrence following surgery alone. J Surg Oncol. 1983;22:95-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 56] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 3. | Russell AH, Pelton J, Reheis CE, Wisbeck WM, Tong DY, Dawson LE. Adenocarcinoma of the colon: an autopsy study with implications for new therapeutic strategies. Cancer. 1985;56:1446-1451. [PubMed] |
| 4. | Welch JP, Donaldson GA. The clinical correlation of an autopsy study of recurrent colorectal cancer. Ann Surg. 1979;189:496-502. [PubMed] |
| 5. | Knorr C, Reingruber B, Meyer T, Hohenberger W, Stremmel C. Peritoneal carcinomatosis of colorectal cancer: incidence, prognosis, and treatment modalities. Int J Colorectal Dis. 2004;19:181-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Glehen O, Kwiatkowski F, Sugarbaker PH, Elias D, Levine EA, De Simone M, Barone R, Yonemura Y, Cavaliere F, Quenet F. Cytoreductive surgery combined with perioperative intraperitoneal chemotherapy for the management of peritoneal carcinomatosis from colorectal cancer: a multi-institutional study. J Clin Oncol. 2004;22:3284-3292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 860] [Cited by in RCA: 882] [Article Influence: 42.0] [Reference Citation Analysis (0)] |
| 7. | Jayne DG, Fook S, Loi C, Seow-Choen F. Peritoneal carcinomatosis from colorectal cancer. Br J Surg. 2002;89:1545-1550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 598] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 8. | Sadeghi B, Arvieux C, Glehen O, Beaujard AC, Rivoire M, Baulieux J, Fontaumard E, Brachet A, Caillot JL, Faure JL. Peritoneal carcinomatosis from non-gynecologic malignancies: results of the EVOCAPE 1 multicentric prospective study. Cancer. 2000;88:358-363. [PubMed] |
| 9. | Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Alberts SR. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol. 2004;22:23-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1779] [Cited by in RCA: 1720] [Article Influence: 81.9] [Reference Citation Analysis (0)] |
| 10. | Jonker DJ, Maroun JA, Kocha W. Survival benefit of chemotherapy in metastatic colorectal cancer: a meta-analysis of randomized controlled trials. Br J Cancer. 2000;82:1789-1794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 67] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 11. | Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335-2342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7832] [Cited by in RCA: 7729] [Article Influence: 368.0] [Reference Citation Analysis (1)] |
| 12. | Mirnezami R, Moran BJ, Harvey K, Cecil T, Chandrakumaran K, Carr N, Mohamed F, Mirnezami AH. Cytoreductive surgery and intraperitoneal chemotherapy for colorectal peritoneal metastases. World J Gastroenterol. 2014;20:14018-14032. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 33] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Sugarbaker PH, Jablonski KA. Prognostic features of 51 colorectal and 130 appendiceal cancer patients with peritoneal carcinomatosis treated by cytoreductive surgery and intraperitoneal chemotherapy. Ann Surg. 1995;221:124-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 430] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 14. | Sugarbaker PH, Stuart OA, Yoo D. Strategies for management of the peritoneal surface component of cancer: cytoreductive surgery plus perioperative intraperitoneal chemotherapy. J Oncol Pharm Pract. 2005;11:111-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 15. | Elias DM, Ouellet JF. Intraperitoneal chemohyperthermia: rationale, technique, indications, and results. Surg Oncol Clin N Am. 2001;10:915-933, xi. [PubMed] |
| 16. | Verwaal VJ, van Ruth S, de Bree E, van Sloothen GW, van Tinteren H, Boot H, Zoetmulder FA. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J Clin Oncol. 2003;21:3737-3743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1396] [Cited by in RCA: 1512] [Article Influence: 68.7] [Reference Citation Analysis (0)] |
| 17. | Elias D, Gilly F, Boutitie F, Quenet F, Bereder JM, Mansvelt B, Lorimier G, Dubè P, Glehen O. Peritoneal colorectal carcinomatosis treated with surgery and perioperative intraperitoneal chemotherapy: retrospective analysis of 523 patients from a multicentric French study. J Clin Oncol. 2010;28:63-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 685] [Cited by in RCA: 737] [Article Influence: 46.1] [Reference Citation Analysis (0)] |
| 18. | Mirnezami R, Mehta AM, Chandrakumaran K, Cecil T, Moran BJ, Carr N, Verwaal VJ, Mohamed F, Mirnezami AH. Cytoreductive surgery in combination with hyperthermic intraperitoneal chemotherapy improves survival in patients with colorectal peritoneal metastases compared with systemic chemotherapy alone. Br J Cancer. 2014;111:1500-1508. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 19. | Verwaal VJ, Bruin S, Boot H, van Slooten G, van Tinteren H. 8-year follow-up of randomized trial: cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann Surg Oncol. 2008;15:2426-2432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 710] [Cited by in RCA: 765] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 20. | Esquivel J, Elias D, Baratti D, Kusamura S, Deraco M. Consensus statement on the loco regional treatment of colorectal cancer with peritoneal dissemination. J Surg Oncol. 2008;98:263-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 152] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 21. | Koh JL, Yan TD, Glenn D, Morris DL. Evaluation of preoperative computed tomography in estimating peritoneal cancer index in colorectal peritoneal carcinomatosis. Ann Surg Oncol. 2009;16:327-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 263] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 22. | Pasqual EM, Bertozzi S, Bacchetti S, Londero AP, Basso SM, Santeufemia DA, Lo Re G, Lumachi F. Preoperative assessment of peritoneal carcinomatosis in patients undergoing hyperthermic intraperitoneal chemotherapy following cytoreductive surgery. Anticancer Res. 2014;34:2363-2368. [PubMed] |
| 23. | Iversen LH, Rasmussen PC, Laurberg S. Value of laparoscopy before cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for peritoneal carcinomatosis. Br J Surg. 2013;100:285-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 105] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 24. | Elias D, Quenet F, Goéré D. Current status and future directions in the treatment of peritoneal dissemination from colorectal carcinoma. Surg Oncol Clin N Am. 2012;21:611-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 25. | Cavaliere F, De Simone M, Virzì S, Deraco M, Rossi CR, Garofalo A, Di Filippo F, Giannarelli D, Vaira M, Valle M. Prognostic factors and oncologic outcome in 146 patients with colorectal peritoneal carcinomatosis treated with cytoreductive surgery combined with hyperthermic intraperitoneal chemotherapy: Italian multicenter study S.I.T.I.L.O. Eur J Surg Oncol. 2011;37:148-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 26. | Pestieau SR, Sugarbaker PH. Treatment of primary colon cancer with peritoneal carcinomatosis: comparison of concomitant vs. delayed management. Dis Colon Rectum. 2000;43:1341-1346; discussion 1341-1346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 122] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 27. | Carmignani CP, Ortega-Perez G, Sugarbaker PH. The management of synchronous peritoneal carcinomatosis and hematogenous metastasis from colorectal cancer. Eur J Surg Oncol. 2004;30:391-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 28. | Füzün M, Sökmen S, Terzi C, Canda AE. Cytoreductive approach to peritoneal carcinomatosis originated from colorectal cancer: Turkish experience. Acta Chir Iugosl. 2006;53:17-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Shen P, Thai K, Stewart JH, Howerton R, Loggie BW, Russell GB, Levine EA. Peritoneal surface disease from colorectal cancer: comparison with the hepatic metastases surgical paradigm in optimally resected patients. Ann Surg Oncol. 2008;15:3422-3432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Mulsow J, Merkel S, Agaimy A, Hohenberger W. Outcomes following surgery for colorectal cancer with synchronous peritoneal metastases. Br J Surg. 2011;98:1785-1791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 44] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 31. | Park SY, Choi GS, Park JS, Kim HJ, Ryuk JP, Yun SH, Kim JG, Kang BW. Laparoscopic cytoreductive surgery and early postoperative intraperitoneal chemotherapy for patients with colorectal cancer peritoneal carcinomatosis: initial results from a single center. Surg Endosc. 2014;28:1555-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 32. | Winer J, Zenati M, Ramalingam L, Jones H, Zureikat A, Holtzman M, Lee K, Ahrendt S, Pingpank J, Zeh HJ. Impact of aggressive histology and location of primary tumor on the efficacy of surgical therapy for peritoneal carcinomatosis of colorectal origin. Ann Surg Oncol. 2014;21:1456-1462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 48] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 33. | Ceelen W, Van Nieuwenhove Y, Putte DV, Pattyn P. Neoadjuvant chemotherapy with bevacizumab may improve outcome after cytoreduction and hyperthermic intraperitoneal chemoperfusion (HIPEC) for colorectal carcinomatosis. Ann Surg Oncol. 2014;21:3023-3028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 72] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 34. | Chua TC, Baker B, Yan TD, Zhao J, Morris DL. Palliative effects of an incomplete cytoreduction combined with perioperative intraperitoneal chemotherapy. Am J Clin Oncol. 2010;33:568-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Matsuda K, Hotta T, Takifuji K, Yamamoto M, Nasu T, Togo N, Oka M, Tabuse K, Yamaue H. Clinical impact of a macroscopically complete resection of colorectal cancer with peritoneal carcinomatosis. Surgery. 2012;151:238-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.2] [Reference Citation Analysis (32)] |
| 36. | Kobayashi H, Kotake K, Funahashi K, Hase K, Hirata K, Iiai T, Kameoka S, Kanemitsu Y, Maeda K, Murata A. Clinical benefit of surgery for stage IV colorectal cancer with synchronous peritoneal metastasis. J Gastroenterol. 2014;49:646-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 37. | Varban O, Levine EA, Stewart JH, McCoy TP, Shen P. Outcomes associated with cytoreductive surgery and intraperitoneal hyperthermic chemotherapy in colorectal cancer patients with peritoneal surface disease and hepatic metastases. Cancer. 2009;115:3427-3436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 38. | Huang CQ, Feng JP, Yang XJ, Li Y. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy improves survival of patients with peritoneal carcinomatosis from colorectal cancer: a case-control study from a Chinese center. J Surg Oncol. 2014;109:730-739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 39. | Chua TC, Morris DL, Saxena A, Esquivel J, Liauw W, Doerfer J, Germer CT, Kerscher AG, Pelz JO. Influence of modern systemic therapies as adjunct to cytoreduction and perioperative intraperitoneal chemotherapy for patients with colorectal peritoneal carcinomatosis: a multicenter study. Ann Surg Oncol. 2011;18:1560-1567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 40. | Mahteme H, Hansson J, Berglund A, Påhlman L, Glimelius B, Nygren P, Graf W. Improved survival in patients with peritoneal metastases from colorectal cancer: a preliminary study. Br J Cancer. 2004;90:403-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 90] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 41. | Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996;82:359-374. [PubMed] |
| 42. | Pomel C, Appleyard TL, Gouy S, Rouzier R, Elias D. The role of laparoscopy to evaluate candidates for complete cytoreduction of peritoneal carcinomatosis and hyperthermic intraperitoneal chemotherapy. Eur J Surg Oncol. 2005;31:540-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 43. | van Oudheusden TR, Braam HJ, Luyer MD, Wiezer MJ, van Ramshorst B, Nienhuijs SW, de Hingh IH. Peritoneal cancer patients not suitable for cytoreductive surgery and HIPEC during explorative surgery: risk factors, treatment options, and prognosis. Ann Surg Oncol. 2015;22:1236-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 44. | Chua TC, Al-Alem I, Saxena A, Liauw W, Morris DL. Surgical cytoreduction and survival in appendiceal cancer peritoneal carcinomatosis: an evaluation of 46 consecutive patients. Ann Surg Oncol. 2011;18:1540-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 45. | Husain A, Bezjak A, Easson A. Malignant ascites symptom cluster in patients referred for paracentesis. Ann Surg Oncol. 2010;17:461-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 46. | Randle RW, Swett KR, Swords DS, Shen P, Stewart JH, Levine EA, Votanopoulos KI. Efficacy of cytoreductive surgery with hyperthermic intraperitoneal chemotherapy in the management of malignant ascites. Ann Surg Oncol. 2014;21:1474-1479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 47. | Garofalo A, Valle M, Garcia J, Sugarbaker PH. Laparoscopic intraperitoneal hyperthermic chemotherapy for palliation of debilitating malignant ascites. Eur J Surg Oncol. 2006;32:682-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 80] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 48. | Valle M, Van der Speeten K, Garofalo A. Laparoscopic hyperthermic intraperitoneal peroperative chemotherapy (HIPEC) in the management of refractory malignant ascites: A multi-institutional retrospective analysis in 52 patients. J Surg Oncol. 2009;100:331-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 49. | Paul Olson TJ, Pinkerton C, Brasel KJ, Schwarze ML. Palliative surgery for malignant bowel obstruction from carcinomatosis: a systematic review. JAMA Surg. 2014;149:383-392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 128] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 50. | Hompes D, Boot H, van Tinteren H, Verwaal V. Unresectable peritoneal carcinomatosis from colorectal cancer: a single center experience. J Surg Oncol. 2011;104:269-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 51. | Rodt AP, Svarrer RO, Iversen LH. Clinical course for patients with peritoneal carcinomatosis excluded from cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. World J Surg Oncol. 2013;11:232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 52. | Goéré D, Souadka A, Faron M, Cloutier AS, Viana B, Honoré C, Dumont F, Elias D. Extent of Colorectal Peritoneal Carcinomatosis: Attempt to Define a Threshold Above Which HIPEC Does Not Offer Survival Benefit: A Comparative Study. Ann Surg Oncol. 2015;22:2958-2964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 153] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 53. | da Silva RG, Sugarbaker PH. Analysis of prognostic factors in seventy patients having a complete cytoreduction plus perioperative intraperitoneal chemotherapy for carcinomatosis from colorectal cancer. J Am Coll Surg. 2006;203:878-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 227] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 54. | Cashin PH, Dranichnikov F, Mahteme H. Cytoreductive surgery and hyperthermic intra-peritoneal chemotherapy treatment of colorectal peritoneal metastases: cohort analysis of high volume disease and cure rate. J Surg Oncol. 2014;110:203-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Kang H, O’Connell JB, Maggard MA, Sack J, Ko CY. A 10-year outcomes evaluation of mucinous and signet-ring cell carcinoma of the colon and rectum. Dis Colon Rectum. 2005;48:1161-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 243] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 56. | Hyngstrom JR, Hu CY, Xing Y, You YN, Feig BW, Skibber JM, Rodriguez-Bigas MA, Cormier JN, Chang GJ. Clinicopathology and outcomes for mucinous and signet ring colorectal adenocarcinoma: analysis from the National Cancer Data Base. Ann Surg Oncol. 2012;19:2814-2821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 265] [Article Influence: 20.4] [Reference Citation Analysis (1)] |
| 57. | Chua TC, Pelz JO, Kerscher A, Morris DL, Esquivel J. Critical analysis of 33 patients with peritoneal carcinomatosis secondary to colorectal and appendiceal signet ring cell carcinoma. Ann Surg Oncol. 2009;16:2765-2770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 58. | van Oudheusden TR, Braam HJ, Nienhuijs SW, Wiezer MJ, van Ramshorst B, Luyer P, de Hingh IH. Poor outcome after cytoreductive surgery and HIPEC for colorectal peritoneal carcinomatosis with signet ring cell histology. J Surg Oncol. 2015;111:237-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 59. | Klaver YL, Lemmens VE, Creemers GJ, Rutten HJ, Nienhuijs SW, de Hingh IH. Population-based survival of patients with peritoneal carcinomatosis from colorectal origin in the era of increasing use of palliative chemotherapy. Ann Oncol. 2011;22:2250-2256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 60. | Chua TC, Yan TD, Saxena A, Morris DL. Should the treatment of peritoneal carcinomatosis by cytoreductive surgery and hyperthermic intraperitoneal chemotherapy still be regarded as a highly morbid procedure?: a systematic review of morbidity and mortality. Ann Surg. 2009;249:900-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 387] [Cited by in RCA: 422] [Article Influence: 26.4] [Reference Citation Analysis (0)] |