Published online Feb 15, 2023. doi: 10.4251/wjgo.v15.i2.251
Peer-review started: September 24, 2022
First decision: October 20, 2022
Revised: November 3, 2022
Accepted: January 10, 2023
Article in press: January 10, 2023
Published online: February 15, 2023
Processing time: 143 Days and 23 Hours
Colorectal cancer (CRC) is among the most prevalent and deadly neoplasms worldwide. According to GLOBOCAN predictions, its incidence will increase from 1.15 million CRC cases in 2020 to 1.92 million cases in 2040. Therefore, a better understanding of the mechanisms involved in CRC development is necessary to improve strategies focused on reducing the incidence, prevalence, and mortality of this oncological pathology. Surgery, chemotherapy, and radio
Core Tip: This review focused on the drugs that may modulate the immune response and show specific efficacy in the treatment of colorectal cancer. We then presented the evidence that immunosuppression or promotion of the immune response is the answer to generating effective treatments with combined chemotherapeutic drugs.
- Citation: Olguin JE, Mendoza-Rodriguez MG, Sanchez-Barrera CA, Terrazas LI. Is the combination of immunotherapy with conventional chemotherapy the key to increase the efficacy of colorectal cancer treatment? World J Gastrointest Oncol 2023; 15(2): 251-267
- URL: https://www.wjgnet.com/1948-5204/full/v15/i2/251.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v15.i2.251
The origin of colorectal cancer (CRC) is heterogeneous. The general classification of CRC is divided into inherited, sporadic, and intestinal bowel diseases. Inherited CRC, which represents approximately 5% of all CRC cases, includes either the presence or absence of colonic polyps, such as Lynch syndrome and serrated polyposis syndrome[1]. Sporadic CRC (approximately 70% of CRC cases) is sustained by environmental and modifiable risk factors, including stress, diet, and age. Sporadic CRC has a monoclonal origin and is characterized by mutation accumulation in oncogenes and tumor suppressor genes. The second pathway of CRC includes the traditional APC-KRAS pathway and the microsatellite instability group, both having an essential role in clinical studies[2]. The third pathway includes intestinal chronic inflammatory diseases, such as Crohn’s disease and ulcerative colitis, which could result in colitis-associated colon cancer[3].
From a biological perspective, this evidence demonstrates that the origin of CRC is diverse. The response to therapies is not always homogeneous in patients. The best treatment should be based on the tumor’s unique characteristics. Effective treatments need to be broad and involve chemical and immunological molecules. The context of the broad causes of CRC development is highly involved in the low effectiveness of either single chemotherapeutics or classical immunotherapy by checkpoints inhibitor (ICI) administration during this oncological pathology. An in-depth and more precise description of the CRC origin and development, including a role for both immune response and inflammation, can be found in[4,5].
CRC is one of the deadliest diseases in the world. Despite advances in diagnosis, treatment strategies remain an essential bottleneck affecting survival, in which the pathological stage represents the most important prognostic factor for patients with CRC. The accurate classification of lesions is the primary tool to decide the most appropriate treatment and therapy[6]. The treatment for early-stage CRC (stage I and stage II) currently consists of resecting the tumor area with regional lymph nodes, which has a 5-year disease-free survival rate of 95%[7]. In the advanced stage of the disease (stages III and above), the rate of disease-free survival drops from 90% to 50% for surgery alone, requiring the administration of chemotherapeutics, and only 17%-20% of these patients ultimately survive[8,9].
5-Fluorouracil (5-FU) has been central in treating advanced CRC since 1957. Unfortunately, the response rate to 5-FU as the first-line chemotherapy in advanced CRC is still only 10%-15%. In contrast, 5-FU combined with other anticancer drugs as adjuvants, such as leucovorin and oxaliplatin (FOLFOX) or leucovorin calcium and irinotecan, increases the effectiveness of 5-FU by 50%[10,11].
5-FU is the third most commonly used chemotherapeutic agent for the treatment of solid malignancies worldwide[12]. Heidelberger synthesized it in the early 1950s as a derivate of fluoro
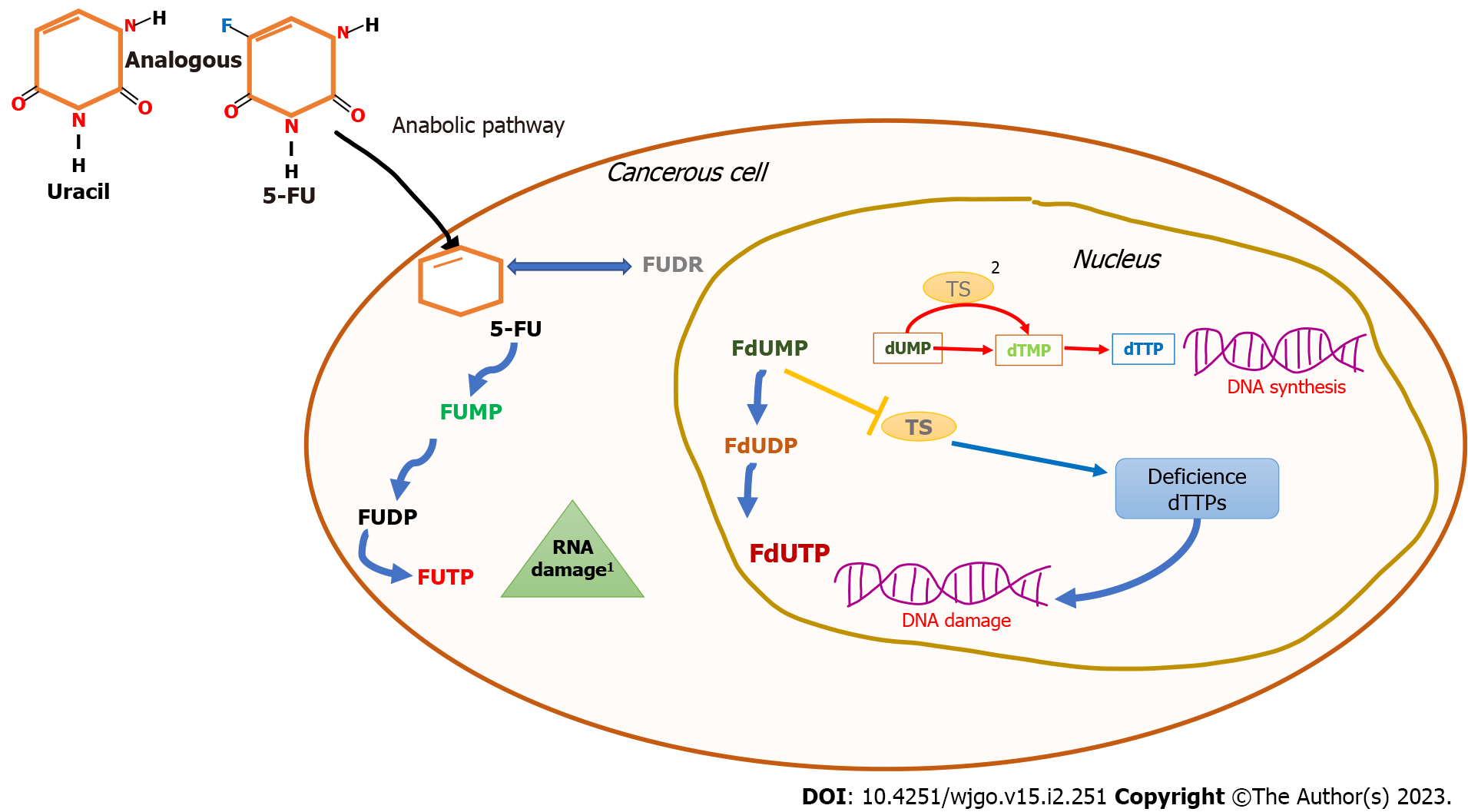
In intravenous administration, 5-FU is incorporated rapidly into the cells through facilitated transport as uracil[15]. Subsequently, its metabolism can be driven in two ways, i.e., via anabolic and catabolic routes, which compete with each other. In sensitive cancerous cells, the anabolic pathway leads to the conversion of this drug into several active metabolites, such as fluorodeoxyuridine monophosphate, fluorodeoxyuridine triphosphate, and fluorouridine triphosphate[16]. The active metabolites interfere with nucleoside metabolism and can be incorporated into RNA and DNA, leading to cytotoxicity and cell death[17,18]. This mechanism is due to its similar structure to pyrimidine, molecules of DNA and RNA, an analog of uracil with a fluorine atom at the C-5 position in place of hydrogen[19]. Fluorodeoxyuridine monophosphate disrupts the function of thymidylate synthase, a key enzyme responsible for providing deoxynucleotide triphosphates, which are necessary for DNA replication and repair, catalyzing the reaction of deoxyuridine monophosphate to deoxythymidine monophosphate synthesis[14,15]. An insufficiency in deoxythymidine monophosphate leads to the depletion of deoxythymidine triphosphate, which perturbs the levels of the other deoxynucleotide triphosphates[16-20] (Figure 1).
5-FU has primarily been used in the treatment of solid cancers of digestive origin, such as colorectal, anal, pancreatic, esophageal, gastric, and ampullary tumors, and less frequently in breast, cervical, and head and neck cancers[21-23]. CRC treatment includes various chemotherapeutic drugs. As the backbone of treatments, 5-FU has been used for more than five decades, and more recently, it has been combined with other chemotherapeutic drugs to potentiate its anticarcinogenic effect[22,24].
The use of alternative broad-spectrum chemotherapeutics in addition to 5-FU has been proposed for colon cancer treatment. Doxorubicin treatment combined with other drugs, such as metformin and sodium oxamate, reduces the proliferation rate of colon cancer cell lines in vitro[25]. However, the use of doxorubicin in patients is limited by the side effects frequently associated with this drug, such as hepatotoxicity, nephrotoxicity, pulmotoxicity, and cardiotoxicity[26,27]. Additionally, doxorubicin can lead to chemoresistance in tumor cells through nuclear factor kappa B translocation to the nucleus and DNA binding because of the damage induced by this drug, triggering the expression of antiapoptotic genes[28].
In CRC, nuclear factor kappa B nuclear translocation is a characteristic in more than 70% of patients, limiting the use of doxorubicin[29]. Another disadvantage of this drug is its anthracycline nature since it is extracted from Streptomyces spp. Cancer cells frequently show rapid resistance to naturally occurring cancer drugs, diminishing their effectiveness, whereas they are more sensitive to antimetabolites, such as 5-FU and cisplatin, among others[30]. The use of other chemotherapeutics, such as tamoxifen, which is highly effective and frequently used in breast cancer treatment, has an adverse effect in the treatment of CRC[31]. Due to the molecular characteristics of each type of cancer, the successful use of tamoxifen in breast cancer lies in its mechanism of action. Approximately 80% of all breast cancers are positive for the estrogen receptor, and tamoxifen inhibits the expression of estrogen-regulated genes by the competitive inhibition of this receptor. Different reports indicate that tamoxifen has the opposite effect on CRC, increasing the risk of developing this type of cancer[31,32].
CRC is a molecularly heterogeneous disease in which genetics and cellular events accumulate to endow tumor cells with aggressive characteristics, including chemotherapy resistance. Chromosomal instability, mismatch repair, and methylator phenotype are the three major pathways involved in acquiring tumorigenesis and a malignant phenotype and could be present in sporadic and inherited CRC[33,34]. The choice of better therapy is based on cancer-related features and patient-related factors, such as the number and localization of metastases, tumor progression, presence or absence of biochemical markers, and comorbidity[35-37]. Despite all these characteristics, treatment based on the antineoplastic effects of 5-FU is the cornerstone of therapy in advanced CRC stages.
Treatment with 5-FU in combination with other drugs, such as oxaliplatin (OXA), irinotecan, capecitabine, bevacizumab, cetuximab, panitumumab, ziv-aflibercept, regorafenib, and ramucirumab, increases its effectiveness and has been approved by the Food and Drug Administration for the management of CRC[38]. Thus, during stages III or IV in resected CRC patients, the use of combination treatment, such as FOLFOX or 5-FU, leucovorin calcium, and irinotecan, is common as a first-line treatment. This strategy significantly increases the survival rate of these patients[39]. The initial chemotherapy scheme and the decision on better combinatory drugs depends on multiple conditions in the patients. In metastatic CRC limited to the liver or lung, surgery and the rapid initiation of chemotherapy appears to be the best option. When CRC cure is not possible, three additional scenarios can arise: (1) Patients with advanced tumors and symptoms require rapid tumor shrinkage to provide palliation, which begins with chemotherapy; (2) Asymptomatic patients with bulky tumor and possible rapid progression are likely to become symptomatic in a short period; and (3) Patients without symptoms but disseminated disease who never had resectable disease but whose tumors remain non-bulky are likely to remain asymptomatic for an extended period. In the last two scenarios, the initiation of chemotherapy can be discussed[22,40]. Previous work in the Nordic population demonstrated that early treatment with 5-FU plus leucovorin in asymptomatic patients with advanced CRC prolonged survival and delayed both disease progression and the onset of symptoms[41]. In another study in Australasian and Canadian populations of asymptomatic patients using the same chemotherapy regimen, no difference was reported between early or delayed chemotherapy use until symptoms appeared[42]. Thus, clinical treatment requires a medical discussion and the patient´s preference when cure is not possible. The spectrum of molecular alterations that offer alternative management for this disease could be explored.
Alterations in genes related to survival, angiogenesis, proliferation, and apoptosis incorporate additional strategies into CRC treatment. The RAS, KRAS, and NRAS genes play an essential role as prognostic and predictive indicators in CRC treatment[43-45]. Mutations in the DNA at position 12 in the KRAS protein are significantly associated with a poor prognosis: a 5-year survival rate of approximately 3%[46]. Patients with this mutation are not candidates to receive treatment with monoclonal antibodies, such as cetuximab or panitumumab, which target the epidermal growth factor receptor (EGFR)[47-50]. Blocking EGFR represents the second line of treatment in patients with wild-type RAS together with the backbone 5-FU, leucovorin calcium, and irinotecan therapy. Therefore, the patient’s genetic and tumor-specific factors need to be considered when choosing chemotherapeutic and combination schemes to avoid resistance and undesired responses to these therapies.
Drug resistance and consequent therapy failure are the main problems clinicians face in treating different neoplasms, which limit the quality of life and long-term remission rates as a consequence of tumor growth and spreading leading to 90% of patients dying[51,52]. Drug resistance is a highly complex process that is commonly classified into two types: intrinsic and acquired. Both types of drug resistance lead to the regulation of molecular mechanisms of chemoresistance, such as the activation of transporter pumps, oncogenes, tumor suppressor genes, mitochondrial alteration, DNA repair, autophagy, epithelial-mesenchymal transition, cancer stemness, and exosomes[53,54].
In the intrinsic phenotype, diverse alterations existing before drug administration in the patient complicate the selection of chemotherapy. The inherent genetic mutations in tumors, such as a KRAS mutation in exon 2 in codon 12 or 13, are the most frequent mutations associated with poor prognosis and drug resistance in CRC[55,56]. Therapies based on the first line of treatment using FOLFOX or 5-FU, leucovorin calcium, and irinotecan plus cetuximab or panitumumab (anti-EGFR) are ineffective in patients with KRAS mutations[57]. Recent studies indicate that mutations in genes related to the pathways that regulate tumor cell survival and proliferation and inhibit apoptosis in tumor cells, such as AKT1 and CTNNB1, contribute to 5-FU chemotherapeutic resistance in CRC. The CTNNB1 gene encodes the β-catenin protein, which plays a crucial role in cancer by activating the Wnt/β-catenin signaling pathway. This pathway is associated with tumorigenesis and CRC resistance, and it upregulates genes and proteins, such as multidrug resistance gene (MDR1) and inhibitor of apoptosis (Bcl2), to induce epithelial-mesenchymal transition and regulate the tumor microenvironment (TME)[58-60].
The heterogeneity of tumors, including CRC, consists of heterogeneity in cancer and infiltrated resident host cells, extracellular matrix, and immune and inflammatory cells, such as macrophages, dendritic cells, myeloid-derived suppressor cells, T cells, mast cells, and natural killer cells. These components comprise the TME, which has a dynamic composition[61]. It is well known that one of the main functions of the TME is to provide a protective function for tumor cells, inducing crosstalk between immune and nonimmune cells that leads to tumor-mediated immunosuppression, supporting tumor growth and survival[62]. Recent reports indicate that the TME in CRC contributes to cancer progression and drug resistance through high interstitial pressure, fibrosis, and the degradation of the therapeutic agent by enzymatic activity and inducing immunosuppression[61,63,64]. These findings indicate that the regulation of immune cells surrounding the tumor has a critical role in the response to therapies for CRC. Thus, chemotherapy and immunotherapies targeting the recovery activity of immune cells are likely necessary to fight CRC.
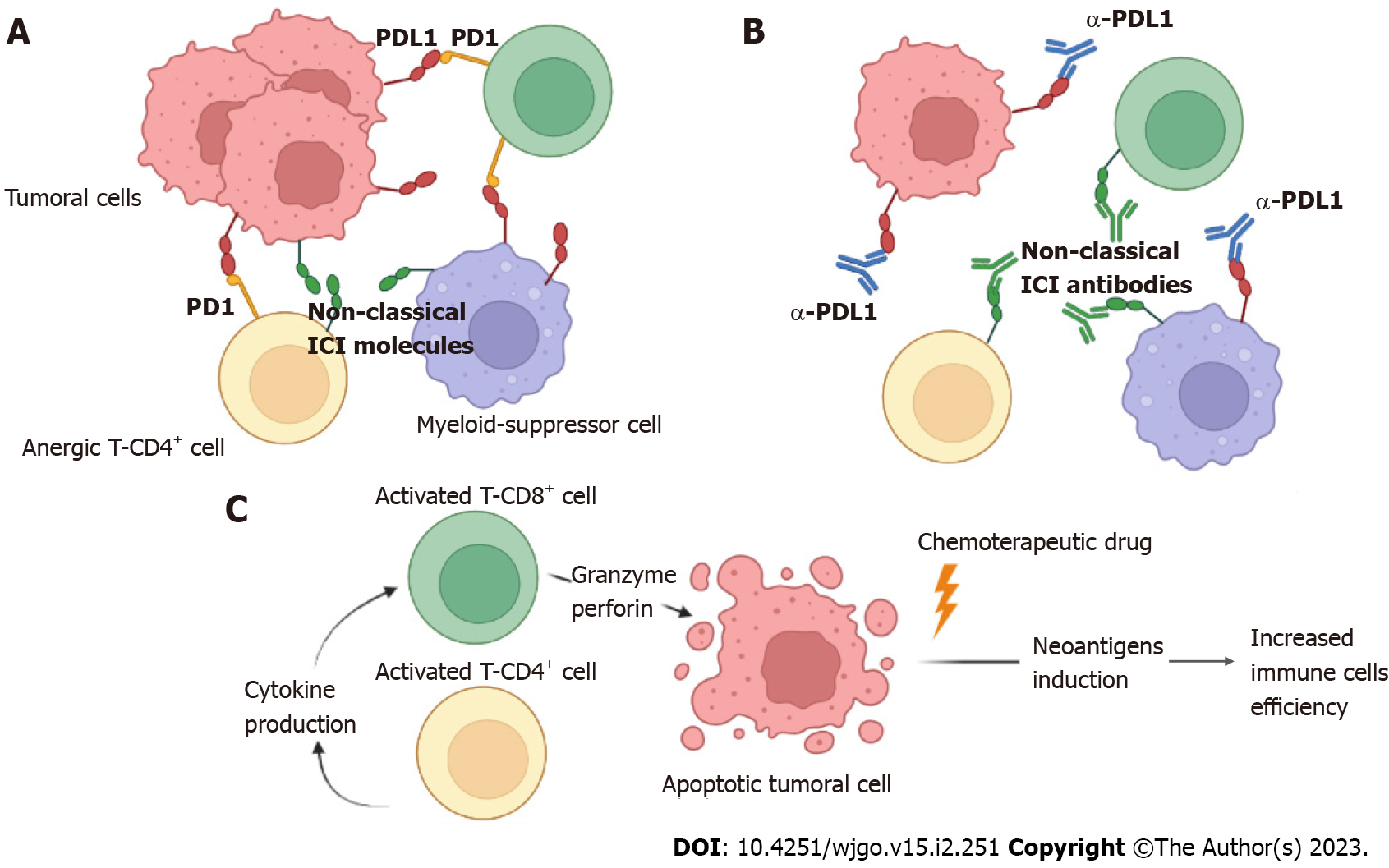
The study of the effect of chemotherapeutic drugs on immune cells is controversial. The central concept here is that chemotherapy reduces the capacity of the immune system to function, but how could a drug affect the capacity and efficiency of the immune response to induce an efficient post-treatment response? Perhaps the “original” concept has a flawed approach. Evidence suggests that after 5-FU treatment in a mouse model, bone marrow cellularity decreases, but platelets and thrombopoietin, which are close to the immune response, rebound[64,65]. Similarly, the serum of patients diagnosed with stage III/IV CRC who had received FOLFOX chemotherapy showed increased levels of heat shock protein 70, which belongs to the damage-associated molecular patterns recognized by innate receptors[66]. Later, in vitro studies showed that the supernatants of dying CRC cells treated with OXA and 5-FU induced a mature phenotype in dendritic cells coexpressing HLA-DR, CD80, and CD86 and producing interleukin-1β, tumor necrosis factor-α, and MIP-1α in a TLR-4-dependent manner[66]. These results strongly suggested that OXA/5-FU treatment induced the activation of the innate immune response during CRC. Additionally, increased numbers of myeloid-derived suppressor cells have been reported in a mouse model of thymoma, and treatment with 5-FU combined with gemcitabine selectively induced apoptosis in myeloid-derived suppressor cells. Consequently, increased antigen-specific CD8+ T cells produced more interferon-γ, generating a T cell antitumor response (Figure 2)[66].
Conversely, high levels of the chemokine CCL20 recruit regulatory T (Treg) cells in CRC patients resistant to FOLFOX[67]. However, in blood samples from metastatic FOLFOX-sensitive CRC patients, a reduced percentage of Foxp3+ Treg cells was recorded after treatment[68]. Therefore, the increase in Treg cells is associated with 5-FU chemoresistance. Thus, evidence of 5-FU chemotherapy suggests a role in the specific and direct reduction of suppressive immune cells during CRC. Additionally, the increased apoptosis-induced death of tumor cells by 5-FU could increase the ability of immune cells to recognize damage-associated molecular patterns released by these dying tumor cells, inducing protective inflammation. Evidence needs to be accumulated in this field to clarify whether chemotherapy may be an inducer of immune cell activation (Figure 3).
The effectiveness of ICI as an immunotherapy treatment has been evaluated in the last decade. The efficacy of these agents is evident in liquid tumors, such as melanoma, leukemia, and solid non-small cell lung carcinoma. Classical ICIs used to treat these oncological pathologies are anti-programmed cell death 1 (PD1), anti-programmed cell death ligand 1 (PDL1), and anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA4) monoclonal antibodies, where anti-CTLA4 has a lower clinical efficacy[68,69] (Table 1).
| Antibody name | Isotype | Target molecule | Effectiveness1 as monotherapy | Effectiveness as combined ICI | Effectiveness as ICI + chemotherapy | Ref. |
| Ipilimumab | IgG1 | CTLA4 | Yes | Well tolerated in combination with nivolumab | No | Suzuki et al[80], 2021; Lenz et al[86], 2022; Cohen et al[90], 2020 |
| Tremelimumab | IgG2 | CTLA4 | No | Yes, durvalumab improved OS and increased lymphoid response | Combined with durvalumab + fluoropyrimidines, oxaliplatin, irinotecan, showed increased OS | Chung et al[77], 2010; Kanikarla Marie et al[85], 2021; Chen et al[102], 2020 |
| Nivolumab | IgG4 | PD1 | Well tolerated | Well tolerated in combination with low ipilimumab dose, with increased OS | Yes | Overman et al[78], 2017; Kawazoe et al[79], 2021; Lenz et al[86], 2022; Morse et al[87], 2019 |
| Pembrolizumab | IgG4 | PD1 | Well tolerated, increased OS | No | There is no evidence | Haag et al[82], 2022 |
| Atezolizumab | IgG1 | PDL1 | There is no evidence | There is no evidence | Safe when combined with cobimetinib, having no effect on OS. Combined with FOLFOX and bevacizumab showed increased progression-free survival, but adverse events were shown | Eng et al[91], 2019; Antoniotti et al[94], 2022 |
| Avelumab | IgG1 | PDL1 | Increased OS but adverse events were shown | There is no evidence | Combined with cetuximab showed increased T cell killing | Haag et al[82], 2022; Stein et al[97], 2021 |
| Durvalumab | IgG1 | PDL1 | Increased progression-free survival, but adverse events were shown | There is no evidence | Safe when combined with MEKi, having no effect on OS | Oh et al[84], 2022 |
Diverse reports suggest single or combined therapy using different antibodies targeted against the same or another molecule. In melanoma therapy, nivolumab, a human immunoglobulin G4 anti-PD1 pathway monoclonal antibody, results in an overall survival (OS) rate of 72.9%, whereas the antineoplastic chemotherapeutic dacarbazine resulted in a survival rate of 42.1%[69]. In metastatic melanoma refractory to chemotherapy, treatment with ipilimumab, an anti-CTLA4 antibody, shows high efficacy when combined with anti-PD1 antibodies (either pembrolizumab or nivolumab)[70]. Additionally, nivolumab and ipilimumab combination treatment prolongs progression-free survival, mainly in patients with tumors testing positive for PDL1 expression[71]. Although in vitro studies suggested that atezolizumab, avelumab, and durvalumab, all anti-PDL1 antibodies, more effectively block PD1/PDL1 signaling (Figure 2)[72], evidence of the use of an anti-PDL1 antibody as a single approved treatment without immunotherapy or chemotherapy combination is insufficient to conclude their role in inducing protection in metastatic melanoma[73]. Immunotherapy with the anti-PDL1 antibody atezolizumab has been approved to treat non-small cell lung carcinoma combined with chemotherapy, and anti-PDL1 has been approved to treat triple-negative breast cancer[74,75]. Therefore, increased PDL1 expression in the TME of melanoma patients is an efficient marker to predict response to anti-PD1 treatments, which must be applied in all types of cancer when this immunotherapy is suggested[76].
Notwithstanding the successful use of immunotherapy in the neoplasms mentioned above, few reports show an influential role for classical monoclonal ICI using anti-CTLA4 and anti-PD1/PDL1 axis antibodies in CRC. Most clinical assays were disappointing. For example, tremelimumab, a human immunoglobulin G2 anti-CTLA4 antibody, did not produce clinically meaningful results when it was used as a monotherapy in patients with refractory metastatic CRC[77] (Table 1).
Another monotherapy treatment using nivolumab in CRC patients with microsatellite instability and FOLFOX chemoresistance showed excellent control of the disease, and patients tolerated this treatment well[78]. CRC patients treated with nivolumab combined with an oral heat shock protein 90 inhibitor showed safety profiles and antitumor activity associated with reduced activity of Treg cells and better response of tumor-infiltrating lymphocytes[79]. Interestingly, a report of a woman treated with nivolumab for melanoma with no hereditary CRC background developed colon carcinoma after 7 years of anti-melanoma treatment. The medical service then decided to switch the treatment to ipilimumab, and after four cycles of monotherapy, the colon tumor was in complete remission[80]. Pembrolizumab used as monotherapy in either microsatellite instability-high or mismatch repair-deficient CRC patients produced improvements in health-related quality of life compared to patients treated with leucovorin, 5-FU, and OXA[81]. In one study of refractory mismatch repair, CRC patients treated with pembrolizumab as monotherapy plus maraviroc, an agonist of CCR5 that promotes the activation and recruitment of macrophages inducing immune cell infiltrate in tumors, showed a beneficial toxicity pattern. The OS was higher than expected[82].
ICI with avelumab monotherapy in unresectable metastatic CRC patients who failed FOLFOX chemotherapy showed an OS of 72.2% at 8.1 mo, which was similar to the effect of ICI monotherapy using either pembrolizumab or nivolumab. However, some patients showed treatment-related adverse events[83]. Durvalumab used as monotherapy in microsatellite-instability high/mismatch repair-deficient metastatic CRC patients whose disease had progressed after chemotherapy showed efficiency and a satisfactory progression-free survival of 58.2%; however, side effects were found in 36.4% of patients[84].
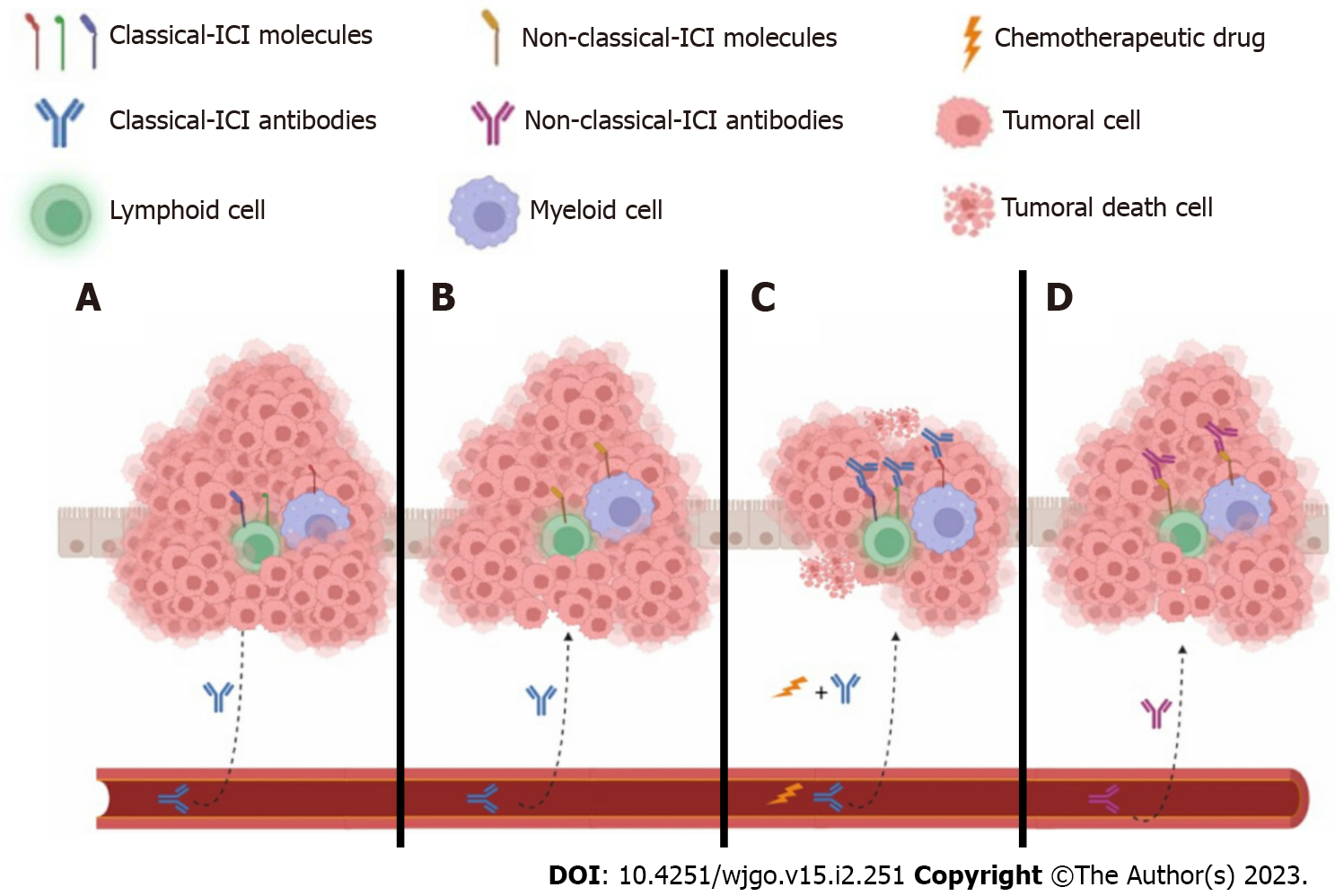
Taken together, these results suggest that contrary to anti-CTLA4, ICI monotherapy blocking the PD1/PDL1 axis has a better effect in high microsatellite instability/mismatch repair-deficient metastatic CRC patients who previously displayed chemoresistance. Additionally, combining ICI PD1/PDL1 monotherapy with either antibodies or immune cell stimulators improves treatment efficacy. However, only a small number of clinical trials show increased OS (Table 1). Most likely, the TME reduces the access of ICI antibodies to the target molecules expressed in either immune or epithelial cells (Figure 3).
Little evidence of the apparent effect of ICI monotherapy on CRC development is available. Conversely, increasing evidence suggests a better outcome using combined ICI, i.e., the use of two monoclonal antibodies targeting CTLA4 or the PD1/PDL1 axis. To improve the immune response, combination treatment with tremelimumab and durmalumab was used in patients for the preoperative management of resectable CRC and liver metastases. These patients improved their OS to 24.5 mo; interestingly, their CD4+, CD8+, and B cells displayed an activated profile[85]. Additionally, first-line treatment consisting of nivolumab plus low-dose ipilimumab treatment in patients with microsatellite instability and metastatic CRC without previous chemotherapy showed that this combination was well tolerated at the primary endpoint, with robust and durable clinical benefit. However, the study is ongoing, and OS data are not yet available[86]. Previous ICI studies using similar inclusion criteria and antibody doses showed that follow-up over 12 mo of combined treatment resulted in an 85% OS[87]. Recently, the combined use of ipilimumab plus nivolumab before surgery in either mismatch repair-deficient or mismatch repair-proficient CRC patients induced an antitumoral response associated with a lack of signs of cancer after surgery with increased infiltration of CD8+PD1+ cells; the authors suggested that this combined ICI therapy may be the standard treatment for mismatch repair-deficient CRC patients[21].
Additionally, circulating tumor DNA detection in the blood of patients with durable and ongoing responses to ipilimumab plus nivolumab could be used as a monitoring response and dynamic marker of this combined ICI treatment[88,89]. Finally, a study attempted to analyze whether pseudo progression was observed in mismatch repair-deficient CRC patients treated with nivolumab plus ipilimumab, showing that this treatment rarely induces and confirming a high disease control rate of 86%[90]. Taken together, this evidence strongly suggests that the combined ICI blockade of CTLA4-PD1/PDL1 in mismatch repair-deficient CRC patients is highly successful. However, these patients represent only a tiny fraction of all CRC-diagnosed patients. Therefore, more clinical trials must be developed to obtain sufficient evidence to conclude the positive effects of the classic ICI combination.
The combined use of atezolizumab plus aobimetinib, a MAPK/ERK kinase 1 and 2 inhibitor that increases CD8+ cell infiltration in tumors of patients with microsatellite-stable metastatic CRC, showed no improvement in OS and was consistent with the safety of using both drugs[91] (Table 1). A similar effect showing only acceptable tolerance to this treatment was observed with durmalumab plus inhibitor of MAPK/ERK kinase in microsatellite-stable metastatic CRC patients[89]. No improved OS was observed using atezolizumab combined with cobimetinib in metastatic CRC patients[92]. A multicenter phase I/II study in June 2017 aimed to analyze the role of durmalumab plus tremelimumab combined with FOLFOX chemotherapy in patients with metastatic CRC, expecting a 6-mo progression-free survival of over 70.7%; however, the authors do not have the final results to date[93].
Recently, the combination of atezolizumab with FOLFOX chemotherapy plus bevacizumab (monoclonal anti-vascular endothelial growth factor antibody) in mismatch repair metastatic CRC patients induced a median progression-free survival of 13 mo, while FOLFOX plus bevacizumab alone resulted in 11 mo of progression-free survival, suggesting that the addition of atezolizumab improves progression-free survival. However, 42% of patients showed neutropenia, and 27% displayed severe adverse events[94]. The same treatment consisting of atezolizumab combined with FOLFOX plus bevacizumab was used in patients with untreated unresectable metastatic CRC, showing no improvement in OS and safety signals[95]. The use of the anti-EGFR antibody cetuximab plus avelumab for treating wild-type RAS metastatic CRC patients was safe; the authors suggested that an analysis of circulating DNA in plasma could be an indicator of the positive effects of this treatment. However, the data are insufficient to show the impact on the OS rate[96].
Treatment with cetuximab plus the ICI avelumab in microsatellite stable metastatic CRC patients showed that subclones of tumors expressing PDL1 mutations mediated the resistance to direct avelumab antitumor effects but also increased T cell killing[97]. An analysis of the neutrophil-to-lymphocyte ratio in the blood of chemorefractory metastatic CRC patients treated with cetuximab plus avelumab showed that a high neutrophil-to-lymphocyte ratio was a poor prognostic factor. Thus, the neutrophil-to-lymphocyte ratio could also be a predictor for the effectiveness of the combined ICI cetuximab plus avelumab[98].
Regorafenib, an inhibitor of protein kinases in tumor angiogenesis used in combination with avelumab in microsatellite stable CRC patients, showed increased infiltration of CD8+ T cells associated with better outcomes, with an OS of 10.8 mo[99]. Metastatic CRC patients who previously received two radiotherapies and who were treated with durvalumab plus tremelimumab before the third round of radiotherapy showed increased circulating, differentiated, and proliferating CD8+ T cells, but the authors concluded that this finding does not meet the prespecified endpoint criteria to consider this combined ICI plus radiotherapy worthwhile for further study; specifically, the authors suggested an objective response rate of at least 25%, but they only obtained a response rate of 8.3%[100].
The addition of FOLFOX-based chemotherapy to avelumab plus an adenovirus vector vaccine capable of inducing a CD4+/CD8+ T cell response in mismatch repair-deficient microsatellite instability-high metastatic CRC patients showed no improvement in progression-free survival[101]. However, The Canadian Cancer Trials Group suggests that combining tremelimumab and durmalumab to treat patients with high microsatellite instability who had previously received chemotherapy (fluoro
It is essential to mention that some research about the combination of chemotherapy and ICI is under development[93]. For example, a phase II trial in 2020 will show whether atezolizumab combined with OXA, radiotherapy, and bevacizumab may increase progression-free survival in microsatellite instability CRC patients[103]. Additionally, in microsatellite instability-high metastatic CRC patients with deficient mismatch repair, a study is currently underway to prove the improvement of disease-free survival by ICI with avelumab plus fluoropyrimidine; the authors suggested that this ICI plus chemotherapy treatment would improve the expected 3-year disease-free survival rate by 12%[104]. Evidence showing that combining ICI with chemotherapy improves treatment efficacy continues to accumulate.
Most clinical trials reported here are recent, and perhaps the evidence is insufficient to conclude that a treatment criterion has already been established. Consequently, evidence supports the hypothesis that the use of classical ICIs improves chemotherapy treatment, mainly in CRC patients with high microsatellite instability. It is crucial for patients who do not have a good prognosis with chemotherapy alone to have a better response with the combination of classical ICI antibodies.
In recent years, mechanisms have begun to be developed to understand and explain why, in some cases, classical immunotherapy is sufficient to generate benefits in some patients. Multiple factors participate in the development of any pathology, such as the patient’s clinical history, genetics, and the ability of their immune cells to act during cancer. We emphasize the importance of the recruitment of immune cells to the TME; in the case of melanoma, non-small cell lung cancer, and leukemia, the capacity of the immune cells to access is greater, which together with ICIs increases the bioavailability of monoclonal antibodies to find the antigens expressed in the required enclosures to be detected and removed or blocked[105]. In contrast, other types of oncological pathologies are available when access to the tumor site is more difficult for both immune cells and classical ICI antibodies, such as CRC[106]. In addition, the causes of CRC are multiple, and we cannot attempt to generalize a unique treatment for all varieties of CRC to reduce the statistics of this oncological pathology on the rise.
The single-cell multiomics technique has shed light on the complexity of the individual immune response elicited against any agent. This approach facilitates the individual characterization of groups of cells by identifying the gene transcripts at a specific time[107]. This technique depends on the efficiency of flow cytometry to distinguish and separate individual cells, the equipment used to amplify mRNA transcripts and synthesize complementary strand DNA, and sequencing equipment, allowing for robust data with high precision and certainty[108]. The advancement of these latest-generation technologies allows not only the expression of the classic ICI (CTLA4, PD1/PDL1) to be distinguished but also the characterization of mRNA transcripts in specific immune or epithelial cell populations at a particular time. These transcripts, already expressed as proteins, can individually be proposed as new nonclassical ICIs in patients[109], generating a wide range of therapeutic targets that, as in the case of CRC, increase the efficiency of previous treatments.
Some surface molecules involved in suppressive functions for activated T cells, Treg cells, macrophages, neutrophils, and epithelial cells have been proposed as immunotherapy targets. Glucocorticoid-induced tumor necrosis factor receptor-related protein (GITR), T cell immunoglobulin and mucin domain 3 (TIM3), LAG3, CD39, CD73, CD47, and SIRP-1α (a do not eat me signal)[110] warrant evaluation as monotherapy or combined therapy in CRC.
TIM3 is an overexpressed inhibitory receptor in active immune cells, including myeloid and lymphoid cells, with suppressive and modulatory characteristics. It is relevant in reducing interferon-γ production by helper T type 1 cells after binding to its ligand, galectin-9[111]. TIM3 is also overexpressed by Treg cells in a colitis-associated colon cancer mouse model[112]. The combination of TIM3 with anti-PD1 ICI antibodies is a good prospect in a murine breast cancer model[113]. Sabatolimab, an anti-TIM3 antibody, has already been used as a treatment in ovarian, CRC, and non-small cell lung cancer combined with an anti-PD1 antibody, being well tolerated and improving antitumor activity[114]. GITR has a role in the immunomodulation of effector T cells and increases tumor resistance[113,115]. Treatment with an anti-GITR antibody combined with pembrolizumab improves the disease control rate compared with anti-GITR used as monotherapy for treating CRC, melanoma, and adrenal carcinoma[116].
Lymphocyte activation gene-3 (LAG3 or CD223) has a structure similar to that of the CD4 molecule, joining major histocompatibility complex II in antigen-presenting cells. However, two of their immunoglobulin-like domains can bind receptors in tumor cells[81]. Blockage of LAG3 induces increased interleukin-2 production and enhances T cell proliferation[117]. Therefore, LAG3 has been proposed as an ICI; the genes LAG3 and IDO1 were shown to be overexpressed in a phase II study of pembrolizumab use as ICI monotherapy in patients with esophageal squamous cell carcinoma. The authors suggested that a combination of ICIs is needed to induce immunity against this tumor[118]. CD73 is an extracellular adenosine receptor expressed in immunosuppressant cells (such as Treg cells), favoring tumor progression[119].
Recently, single-cell RNA sequencing in a colitis-associated CRC murine model showed that an anti-CD73 antibody has a significant role in improving the anticancer functions of Treg cells, and exhausted CD8+ T cells became activated CD8+ T cells. In contrast, anti-PD1 antibodies in the same model depleted Treg cells and M2 macrophages[120], suggesting a synergistic role for the new ICI anti-CD73 that may improve the positive effects of anti-PD1 monotherapy. Ex vivo samples of blood and tumors from microsatellite instability CRC patients showed that atezolizumab alone could reactivate T cells.
Furthermore, adding tiragolumab, an anti-TIGIT antibody, restored intraepithelial CD4 T and CD8 T cell function by favoring interferon-γ and tumor necrosis factor-α production[120,121]. TIGIT is a receptor upregulated in natural killer and activated T cells when the modulation of their effector abilities is necessary for the microenvironment, such as cancer. It is also overexpressed in Treg cells[122]. The use of avelumab plus the adoptive transfer of autologous dendritic cell vaccine in chemotherapy-treated mismatch repair-proficient metastatic CRC patients had a successful result because this treatment was well tolerated. Furthermore, treatment was terminated early because 11% of patients were disease free at 6 mo, and progression-free survival was increased by 40%[123]. This evidence also suggests that new ICI research could open other possibilities for specific and beneficial treatment for CRC patients because either immune or epithelial cells may express nonclassical ICI molecules (Figure 2).
Chemotherapy using 5-FU remains the primary treatment for CRC, despite its high toxicity and low efficacy. New strategies targeting ICIs have been useful in some oncological pathologies; however, evidence showing the effectiveness of classical ICI monotherapy in CRC is scarce. A combination of classical ICI antibodies targeting CTLA4 and PD1/PDL1 molecules showed stronger efficacy for CRC treatment. Finally, classical ICI plus conventional chemotherapy is effective, as evidenced by increased OS, but these strategies are not yet well established, and some clinical studies are ongoing. Evidence suggests that chemotherapy produces neoantigens, increasing tumor immunogenicity that may activate immune responses[124]. This increased immunogenicity is likely the reason for a better response when classical ICI plus chemotherapy is used and may represent a pathway to design new therapeutic strategies aimed at improving the response in CRC patients based on immunological reactivation combined with conventional chemotherapy. Knowledge of the TME in CRC is essential to understand immunosuppression. New options for nonclassical ICIs obtained by single-cell sequencing are shedding light on this area and will probably improve the effectiveness of many treatments.
We are possibly on the verge of major findings in the study of CRC, where the immune response will continue playing a leading role and where new proposals with nonclassical ICIs may reduce the current statistics and poor prognoses for this oncological pathology.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: Mexico
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Tantau AI, Romania; Wismayer R, Uganda S-Editor: Gao CC L-Editor: Filipodia P-Editor: Gao CC
| 1. | Øines M, Helsingen LM, Bretthauer M, Emilsson L. Epidemiology and risk factors of colorectal polyps. Best Pract Res Clin Gastroenterol. 2017;31:419-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 92] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 2. | Yamagishi H, Kuroda H, Imai Y, Hiraishi H. Molecular pathogenesis of sporadic colorectal cancers. Chin J Cancer. 2016;35:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 111] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 3. | Shah SC, Itzkowitz SH. Colorectal Cancer in Inflammatory Bowel Disease: Mechanisms and Management. Gastroenterology. 2022;162:715-730.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 436] [Article Influence: 145.3] [Reference Citation Analysis (0)] |
| 4. | Medina-Andrade I, Olguín JE, Rodriguez T, Terrazas LI. Chapter 7- The inflammation during colorectal cancer: A friend or a foe? In: Morales-Montor J, Segovia-Mendoza M. Breaking Tolerance to Anti-Cancer Cell-Mediated Immunotherapy in Resistant Cancer: From the Lab Bench Work to Its Clinical Perspectives. Academic Press, 2021: 103-129. |
| 5. | Morales-Montor J. The Innate Immune System in Health and Disease: From the Lab Bench Work to Its Clinical Implications. Volume 1. Nova Science Publishers Incorporated, 2022. |
| 6. | Wolpin BM, Mayer RJ. Systemic treatment of colorectal cancer. Gastroenterology. 2008;134:1296-1310. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 369] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 7. | Osterman E, Glimelius B. Recurrence Risk After Up-to-Date Colon Cancer Staging, Surgery, and Pathology: Analysis of the Entire Swedish Population. Dis Colon Rectum. 2018;61:1016-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 131] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 8. | Wilkinson NW, Yothers G, Lopa S, Costantino JP, Petrelli NJ, Wolmark N. Long-term survival results of surgery alone vs surgery plus 5-fluorouracil and leucovorin for stage II and stage III colon cancer: pooled analysis of NSABP C-01 through C-05. A baseline from which to compare modern adjuvant trials. Ann Surg Oncol. 2010;17:959-966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 9. | Böckelman C, Engelmann BE, Kaprio T, Hansen TF, Glimelius B. Risk of recurrence in patients with colon cancer stage II and III: a systematic review and meta-analysis of recent literature. Acta Oncol. 2015;54:5-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 281] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 10. | Gu J, Li Z, Zhou J, Sun Z, Bai C. Response prediction to oxaliplatin plus 5-fluorouracil chemotherapy in patients with colorectal cancer using a four-protein immunohistochemical model. Oncol Lett. 2019;18:2091-2101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 11. | Tournigand C, André T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G, Landi B, Colin P, Louvet C, de Gramont A. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22:229-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2282] [Cited by in RCA: 2199] [Article Influence: 104.7] [Reference Citation Analysis (1)] |
| 12. | Grem JL. 5-Fluorouracil: forty-plus and still ticking. A review of its preclinical and clinical development. Invest New Drugs. 2000;18:299-313. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 282] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 13. | Heidelberger C, Chaudhuri NK, Danneberg P, Mooren D, Griesbach L, Duschinsky R, Schnitzer RJ, Pleven E, Scheiner J. Fluorinated pyrimidines, a new class of tumour-inhibitory compounds. Nature. 1957;179:663-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1089] [Cited by in RCA: 1024] [Article Influence: 15.1] [Reference Citation Analysis (1)] |
| 14. | Rutman RJ, Cantarow A, Paschkis KE. The catabolism of uracil in vivo and in vitro. J Biol Chem. 1954;210:321-329. [PubMed] |
| 15. | Wohlhueter RM, McIvor RS, Plagemann PG. Facilitated transport of uracil and 5-fluorouracil, and permeation of orotic acid into cultured mammalian cells. J Cell Physiol. 1980;104:309-319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 117] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 16. | Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3:330-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3255] [Cited by in RCA: 3623] [Article Influence: 164.7] [Reference Citation Analysis (0)] |
| 17. | Thomas DM, Zalcberg JR. 5-fluorouracil: a pharmacological paradigm in the use of cytotoxics. Clin Exp Pharmacol Physiol. 1998;25:887-895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 100] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Noordhuis P, Holwerda U, Van der Wilt CL, Van Groeningen CJ, Smid K, Meijer S, Pinedo HM, Peters GJ. 5-Fluorouracil incorporation into RNA and DNA in relation to thymidylate synthase inhibition of human colorectal cancers. Ann Oncol. 2004;15:1025-1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 181] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 19. | Rutman RJ, Cantarow A, Paschkis KE. Studies in 2-acetylaminofluorene carcinogenesis. III. The utilization of uracil-2-C14 by preneoplastic rat liver and rat hepatoma. Cancer Res. 1954;14:119-123. [PubMed] |
| 20. | Miura K, Kinouchi M, Ishida K, Fujibuchi W, Naitoh T, Ogawa H, Ando T, Yazaki N, Watanabe K, Haneda S, Shibata C, Sasaki I. 5-fu metabolism in cancer and orally-administrable 5-fu drugs. Cancers (Basel). 2010;2:1717-1730. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 178] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 21. | Chalabi M, Fanchi LF, Dijkstra KK, Van den Berg JG, Aalbers AG, Sikorska K, Lopez-Yurda M, Grootscholten C, Beets GL, Snaebjornsson P, Maas M, Mertz M, Veninga V, Bounova G, Broeks A, Beets-Tan RG, de Wijkerslooth TR, van Lent AU, Marsman HA, Nuijten E, Kok NF, Kuiper M, Verbeek WH, Kok M, Van Leerdam ME, Schumacher TN, Voest EE, Haanen JB. Neoadjuvant immunotherapy leads to pathological responses in MMR-proficient and MMR-deficient early-stage colon cancers. Nat Med. 2020;26:566-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 885] [Article Influence: 177.0] [Reference Citation Analysis (0)] |
| 22. | Vodenkova S, Buchler T, Cervena K, Veskrnova V, Vodicka P, Vymetalkova V. 5-fluorouracil and other fluoropyrimidines in colorectal cancer: Past, present and future. Pharmacol Ther. 2020;206:107447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 597] [Article Influence: 99.5] [Reference Citation Analysis (0)] |
| 23. | Robertson J, Barr R, Shulman LN, Forte GB, Magrini N. Essential medicines for cancer: WHO recommendations and national priorities. Bull World Health Organ. 2016;94:735-742. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 87] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 24. | Sargent D, Sobrero A, Grothey A, O'Connell MJ, Buyse M, Andre T, Zheng Y, Green E, Labianca R, O'Callaghan C, Seitz JF, Francini G, Haller D, Yothers G, Goldberg R, de Gramont A. Evidence for cure by adjuvant therapy in colon cancer: observations based on individual patient data from 20,898 patients on 18 randomized trials. J Clin Oncol. 2009;27:872-877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 471] [Cited by in RCA: 507] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 25. | Coronel-Hernández J, Salgado-García R, Cantú-De León D, Jacobo-Herrera N, Millan-Catalan O, Delgado-Waldo I, Campos-Parra AD, Rodríguez-Morales M, Delgado-Buenrostro NL, Pérez-Plasencia C. Combination of Metformin, Sodium Oxamate and Doxorubicin Induces Apoptosis and Autophagy in Colorectal Cancer Cells via Downregulation HIF-1α. Front Oncol. 2021;11:594200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 27] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 26. | Soulières D, Greer W, Magliocco AM, Huntsman D, Young S, Tsao MS, Kamel-Reid S. KRAS mutation testing in the treatment of metastatic colorectal cancer with anti-EGFR therapies. Curr Oncol. 2010;17 Suppl 1:S31-S40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Hasbal-Celikok G, Aksoy-Sagirli P, Altiparmak-Ulbegi G, Can A. Identification of AKT1/β-catenin mutations conferring cetuximab and chemotherapeutic drug resistance in colorectal cancer treatment. Oncol Lett. 2021;21:209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Tong J, Ganguly PK, Singal PK. Myocardial adrenergic changes at two stages of heart failure due to adriamycin treatment in rats. Am J Physiol. 1991;260:H909-H916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 25] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 29. | Injac R, Radic N, Govedarica B, Perse M, Cerar A, Djordjevic A, Strukelj B. Acute doxorubicin pulmotoxicity in rats with malignant neoplasm is effectively treated with fullerenol C60(OH)24 through inhibition of oxidative stress. Pharmacol Rep. 2009;61:335-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 48] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 30. | Bednarski BK, Baldwin AS Jr, Kim HJ. Addressing reported pro-apoptotic functions of NF-kappaB: targeted inhibition of canonical NF-kappaB enhances the apoptotic effects of doxorubicin. PLoS One. 2009;4:e6992. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 31. | Lind DS, Hochwald SN, Malaty J, Rekkas S, Hebig P, Mishra G, Moldawer LL, Copeland EM 3rd, Mackay S. Nuclear factor-kappa B is upregulated in colorectal cancer. Surgery. 2001;130:363-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 218] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 32. | Nooter K, Stoter G. Molecular mechanisms of multidrug resistance in cancer chemotherapy. Pathol Res Pract. 1996;192:768-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 118] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 33. | Chen S, Liu H, Li J, Yang G. Risk of Gastric and Colorectal Cancer After Tamoxifen Use for Breast Cancer: A Systematic Review and Meta-Analysis. J Clin Gastroenterol. 2015;49:666-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Goodsell DS. The molecular perspective: tamoxifen and the estrogen receptor. Oncologist. 2002;7:163-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Wright M, Beaty JS, Ternent CA. Molecular Markers for Colorectal Cancer. Surg Clin North Am. 2017;97:683-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 36. | Grady WM, Carethers JM. Genomic and epigenetic instability in colorectal cancer pathogenesis. Gastroenterology. 2008;135:1079-1099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 717] [Cited by in RCA: 716] [Article Influence: 42.1] [Reference Citation Analysis (0)] |
| 37. | Mármol I, Sánchez-de-Diego C, Pradilla Dieste A, Cerrada E, Rodriguez Yoldi MJ. Colorectal Carcinoma: A General Overview and Future Perspectives in Colorectal Cancer. Int J Mol Sci. 2017;18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 520] [Cited by in RCA: 903] [Article Influence: 112.9] [Reference Citation Analysis (2)] |
| 38. | Van Cutsem E, Nordlinger B, Cervantes A; ESMO Guidelines Working Group. Advanced colorectal cancer: ESMO Clinical Practice Guidelines for treatment. Ann Oncol. 2010;21 Suppl 5:v93-v97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 353] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 39. | Van Cutsem E, Cervantes A, Nordlinger B, Arnold D; ESMO Guidelines Working Group. Metastatic colorectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2014;25 Suppl 3:iii1-iii9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 658] [Cited by in RCA: 797] [Article Influence: 72.5] [Reference Citation Analysis (0)] |
| 40. | Goldstein DA, Zeichner SB, Bartnik CM, Neustadter E, Flowers CR. Metastatic Colorectal Cancer: A Systematic Review of the Value of Current Therapies. Clin Colorectal Cancer. 2016;15:1-6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 67] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 41. | Aparicio J, Fernandez-Martos C, Vincent JM, Maestu I, Llorca C, Busquier I, Campos JM, Perez-Enguix D, Balcells M. FOLFOX alternated with FOLFIRI as first-line chemotherapy for metastatic colorectal cancer. Clin Colorectal Cancer. 2005;5:263-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 42. | Chakrabarti S, Peterson CY, Sriram D, Mahipal A. Early stage colon cancer: Current treatment standards, evolving paradigms, and future directions. World J Gastrointest Oncol. 2020;12:808-832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 91] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 43. | Glimelius B, Graf W, Hoffman K, Påhlman L, Sjödén PO, Wennberg A. General condition of asymptomatic patients with advanced colorectal cancer receiving palliative chemotherapy. A longitudinal study. Acta Oncol. 1992;31:645-651. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 31] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 44. | Ackland SP, Jones M, Tu D, Simes J, Yuen J, Sargeant AM, Dhillon H, Goldberg RM, Abdi E, Shepherd L, Moore MJ. A meta-analysis of two randomised trials of early chemotherapy in asymptomatic metastatic colorectal cancer. Br J Cancer. 2005;93:1236-1243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 46] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 45. | Hu Y, Tao SY, Deng JM, Hou ZK, Liang JQ, Huang QG, Li LH, Li HB, Chen YM, Yi H, Chen XL, Liu H. Prognostic Value of NRAS Gene for Survival of Colorectal Cancer Patients: A Systematic Review and Meta-Analysis. Asian Pac J Cancer Prev. 2018;19:3001-3008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 46. | Peng J, Lv J, Peng J. KRAS mutation is predictive for poor prognosis in rectal cancer patients with neoadjuvant chemoradiotherapy: a systemic review and meta-analysis. Int J Colorectal Dis. 2021;36:1781-1790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 47. | Dai D, Wang Y, Zhu L, Jin H, Wang X. Prognostic value of KRAS mutation status in colorectal cancer patients: a population-based competing risk analysis. PeerJ. 2020;8:e9149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 48. | Jones RP, Sutton PA, Evans JP, Clifford R, McAvoy A, Lewis J, Rousseau A, Mountford R, McWhirter D, Malik HZ. Specific mutations in KRAS codon 12 are associated with worse overall survival in patients with advanced and recurrent colorectal cancer. Br J Cancer. 2017;116:923-929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 119] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 49. | Van Cutsem E, Lenz HJ, Köhne CH, Heinemann V, Tejpar S, Melezínek I, Beier F, Stroh C, Rougier P, van Krieken JH, Ciardiello F. Fluorouracil, leucovorin, and irinotecan plus cetuximab treatment and RAS mutations in colorectal cancer. J Clin Oncol. 2015;33:692-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 564] [Cited by in RCA: 631] [Article Influence: 63.1] [Reference Citation Analysis (0)] |
| 50. | Peeters M, Oliner KS, Price TJ, Cervantes A, Sobrero AF, Ducreux M, Hotko Y, André T, Chan E, Lordick F, Punt CJ, Strickland AH, Wilson G, Ciuleanu TE, Roman L, Van Cutsem E, He P, Yu H, Koukakis R, Terwey JH, Jung AS, Sidhu R, Patterson SD. Analysis of KRAS/NRAS Mutations in a Phase III Study of Panitumumab with FOLFIRI Compared with FOLFIRI Alone as Second-line Treatment for Metastatic Colorectal Cancer. Clin Cancer Res. 2015;21:5469-5479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 148] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 51. | Bokemeyer C, Köhne CH, Ciardiello F, Lenz HJ, Heinemann V, Klinkhardt U, Beier F, Duecker K, van Krieken JH, Tejpar S. FOLFOX4 plus cetuximab treatment and RAS mutations in colorectal cancer. Eur J Cancer. 2015;51:1243-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 189] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 52. | Ikoma T, Shimokawa M, Kotaka M, Matsumoto T, Nagai H, Boku S, Shibata N, Yasui H, Satake H. Clinical and prognostic features of patients with detailed RAS/BRAF-mutant colorectal cancer in Japan. BMC Cancer. 2021;21:518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 53. | Housman G, Byler S, Heerboth S, Lapinska K, Longacre M, Snyder N, Sarkar S. Drug resistance in cancer: an overview. Cancers (Basel). 2014;6:1769-1792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1265] [Cited by in RCA: 1681] [Article Influence: 152.8] [Reference Citation Analysis (0)] |
| 54. | Alfarouk KO, Stock CM, Taylor S, Walsh M, Muddathir AK, Verduzco D, Bashir AH, Mohammed OY, Elhassan GO, Harguindey S, Reshkin SJ, Ibrahim ME, Rauch C. Resistance to cancer chemotherapy: failure in drug response from ADME to P-gp. Cancer Cell Int. 2015;15:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 425] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 55. | Lu C, Shervington A. Chemoresistance in gliomas. Mol Cell Biochem. 2008;312:71-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 102] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 56. | Brasseur K, Gévry N, Asselin E. Chemoresistance and targeted therapies in ovarian and endometrial cancers. Oncotarget. 2017;8:4008-4042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 146] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 57. | Zhu G, Pei L, Xia H, Tang Q, Bi F. Role of oncogenic KRAS in the prognosis, diagnosis and treatment of colorectal cancer. Mol Cancer. 2021;20:143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 250] [Article Influence: 62.5] [Reference Citation Analysis (1)] |
| 58. | Yoon HH, Tougeron D, Shi Q, Alberts SR, Mahoney MR, Nelson GD, Nair SG, Thibodeau SN, Goldberg RM, Sargent DJ, Sinicrope FA; Alliance for Clinical Trials in Oncology. KRAS codon 12 and 13 mutations in relation to disease-free survival in BRAF-wild-type stage III colon cancers from an adjuvant chemotherapy trial (N0147 alliance). Clin Cancer Res. 2014;20:3033-3043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 118] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 59. | Yuan S, Tao F, Zhang X, Zhang Y, Sun X, Wu D. Role of Wnt/β-Catenin Signaling in the Chemoresistance Modulation of Colorectal Cancer. Biomed Res Int. 2020;2020:9390878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 60. | Bugter JM, Fenderico N, Maurice MM. Mutations and mechanisms of WNT pathway tumour suppressors in cancer. Nat Rev Cancer. 2021;21:5-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 289] [Article Influence: 72.3] [Reference Citation Analysis (0)] |
| 61. | Gallo G, Vescio G, De Paola G, Sammarco G. Therapeutic Targets and Tumor Microenvironment in Colorectal Cancer. J Clin Med. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 62. | Saleh R, Elkord E. Acquired resistance to cancer immunotherapy: Role of tumor-mediated immunosuppression. Semin Cancer Biol. 2020;65:13-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 176] [Article Influence: 29.3] [Reference Citation Analysis (0)] |
| 63. | Fang H, Declerck YA. Targeting the tumor microenvironment: from understanding pathways to effective clinical trials. Cancer Res. 2013;73:4965-4977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 210] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 64. | Gaggianesi M, Di Franco S, Pantina VD, Porcelli G, D'Accardo C, Verona F, Veschi V, Colarossi L, Faldetta N, Pistone G, Bongiorno MR, Todaro M, Stassi G. Messing Up the Cancer Stem Cell Chemoresistance Mechanisms Supported by Tumor Microenvironment. Front Oncol. 2021;11:702642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 65. | Li X, Slayton WB. Molecular mechanisms of platelet and stem cell rebound after 5-fluorouracil treatment. Exp Hematol. 2013;41:635-645.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 66. | Fang H, Ang B, Xu X, Huang X, Wu Y, Sun Y, Wang W, Li N, Cao X, Wan T. TLR4 is essential for dendritic cell activation and anti-tumor T-cell response enhancement by DAMPs released from chemically stressed cancer cells. Cell Mol Immunol. 2014;11:150-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 158] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 67. | Wang D, Yang L, Yu W, Wu Q, Lian J, Li F, Liu S, Li A, He Z, Liu J, Sun Z, Yuan W, Zhang Y. Colorectal cancer cell-derived CCL20 recruits regulatory T cells to promote chemoresistance via FOXO1/CEBPB/NF-κB signaling. J Immunother Cancer. 2019;7:215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 150] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 68. | Bagchi S, Yuan R, Engleman EG. Immune Checkpoint Inhibitors for the Treatment of Cancer: Clinical Impact and Mechanisms of Response and Resistance. Annu Rev Pathol. 2021;16:223-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 1324] [Article Influence: 264.8] [Reference Citation Analysis (0)] |
| 69. | Robert C, Long GV, Brady B, Dutriaux C, Maio M, Mortier L, Hassel JC, Rutkowski P, McNeil C, Kalinka-Warzocha E, Savage KJ, Hernberg MM, Lebbé C, Charles J, Mihalcioiu C, Chiarion-Sileni V, Mauch C, Cognetti F, Arance A, Schmidt H, Schadendorf D, Gogas H, Lundgren-Eriksson L, Horak C, Sharkey B, Waxman IM, Atkinson V, Ascierto PA. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3964] [Cited by in RCA: 4342] [Article Influence: 434.2] [Reference Citation Analysis (0)] |
| 70. | Pires da Silva I, Ahmed T, Reijers ILM, Weppler AM, Betof Warner A, Patrinely JR, Serra-Bellver P, Allayous C, Mangana J, Nguyen K, Zimmer L, Trojaniello C, Stout D, Lyle M, Klein O, Gerard CL, Michielin O, Haydon A, Ascierto PA, Carlino MS, Lebbe C, Lorigan P, Johnson DB, Sandhu S, Lo SN, Blank CU, Menzies AM, Long GV. Ipilimumab alone or ipilimumab plus anti-PD-1 therapy in patients with metastatic melanoma resistant to anti-PD-(L)1 monotherapy: a multicentre, retrospective, cohort study. Lancet Oncol. 2021;22:836-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 134] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 71. | Larkin J, Chiarion-Sileni V, Gonzalez R, Grob JJ, Cowey CL, Lao CD, Schadendorf D, Dummer R, Smylie M, Rutkowski P, Ferrucci PF, Hill A, Wagstaff J, Carlino MS, Haanen JB, Maio M, Marquez-Rodas I, McArthur GA, Ascierto PA, Long GV, Callahan MK, Postow MA, Grossmann K, Sznol M, Dreno B, Bastholt L, Yang A, Rollin LM, Horak C, Hodi FS, Wolchok JD. Combined Nivolumab and Ipilimumab or Monotherapy in Untreated Melanoma. N Engl J Med. 2015;373:23-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6070] [Cited by in RCA: 6194] [Article Influence: 619.4] [Reference Citation Analysis (0)] |
| 72. | De Sousa Linhares A, Battin C, Jutz S, Leitner J, Hafner C, Tobias J, Wiedermann U, Kundi M, Zlabinger GJ, Grabmeier-Pfistershammer K, Steinberger P. Therapeutic PD-L1 antibodies are more effective than PD-1 antibodies in blocking PD-1/PD-L1 signaling. Sci Rep. 2019;9:11472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 121] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 73. | de Azevedo SJ, de Melo AC, Roberts L, Caro I, Xue C, Wainstein A. First-line atezolizumab monotherapy in patients with advanced BRAF(V600) wild-type melanoma. Pigment Cell Melanoma Res. 2021;34:973-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 74. | Schmid P, Adams S, Rugo HS, Schneeweiss A, Barrios CH, Iwata H, Diéras V, Hegg R, Im SA, Shaw Wright G, Henschel V, Molinero L, Chui SY, Funke R, Husain A, Winer EP, Loi S, Emens LA; IMpassion130 Trial Investigators. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. N Engl J Med. 2018;379:2108-2121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2345] [Cited by in RCA: 2996] [Article Influence: 428.0] [Reference Citation Analysis (0)] |
| 75. | West H, McCleod M, Hussein M, Morabito A, Rittmeyer A, Conter HJ, Kopp HG, Daniel D, McCune S, Mekhail T, Zer A, Reinmuth N, Sadiq A, Sandler A, Lin W, Ochi Lohmann T, Archer V, Wang L, Kowanetz M, Cappuzzo F. Atezolizumab in combination with carboplatin plus nab-paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019;20:924-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 1182] [Article Influence: 197.0] [Reference Citation Analysis (0)] |
| 76. | O'Donnell JS, Long GV, Scolyer RA, Teng MW, Smyth MJ. Resistance to PD1/PDL1 checkpoint inhibition. Cancer Treat Rev. 2017;52:71-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 434] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 77. | Chung KY, Gore I, Fong L, Venook A, Beck SB, Dorazio P, Criscitiello PJ, Healey DI, Huang B, Gomez-Navarro J, Saltz LB. Phase II study of the anti-cytotoxic T-lymphocyte-associated antigen 4 monoclonal antibody, tremelimumab, in patients with refractory metastatic colorectal cancer. J Clin Oncol. 2010;28:3485-3490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 230] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 78. | Overman MJ, McDermott R, Leach JL, Lonardi S, Lenz HJ, Morse MA, Desai J, Hill A, Axelson M, Moss RA, Goldberg MV, Cao ZA, Ledeine JM, Maglinte GA, Kopetz S, André T. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): an open-label, multicentre, phase 2 study. Lancet Oncol. 2017;18:1182-1191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1775] [Cited by in RCA: 2077] [Article Influence: 259.6] [Reference Citation Analysis (0)] |
| 79. | Kawazoe A, Itahashi K, Yamamoto N, Kotani D, Kuboki Y, Taniguchi H, Harano K, Naito Y, Suzuki M, Fukutani M, Higuchi T, Ikeno T, Wakabayashi M, Sato A, Koyama S, Nishikawa H, Shitara K. TAS-116 (Pimitespib), an Oral HSP90 Inhibitor, in Combination with Nivolumab in Patients with Colorectal Cancer and Other Solid Tumors: An Open-Label, Dose-Finding, and Expansion Phase Ib Trial (EPOC1704). Clin Cancer Res. 2021;27:6709-6715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 33] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 80. | Suzuki S, Kawakami H, Miike T, Yamamoto S. Complete Remission of Colon Cancer with Ipilimumab Monotherapy. Intern Med. 2021;60:957-958. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 81. | Andrews LP, Marciscano AE, Drake CG, Vignali DA. LAG3 (CD223) as a cancer immunotherapy target. Immunol Rev. 2017;276:80-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 687] [Article Influence: 85.9] [Reference Citation Analysis (0)] |
| 82. | Haag GM, Springfeld C, Grün B, Apostolidis L, Zschäbitz S, Dietrich M, Berger AK, Weber TF, Zoernig I, Schaaf M, Waberer L, Müller DW, Al-Batran SE, Halama N, Jaeger D. Pembrolizumab and maraviroc in refractory mismatch repair proficient/microsatellite-stable metastatic colorectal cancer - The PICCASSO phase I trial. Eur J Cancer. 2022;167:112-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 68] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 83. | Kim JH, Kim SY, Baek JY, Cha YJ, Ahn JB, Kim HS, Lee KW, Kim JW, Kim TY, Chang WJ, Park JO, Kim J, Kim JE, Hong YS, Kim YH, Kim TW. A Phase II Study of Avelumab Monotherapy in Patients with Mismatch Repair-Deficient/Microsatellite Instability-High or POLE-Mutated Metastatic or Unresectable Colorectal Cancer. Cancer Res Treat. 2020;52:1135-1144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 84. | Oh CR, Kim JE, Hong YS, Kim SY, Ahn JB, Baek JY, Lee MA, Kang MJ, Cho SH, Beom SH, Kim TW. Phase II study of durvalumab monotherapy in patients with previously treated microsatellite instability-high/mismatch repair-deficient or POLE-mutated metastatic or unresectable colorectal cancer. Int J Cancer. 2022;150:2038-2045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 85. | Kanikarla Marie P, Haymaker C, Parra ER, Kim YU, Lazcano R, Gite S, Lorenzini D, Wistuba II, Tidwell RSS, Song X, Foo WC, Maru DM, Chun YS, Futreal A, Kee B, Menter D, Solis L, Tzeng CW, Parseghian C, Raghav K, Morris V, Chang CC, Jenq R, Tam A, Bernatchez C, Kopetz S, Vauthey JN, Overman MJ. Pilot Clinical Trial of Perioperative Durvalumab and Tremelimumab in the Treatment of Resectable Colorectal Cancer Liver Metastases. Clin Cancer Res. 2021;27:3039-3049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 86. | Lenz HJ, Van Cutsem E, Luisa Limon M, Wong KYM, Hendlisz A, Aglietta M, García-Alfonso P, Neyns B, Luppi G, Cardin DB, Dragovich T, Shah U, Abdullaev S, Gricar J, Ledeine JM, Overman MJ, Lonardi S. First-Line Nivolumab Plus Low-Dose Ipilimumab for Microsatellite Instability-High/Mismatch Repair-Deficient Metastatic Colorectal Cancer: The Phase II CheckMate 142 Study. J Clin Oncol. 2022;40:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 412] [Article Influence: 137.3] [Reference Citation Analysis (0)] |
| 87. | Morse MA, Overman MJ, Hartman L, Khoukaz T, Brutcher E, Lenz HJ, Atasoy A, Shangguan T, Zhao H, El-Rayes B. Safety of Nivolumab plus Low-Dose Ipilimumab in Previously Treated Microsatellite Instability-High/Mismatch Repair-Deficient Metastatic Colorectal Cancer. Oncologist. 2019;24:1453-1461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 74] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 88. | Kasi PM, Budde G, Krainock M, Aushev VN, Koyen Malashevich A, Malhotra M, Olshan P, Billings PR, Aleshin A. Circulating tumor DNA (ctDNA) serial analysis during progression on PD-1 blockade and later CTLA-4 rescue in patients with mismatch repair deficient metastatic colorectal cancer. J Immunother Cancer. 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 89. | Johnson B, Haymaker CL, Parra ER, Soto LMS, Wang X, Thomas JV, Dasari A, Morris VK, Raghav K, Vilar E, Kee BK, Eng C, Parseghian CM, Wolff RA, Lee Y, Lorenzini D, Laberiano-Fernandez C, Verma A, Lang W, Wistuba II, Futreal A, Kopetz S, Overman MJ. Phase II study of durvalumab (anti-PD-L1) and trametinib (MEKi) in microsatellite stable (MSS) metastatic colorectal cancer (mCRC). J Immunother Cancer. 2022;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 31] [Reference Citation Analysis (0)] |
| 90. | Cohen R, Bennouna J, Meurisse A, Tournigand C, De La Fouchardière C, Tougeron D, Borg C, Mazard T, Chibaudel B, Garcia-Larnicol ML, Svrcek M, Vernerey D, Menu Y, André T. RECIST and iRECIST criteria for the evaluation of nivolumab plus ipilimumab in patients with microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: the GERCOR NIPICOL phase II study. J Immunother Cancer. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 91. | Eng C, Kim TW, Bendell J, Argilés G, Tebbutt NC, Di Bartolomeo M, Falcone A, Fakih M, Kozloff M, Segal NH, Sobrero A, Yan Y, Chang I, Uyei A, Roberts L, Ciardiello F; IMblaze370 Investigators. Atezolizumab with or without cobimetinib vs regorafenib in previously treated metastatic colorectal cancer (IMblaze370): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2019;20:849-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 408] [Article Influence: 68.0] [Reference Citation Analysis (0)] |
| 92. | Hellmann MD, Kim TW, Lee CB, Goh BC, Miller WH Jr, Oh DY, Jamal R, Chee CE, Chow LQM, Gainor JF, Desai J, Solomon BJ, Das Thakur M, Pitcher B, Foster P, Hernandez G, Wongchenko MJ, Cha E, Bang YJ, Siu LL, Bendell J. Phase Ib study of atezolizumab combined with cobimetinib in patients with solid tumors. Ann Oncol. 2019;30:1134-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 131] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 93. | Fumet JD, Isambert N, Hervieu A, Zanetta S, Guion JF, Hennequin A, Rederstorff E, Bertaut A, Ghiringhelli F. Phase Ib/II trial evaluating the safety, tolerability and immunological activity of durvalumab (MEDI4736) (anti-PD-L1) plus tremelimumab (anti-CTLA-4) combined with FOLFOX in patients with metastatic colorectal cancer. ESMO Open. 2018;3:e000375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 94. | Antoniotti C, Rossini D, Pietrantonio F, Catteau A, Salvatore L, Lonardi S, Boquet I, Tamberi S, Marmorino F, Moretto R, Ambrosini M, Tamburini E, Tortora G, Passardi A, Bergamo F, Kassambara A, Sbarrato T, Morano F, Ritorto G, Borelli B, Boccaccino A, Conca V, Giordano M, Ugolini C, Fieschi J, Papadopulos A, Massoué C, Aprile G, Antonuzzo L, Gelsomino F, Martinelli E, Pella N, Masi G, Fontanini G, Boni L, Galon J, Cremolini C; GONO Foundation Investigators. Upfront FOLFOXIRI plus bevacizumab with or without atezolizumab in the treatment of patients with metastatic colorectal cancer (AtezoTRIBE): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol. 2022;23:876-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 157] [Article Influence: 52.3] [Reference Citation Analysis (0)] |
| 95. | Tabernero J, Grothey A, Arnold D, de Gramont A, Ducreux M, O'Dwyer P, Tahiri A, Gilberg F, Irahara N, Schmoll HJ, Van Cutsem E. MODUL cohort 2: an adaptable, randomized, signal-seeking trial of fluoropyrimidine plus bevacizumab with or without atezolizumab maintenance therapy for BRAF(wt) metastatic colorectal cancer. ESMO Open. 2022;7:100559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 96. | Martinelli E, Martini G, Famiglietti V, Troiani T, Napolitano S, Pietrantonio F, Ciardiello D, Terminiello M, Borrelli C, Vitiello PP, De Braud F, Morano F, Avallone A, Normanno N, Nappi A, Maiello E, Latiano T, Falcone A, Cremolini C, Rossini D, Santabarbara G, Pinto C, Santini D, Cardone C, Zanaletti N, Di Liello A, Renato D, Esposito L, Marrone F, Ciardiello F. Cetuximab Rechallenge Plus Avelumab in Pretreated Patients With RAS Wild-type Metastatic Colorectal Cancer: The Phase 2 Single-Arm Clinical CAVE Trial. JAMA Oncol. 2021;7:1529-1535. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 115] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 97. | Stein A, Simnica D, Schultheiß C, Scholz R, Tintelnot J, Gökkurt E, von Wenserski L, Willscher E, Paschold L, Sauer M, Lorenzen S, Riera-Knorrenschild J, Depenbusch R, Ettrich TJ, Dörfel S, Al-Batran SE, Karthaus M, Pelzer U, Waberer L, Hinke A, Bauer M, Massa C, Seliger B, Wickenhauser C, Bokemeyer C, Hegewisch-Becker S, Binder M. PD-L1 targeting and subclonal immune escape mediated by PD-L1 mutations in metastatic colorectal cancer. J Immunother Cancer. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 98. | Ciardiello D, Famiglietti V, Napolitano S, Esposito L, Pietrantonio F, Avallone A, Maiello E, Cremolini C, Troiani T, Martinelli E, Ciardiello F, Martini G. Final results of the CAVE trial in RAS wild type metastatic colorectal cancer patients treated with cetuximab plus avelumab as rechallenge therapy: Neutrophil to lymphocyte ratio predicts survival. Clin Colorectal Cancer. 2022;21:141-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 99. | Cousin S, Cantarel C, Guegan JP, Gomez-Roca C, Metges JP, Adenis A, Pernot S, Bellera C, Kind M, Auzanneau C, Le Loarer F, Soubeyran I, Bessede A, Italiano A. Regorafenib-Avelumab Combination in Patients with Microsatellite Stable Colorectal Cancer (REGOMUNE): A Single-arm, Open-label, Phase II Trial. Clin Cancer Res. 2021;27:2139-2147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 100. | Segal NH, Cercek A, Ku G, Wu AJ, Rimner A, Khalil DN, Reidy-Lagunes D, Cuaron J, Yang TJ, Weiser MR, Romesser PB, Stadler ZK, Varghese AM, Ganesh K, Yaeger R, Connell LC, Faleck D, Abou-Alfa GK, Mcauliffe KC, Vaiskauskas P, Solter ML, Ogle M, Adamow MJ, Holland A, Vedantam P, Wong P, Merghoub T, Vakiani E, Hollmann TJ, Juluru K, Chou JF, Capanu M, Erinjeri J, Solomon S, Yamada Y, Kemeny N, Crane CH, Saltz LB. Phase II Single-arm Study of Durvalumab and Tremelimumab with Concurrent Radiotherapy in Patients with Mismatch Repair-proficient Metastatic Colorectal Cancer. Clin Cancer Res. 2021;27:2200-2208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 65] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 101. | Redman JM, Tsai YT, Weinberg BA, Donahue RN, Gandhy S, Gatti-Mays ME, Abdul Sater H, Bilusic M, Cordes LM, Steinberg SM, Marte JL, Jochems C, Kim SS, Marshall JL, McMahon S, Redmond E, Schlom J, Gulley JL, Strauss J. A Randomized Phase II Trial of mFOLFOX6 + Bevacizumab Alone or with AdCEA Vaccine + Avelumab Immunotherapy for Untreated Metastatic Colorectal Cancer. Oncologist. 2022;27:198-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 23] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 102. | Chen EX, Jonker DJ, Loree JM, Kennecke HF, Berry SR, Couture F, Ahmad CE, Goffin JR, Kavan P, Harb M, Colwell B, Samimi S, Samson B, Abbas T, Aucoin N, Aubin F, Koski SL, Wei AC, Magoski NM, Tu D, O'Callaghan CJ. Effect of Combined Immune Checkpoint Inhibition vs Best Supportive Care Alone in Patients With Advanced Colorectal Cancer: The Canadian Cancer Trials Group CO.26 Study. JAMA Oncol. 2020;6:831-838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 209] [Cited by in RCA: 282] [Article Influence: 56.4] [Reference Citation Analysis (0)] |
| 103. | Randrian V, Pernot S, Le Malicot K, Catena V, Baumgaertner I, Tacher V, Forestier J, Hautefeuille V, Tabouret-Viaud C, Gagnaire A, Mitry E, Guiu B, Aparicio T, Smith D, Dhomps A, Tasu JP, Perdrisot R, Edeline J, Capron C, Cheze-Le Rest C, Emile JF, Laurent-Puig P, Bejan-Angoulvant T, Sokol H, Lepage C, Taieb J, Tougeron D. FFCD 1709-SIRTCI phase II trial: Selective internal radiation therapy plus Xelox, Bevacizumab and Atezolizumab in liver-dominant metastatic colorectal cancer. Dig Liver Dis. 2022;54:857-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 104. | Lau D, Kalaitzaki E, Church DN, Pandha H, Tomlinson I, Annels N, Gerlinger M, Sclafani F, Smith G, Begum R, Crux R, Gillbanks A, Wordsworth S, Chau I, Starling N, Cunningham D, Dhillon T. Rationale and design of the POLEM trial: avelumab plus fluoropyrimidine-based chemotherapy as adjuvant treatment for stage III mismatch repair deficient or POLE exonuclease domain mutant colon cancer: a phase III randomised study. ESMO Open. 2020;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 105. | Ryman JT, Meibohm B. Pharmacokinetics of Monoclonal Antibodies. CPT Pharmacometrics Syst Pharmacol. 2017;6:576-588. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 349] [Cited by in RCA: 530] [Article Influence: 66.3] [Reference Citation Analysis (0)] |
| 106. | Wang J, Giragossian C, Hansel S. Analyze impact of tumor-associated kinetics on antibody delivery in solid tumors with a physiologically based pharmacokinetics/pharmacodynamics model. Eur J Pharm Biopharm. 2021;168:110-121. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 107. | Gawel DR, Serra-Musach J, Lilja S, Aagesen J, Arenas A, Asking B, Bengnér M, Björkander J, Biggs S, Ernerudh J, Hjortswang H, Karlsson JE, Köpsen M, Lee EJ, Lentini A, Li X, Magnusson M, Martínez-Enguita D, Matussek A, Nestor CE, Schäfer S, Seifert O, Sonmez C, Stjernman H, Tjärnberg A, Wu S, Åkesson K, Shalek AK, Stenmarker M, Zhang H, Gustafsson M, Benson M. A validated single-cell-based strategy to identify diagnostic and therapeutic targets in complex diseases. Genome Med. 2019;11:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 108. | Hu P, Zhang W, Xin H, Deng G. Single Cell Isolation and Analysis. Front Cell Dev Biol. 2016;4:116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 161] [Cited by in RCA: 219] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 109. | Gibney GT, Weiner LM, Atkins MB. Predictive biomarkers for checkpoint inhibitor-based immunotherapy. Lancet Oncol. 2016;17:e542-e551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1167] [Cited by in RCA: 1271] [Article Influence: 141.2] [Reference Citation Analysis (0)] |
| 110. | Dancsok AR, Gao D, Lee AF, Steigen SE, Blay JY, Thomas DM, Maki RG, Nielsen TO, Demicco EG. Tumor-associated macrophages and macrophage-related immune checkpoint expression in sarcomas. Oncoimmunology. 2020;9:1747340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 125] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 111. | Solinas C, De Silva P, Bron D, Willard-Gallo K, Sangiolo D. Significance of TIM3 expression in cancer: From biology to the clinic. Semin Oncol. 2019;46:372-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 112. | Olguín JE, Medina-Andrade I, Molina E, Vázquez A, Pacheco-Fernández T, Saavedra R, Pérez-Plasencia C, Chirino YI, Vaca-Paniagua F, Arias-Romero LE, Gutierrez-Cirlos EB, León-Cabrera SA, Rodriguez-Sosa M, Terrazas LI. Early and Partial Reduction in CD4(+)Foxp3(+) Regulatory T Cells during Colitis-Associated Colon Cancer Induces CD4(+) and CD8(+) T Cell Activation Inhibiting Tumorigenesis. J Cancer. 2018;9:239-249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 113. | Sakuishi K, Ngiow SF, Sullivan JM, Teng MW, Kuchroo VK, Smyth MJ, Anderson AC. TIM3(+)FOXP3(+) regulatory T cells are tissue-specific promoters of T-cell dysfunction in cancer. Oncoimmunology. 2013;2:e23849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 202] [Cited by in RCA: 261] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 114. | Curigliano G, Gelderblom H, Mach N, Doi T, Tai D, Forde PM, Sarantopoulos J, Bedard PL, Lin CC, Hodi FS, Wilgenhof S, Santoro A, Sabatos-Peyton CA, Longmire TA, Xyrafas A, Sun H, Gutzwiller S, Manenti L, Naing A. Phase I/Ib Clinical Trial of Sabatolimab, an Anti-TIM-3 Antibody, Alone and in Combination with Spartalizumab, an Anti-PD-1 Antibody, in Advanced Solid Tumors. Clin Cancer Res. 2021;27:3620-3629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 217] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 115. | Tian J, Zhang B, Rui K, Wang S. The Role of GITR/GITRL Interaction in Autoimmune Diseases. Front Immunol. 2020;11:588682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 116. | Geva R, Voskoboynik M, Dobrenkov K, Mayawala K, Gwo J, Wnek R, Chartash E, Long GV. First-in-human phase 1 study of MK-1248, an anti-glucocorticoid-induced tumor necrosis factor receptor agonist monoclonal antibody, as monotherapy or with pembrolizumab in patients with advanced solid tumors. Cancer. 2020;126:4926-4935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 117. | Huard B, Tournier M, Hercend T, Triebel F, Faure F. Lymphocyte-activation gene 3/major histocompatibility complex class II interaction modulates the antigenic response of CD4+ T lymphocytes. Eur J Immunol. 1994;24:3216-3221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 178] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 118. | de Klerk LK, Patel AK, Derks S, Pectasides E, Augustin J, Uduman M, Raman N, Akarca FG, McCleary NJ, Cleary JM, Rubinson DA, Clark JW, Fitzpatrick B, Brais LK, Cavanaugh ME, Rode AJ, Jean MG, Lizotte PH, Nazzaro MJ, Severgnini M, Zheng H, Fuchs CS, Enzinger PC, Bass AJ. Phase II study of pembrolizumab in refractory esophageal cancer with correlates of response and survival. J Immunother Cancer. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 119. | Kim M, Min YK, Jang J, Park H, Lee S, Lee CH. Single-cell RNA sequencing reveals distinct cellular factors for response to immunotherapy targeting CD73 and PD-1 in colorectal cancer. J Immunother Cancer. 2021;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 120. | Turiello R, Pinto A, Morello S. CD73: A Promising Biomarker in Cancer Patients. Front Pharmacol. 2020;11:609931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 121. | Thibaudin M, Limagne E, Hampe L, Ballot E, Truntzer C, Ghiringhelli F. Targeting PD-L1 and TIGIT could restore intratumoral CD8 T cell function in human colorectal cancer. Cancer Immunol Immunother. 2022;71:2549-2563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 122. | Chauvin JM, Zarour HM. TIGIT in cancer immunotherapy. J Immunother Cancer. 2020;8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 430] [Cited by in RCA: 486] [Article Influence: 97.2] [Reference Citation Analysis (0)] |
| 123. | Español-Rego M, Fernández-Martos C, Elez E, Foguet C, Pedrosa L, Rodríguez N, Ruiz-Casado A, Pineda E, Cid J, Cabezón R, Oliveres H, Lozano M, Ginés A, García-Criado A, Ayuso JR, Pagés M, Cuatrecasas M, Torres F, Thomson T, Cascante M, Benítez-Ribas D, Maurel J. A Phase I-II multicenter trial with Avelumab plus autologous dendritic cell vaccine in pre-treated mismatch repair-proficient (MSS) metastatic colorectal cancer patients; GEMCAD 1602 study. Cancer Immunol Immunother. 2022;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 124. | Zhang Z, Lu M, Qin Y, Gao W, Tao L, Su W, Zhong J. Neoantigen: A New Breakthrough in Tumor Immunotherapy. Front Immunol. 2021;12:672356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 156] [Article Influence: 39.0] [Reference Citation Analysis (0)] |