Published online Aug 15, 2022. doi: 10.4251/wjgo.v14.i8.1388
Peer-review started: November 11, 2021
First decision: December 27, 2021
Revised: January 6, 2022
Accepted: July 22, 2022
Article in press: July 22, 2022
Published online: August 15, 2022
Processing time: 272 Days and 5.4 Hours
The KAI1/CD82 gene inhibits the metastasis of most tumors and is remarkably correlated with tumor invasion and prognosis. Cell metabolism dysregulation is an important cause of tumor occurrence, development, and metastasis. As one of the important characteristics of tumors, cell metabolism dysregulation is attracting increasing research attention. Phospholipids are an indispensable substance in the metabolism in various tumor cells. Phospholipid metabolites have become important cell signaling molecules. The pathological role of lysophosphatidic acid (LPA) in tumors was identified in the early 1990s. Currently, LPA inhibitors have entered clinical trials but are not yet used in clinical treatment. Autotaxin (ATX) has lysophospholipase D (lysoPLD) activity and can regulate LPA levels in vivo. The LPA receptor family and ATX/lysoPLD are abnormally expressed in various gastrointestinal tumors. According to our recent pre-experimental results, KAI1/CD82 might inhibit the migration and metastasis of cancer cells by regulating the ATX-LPA axis. However, no relevant research has been reported. Clarifying the mechanism of ATX-LPA in the inhibition of cancer metastasis by KAI1/CD82 will provide an important theoretical basis for targeted cancer therapy. In this paper, the molecular compositions of the KAI1/CD82 gene and the ATX-LPA axis, their physiological functions in tumors, and their roles in gastrointestinal cancers and target therapy are reviewed.
Core Tip: The KAI1/CD82 gene inhibits the metastasis of most tumors and is significantly correlated with their invasion and prognosis. According to our recent pre-experimental results, we speculated that KAI1/CD82 might inhibit the migration and metastasis of cancer cells by regulating autotaxin (ATX)-lysophosphatidic acid (LPA) axis. However, no relevant research has been reported. To clarify the mechanism of ATX-LPA in KAI1/CD82 inhibition of cancer metastasis will provide an important theoretical basis for targeted cancer therapy, and further research is necessary. In this paper, the molecular composition of the KAI1/CD82 gene and ATX-LPA axis, their physiological functions in tumors, and their roles in gastrointestinal cancers and target therapy are reviewed.
- Citation: Wang S, Chen J, Guo XZ. KAI1/CD82 gene and autotaxin-lysophosphatidic acid axis in gastrointestinal cancers. World J Gastrointest Oncol 2022; 14(8): 1388-1405
- URL: https://www.wjgnet.com/1948-5204/full/v14/i8/1388.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v14.i8.1388
The KAI1/CD82 gene is an important tumor suppressor gene. As a metastasis-related suppressor gene of prostate cancer discovered by Dong et al[1] in 1995, KAI1/CD82 is located on human chromosome 11p11.2 and consists of l0 exons and 9 introns with a length of about 80 kb. The protein encoded by this gene is composed of 267 amino acids residues and has a relative molecular weight of 29600 Da. KAI1/CD82 is a member of the transmembrane 4 superfamily (TM4SF). TM4SF proteins promote the interactions between cells and the extracellular matrix, enhance the cohesion between tumor cells, reduce phagocytosis and invasion, and inhibit tumor cell metastasis. Cell dysmetabolism is an important cause of tumor occurrence, development, and metastasis. As one of the hallmarks of cancer, cell dysmetabolism has increasingly attracted the attention of researchers in recent years. Phospholipid is an indispensable substance in cell metabolism and participates in the metabolism of various tumor cells. Phospholipid metabolites have become important cell signaling molecules. Lysophosphatidic acid (LPA) is secreted by platelets, fibroblasts, cancer cells, and fat cells and is a multifunctional “phos
KAI1 (named after Anticancer Kang Ai) is a tumor-suppressor gene first discovered by Dong et al[1] in 1995 on chromosome 11 of rabbit AT6.1 metastatic prostate cancer cells. Later, researchers confirmed that KAI1 has the same structure as the CD82 gene; therefore, it was named KAI1/CD82. The 5’-end promoter region of the KAI1/CD82 gene is 735 bp long and rich in CpG island with nine transcription factor-specific protein SPI binding sites, five AP2 binding sites, and tcF-1, Myb, and MEP.1 binding sites, which suggests that the gene is regulated by multiple mechanisms[2,3]. KAI1/CD82 is located on the cell membrane and is a member of TM4SF, which comprises four conservative hydrophobic transmembrane domains (TM1–TM4) and one extracellular glycosyl-based binding site. This structure indicates that KAI1/CD82, like other TM4SF members, can affect plasma membrane molecular rearrangement, cell aggregation, adhesion, and migration, and other physiological and pathological activities through various mechanisms, as well as inhibit the migration and metastasis of various malignant tumors[4].
ATX is a secretory glycoprotein called autocrine motility factor. ATX was first identified in A2058 melanoma cells and induces cell migration through the pertussis toxin G protein[5]. ATX has phosphodiesterase activity[6], and LPA is catalyzed by lysophosphatidylcholine (LPC)[7]. LPA is a multifunctional “phospholipid messenger” secreted by platelets, fibroblasts, adipocytes, and cancer cells. Although LPA is the simplest phospholipid, it is not a simple biomolecule. LPA has six G-protein-coupled receptors that mediate several physiological and pathological processes, including embryogenesis, wound healing, chronic inflammation, cancer progression, and treatment tolerance[8]. In tumor tissues, LPA binds to LPARs on the cell surface to induce intracellular signal transduction, which in turn regulates tumor cell proliferation, adhesion, migration, and invasion[7]. At present, ATX-LPA target inhibitors are not yet used as a therapeutic measure clinically, and the therapeutic effects of LPA monoclonal antibodies, LPAR antagonists, and ATX inhibitors are still being explored.
ATX is also called extracellular pyrophosphatase/phosphodiesterase (ENPP)2 because of its 47%-55% homology with pc-1/NPP1 and B-10/NPP3 amino acid sequences in the ENPP family. ATX is a multidomain protein[9], and lysophospholipase D (lysoPLD) catalyzes LPA formation[10]. ATX has a slightly U-shaped hydrophobic pocket in the catalytic region, which tends to contain unsaturated substrates, such as unsaturated fatty acids[11], and all five selective splicing isomers have catalytic activity[12,13]. Therefore, its affinity with LPC is strong. Although LPA can be produced by other processes, such as phospholipase A2, Ca2+-independent phospholipase A2, and phosphatidate[14-16], ATX is still the main pathway of extracellular LPA generation.
Serum contains 2-20 μm LPA, and its metabolites extensively affect biological activities inside and outside cells[17]. LPA is one of the smallest glycerophosphatides and comprises three domains: Phosphate head, linker, and lipophilic terminal. The function of the phosphoric head is to activate the receptor; the lipophilic terminal sequence determines its biological activity; and the head and tail are linked by acyl, alkyl, or alkenyl groups[18]. Its free hydroxyl and phosphate groups make LPA more soluble in water than long-chain phospholipids, which likely contributes to its biological activities. The family of lipid phosphate phosphohydrolases (LPPs) dephosphorylates LPA[19,20].
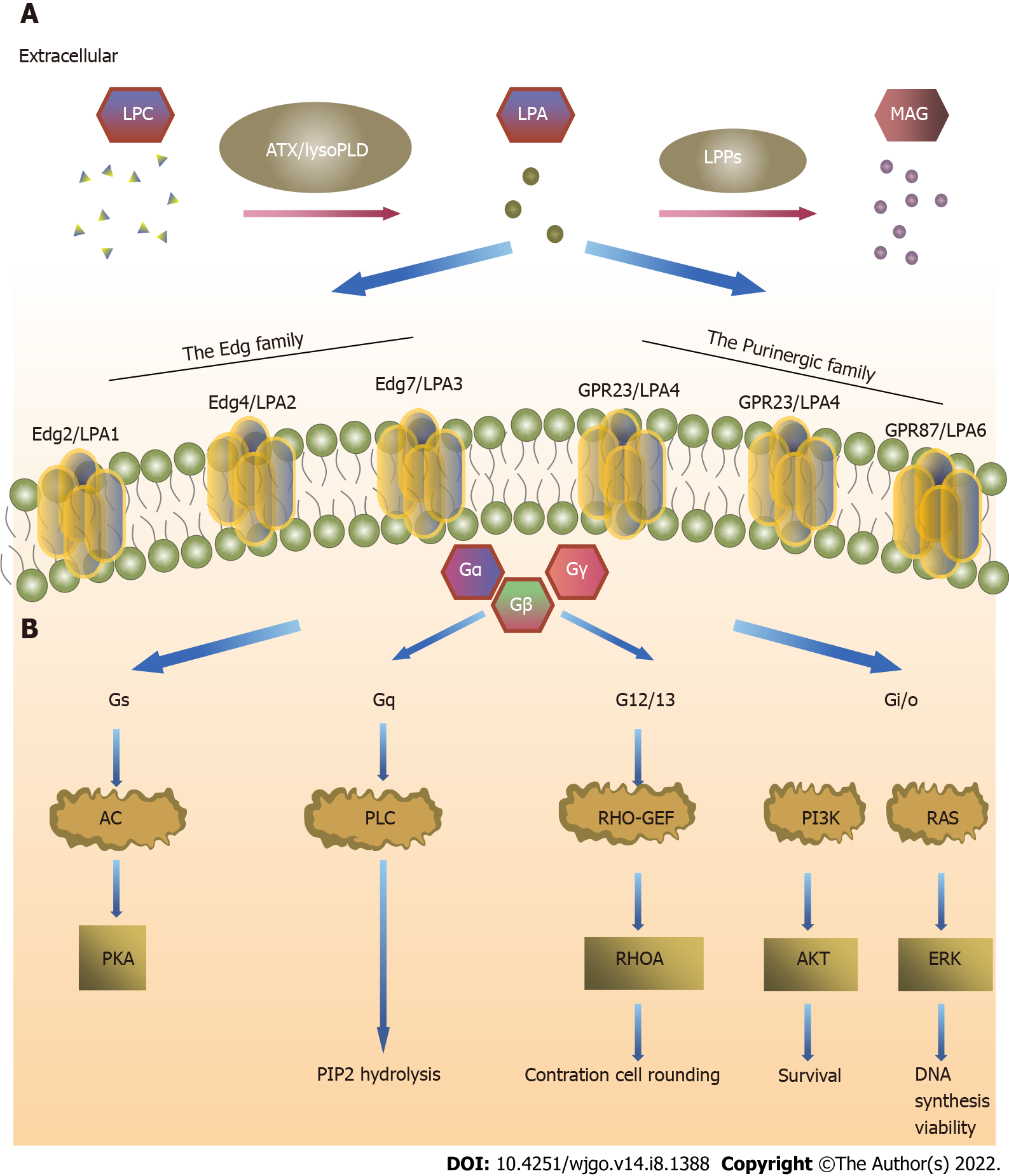
LPARs are divided into two subfamilies: LPA1-3 receptors belonging to the endothelial cell differentiation gene (Edg) family, and LPA4-6 receptors belonging to the purine (P2Y) receptor family[9,21]. LPA1 (Edg2) has 50%-60% amino acid homology with LPA2 (Edg4) and LPA3 (Edg7). LPA1 and LPA2 need to pass through the Gi/O, Gq/11, and G12/13 signaling pathways, whereas LPA3 passes only through the Gi/O and Gq/11 signaling pathways[22]. The function of Gi/O is to stimulate mitotic division through the Ras-Raf-MAPK signaling pathway and promote tumor cell survival through the PI3K-Akt signaling pathway[23,24]. LPA4 (P2Y9/GPR23), LPA5 (GPR92), and LPA6 (P2Y5) have 35%-55% amino acid homology. LPA4 acts through the Gs, Gi/O, Gq/11, and G12/13 signaling pathways and is the only LPAR that activates adenosine cyclase and leads to cyclic adenosine monophosphate elevation. LPA5 plays a role through the Gq/11 and G12/13 signaling pathways, whereas LPA6 plays a role through the G12/13 activation of the Rho signaling pathways[22]. The effect of LPARs on tumors depends on the G protein signaling pathway that it activates[25].
Low KAI1 expression accelerates tumor invasion and metastasis[26]. In 2017, a meta-analysis involving 31 studies showed that high KAI1 expression is significantly associated with overall survival (OS) [hazard ratio (HR) = 0.56, 95% confidence interval (CI): 0.47-0.67] and disease-free/relapse-free/progression-free survival (PFS) (HR = 0.42, 95%CI: 0.30-0.59) in patients with cancer. In addition, they performed a subgroup analysis showing that KAI1/CD82 is associated with a good prognosis in patients with cancer. KAI1/CD82 may be a promising biomarker for predicting the prognosis of patients with malignant tumors, and its biological function has important research value for this topic[27]. The Human Protein Atlas is an outstanding initiative associated to the Human Proteome Project, which has made available valuable information about the functional and pathological aspects of about 17000 proteins. In particular, they are able to propose scores that suggest the prognostic value of proteins in diseases based on the expression levels of these proteins in healthy and diseased tissues. Considering that only 31 studies were included in the meta-analysis, more studies may be needed in the future to verify whether KAI1 can be used as a prognostic factor. KAI1/CD82 may inhibit cell metastasis and migration through two pathways. The first is that KAI1/CD82 inhibits cell migration as an initiating signal. However, the possibility of this pathway is low because of the simple structure of KAI1/CD82 and the lack of corresponding enzymes in the cytoplasm. However, evidence also indicates that KAI1/CD82 may be an initiating signal[28,29]. KAI1/CD82 is crosslinked with monoclonal antibody to induce morphological changes and signal transduction[30]. Integrins are also essential for cell adhesion and migration, and KAI1/CD82 is associated with several integrins, including α3β1, α4β1, α5β1, α6β1, and αLβ2[31-35], which may also be one of the pathways through which KAI1/CD82 inhibits tumor. Epidermal growth factor receptor (EGFR) is a member of the ErbB family. In tumor tissues, the receptors and ligand of the ErbB pathway are overproduced and overactivated. Odintsova et al[36] found that KAI1/CD82 is correlated with EGFR, ErbB2, and ErbB3 and inhibits the endocytosis of the EGF signaling pathway and EGFR. KAI1/CD82 redistributes molecules on the cell membrane surface; KAI1/CD82 overexpression results in the redistribution and aggregation of urokinase-type plasminogen activator receptor (uPAR) into a stable α5β1 complex. Moreover, KAI1/CD82 overexpression also results in the redistribution of EGFR and gangliosides in the plasma membrane. However, whether the redistribution of these substances is related to KAI1/CD82 tumor inhibition remains unknown[37].
LPA signals can be roughly divided into three parts, namely, ATX, LPARs, and LPP of extracellular LPA[38,39]. ATX has lysoPLD activity and promotes LPA generation in blood[40,41]. Many tumor cells secrete ATX[42], LPAR expression is higher on tumor cell surfaces than on normal cells, and LPP expression is lower in tumor cells than in normal cells. Understanding the metabolic pathway of the ATX-LPA-LPP axis in the tumor microenvironment (TME) is important to study its target therapy (Figure 1).
The TME is produced by tumor cells, such as neuroblastoma[43], glioblastoma[44], liver cancer[45], B-cell lymphoma[46], melanoma[47], kidney cancer[48], thyroid cancer[49], breast cancer, and non-small cell lung cancer[50], as well as stromal cells such as fibroblasts and adipocytes[51-53]. How to regulate ATX expression remains unclear. ENPP overexpression may be one of the reasons for ATX upregulation in cancer tissues[54]. The Cancer Genome Atlas shows that ENPP overexpression is present in serous ovarian cystadenocarcinoma (about 33%) and invasive breast carcinoma (about 20%). The ENPP2 gene is overexpressed in hepatocellular carcinoma (HCC; about 20%), lung adenocarcinoma (about 11%), bladder transitional cell carcinoma (about 10%), and head and neck squamous cell carcinoma (about 10%)[13]. Moreover, ATX is involved in the physiological wound-healing response, and ATX levels are increased in some inflammatory diseases[55]. Park et al[56] found that the levels of interleukin (IL)-4, IL-5, and ATX increase in patients with asthma who received bronchoalveolar lavage fluid when stimulated by allergens. ATX induces pro-inflammatory cytokines, such as tumor necrosis factor (TNF)-α and IL-1β[57,58], NOD receptor family (NLRP3), ATM kinase, ATR protein kinase, and nuclear transcription factor-kappa B (NF-κB)[59]. At present, although ATX research has made some progress, the overall understanding remains limited.
LPA is present in intracellular and extracellular fluids (blood, ascites, follicular fluid, saliva, etc.)[56]. In 1989, van Corven et al[60] found that LPA may be involved in cell diffusion and migration. Two years later, Merchant et al[61] found increased LPA levels in malignant colon tumor tissues. LPA may be a simple lipid, but it is involved in all aspects of tumor development; it stimulates proliferative signals[62], prevents growth inhibition and resists apoptosis[63,64], regulates telomerase[64], promotes vascular endothelial growth factor (VEGF)-A and VEGF-C, and induces angiogenesis[65-67]. LPA induces the gene instability caused by reactive oxygen species and stimulates the production of inflammatory factors, such as COX-2, IL, and TNF-α[68,69]. LPA activates at least three signaling pathways: (1) Promotes phosphoinositol hydrolysis and therefore activates protein kinase C (PKC) and Ca2+ mobilization; (2) Promotes the release of guanosine triphosphate (GTP); and (3) Inhibits adenylate cyclase activity. In recent years, the activation of the downstream signaling Ras pathway may promote LPA fibrogenesis[70]. Moreover, MAK-related kinase, as an effector of RhoC, regulates LPA-induced cell invasion through myosin, extracellular signal-regulated kinase (ERK), and P38[71], whereas LPA induces the G12/13-Rhoa-Rock signaling pathway to mediate focal adhesion kinase autophosphorylation and promote tumor cell migration[72]. Furthermore, Lee et al[73] found that LPA interacts with T lymphocytes, B lymphocytes, acidic granulocytes, neutrophils, macrophages, mast cells, dendritic cells, and natural killer cells in the immune system and blood. Currently, no clinical treatment for LPA target is available, and the study of TME’s molecular mechanism is helpful to guide clinical treatment.
LPA is hydrolyzed and inactivated by LPPs. Studies have found that LPP1 and LPP3 are reduced in various tumor tissues[74]. LPPs activate ERK signaling by thrombin; induce LPP1 and LPP2 overexpression; and attenuate cell migration, cell differentiation, and angiogenesis[75]. Pilquil et al[76] found that increased LPP1 expression weakens PLD activation, which is an intermediate substance necessary for LPA to stimulate cell migration. LPP1 also weakens fibroblast migration. Tanyi et al[77] found that LPP3 reduces cell apoptosis, decreases the migration ability of transfected LPP3 cells, and slows down tumor growth in vivo and in vitro.
LPA1: LPA1 is the most widely expressed Edg LPAR in tissues[69]. LPA signaling through LPA1 regulates a variety of malignant properties in cancer cells[78]. Murph et al[79] found that LPA1 downregulates the tumor suppressor gene p53 and weakens its inhibitory effect. Marshall et al[80] found that the tumor-suppressor gene Nm23 could inhibit LPA1 expression. Additionally, Stadler et al[81] found that LPA1 is a signaling receptor downstream of fibroblast growth factor receptor 4 (FGFR4) that promotes cell transformation of cells into fibroblasts, which are one of the main components of TME matrix. LPA1 preferentially binds to Gα Q proteins in tumors to activate PKC. PKC is involved in many cellular processes, including proliferation and metastasis. Valdés-Rives et al[82] found that when the LPA1/PKCα signaling pathway is blocked, the number of cells is reduced; this finding suggests a correlation between LPA1 and PKCα in glioblastoma multiforme growth. Stadler et al[81] found that patients with high expression of the LPA1 receptor for R388 FGFR4 phenotype are more likely to develop cancer. Lin et al[83] found that LPA1 signaling mediates tumor lymphangiogenesis by promoting calreticulin expression in prostate cancer. Elevated LPA1 receptors also contribute to cancer development.
LPA2: LPA2 is elevated in tumor tissue[84]. Studies showed that LPA2 is associated with many human tumors, and the binding of LPA2 with its ligand, LPA, can activate the LPA signaling pathway and promote cell proliferation and malignant transformation. For example, the high expression level of the LPA2 receptor in breast cancer suggests a poor prognosis[85]. The high expression of LPA2 mRNA in HCC is related to the low differentiation of cancer cells[86], and the high expression of LPA2 receptor in colon cancer cells promotes the acquisition of drug resistance and the failure of anticancer drugs[87]. LPA2-mediated signaling plays an important role in the enhancement of the chemoresistance of A375 cells treated with anticancer drugs[78]. Ren et al[88] transfected SGC-7901 gastric cancer (GC) cells with LPA2 expression vector and found that the expression of E-cadherin gradually decreases and the expression of vimentin gradually increases with the increase in LPA2 level. These findings suggest that LPA2 is involved in the epithelial-mesenchymal transition (EMT) process of GC cells. GC cells with increased LPA2 level are likely to metastasize. Dong et al[89] believed that an effective drug that can inhibit LPA2gene expression, inhibit GC cell proliferation, and promote apoptosis might be a potential new target for GC treatment. Xu et al[90] found that thyroid receptor interacting protein 6 activates LPA2 and its downstream signal and therefore promotes cell adhesion and migration. The carcinogenic mechanism of LPA2 is still unknown, and most studies have focused on the LPA stimulation of the expression of cytokines, such as IL-6, VEGF, hypoxia-inducible factor 1α, C-MyC, cyclin D1, Kruppel-like factor 5, and COX-2. Moreover, Na+/H+ regulatory factor 2 (NHERF-2) may enhance LPA2gene expression and other LPA-induced cellular processes[91].
LPA3: Research found that LPA3 promotes cancer cell proliferation and metastasis. Zhao et al[92] found that the high expression of the LPA3 protein is considerably correlated with the occurrence and recurrence of epithelial ovarian cancer. Hayashi et al[93] and Kitayoshi et al[94] found that LPA3 inhibits tumor cell migration. Sun et al[95] found that LPA3 overexpression is associated with lymph node metastasis and the loss of the expression of estrogen receptor, progesterone receptor, and human EGFR2. Studies found that LPA3 may be related to the activation of the YAP protein in breast cancer and that LPA3 overexpression may promote the activation of YAP protein and the proliferation and metastasis of breast cancer cells. Fang et al[96] found that LPA3 affects B cell lymphoma (Bcl)-2 and Bax expression; therefore, it affects the Bcl-2/Bax ratio, inhibits the apoptosis of ovarian cancer cells, and promotes the development of ovarian cancer. The vasodilator-stimulated phospho-protein phosphorylation induced by LPA receptor is a key mediator of migration initiation. LPA3 plays a role in cellular motility and may contribute to cell invasion and metastasis[97].
LPA4-6: LPA4 may be involved in the invasion and metastasis of breast cancer cells, and the migration and invasion ability may involve the regulation of MMP2 and MMP9 protein expression. Takara et al[98] found that LPA4 is involved in the formation of vascular networks. LPA4 activation induces the subcellular binding of circumferential actin and enhances the linear adhesion of vascular-endothelial cadherin in endothelial cells. Studies found that LPA5 knockout cells show high motor activity. The gelatinase spectrum shows that LPA5 inhibits the activation of MMP2. LPA5 also inhibits the cellular motility of endothelial cells, which is correlated to the expression level of the VEGF gene[99]. However, Tsujino et al[100] found no mutation in the LPA5gene in colon cancer cells DLD1, SW480, HCT116, CACO-2, SW48, and LoVo. LPA6-mediated tube formation, which reflects the stabilization of barrier integrity, was confirmed by in vitro angiogenesis assay. By contrast, LPA6-mediated protective actions are associated with the activation of Src and Rap1 and attenuated by the abrogation of their activities[101]. A considerable correlation between LPA6 and PIM-3 expression levels is also observed in patients with HCC. Furthermore, the biological roles of LPA4-6 remain unknown[102,103].
Pancreatic cancer (PC) is the seventh most common cancer worldwide and causes more than 300000 deaths a year[104]. The 5-year survival rate of PC is only 3%-5%. In the early stages of PC, it directly invades peripancreatic tissues or metastasizes to organs near and far via lymphatic and/or blood vessels. More than 80% of patients with PC are initially diagnosed at advanced stages, lose the chance of surgical treatment, and have poor radiotherapy and chemotherapy effects. In 1996, Guo et al[105] found that the expression of KAI1/CD82 mRNA in early pancreatic tumors (I and II) is significantly higher than that in advanced tumors (III and IV) with lymph node metastasis or distant metastasis (P < 0.01), and the KAI1 mRNA level in poorly differentiated tumors is significantly higher than that in moderately differentiated or well-differentiated tumors (P < 0.05). Friess et al[106] and Xu et al[107] also found similar results. Subsequent studies have shown that low KAI1/CD82 level is associated with the inhibition of PC cell invasion and metastasis, and the KAIl/CD82 gene may control PC cell metastasis by inhibiting cancer cell invasion and motor function[108-111].
KAIl/CD82 protein, a member of TM4SF, has been accepted for its inhibitory effect on tumor metastasis; the mechanism of this effect has not yet been clearly explained, but it may be related to its localization on the cell membrane, extensive glycosylation, and cell-cell and cell-extracellular matrix interactions. Mashimo et al[112] found that the loss of p53 leads to the downregulation of the KAI1/CD82 gene and promotes cancer metastasis. KAI1 may inhibit the metastasis of the PC cells PANC-1 and Miapaca-2, caused by hepatocyte growth factor (HGF) by downregulating sphingosine kinase (SphK) expression. After they were infected with the KAI1 gene, the PANC-1 and Miapaca-2 cells induced by HGF had decreased invasive ability in the Boyden chamber assay. KAI1 overexpression in cells leads to the deactivation of SphK and a decreased level of intracellular sphingosine-1-phosphate[108]. Liu et al[108] found that KAI1/CD82 induces the downregulation of VEGF-C expression through the Src/STAT3 signaling pathway, which may also inhibit the lymph node metastasis of PC. Wu et al[111] found that KAI1 induces the expression of the autophagy proteins LC3 and Beclin1, and further confirmed that KAI1 could induce autophagy in the human PC cell line MiAPACA-2 and therefore promote cell apoptosis and inhibit proliferation. EMT plays an important role in the pathogenesis of PC. KAI1 reverses the expression of EMT-related factors, such as Snail, Vimentin, MMP2, and MMP9 (P < 0.05), and inhibits PC cell metastasis and invasion. In conclusion, KAI1 may be a new potential therapeutic target for PC in the future.
HCC is a common malignant tumor with the second highest mortality rate in China. Rapid intrahepatic and extrahepatic metastases lead to poor prognosis[113]. Zhang et al[114] found that the combined detection of KAI1 and VEGF can greatly improve the diagnostic efficiency for HCC. Mu et al[115] found that KAI1/CD82 suppresses the HGF-induced migration of hepatoma cells via SphK1 downregulation. HGF induces hepatoma cell migration through cellular SphK1 activation. The adenovirus-mediated gene transfer of KAI1 downregulates SphK1 expression and suppresses the HGF-induced migration of SMMC-7721 human HCC cells. Guo et al[116] found that the wTP53 fusion gene and JunB inhibit tumor cell invasiveness and promote tumor cell apoptosis by regulating KAI1/CD82 expression. Si et al[117] and Yang et al[118] found that changing KAI1 expression could alter the migration and invasion ability of MHCC97-H in HCC cells. Xu et al[119] found that KAI1 is negatively correlated with tumor grade, venous invasion, lymph node metastasis, intrahepatic metastasis, and TNM stage and positively correlated with patients’ OS. KAI1/CD82 may also play an important role in HCC metastasis and prognosis.
GC is one of the most common malignant tumors. Although GC-related morbidity has shown a downward trend in recent years, the mortality rate remains high[120,121]. KAI1 has been studied to identify novel therapeutic targets[122-126]. Ilhan et al[122] and Knoener et al[123] found that KAI1/CD82 is negative in all tissues with distant metastasis or tissues in stage IV GC with statistical significance (P < 0.05). KAI1 inhibits tumor growth and metastasis and is a prognostic factor for patients with GC. Hinoda et al[124] found that the positive rate of KAI1/CD82 in patients with stages Ia-IIIa GC is 16.6% (8/48), and all patients with stages IIIb-IVb GC are negative for KAI1/CD82 (0%, 0/25; P = 0.05). KAI1/CD82 is highly expressed in normal gastric epithelial cells. In GC, KAI1/CD82 expression decreases with increased tumor differentiation, tumor invasion depth, and lymph node metastasis[127,128]. Guan et al[129] found that reduced KAI1/CD82 expression promotes lymph node metastasis and liver metastasis in patients with GC. The detection of KAI1/CD82 mRNA expression level can be used as a prognostic index for patients with GC.
Colorectal cancer (CRC) is a common malignant tumor, and metastasis is the main cause of its poor prognosis. KAI1 may affect cellular connectivity and may be related to its metastasis. KAI1 may be a new therapeutic target for CRC[130,131]. KAI1 mRNA and protein are increased in early CRC tumors, decreased in late CRC tumors, and no longer expressed in distant metastasis[132]. Integrin-α3 and TAp73 regulate CRC invasion and metastasis by regulating KAI1 transcription[133,134].
The expression of ATX in PC remains unclear, and its molecular biological mechanism has not yet been reported. Ryder et al[135] and Nakai et al[136] found that ATX expression is increased in PC tissues, but it is more increased in chronic pancreatitis or pancreatic cysts than in PC. Quan et al[137] found that TNF-α, NF-κB, Wnt/β-catenin pathway, V-Jun, EGF, and B-FGF are all activated or abnormally expressed in PC tissues, which may provide a direction for future research on mechanisms. LPA activates downstream signaling pathways, such as PI3K/AKT, RAS/ERK, Rho, and Hippo, and promotes PC cell proliferation, migration, and invasion[138,139]. Additionally, LPA is remarkably increased in the serum and ascites[140,141], which suggests that ATX activity is elevated in patients with PC.
ATX catalyzes LPA synthesis from LPC and exerts biological effects through the receptors LPA1-6. Fukushima et al[142] found that the invasion ability of PANC-R9 cells is 15 times that of PANC-1 cells, LPA1 expression in PANC-R9 cells is remarkably higher than that in PANC-1 cells, and LPA3 is decreased. Kato et al[143] also found that LPA1 and LPA3 play opposite roles in PC cell migration. Tsujiuchi et al[144], Komachi et al[145], and Yamada et al[146] found that LPA1 induces PC cell migration. Liao et al[141] and Yoshikawa et al[147] found that LPA2 may induce PC cell migration by enhancing the proto-oncogene K-RAS pathway. However, Komachi et al[145] found that LPA2 may inhibit PC cell migration through the conjugated G12/13/Rho signaling pathway. Ishii et al[148] conducted a cell activity assay after LPARs were knocked out from PANC-1 cells (PANC-SH4, PANC-SH5, and PANC-SH6 cells). They found that PANC-SH4 and PANC-SH5 enhance cell migration ability, whereas PANC-SH6 inhibits cell migration. Currently, few studies have been conducted on the molecular biology of LPAR and PC, and further research is needed.
The main risk factors for HCC are hepatitis virus infection; alcohol consumption; and metabolic disorders, such as obesity, diabetes, and non-alcoholic fatty liver disease[149]. The abnormal expression of the ATX-LPA axis may cause liver metabolism disorder and induce steatohepatitis and liver cancer[150,151]. The ATX-LPA axis is currently considered one of the most promising signaling pathways in liver cancer[152]. Watanabe et al[153] found elevated ATX and LPA levels in hepatic fibrosis tissues. Memet et al[149] found that high expression of ATX in HCC is an independent prognostic factor (HR = 13.70, 95%CI: 3.26-57.62, P = 0.0004), and high expression of ATX (+3) also increases the risk of death by eight-fold. Wu et al[154] found that ATX is significantly elevated in Hep3B and Huh7 cells. Park et al[155] found that LPA1 is significantly elevated in liver cancer. LPA3 may be highly expressed in HCC tissues through the lPA3-GI-ERK signaling pathway[156]. Enooku et al[86] found that increased LPA2 mRNA level may be associated with the low differentiation degree of HCC. Okabe et al[157] found that LPA3 induces the invasion of rabbit RH7777 hepatoma cells. LPA6 is not expressed in normal tissues but is expressed in liver cancer tissues. Zheng et al[158] found that nuclear receptor coactivator 3 induces the acetylation of histone 3-LYS-27 at the LPA6 site after HGF treatment and inhibits LPA6 transcription. High LPA6 expression promotes HCC proliferation. Lippolis et al[159] found that high LPA6 expression promotes the development of HCC with poor prognosis. Gnocchi et al[160] and Mazzocca et al[161] found that LPA6 may be an important therapeutic target for HCC, although LPA6 overexpression promotes HCC cell growth.
The role of ATX-LPA axis in GC invasion and metastasis remains to be explored. Zeng et al[162] found that LPA is increased in GC tissue samples with peritoneal metastasis (P = 0.046) and is significantly increased in ascites (P < 0.001). Serum LPA decreases after chemotherapy (P = 0.028). PFS and OS are significantly decreased in an ascites LPA > 24000 ng/mL group (P < 0.001). Ramachandran et al[163] and Shida et al[164] found that LPA upregulates SphK1 through the ERK1 signaling pathway. Kim et al[165] found that LPA can induce uPAR to stimulate the downstream signaling pathways, rho-family GTPase, JNK, AP-1, and NF-κB. Budnik[18] found that LPA upregulates human epidermal growth factor receptor 2 expression in GC cells and promotes GC cell invasion. LPA promotes cell proliferation, but the molecular biological mechanism between LPA and GC still needs further exploration, and LPA may become a new target for GC treatment.
CRC is the fourth leading cause of cancer deaths in the world[166]. Kazama et al[167] found that ATX overexpression is associated with tumor angiogenesis in the early stage of colon cancer. LPA may stimulate the proliferation and migration of CRC cells through the EGFR pathway. It may also promote hcT-116 colon cancer cell migration by regulating the cell cycle through the rho-Rock and STAT3 pathways. Whether LPA1 stimulates colon cancer cell proliferation remains controversial. A study found that HCT116 and LS174T cells with LPA1 knockout do not affect the spread of cancer cells[168], and DLD cancer cells are affected when they spread[169,170]. LPA2 promotes the spread and migration of colon cancer by regulating the NHERF-2 pathway[171,172]; therefore, LPA2 may be one of the therapeutic targets for CRC in the future[173]. Shida et al[174] found that LPA3 mRNA is micro-expressed in normal and tumor tissues. Fukui et al[175] found that the expression levels of VEGF-A and VEGF-C are increased in HCT-SH3-3 cells with LPA3 knockout, and LPA3 inhibits the metastasis of HCT116 colon cancer cells. Takahashi et al[176] found that LPA4 and LPA6 inhibit the activities of DLD1 and HCT116 colon cancer cells. Studies on ATX-LPA axis target inhibitors and colon malignancies are still few and require further exploration.
Most studies have shown that KAI1/CD82 inhibits tumor metastasis and migration, but knowledge about KAI1/CD82 antibody reagents is still lacking. Custer et al[177] found that the KAI1 polyclonal antibody produced by rabbits is expressed similarly in normal tissues of mice and humans and could specifically detect mouse KAI1/CD82 protein. KAI1/CD82 is a novel tumor therapeutic target, and more KAI1/CD82 antibodies are expected to be developed in the future[178,179].
ATX inhibitors decrease serum LPA levels by more than 95%[180]. Oral ATX inhibitors have better bioavailability owing to their low hydrophobicity and slow degradation in vivo[181]. PF-8380 is the first ATX inhibitor to permanently reduce LPA levels in vivo. Bhave et al[182] and Schleicher et al[183] found that PF-8380 reduces lPA-induced inflammation and delays tumor growth for more than 20 d in a mouse model of glioblastoma multiforme. Tang et al[184] found that the inhibition of GLPG1690 on ATX enhances the efficacy of chemoradiotherapy in mouse breast cancer models. ONO-8430506 is also a highly effective ATX inhibitor, and the oral administration of 30 mg/kg ONO-8430506 effectively reduces serum ATX and LPA levels in rats[185]. ONO-8430506 in combination with adriamycin delays the growth time of orthotopic 4T1 breast tumors in 60% Balb/C mice by about 10 d and reduces the growth time of 70% tumors by about 17 d[186,187]. Cholera toxin treatment increases the expression of the anti-inflammatory cytokines IL-4 and IL-10 and inhibits ATX mRNA[188], and the knockdown of ATX mRNA inhibits the growth of Hep3B and Huh7 hepatoma cells[189]. Gupte et al[190] found that ATX inhibitors, such as 4-pentadecylbenzylphosphonic acid, reduce plasma LPA levels by 50%. Plasma LPA in ATX-KO mice lacking dominant heterozygosity is reduced by 50%. ATX inhibitors have not shown remarkable side effects to date.
Antibody interventional therapy is superior to traditional therapy, and its antibody bioavailability and receptor binding are longer than other therapies[191]. Goldshmit et al[192] found that monoclonal antibody B3 can reduce inflammation and glial cell death and improve neuronal function. Monoclonal antibody B3, also known as lpathomab, reduces IL-6 expression and the lesion area and has improved function in a mouse model of traumatic brain injury[193].
Many LPA receptor antagonists have been found, but few work in vivo. LPA receptor antagonists are divided into lipid and small-molecule inhibitors, which are derived from fibrosis model studies[194]. BrP-LPA, a pan-LPAR antagonist, was used to treat breast MDA-MB-231 cancer cells[195]. Through LPAR2, BrP-LPA may also sensitize vascular endothelial cells in mouse GL-261 glioma cells to improve malignant glioma response to radiation therapy[183]. LPA accelerates pulmonary fibrosis through LPA1, and the LPA1 antagonist AM966 can inhibit bleomycin-induced idiopathic pulmonary fibrosis. Zhao et al[196] found that Ki16425 (LPA1 and LPA3 antagonist) and ono7300243 (LPA1 antagonist) completely block LPA-induced actions. Recently, lysophospholipid GPCR genes have been used to develop receptor subtype-selective agonists and antagonists. The discovery of FTY720, a novel immune modulator, along with other chemical tools, has provided a means of elucidating the functions of each lysophospholipid GPCR on an organ and the whole body level[197]. In some cancers, targeting LPAR5 is considered a good option against cancer development[87,198]. LPAR5 antagonist TCLPA5 attenuates the proliferation and migration of thyroid carcinoma cells[199]. In addition, the loss of LPA5 in mouse B16-F10 melanoma results in fewer lung metastases[200], which suggests that the drug inhibition of LPA5 can also control melanoma-mediated metastasis. MP-LPA analogs exhibit an unanticipated pattern of partial agonist/antagonist activity for the LPA G protein-coupled receptor family and the intracellular LPA receptor peroxisome proliferator-activated receptors-γ[201]. Currently, all are based on LPA1, LPA2, or LPA1/3 dual antagonists[194]. However, the development of PAN-LPA receptor antagonists may be a more effective approach owing to the complexity of LPAR signals[202].
This paper systematically reviews the physiological functions of the KAI1/CD82 gene and the ATX-LPA axis in tumors, as well as their roles in digestive system tumors and targeted therapies. The results demonstrate that KAI1/CD82 is indeed an important inhibitor of tumor metastasis. Further elucidation of the molecular mechanism and regulatory network of KAI1/CD82 and the inhibition of tumor metastasis is needed to discover the molecular markers of pancreatic tumor metastasis, adopt effective strategies to treat PC and prevent PC metastasis, and provide a new approach for the diagnosis and treatment of patients with refractory PC. Although the ATX-LPA axis is considered an important target of cancer, its clinical application is still faced with obstacles. LPA is degraded quickly in the body, and many other factors, such as diet, smoking, and alcohol consumption, can affect the detection results. Other lipids may also generate LPA during extraction, storage, and detection. Therefore, many technical problems need to be overcome in LPA detection. In recent years, clinical trials on the ATX-LPA axis have begun. LPA monoclonal antibodies, LPA receptor antagonists, and ATX inhibitors may become feasible treatment measures. Moreover, ATX-LPA axis-targeted therapy may affect the efficacy of existing chemical drugs. Therefore, an in-depth exploration of specific biomarkers related to LPA activity should be conducted to track disease progression during LPA treatment and ensure the rational application of drugs.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author's Membership in Professional Societies: Director, chief physician, Department of Gastroenterology, General Hospital of Northern Theater Command; Director of biliopancreatic and Endoscopic Diagnosis and Treatment Center of PLA and Key Laboratory of Liaoning Province; Director of Liaoning Institute of Digestive Diseases; Vice chairman of Pancreatic Disease Branch, Chinese Medical Doctor Association; Standing member of Gastroenterology Society, Chinese Medical Association; Standing member of Gastroenterologist Branch, Chinese Medical Doctor Association; Leader of pancreatology Group, Gastroenterology Branch, Chinese Medical Association.
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Corrales FJ, Spain; Corrales FJ, Spain; Jiang L, China S-Editor: Wang JJ L-Editor: Wang TQ P-Editor: Wang JJ
| 1. | Dong JT, Lamb PW, Rinker-Schaeffer CW, Vukanovic J, Ichikawa T, Isaacs JT, Barrett JC. KAI1, a metastasis suppressor gene for prostate cancer on human chromosome 11p11.2. Science. 1995;268:884-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 548] [Cited by in RCA: 546] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 2. | Malik FA, Sanders AJ, Jiang WG. KAI-1/CD82, the molecule and clinical implication in cancer and cancer metastasis. Histol Histopathol. 2009;24:519-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 3. | Dong JT, Isaacs WB, Barrett JC, Isaacs JT. Genomic organization of the human KAI1 metastasis-suppressor gene. Genomics. 1997;41:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 54] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 4. | Zhang XA, He B, Zhou B, Liu L. Requirement of the p130CAS-Crk coupling for metastasis suppressor KAI1/CD82-mediated inhibition of cell migration. J Biol Chem. 2003;278:27319-27328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 66] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 5. | Tang X, Benesch MGK, Brindley DN. Role of the autotaxin-lysophosphatidate axis in the development of resistance to cancer therapy. Biochim Biophys Acta Mol Cell Biol Lipids. 2020;1865:158716. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 6. | Lee HY, Bae GU, Jung ID, Lee JS, Kim YK, Noh SH, Stracke ML, Park CG, Lee HW, Han JW. Autotaxin promotes motility via G protein-coupled phosphoinositide 3-kinase gamma in human melanoma cells. FEBS Lett. 2002;515:137-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 7. | Leblanc R, Peyruchaud O. New insights into the autotaxin/LPA axis in cancer development and metastasis. Exp Cell Res. 2015;333:183-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 8. | Benesch MG, Tang X, Venkatraman G, Bekele RT, Brindley DN. Recent advances in targeting the autotaxin-lysophosphatidate-lipid phosphate phosphatase axis in vivo. J Biomed Res. 2016;30:272-284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 9. | Houben AJ, Moolenaar WH. Autotaxin and LPA receptor signaling in cancer. Cancer Metastasis Rev. 2011;30:557-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 192] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 10. | Stefan C, Jansen S, Bollen M. NPP-type ectophosphodiesterases: unity in diversity. Trends Biochem Sci. 2005;30:542-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 297] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 11. | Nishimasu H, Okudaira S, Hama K, Mihara E, Dohmae N, Inoue A, Ishitani R, Takagi J, Aoki J, Nureki O. Crystal structure of autotaxin and insight into GPCR activation by lipid mediators. Nat Struct Mol Biol. 2011;18:205-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 197] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 12. | Boutin JA, Ferry G. Autotaxin. Cell Mol Life Sci. 2009;66:3009-3021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 13. | Hashimoto T, Okudaira S, Igarashi K, Hama K, Yatomi Y, Aoki J. Identification and biochemical characterization of a novel autotaxin isoform, ATXδ, with a four-amino acid deletion. J Biochem. 2012;151:89-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 14. | Li H, Zhao Z, Wei G, Yan L, Wang D, Zhang H, Sandusky GE, Turk J, Xu Y. Group VIA phospholipase A2 in both host and tumor cells is involved in ovarian cancer development. FASEB J. 2010;24:4103-4116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 15. | Zhao X, Wang D, Zhao Z, Xiao Y, Sengupta S, Zhang R, Lauber K, Wesselborg S, Feng L, Rose TM, Shen Y, Zhang J, Prestwich G, Xu Y. Caspase-3-dependent activation of calcium-independent phospholipase A2 enhances cell migration in non-apoptotic ovarian cancer cells. J Biol Chem. 2006;281:29357-29368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 91] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 16. | Fourcade O, Simon MF, Viodé C, Rugani N, Leballe F, Ragab A, Fournié B, Sarda L, Chap H. Secretory phospholipase A2 generates the novel lipid mediator lysophosphatidic acid in membrane microvesicles shed from activated cells. Cell. 1995;80:919-927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 398] [Cited by in RCA: 419] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 17. | Moolenaar WH. Lysophosphatidic acid, a multifunctional phospholipid messenger. J Biol Chem. 1995;270:12949-12952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 453] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 18. | Budnik LT. Lysophosphatidic acid, LPA: a bad boy becomes good. Reprod Biol Endocrinol. 2003;1:37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 19. | Wang FQ, Ariztia EV, Boyd LR, Horton FR, Smicun Y, Hetherington JA, Smith PJ, Fishman DA. Lysophosphatidic acid (LPA) effects on endometrial carcinoma in vitro proliferation, invasion, and matrix metalloproteinase activity. Gynecol Oncol. 2010;117:88-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Aoki J. Mechanisms of lysophosphatidic acid production. Semin Cell Dev Biol. 2004;15:477-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 234] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 21. | Aoki J, Inoue A, Okudaira S. Two pathways for lysophosphatidic acid production. Biochim Biophys Acta. 2008;1781:513-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 362] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 22. | Yung YC, Stoddard NC, Chun J. LPA receptor signaling: pharmacology, physiology, and pathophysiology. J Lipid Res. 2014;55:1192-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 565] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 23. | Zhang G, Cheng Y, Zhang Q, Li X, Zhou J, Wang J, Wei L. ATXLPA axis facilitates estrogeninduced endometrial cancer cell proliferation via MAPK/ERK signaling pathway. Mol Med Rep. 2018;17:4245-4252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 24. | Riaz A, Huang Y, Johansson S. G-Protein-Coupled Lysophosphatidic Acid Receptors and Their Regulation of AKT Signaling. Int J Mol Sci. 2016;17:215. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 25. | Valdés-Rives SA, González-Arenas A. Autotaxin-Lysophosphatidic Acid: From Inflammation to Cancer Development. Mediators Inflamm. 2017;2017:9173090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 80] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 26. | Jackson P, Marreiros A, Russell PJ. KAI1 tetraspanin and metastasis suppressor. Int J Biochem Cell Biol. 2005;37:530-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Zhu J, Miao C, Liu S, Tian Y, Zhang C, Liang C, Xu A, Cao Q, Wang Z. Prognostic role of CD82/KAI1 in multiple human malignant neoplasms: a meta-analysis of 31 studies. Onco Targets Ther. 2017;10:5805-5816. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 28. | Waterhouse R, Ha C, Dveksler GS. Murine CD9 is the receptor for pregnancy-specific glycoprotein 17. J Exp Med. 2002;195:277-282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 72] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 29. | Crotta S, Stilla A, Wack A, D'Andrea A, Nuti S, D'Oro U, Mosca M, Filliponi F, Brunetto RM, Bonino F, Abrignani S, Valiante NM. Inhibition of natural killer cells through engagement of CD81 by the major hepatitis C virus envelope protein. J Exp Med. 2002;195:35-41. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 342] [Cited by in RCA: 325] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 30. | Nojima Y, Hirose T, Tachibana K, Tanaka T, Shi L, Doshen J, Freeman GJ, Schlossman SF, Morimoto C. The 4F9 antigen is a member of the tetra spans transmembrane protein family and functions as an accessory molecule in T cell activation and adhesion. Cell Immunol. 1993;152:249-260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 31. | Ono M, Handa K, Withers DA, Hakomori S. Motility inhibition and apoptosis are induced by metastasis-suppressing gene product CD82 and its analogue CD9, with concurrent glycosylation. Cancer Res. 1999;59:2335-2339. [PubMed] |
| 32. | Lee JH, Seo YW, Park SR, Kim YJ, Kim KK. Expression of a splice variant of KAI1, a tumor metastasis suppressor gene, influences tumor invasion and progression. Cancer Res. 2003;63:7247-7255. [PubMed] |
| 33. | Iwata S, Kobayashi H, Miyake-Nishijima R, Sasaki T, Souta-Kuribara A, Nori M, Hosono O, Kawasaki H, Tanaka H, Morimoto C. Distinctive signaling pathways through CD82 and beta1 integrins in human T cells. Eur J Immunol. 2002;32:1328-1337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 34. | Mannion BA, Berditchevski F, Kraeft SK, Chen LB, Hemler ME. Transmembrane-4 superfamily proteins CD81 (TAPA-1), CD82, CD63, and CD53 specifically associated with integrin alpha 4 beta 1 (CD49d/CD29). J Immunol. 1996;157:2039-2047. [PubMed] |
| 35. | Sugiura T, Berditchevski F. Function of alpha3beta1-tetraspanin protein complexes in tumor cell invasion. Evidence for the role of the complexes in production of matrix metalloproteinase 2 (MMP-2). J Cell Biol. 1999;146:1375-1389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 153] [Cited by in RCA: 158] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 36. | Odintsova E, Sugiura T, Berditchevski F. Attenuation of EGF receptor signaling by a metastasis suppressor, the tetraspanin CD82/KAI-1. Curr Biol. 2000;10:1009-1012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 162] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 37. | Liu WM, Zhang XA. KAI1/CD82, a tumor metastasis suppressor. Cancer Lett. 2006;240:183-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 108] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 38. | Brindley DN, Lin FT, Tigyi GJ. Role of the autotaxin-lysophosphatidate axis in cancer resistance to chemotherapy and radiotherapy. Biochim Biophys Acta. 2013;1831:74-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 104] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 39. | Bekele R, David S. Role of autotaxin and lysophosphatidate in cancer progression and resistance to chemotherapy and radiotherapy. Clin Lipidol. 2012;7:313-328. [RCA] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 40. | Tokumura A, Majima E, Kariya Y, Tominaga K, Kogure K, Yasuda K, Fukuzawa K. Identification of human plasma lysophospholipase D, a lysophosphatidic acid-producing enzyme, as autotaxin, a multifunctional phosphodiesterase. J Biol Chem. 2002;277:39436-39442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 576] [Cited by in RCA: 603] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 41. | Umezu-Goto M, Kishi Y, Taira A, Hama K, Dohmae N, Takio K, Yamori T, Mills GB, Inoue K, Aoki J, Arai H. Autotaxin has lysophospholipase D activity leading to tumor cell growth and motility by lysophosphatidic acid production. J Cell Biol. 2002;158:227-233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 741] [Cited by in RCA: 770] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 42. | Gotoh M, Fujiwara Y, Yue J, Liu J, Lee S, Fells J, Uchiyama A, Murakami-Murofushi K, Kennel S, Wall J, Patil R, Gupte R, Balazs L, Miller DD, Tigyi GJ. Controlling cancer through the autotaxin-lysophosphatidic acid receptor axis. Biochem Soc Trans. 2012;40:31-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 43. | Kawagoe H, Stracke ML, Nakamura H, Sano K. Expression and transcriptional regulation of the PD-Ialpha/autotaxin gene in neuroblastoma. Cancer Res. 1997;57:2516-2521. [PubMed] |
| 44. | Hoelzinger DB, Mariani L, Weis J, Woyke T, Berens TJ, McDonough WS, Sloan A, Coons SW, Berens ME. Gene expression profile of glioblastoma multiforme invasive phenotype points to new therapeutic targets. Neoplasia. 2005;7:7-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 252] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 45. | Kostadinova L, Shive CL, Anthony DD. Elevated Autotaxin and LPA Levels During Chronic Viral Hepatitis and Hepatocellular Carcinoma Associate with Systemic Immune Activation. Cancers (Basel). 2019;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 46. | Masuda A, Nakamura K, Izutsu K, Igarashi K, Ohkawa R, Jona M, Higashi K, Yokota H, Okudaira S, Kishimoto T, Watanabe T, Koike Y, Ikeda H, Kozai Y, Kurokawa M, Aoki J, Yatomi Y. Serum autotaxin measurement in haematological malignancies: a promising marker for follicular lymphoma. Br J Haematol. 2008;143:60-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 93] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 47. | Stracke ML, Krutzsch HC, Unsworth EJ, Arestad A, Cioce V, Schiffmann E, Liotta LA. Identification, purification, and partial sequence analysis of autotaxin, a novel motility-stimulating protein. J Biol Chem. 1992;267:2524-2529. [PubMed] |
| 48. | Stassar MJ, Devitt G, Brosius M, Rinnab L, Prang J, Schradin T, Simon J, Petersen S, Kopp-Schneider A, Zöller M. Identification of human renal cell carcinoma associated genes by suppression subtractive hybridization. Br J Cancer. 2001;85:1372-1382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 117] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 49. | Kehlen A, Englert N, Seifert A, Klonisch T, Dralle H, Langner J, Hoang-Vu C. Expression, regulation and function of autotaxin in thyroid carcinomas. Int J Cancer. 2004;109:833-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 89] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 50. | Yang Y, Mou Lj, Liu N, Tsao MS. Autotaxin expression in non-small-cell lung cancer. Am J Respir Cell Mol Biol. 1999;21:216-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 51. | Su SC, Hu X, Kenney PA, Merrill MM, Babaian KN, Zhang XY, Maity T, Yang SF, Lin X, Wood CG. Autotaxin-lysophosphatidic acid signaling axis mediates tumorigenesis and development of acquired resistance to sunitinib in renal cell carcinoma. Clin Cancer Res. 2013;19:6461-6472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 52. | Federico L, Ren H, Mueller PA, Wu T, Liu S, Popovic J, Blalock EM, Sunkara M, Ovaa H, Albers HM, Mills GB, Morris AJ, Smyth SS. Autotaxin and its product lysophosphatidic acid suppress brown adipose differentiation and promote diet-induced obesity in mice. Mol Endocrinol. 2012;26:786-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 57] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 53. | Benesch MG, Tang X, Dewald J, Dong WF, Mackey JR, Hemmings DG, McMullen TP, Brindley DN. Tumor-induced inflammation in mammary adipose tissue stimulates a vicious cycle of autotaxin expression and breast cancer progression. FASEB J. 2015;29:3990-4000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 73] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 54. | Benesch MGK, MacIntyre ITK, McMullen TPW, Brindley DN. Coming of Age for Autotaxin and Lysophosphatidate Signaling: Clinical Applications for Preventing, Detecting and Targeting Tumor-Promoting Inflammation. Cancers (Basel). 2018;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 55. | Benesch MG, Ko YM, McMullen TP, Brindley DN. Autotaxin in the crosshairs: taking aim at cancer and other inflammatory conditions. FEBS Lett. 2014;588:2712-2727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 99] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 56. | Park GY, Lee YG, Berdyshev E, Nyenhuis S, Du J, Fu P, Gorshkova IA, Li Y, Chung S, Karpurapu M, Deng J, Ranjan R, Xiao L, Jaffe HA, Corbridge SJ, Kelly EA, Jarjour NN, Chun J, Prestwich GD, Kaffe E, Ninou I, Aidinis V, Morris AJ, Smyth SS, Ackerman SJ, Natarajan V, Christman JW. Autotaxin production of lysophosphatidic acid mediates allergic asthmatic inflammation. Am J Respir Crit Care Med. 2013;188:928-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 108] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 57. | Benesch MG, Zhao YY, Curtis JM, McMullen TP, Brindley DN. Regulation of autotaxin expression and secretion by lysophosphatidate and sphingosine 1-phosphate. J Lipid Res. 2015;56:1134-1144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 99] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 58. | Balogh A, Shimizu Y, Lee SC, Norman DD, Gangwar R, Bavaria M, Moon C, Shukla P, Rao R, Ray R, Naren AP, Banerjee S, Miller DD, Balazs L, Pelus L, Tigyi G. The autotaxin-LPA2 GPCR axis is modulated by γ-irradiation and facilitates DNA damage repair. Cell Signal. 2015;27:1751-1762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 59. | Meng G, Tang X, Yang Z, Benesch MGK, Marshall A, Murray D, Hemmings DG, Wuest F, McMullen TPW, Brindley DN. Implications for breast cancer treatment from increased autotaxin production in adipose tissue after radiotherapy. FASEB J. 2017;31:4064-4077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 60. | van Corven EJ, Groenink A, Jalink K, Eichholtz T, Moolenaar WH. Lysophosphatidate-induced cell proliferation: identification and dissection of signaling pathways mediated by G proteins. Cell. 1989;59:45-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 623] [Cited by in RCA: 678] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 61. | Merchant TE, Kasimos JN, de Graaf PW, Minsky BD, Gierke LW, Glonek T. Phospholipid profiles of human colon cancer using 31P magnetic resonance spectroscopy. Int J Colorectal Dis. 1991;6:121-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 59] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 62. | Xu Y, Gaudette DC, Boynton JD, Frankel A, Fang XJ, Sharma A, Hurteau J, Casey G, Goodbody A, Mellors A. Characterization of an ovarian cancer activating factor in ascites from ovarian cancer patients. Clin Cancer Res. 1995;1:1223-1232. [PubMed] |
| 63. | Deng W, Wang DA, Gosmanova E, Johnson LR, Tigyi G. LPA protects intestinal epithelial cells from apoptosis by inhibiting the mitochondrial pathway. Am J Physiol Gastrointest Liver Physiol. 2003;284:G821-G829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 68] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 64. | Sui Y, Yang Y, Wang J, Li Y, Ma H, Cai H, Liu X, Zhang Y, Wang S, Li Z, Zhang X, Liu R, Yan Y, Xue C, Shi X, Tan L, Ren J. Lysophosphatidic Acid Inhibits Apoptosis Induced by Cisplatin in Cervical Cancer Cells. Biomed Res Int. 2015;2015:598386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 65. | Sutphen R, Xu Y, Wilbanks GD, Fiorica J, Grendys EC Jr, LaPolla JP, Arango H, Hoffman MS, Martino M, Wakeley K, Griffin D, Blanco RW, Cantor AB, Xiao YJ, Krischer JP. Lysophospholipids are potential biomarkers of ovarian cancer. Cancer Epidemiol Biomarkers Prev. 2004;13:1185-1191. [PubMed] |
| 66. | Kim KS, Sengupta S, Berk M, Kwak YG, Escobar PF, Belinson J, Mok SC, Xu Y. Hypoxia enhances lysophosphatidic acid responsiveness in ovarian cancer cells and lysophosphatidic acid induces ovarian tumor metastasis in vivo. Cancer Res. 2006;66:7983-7990. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 123] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 67. | Ren J, Xiao YJ, Singh LS, Zhao X, Zhao Z, Feng L, Rose TM, Prestwich GD, Xu Y. Lysophosphatidic acid is constitutively produced by human peritoneal mesothelial cells and enhances adhesion, migration, and invasion of ovarian cancer cells. Cancer Res. 2006;66:3006-3014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 154] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 68. | Barekzi E, Roman J, Hise K, Georas S, Steinke JW. Lysophosphatidic acid stimulates inflammatory cascade in airway epithelial cells. Prostaglandins Leukot Essent Fatty Acids. 2006;74:357-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 69. | Spangelo BL, Jarvis WD. Lysophosphatidylcholine stimulates interleukin-6 release from rat anterior pituitary cells in vitro. Endocrinology. 1996;137:4419-4426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 70. | Seufferlein T, Rozengurt E. Lysophosphatidic acid stimulates tyrosine phosphorylation of focal adhesion kinase, paxillin, and p130. Signaling pathways and cross-talk with platelet-derived growth factor. J Biol Chem. 1994;269:9345-9351. [PubMed] |
| 71. | Korkina O, Dong Z, Marullo A, Warshaw G, Symons M, Ruggieri R. The MLK-related kinase (MRK) is a novel RhoC effector that mediates lysophosphatidic acid (LPA)-stimulated tumor cell invasion. J Biol Chem. 2013;288:5364-5373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 72. | Bian D, Mahanivong C, Yu J, Frisch SM, Pan ZK, Ye RD, Huang S. The G12/13-RhoA signaling pathway contributes to efficient lysophosphatidic acid-stimulated cell migration. Oncogene. 2006;25:2234-2244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 96] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 73. | Lee SC, Dacheux MA, Norman DD, Balázs L, Torres RM, Augelli-Szafran CE, Tigyi GJ. Regulation of Tumor Immunity by Lysophosphatidic Acid. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 74. | Chatterjee I, Humtsoe JO, Kohler EE, Sorio C, Wary KK. Lipid phosphate phosphatase-3 regulates tumor growth via β-catenin and CYCLIN-D1 signaling. Mol Cancer. 2011;10:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 75. | Samadi N, Bekele R, Capatos D, Venkatraman G, Sariahmetoglu M, Brindley DN. Regulation of lysophosphatidate signaling by autotaxin and lipid phosphate phosphatases with respect to tumor progression, angiogenesis, metastasis and chemo-resistance. Biochimie. 2011;93:61-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 89] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 76. | Pilquil C, Dewald J, Cherney A, Gorshkova I, Tigyi G, English D, Natarajan V, Brindley DN. Lipid phosphate phosphatase-1 regulates lysophosphatidate-induced fibroblast migration by controlling phospholipase D2-dependent phosphatidate generation. J Biol Chem. 2006;281:38418-38429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 48] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 77. | Tanyi JL, Hasegawa Y, Lapushin R, Morris AJ, Wolf JK, Berchuck A, Lu K, Smith DI, Kalli K, Hartmann LC, McCune K, Fishman D, Broaddus R, Cheng KW, Atkinson EN, Yamal JM, Bast RC, Felix EA, Newman RA, Mills GB. Role of decreased levels of lipid phosphate phosphatase-1 in accumulation of lysophosphatidic acid in ovarian cancer. Clin Cancer Res. 2003;9:3534-3545. [PubMed] |
| 78. | Minami K, Ueda N, Ishimoto K, Tsujiuchi T. Lysophosphatidic acid receptor-2 (LPA2)-mediated signaling enhances chemoresistance in melanoma cells treated with anticancer drugs. Mol Cell Biochem. 2020;469:89-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 79. | Murph MM, Hurst-Kennedy J, Newton V, Brindley DN, Radhakrishna H. Lysophosphatidic acid decreases the nuclear localization and cellular abundance of the p53 tumor suppressor in A549 lung carcinoma cells. Mol Cancer Res. 2007;5:1201-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 80. | Marshall JC, Collins J, Marino N, Steeg P. The Nm23-H1 metastasis suppressor as a translational target. Eur J Cancer. 2010;46:1278-1282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 81. | Stadler CR, Knyazev P, Bange J, Ullrich A. FGFR4 GLY388 isotype suppresses motility of MDA-MB-231 breast cancer cells by EDG-2 gene repression. Cell Signal. 2006;18:783-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 82. | Valdés-Rives SA, de la Fuente-Granada M, Velasco-Velázquez MA, González-Flores O, González-Arenas A. LPA1 receptor activation induces PKCα nuclear translocation in glioblastoma cells. Int J Biochem Cell Biol. 2019;110:91-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 83. | Lin YC, Chen CC, Chen WM, Lu KY, Shen TL, Jou YC, Shen CH, Ohbayashi N, Kanaho Y, Huang YL, Lee H. LPA1/3 signaling mediates tumor lymphangiogenesis through promoting CRT expression in prostate cancer. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863:1305-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 84. | Kitayama J, Shida D, Sako A, Ishikawa M, Hama K, Aoki J, Arai H, Nagawa H. Over-expression of lysophosphatidic acid receptor-2 in human invasive ductal carcinoma. Breast Cancer Res. 2004;6:R640-R646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 88] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 85. | Li M, Xiao D, Zhang J, Qu H, Yang Y, Yan Y, Liu X, Wang J, Liu L, Duan X. Expression of LPA2 is associated with poor prognosis in human breast cancer and regulates HIF-1α expression and breast cancer cell growth. Oncol Rep. 2016;36:3479-3487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 86. | Enooku K, Uranbileg B, Ikeda H, Kurano M, Sato M, Kudo H, Maki H, Koike K, Hasegawa K, Kokudo N, Yatomi Y. Higher LPA2 and LPA6 mRNA Levels in Hepatocellular Carcinoma Are Associated with Poorer Differentiation, Microvascular Invasion and Earlier Recurrence with Higher Serum Autotaxin Levels. PLoS One. 2016;11:e0161825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 87. | Ishimoto K, Minami A, Minami K, Ueda N, Tsujiuchi T. Different effects of lysophosphatidic acid receptor-2 (LPA2) and LPA5 on the regulation of chemoresistance in colon cancer cells. J Recept Signal Transduct Res. 2021;41:93-98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 88. | Ren Z, Zhang C, Ma L, Zhang X, Shi S, Tang D, Xu J, Hu Y, Wang B, Zhang F, Zheng H. Lysophosphatidic acid induces the migration and invasion of SGC-7901 gastric cancer cells through the LPA2 and Notch signaling pathways. Int J Mol Med. 2019;44:67-78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 89. | Dong S, Li GX, Fang JH, Chen X, Sun YT. Advances in understanding of relationship between Hhip and Lpar2 gene expression and gastric cancer. Shijie Huaren Xiaohua Zazhi. 2021;29:1049-1054. [RCA] [DOI] [Full Text] [Cited by in CrossRef: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 90. | Xu J, Lai YJ, Lin WC, Lin FT. TRIP6 enhances lysophosphatidic acid-induced cell migration by interacting with the lysophosphatidic acid 2 receptor. J Biol Chem. 2004;279:10459-10468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 80] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 91. | Lin FT, Lai YJ. Regulation of the LPA2 receptor signaling through the carboxyl-terminal tail-mediated protein-protein interactions. Biochim Biophys Acta. 2008;1781:558-562. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 92. | Zhao P, Yun Q, Li R, Yan Y, Wang Y, Sun H, Damirin A. LPA3 is a precise therapeutic target and potential biomarker for ovarian cancer. Med Oncol. 2022;39:17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 93. | Hayashi M, Okabe K, Yamawaki Y, Teranishi M, Honoki K, Mori T, Fukushima N, Tsujiuchi T. Loss of lysophosphatidic acid receptor-3 enhances cell migration in rat lung tumor cells. Biochem Biophys Res Commun. 2011;405:450-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 94. | Kitayoshi M, Fukui R, Tanabe E, Kato K, Yoshikawa K, Fukushima N, Tsujiuchi T. Different effects on cell proliferation and migration abilities of endothelial cells by LPA₁ and LPA₃ in mammary tumor FM3A cells. J Recept Signal Transduct Res. 2012;32:209-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 95. | Sun K, Cai H, Duan X, Yang Y, Li M, Qu J, Zhang X, Wang J. Aberrant expression and potential therapeutic target of lysophosphatidic acid receptor 3 in triple-negative breast cancers. Clin Exp Med. 2015;15:371-380. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 96. | Fang X, Yu S, Bast RC, Liu S, Xu HJ, Hu SX, LaPushin R, Claret FX, Aggarwal BB, Lu Y, Mills GB. Mechanisms for lysophosphatidic acid-induced cytokine production in ovarian cancer cells. J Biol Chem. 2004;279:9653-9661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 152] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 97. | Hasegawa Y, Murph M, Yu S, Tigyi G, Mills GB. Lysophosphatidic acid (LPA)-induced vasodilator-stimulated phosphoprotein mediates lamellipodia formation to initiate motility in PC-3 prostate cancer cells. Mol Oncol. 2008;2:54-69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 98. | Takara K, Eino D, Ando K, Yasuda D, Naito H, Tsukada Y, Iba T, Wakabayashi T, Muramatsu F, Kidoya H, Fukuhara S, Mochizuki N, Ishii S, Kishima H, Takakura N. Lysophosphatidic Acid Receptor 4 Activation Augments Drug Delivery in Tumors by Tightening Endothelial Cell-Cell Contact. Cell Rep. 2017;20:2072-2086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 99. | Araki M, Kitayoshi M, Dong Y, Hirane M, Ozaki S, Mori S, Fukushima N, Honoki K, Tsujiuchi T. Inhibitory effects of lysophosphatidic acid receptor-5 on cellular functions of sarcoma cells. Growth Factors. 2014;32:117-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 100. | Tsujino M, Fujii M, Okabe K, Mori T, Fukushima N, Tsujiuchi T. Differential expressions and DNA methylation patterns of lysophosphatidic acid receptor genes in human colon cancer cells. Virchows Arch. 2010;457:669-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 101. | Kimura T, Mogi C, Sato K, Tomura H, Ohta H, Im DS, Kuwabara A, Kurose H, Murakami M, Okajima F. p2y5/LPA(6) attenuates LPA(1)-mediated VE-cadherin translocation and cell-cell dissociation through G(12/13) protein-Src-Rap1. Cardiovasc Res. 2011;92:149-158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 102. | Mukherjee A, Wu J, Barbour S, Fang X. Lysophosphatidic acid activates lipogenic pathways and de novo lipid synthesis in ovarian cancer cells. J Biol Chem. 2012;287:24990-25000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 103. | Fukushima N, Ishii S, Tsujiuchi T, Kagawa N, Katoh K. Comparative analyses of lysophosphatidic acid receptor-mediated signaling. Cell Mol Life Sci. 2015;72:2377-2394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 104. | Wu DH, Liu L, Chen LH, Ding YQ. Expression of KAI1/CD82 in human colorectal tumor. Di Yi Jun Yi Da Xue Xue Bao. 2003;23:714-715, 719. [PubMed] |
| 105. | Guo X, Friess H, Graber HU, Kashiwagi M, Zimmermann A, Korc M, Büchler MW. KAI1 expression is up-regulated in early pancreatic cancer and decreased in the presence of metastases. Cancer Res. 1996;56:4876-4880. [PubMed] |
| 106. | Friess H, Guo XZ, Berberat P, Graber HU, Zimmermann A, Korc M, Büchler MW. Reduced KAI1 expression in pancreatic cancer is associated with lymph node and distant metastases. Int J Cancer. 1998;79:349-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 107. | Xu JH, Guo XZ, Ren LN, Shao LC, Liu MP. KAI1 is a potential target for anti-metastasis in pancreatic cancer cells. World J Gastroenterol. 2008;14:1126-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 27] [Cited by in RCA: 31] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 108. | Liu X, Guo XZ, Zhang WW, Lu ZZ, Zhang QW, Duan HF, Wang LS. KAI1 inhibits HGF-induced invasion of pancreatic cancer by sphingosine kinase activity. Hepatobiliary Pancreat Dis Int. 2011;10:201-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 109. | Li H, Li J, Liu X, Chen J, Wu C, Guo X. Effect of PTEN and KAI1 gene overexpression on the proliferation, metastasis and radiosensitivity of ASPC1 pancreatic cancer cells under hypoxic conditions. Mol Med Rep. 2014;10:1973-1977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 110. | Liu X, Guo XZ, Li HY, Chen J, Ren LN, Wu CY. KAI1 inhibits lymphangiogenesis and lymphatic metastasis of pancreatic cancer in vivo. Hepatobiliary Pancreat Dis Int. 2014;13:87-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 111. | Wu CY, Guo XZ, Li HY. Hypoxia and Serum deprivation protected MiaPaCa-2 cells from KAI1-induced proliferation inhibition through autophagy pathway activation in solid tumors. Clin Transl Oncol. 2015;17:201-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 112. | Mashimo T, Watabe M, Hirota S, Hosobe S, Miura K, Tegtmeyer PJ, Rinker-Shaeffer CW, Watabe K. The expression of the KAI1 gene, a tumor metastasis suppressor, is directly activated by p53. Proc Natl Acad Sci U S A. 1998;95:11307-11311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 98] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 113. | Tang ZY. Hepatocellular carcinoma surgery-review of the past and prospects for the 21st century. J Surg Oncol. 2005;91:95-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 114. | Zhang W, Zhao CG, Sun HY, Zheng WE, Chen H. Expression characteristics of KAI1 and vascular endothelial growth factor and their diagnostic value for hepatocellular carcinoma. Gut Liver. 2014;8:536-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 115. | Mu Z, Wang H, Zhang J, Li Q, Wang L, Guo X. KAI1/CD82 suppresses hepatocyte growth factor-induced migration of hepatoma cells via upregulation of Sprouty2. Sci China C Life Sci. 2008;51:648-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 116. | Guo C, Liu Q, Zhang L, Yang X, Song T, Yao Y. Double lethal effects of fusion gene of wild-type p53 and JunB on hepatocellular carcinoma cells. J Huazhong Univ Sci Technolog Med Sci. 2012;32:663-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 117. | Si SH, Yang JM, Peng ZH, Luo YH, Zhou P. Effects of KAI1 gene on growth and invasion of human hepatocellular carcinoma MHCC97-H cells. World J Gastroenterol. 2004;10:2019-2023. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 118. | Yang JM, Peng ZH, Si SH, Liu WW, Luo YH, Ye ZY. KAI1 gene suppresses invasion and metastasis of hepatocellular carcinoma MHCC97-H cells in vitro and in animal models. Liver Int. 2008;28:132-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 119. | Xu J, Zhang Y, Wang Y, Tao X, Cheng L, Wu S, Tao Y. Correlation of KAI1, CD133 and vasculogenic mimicry with the prediction of metastasis and prognosis in hepatocellular carcinoma. Int J Clin Exp Pathol. 2018;11:3638-3646. [PubMed] |
| 120. | Lai JF, Xu WN, Noh SH, Lu WQ. Effect of World Health Organization (WHO) Histological Classification on Predicting Lymph Node Metastasis and Recurrence in Early Gastric Cancer. Med Sci Monit. 2016;22:3147-3153. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 121. | Ang TL, Fock KM. Clinical epidemiology of gastric cancer. Singapore Med J. 2014;55:621-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 274] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 122. | Ilhan O, Celik SY, Han U, Onal B. Use of KAI-1 as a prognostic factor in gastric carcinoma. Eur J Gastroenterol Hepatol. 2009;21:1369-1372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 123. | Knoener M, Krech T, Puls F, Lehmann U, Kreipe H, Christgen M. Limited value of KAI1/CD82 protein expression as a prognostic marker in human gastric cancer. Dis Markers. 2012;32:337-342. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 124. | Hinoda Y, Adachi Y, Takaoka A, Mitsuuchi H, Satoh Y, Itoh F, Kondoh Y, Imai K. Decreased expression of the metastasis suppressor gene KAI1 in gastric cancer. Cancer Lett. 1998;129:229-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 125. | Chen M, Towers LN, O'Connor KL. LPA2 (EDG4) mediates Rho-dependent chemotaxis with lower efficacy than LPA1 (EDG2) in breast carcinoma cells. Am J Physiol Cell Physiol. 2007;292:C1927-C1933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 126. | Xu L, Hou Y, Tu G, Chen Y, Du YE, Zhang H, Wen S, Tang X, Yin J, Lang L, Sun K, Yang G, Liu M. Nuclear Drosha enhances cell invasion via an EGFR-ERK1/2-MMP7 signaling pathway induced by dysregulated miRNA-622/197 and their targets LAMC2 and CD82 in gastric cancer. Cell Death Dis. 2017;8:e2642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 127. | Tsutsumi S, Shimura T, Morinaga N, Mochiki E, Asao T, Kuwano H. Loss of KAI1 expression in gastric cancer. Hepatogastroenterology. 2005;52:281-284. [PubMed] |
| 128. | Zheng HC, Wang MC, Li JY, Yang XF, Sun JM, Xin Y. Expression of maspin and kai1 and their clinicopathological significance in carcinogenesis and progression of gastric cancer. Chin Med Sci J. 2004;19:193-198. [PubMed] |
| 129. | Guan-Zhen Y, Ying C, Can-Rong N, Guo-Dong W, Jian-Xin Q, Jie-Jun W. Reduced protein expression of metastasis-related genes (nm23, KISS1, KAI1 and p53) in lymph node and liver metastases of gastric cancer. Int J Exp Pathol. 2007;88:175-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 61] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 130. | Zhu B, Zhou L, Yu L, Wu S, Song W, Gong X, Wang D. Evaluation of the correlation of vasculogenic mimicry, ALDH1, KAI1 and microvessel density in the prediction of metastasis and prognosis in colorectal carcinoma. BMC Surg. 2017;17:47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 33] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 131. | Lu G, Zhou L, Zhang X, Zhu B, Wu S, Song W, Gong X, Wang D, Tao Y. The expression of metastasis-associated in colon cancer-1 and KAI1 in gastric adenocarcinoma and their clinical significance. World J Surg Oncol. 2016;14:276. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 132. | Yang JL, Jackson P, Yu Y, Russell PJ, Markovic B, Crowe PJ. Expression of the KAI1 metastasis suppressor gene in non-metastatic versus metastatic human colorectal cancer. Anticancer Res. 2002;22:3337-3342. [PubMed] |
| 133. | Bae WK, Hong CS, Park MR, Sun EG, Lee JH, Kang K, Ryu KH, Shim HJ, Hwang JE, Cho SH, Chung IJ. TAp73 inhibits cell invasion and migration by directly activating KAI1 expression in colorectal carcinoma. Cancer Lett. 2018;415:106-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 134. | Hashida H, Takabayashi A, Tokuhara T, Taki T, Kondo K, Kohno N, Yamaoka Y, Miyake M. Integrin alpha3 expression as a prognostic factor in colon cancer: association with MRP-1/CD9 and KAI1/CD82. Int J Cancer. 2002;97:518-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 52] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 135. | Ryder NM, Guha S, Hines OJ, Reber HA, Rozengurt E. G protein-coupled receptor signaling in human ductal pancreatic cancer cells: neurotensin responsiveness and mitogenic stimulation. J Cell Physiol. 2001;186:53-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 136. | Nakai Y, Ikeda H, Nakamura K, Kume Y, Fujishiro M, Sasahira N, Hirano K, Isayama H, Tada M, Kawabe T, Komatsu Y, Omata M, Aoki J, Koike K, Yatomi Y. Specific increase in serum autotaxin activity in patients with pancreatic cancer. Clin Biochem. 2011;44:576-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 137. | Quan M, Cui JJ, Feng X, Huang Q. The critical role and potential target of the autotaxin/lysophosphatidate axis in pancreatic cancer. Tumour Biol. 2017;39:1010428317694544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 138. | Yang S, Zhang L, Purohit V, Shukla SK, Chen X, Yu F, Fu K, Chen Y, Solheim J, Singh PK, Song W, Dong J. Active YAP promotes pancreatic cancer cell motility, invasion and tumorigenesis in a mitotic phosphorylation-dependent manner through LPAR3. Oncotarget. 2015;6:36019-36031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 139. | Tveteraas IH, Aasrum M, Brusevold IJ, Ødegård J, Christoffersen T, Sandnes D. Lysophosphatidic acid induces both EGFR-dependent and EGFR-independent effects on DNA synthesis and migration in pancreatic and colorectal carcinoma cells. Tumour Biol. 2016;37:2519-2526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 140. | Gardner JA, Ha JH, Jayaraman M, Dhanasekaran DN. The gep proto-oncogene Gα13 mediates lysophosphatidic acid-mediated migration of pancreatic cancer cells. Pancreas. 2013;42:819-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 141. | Liao Y, Mu G, Zhang L, Zhou W, Zhang J, Yu H. Lysophosphatidic acid stimulates activation of focal adhesion kinase and paxillin and promotes cell motility, via LPA1-3, in human pancreatic cancer. Dig Dis Sci. 2013;58:3524-3533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 142. | Fukushima K, Otagaki S, Takahashi K, Minami K, Ishimoto K, Fukushima N, Honoki K, Tsujiuchi T. Promotion of cell-invasive activity through the induction of LPA receptor-1 in pancreatic cancer cells. J Recept Signal Transduct Res. 2018;38:367-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 143. | Kato K, Yoshikawa K, Tanabe E, Kitayoshi M, Fukui R, Fukushima N, Tsujiuchi T. Opposite roles of LPA1 and LPA3 on cell motile and invasive activities of pancreatic cancer cells. Tumour Biol. 2012;33:1739-1744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 32] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 144. | Tsujiuchi T, Furukawa M, Obo Y, Yamasaki A, Hotta M, Kusunoki C, Suyama N, Mori T, Honoki K, Fukushima N. Infrequent mutation of lysophosphatidic Acid receptor-1 gene in hamster pancreatic duct adenocarcinomas and established cell lines. J Toxicol Pathol. 2009;22:89-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 145. | Komachi M, Tomura H, Malchinkhuu E, Tobo M, Mogi C, Yamada T, Kimura T, Kuwabara A, Ohta H, Im DS, Kurose H, Takeyoshi I, Sato K, Okajima F. LPA1 receptors mediate stimulation, whereas LPA2 receptors mediate inhibition, of migration of pancreatic cancer cells in response to lysophosphatidic acid and malignant ascites. Carcinogenesis. 2009;30:457-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 53] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 146. | Yamada T, Sato K, Komachi M, Malchinkhuu E, Tobo M, Kimura T, Kuwabara A, Yanagita Y, Ikeya T, Tanahashi Y, Ogawa T, Ohwada S, Morishita Y, Ohta H, Im DS, Tamoto K, Tomura H, Okajima F. Lysophosphatidic acid (LPA) in malignant ascites stimulates motility of human pancreatic cancer cells through LPA1. J Biol Chem. 2004;279:6595-6605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 146] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 147. | Yoshikawa K, Tanabe E, Shibata A, Inoue S, Kitayoshi M, Okimoto S, Fukushima N, Tsujiuchi T. Involvement of oncogenic K-ras on cell migration stimulated by lysophosphatidic acid receptor-2 in pancreatic cancer cells. Exp Cell Res. 2013;319:105-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 148. | Ishii S, Hirane M, Fukushima K, Tomimatsu A, Fukushima N, Tsujiuchi T. Diverse effects of LPA4, LPA5 and LPA6 on the activation of tumor progression in pancreatic cancer cells. Biochem Biophys Res Commun. 2015;461:59-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 149. | Memet I, Tsalkidou E, Tsaroucha AK, Lambropoulou M, Chatzaki E, Trypsianis G, Schizas D, Pitiakoudis M, Simopoulos C. Autotaxin Expression in Hepatocellular Carcinoma. J Invest Surg. 2018;31:359-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 150. | Ertle J, Dechêne A, Sowa JP, Penndorf V, Herzer K, Kaiser G, Schlaak JF, Gerken G, Syn WK, Canbay A. Non-alcoholic fatty liver disease progresses to hepatocellular carcinoma in the absence of apparent cirrhosis. Int J Cancer. 2011;128:2436-2443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 383] [Article Influence: 27.4] [Reference Citation Analysis (1)] |
| 151. | Alexander J, Torbenson M, Wu TT, Yeh MM. Non-alcoholic fatty liver disease contributes to hepatocarcinogenesis in non-cirrhotic liver: a clinical and pathological study. J Gastroenterol Hepatol. 2013;28:848-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (2)] |
| 152. | Lopane C, Agosti P, Gigante I, Sabbà C, Mazzocca A. Implications of the lysophosphatidic acid signaling axis in liver cancer. Biochim Biophys Acta Rev Cancer. 2017;1868:277-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 153. | Watanabe N, Ikeda H, Nakamura K, Ohkawa R, Kume Y, Tomiya T, Tejima K, Nishikawa T, Arai M, Yanase M, Aoki J, Arai H, Omata M, Fujiwara K, Yatomi Y. Plasma lysophosphatidic acid level and serum autotaxin activity are increased in liver injury in rats in relation to its severity. Life Sci. 2007;81:1009-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 154. | Wu JM, Xu Y, Skill NJ, Sheng H, Zhao Z, Yu M, Saxena R, Maluccio MA. Autotaxin expression and its connection with the TNF-alpha-NF-kappaB axis in human hepatocellular carcinoma. Mol Cancer. 2010;9:71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 155. | Park SY, Jeong KJ, Panupinthu N, Yu S, Lee J, Han JW, Kim JM, Lee JS, Kang J, Park CG, Mills GB, Lee HY. Lysophosphatidic acid augments human hepatocellular carcinoma cell invasion through LPA1 receptor and MMP-9 expression. Oncogene. 2011;30:1351-1359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 111] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 156. | Zuckerman V, Sokolov E, Swet JH, Ahrens WA, Showlater V, Iannitti DA, Mckillop IH. Expression and function of lysophosphatidic acid receptors (LPARs) 1 and 3 in human hepatic cancer progenitor cells. Oncotarget. 2016;7:2951-2967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 157. | Okabe K, Hayashi M, Yamawaki Y, Teranishi M, Honoki K, Mori T, Fukushima N, Tsujiuchi T. Possible involvement of lysophosphatidic acid receptor-5 gene in the acquisition of growth advantage of rat tumor cells. Mol Carcinog. 2011;50:635-642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 158. | Zheng X, Jia Y, Qiu L, Zeng X, Xu L, Wei M, Huang C, Liu C, Chen L, Han J. A potential target for liver cancer management, lysophosphatidic acid receptor 6 (LPAR6), is transcriptionally up-regulated by the NCOA3 coactivator. J Biol Chem. 2020;295:1474-1488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 159. | Lippolis R, Gnocchi D, Santacroce L, Siciliano RA, Mazzeo MF, Scacco S, Sabbà C, Mazzocca A. A distinctive protein signature induced by lysophosphatidic acid receptor 6 (LPAR6) expression in hepatocellular carcinoma cells. Biochem Biophys Res Commun. 2020;526:1150-1156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 160. | Gnocchi D, Kapoor S, Nitti P, Cavalluzzi MM, Lentini G, Denora N, Sabbà C, Mazzocca A. Novel lysophosphatidic acid receptor 6 antagonists inhibit hepatocellular carcinoma growth through affecting mitochondrial function. J Mol Med (Berl). 2020;98:179-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 161. | Mazzocca A, Dituri F, De Santis F, Filannino A, Lopane C, Betz RC, Li YY, Mukaida N, Winter P, Tortorella C, Giannelli G, Sabbà C. Lysophosphatidic acid receptor LPAR6 supports the tumorigenicity of hepatocellular carcinoma. Cancer Res. 2015;75:532-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 162. | Zeng R, Li B, Huang J, Zhong M, Li L, Duan C, Zeng S, Liu W, Lu J, Tang Y, Zhou L, Liu Y, Li J, He Z, Wang Q, Dai Y. Lysophosphatidic Acid is a Biomarker for Peritoneal Carcinomatosis of Gastric Cancer and Correlates with Poor Prognosis. Genet Test Mol Biomarkers. 2017;21:641-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 163. | Ramachandran S, Shida D, Nagahashi M, Fang X, Milstien S, Takabe K, Spiegel S. Lysophosphatidic acid stimulates gastric cancer cell proliferation via ERK1-dependent upregulation of sphingosine kinase 1 transcription. FEBS Lett. 2010;584:4077-4082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 164. | Shida D, Fang X, Kordula T, Takabe K, Lépine S, Alvarez SE, Milstien S, Spiegel S. Cross-talk between LPA1 and epidermal growth factor receptors mediates up-regulation of sphingosine kinase 1 to promote gastric cancer cell motility and invasion. Cancer Res. 2008;68:6569-6577. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 107] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 165. | Kim MH, Park JS, Chang HJ, Baek MK, Kim HR, Shin BA, Ahn BW, Jung YD. Lysophosphatidic acid promotes cell invasion by up-regulating the urokinase-type plasminogen activator receptor in human gastric cancer cells. J Cell Biochem. 2008;104:1102-1112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 166. | Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3058] [Cited by in RCA: 2537] [Article Influence: 317.1] [Reference Citation Analysis (3)] |
| 167. | Kazama S, Kitayama J, Aoki J, Mori K, Nagawa H. Immunohistochemical detection of autotaxin (ATX)/lysophospholipase D (lysoPLD) in submucosal invasive colorectal cancer. J Gastrointest Cancer. 2011;42:204-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 168. | Yang M, Zhong WW, Srivastava N, Slavin A, Yang J, Hoey T, An S. G protein-coupled lysophosphatidic acid receptors stimulate proliferation of colon cancer cells through the {beta}-catenin pathway. Proc Natl Acad Sci U S A. 2005;102:6027-6032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 154] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 169. | Shida D, Kitayama J, Yamaguchi H, Okaji Y, Tsuno NH, Watanabe T, Takuwa Y, Nagawa H. Lysophosphatidic acid (LPA) enhances the metastatic potential of human colon carcinoma DLD1 cells through LPA1. Cancer Res. 2003;63:1706-1711. [PubMed] |
| 170. | Takahashi K, Fukushima K, Otagaki S, Ishimoto K, Minami K, Fukushima N, Honoki K, Tsujiuchi T. Effects of LPA1 and LPA6 on the regulation of colony formation activity in colon cancer cells treated with anticancer drugs. J Recept Signal Transduct Res. 2018;38:71-75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 171. | Lee SJ, Ritter SL, Zhang H, Shim H, Hall RA, Yun CC. MAGI-3 competes with NHERF-2 to negatively regulate LPA2 receptor signaling in colon cancer cells. Gastroenterology. 2011;140:924-934. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 172. | Yun CC, Sun H, Wang D, Rusovici R, Castleberry A, Hall RA, Shim H. LPA2 receptor mediates mitogenic signals in human colon cancer cells. Am J Physiol Cell Physiol. 2005;289:C2-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 99] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 173. | Lin S, Wang D, Iyer S, Ghaleb AM, Shim H, Yang VW, Chun J, Yun CC. The absence of LPA2 attenuates tumor formation in an experimental model of colitis-associated cancer. Gastroenterology. 2009;136:1711-1720. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 104] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 174. | Shida D, Watanabe T, Aoki J, Hama K, Kitayama J, Sonoda H, Kishi Y, Yamaguchi H, Sasaki S, Sako A, Konishi T, Arai H, Nagawa H. Aberrant expression of lysophosphatidic acid (LPA) receptors in human colorectal cancer. Lab Invest. 2004;84:1352-1362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 117] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 175. | Fukui R, Tanabe E, Kitayoshi M, Yoshikawa K, Fukushima N, Tsujiuchi T. Negative regulation of cell motile and invasive activities by lysophosphatidic acid receptor-3 in colon cancer HCT116 cells. Tumour Biol. 2012;33:1899-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 176. | Takahashi K, Fukushima K, Onishi Y, Inui K, Node Y, Fukushima N, Honoki K, Tsujiuchi T. Lysophosphatidic acid (LPA) signaling via LPA4 and LPA6 negatively regulates cell motile activities of colon cancer cells. Biochem Biophys Res Commun. 2017;483:652-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 177. | Custer MC, Risinger JI, Hoover S, Simpson RM, Patterson T, Barrett JC. Characterization of an antibody that can detect the Kai1/CD82 murine metastasis suppressor. Prostate. 2006;66:567-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 178. | Iiizumi M, Bandyopadhyay S, Watabe K. Interaction of Duffy antigen receptor for chemokines and KAI1: a critical step in metastasis suppression. Cancer Res. 2007;67:1411-1414. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 179. | Tonoli H, Barrett JC. CD82 metastasis suppressor gene: a potential target for new therapeutics? Trends Mol Med. 2005;11:563-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 180. | Gierse J, Thorarensen A, Beltey K, Bradshaw-Pierce E, Cortes-Burgos L, Hall T, Johnston A, Murphy M, Nemirovskiy O, Ogawa S, Pegg L, Pelc M, Prinsen M, Schnute M, Wendling J, Wene S, Weinberg R, Wittwer A, Zweifel B, Masferrer J. A novel autotaxin inhibitor reduces lysophosphatidic acid levels in plasma and the site of inflammation. J Pharmacol Exp Ther. 2010;334:310-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 184] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 181. | North EJ, Howard AL, Wanjala IW, Pham TC, Baker DL, Parrill AL. Pharmacophore development and application toward the identification of novel, small-molecule autotaxin inhibitors. J Med Chem. 2010;53:3095-3105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 182. | Bhave SR, Dadey DY, Karvas RM, Ferraro DJ, Kotipatruni RP, Jaboin JJ, Hallahan AN, Dewees TA, Linkous AG, Hallahan DE, Thotala D. Autotaxin Inhibition with PF-8380 Enhances the Radiosensitivity of Human and Murine Glioblastoma Cell Lines. Front Oncol. 2013;3:236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 43] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 183. | Schleicher SM, Thotala DK, Linkous AG, Hu R, Leahy KM, Yazlovitskaya EM, Hallahan DE. Autotaxin and LPA receptors represent potential molecular targets for the radiosensitization of murine glioma through effects on tumor vasculature. PLoS One. 2011;6:e22182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 184. | Tang X, Wuest M, Benesch MGK, Dufour J, Zhao Y, Curtis JM, Monjardet A, Heckmann B, Murray D, Wuest F, Brindley DN. Inhibition of Autotaxin with GLPG1690 Increases the Efficacy of Radiotherapy and Chemotherapy in a Mouse Model of Breast Cancer. Mol Cancer Ther. 2020;19:63-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 185. | Saga H, Ohhata A, Hayashi A, Katoh M, Maeda T, Mizuno H, Takada Y, Komichi Y, Ota H, Matsumura N, Shibaya M, Sugiyama T, Nakade S, Kishikawa K. A novel highly potent autotaxin/ENPP2 inhibitor produces prolonged decreases in plasma lysophosphatidic acid formation in vivo and regulates urethral tension. PLoS One. 2014;9:e93230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 186. | Venkatraman G, Benesch MG, Tang X, Dewald J, McMullen TP, Brindley DN. Lysophosphatidate signaling stabilizes Nrf2 and increases the expression of genes involved in drug resistance and oxidative stress responses: implications for cancer treatment. FASEB J. 2015;29:772-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 79] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 187. | Benesch MG, Tang X, Maeda T, Ohhata A, Zhao YY, Kok BP, Dewald J, Hitt M, Curtis JM, McMullen TP, Brindley DN. Inhibition of autotaxin delays breast tumor growth and lung metastasis in mice. FASEB J. 2014;28:2655-2666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 188. | Kehlen A, Lauterbach R, Santos AN, Thiele K, Kabisch U, Weber E, Riemann D, Langner J. IL-1 beta- and IL-4-induced down-regulation of autotaxin mRNA and PC-1 in fibroblast-like synoviocytes of patients with rheumatoid arthritis (RA). Clin Exp Immunol. 2001;123:147-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 189. | Xia Q, Deng AM, Wu SS, Zheng M. Cholera toxin inhibits human hepatocarcinoma cell proliferation in vitro via suppressing ATX/LPA axis. Acta Pharmacol Sin. 2011;32:1055-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 190. | Gupte R, Patil R, Liu J, Wang Y, Lee SC, Fujiwara Y, Fells J, Bolen AL, Emmons-Thompson K, Yates CR, Siddam A, Panupinthu N, Pham TC, Baker DL, Parrill AL, Mills GB, Tigyi G, Miller DD. Benzyl and naphthalene methylphosphonic acid inhibitors of autotaxin with anti-invasive and anti-metastatic activity. ChemMedChem. 2011;6:922-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 191. | Beck A, Wurch T, Bailly C, Corvaia N. Strategies and challenges for the next generation of therapeutic antibodies. Nat Rev Immunol. 2010;10:345-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 631] [Cited by in RCA: 635] [Article Influence: 42.3] [Reference Citation Analysis (0)] |
| 192. | Goldshmit Y, Matteo R, Sztal T, Ellett F, Frisca F, Moreno K, Crombie D, Lieschke GJ, Currie PD, Sabbadini RA, Pébay A. Blockage of lysophosphatidic acid signaling improves spinal cord injury outcomes. Am J Pathol. 2012;181:978-992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 55] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 193. | Crack PJ, Zhang M, Morganti-Kossmann MC, Morris AJ, Wojciak JM, Fleming JK, Karve I, Wright D, Sashindranath M, Goldshmit Y, Conquest A, Daglas M, Johnston LA, Medcalf RL, Sabbadini RA, Pébay A. Anti-lysophosphatidic acid antibodies improve traumatic brain injury outcomes. J Neuroinflammation. 2014;11:37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 194. | Budd DC, Qian Y. Development of lysophosphatidic acid pathway modulators as therapies for fibrosis. Future Med Chem. 2013;5:1935-1952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 195. | Zhang H, Xu X, Gajewiak J, Tsukahara R, Fujiwara Y, Liu J, Fells JI, Perygin D, Parrill AL, Tigyi G, Prestwich GD. Dual activity lysophosphatidic acid receptor pan-antagonist/autotaxin inhibitor reduces breast cancer cell migration in vitro and causes tumor regression in vivo. Cancer Res. 2009;69:5441-5449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 196. | Zhao PF, Wu S, Li Y, Bao G, Pei JY, Wang YW, Ma Q, Sun HJ, Damirin A. LPA receptor1 antagonists as anticancer agents suppress human lung tumours. Eur J Pharmacol. 2020;868:172886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 197. | Im DS. Pharmacological tools for lysophospholipid GPCRs: development of agonists and antagonists for LPA and S1P receptors. Acta Pharmacol Sin. 2010;31:1213-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 198. | Minami K, Ueda N, Maeda H, Ishimoto K, Otagaki S, Tsujiuchi T. Modulation of chemoresistance by lysophosphatidic acid (LPA) signaling through LPA5 in melanoma cells treated with anticancer drugs. Biochem Biophys Res Commun. 2019;517:359-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 199. | Zhao WJ, Zhu LL, Yang WQ, Xu SJ, Chen J, Ding XF, Liang Y, Chen G. LPAR5 promotes thyroid carcinoma cell proliferation and migration by activating class IA PI3K catalytic subunit p110β. Cancer Sci. 2021;112:1624-1632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 200. | Lee SC, Fujiwara Y, Liu J, Yue J, Shimizu Y, Norman DD, Wang Y, Tsukahara R, Szabo E, Patil R, Banerjee S, Miller DD, Balazs L, Ghosh MC, Waters CM, Oravecz T, Tigyi GJ. Autotaxin and LPA1 and LPA5 receptors exert disparate functions in tumor cells versus the host tissue microenvironment in melanoma invasion and metastasis. Mol Cancer Res. 2015;13:174-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 201. | Prestwich GD, Gajewiak J, Zhang H, Xu X, Yang G, Serban M. Phosphatase-resistant analogues of lysophosphatidic acid: agonists promote healing, antagonists and autotaxin inhibitors treat cancer. Biochim Biophys Acta. 2008;1781:588-594. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 202. | Swaney JS, Chapman C, Correa LD, Stebbins KJ, Bundey RA, Prodanovich PC, Fagan P, Baccei CS, Santini AM, Hutchinson JH, Seiders TJ, Parr TA, Prasit P, Evans JF, Lorrain DS. A novel, orally active LPA(1) receptor antagonist inhibits lung fibrosis in the mouse bleomycin model. Br J Pharmacol. 2010;160:1699-1713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 209] [Article Influence: 13.9] [Reference Citation Analysis (0)] |