Published online Oct 15, 2013. doi: 10.4251/wjgo.v5.i10.195
Revised: August 30, 2013
Accepted: September 3, 2013
Published online: October 15, 2013
Processing time: 113 Days and 6.8 Hours
Gangliocytic paraganglioma is a rare neuroendocrine tumor predominantly arising in the second part of the duodenum with rare local recurrence or metastasis to regional lymph nodes. A 92-year-old female presented with obstructive jaundice. On exam she had pale conjunctiva and icteric sclera. Abdominal examination revealed tenderness in the upper abdomen. Laboratory data was consistent with obstructive jaundice. Computed tomography of the abdomen revealed a dilated gall bladder and a common bile duct (CBD) with no evidence of liver lesions or pancreatic head mass. Endoscopic ultrasonography revealed a 1 cm isoechoic submucosal nodule at the periampullary area, dilated CBD (9 mm), a prominent pancreatic duct (4.1 mm) and a hydropic gall bladder with no stones. Endoscopic retrograde cholangiopancreaticography was performed to relieve obstruction and showed a 1 cm periampullary mass which underwent an en-bloc snare resection. Histopathology analyses with immunohistochemical stains were positive for cytokeratin, synaptophysin, S-100 protein, neuron specific enolase and negative for actin and desmin consistent with periampullary gangliocytic paraganglioma. Periampullary gangliocytic paraganglioma is a rare benign tumor of the small bowel. Common presentation includes abdominal pain and obstructive jaundice which should be included in differential diagnosis of obstructive jaundice. Endoscopic resection is a curative therapy in the absence of local invasion or distant metastasis.
Core tip: This case report shed some light on a rare cause of obstructive jaundice in elderly patients. The disease is rare but should be considered in the differential diagnosis of biliary obstruction. The literature provided summarizes several outcomes of case presentation with this disorder and provide input on some of the aggressive feature of this disorder.
- Citation: Sathyamurthy A, Choudhary A, Ng D, Okponobi S, Diaz-Arias A, Grewal A, Hammoud GM. Obstructive jaundice due to a rare periampullary tumor. World J Gastrointest Oncol 2013; 5(10): 195-197
- URL: https://www.wjgnet.com/1948-5204/full/v5/i10/195.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v5.i10.195
Gangliocytic paraganglioma is a rare neuroendocrine tumor predominantly arising in the second part of the duodenum with rare local recurrence or metastasis to regional lymph nodes. We present a case of a 92-year-old female with abdominal pain, obstructive jaundice and a mass in the second portion of the duodenum, near the papillary region.
A 92-year-old female presented with upper abdominal pain associated with nausea, vomiting and jaundice for 4 d. She had no history of fever, chills, melena, hematemesis or weight loss. Her past medical history was significant for diabetes, hypertension, gastroesophageal reflux, chronic renal insufficiency and hypothyroidism.
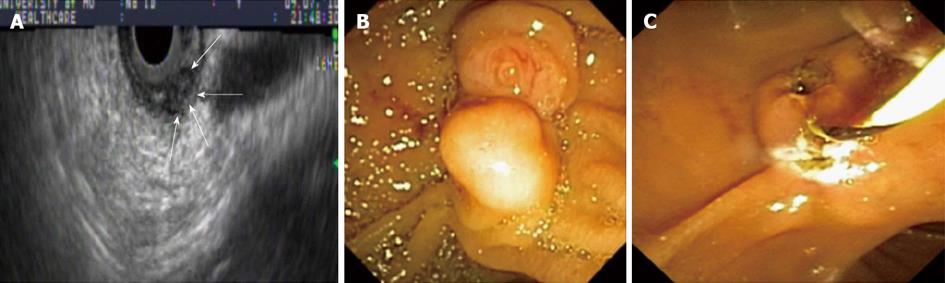
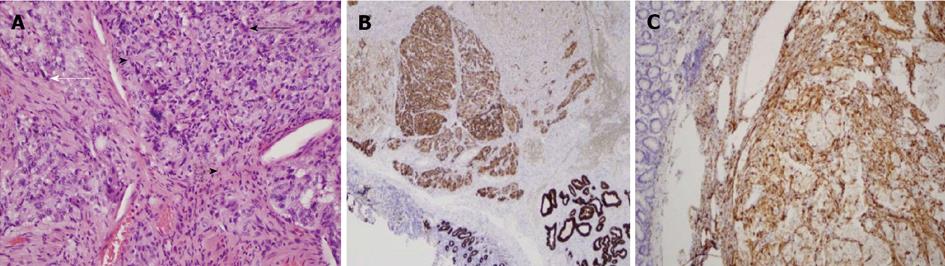
Physical examination revealed pale conjunctiva and icteric sclera with no cervical lymphadenopathy. Abdominal examination revealed tenderness in the upper abdomen with no rebound tenderness. She had no hepatosplenomegaly or palpable masses. Bowel sounds were present. Laboratory data were significant for hemoglobin 9.7 g/dL, total bilirubin 2.4 mg/dL, aspartate aminotransferase 166 U/L, alanine aminotransferase 465 U/L, and alkaline phosphatase 515 U/L. Her white cell count, serum amylase and lipase were normal. Computed tomography of abdomen revealed a dilated gallbladder and common bile duct (CBD = 9 mm) with no evidence of liver lesions or pancreatic head mass. Endoscopic ultrasound (EUS) revealed a well-defined, 1 cm × 1.5 cm heterogeneous, isoechoic, periampullary submucosal nodule (Figure 1A). The nodule appears to cause an extrinsic compression of the CBD at the ampullary orifice. The CBD was dilated at 10 mm in diameter and the pancreatic duct (PD) appeared mildly prominent and measured 4.1 mm in diameter. The gallbladder appeared hydropic with no stones. The lesion did not appear to invade the CBD, PD or muscularis propria layer of the duodenal wall (Figure 1A). Endoscopic retrograde cholangiogram with biliary sphincterotomy was performed to relieve jaundice and showed a 1.5 cm × 2.0 cm periampullary nodule that partially obstruct the orifice of the major papilla (Figure 1B) which underwent en-bloc endoscopic mucosal resection with electrocautery snare (Figure 1C). Upon follow up, jaundice resolved once resection of the lesion was performed. There were no lymph nodes seen on EUS examination. Histopathology analyses with immunohistochemical stains were positive for cytokeratin (Figure 2B), synaptophysin, S-100 protein (Figure 2C), neuron specific enolase and negative for actin and desmin confirming the diagnosis of periampullary gangliocytic paraganglioma (Figure 2A). The margins were free of tumor and there were no histologic findings of aggressive behavior such as mitosis and/or pleomorphism.
Gangliocytic paragangliomas are exceedingly rare tumors that arise in close proximity to the papilla of Vater and 90% are found in the second part of the duodenum[1]. The disease is common in the 5th decade and the incidence is slightly higher in males with M:F ratio 1.8:1. Gangliocytic paragangliomas are epithelial (submucosal) tumors with three histological cell types namely epithelioid, ganglion and spindle cells[2].
Typical presentation is abdominal pain, gastrointestinal outlet obstruction and bleeding. However, obstructive jaundice is not common. In our case, jaundice was presumed secondary to mechanical obstruction of the ampullary orifice by the tumor. Immunohistochemistry is positive for cytokeratin, synaptophysin, neuron specific antigen and S-100 protein[2]. Endoscopic ultrasonography is useful for preoperative differential diagnosis such as gastrointestinal stromal tumors, carcinoids and periampullary adenoma. The disease generally follows a benign course with rare invasive growth patterns and lymph node metastasis[3,4].
Histologic findings such as increase mitosis, pleomorphism, infiltrative margin and lymph node metastasis are suggestive of potential malignant features[5,6]. If feasible, endoscopic resection is a curative therapy in the absence of local invasion or distant metastasis. Table 1 shows summary of case reports, findings and outcome from selected publications[5-10].
| Author | Presentation | Endoscopic findings | Outcome | Conclusion |
| Kwon et al[5] | 56 yr old male with melena | EGD-tumor of ampulla of Vater with bleeding on surface | Pancreaticoduodenectomy | If followed up after a diagnosis, local excision can be curative, avoiding surgery or lymph node dissection |
| Okubo et al[6] | 61 yr old male with epigastric pain and melena | EGD, ERCP, EUS-tumor of papilla of Vater | Pylorus-preserving Pancreaticoduodenectomy and lymph node dissection; Lymph nodes positive | Do not limit to local resection, as disease recurrence, lymph node involvement or distant metastases may occur |
| Witkiewicz et al[7] | 38 yr old female with right upper quadrant abdominal pain | EGD-mass in duodenum near ampulla of Vater | Endoscopic excision of mass followed by pylorus-preserving pancreaticoduodenectomy as margin was positive | It may recur or metastasize; hence pancreaticoduodenectomy with lymph node dissection might be indicated for large lesions with infiltrative margin or lesions with pleomorphism and mitoses |
| Morita et al[8] | 53 yr old male with incidental finding on EGD | EUS-submucosal tumor in the 3rd-4th layer | Endoscopic mucosal resection | Endoscopic removal is an alternative to surgical resection if no local or distant invasion |
| Sakhuja et al[9] | 33 yr old male with obstructive jaundice | ERCP-periampullary growth | Pancreaticoduodenectomy | Recognize and diagnose this rare benign entity (with 3 components on H and E sections) |
| Evans et al[10] | 56 yr old male with epigastric pain, vomiting and obstructive jaundice | EGD-pedunculated ampullary tumor | Pylorus-preserving total pancreatectomy | Benign entity-2 yr post procedure no recurrence of tumor |
P- Reviewers El-Karaksy HM, Iacono C, Nakai Y S- Editor Wen LL L- Editor A E- Editor Liu XM
| 1. | Scheithauer BW, Nora FE, LeChago J, Wick MR, Crawford BG, Weiland LH, Carney JA. Duodenal gangliocytic paraganglioma. Clinicopathologic and immunocytochemical study of 11 cases. Am J Clin Pathol. 1986;86:559-565. [PubMed] |
| 2. | Perrone T, Sibley RK, Rosai J. Duodenal gangliocytic paraganglioma. An immunohistochemical and ultrastructural study and a hypothesis concerning its origin. Am J Surg Pathol. 1985;9:31-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 125] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 3. | Dookhan DB, Miettinen M, Finkel G, Gibas Z. Recurrent duodenal gangliocytic paraganglioma with lymph node metastases. Histopathology. 1993;22:399-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 59] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 4. | Hashimoto S, Kawasaki S, Matsuzawa K, Harada H, Makuuchi M. Gangliocytic paraganglioma of the papilla of Vater with regional lymph node metastasis. Am J Gastroenterol. 1992;87:1216-1218. [PubMed] |
| 5. | Kwon J, Lee SE, Kang MJ, Jang JY, Kim SW. A case of gangliocytic paraganglioma in the ampulla of Vater. World J Surg Oncol. 2010;8:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Okubo Y, Yokose T, Tuchiya M, Mituda A, Wakayama M, Hasegawa C, Sasai D, Nemoto T, Shibuya K. Duodenal gangliocytic paraganglioma showing lymph node metastasis: a rare case report. Diagn Pathol. 2010;5:27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 7. | Witkiewicz A, Galler A, Yeo CJ, Gross SD. Gangliocytic paraganglioma: case report and review of the literature. J Gastrointest Surg. 2007;11:1351-1354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 8. | Morita T, Tamura S, Yokoyama Y, Onishi T, Kuratani Y, Mizuta H, Onishi S. Endoscopic resection of a duodenal gangliocytic paraganglioma. Dig Dis Sci. 2007;52:1400-1404. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 9. | Sakhuja P, Malhotra V, Gondal R, Dutt N, Choudhary A. Periampullary gangliocytic paraganglioma. J Clin Gastroenterol. 2001;33:154-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Evans JD, Wilson PG, Barber PC, Neoptolemos JP. Duodenal gangliocytic paraganglioma presenting as an ampullary tumor. Int J Pancreatol. 1996;20:131-134. [PubMed] |