Published online Feb 15, 2024. doi: 10.4251/wjgo.v16.i2.273
Peer-review started: October 6, 2023
First decision: December 5, 2023
Revised: December 13, 2023
Accepted: January 8, 2024
Article in press: January 8, 2024
Published online: February 15, 2024
Processing time: 118 Days and 19.8 Hours
Advanced hepatocellular carcinoma (HCC) is a severe malignancy that poses a serious threat to human health. Owing to challenges in early diagnosis, most patients lose the opportunity for radical treatment when diagnosed. Nonetheless, recent advancements in cancer immunotherapy provide new directions for the treatment of HCC. For instance, monoclonal antibodies against immune check
Core Tip: Although the incidence of hepatocellular carcinoma (HCC) in China is decreasing in all age groups, the overall treatment effect of a single immune checkpoint inhibitors regimen is often poor due to the obvious immunosuppressive features of HCC. Therefore, we review the current status, research progress and future directions of different immunotherapy strategies in HCC treatment.
- Citation: Pan D, Liu HN, Qu PF, Ma X, Ma LY, Chen XX, Wang YQ, Qin XB, Han ZX. Progress in the treatment of advanced hepatocellular carcinoma with immune combination therapy. World J Gastrointest Oncol 2024; 16(2): 273-286
- URL: https://www.wjgnet.com/1948-5204/full/v16/i2/273.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v16.i2.273
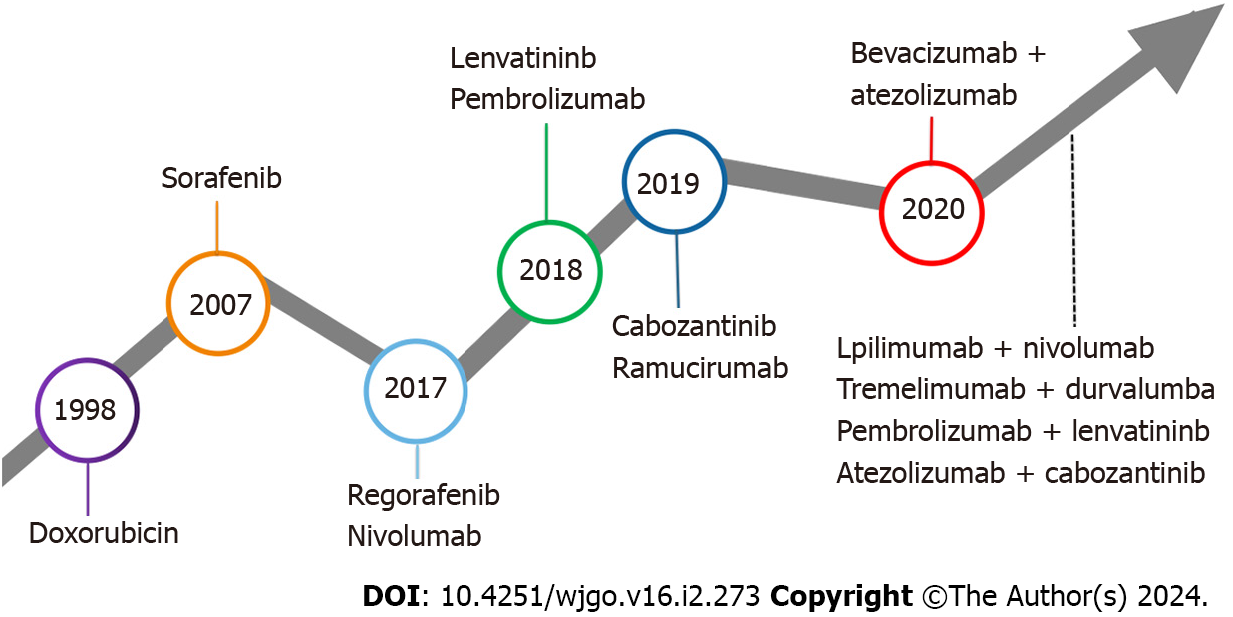
Hepatocellular carcinoma (HCC) remains among the most prevalent malignant tumors worldwide[1] and represents the primary form of liver cancer[2]. Diagnosis typically occurs during the middle to late stages for the majority of patients with HCC[3]. For patients with mid to late-stage HCC diagnosis, systemic therapy is the main treatment method[4]. The Food and Drug Administration (FDA) has undergone an extensive approval process for systematic liver cancer treat
Establishing an immuno-suppressive state within the tumor microenvironment (TME) stands as a critical capability of malignant tumors[11]. Recent advancements in comprehending the immune escape mechanisms of malignancies have led to the development of various immune drugs, yielding promising outcomes[12-15]. Immunotherapy is a systemic treatment approach that stimulates the human immune system, enhances immune response, strengthens immune cells’ capability to resist tumor cells, and overcomes tumor cells’ evasion of immune surveillance[16]. Tumor immunotherapy, a highly anticipated innovative therapy, holds broad application prospects in mid to late-stage HCC, with numerous notable clinical trials in HCC immunotherapy (Table 1).
| Study | NCT ID | Study phase | Patient population | Sample size | Intervention measures | Finding | Region |
| Checkmate 040 | NCT01658878 | I/II | Advanced HCC with or without chronic viral hepatits | 659 | Envafolimab/Pembrolizumab | ORR: 15% vs 20%; DCR: 58% vs 64%; median TTP: 3.4 months vs 4.1 months; median DOR: 17 months vs 9.9 months | Asia, Europe, United States |
| Keynote 224 | NCT02702414 | II | Advanced HCC | 156 | Pembrolizumab | ORR: 17%; DCR: 62%; mPFS: 4.9 months; mOS: 12.9% | Asia, Europe, North America |
| Reflect | NCT01761266 | Ⅲ | Unresectable HCC | 954 | Lenvatinib/Sorafenib | mOS: 13.6 months vs 12.3 months; mPFS: 7.4 months vs 3.7 months; medianTTP: 8.9 months vs 3.7 months; ORR: 24.1% vs 9.2% | Asia, Europe, North America |
| Keynote 524 | NCT03006926 | I | HCC | 104 | Pembrolizumab + Lenvatinib | ORR: 46% vs 36%; DCR: 88% vs 88%; median DOR: 8.6 months vs 12.6 months; mPFS: 9.3 months vs 8.6 months; mOS: 22 months vs 22 months | United States, France, Italy, Japan |
| Leap002 | NCT03713593 | III | Advanced HCC | 794 | Pembrolizumab + Lenvatinib/Lenvatinib | mOS: 21.1 months vs 19.0 months; mPFS: 8.2 months vs 8.0 months; ORR: 26.1% vs 17.5%; DCR: 81.3% vs 78.4% | Asia, Europe, United States |
| Study117 | NCT03418922 | Ib | HCC | 30 | Lenvatinib + Nivolumab | ORR: 37.5% vs 7.7%; DCR: 62.5% vs 69.2%; 12 mPFS: 30.0% vs 49.2%; 12 mOS: 52.1% vs 51.3% | NA |
| COSMIC-312 | NCT03755791 | III | Advanced HCC without prior systemic anticancer therapy | 837 | Cabozantinib + Atezolizumab/Cabozantinib + Sorafenib | mPFS: 6.8 months vs 4.2 months; mOS: 15.4 months vs 15.5 months; ORR: 13% vs 5%; DCR: 82% vs 63% | Asia, Europe, United States |
| RESCUE | NCT03463876 | Ⅱ | Advanced HCC | 190 | Camrelizumabfor + Apatinib | ORR: 34.3% vs 23.8%; DCR: 77.1% vs 75.8%; median DOR: 14.8 months vs NE; mPFS: 5.7 months vs 5.5 months | NA |
| Imbrave 150 | NCT03434379 | III | locally advanced or metastatic HCC | 558 | Atezolizumab + Bevacizumab/Sorafenib | ORR: 33.2% vs 13.3%; DCR: 72.3% vs 55.1%; median DOR: NE vs 6.3 months | Asia, Europe, United States |
| ORIENT-32 | NCT03794440 | II/III | Advanced HCC | 571 | Sintilimab + Bevacizumab/Sorafenib | mOS: NE vs 10.4 months; mPFS: 6.9 months vs 4.3 months; ORR: | China |
| Study 22 | NCT02519348 | Ⅱ | Advanced HCC | 332 | Tremelimumab + Durvalumab/Tremelimumab/Durvalumab | mOS: 18.7 months vs 13.6 months vs 15.1 months vs 11.3 months; DCR: 34% vs 39% vs 34% vs 31%; ORR: 24% vs 10.6% vs 7.2% vs 9.5%; median DOR: NR vs 11.17 months vs 23.95 months vs 13.21 months | Asia, United States, Italy |
| HIMALAYA | NCT03298451 | III | No prior systemic therapy for unresectable HCC | 1504 | Tremelimumab/Durvalumab | OS: 16.4 months vs 13.8 months; ORR: 20.1% vs 5.1% | Asia, Europe, United States |
| Checkmate-9DW | NCT04039607 | III | No prior systemic therapy for Advanced HCC | 732 | Nivolumab + Pembrolizumab/Sorafenib/Lenvatinib | NE | Asia, Europe, United States |
| CHANCE001 | NCT04975932 | Retrospective study | HCC | 556 | TACE + PD-L1 inhibitors + Inhibitor molecule targeted drugs/TACE | mPFS: 9.5 months vs 8.0 months; mOS: 19.2 months vs 15.7 months; ORR: 60.1% vs 32.0% | China |
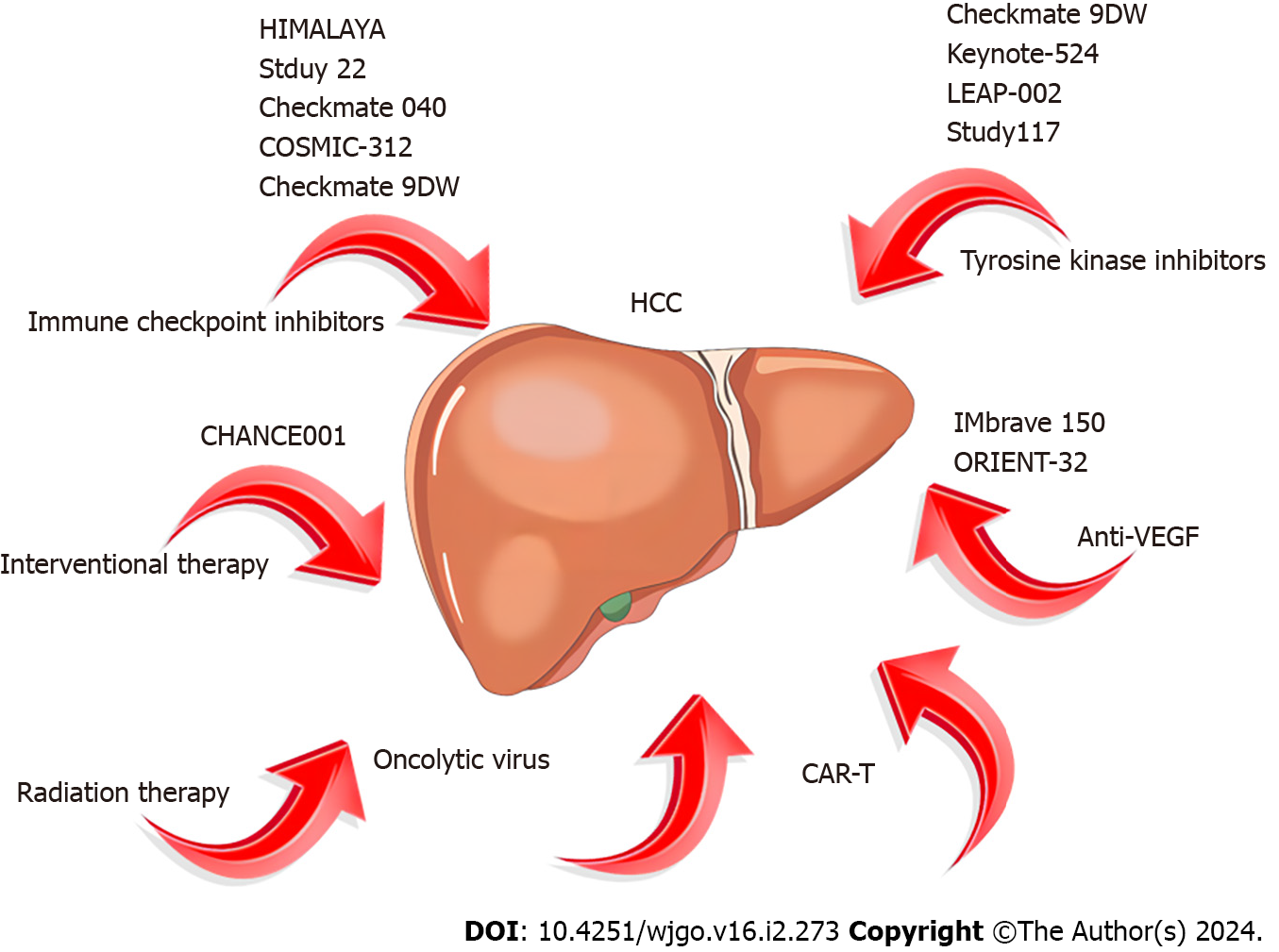
Currently, diverse immunotherapy methods exist for HCC, including immune checkpoint inhibitors (ICIs), peptide vaccines, dendritic cell vaccines, chimeric antigen receptor T cells (CAR-T), and oncolytic viruses[17]. However, ICIs have emerged as the primary research focus in HCC treatment[18]. We conducted a comprehensive review of completed ICI treatment trials for HCC in the past five years using the Clinical Trials website (ClinicalTrials.gov; Table 2).
| NCT ID | Study phase | Patient population | Intervention measures | Region |
| NCT04393220 | II | Advanced HCC | Bevacizumab + Nivolumab | China |
| NCT03785210 | II | Primary HCC or liver dominant metastatic cancer from colorectal or pancreatic cancers | Nivolumab + Tadalafil + Vancomycin | United States |
| NCT03939975 | II | Advanced HCC | Pembrolizumab/Nivolumab/JS001 | China |
| NCT04172506 | I/II | Advanced solid tumors | AK105 | China |
| NCT03419897 | II | Previously treated HCC unresectable carcinoma | Atezolizumab | China, Europe |
| NCT05471674 | II | Borderline resectable HCC | Nivolumab | China |
| NCT03980288 | I | Advanced HCC | CAR-GPC3 T Cell | China |
| NCT03841110 | I | Advanced solid tumors | FT500/FT500 + Nivolumab/FT500 + Pembrolizumab/FT500 + Atezolizumab/FT500 + IL-2 + Nivolumab/FT500 + IL-2 + Pembrolizumab/FT500 + IL-2 + Atezolizumab | United States |
| NCT04161911 | NA | Advanced HCC | Nivolumab | United States |
| NCT03735628 | I | Advanced solid tumors | Copanlisib + Nivolumab | United States, Canada |
| NCT04310709 | II | Unresectable or metastatic HCC | Regorafenib/Nivolumab | Korea |
| NCT03299946 | I | Locally advanced HCC | Cabozantinib/Nivolumab | United States |
| NCT05535998 | Retrospective study | HCC with portal vein tumor thrombus | TACE-HAIC + TKIs + PD-1 inhibitors/TACE | China |
| NCT03849469 | I | Advanced solid tumors | Pembrolizumab | United States |
| NCT03652077 | I | Advanced solid tumors | INCAGN02390 (TIM3 inhibitors) | United States |
ICIs represent monoclonal antibodies that obstruct checkpoint proteins from binding to their ligands, reactivating and maintaining the tumor immune cycle, thereby enabling T cells to eliminate tumor cells[19]. Currently, three types of ICIs, namely programmed cell death receptor 1 programmed cell death protein 1/death ligand-1 (PD-1/PD-L1) inhibitors and cytotoxic t-lymphocyte associated antigen-4 (CTLA-4) inhibitors, have garnered FDA approval for HCC treatment[20]. The data from the Checkmate 040 study phase I-II revealed an objective response rate (ORR) of 14%, a disease control rate (DCR) of 56%, and an average OS of 15.6 months in patients with advanced HCC treated with nivolumab monotherapy[9]. The keynote-224 study documented the clinical efficacy of pembrolizumab in the treatment of patients with HCC who had undergone prior sorafenib treatment, showcasing a 17% ORR and a median OS (mOS) of 13 months[21]. Following these findings, the FDA has approved pembrolizumab and nivolumab for second-line monotherapy in patients with HCC. However, despite positive outcomes from Phase I and Phase II clinical studies, subsequent Phase III randomized controlled trials for nivolumab and pembrolizumab failed to meet their primary endpoints[22-23]. Consequently, the FDA retracted the indication for nivolumab monotherapy as a second-line treatment for patients with HCC. The response rate for ICI monotherapy ranges from 15% to 23%, escalating to approximately 30% after combination therapy[24]. Although ICIs have exhibited clinical efficacy in HCC treatment, the limited response rate with monotherapy necessitates exploring combined strategies to further enhance treatment efficacy. In recent years, ICIS monotherapy and combination therapy have emerged as crucial pillars in HCC treatment (Figure 2). In this review, we explore current immunotherapy approaches and future directions.
Lenvatinib, a selective, multi-target drug [such as VEGFR 1-3, FGFR 1-4, PDGFR-α, tyrosine kinase inhibitors (TKI) of RET and KIT][25] that has gained approval from the European Drug Administration, FDA, and the National Medical Products Administration (NMPA) as a first-line treatment for advanced HCC in 2018, based on the Reflect study[26], along with lenvatinib and joining sorafenib as the standard first-line therapy. TKIs possess anti-immunosuppressive properties, wherein tumor-associated macrophages (TAM) are reduced and CD8 + T cell proliferation and activation are enhanced[27,28]. In the TME, TAMs are pivotal immune modulators implicated in tumor immune escape by producing cytokines like interleukin-10, prostaglandin E2, and transforming growth factor. Therefore, inhibiting TAM production supports immune activation[29,30].
In 2020, Finn et al[31] reported the results of a single-arm Phase Ib clinical trial (keynote-524)[31]. The trial enrolled 140 patients with advanced HCC as the study subjects, showing that pembrolizumab combined with lenvatinib as a first-line treatment regimen achieved an ORR of 36% (RECIST 1.1 standard) and 46% (mRECIST standard). Additionally, this combination significantly extended the median progressive free survival (PFS) to 9.3 months (95%CI = 5.6-9.7) and mOS to 22 months [95%CI = 20.4-not estimable (NE)]. Based on these promising findings, the LEAP-002 study[32], a randomized, controlled, double-blind large phase III trial, compared pembrolizumab combined with lenvatinib (1:1 ratio) against lenvatinib alone in 794 patients with HCC who did not undergo systemic treatment. The results indicated a significantly higher mOS (21.2 months) with pembrolizumab combined with lenvatinib compared to lenvatinib alone (19 months) [hazard ratio (HR) = 0.840, P = 0.0227], and it also significantly improved median PFS (mPFS) (8.2 months vs 8.0 months, HR = 0.867, P = 0.0466). The ORR for combination therapy was 26.1%, surpassing the monotherapy group’s 17.5%. However, while combination therapy showed trends toward improved OS and PFS, it did not achieve the predetermined statistical significance in efficacy.
Sudy117[33] assessed 30 patients with advanced HCC treated with lenvatinib combined with nivolumab. The primary endpoint of the study was tolerance and safety, while the secondary endpoint was ORR. All patients experienced AEs, predominately hand and foot syndrome (56.7%) and dysphonia (53.3%), which were manageable. The combination therapy demonstrated a remarkable ORR of 76.7% and a DCR of 96.7%, establishing its tolerability and robust anti-tumor effects.
Kelley et al[34] evaluated the efficacy and safety of cabozantinib combined with atezolizumab vs sorafenib alone in treating advanced HCC (COSMIC-312)[34]. The results revealed significantly superior mPFS in the cabiralizumab reduction (40 mg) + atezolizumab group compared to sorafenib monotherapy (6.8 months vs 4.2 months, P = 0.0012), yet mid-term analysis showed no significant difference in the mOS between the two groups (15.4 months vs 15.5 months, P = 0.44).
The combination therapy of domestic programmed cell death protein 1 (PD-1) inhibitor camrelizumab and apatinib (Rescue)[35] displayed notable efficacy in advanced HCC treatment. Compared to sorafenib alone, the combination of camrelizumab and apatinib as first-line treatment for HCC significantly prolonged both PFS and OS. Notably, the OS rate at 12 months reached approximately 75%, with a mPFS of 5.6 months and a mOS of 20.1 months. Additionally, the ORR and DCR for this combined treatment regimen were 25.4% and 78.3%, respectively. Grade 3-4 AEs were experienced by 77.4% of patients, primarily manifesting as hypertension (34.2%). On January 31, 2023, with NMPA approval, camrelizumab combined with apatinib became the first-line treatment for unresectable or metastatic liver cancer in China, establishing the "Shuang ai" combination as China’s first approved PD-1 inhibitor + small molecule anti-angiogenic drug regimen for advanced HCC.
Vasoactive endothelial growth factor (VEGF), originating from tumor cells and the surrounding matrix[36], not only facilitates tumor angiogenesis but also suppresses dendritic cell antigen-presenting function, hinders T cell activation and infiltration, and fosters regulatory T cells and myeloid suppressive cells, thereby disrupting the anti-tumor immune response[37,38].
The IMbrave150 study[39], conducted in over 70 countries, was a phase III, multicenter open-ended study. comparing the efficacy and safety of atezolizumab combined with bevacizumab (T + A scheme) vs sorafenib alone in patients with advanced HCC who had not received prior systemic treatment. Results revealed a mOS of 19.2 months with the T + A regimen and 13.4 months with sorafenib monotherapy (P < 0.001), and mPFS of 6.9 and 4.3 months, respectively (P < 0.001). The Chinese subgroup exhibited a mOS of 24.0 months. The incidence of grade 3-4 AEs in the T + A regimen (43%) was similar to sorafenib monotherapy (43%). The study also found that the ORR of the T + A combination reached 30%. Additionally, the T + A regimen significantly improved patients’ quality of life compared to sorafenib alone (HR = 0.63, 95%CI: 0.46-0.85), leading to its approval as a first-line treatment for HCC.
The ORIENT-32 phase III clinical trial[40] assessed sintilimab combined with bevacizumab vs sorafenib in first-line treatment for hepatitis B-related advanced liver cancer. The combined regimen significantly improved the mOS (NE vs 10.4 months, HR = 0.57, P < 0.0001) and mPFS (4.6 months vs 2.8 months, HR 0.56, P < 0.0001) compared to sorafenib alone. This study encompassed a Chinese population, reflecting real-world scenarios in China. Consequently, the Chinese Society of Clinical Oncology (CSCO) guideline included "sintilimab in combination with bevacizumab biosimilar" as a treatment option.
In 2022, ESMO reported on the CARES-310 study, a multicentre open phase III clinical trial[41] involving 543 patients with advanced HCC randomly assigned to apatinib + karelizumab or sorafenib treatment groups in a 1:1 ratio. The results indicated that the abatinib + karelizumab group improved mPFS (5.6 months vs 3.7 months) and increased mOS (22.1 months vs 15.2 months) compared to the sorafenib group. Consequently, CSCO has now adopted "apatinib + carilizumab" as a treatment option.
A phase Ib/II study[42] explored tivozanib combined with duvalizumab in second-line treatment for advanced HCC following initial treatment or after T + A progression. With a total of 27 enrolled patients, the median follow-up time was 13.2 months for group A and 3.4 months for group B. Data were available for 25 of the 27 patients enrolled in group A, revealing an ORR of 25% and a 1-year OS of 76%.
Ren et al[43] reported a phase II clinical study evaluated serplulimab in combination with bevacizumab biosimilar in advanced HCC across four groups. Groups A, B, and C received 3 mg/kg serplulimab with 5 mg/kg HLX04 (beva
Grade 3 or higher treatment-emergent adverse event incidence was 70.0%, 57.1% in groups A and B, respectively. Most immune-related adverse events were grade ≤ 3. Yet the overall safety of serplulimab in combination with bevacizumab was manageable. Based on several key efficacy indicators such as ORR, PFS, and OS, the first-line application of serplulimab combined with bevacizumab biosimilar appeared more beneficial than second-line use, highlighting its preference as an immunological combination regimen for patients with advanced HCC at the time of diagnosis.
Studies have found that CTLA-4 inhibitors and PD-1/PD-L1 inhibitors target different sites and timeframes within the same T cell[44]. CTLA-4 inhibitors activate T cells in lymph nodes, while PD-1 inhibitors reactivate T cells suppressed in the TME[45,46]. Combining these two agents can concurrently stimulate the same T cell, whether in the activated site (lymph node) or TME, ensuring continuous T cell stimulation over a prolonged period[47]. This combination may intensify co-stimulation, further enhancing T cell activation. Based on this theoretical foundation, various clinical studies exploring dual immune combination therapy for HCC have been conducted.
The Checkmate 040[48] study initially demonstrated the substantial treatment efficacy of nivolumab and ipilimumab, reporting a 31% ORR with a mOS of 22.8 months. It is the earliest clinical study of HCC immunotherapy and plays a pivotal role in pioneering the development of dual immunotherapy.
ASCO 2023 reported another study evaluating the efficacy of nivolumab combined with ipilimumab as a backline treatment for HCC[49]. The results showed that of the 30 evaluable patients in the cohort experiencing ICIs, 1 achieved confirmed partial response (PR) and 14 achieved stable disease (SD), with ORR of 3.3% and DCR of 50.0%. Of the 30 evaluable patients in the ICIs-nave cohort, 4 achieved confirmed PR and 10 achieved SD, with an ORR of 13.3% and DCR of 46.7%. Furthermore, the mPFS was 2.4 and 3.1 months for the ICIs- and ICIs-naïve cohorts, respectively. Although mOS was not reached in either group, these results offer hope to patients with advanced HCC.
A global, open-label, randomized controlled phase I/II trial[50] involving multiple countries enrolled 332 patients with advanced HCC across four treatment groups to evaluate the toxicity and efficacy of tremelimumab and duvalizumab combination therapy in first and second-line treatments. The tremelimumab (300 mg) plus Furvalumab (1500 mg) followed by the durvalumab (1500 mg) group exhibited the most significant benefit, achieving a mOS of 18.73 months and a 24% ORR. The dual immunotherapy group had a lower incidence of grade 3-4 treatment-related AE (37.8%) compared to the tremelimumab monotherapy group. This study suggests that the combination of tremelimumab with nivolumab significantly improves OS with good safety profiles. Subsequently, based on these favorable results, the Phase III HIMALAYA study was initiated to evaluate duvalizumab + tramelumab and sorafenib in first-line HCC treatment[51]. The study also introduced the STRIDE regimen (conventional interval administration of duvalizumab + single high initiation dose of tremelimumab). The STRIDE regimen reduced the risk of death by 22% (HR = 0.78, 95%CI = 0.65-0.93), with a mOS of 16.4 months comparable to sorafenib alone (13.8 months). In terms of ORR, the ORR of the STRIDE regimen group was three times higher than that of the sorafenib monotherapy group (20.1% vs 5.1%), the median duration of remission was up to 22.3 months, and the incidence of ≥ grade 3 treatment-related AEs was 30% lower than that of the control group, without increasing serious liver injury and bleeding events. It became the first dual immunization combined with sorafenib study to show positive OS results in the first-line treatment of HCC, leading to the National Comprehensive Cancer Network Guidelines including "duvalizumab + tremelimumab" as a treatment option (Class 1 recommendation).
Advancing research has introduced bispecific antibodies targeting both CTLA-4 and PD-1 simultaneously, potentially offering lower toxicity than dual immune drug combinations[52]. China’s first bispecific antibody drug, candonilimab (AK104)[53], showed promise in a phase II clinical trial when combined with lenvatinib for advanced HCC first-line treatment[54]. A total of 30 patients were included in the study. Among 18 patients with evaluable anti-tumor activity, the post-treatment ORR was 44.4% and DCR was 77.8%. Moreover, the mPFS has not been reached thus far.
Tebotelimab, a bispecific antibody targeting PD-1 and LAG-3, demonstrated good safety profiles and modest efficacy in a multicentre Phase I-II clinical trial[55] involving patients with treated HCC in a dose-escalation phase and a dose-expansion phase. It enrolled 30 immunotherapy-treated patients and 30 immunotherapy-naïve patients. Overall, the safety profile of the treatment was good, with a 19% incidence of treatment-related AEs at G3 and above in the dose-escalation phase, similar to that of PD-1 antibodies in general. In terms of efficacy, the ORR was 3% in immunotherapy-treated patients and 13.3% in immunotherapy-naïve patients. Thus, its efficacy appears to have a slight advantage over PD-1 antibodies.
Additionally, other ICIs beyond those targeting PD-1/PD-L1 and CTLA-4 have likewise demonstrated favorable outcomes. A phase II study[56] explored the efficacy of cobolimab (TIM-3 antibody) in combination with dostarlimab for first-line HCC treatment, exhibiting a 46% ORR (n = 13, with six patients experiencing remission)
Another phase II study[57] investigated bavituximab (phosphatidylinositide antibody) combined with pembrolizumab for first-line advanced HCC treatment, revealing a 32% ORR. This study also identified a diagnostic marker for the immune microenvironment. A total of 19 patients were monitored for marker identification, and 5 of the 8 positive patients experienced remission, while only 1 of the 11 negative patients experienced remission and 9 experienced tumor progression. Thus, such a dual immune combination regimen has notable potential in HCC.
The landscape of first-line treatment has undergone significant improvement with the approval of the T + A scheme and sindilimab + bevacizumab regimens, marking a successful blend of targeted and immune therapy in HCC treatment. Recent years have witnessed a deeper exploration of combinational therapies, shining a light on the role of interventional therapy. Various triple schemes involving targeted + immune + interventional therapies have showcased notable efficacy in treating HCC.
Transcatheter arterial chemoembolization (TACE) remains the standard treatment for mid-term HCC and has also been applied to advanced HCC[58-64]. While TACE exhibits a local anti-tumor effect and boosts the anti-tumor immune response, its induced ischemic and hypoxic conditions often trigger VEGF overexpression, ultimately leading to HCC neovascularization[65,66]. Targeted drugs not only inhibit angiogenesis, but also exert immunomodulatory effects on the TME[27,67]. Hence, the synergy between targeted, immune, and interventional therapies is evident. Cai et al[68] reported that compared to TACE combined with lenvatinib alone[68], the addition of PD-1 inhibitors significantly prolonged mOS in patients with advanced HCC (16.9 months vs 12.1 months, P = 0.009), with manageable AEs. Cao et al's study[69] observed that for unresectable HCC, the treatment regimen of TACE combined with lenvatinib and sintilimab resulted in a mOS of 23.6 months and an ORR of 46.7%[69]. Notably, the CHANCE001 study, a large sample, multicenter retrospective study, compared the clinical efficacy of TACE combined with targeted PD-1 inhibitors vs TACE alone[70]. Results demonstrated significantly longer mOS (19.2 months vs 15.7 months, P = 0.001) and PFS (9.5 months vs 8.5 months, P = 0.002) in the combination therapy group. The ORR was 60.1% for the combination therapy group compared to 32.0% for the TACE group, marking it as the largest sample size study on TACE combined with immunotherapy and targeted drug therapy for HCC.
Hepatocellular arterial infusion chemotherapy (HAIC) is another interventional method for the treatment of HCC, delivering chemotherapy drugs directly to the arterial branches related to HCC, thereby increasing local drug concentration[71]. Meta-analyses indicates superior efficacy of HAIC over TACE[72-74]. Lai et al[75] observed that after treatment with the HAIC combined with an envatinib and toripalimab regimen, patients with advanced HCC achieved a mOS of up to 17.9 months, with an ORR of 66.7%, and experienced minimal overlapping toxicities.
Bae et al[76] reported a study on local high-dose autologous Natural killer (NK) cells combined with HAIC for HCC treatment. Among the 11 enrolled patients, the ORR was 63.6% and the DCR was 81.8%. The mPFS and OS were 10.3 and 41.6 months, respectively. No compensatory losses or serious AEs occurred during HAIC, and no AEs related to NK cell infusion were noted.
Compared to invasive techniques like TACE, the integration of radiation therapy and immunotherapy stands out for its non-invasiveness, high safety profile, and easy acceptance by patients, gradually becoming a research hotspot in the comprehensive treatment of HCC. Radiation therapy excels in exposing more tumor-related antigens and releasing damage-related molecules, thus promoting an in situ vaccine effect. It also reshapes the TME, enhancing dendritic cell antigen presentation, activating CD8 + T cells, and improving cytotoxicity[77,78]. Moreover, radiation therapy also promotes the secretion of cytokines by recruiting cytotoxic T lymphocytes, thereby promoting the infiltration of immune killer cells into tumor tissue[79].
The START-FIT phase II clinical trial[80] explored the efficacy and safety of TACE + stereotactic body radiation therapy (SBRT) in combination with avelumab for HCC treatment. Results showcased a 42% CR, 24% PR, and a 67% ORR. PFS stood out at 20.7 months and OS at 30.3 months, with a local control rate of 92% at two years. Although clinical studies with avelumab in HCC are limited, the exploration continues.
The CA 209-678 study evaluating Y90 radiation embolization combined with navolizumab treatment[81] revealed an 81% regression of radiation field target lesions, achieving an ORR of 30.6% in all patients with HCC and 43.5% in those limited to intrahepatic lesions. Zhong et al[82] conducted a retrospective study on stage C patients with HCC in Barcelona, exploring the safety and clinical outcomes of a triple regimen involving radiotherapy, targeted immunotherapy, and other therapies. Sixteen patients received a treatment regimen comprising PD-1 inhibitor, anti-angiogenesis, and radiotherapy. According to the mRECIST standard, six patients experienced PR and seven exhibited SD, with no CRs observed. The ORR and DCR were 40.0% and 86.7%, respectively. Additionally, a study by Luke et al[83] assessing the safety of SBRT + pembrolizumab in patients with advanced solid tumors (including HCC) revealed a comparable adverse reaction rate between the radiation + immunotherapy regimen and radiation therapy alone, with a dose-limiting adverse reaction rate of 9.7%.
Oncolytic viruses are capable of selectively targeting and killing cancer cells and hold promise in cancer treatment. They exist in nature or can be obtained through genetic engineering[84]. Despite their potential, their therapeutic efficacy needs improvement as the body tends to swiftly eliminate these viruses[85]. Hence, trials often combine oncolytic viruses with other treatments to enhance their anti-tumor effects. The goal is to leverage the dual efficacy of oncolytic viruses and immune cells in combating cancer. This subtype of immunotherapy is widely combined with ICIs in both preclinical and clinical studies, yet application in the field of HCC remains limited, mostly in the developmental phase.
An ongoing clinical trial is recruiting patients to evaluate the efficacy and safety of SynOV 1.1 adenovirus injection combined with tislelizumab in patients with advanced HCC through a single-arm, open-label, and dose-increasing evaluation.
As molecular biology and cellular immunology advance, CAR-T therapy emerges as a novel approach in cellular immunotherapy, widely used in hematologic malignancies and solid tumors. In exploring CAR-T targets for HCC, Glypican-3, MUC-1, EpCAM, and CD147 stand out[86]. However, there is currently a lack of clinical research on the combination of CAR-T and ICIs for the treatment of mid to late-stage HCC. The future focus will remain in this direction. Furthermore, attention should be paid to the limitations of clinical trials. For example, most clinical trials overlook the etiology of HCC, and incorporating assessments of patients’ quality of life post-treatment could illuminate the negative impacts of appropriate treatment and clinical decisions. Unfortunately, the main endpoints of most trials do not include quality-of-life assessments, necessitating solutions for better evaluation (Table 3).
| Limitations | Solution |
| Most clinical trials do not take into account the etiology of HCC | Subgroup analysis according on real-world causes |
| Insufficient exploratory analysis of subgroups during safety evaluation | Further analysis will be performed based on the subject's characteristics (e.g., height, weight, underlying disease) or the subject's population (e.g., sex, age) |
| The sample size of some studies was too small and easy to shift the results | Strict adherence to the inclusion and exclusion criteria, increasing the sample size |
| Assessing QOL after treatment could help assess the negative impact and clinical decisions of appropriate treatment, but the primary endpoint of most experiments does not include QOL | The primary endpoint, in addition to tumor treatment response and imaging assessment |
Although the incidence of HCC in various age groups in China is on a downward trend[87], the immunosuppressive characteristics of HCC often yield poor treatment outcomes with single ICI regimens. With continued advances in molecular biology, several drugs have gained approval for HCC treatment (Table 4). In the era predominantly focused on anti-angiogenic drug therapy, the publication of initial CARES-310 study data[41] at ESMO 2022 brought about positive dual endpoints- PFS and OS- inspiring hope in clinics for immunotherapy combined with an anti-angiogenic small molecule TKI. The significant strides made in molecularly targeted therapy and immune checkpoint therapy for advanced HCC will benefit many patients, yet they also pose challenges in drug selection and sequencing. As these drugs eventually become available, combination therapy using targeted therapies and ICIs is expected to yield better outcomes than current treatments. Currently, various immunological combination regimens for HCC exist, but direct comparison between different clinical studies is challenging, lacking "head-to-head" trials. Thus, selecting the appropriate treatment regimen has become a new challenge. A meta-analysis[88] evaluated the clinical benefits of each first-line regimen, highlighting atirizumab in combination with bevacizumab and sindilizumab in combination with bevacizumab as having significant clinical benefits. Furthermore, the study encompassed four targeted immune combination regimen trials, indicating no significant difference in mOS among the first-line combination regimens. However, atilizumab in combination with bevacizumab, sindilizumab in combination with IBI305 (a bevacizumab analog), and karelizumab in combination with abatinib significantly prolonged patients' mPFS vs sorafenib, while tesilizumab in combination with duvarizumab vs sorafenib did not significantly prolong patient mPFS. In terms of safety, atelizumab in combination with bevacizumab exhibited a significantly lower risk of treatment-related adverse events (TRAEs) compared to karelizumab + apatinib, sindilizumab + lBl305, and sorafenib, and a similar risk to tesirizumab + doxorubicin. Additionally, carilizumab + apatinib had a significantly higher RR for greater than grade 3 TRAEs compared to atalizumab + bevacizumab. With sorafenib no longer deemed the standard control arm, future trials aim to challenge new benchmarks by exploring further treatment options for patients with HCC, utilizing first-line immunotherapy-based combinations. In conclusion, the necessity for cross-disciplinary systemic therapy for inoperable HCC will intensify in the future. Cellular immunotherapy technologies, including CAR-T cells (except for the three launched CAR-T variants), dendritic cell vaccines, NK cells, tumor infiltrating lymphocytes cells, T cell receptor-gene engineered T cells therapy, and cancer vaccines, are in clinical trial stages and are yet to gain approval for use in Chinese hospitals. The future holds numerous unexplored facets in HCC immunotherapy. First, recent studies indicate a lower immune therapy response rate in non-viral-associated liver cancer compared to viral-associated liver cancer, urging exploration into the etiological mechanisms of HCC for selecting combined immunotherapy[89,90]. Second, while alpha-fetoprotein remains a valuable biomarker for guiding treatment and prognosis prediction in HCC[11], discovering other biomarkers for efficacy prediction in screening patients with HCC could significantly benefit immunotherapy. Lastly, active research on the mechanisms underlying acquired drug resistance in immune combination regimens is necessary to guide further investigation.
| Drug target | Drug name | Drug launch time | Listed in China |
| PD-1 | Nivolumab | 2017 | No |
| PD-1 | Pembrolizumab | 2018 | Yes |
| CTLA-4 | Ipilimumab | 2020 | No |
| PD-1 | Tislelizumab | 2021 | Yes |
| PD-1 | Sintilimab | 2021 | Yes |
| PD-1 + CTLA-4 | Tremelimumab + durvalumab | 2022 | No |
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Gupta T, India; Seledtsov V, United States S-Editor: Li L L-Editor: A P-Editor: Li L
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75126] [Cited by in RCA: 64219] [Article Influence: 16054.8] [Reference Citation Analysis (174)] |
| 2. | Balogh J, Victor D 3rd, Asham EH, Burroughs SG, Boktour M, Saharia A, Li X, Ghobrial RM, Monsour HP Jr. Hepatocellular carcinoma: a review. J Hepatocell Carcinoma. 2016;3:41-53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 755] [Cited by in RCA: 806] [Article Influence: 89.6] [Reference Citation Analysis (0)] |
| 3. | Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16:589-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2184] [Cited by in RCA: 2881] [Article Influence: 480.2] [Reference Citation Analysis (17)] |
| 4. | Leyh C, Ehmer U, Roessler D, Philipp AB, Reiter FP, Jeliazkova P, Jochheim LS, Jeschke M, Hammig J, Ludwig JM, Theysohn JM, Geier A, Lange CM. Sorafenib Versus Lenvatinib-Based Sequential Systemic Therapy for Advanced Hepatocellular Carcinoma: A Real-World Analysis. Cancers (Basel). 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Shen J, Shen H, Ke L, Chen J, Dang X, Liu B, Hua Y. Knowledge Mapping of Immunotherapy for Hepatocellular Carcinoma: A Bibliometric Study. Front Immunol. 2022;13:815575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 87] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 6. | Kim DW, Talati C, Kim R. Hepatocellular carcinoma (HCC): beyond sorafenib-chemotherapy. J Gastrointest Oncol. 2017;8:256-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 243] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 7. | Abou-Alfa GK, Johnson P, Knox JJ, Capanu M, Davidenko I, Lacava J, Leung T, Gansukh B, Saltz LB. Doxorubicin plus sorafenib vs doxorubicin alone in patients with advanced hepatocellular carcinoma: a randomized trial. JAMA. 2010;304:2154-2160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 333] [Cited by in RCA: 340] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 8. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10242] [Article Influence: 602.5] [Reference Citation Analysis (2)] |
| 9. | El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, Chopra A, Anderson J, Dela Cruz C, Lang L, Neely J, Tang H, Dastani HB, Melero I. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017;389:2492-2502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3278] [Cited by in RCA: 3296] [Article Influence: 412.0] [Reference Citation Analysis (1)] |
| 10. | Nakano S, Eso Y, Okada H, Takai A, Takahashi K, Seno H. Recent Advances in Immunotherapy for Hepatocellular Carcinoma. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 11. | Laface C, Ranieri G, Maselli FM, Ambrogio F, Foti C, Ammendola M, Laterza M, Cazzato G, Memeo R, Mastrandrea G, Lioce M, Fedele P. Immunotherapy and the Combination with Targeted Therapies for Advanced Hepatocellular Carcinoma. Cancers (Basel). 2023;15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 12. | Wei Q, Taskén K. Immunoregulatory signal networks and tumor immune evasion mechanisms: insights into therapeutic targets and agents in clinical development. Biochem J. 2022;479:2219-2260. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 13. | Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, Wu X, Ma J, Zhou M, Li X, Li Y, Li G, Xiong W, Guo C, Zeng Z. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer. 2019;18:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 413] [Cited by in RCA: 986] [Article Influence: 164.3] [Reference Citation Analysis (0)] |
| 14. | Liu Y, Cao X. Immunosuppressive cells in tumor immune escape and metastasis. J Mol Med (Berl). 2016;94:509-522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 273] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 15. | Vinay DS, Ryan EP, Pawelec G, Talib WH, Stagg J, Elkord E, Lichtor T, Decker WK, Whelan RL, Kumara HMCS, Signori E, Honoki K, Georgakilas AG, Amin A, Helferich WG, Boosani CS, Guha G, Ciriolo MR, Chen S, Mohammed SI, Azmi AS, Keith WN, Bilsland A, Bhakta D, Halicka D, Fujii H, Aquilano K, Ashraf SS, Nowsheen S, Yang X, Choi BK, Kwon BS. Immune evasion in cancer: Mechanistic basis and therapeutic strategies. Semin Cancer Biol. 2015;35:S185-S198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 758] [Cited by in RCA: 1105] [Article Influence: 110.5] [Reference Citation Analysis (0)] |
| 16. | Yang Y. Cancer immunotherapy: harnessing the immune system to battle cancer. J Clin Invest. 2015;125:3335-3337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 991] [Article Influence: 99.1] [Reference Citation Analysis (0)] |
| 17. | Mizukoshi E, Kaneko S. Immune cell therapy for hepatocellular carcinoma. J Hematol Oncol. 2019;12:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 93] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 18. | Schachtschneider KM, Schwind RM, Darfour-Oduro KA, De AK, Rund LA, Singh K, Principe DR, Guzman G, Ray CE Jr, Ozer H, Gaba RC, Schook LB. A validated, transitional and translational porcine model of hepatocellular carcinoma. Oncotarget. 2017;8:63620-63634. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 19. | Suresh D, Srinivas AN, Prashant A, Harikumar KB, Kumar DP. Therapeutic options in hepatocellular carcinoma: a comprehensive review. Clin Exp Med. 2023;23:1901-1916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 20. | Abd El Aziz MA, Facciorusso A, Nayfeh T, Saadi S, Elnaggar M, Cotsoglou C, Sacco R. Immune Checkpoint Inhibitors for Unresectable Hepatocellular Carcinoma. Vaccines (Basel). 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 21. | Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, Knox J, Daniele B, Webber AL, Ebbinghaus SW, Ma J, Siegel AB, Cheng AL, Kudo M; keynote-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (keynote-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018;19:940-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1184] [Cited by in RCA: 1891] [Article Influence: 270.1] [Reference Citation Analysis (0)] |
| 22. | Yau T, Park JW, Finn RS, Cheng AL, Mathurin P, Edeline J, Kudo M, Han KH, Harding JJ, Merle P, Rosmorduc O, Wyrwicz L, Schott E, Choo SP, Kelley RK, Begic D, Chen G, Neely J, Anderson J, Sangro B. LBA38_PR- CheckMate 459: A randomized, multi-center phase III study of nivolumab (NIVO) vs sorafenib (SOR) as first-line (1L) treatment in patients (pts) with advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2019;v874-v875. [RCA] [DOI] [Full Text] [Cited by in Crossref: 426] [Cited by in RCA: 433] [Article Influence: 72.2] [Reference Citation Analysis (0)] |
| 23. | Finn RS, Ryoo BY, Merle P, Kudo M, Bouattour M, Lim HY, Breder V, Edeline J, Chao Y, Ogasawara S, Yau T, Garrido M, Chan SL, Knox J, Daniele B, Ebbinghaus SW, Chen E, Siegel AB, Zhu AX, Cheng AL; keynote-240 investigators. Pembrolizumab As Second-Line Therapy in Patients With Advanced Hepatocellular Carcinoma in keynote-240: A Randomized, Double-Blind, Phase III Trial. J Clin Oncol. 2020;38:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1365] [Cited by in RCA: 1336] [Article Influence: 267.2] [Reference Citation Analysis (0)] |
| 24. | Pinter M, Jain RK, Duda DG. The Current Landscape of Immune Checkpoint Blockade in Hepatocellular Carcinoma: A Review. JAMA Oncol. 2021;7:113-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 252] [Article Influence: 63.0] [Reference Citation Analysis (1)] |
| 25. | Sun L, Xu X, Meng F, Liu Q, Wang H, Li X, Li G, Chen F. Lenvatinib plus transarterial chemoembolization with or without immune checkpoint inhibitors for unresectable hepatocellular carcinoma: A review. Front Oncol. 2022;12:980214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 26. | Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G, Jassem J, Blanc JF, Vogel A, Komov D, Evans TRJ, Lopez C, Dutcus C, Guo M, Saito K, Kraljevic S, Tamai T, Ren M, Cheng AL. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3128] [Cited by in RCA: 3805] [Article Influence: 543.6] [Reference Citation Analysis (1)] |
| 27. | Kimura T, Kato Y, Ozawa Y, Kodama K, Ito J, Ichikawa K, Yamada K, Hori Y, Tabata K, Takase K, Matsui J, Funahashi Y, Nomoto K. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model. Cancer Sci. 2018;109:3993-4002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 258] [Article Influence: 36.9] [Reference Citation Analysis (0)] |
| 28. | Kato Y, Tabata K, Kimura T, Yachie-Kinoshita A, Ozawa Y, Yamada K, Ito J, Tachino S, Hori Y, Matsuki M, Matsuoka Y, Ghosh S, Kitano H, Nomoto K, Matsui J, Funahashi Y. Lenvatinib plus anti-PD-1 antibody combination treatment activates CD8+ T cells through reduction of tumor-associated macrophage and activation of the interferon pathway. PLoS One. 2019;14:e0212513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 242] [Cited by in RCA: 354] [Article Influence: 59.0] [Reference Citation Analysis (0)] |
| 29. | Cheng AL, Hsu C, Chan SL, Choo SP, Kudo M. Challenges of combination therapy with immune checkpoint inhibitors for hepatocellular carcinoma. J Hepatol. 2020;72:307-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 368] [Article Influence: 73.6] [Reference Citation Analysis (1)] |
| 30. | Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY, Xiao YS, Xu Y, Li YW, Tang ZY. Intratumoral balance of regulatory and cytotoxic T cells is associated with prognosis of hepatocellular carcinoma after resection. J Clin Oncol. 2007;25:2586-2593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 787] [Cited by in RCA: 891] [Article Influence: 49.5] [Reference Citation Analysis (0)] |
| 31. | Finn RS, Ikeda M, Zhu AX, Sung MW, Baron AD, Kudo M, Okusaka T, Kobayashi M, Kumada H, Kaneko S, Pracht M, Mamontov K, Meyer T, Kubota T, Dutcus CE, Saito K, Siegel AB, Dubrovsky L, Mody K, Llovet JM. Phase Ib Study of Lenvatinib Plus Pembrolizumab in Patients With Unresectable Hepatocellular Carcinoma. J Clin Oncol. 2020;38:2960-2970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 661] [Cited by in RCA: 871] [Article Influence: 174.2] [Reference Citation Analysis (0)] |
| 32. | Finn RS, Kudo M, Merle P, Meyer T, Qin S, Ikeda M, Xu R, Edeline J, Ryoo BY, Ren Z, Cheng AL, Galle PR, Kaneko S, Kumada H, Wang A, Mody K, Dubrovsky L, Siegel AB, Llovet J. LBA34 Primary results from the phase III LEAP-002 study: Lenvatinib plus pembrolizumab vs lenvatinib as first-line (1L) therapy for advanced hepatocellular carcinoma (aHCC). Ann Oncol. 2022;S1401. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 145] [Article Influence: 48.3] [Reference Citation Analysis (0)] |
| 33. | Kudo M, Ikeda M, Motomura K, Okusaka T, Kato N, Dutcus CE, Hisai T, Suzuki M, Ikezawa H, Iwata T, Kumada H, Kobayashi M. A phase Ib study of lenvatinib (LEN) plus nivolumab (NIV) in patients (pts) with unresectable hepatocellular carcinoma (uHCC): Study 117. J Clin Oncol. 2020;38:513. [RCA] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 34. | Kelley RK, Rimassa L, Cheng AL, Kaseb A, Qin S, Zhu AX, Chan SL, Melkadze T, Sukeepaisarnjaroen W, Breder V, Verset G, Gane E, Borbath I, Rangel JDG, Ryoo BY, Makharadze T, Merle P, Benzaghou F, Banerjee K, Hazra S, Fawcett J, Yau T. Cabozantinib plus atezolizumab versus sorafenib for advanced hepatocellular carcinoma (COSMIC-312): a multicentre, open-label, randomised, phase 3 trial. Lancet Oncol. 2022;23:995-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 348] [Article Influence: 116.0] [Reference Citation Analysis (1)] |
| 35. | Xu J, Shen J, Gu S, Zhang Y, Wu L, Wu J, Shao G, Xu L, Yin T, Liu J, Ren Z, Xiong J, Mao X, Zhang L, Yang J, Li L, Chen X, Wang Z, Gu K, Pan Z, Ma K, Zhou X, Yu Z, Li E, Yin G, Zhang X, Wang S, Wang Q. Camrelizumab in Combination with Apatinib in Patients with Advanced Hepatocellular Carcinoma (RESCUE): A Nonrandomized, Open-label, Phase II Trial. Clin Cancer Res. 2021;27:1003-1011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 397] [Article Influence: 99.3] [Reference Citation Analysis (0)] |
| 36. | Apte RS, Chen DS, Ferrara N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell. 2019;176:1248-1264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1864] [Cited by in RCA: 1756] [Article Influence: 292.7] [Reference Citation Analysis (0)] |
| 37. | Courau T, Nehar-Belaid D, Florez L, Levacher B, Vazquez T, Brimaud F, Bellier B, Klatzmann D. TGF-β and VEGF cooperatively control the immunotolerant tumor environment and the efficacy of cancer immunotherapies. JCI Insight. 2016;1:e85974. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 38. | Ranieri G, Marech I, Lorusso V, Goffredo V, Paradiso A, Ribatti D, Gadaleta CD. Molecular targeting agents associated with transarterial chemoembolization or radiofrequency ablation in hepatocarcinoma treatment. World J Gastroenterol. 2014;20:486-497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 17] [Cited by in RCA: 21] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 39. | Finn RS, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Kudo M, Breder V, Merle P, Kaseb AO, Li D, Verret W, Xu DZ, Hernandez S, Liu J, Huang C, Mulla S, Wang Y, Lim HY, Zhu AX, Cheng AL; IMbrave150 Investigators. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med. 2020;382:1894-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2542] [Cited by in RCA: 4645] [Article Influence: 929.0] [Reference Citation Analysis (2)] |
| 40. | Ren Z, Xu J, Bai Y, Xu A, Cang S, Du C, Li Q, Lu Y, Chen Y, Guo Y, Chen Z, Liu B, Jia W, Wu J, Wang J, Shao G, Zhang B, Shan Y, Meng Z, Gu S, Yang W, Liu C, Shi X, Gao Z, Yin T, Cui J, Huang M, Xing B, Mao Y, Teng G, Qin Y, Xia F, Yin G, Yang Y, Chen M, Wang Y, Zhou H, Fan J; ORIENT-32 study group. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2-3 study. Lancet Oncol. 2021;22:977-990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 663] [Article Influence: 165.8] [Reference Citation Analysis (1)] |
| 41. | Qin S, Chan LS, Gu S, Bai Y, Ren Z, Lin X, Chen Z, Jia W, Jin Y, Guo Y, Sultanbaev AV, Pazgan-Simon M, Pisetska M, Liang X, Chen C, Nie Z, Wang L, Cheng AL, Kaseb A, Vogel A. LBA35 Camrelizumb(C) plus rivoceranib (R) vs sorafenib (S) as first-line therapy for unresectable hepatocellular carcinoma (uHCC): a randomized, phase III trial. Ann Oncol. 2022;33:S1401-S1402. [RCA] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 72] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 42. | Li D, Dayyani F, Mahalingam D, Rowe J, Abrams T, Kasturi V, Iyer R. Final results of a phase 1b/2 study of tivozanib in combination with durvalumab in patients with advanced hepatocellular carcinoma in both previously untreated patients and patients progressing on atezolizumab and bevacizumab. Journal of Clinical Oncology. 2023;41:585-585. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 43. | Ren Z, Shao G, Shen J, Zhang L, Zhu X, Fang W, Sun G, Bai Y, Wu J, Liu L, Yuan Y, Zhang J, Li Z, Yin T, Hou X, Wang Q, Zhu J, Fan J. Phase 2 Study of the PD-1 Inhibitor Serplulimab plus the Bevacizumab Biosimilar HLX04 in Patients with Previously Treated Advanced Hepatocellular Carcinoma. Liver Cancer. 2023;2:116-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 44. | Alden SL, Lim M, Kao C, Shu D, Singal AG, Noonan A, Griffith P, Baretti M, Ho WJ, Kamel I, Yarchoan M, Hsiehchen D. Salvage Ipilimumab plus Nivolumab after Anti-PD-1/PD-L1 Therapy in Advanced Hepatocellular Carcinoma. Cancer Res Commun. 2023;3:1312-1317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 45. | Wei SC, Levine JH, Cogdill AP, Zhao Y, Anang NAS, Andrews MC, Sharma P, Wang J, Wargo JA, Pe'er D, Allison JP. Distinct Cellular Mechanisms Underlie Anti-CTLA-4 and Anti-PD-1 Checkpoint Blockade. Cell. 2017;170:1120-1133.e17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 700] [Cited by in RCA: 975] [Article Influence: 121.9] [Reference Citation Analysis (0)] |
| 46. | Parry RV, Chemnitz JM, Frauwirth KA, Lanfranco AR, Braunstein I, Kobayashi SV, Linsley PS, Thompson CB, Riley JL. CTLA-4 and PD-1 receptors inhibit T-cell activation by distinct mechanisms. Mol Cell Biol. 2005;25:9543-9553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1248] [Cited by in RCA: 1487] [Article Influence: 74.4] [Reference Citation Analysis (0)] |
| 47. | Han Y, Chen Z, Yang Y, Jiang Z, Gu Y, Liu Y, Lin C, Pan Z, Yu Y, Jiang M, Zhou W, Cao X. Human CD14+ CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2,3-dioxygenase production in hepatocellular carcinoma. Hepatology. 2014;59:567-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 184] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 48. | Zhou G, Sprengers D, Boor PPC, Doukas M, Schutz H, Mancham S, Pedroza-Gonzalez A, Polak WG, de Jonge J, Gaspersz M, Dong H, Thielemans K, Pan Q, IJzermans JNM, Bruno MJ, Kwekkeboom J. Antibodies Against Immune Checkpoint Molecules Restore Functions of Tumor-Infiltrating T Cells in Hepatocellular Carcinomas. Gastroenterology. 2017;153:1107-1119.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 318] [Article Influence: 39.8] [Reference Citation Analysis (0)] |
| 49. | Yau T, Kang YK, Kim TY, El-Khoueiry AB, Santoro A, Sangro B, Melero I, Kudo M, Hou MM, Matilla A, Tovoli F, Knox JJ, Ruth He A, El-Rayes BF, Acosta-Rivera M, Lim HY, Neely J, Shen Y, Wisniewski T, Anderson J, Hsu C. Efficacy and Safety of Nivolumab Plus Ipilimumab in Patients With Advanced Hepatocellular Carcinoma Previously Treated With Sorafenib: The CheckMate 040 Randomized Clinical Trial. JAMA Oncol. 2020;6:e204564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 908] [Cited by in RCA: 959] [Article Influence: 191.8] [Reference Citation Analysis (0)] |
| 50. | Kelley RK, Sangro B, Harris W, Ikeda M, Okusaka T, Kang YK, Qin S, Tai DW, Lim HY, Yau T, Yong WP, Cheng AL, Gasbarrini A, Damian S, Bruix J, Borad M, Bendell J, Kim TY, Standifer N, He P, Makowsky M, Negro A, Kudo M, Abou-Alfa GK. Safety, Efficacy, and Pharmacodynamics of Tremelimumab Plus Durvalumab for Patients With Unresectable Hepatocellular Carcinoma: Randomized Expansion of a Phase I/II Study. J Clin Oncol. 2021;39:2991-3001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 325] [Article Influence: 81.3] [Reference Citation Analysis (0)] |
| 51. | Rimassa L, Finn RS, Sangro B. Combination immunotherapy for hepatocellular carcinoma. J Hepatol. 2023;79:506-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 164] [Reference Citation Analysis (0)] |
| 52. | Wei L, Tian Y, Chen X, Guo X, Chen C, Zheng Y, Xu J, Ye X. Data mining and analysis for emicizumab adverse event signals based on the Food and Drug Administration Adverse Event Reporting System database. Int J Clin Pharm. 2023;45:622-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 53. | Peng J, Zhu Q, Peng Z, Chen Z, Liu Y, Liu B. Patients with positive HER-2 amplification advanced gastroesophageal junction cancer achieved complete response with combined chemotherapy of AK104/cadonilimab (PD-1/CTLA-4 bispecific): A case report. Front Immunol. 2022;13:1049518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 54. | Pang X, Huang Z, Zhong T, Zhang P, Wang ZM, Xia M, Li B. Cadonilimab, a tetravalent PD-1/CTLA-4 bispecific antibody with trans-binding and enhanced target binding avidity. MAbs. 2023;15:2180794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 79] [Article Influence: 39.5] [Reference Citation Analysis (0)] |
| 55. | Ren Z, Guo Y, Bai Y, Ying J, Meng Z, Chen Z, Gu S, Zhang J, Liang J, Hou X, Li W, Xu A, Hao C, Xing R, Zhang X, Zhang D, Chan S. Tebotelimab, a pD-1/LAG-3 bispecific antibody, in patients with advanced hepatocellular carcinoma who had failed prior targeted therapy and/or immunotherapy: An open-label, single-arm, phase 1/2 dose-escalation and expansion study. Journal of Clinical Oncology. 2023;41:578-578. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 56. | Acoba J, Rho Y, Fukaya E. Phase iI study of cobolimab in combination with dostarlimab for the treatment of advanced hepatocellular carcinoma. Journal of Clinical Oncology. 2023;41:580-580. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 57. | Hsieh D, Kainthla R, Zhu H, Beg M. Phase 2 trial of pembrolizumab (pembro) and bavituximab (bavi) in advanced hepatocellular carcinoma (HCC). Journal of Clinical Oncology. 2023;41:584-584. [RCA] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 58. | Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020-1022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5972] [Cited by in RCA: 6565] [Article Influence: 468.9] [Reference Citation Analysis (1)] |
| 59. | Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, Gores G. Hepatocellular carcinoma. Nat Rev Dis Primers. 2016;2:16018. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1933] [Cited by in RCA: 1865] [Article Influence: 207.2] [Reference Citation Analysis (4)] |
| 60. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol. 2018;69:182-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5593] [Cited by in RCA: 6019] [Article Influence: 859.9] [Reference Citation Analysis (3)] |
| 61. | Marrero JA, Kulik LM, Sirlin CB, Zhu AX, Finn RS, Abecassis MM, Roberts LR, Heimbach JK. Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology. 2018;68:723-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2121] [Cited by in RCA: 3222] [Article Influence: 460.3] [Reference Citation Analysis (1)] |
| 62. | Llovet JM, Villanueva A, Marrero JA, Schwartz M, Meyer T, Galle PR, Lencioni R, Greten TF, Kudo M, Mandrekar SJ, Zhu AX, Finn RS, Roberts LR; AASLD Panel of Experts on Trial Design in HCC. Trial Design and Endpoints in Hepatocellular Carcinoma: AASLD Consensus Conference. Hepatology. 2021;73:158-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 274] [Article Influence: 68.5] [Reference Citation Analysis (0)] |
| 63. | Park JW, Chen M, Colombo M, Roberts LR, Schwartz M, Chen PJ, Kudo M, Johnson P, Wagner S, Orsini LS, Sherman M. Global patterns of hepatocellular carcinoma management from diagnosis to death: the BRIDGE Study. Liver Int. 2015;35:2155-2166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 569] [Cited by in RCA: 934] [Article Influence: 93.4] [Reference Citation Analysis (0)] |
| 64. | Zhou J, Sun H, Wang Z, Cong W, Wang J, Zeng M, Zhou W, Bie P, Liu L, Wen T, Han G, Wang M, Liu R, Lu L, Ren Z, Chen M, Zeng Z, Liang P, Liang C, Yan F, Wang W, Ji Y, Yun J, Cai D, Chen Y, Cheng W, Cheng S, Dai C, Guo W, Hua B, Huang X, Jia W, Li Y, Liang J, Liu T, Lv G, Mao Y, Peng T, Ren W, Shi H, Shi G, Tao K, Wang X, Xiang B, Xing B, Xu J, Yang J, Yang Y, Ye S, Yin Z, Zhang B, Zhang L, Zhang S, Zhang T, Zhao Y, Zheng H, Zhu J, Zhu K, Shi Y, Xiao Y, Dai Z, Teng G, Cai J, Cai X, Li Q, Shen F, Qin S, Dong J, Fan J. Guidelines for the Diagnosis and Treatment of Hepatocellular Carcinoma (2019 Edition). Liver Cancer. 2020;9:682-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 410] [Cited by in RCA: 568] [Article Influence: 113.6] [Reference Citation Analysis (1)] |
| 65. | Chang Y, Jeong SW, Young Jang J, Jae Kim Y. Recent Updates of Transarterial Chemoembolilzation in Hepatocellular Carcinoma. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 200] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 66. | Cheu JW, Wong CC. Mechanistic Rationales Guiding Combination Hepatocellular Carcinoma Therapies Involving Immune Checkpoint Inhibitors. Hepatology. 2021;74:2264-2276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 67. | Yi C, Chen L, Lin Z, Liu L, Shao W, Zhang R, Lin J, Zhang J, Zhu W, Jia H, Qin L, Lu L, Chen J. Lenvatinib Targets FGF Receptor 4 to Enhance Antitumor Immune Response of Anti-Programmed Cell Death-1 in HCC. Hepatology. 2021;74:2544-2560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 212] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 68. | Cai M, Huang W, Huang J, Shi W, Guo Y, Liang L, Zhou J, Lin L, Cao B, Chen Y, Zhu K. Transarterial Chemoembolization Combined With Lenvatinib Plus PD-1 Inhibitor for Advanced Hepatocellular Carcinoma: A Retrospective Cohort Study. Front Immunol. 2022;13:848387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 121] [Article Influence: 40.3] [Reference Citation Analysis (0)] |
| 69. | Cao F, Yang Y, Si T, Luo J, Zeng H, Zhang Z, Feng D, Chen Y, Zheng J. The Efficacy of TACE Combined With Lenvatinib Plus Sintilimab in Unresectable Hepatocellular Carcinoma: A Multicenter Retrospective Study. Front Oncol. 2021;11:783480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 68] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 70. | Zhu HD, Li HL, Huang MS, Yang WZ, Yin GW, Zhong BY, Sun JH, Jin ZC, Chen JJ, Ge NJ, Ding WB, Li WH, Huang JH, Mu W, Gu SZ, Li JP, Zhao H, Wen SW, Lei YM, Song YS, Yuan CW, Wang WD, Huang M, Zhao W, Wu JB, Wang S, Zhu X, Han JJ, Ren WX, Lu ZM, Xing WG, Fan Y, Lin HL, Zhang ZS, Xu GH, Hu WH, Tu Q, Su HY, Zheng CS, Chen Y, Zhao XY, Fang ZT, Wang Q, Zhao JW, Xu AB, Xu J, Wu QH, Niu HZ, Wang J, Dai F, Feng DP, Li QD, Shi RS, Li JR, Yang G, Shi HB, Ji JS, Liu YE, Cai Z, Yang P, Zhao Y, Zhu XL, Lu LG, Teng GJ; CHANCE001 Investigators. Transarterial chemoembolization with PD-(L)1 inhibitors plus molecular targeted therapies for hepatocellular carcinoma (CHANCE001). Signal Transduct Target Ther. 2023;8:58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 148] [Article Influence: 74.0] [Reference Citation Analysis (1)] |
| 71. | Bartkowski R, Berger MR, Aguiar JL, Henne TH, Dörsam J, Geelhaar GH, Schlag P, Herfarth C. Experiments on the efficacy and toxicity of locoregional chemotherapy of liver tumors with 5-fluoro-2'-deoxyuridine (FUDR) and 5-fluorouracil (5-FU) in an animal model. J Cancer Res Clin Oncol. 1986;111:42-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 45] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 72. | Liu H, Qin X, Jiang H, Sun C, Wu M, Xu Z, Lu T, Ma X, Han Z. Comparison of Hepatic Arterial Infusion Chemotherapy and Transarterial Chemoembolization for Advanced Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. J Gastrointestin Liver Dis. 2022;31:336-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Reference Citation Analysis (0)] |
| 73. | Li S, Xu J, Zhang H, Hong J, Si Y, Yang T, He Y, Ng DM, Zheng D. The Role of Hepatic Arterial Infusion Chemotherapy in the Treatment of Hepatocellular Carcinoma: A Systematic Review and Meta-Analysis. Chemotherapy. 2021;66:124-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 74. | Liu J, Zhang J, Wang Y, Shu G, Lou C, Du Z. HAIC versus TACE for patients with unresectable hepatocellular carcinoma: A systematic review and meta-analysis. Medicine (Baltimore). 2022;101:e32390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 75. | Lai Z, He M, Bu X, Xu Y, Huang Y, Wen D, Li Q, Xu L, Zhang Y, Wei W, Chen M, Kan A, Shi M. Lenvatinib, toripalimab plus hepatic arterial infusion chemotherapy in patients with high-risk advanced hepatocellular carcinoma: A biomolecular exploratory, phase II trial. Eur J Cancer. 2022;174:68-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 110] [Reference Citation Analysis (0)] |
| 76. | Bae WK, Lee BC, Kim HJ, Lee JJ, Chung IJ, Cho SB, Koh YS. A Phase I Study of Locoregional High-Dose Autologous Natural Killer Cell Therapy With Hepatic Arterial Infusion Chemotherapy in Patients With Locally Advanced Hepatocellular Carcinoma. Front Immunol. 2022;13:879452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 77. | Bernstein MB, Krishnan S, Hodge JW, Chang JY. Immunotherapy and stereotactic ablative radiotherapy (ISABR): a curative approach? Nat Rev Clin Oncol. 2016;13:516-524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 303] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 78. | Jiang W, Chan CK, Weissman IL, Kim BYS, Hahn SM. Immune Priming of the Tumor Microenvironment by Radiation. Trends Cancer. 2016;2:638-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 130] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 79. | Chiang CL, Chan ACY, Chiu KWH, Kong FS. Combined Stereotactic Body Radiotherapy and Checkpoint Inhibition in Unresectable Hepatocellular Carcinoma: A Potential Synergistic Treatment Strategy. Front Oncol. 2019;9:1157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 84] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 80. | Chiang CL, Chiu KWH, Chan KSK, Lee FAS, Li JCB, Wan CWS, Dai WC, Lam TC, Chen W, Wong NSM, Cheung ALY, Lee VWY, Lau VWH, El Helali A, Man K, Kong FMS, Lo CM, Chan AC. Sequential transarterial chemoembolisation and stereotactic body radiotherapy followed by immunotherapy as conversion therapy for patients with locally advanced, unresectable hepatocellular carcinoma (START-FIT): a single-arm, phase 2 trial. Lancet Gastroenterol Hepatol. 2023;8:169-178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 67] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 81. | Tai D, Loke K, Gogna A, Kaya NA, Tan SH, Hennedige T, Ng D, Irani F, Lee J, Lim JQ, Too CW, Ng MCH, Tham CK, Lam J, Koo SL, Chong HS, Goh GB, Huang HL, Venkatanarasimha N, Lo R, Chow PKH, Goh BKP, Chung A, Toh HC, Thng CH, Lim TKH, Yeong J, Zhai W, Chan CY, Choo SP. Radioembolisation with Y90-resin microspheres followed by nivolumab for advanced hepatocellular carcinoma (CA 209-678): a single arm, single centre, phase 2 trial. Lancet Gastroenterol Hepatol. 2021;6:1025-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 82. | Zhong L, Wu D, Peng W, Sheng H, Xiao Y, Zhang X, Wang Y. Safety of PD-1/PD-L1 Inhibitors Combined With Palliative Radiotherapy and Anti-Angiogenic Therapy in Advanced Hepatocellular Carcinoma. Front Oncol. 2021;11:686621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 83. | Luke JJ, Lemons JM, Karrison TG, Pitroda SP, Melotek JM, Zha Y, Al-Hallaq HA, Arina A, Khodarev NN, Janisch L, Chang P, Patel JD, Fleming GF, Moroney J, Sharma MR, White JR, Ratain MJ, Gajewski TF, Weichselbaum RR, Chmura SJ. Safety and Clinical Activity of Pembrolizumab and Multisite Stereotactic Body Radiotherapy in Patients With Advanced Solid Tumors. J Clin Oncol. 2018;36:1611-1618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 456] [Article Influence: 65.1] [Reference Citation Analysis (0)] |
| 84. | Mondal M, Guo J, He P, Zhou D. Recent advances of oncolytic virus in cancer therapy. Hum Vaccin Immunother. 2020;16:2389-2402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 85. | Raja J, Ludwig JM, Gettinger SN, Schalper KA, Kim HS. Oncolytic virus immunotherapy: future prospects for oncology. J Immunother Cancer. 2018;6:140. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 218] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 86. | Zhang R, Zhang Z, Liu Z, Wei D, Wu X, Bian H, Chen Z. Adoptive cell transfer therapy for hepatocellular carcinoma. Front Med. 2019;13:3-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 46] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 87. | Zheng R, Qu C, Zhang S, Zeng H, Sun K, Gu X, Xia C, Yang Z, Li H, Wei W, Chen W, He J. Liver cancer incidence and mortality in China: Temporal trends and projections to 2030. Chin J Cancer Res. 2018;30:571-579. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 241] [Article Influence: 34.4] [Reference Citation Analysis (1)] |
| 88. | Cappuyns S, Corbett V, Yarchoan M, Finn RS, Llovet JM. Critical Appraisal of Guideline Recommendations on Systemic Therapies for Advanced Hepatocellular Carcinoma: A Review. JAMA Oncol. 2023;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 82] [Cited by in RCA: 69] [Article Influence: 69.0] [Reference Citation Analysis (0)] |
| 89. | Pfister D, Núñez NG, Pinyol R, Govaere O, Pinter M, Szydlowska M, Gupta R, Qiu M, Deczkowska A, Weiner A, Müller F, Sinha A, Friebel E, Engleitner T, Lenggenhager D, Moncsek A, Heide D, Stirm K, Kosla J, Kotsiliti E, Leone V, Dudek M, Yousuf S, Inverso D, Singh I, Teijeiro A, Castet F, Montironi C, Haber PK, Tiniakos D, Bedossa P, Cockell S, Younes R, Vacca M, Marra F, Schattenberg JM, Allison M, Bugianesi E, Ratziu V, Pressiani T, D'Alessio A, Personeni N, Rimassa L, Daly AK, Scheiner B, Pomej K, Kirstein MM, Vogel A, Peck-Radosavljevic M, Hucke F, Finkelmeier F, Waidmann O, Trojan J, Schulze K, Wege H, Koch S, Weinmann A, Bueter M, Rössler F, Siebenhüner A, De Dosso S, Mallm JP, Umansky V, Jugold M, Luedde T, Schietinger A, Schirmacher P, Emu B, Augustin HG, Billeter A, Müller-Stich B, Kikuchi H, Duda DG, Kütting F, Waldschmidt DT, Ebert MP, Rahbari N, Mei HE, Schulz AR, Ringelhan M, Malek N, Spahn S, Bitzer M, Ruiz de Galarreta M, Lujambio A, Dufour JF, Marron TU, Kaseb A, Kudo M, Huang YH, Djouder N, Wolter K, Zender L, Marche PN, Decaens T, Pinato DJ, Rad R, Mertens JC, Weber A, Unger K, Meissner F, Roth S, Jilkova ZM, Claassen M, Anstee QM, Amit I, Knolle P, Becher B, Llovet JM, Heikenwalder M. NASH limits anti-tumour surveillance in immunotherapy-treated HCC. Nature. 2021;592:450-456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 832] [Article Influence: 208.0] [Reference Citation Analysis (1)] |
| 90. | Ji F, Nguyen MH. Cabozantinib plus atezolizumab in advanced hepatocellular carcinoma and the role of adjuvant antiviral therapy. Lancet Oncol. 2022;23:962-963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |